Indocyanine Green (ICG) and Colorectal Surgery: A Literature Review on Qualitative and Quantitative Methods of Usage
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Recto-Colonic Perfusion
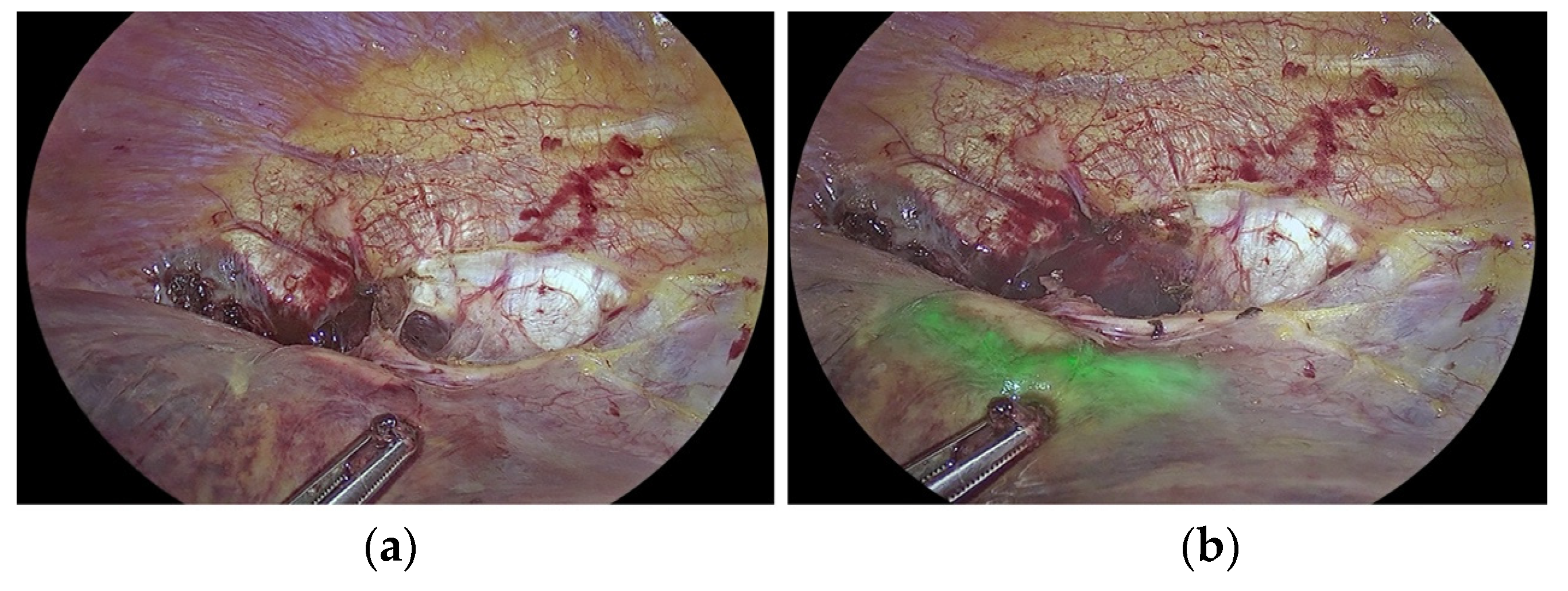
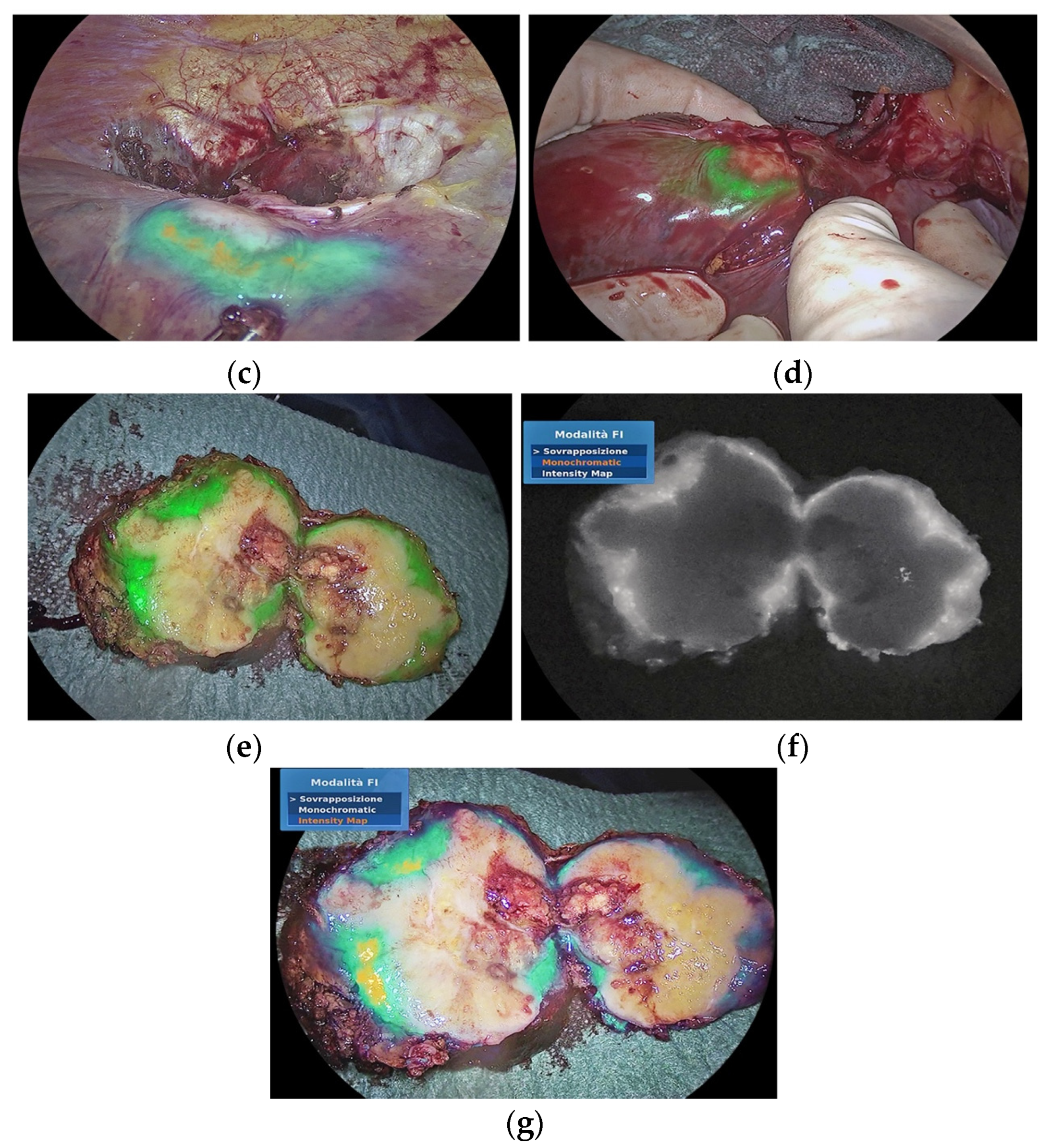
3.2. Improving the Detection of Cancer Tissue and Targeted Treatment
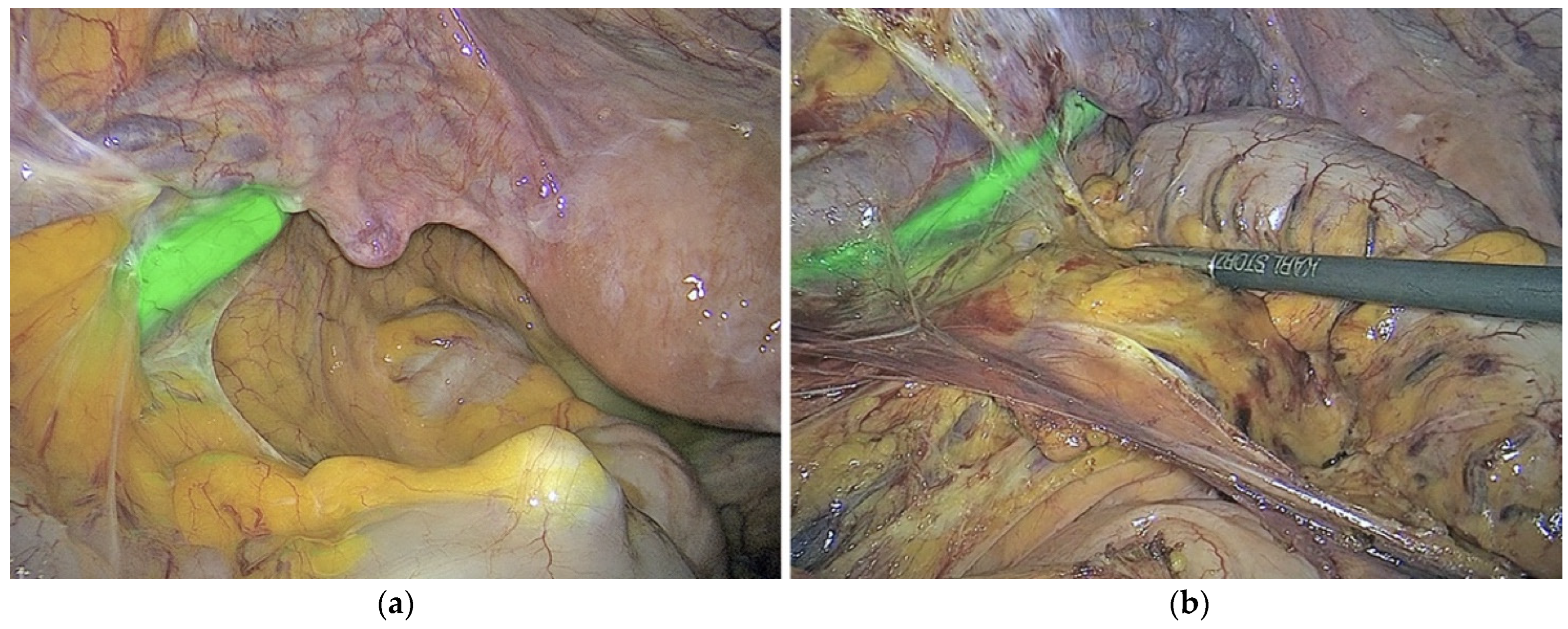
3.3. Lymph Nodes: Mapping and Metastasis
3.4. Inflammatory Bowel Disease (IBD)
3.5. Studies Quantifying Perfusion through the Association with Fluorescence and Other Methods
3.6. Estimation of Optimal Resection in Relation to Liver Function for Colorectal Metastases
3.7. Flap Assessment
3.8. Experimental Research and Results Foreshadow Future Clinical Achievements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ottobrini, L.; Martelli, C.; Lucignani, G. Optical Imaging Agents. Mol. Imaging 2021, 603–625. [Google Scholar] [CrossRef]
- Pirovano, G.; Roberts, S.; Kossatz, S.; Reiner, T. Optical Imaging Modalities: Principles and Applications in Preclinical Research and Clinical Settings. J. Nucl. Med. 2020, 61, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Huang, B.; Hu, S.; An, Y.; Li, Y.; Wang, Y.; Gu, N. Indocyanine green assembled free oxygen-nanobubbles towards enhanced near-infrared induced photodynamic therapy. Nano Res. 2022, 15, 4285–4293. [Google Scholar] [CrossRef]
- Lee, E.H.; Lee, M.K.; Lim, S.J. Enhanced stability of indocyanine green by encapsulation in zein-phosphatidylcholine hybrid nanoparticles for use in the phototherapy of cancer. Pharmaceutics 2021, 13, 305. [Google Scholar] [CrossRef]
- Qing, W.; Xing, X.; Feng, D.; Chen, R.; Liu, Z. Indocyanine green loaded pH-responsive bortezomib supramolecular hydrogel for synergistic chemo-photothermal/photodynamic colorectal cancer therapy. Photodiagn. Photodyn. Ther. 2021, 36, 102521. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, V.; Cao, T.G.N.; Choi, D.G.; Kang, H.C.; Shim, M.S. Non-ionic polysorbate-based nanoparticles for efficient combination chemo/photothermal/photodynamic therapy. J. Ind. Eng. Chem. 2020, 88, 260–267. [Google Scholar] [CrossRef]
- Bi, Z.; Huang, L.; Han, M.; Ma, J.; Wang, P. One-pot preparation of small lipid-indocyanine green nanoparticles to induced intracellular oxidative/thermal stress damage for effective colorectal cancer therapy. Micro Nano Lett. 2021, 16, 636–642. [Google Scholar] [CrossRef]
- Choi, H.W.; Lim, J.H.; Kim, C.W.; Lee, E.; Kim, J.-M.; Chang, K.; Chung, B.G. Near-Infrared Light-Triggered Generation of Reactive Oxygen Species and Induction of Local Hyperthermia from Indocyanine Green Encapsulated Mesoporous Silica-Coated Graphene Oxide for Colorectal Cancer Therapy. Antioxidants 2022, 11, 174. [Google Scholar] [CrossRef]
- Liu, C.; Ruan, C.; Shi, R.; Jiang, B.P.; Ji, S.; Shen, X.C. A near infrared-modulated thermosensitive hydrogel for stabilization of indocyanine green and combinatorial anticancer phototherapy. Biomater. Sci. 2019, 7, 1705–1715. [Google Scholar] [CrossRef]
- Ren, L.; Nie, J.; Wei, J.; Li, Y.; Yin, J.; Yang, X.; Chen, G. RGD-targeted redox responsive nano micelle: Co-loading docetaxel and indocyanine green to treat the tumor. Drug Deliv. 2021, 28, 2024–2032. [Google Scholar] [CrossRef]
- Wu, H.; You, C.; Jiao, J.; Chen, F.; Sun, B.; Zhu, X. A novel near-infrared triggered dual-targeted nanoplatform for mitochondrial combined photothermal-chemotherapy of cancer in vitro. Nanotechnology 2019, 30, 035601. [Google Scholar] [CrossRef]
- Zhou, X.; Liang, J.; Liu, Q.; Huang, D.; Xu, J.; Gu, H.; Xue, W. Codelivery of epigallocatechin-3-gallate and diallyl trisulfide by near-infrared light-responsive mesoporous polydopamine nanoparticles for enhanced antitumor efficacy. Int. J. Pharm. 2021, 592, 120020. [Google Scholar] [CrossRef] [PubMed]
