The Immunomodulatory Role of Microbiota in Rheumatic Heart Disease: What Do We Know and What Can We Learn from Other Rheumatic Diseases?
Abstract
:1. Introduction
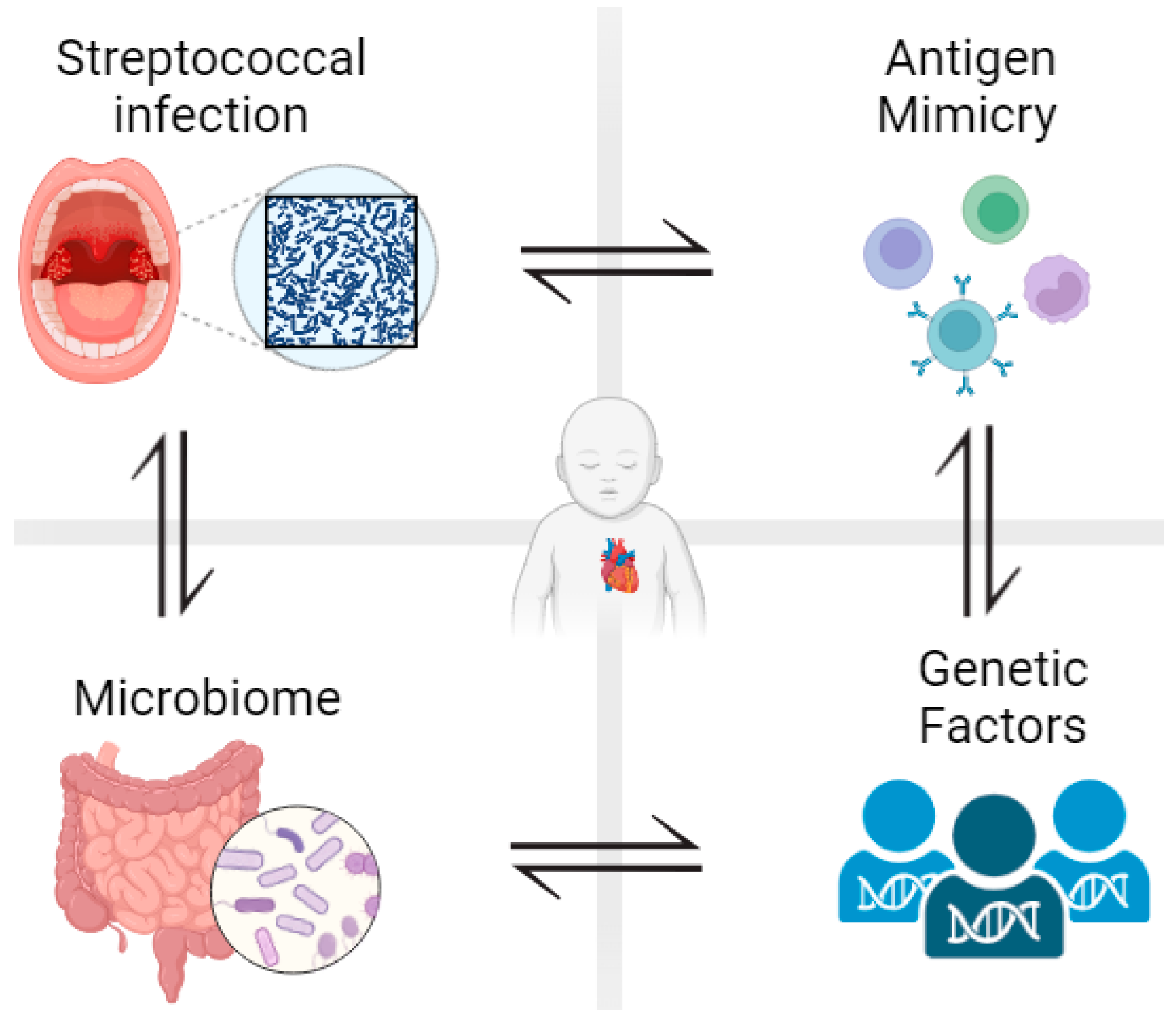
1.1. Rheumatic Heart Disease
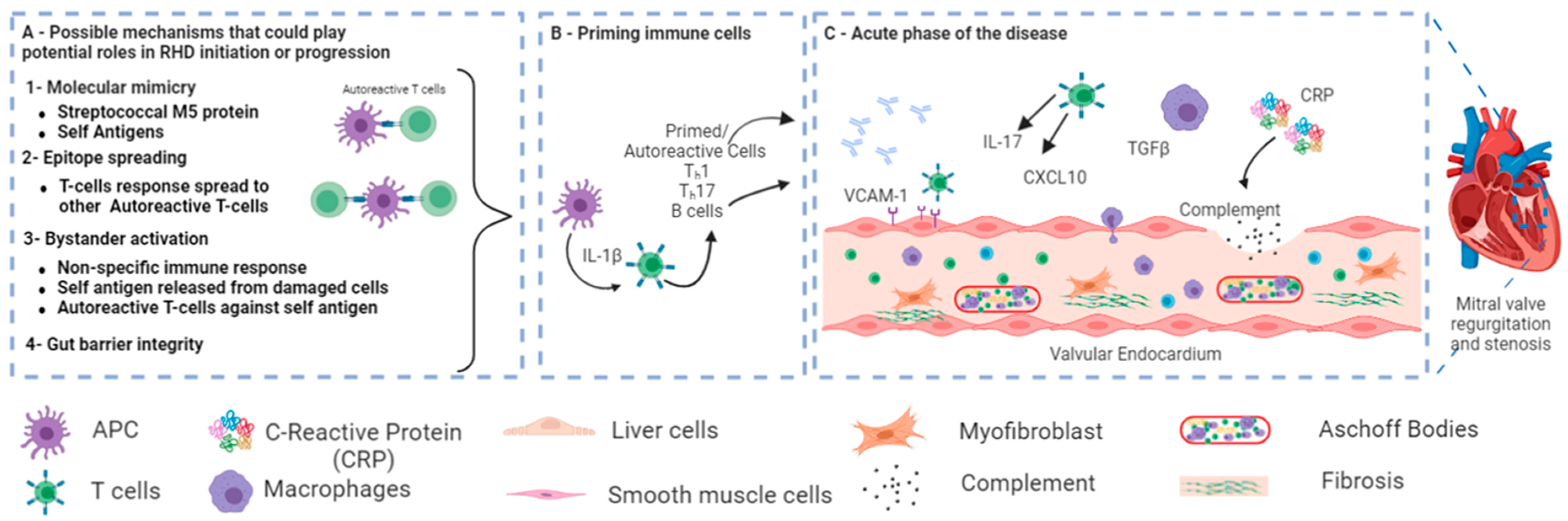
1.2. Signaling Pathway in RHD
2. Human Microbiota
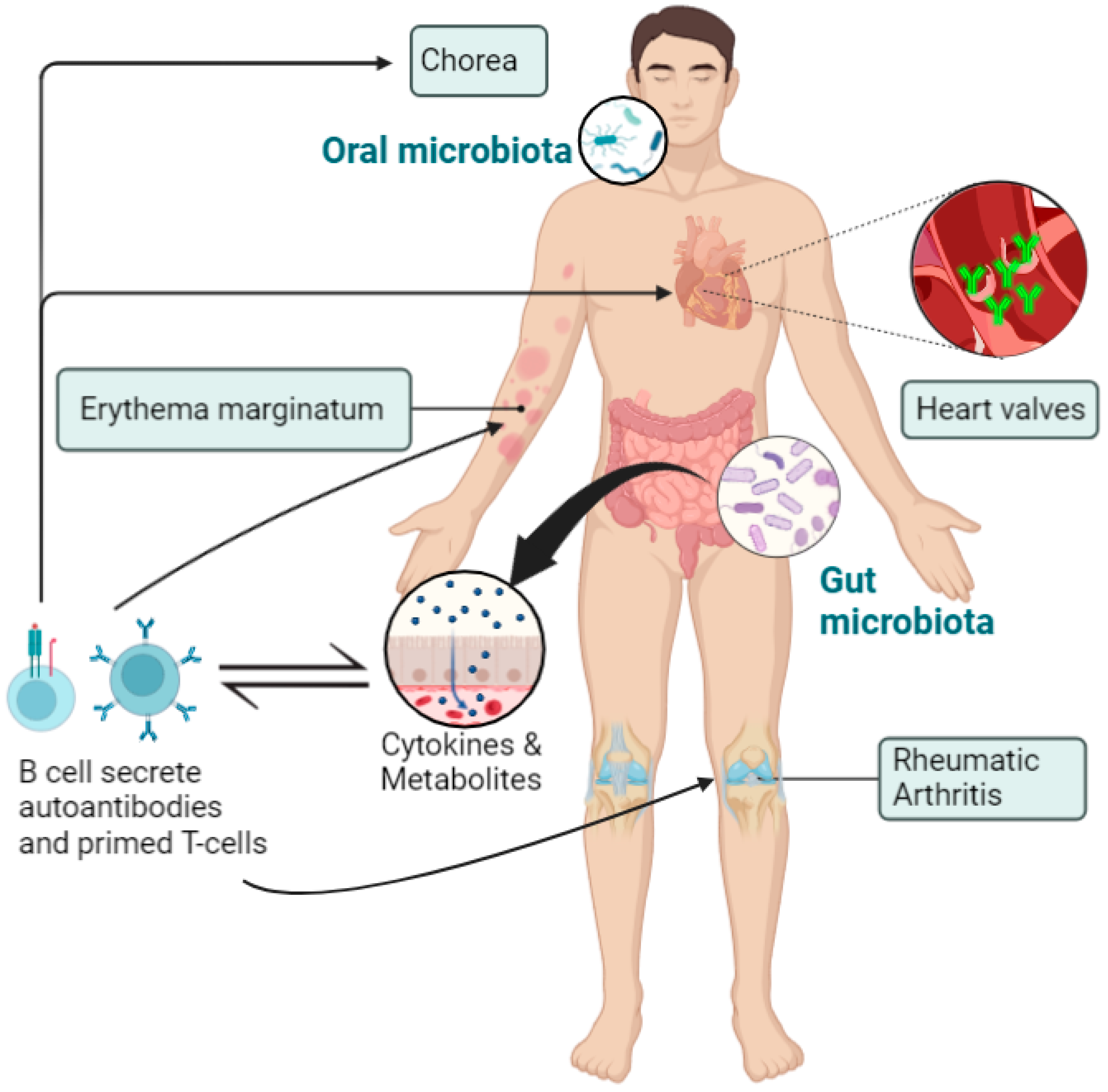
3. The Role of Microbiota in the Pathogenesis of RHD
4. Lessons from Other Rheumatic Disorders—The Role of Microbiota
4.1. Joint Involvement and Rheumatoid Arthritis
4.2. Skin Involvement and Dermatomyositis
4.3. Neurological Involvement and Fibromyalgia Syndrome
5. Probiotics Use in Rheumatic Diseases
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marijon, E.; Mirabel, M.; Celermajer, D.S.; Jouven, X. Rheumatic Heart Disease. Lancet 2012, 379, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Yu, D.; Lu, Q.; Zheng, Y.; Yang, Y. The Rise and Fall of Acute Rheumatic Fever and Rheumatic Heart Disease: A Mini Review. Front. Cardiovasc. Med. 2023, 10, 1183606. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, M.W. Rheumatic Fever, Autoimmunity and Molecular Mimicry: The Streptococcal Connection. Int. Rev. Immunol. 2014, 33, 314–329. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, L.; Kalil, J. Rheumatic Heart Disease: Molecules Involved in Valve Tissue Inflammation Leading to the Autoimmune Process and Anti-S. Pyogenes Vaccine. Front. Immunol. 2013, 4, 352. [Google Scholar] [CrossRef]
- Bryant, P.A.; Robins-Browne, R.; Carapetis, J.R.; Curtis, N. Some of the People, Some of the Time: Susceptibility to Acute Rheumatic Fever. Circulation 2009, 119, 742–753. [Google Scholar] [CrossRef] [PubMed]
- Muhamed, B.; Parks, T.; Sliwa, K. Genetics of Rheumatic Fever and Rheumatic Heart Disease. Nat. Rev. Cardiol. 2020, 17, 145–154. [Google Scholar] [CrossRef]
- Abdallah, A.M.; Abu-Madi, M. The Genetic Control of the Rheumatic Heart: Closing the Genotype-Phenotype Gap. Front. Med. 2021, 8, 611036. [Google Scholar] [CrossRef]
- Poddighe, D.; Comi, E.V.; Brambilla, I.; Licari, A.; Bruni, P.; Marseglia, G.L. Increased Total Serum Immunoglobulin E in Children Developing Mycoplasma Pneumoniae-Related Extra-Pulmonary Diseases. Iran. J. Allergy Asthma Immunol. 2018, 17, 490–496. [Google Scholar] [CrossRef]
- Gonciarz, W.; Tomaszewska, A.; Krupa, A.; Rechciński, T.; Chałubiński, M.; Broncel, M.; Chmiela, M. Antibodies towards TVLLPVIFF Amino Acid Sequence of TNF Receptor Induced by Helicobacter Pylori in Patients with Coronary Heart Disease. J. Clin. Med. 2022, 11, 2545. [Google Scholar] [CrossRef]
- Guilherme, L.; Kalil, J.; Cunningham, M. Molecular Mimicry in the Autoimmune Pathogenesis of Rheumatic Heart Disease. Autoimmunity 2006, 39, 31–39. [Google Scholar] [CrossRef]
- Guilherme, L.; Köhler, K.F.; Postol, E.; Kalil, J. Genes, Autoimmunity and Pathogenesis of Rheumatic Heart Disease. Ann. Pediatr. Cardiol. 2011, 4, 13–21. [Google Scholar] [CrossRef]