- Obinu, A.; Gavini, E.; Rassu, G.; Riva, F.; Calligaro, A.; Bonferoni, M.C.; Maestri, M.; Giunchedi, P. Indocyanine green loaded polymeric nanoparticles: Physicochemical characterization and interaction studies with caco-2 cell line by light and transmission electron microscopy. Nanomaterials 2020, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Xu, C.; Yang, S.; Zhang, Z.; Wei, Z.; Wu, M.; Xue, F. Vehicle-Free Nanotheranostic Self-Assembled from Clinically Approved Dyes for Cancer Fluorescence Imaging and Photothermal/Photodynamic Combinational Therapy. Pharmaceutics 2020, 14, 1074. [Google Scholar] [CrossRef] [PubMed]
- Morales-Conde, S.; Licardie, E.; Alarcón, I.; Balla, A. Indocyanine green (ICG) fluorescence guide for the use and indications in general surgery: Recommendations based on the descriptive review of the literature and the analysis of experience. Cirugía Española 2022, 100, 534–554. [Google Scholar] [CrossRef]
- Ghuman, A.; Kavalukas, S.; Sharp, S.P.; Wexner, S.D. Clinical role of fluorescence imaging in colorectal surgery—An updated review. Expert Rev. Med. Devices 2020, 17, 1277–1283. [Google Scholar] [CrossRef]
- Thammineedi, S.R.; Saksena, A.R.; Nusrath, S.; Iyer, R.R.; Shukla, S.; Patnaik, S.C.; Reddy, R.P.; Boleneni, N.; Sharma, R.M.; Smith, L.; et al. Fluorescence-guided cancer surgery—A new paradigm. J. Surg. Oncol. 2021, 123, 1679–1698. [Google Scholar] [CrossRef]
- Martínez-López, E.; Martínez-Pérez, A.; Navarro-Martínez, S.; Sebastián-Tomás, J.C.; de’Angelis, N.; García-Granero, E. Real-time fluorescence image-guided gastrointestinal oncologic surgery: Towards a new era. World J. Gastrointest. Oncol. 2021, 13, 1029–1042. [Google Scholar] [CrossRef]
- Heeman, W.; Vonk, J.; Ntziachristos, V.; Pogue, B.W.; Dierckx, R.A.J.O.; Kruijff, S.; van Dam, G.M. A guideline for clinicians performing clinical studies with fluorescence imaging. J. Nucl. Med. 2022, 63, 640–645. [Google Scholar] [CrossRef]
- Lütken, C.D.; Achiam, M.P.; Svendsen, M.B.; Boni, L.; Nerup, N. Optimizing quantitative fluorescence angiography for visceral perfusion assessment. Surg. Endosc. 2020, 34, 5223–5233. [Google Scholar] [CrossRef]
- Noltes, M.E.; Metman, M.J.H.; Heeman, W.; Rotstein, L.; van Ginhoven, T.M.; Vriens, M.R.; Engelsman, A.F.; Boerma, E.C.; Brouwers, A.H.; van Dam, G.M.; et al. A Novel and Generic Workflow of Indocyanine Green Perfusion Assessment Integrating Standardization and Quantification Toward Clinical Implementation. Ann. Surg. 2021, 274, E659–E663. [Google Scholar] [CrossRef] [PubMed]
- Slooter, M.D.; Mansvelders, M.S.E.; Bloemen, P.R.; Gisbertz, S.S.; Bemelman, W.A.; Tanis, P.J.; Hompes, R.; van Berge Henegouwen, M.I.; de Bruin, D.M. Defining indocyanine green fluorescence to assess anastomotic perfusion during gastrointestinal surgery: Systematic review. BJS Open 2021, 5, zraa074. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.M.; Son, G.M.; Lee, I.Y.; Park, S.H.; Kim, N.S.; Baek, K.R. Optimization of indocyanine green angiography for colon perfusion during laparoscopic colorectal surgery. Color. Dis. 2021, 23, 1848–1859. [Google Scholar] [CrossRef] [PubMed]
- Gosvig, K.; Jensen, S.S.; Qvist, N.; Nerup, N.; Agnus, V.; Diana, M.; Ellebæk, M.B. Quantification of ICG fluorescence for the evaluation of intestinal perfusion: Comparison between two software-based algorithms for quantification. Surg. Endosc. 2020, 35, 5043–5050. [Google Scholar] [CrossRef] [PubMed]
- Gorpas, D.; Ntziachristos, V.; Tian, J. Principles and Practice of Intraoperative Fluorescence Imaging. Mol. Imaging 2021, 143–152. [Google Scholar] [CrossRef]
- Gomez-Rosado, J.C.; Valdes-Hernandez, J.; Cintas-Catena, J.; Cano-Matias, A.; Perez-Sanchez, A.; Del Rio-Lafuente, F.J.; Torres-Arcos, C.; Lara-Fernandez, Y.; Capitan-Morales, L.C.; Oliva-Mompean, F. Feasibility of quantitative analysis of colonic perfusion using indocyanine green to prevent anastomotic leak in colorectal surgery. Surg. Endosc. 2022, 36, 1688–1695. [Google Scholar] [CrossRef]
- Son, G.M.; Kwon, M.S.; Kim, Y.; Kim, J.; Kim, S.H.; Lee, J.W. Quantitative analysis of colon perfusion pattern using indocyanine green (ICG) angiography in laparoscopic colorectal surgery. Surg. Endosc. 2018, 33, 1640–1649. [Google Scholar] [CrossRef]