- Guilherme, L.; Cury, P.; Demarchi, L.M.F.; Coelho, V.; Abel, L.; Lopez, A.P.; Oshiro, S.E.; Aliotti, S.; Cunha-Neto, E.; Pomerantzeff, P.M.A.; et al. Rheumatic Heart Disease: Proinflammatory Cytokines Play a Role in the Progression and Maintenance of Valvular Lesions. Am. J. Pathol. 2004, 165, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Engel, M.E.; Stander, R.; Vogel, J.; Adeyemo, A.A.; Mayosi, B.M. Genetic Susceptibility to Acute Rheumatic Fever: A Systematic Review and Meta-Analysis of Twin Studies. PLoS ONE 2011, 6, e25326. [Google Scholar] [CrossRef] [PubMed]
- Selmi, C.; Lu, Q.; Humble, M.C. Heritability versus the Role of the Environment in Autoimmunity. J. Autoimmun. 2012, 39, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Muhamed, B.; Shaboodien, G.; Engel, M.E. Genetic Variants in Rheumatic Fever and Rheumatic Heart Disease. Am. J. Med. Genet. 2020, 184, 159–177. [Google Scholar] [CrossRef]
- Shi, X.-R.; Chen, B.-Y.; Lin, W.-Z.; Li, Y.-L.; Wang, Y.-L.; Liu, Y.; Huang, J.-J.; Zhang, W.-W.; Ma, X.-X.; Shao, S.; et al. Microbiota in Gut, Oral Cavity, and Mitral Valves Are Associated With Rheumatic Heart Disease. Front. Cell. Infect. Microbiol. 2021, 11, 643092. [Google Scholar] [CrossRef]
- Dong, H.; Sun, Y.; Shan, F.; Sun, Q.; Yang, B. Down-Regulation of MiR-101 Contributes to Rheumatic Heart Disease Through Up-Regulating TLR2. Med. Sci. Monit. 2015, 21, 1500–1506. [Google Scholar] [CrossRef]
- Xian, S.; Zeng, Z. Signalling Pathways Implicated in the Pathogenesis of Rheumatic Heart Disease (Review). Exp. Ther. Med. 2021, 21, 76. [Google Scholar] [CrossRef]
- Martin, W.J.; Steer, A.C.; Smeesters, P.R.; Keeble, J.; Inouye, M.; Carapetis, J.; Wicks, I.P. Post-Infectious Group A Streptococcal Autoimmune Syndromes and the Heart. Autoimmun. Rev. 2015, 14, 710–725. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, X.; Chen, X.; Gu, J.; Li, F.; Zhang, W.; Zheng, Y. Role of the MAPKs/TGF-Β1/TRAF6 Signaling Pathway in Atrial Fibrosis of Patients with Chronic Atrial Fibrillation and Rheumatic Mitral Valve Disease. Cardiology 2014, 129, 216–223. [Google Scholar] [CrossRef]
- Zhao, Z.; He, D.; Ling, F.; Chu, T.; Huang, D.; Wu, H.; Ge, J. CD4+ T Cells and TGFβ1/MAPK Signal Pathway Involved in the Valvular Hyperblastosis and Fibrosis in Patients with Rheumatic Heart Disease. Exp. Mol. Pathol. 2020, 114, 104402. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Wasson, M.; Baro, L.; Chaliha, M.S.; Toor, D. Lectin Complement Pathway Components as Risk Factors for Rheumatic Heart Disease in Assam, India. Human. Gene 2022, 34, 201102. [Google Scholar] [CrossRef]
- Beltrame, M.H.; Catarino, S.J.; Goeldner, I.; Boldt, A.B.W.; de Messias-Reason, I.J. The Lectin Pathway of Complement and Rheumatic Heart Disease. Front. Pediatr. 2014, 2, 148. [Google Scholar] [CrossRef] [PubMed]
- Mattos-Graner, R.O.; Klein, M.I.; Alves, L.A. The Complement System as a Key Modulator of the Oral Microbiome in Health and Disease. Crit. Rev. Microbiol. 2023, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Jovel, J.; Dieleman, L.A.; Kao, D.; Mason, A.L.; Wine, E. The Human Gut Microbiome in Health and Disease. Metagenomics 2018, 197–213. [Google Scholar]
- Ursell, L.K.; Clemente, J.C.; Rideout, J.R.; Gevers, D.; Caporaso, J.G.; Knight, R. The Interpersonal and Intrapersonal Diversity of Human-Associated Microbiota in Key Body Sites. J. Allergy Clin. Immunol. 2012, 129, 1204–1208. [Google Scholar] [CrossRef]
- Wastyk, H.C.; Fragiadakis, G.K.; Perelman, D.; Dahan, D.; Merrill, B.D.; Feiqiao, B.Y.; Topf, M.; Gonzalez, C.G.; Van Treuren, W.; Han, S. Gut-Microbiota-Targeted Diets Modulate Human Immune Status. Cell 2021, 184, 4137–4153.e14. [Google Scholar] [CrossRef]
- Asnicar, F.; Berry, S.E.; Valdes, A.M.; Nguyen, L.H.; Piccinno, G.; Drew, D.A.; Leeming, E.; Gibson, R.; Le Roy, C.; Khatib, H.A.; et al. Microbiome Connections with Host Metabolism and Habitual Diet from 1,098 Deeply Phenotyped Individuals. Nat. Med. 2021, 27, 321–332. [Google Scholar] [CrossRef]
- Quigley, E.M. Gut Bacteria in Health and Disease. Gastroenterol. Hepatol. 2013, 9, 560. [Google Scholar]
- Robles-Alonso, V.; Guarner, F. Progress in the Knowledge of the Intestinal Human Microbiota. Nutr. Hosp. 2013, 28, 553–557. [Google Scholar]
- Gomaa, E.Z. Human Gut Microbiota/Microbiome in Health and Diseases: A Review. Antonie Van Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Bäckhed, F.; Landmesser, U.; Hazen, S.L. Intestinal Microbiota in Cardiovascular Health and Disease: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 2089–2105. [Google Scholar] [CrossRef]
- Karlsson, F.H.; Tremaroli, V.; Nookaew, I.; Bergström, G.; Behre, C.J.; Fagerberg, B.; Nielsen, J.; Bäckhed, F. Gut Metagenome in European Women with Normal, Impaired and Diabetic Glucose Control. Nature 2013, 498, 99–103. [Google Scholar] [CrossRef]
- Cignarella, F.; Cantoni, C.; Ghezzi, L.; Salter, A.; Dorsett, Y.; Chen, L.; Phillips, D.; Weinstock, G.M.; Fontana, L.; Cross, A.H.; et al. Intermittent Fasting Confers Protection in CNS Autoimmunity by Altering the Gut Microbiota. Cell. Metab. 2018, 27, 1222–1235.e6. [Google Scholar] [CrossRef] [PubMed]
- Bui, F.Q.; Almeida-da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between Periodontal Pathogens and Systemic Disease. Biomed. J. 2019, 42, 27–35. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Li, H.; Ni, C.; Du, Z.; Yan, F. Human Oral Microbiota and Its Modulation for Oral Health. Biomed. Pharmacother. 2018, 99, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Darveau, R.P.; Curtis, M.A. The Keystone-Pathogen Hypothesis. Nat. Rev. Microbiol. 2012, 10, 717–725. [Google Scholar] [CrossRef]
- Read, E.; Curtis, M.A.; Neves, J.F. The Role of Oral Bacteria in Inflammatory Bowel Disease. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, A.; Lumngwena, E.N.; Ntusi, N.A.B. The Oral Microbiome in the Pathophysiology of Cardiovascular Disease. Nat. Rev. Cardiol. 2023, 20, 386–403. [Google Scholar] [CrossRef] [PubMed]
- Poddighe, D.; Kushugulova, A. Salivary Microbiome in Pediatric and Adult Celiac Disease. Front. Cell. Infect. Microbiol. 2021, 11, 625162. [Google Scholar] [CrossRef]