- Nerup, N.; Svendsen, M.B.S.; Rønn, J.H.; Konge, L.; Svendsen, L.B.; Achiam, M.P. Quantitative fluorescence angiography aids novice and experienced surgeons in performing intestinal resection in well-perfused tissue. Surg. Endosc. 2021, 36, 2373–2381. [Google Scholar] [CrossRef]
- Tang, G.; Du, D.; Tao, J.; Wei, Z. Effect of Indocyanine Green Fluorescence Angiography on Anastomotic Leakage in Patients Undergoing Colorectal Surgery: A Meta-Analysis of Randomized Controlled Trials and Propensity-Score-Matched Studies. Front. Surg. 2022, 9, 815753. [Google Scholar] [CrossRef]
- Vargas, H.D.; Margolin, D.A. Anastomotic Construction. In The ASCRS Textbook of Colon and Rectal Surgery; Springer: Cham, Switzerland, 2022; pp. 157–187. [Google Scholar] [CrossRef]
- Trastulli, S.; Munzi, G.; Desiderio, J.; Cirocchi, R.; Rossi, M.; Parisi, A. Indocyanine green fluorescence angiography versus standard intraoperative methods for prevention of anastomotic leak in colorectal surgery: Meta-analysis. Br. J. Surg. 2021, 108, 359–372. [Google Scholar] [CrossRef]
- Neddermeyer, M.; Kanngießer, V.; Maurer, E.; Bartsch, D.K. Indocyanine Green Near-Infrared Fluoroangiography Is a Useful Tool in Reducing the Risk of Anastomotic Leakage Following Left Colectomy. Front. Surg. 2022, 9, 850256. [Google Scholar] [CrossRef] [PubMed]
- Brescia, A.; Muttillo, E.M.; Angelicone, I.; Madaffari, I.; Maggi, F.; Sperduti, I.; Gasparrini, M.; Osti, M.F. The Role of Indocyanine Green in Laparoscopic Low Anterior Resections for Rectal Cancer Previously Treated with Chemo-radiotherapy: A Single-center Retrospective Analysis. Anticancer. Res. 2022, 42, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Meijer, R.P.J.; van Manen, L.; Hartgrink, H.H.; Burggraaf, J.; Gioux, S.; Vahrmeijer, A.L.; Mieog, J.S.D. Quantitative dynamic near-infrared fluorescence imaging using indocyanine green for analysis of bowel perfusion after mesenteric resection. J. Biomed. Opt. 2021, 26, 060501. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.C.; Lee, J.L.; Park, S.H. Interpretative Guidelines and Possible Indications for Indocyanine Green Fluorescence Imaging in Robot-Assisted Sphincter-Saving Operations. Dis. Colon Rectum 2017, 60, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Hamabe, A.; Okita, K.; Nishidate, T.; Okuya, K.; Usui, A.; Akizuki, E.; Satoyoshi, T.; Takemasa, I. Efficacy of indocyanine green fluorescence angiography in preventing anastomotic leakage after laparoscopic colorectal cancer surgery. Int. J. Color. Dis. 2019, 35, 269–275. [Google Scholar] [CrossRef]
- Higashijima, J.; Yashimoto, T.; Eto, S.; Kashihara, H.; Takasu, C.; Nishi, M.; Tokunaga, T.; Yoshikawa, K.; Shimada, M. Strategy to avoid anastomotic leakage in laparoscopic colorectal resection using the indocyanine green fluorescence system. Int. Surg. 2021, 105, 714–719. [Google Scholar] [CrossRef]
- Aiba, T.; Uehara, K.; Ogura, A.; Tanaka, A.; Yonekawa, Y.; Hattori, N.; Nakayama, G.; Kodera, Y.; Ebata, T.; Nagino, M. The significance of the time to arterial perfusion in intraoperative ICG angiography during colorectal surgery. Surg. Endosc. 2021, 35, 7227–7235. [Google Scholar] [CrossRef]
- Kawada, K.; Wada, T.; Yamamoto, T.; Itatani, Y.; Hida, K.; Obama, K. Correlation between Colon Perfusion and Postoperative Fecal Output through a Transanal Drainage Tube during Laparoscopic Low Anterior Resection. Cancers 2022, 14, 2328. [Google Scholar] [CrossRef]
- Gräfitsch, A.; Kirchhoff, P.; Soysal, S.D.; Däster, S.; Hoffmann, H. Dynamic Serosal Perfusion Assessment during Colorectal Resection Using Visible Light Spectroscopy. Eur. Surg. Res. 2021, 62, 25–31. [Google Scholar] [CrossRef]
- Seeliger, B.; Agnus, V.; Mascagni, P.; Barberio, M.; Longo, F.; Lapergola, A.; Mutter, D.; Klymchenko, A.S.; Chand, M.; Marescaux, J.; et al. Simultaneous computer-assisted assessment of mucosal and serosal perfusion in a model of segmental colonic ischemia. Surg. Endosc. 2020, 34, 4818–4827. [Google Scholar] [CrossRef]
- Han, S.R.; Lee, C.S.; Bae, J.H.; Lee, H.J.; Yoon, M.R.; Al-Sawat, A.; Lee, D.S.; Lee, I.K.; Lee, Y.S. Quantitative evaluation of colon perfusion after high versus low ligation in rectal surgery by indocyanine green: A pilot study. Surg. Endosc. 2022, 36, 3511–3519. [Google Scholar] [CrossRef]