- Ezzamouri, B.; Shoaie, S.; Ledesma-Amaro, R. Synergies of Systems Biology and Synthetic Biology in Human Microbiome Studies. Front. Microbiol. 2021, 12, 681982. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Rigatto, K.; Gazzana, M.B.; Knorst, M.M.; Richards, E.M.; Pepine, C.J.; Raizada, M.K. Altered Gut Microbiome Profile in Patients with Pulmonary Arterial Hypertension. Hypertension 2020, 75, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Kummen, M.; Mayerhofer, C.C.; Vestad, B.; Broch, K.; Awoyemi, A.; Storm-Larsen, C.; Ueland, T.; Yndestad, A.; Hov, J.R.; Trøseid, M. Gut Microbiota Signature in Heart Failure Defined from Profiling of 2 Independent Cohorts. J. Am. Coll. Cardiol. 2018, 71, 1184–1186. [Google Scholar] [CrossRef]
- Li, H.; Xu, H.; Li, Y.; Jiang, Y.; Hu, Y.; Liu, T.; Tian, X.; Zhao, X.; Zhu, Y.; Wang, S. Alterations of Gut Microbiota Contribute to the Progression of Unruptured Intracranial Aneurysms. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Koren, O.; Spor, A.; Felin, J.; Fåk, F.; Stombaugh, J.; Tremaroli, V.; Behre, C.J.; Knight, R.; Fagerberg, B.; Ley, R.E. Human Oral, Gut, and Plaque Microbiota in Patients with Atherosclerosis. Proc. Natl. Acad. Sci. USA 2011, 108, 4592–4598. [Google Scholar] [CrossRef]
- Ziebolz, D.; Jahn, C.; Pegel, J.; Semper-Pinnecke, E.; Mausberg, R.F.; Waldmann-Beushausen, R.; Schöndube, F.A.; Danner, B.C. Periodontal Bacteria DNA Findings in Human Cardiac Tissue—Is There a Link of Periodontitis to Heart Valve Disease? Int. J. Cardiol. 2018, 251, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal Diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Toscano, M.; De Grandi, R.; Stronati, L.; De Vecchi, E.; Drago, L. Effect of Lactobacillus Rhamnosus HN001 and Bifidobacterium Longum BB536 on the Healthy Gut Microbiota Composition at Phyla and Species Level: A Preliminary Study. World J. Gastroenterol. 2017, 23, 2696–2704. [Google Scholar] [CrossRef]
- Wang, W.; Chen, L.; Zhou, R.; Wang, X.; Song, L.; Huang, S.; Wang, G.; Xia, B. Increased Proportions of Bifidobacterium and the Lactobacillus Group and Loss of Butyrate-Producing Bacteria in Inflammatory Bowel Disease. J. Clin. Microbiol. 2014, 52, 398–406. [Google Scholar] [CrossRef]
- He, Z.; Shao, T.; Li, H.; Xie, Z.; Wen, C. Alterations of the Gut Microbiome in Chinese Patients with Systemic Lupus Erythematosus. Gut Pathog. 2016, 8, 64. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short Chain Fatty Acids in Human Gut and Metabolic Health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van Der Veeken, J.; Deroos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J. Metabolites Produced by Commensal Bacteria Promote Peripheral Regulatory T-Cell Generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.T.; Cresci, G.A.M. The Immunomodulatory Functions of Butyrate. J. Inflamm. Res. 2021, 14, 6025–6041. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Qie, Y.; Park, J.; Kim, C.H. Gut Microbial Metabolites Fuel Host Antibody Responses. Cell. Host Microbe 2016, 20, 202–214. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Li, F.; Xie, F.; Liu, M.; Shi, J.; Dong, N. Circulating Follicular T Helper Cells and Humoral Reactivity in Rheumatic Heart Disease. Life Sci. 2020, 245, 117390. [Google Scholar] [CrossRef]
- Gong, D.; Zhang, L.; Zhang, Y.; Wang, F.; Zhao, Z.; Zhou, X. Gut Microbial Metabolite Trimethylamine N-Oxide Is Related to Thrombus Formation in Atrial Fibrillation Patients. Am. J. Med. Sci. 2019, 358, 422–428. [Google Scholar] [CrossRef]
- Maharaj, B.; Vayej, A.C. Oral Health of Patients with Severe Rheumatic Heart Disease. Cardiovasc. J. Afr. 2012, 23, 336–339. [Google Scholar] [CrossRef]
- Tandon, R.; Sharma, M.; Chandrashekhar, Y.; Kotb, M.; Yacoub, M.H.; Narula, J. Revisiting the Pathogenesis of Rheumatic Fever and Carditis. Nat. Rev. Cardiol. 2013, 10, 171–177. [Google Scholar] [CrossRef]
- Khan, I.; Khan, I.; Jianye, Z.; Xiaohua, Z.; Khan, M.; Hilal, M.G.; Kakakhel, M.A.; Mehmood, A.; Lizhe, A.; Zhiqiang, L. Exploring Blood Microbial Communities and Their Influence on Human Cardiovascular Disease. J. Clin. Lab. Anal. 2022, 36, e24354. [Google Scholar] [CrossRef]
- Tobón, G.J.; Youinou, P.; Saraux, A. The Environment, Geo-Epidemiology, and Autoimmune Disease: Rheumatoid Arthritis. J. Autoimmun. 2010, 35, 10–14. [Google Scholar] [CrossRef]
- Chu, X.-J.; Cao, N.-W.; Zhou, H.-Y.; Meng, X.; Guo, B.; Zhang, H.-Y.; Li, B.-Z. The Oral and Gut Microbiome in Rheumatoid Arthritis Patients: A Systematic Review. Rheumatology 2021, 60, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Artacho, A.; Isaac, S.; Nayak, R.; Flor-Duro, A.; Alexander, M.; Koo, I.; Manasson, J.; Smith, P.B.; Rosenthal, P.; Homsi, Y.; et al. The Pretreatment Gut Microbiome Is Associated with Lack of Response to Methotrexate in New-Onset Rheumatoid Arthritis. Arthritis Rheumatol. 2021, 73, 931–942. [Google Scholar] [CrossRef] [PubMed]
- Lehenaff, R.; Tamashiro, R.; Nascimento, M.M.; Lee, K.; Jenkins, R.; Whitlock, J.; Li, E.C.; Sidhu, G.; Anderson, S.; Progulske-Fox, A.; et al. Subgingival Microbiome of Deep and Shallow Periodontal Sites in Patients with Rheumatoid Arthritis: A Pilot Study. BMC Oral. Health 2021, 21, 248. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Do, T.; Mankia, K.; Meade, J.; Hunt, L.; Clerehugh, V.; Speirs, A.; Tugnait, A.; Emery, P.; Devine, D. Dysbiosis in the Oral Microbiomes of Anti-CCP Positive Individuals at Risk of Developing Rheumatoid Arthritis. Ann. Rheum. Dis. 2021, 80, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Matei, D.E.; Menon, M.; Alber, D.G.; Smith, A.M.; Nedjat-Shokouhi, B.; Fasano, A.; Magill, L.; Duhlin, A.; Bitoun, S.; Gleizes, A.; et al. Intestinal Barrier Dysfunction Plays an Integral Role in Arthritis Pathology and Can Be Targeted to Ameliorate Disease. Med 2021, 2, 864–883.e9. [Google Scholar] [CrossRef]
- Chen, J.; Wright, K.; Davis, J.M.; Jeraldo, P.; Marietta, E.V.; Murray, J.; Nelson, H.; Matteson, E.L.; Taneja, V. An Expansion of Rare Lineage Intestinal Microbes Characterizes Rheumatoid Arthritis. Genome Med. 2016, 8, 43. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y.; et al. The Oral and Gut Microbiomes Are Perturbed in Rheumatoid Arthritis and Partly Normalized after Treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M.; et al. Dysbiosis Contributes to Arthritis Development via Activation of Autoreactive T Cells in the Intestine. Arthritis Rheumatol. 2016, 68, 2646–2661. [Google Scholar] [CrossRef]