- Munechika, T.; Kajitani, R.; Matsumoto, Y.; Nagano, H.; Komono, A.; Aisu, N.; Morimoto, M.; Yoshimatsu, G.; Yoshida, Y.; Hasegawa, S. Safety and effectiveness of high ligation of the inferior mesenteric artery for cancer of the descending colon under indocyanine green fluorescence imaging: A pilot study. Surg. Endosc. 2020, 35, 1696–1702. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, H.; Matsuda, K.; Hayami, S.; Tamura, K.; Mitani, Y.; Mizumoto, Y.; Nakamura, Y.; Murakami, D.; Ueno, M.; Yokoyama, S.; et al. Quantitative Indocyanine Green Fluorescence Imaging Used to Predict Anastomotic Leakage Focused on Rectal Stump During Laparoscopic Anterior Resection. J. Laparoendosc. Adv. Surg. Tech. 2020, 30, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Hara, K.; Kitagawa, T.; Okamoto, A.; Marukuchi, R.; Ito, R.; Nakabayashi, Y. Fluorescence vessel and ureter navigation during laparoscopic lateral lymph node dissection. Langenbeck’s Arch. Surg. 2022, 407, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Gila-Bohórquez, A.; Gómez-Menchero, J.; García-Moreno, J.L.; Suárez-Grau, J.M.; Guadalajara-Jurado, J.F. Utility of indocyanine green for intraoperative localization of ureter in complex colo-rectal surgery. Cirugía Española 2019, 97, 233–234. [Google Scholar] [CrossRef]
- Kim, H.J.; Choi, G.S.; Park, J.S.; Park, S.Y.; Lee, S.M.; Song, S.H. Stepwise Improvement of Surgical Quality in Robotic Lateral Pelvic Node Dissection: Lessons From 100 Consecutive Patients With Locally Advanced Rectal Cancer. Dis. Colon Rectum 2022, 65, 599–607. [Google Scholar] [CrossRef]
- Kumar, S.P.; Ahmed, Z.; Mudambadithaya, R.R.; Shetty, M.G.; Rao, G.V.; Rao, T.S.; Rebala, P. Indocyanine Green Near Infrared Fluorescence Imaging for Colonic Conduit Perfusion Assessment in Colon Pull-up for Corrosive Oesophageal Strictures. Indian J. Surg. 2022, 84, 1269–1275. [Google Scholar] [CrossRef]
- Spagnolo, E.; Zapardiel, I.; Gorostidi, M. Role of fluorescence imaging for intraoperative intestinal assessment in gynecological surgery: A systematic review. Minim. Invasive Ther. Allied Technol. 2022, 31, 992–999. [Google Scholar] [CrossRef]
- Ianieri, M.M.; Della Corte, L.; Campolo, F.; Cosentino, F.; Catena, U.; Bifulco, G.; Scambia, G. Indocyanine green in the surgical management of endometriosis: A systematic review. Acta Obstet. Gynecol. Scand. 2020, 100, 189–199. [Google Scholar] [CrossRef]
- Raimondo, D.; Maletta, M.; Malzoni, M.; Cosentino, F.; Scambia, G.; Falcone, F.; Coppola, M.; Turco, L.C.; Borghese, G.; Raffone, A.; et al. Indocyanine green fluorescence angiography after full-thickness bowel resection for rectosigmoid endometriosis: A multicentric experience with quantitative analysis. Int. J. Gynecol. Obstet. 2021, 158, 679–688. [Google Scholar] [CrossRef]
- Li, H.; Hou, X.; Lin, R.; Fan, M.; Pang, S.; Jiang, L.; Liu, Q.; Fu, L. Advanced endoscopic methods in gastrointestinal diseases: A systematic review. Quant. Imaging Med. Surg. 2019, 9, 905–920. [Google Scholar] [CrossRef]
- González-Abós, C.; Selva, A.B.; de Lacy, F.B.; Valverde, S.; Almenara, R.; Lacy, A.M. Quantitative Indocyanine Green Fluorescence Imaging Assessment for Nonmucinous Peritoneal Metastases: Preliminary Results of the ICCP Study. Dis. Colon Rectum 2021, 65, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Moran, B.J. Detecting and Managing Colorectal Peritoneal Metastases: Some “Light at the End of the Tunnel”. Dis. Colon Rectum 2021, 65, 301–303. [Google Scholar] [CrossRef] [PubMed]
- Lieto, E.; Auricchio, A.; Cardella, F.; Mabilia, A.; Basile, N.; Castellano, P.; Orditura, M.; Galizia, G. Fluorescence-Guided Surgery in the Combined Treatment of Peritoneal Carcinomatosis from Colorectal Cancer: Preliminary Results and Considerations. World J. Surg. 2017, 42, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Lwin, T.M.; Turner, M.A.; Amirfakhri, S.; Nishino, H.; Hoffman, R.M.; Bouvet, M. Fluorescence Molecular Targeting of Colon Cancer to Visualize the Invisible. Cells 2022, 11, 249. [Google Scholar] [CrossRef]
- Mieog, J.S.D.; Achterberg, F.B.; Zlitni, A.; Hutteman, M.; Burggraaf, J.; Swijnenburg, R.J.; Gioux, S.; Vahrmeijer, A.L. Fundamentals and developments in fluorescence-guided cancer surgery. Nat. Rev. Clin. Oncol. 2021, 19, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Privitera, L.; Paraboschi, I.; Dixit, D.; Arthurs, O.J.; Giuliani, S. Image-guided surgery and novel intraoperative devices for enhanced visualisation in general and paediatric surgery: A review. Innov. Surg. Sci. 2022, 6, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, T.; Nakamura, Y.A.; Choyke, P.L.; Kobayashi, H. Fluorescence-guided surgery. Front. Oncol. 2017, 7, 314. [Google Scholar] [CrossRef]
- Tipirneni, K.E.; Warram, J.M.; Moore, L.S.; Prince, A.C.; de Boer, E.; Jani, A.H.; Wapnir, I.L.; Liao, J.C.; Bouvet, M.; Behnke, N.K.; et al. Oncologic Procedures Amenable to Fluorescence-guided Surgery. Ann. Surg. 2017, 266, 36–47. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, G.; Zeng, Z.; Pu, K. Activatable molecular probes for fluorescence-guided surgery, endoscopy and tissue biopsy. Chem. Soc. Rev. 2022, 51, 566–593. [Google Scholar] [CrossRef]
- Liu, H.; Xu, C.; Meng, M.; Li, S.; Sheng, S.; Zhang, S.; Ni, W.; Tian, H.; Wang, Q. Metal-organic framework-mediated multifunctional nanoparticles for combined chemo-photothermal therapy and enhanced immunotherapy against colorectal cancer. Acta Biomater. 2022, 144, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Villegas-Tovar, E.; Jimenez-Lillo, J.; Jimenez-Valerio, V.; Diaz-Giron-Gidi, A.; Faes-Petersen, R.; Otero-Piñeiro, A.; De Lacy, F.B.; Martinez-Portilla, R.J.; Lacy, A.M. Performance of Indocyanine green for sentinel lymph node mapping and lymph node metastasis in colorectal cancer: A diagnostic test accuracy meta-analysis. Surg. Endosc. 2020, 34, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Torres, V.C.; He, Y.; Xu, X.; Basheer, Y.; Papavasiliou, G.; Samkoe, K.S.; Brankov, J.G.; Tichauer, K.M. Intraoperative Detection of Micrometastases in Whole Excised Lymph Nodes Using Fluorescent Paired-Agent Imaging Principles: Identification of a Suitable Staining and Rinsing Protocol. Mol. Imaging Biol. 2021, 23, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Lobbes, L.A.; Berns, S.; Warschkow, R.; Schmidt, L.R.; Schineis, C.; Strobel, R.M.; Lauscher, J.C.; Beyer, K.; Weixler, B. Perfusion Visualization during Ileal J-Pouch Formation—A Proposal for the Standardization of Intraoperative Imaging with Indocyanine Green Near-Infrared Fluorescence and a Postoperative Follow-Up in IBD Surgery. Life 2022, 12, 668. [Google Scholar] [CrossRef]
- Spinelli, A.; Carvello, M.; Kotze, P.G.; Maroli, A.; Montroni, I.; Montorsi, M.; Buchs, N.C.; Ris, F. Ileal pouch–anal anastomosis with fluorescence angiography: A case-matched study. Color. Dis. 2019, 21, 827–832. [Google Scholar] [CrossRef]
- Soares, A.S.; Bano, S.; Clancy, N.T.; Stoyanov, D.; Lovat, L.B.; Chand, M. Multisensor perfusion assessment cohort study: Preliminary evidence toward a standardized assessment of indocyanine green fluorescence in colorectal surgery. Surgery 2022, 172, 69–73. [Google Scholar] [CrossRef]
- Egi, H.; Ohnishi, K.; Akita, S.; Sugishita, H.; Ogi, Y.; Yoshida, M.; Koga, S.; Kikuchi, S.; Matsumoto, H.; Kuwabara, J.; et al. The arrival time of indocyanine green in tissues can be a quantitative index because of its correlation with tissue oxygen saturation: A clinical pilot study. Asian J. Endosc. Surg. 2022, 15, 432–436. [Google Scholar] [CrossRef]
- Mehdorn, M.; Ebel, S.; Köhler, H.; Gockel, I.; Jansen-Winkeln, B. Hyperspectral imaging and indocyanine green fluorescence angiography in acute mesenteric ischemia: A case report on how to visualize intestinal perfusion. Int. J. Surg. Case Rep. 2021, 82, 105853. [Google Scholar] [CrossRef]
- Barberio, M.; Felli, E.; Seyller, E.; Longo, F.; Chand, M.; Gockel, I.; Geny, B.; Swanström, L.; Marescaux, J.; Agnus, V.; et al. Quantitative fluorescence angiography versus hyperspectral imaging to assess bowel ischemia: A comparative study in enhanced reality. Surgery 2020, 168, 178–184. [Google Scholar] [CrossRef]