- Scher, J.U.; Sczesnak, A.; Longman, R.S.; Segata, N.; Ubeda, C.; Bielski, C.; Rostron, T.; Cerundolo, V.; Pamer, E.G.; Abramson, S.B.; et al. Expansion of Intestinal Prevotella Copri Correlates with Enhanced Susceptibility to Arthritis. Elife 2013, 2, e01202. [Google Scholar] [CrossRef]
- Gilis, E.; Mortier, C.; Venken, K.; Debusschere, K.; Vereecke, L.; Elewaut, D. The Role of the Microbiome in Gut and Joint Inflammation in Psoriatic Arthritis and Spondyloarthritis. J. Rheumatol. Suppl. 2018, 94, 36–39. [Google Scholar] [CrossRef]
- Wen, C.; Zheng, Z.; Shao, T.; Liu, L.; Xie, Z.; Le Chatelier, E.; He, Z.; Zhong, W.; Fan, Y.; Zhang, L.; et al. Quantitative Metagenomics Reveals Unique Gut Microbiome Biomarkers in Ankylosing Spondylitis. Genome Biol. 2017, 18, 142. [Google Scholar] [CrossRef] [PubMed]
- Guggino, G.; Mauro, D.; Rizzo, A.; Alessandro, R.; Raimondo, S.; Bergot, A.S.; Rahman, M.A.; Ellis, J.J.; Milling, S.; Lories, R.; et al. Inflammasome Activation in Ankylosing Spondylitis is Associated with Gut Dysbiosis. Arthritis Rheumatol. 2021, 73, 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Zhao, H.; Xiao, X.Y.; Chen, B.D.; Guo, R.J.; Wang, Q.; Chen, H.; Zhao, L.D.; Zhang, C.C.; Jiao, Y.H.; et al. Metagenomic Profiling of the Pro-Inflammatory Gut Microbiota in Ankylosing Spondylitis. J. Autoimmun. 2020, 107, 102360. [Google Scholar] [CrossRef] [PubMed]
- Fuggle, N.R.; Smith, T.O.; Kaul, A.; Sofat, N. Hand to Mouth: A Systematic Review and Meta-Analysis of the Association between Rheumatoid Arthritis and Periodontitis. Front. Immunol. 2016, 7, 80. [Google Scholar] [CrossRef]
- Eriksson, K.; Fei, G.; Lundmark, A.; Benchimol, D.; Lee, L.; Hu, Y.O.O.; Kats, A.; Saevarsdottir, S.; Catrina, A.I.; Klinge, B.; et al. Periodontal Health and Oral Microbiota in Patients with Rheumatoid Arthritis. J. Clin. Med. 2019, 8, 630. [Google Scholar] [CrossRef]
- Beyer, K.; Zaura, E.; Brandt, B.W.; Buijs, M.J.; Brun, J.G.; Crielaard, W.; Bolstad, A.I. Subgingival Microbiome of Rheumatoid Arthritis Patients in Relation to Their Disease Status and Periodontal Health. PLoS ONE 2018, 13, e0202278. [Google Scholar] [CrossRef]
- du Teil Espina, M.; Gabarrini, G.; Harmsen, H.J.M.; Westra, J.; van Winkelhoff, A.J.; van Dijl, J.M. Talk to Your Gut: The Oral-Gut Microbiome Axis and Its Immunomodulatory Role in the Etiology of Rheumatoid Arthritis. FEMS Microbiol. Rev. 2019, 43, 1–18. [Google Scholar] [CrossRef]
- Huang, C.; Yi, X.; Long, H.; Zhang, G.; Wu, H.; Zhao, M.; Lu, Q. Disordered Cutaneous Microbiota in Systemic Lupus Erythematosus. J. Autoimmun. 2020, 108, 102391. [Google Scholar] [CrossRef]
- Zeeuwen, P.L.; Kleerebezem, M.; Timmerman, H.M.; Schalkwijk, J. Microbiome and Skin Diseases. Curr. Opin. Allergy Clin. Immunol. 2013, 13, 514–520. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Cao, N.W.; Guo, B.; Chen, W.J.; Tao, J.H.; Chu, X.J.; Meng, X.; Zhang, T.X.; Li, B.Z. Systemic Lupus Erythematosus Patients Have a Distinct Structural and Functional Skin Microbiota Compared with Controls. Lupus 2021, 30, 1553–1564. [Google Scholar] [CrossRef]