- Quero, G.; Lapergola, A.; Barberio, M.; Seeliger, B.; Saccomandi, P.; Guerriero, L.; Mutter, D.; Saadi, A.; Worreth, M.; Marescaux, J.; et al. Discrimination between arterial and venous bowel ischemia by computer-assisted analysis of the fluorescent signal. Surg. Endosc. 2018, 33, 1988–1997. [Google Scholar] [CrossRef]
- Pfahl, A.; Radmacher, G.K.; Köhler, H.; Maktabi, M.; Neumuth, T.; Melzer, A.; Gockel, I.; Chalopin, C.; Jansen-Winkeln, B. Combined indocyanine green and quantitative perfusion assessment with hyperspectral imaging during colorectal resections. Biomed. Opt. Express 2022, 13, 3145–3160. [Google Scholar] [CrossRef] [PubMed]
- Jansen-Winkeln, B.; Dvorak, M.; Köhler, H.; Maktabi, M.; Mehdorn, M.; Chalopin, C.; Diana, M.; Gockel, I.; Barberio, M. Border Line Definition Using Hyperspectral Imaging in Colorectal Resections. Cancers 2022, 14, 1188. [Google Scholar] [CrossRef] [PubMed]
- Kojima, S.; Sakamoto, T.; Nagai, Y.; Matsui, Y.; Nambu, K.; Masamune, K. Laser Speckle Contrast Imaging for Intraoperative Quantitative Assessment of Intestinal Blood Perfusion During Colorectal Surgery: A Prospective Pilot Study. Surg. Innov. 2019, 26, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Rønn, J.H.; Nerup, N.; Strandby, R.B.; Svendsen, M.B.S.; Ambrus, R.; Svendsen, L.B.; Achiam, M.P. Laser speckle contrast imaging and quantitative fluorescence angiography for perfusion assessment. Langenbeck’s Arch. Surg. 2019, 404, 505–515. [Google Scholar] [CrossRef]
- Joosten, J.J.; Longchamp, G.; Khan, M.F.; Lameris, W.; van Berge Henegouwen, M.I.; Bemelman, W.A.; Cahill, R.A.; Hompes, R.; Ris, F. The use of fluorescence angiography to assess bowel viability in the acute setting: An international, multi-centre case series. Surg. Endosc. 2022, 36, 7369–7375. [Google Scholar] [CrossRef]
- Vaassen, H.; Wermelink, B.; Geelkerken, B.; Lips, D. Fluorescence Angiography for Peri-Operative Assessment of Bowel Viability in Patients with Mesenteric Ischaemia. EJVES Vasc. Forum 2022, 54, e53–e54. [Google Scholar] [CrossRef]
- Park, S.H.; Park, H.M.; Baek, K.R.; Ahn, H.M.; Lee, I.Y.; Son, G.M. Artificial intelligence based real-time microcirculation analysis system for laparoscopic colorectal surgery. World J. Gastroenterol. 2020, 26, 6945–6962. [Google Scholar] [CrossRef]
- D’Urso, A.; Agnus, V.; Barberio, M.; Seeliger, B.; Marchegiani, F.; Charles, A.L.; Geny, B.; Marescaux, J.; Mutter, D.; Diana, M. Computer-assisted quantification and visualization of bowel perfusion using fluorescence-based enhanced reality in left-sided colonic resections. Surg. Endosc. 2020, 35, 4321–4331. [Google Scholar] [CrossRef]
- Tokunaga, T.; Shimada, M.; Higashijima, J.; Yoshikawa, K.; Nishi, M.; Kashihara, H.; Takasu, C.; Ishikawa, D.; Yoshimoto, T. Intraoperative thermal imaging for evaluating blood perfusion during laparoscopic colorectal surgery. Surg. Laparosc. Endosc. Percutaneous Tech. 2021, 31, 281–284. [Google Scholar] [CrossRef]
- Knospe, L.; Gockel, I.; Jansen-Winkeln, B.; Thieme, R.; Niebisch, S.; Moulla, Y.; Stelzner, S.; Lyros, O.; Diana, M.; Marescaux, J.; et al. New intraoperative imaging in oncological visceral surgery. Best Pract. Onkol. 2021, 16, 268–276. [Google Scholar] [CrossRef]
- Morris-Stiff, G.; Gomez, D.; Prasad, R. Quantitative assessment of hepatic function and its relevance to the liver surgeon. J. Gastrointest. Surg. 2008, 13, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Asano, Y.; Ito, M.; Arakawa, S.; Kawabe, N.; Shimura, M.; Koike, D.; Hayashi, C.; Ochi, T.; Kamio, K.; et al. Right hepatectomy with preservation of the entire caudate lobe in patients with metastatic liver: A case of a new hepatectomy technique and treatment strategy for patients with marginal liver function. BMC Surg. 2022, 22, 17. [Google Scholar] [CrossRef]