- Bae, S.S.; Dong, T.S.; Wang, J.; Lagishetty, V.; Katzka, W.; Jacobs, J.P.; Charles-Schoeman, C. Altered Gut Microbiome in Patients with Dermatomyositis. ACR Open Rheumatol. 2022, 4, 658–670. [Google Scholar] [CrossRef] [PubMed]
- Bercik, P.; Collins, S.M. The Effects of Inflammation, Infection and Antibiotics on the Microbiota-Gut-Brain Axis. Adv. Exp. Med. Biol. 2014, 817, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990, 33, 160–172. [Google Scholar] [CrossRef]
- Glass, J.M. Review of Cognitive Dysfunction in Fibromyalgia: A Convergence on Working Memory and Attentional Control Impairments. Rheum. Dis. Clin. North. Am. 2009, 35, 299–311. [Google Scholar] [CrossRef]
- Minerbi, A.; Gonzalez, E.; Brereton, N.J.B.; Anjarkouchian, A.; Dewar, K.; Fitzcharles, M.A.; Chevalier, S.; Shir, Y. Altered Microbiome Composition in Individuals with Fibromyalgia. Pain 2019, 160, 2589–2602. [Google Scholar] [CrossRef] [PubMed]
- Freidin, M.B.; Stalteri, M.A.; Wells, P.M.; Lachance, G.; Baleanu, A.F.; Bowyer, R.C.E.; Kurilshikov, A.; Zhernakova, A.; Steves, C.J.; Williams, F.M.K. An Association between Chronic Widespread Pain and the Gut Microbiome. Rheumatology 2021, 60, 3727–3737. [Google Scholar] [CrossRef]
- Clos-Garcia, M.; Andrés-Marin, N.; Fernández-Eulate, G.; Abecia, L.; Lavín, J.L.; van Liempd, S.; Cabrera, D.; Royo, F.; Valero, A.; Errazquin, N.; et al. Gut Microbiome and Serum Metabolome Analyses Identify Molecular Biomarkers and Altered Glutamate Metabolism in Fibromyalgia. EBioMedicine 2019, 46, 499–511. [Google Scholar] [CrossRef]
- He, J.; Chu, Y.; Li, J.; Meng, Q.; Liu, Y.; Jin, J.; Wang, Y.; Wang, J.; Huang, B.; Shi, L.; et al. Intestinal Butyrate-Metabolizing Species Contribute to Autoantibody Production and Bone Erosion in Rheumatoid Arthritis. Sci. Adv. 2022, 8, eabm1511. [Google Scholar] [CrossRef]
- Audo, R.; Sanchez, P.; Rivière, B.; Mielle, J.; Tan, J.; Lukas, C.; Macia, L.; Morel, J.; Immediato Daien, C. Rheumatoid Arthritis Is Associated with Increased Gut Permeability and Bacterial Translocation Which Are Reversed by Inflammation Control. Rheumatology 2022, 62, keac454. [Google Scholar] [CrossRef]
- Yao, Y.; Cai, X.; Fei, W.; Ye, Y.; Zhao, M.; Zheng, C. The Role of Short-Chain Fatty Acids in Immunity, Inflammation and Metabolism. Crit. Rev. Food Sci. Nutr. 2022, 62, 1–12. [Google Scholar] [CrossRef]
- Muhamad Rizal, N.S.; Neoh, H.; Ramli, R.; Periyasamy, P.R.; Hanafiah, A.; Abdul Samat, M.N.; Tan, T.L.; Wong, K.K.; Nathan, S.; Chieng, S.; et al. Advantages and Limitations of 16S RRNA Next-Generation Sequencing for Pathogen Identification in the Diagnostic Microbiology Laboratory: Perspectives from a Middle-Income Country. Diagnostics 2020, 10, 816. [Google Scholar] [CrossRef]