- Li, Y.; You, Q.; Wang, Z.; Cao, Y.; Butch, C.J.; Guissi, N.E.I.; Cai, H.; Wang, Y.; Lu, Q. A study on setting standards for near-infrared fluorescence-image guided surgery (NIRFGS) time lapse monitoring based on preoperative liver function assessment. Ann. Transl. Med. 2022, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Lobbes, L.A.; Hoveling, R.J.M.; Schmidt, L.R.; Berns, S.; Weixler, B. Objective Perfusion Assessment in Gracilis Muscle Interposition—A Novel Software-Based Approach to Indocyanine Green Derived Near-Infrared Fluorescence in Reconstructive Surgery. Life 2022, 12, 278. [Google Scholar] [CrossRef] [PubMed]
- Bach Korsholm Knudsen, K.; Nerup, N.; Thorup, J.; Thymann, T.; Sangild, P.T.; Svendsen, L.B.; Achiam, M.; Svendsen, M.B.S.; Lauritsen, T.; Leth Maroun, L.; et al. Intestinal perfusion assessed by quantitative fluorescence angiography in piglets with necrotizing enterocolitis. J. Pediatr. Surg. 2022, 57, 747–752. [Google Scholar] [CrossRef]
- Dip, F.; Bregoli, P.; Falco, J.; White, K.P.; Rosenthal, R.J. Nerve autofluorescence in near-ultraviolet light markedly enhances nerve visualization in vivo. Surg. Endosc. 2021, 36, 1999–2005. [Google Scholar] [CrossRef]
- Barth, C.W.; Amling, C.L.; Gibbs, S.L. Fluorescent nerve identification in resected human tissue specimens. SPIE Proc. 2019, 10862, 108620R. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, D.; Li, J.; Li, F.; Wei, R.; Jiang, G.; Xu, H.; Wang, X.; Zhou, Y.; Xi, L. A novel ICG-labeled cyclic TMTP1 peptide dimer for sensitive tumor imaging and enhanced photothermal therapy in vivo. Eur. J. Med. Chem. 2022, 227, 113935. [Google Scholar] [CrossRef]
- Gulfam, M.; Jo, S.H.; Jo, S.W.; Vu, T.T.; Park, S.H.; Lim, K.T. Highly porous and injectable hydrogels derived from cartilage acellularized matrix exhibit reduction and NIR light dual-responsive drug release properties for application in antitumor therapy. NPG Asia Mater. 2022, 14, 8. [Google Scholar] [CrossRef]
- Polom, W.; Migaczewski, M.; Skokowski, J.; Swierblewski, M.; Cwalinski, T.; Kalinowski, L.; Pedziwiatr, M.; Matuszewski, M.; Polom, K. Multispectral Imaging Using Fluorescent Properties of Indocyanine Green and Methylene Blue in Colorectal Surgery—Initial Experience. J. Clin. Med. 2022, 11, 368. [Google Scholar] [CrossRef]
- van Beurden, F.; van Willigen, D.M.; Vojnovic, B.; van Oosterom, M.N.; Brouwer, O.R.; der Poel, H.G.V.; Kobayashi, H.; van Leeuwen, F.W.B.; Buckle, T. Multi-Wavelength Fluorescence in Image-Guided Surgery, Clinical Feasibility and Future Perspectives. Mol. Imaging 2020, 19, 1536012120962333. [Google Scholar] [CrossRef]
- Lee, S.S.; Kim, H.; Sohn, D.K.; Eom, J.B.; Seo, Y.S.; Yoon, H.M.; Choi, Y. Indocyanine green-loaded injectable alginate hydrogel as a marker for precision cancer surgery. Quant. Imaging Med. Surg. 2020, 10, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Marston, J.C.; Kennedy, G.D.; Lapi, S.E.; Hartman, Y.E.; Richardson, M.T.; Modi, H.M.; Warram, J.M. Panitumumab-IRDye800CW for Fluorescence-Guided Surgical Resection of Colorectal Cancer. J. Surg. Res. 2019, 239, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Kan, X.; Zhou, G.; Zhang, F.; Ji, H.; Zheng, H.; Chick, J.F.B.; Valji, K.; Zheng, C.; Yang, X. Interventional optical imaging permits instant visualization of pathological zones of ablated tumor periphery and residual tumor detection. Cancer Res. 2021, 81, 4594–4602. [Google Scholar] [CrossRef] [PubMed]
- Welch, M.G.; Margolis, K.G.; Li, Z.; Gershon, M.D. Oxytocin regulates gastrointestinal motility, inflammation, macromolecular permeability, and mucosal maintenance in mice. Am. J. Physiol. Liver Physiol. 2014, 307, G848–G862. [Google Scholar] [CrossRef]
- Padurariu, M.; Antioch, I.; Ciobica, A.; Lefter, R.; Simion, L. Intranasal oxytocin in Autism: Models, pain and oxidative stress. Rev. Chim. 2017, 68, 1879–1883. [Google Scholar] [CrossRef]
- Bergenheim, F.; Seidelin, J.B.; Pedersen, M.T.; Mead, B.E.; Jensen, K.B.; Karp, J.M.; Nielsen, O.H. Fluorescence-based tracing of transplanted intestinal epithelial cells using confocal laser endomicroscopy. Stem Cell Res. Ther. 2019, 10, 148. [Google Scholar] [CrossRef]
- Biochemical Effects of Biological Supports-Included Stem Cells on Eye Cells Development|Publons. Available online: https://publons.com/publon/18624290/ (accessed on 25 May 2022).