- Bron, P.A.; Kleerebezem, M.; Brummer, R.-J.; Cani, P.D.; Mercenier, A.; MacDonald, T.T.; Garcia-Ródenas, C.L.; Wells, J.M. Can Probiotics Modulate Human Disease by Impacting Intestinal Barrier Function? Br. J. Nutr. 2017, 117, 93–107. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, G.L.V.; Leite, A.Z.; Higuchi, B.S.; Gonzaga, M.I.; Mariano, V.S. Intestinal Dysbiosis and Probiotic Applications in Autoimmune Diseases. Immunology 2017, 152, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, S.; Mahmoudi, M.; Momtazi, A.A.; Sahebkar, A.; Doulabi, H.; Rastin, M. Tolerogenic Probiotics: Potential Immunoregulators in Systemic Lupus Erythematosus. J. Cell. Physiol. 2017, 232, 1994–2007. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Zhang, H.; Liao, X.; Lin, K.; Liu, H.; Edwards, M.R.; Ahmed, S.A.; Yuan, R.; Li, L.; Cecere, T.E. Control of Lupus Nephritis by Changes of Gut Microbiota. Microbiome 2017, 5, 1–12. [Google Scholar] [CrossRef]
- Hulander, E.; Bärebring, L.; Turesson Wadell, A.; Gjertsson, I.; Calder, P.C.; Winkvist, A.; Lindqvist, H.M. Diet Intervention Improves Cardiovascular Profile in Patients with Rheumatoid Arthritis: Results from the Randomized Controlled Cross-over Trial ADIRA. Nutr. J. 2021, 20, 9. [Google Scholar] [CrossRef] [PubMed]
- Zamani, B.; Golkar, H.R.; Farshbaf, S.; Emadi-Baygi, M.; Tajabadi-Ebrahimi, M.; Jafari, P.; Akhavan, R.; Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z. Clinical and Metabolic Response to Probiotic Supplementation in Patients with Rheumatoid Arthritis: A Randomized, Double-blind, Placebo-controlled Trial. Int. J. Rheum. Dis. 2016, 19, 869–879. [Google Scholar] [CrossRef]
- Lee, S.; Koh, J.; Chang, Y.; Kim, H.Y.; Chung, D.H. Invariant NKT Cells Functionally Link Microbiota-Induced Butyrate Production and Joint Inflammation. J. Immunol. 2019, 203, 3199–3208. [Google Scholar] [CrossRef]
- Hui, W.; Yu, D.; Cao, Z.; Zhao, X. Butyrate Inhibit Collagen-Induced Arthritis via Treg/IL-10/Th17 Axis. Int. Immunopharmacol. 2019, 68, 226–233. [Google Scholar] [CrossRef]
- Oliviero, F.; Spinella, P. Benefits of Probiotics in Rheumatic Diseases. Front. Nutr. 2020, 7, 157. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohil, A.; Abdalla, W.; Ibrahim, W.N.; Al-Harbi, K.M.; Al-Haidose, A.; Al-Asmakh, M.; Abdallah, A.M. The Immunomodulatory Role of Microbiota in Rheumatic Heart Disease: What Do We Know and What Can We Learn from Other Rheumatic Diseases? Medicina 2023, 59, 1629. https://doi.org/10.3390/medicina59091629
Kohil A, Abdalla W, Ibrahim WN, Al-Harbi KM, Al-Haidose A, Al-Asmakh M, Abdallah AM. The Immunomodulatory Role of Microbiota in Rheumatic Heart Disease: What Do We Know and What Can We Learn from Other Rheumatic Diseases? Medicina. 2023; 59(9):1629. https://doi.org/10.3390/medicina59091629
Chicago/Turabian StyleKohil, Amira, Wafa Abdalla, Wisam N. Ibrahim, Khalid M. Al-Harbi, Amal Al-Haidose, Maha Al-Asmakh, and Atiyeh M. Abdallah. 2023. "The Immunomodulatory Role of Microbiota in Rheumatic Heart Disease: What Do We Know and What Can We Learn from Other Rheumatic Diseases?" Medicina 59, no. 9: 1629. https://doi.org/10.3390/medicina59091629
APA StyleKohil, A., Abdalla, W., Ibrahim, W. N., Al-Harbi, K. M., Al-Haidose, A., Al-Asmakh, M., & Abdallah, A. M. (2023). The Immunomodulatory Role of Microbiota in Rheumatic Heart Disease: What Do We Know and What Can We Learn from Other Rheumatic Diseases? Medicina, 59(9), 1629. https://doi.org/10.3390/medicina59091629