- Woo, Y.; Chaurasiya, S.; O’Leary, M.; Han, E.; Fong, Y. Fluorescent imaging for cancer therapy and cancer gene therapy. Mol. Ther. Oncolytics 2021, 23, 231–238. [Google Scholar] [CrossRef]
- De Galitiis, F.; Cannita, K.; Tessitore, A.; Martella, F.; Di Rocco, Z.C.; Russo, A.; Adamo, V.; Iacobelli, S.; Martinotti, S.; Marchetti, P.; et al. Consorzio Interuniversitario Nazionale Bio-Oncologia Novel P53 mutations detected by FAMA in colorectal cancers. Ann. Oncol. 2006, 17 (Suppl. 7), vii78–vii83. [Google Scholar] [CrossRef]
- Murarasu, D.; Puiu, L.; Mihalcea, C.E.; Aldea Pitica, I.M.; Mambet, C.; Radu, E.L.; Matei, L.; Dragu, D.L.; Simion, L.; Marincas, M.A. Characterization of TP53 polymorphisms in the Romania colorectal cancer patients. Rom. Biotechnol. Lett. 2017, 23, 14124–14134. [Google Scholar]
- Murarasu, D.; Puiu, L.; Aldea Pitica, I.M.; Mambet, C.; Mihalcea, C.E.; Marincas, M.A.; Cinca, S.; Brasoveanu, L.; Diaconu, C.C. TP53 somatic mutations and LOH profile in colorectal cancer in Romania. Rom. Biotechnol. Lett. 2018, 23, 13530. [Google Scholar]
- Xie, T.; Qi, Y.; Li, Y.; Zhang, F.; Li, W.; Zhong, D.; Tang, Z.; Zhou, M. Ultrasmall Ga-ICG nanoparticles based gallium ion/photodynamic synergistic therapy to eradicate biofilms and against drug-resistant bacterial liver abscess. Bioact. Mater. 2021, 6, 3812–3823. [Google Scholar] [CrossRef] [PubMed]
- Marincaş, A.M.; Cirimbei, C.; Toba, M.E.; Filimon, B.; Păun, L.; Brătucu, E. Retroperitoneal pyogenic infections with uncertain etiopathogenesis. Diagnostic and therapeutic difficulties. Chirurgia 2006, 101, 583–592. [Google Scholar] [PubMed]
- Gherghe, M.; Mutuleanu, M.-D.; Stanciu, A.E.; Irimescu, I.; Lazar, A.; Bacinschi, X.; Anghel, R.M. Quantitative Analysis of SPECT-CT Data in Metastatic Breast Cancer Patients-The Clinical Significance. Cancers 2022, 14, 273. [Google Scholar] [CrossRef] [PubMed]
- Voinea, S.; Sandru, A.; Gherghe, M.; Blidaru, A. Peculiarities of lymphatic drainage in cutaneous malignant melanoma: Clinical experience in 75 cases. Chirurgia 2014, 109, 26–33. [Google Scholar]
- Sandru, A.; Bordea, C.I.; Voinea, S.C.; Gherghe, M.; Albert, P.; Condrea, I.; Blidaru, A. Actualităţi în tratamentul chirurgical al melanomului malign cutanat [Latest approaches in the surgical treatment of cutaneous malignant melanoma]. Chirurgia 2011, 106, 301–308. (In Romanian) [Google Scholar]
- Saha, S.; Elgamal, M.; Cherry, M.; Buttar, R.; Pentapati, S.; Mukkamala, S.; Devisetty, K.; Kaushal, S.; Alnounou, M.; Singh, T.; et al. Challenging the conventional treatment of colon cancer by sentinel lymph node mapping and its role of detecting micrometastases for adjuvant chemotherapy. Clin. Exp. Metastasis 2018, 35, 463–469. [Google Scholar] [CrossRef]
- Garoufalia, Z.; Wexner, S.D. Indocyanine Green Fluorescence Guided Surgery in Colorectal Surgery. J. Clin. Med. 2023, 12, 494. [Google Scholar] [CrossRef]
- Hardy, N.P.; MacAonghusa, P.; Dalli, J.; Gallagher, G.; Epperlein, J.P.; Shields, C.; Mulsow, J.; Rogers, A.C.; Brannigan, A.E.; Conneely, J.B.; et al. Clinical application of machine learning and computer vision to indocyanine green quantification for dynamic intraoperative tissue characterisation: How to do it. Surg. Endosc. 2023, 37, 6361–6370. [Google Scholar] [CrossRef]
- Dip, F.; Menzo, E.L.; Bouvet, M.; Schols, R.M.; Sherwinter, D.; Wexner, S.D.; White, K.P.; Rosenthal, R.J. Intraoperative fluorescence imaging in different surgical fields: Consensus among 140 intercontinental experts. Surgery 2022, 172, S54–S59. [Google Scholar] [CrossRef]

| Name of the Compound | Mechanism | Property/Properties |
|---|---|---|
| ICG-NBs-O2 | ICG + free O2 nanobubbles | Exhibited better aqueous solution stability compared with free ICG. Assessed in the PDT of cancer. |
L-ICG NPs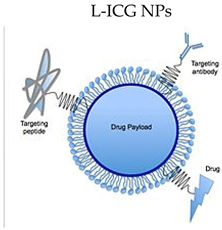 | ICG + nanoparticles | ICG’s concentration-dependent aggregation, short half-life, poor photostability, low hydrolytic stability, non-specific protein binding, and non-specific targeting limit its theranostic use in cancer therapy. ICG in NP platforms may solve these issues. |
Phosphatidylcholine-line nanoparticles (PC-NPs) and zein particles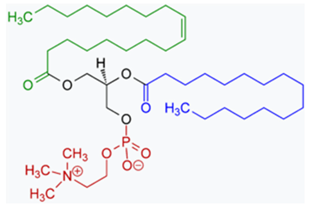 1-Oleoyl-2-palmitoyl-phosphatidylcholine | ICG + PC-NP or ICG + Z PC-NPs | It has been partially effective to stabilise ICG by encapsulating it in liposomes, phosphatidylcholine nanoparticles (PC-NP). The Z/PC-NP also inhibited ICG degradation more effectively than the PC-NP. |
Bortezomib | ICG + Bortezomib | Bortezomib is known as an anti-neoplasm drug involved in the treatment of multiple myeloma and mantle cell lymphoma, among others |
Piperlongumine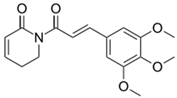 | ICG + Piperlongumine | Piperlongumine (also known as piplartine or piperlongumin) is an amide alkaloid found in the fruit of the long pepper (Piper longum) plant, which is naturally found in southeast Asia. After the excision of tumours, an alginate containing piperlongumine will be used to neutralise any remaining cancer cells. The hydrogel demonstrated efficacy in laboratory and animal investigations, and Phase I human clinical trials are programmed to start to begin in 2023 (according to the web page: https://en.wikipedia.org/wiki/Piperlongumine, accessed on 01 July 2023) |
| The Field | The Time | The Main Applications |
|---|---|---|
| Plastic surgery | 20–60 s | ICG angiography in the assessment of free flaps, pedicled flaps, or large skin paddles |
| Colorectal surgery | 30–60 s | Perfusion evaluation, intraoperative ureteral visualisation, sentinel node detection, lymphatic drainage visualisation. |
| Lymphedema surgery, thyroid and parathyroid surgery | <1 min | Indocyanine green (ICG) lymphography is used for lymphovenous anastomosis (LVA) imaging. NIR ICG fluorescence imaging is promising for real-time parathyroid gland location during thyroid surgery. |
| Gastric surgery | 11–30 min | ICG fluorescence angiography determines the primary blood supply to the proximal stomach before dissection during sleeve gastrectomy to prevent damaging/injury of these arteries. |
| Laparoscopic surgery | >30 min | Laparoscopic surgery introduced ICG fluorescence imaging to increase anatomy visualisation and to decrease the probability of injury to vital structures during dissections. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simion, L.; Ionescu, S.; Chitoran, E.; Rotaru, V.; Cirimbei, C.; Madge, O.-L.; Nicolescu, A.C.; Tanase, B.; Dicu-Andreescu, I.-G.; Dinu, D.M.; et al. Indocyanine Green (ICG) and Colorectal Surgery: A Literature Review on Qualitative and Quantitative Methods of Usage. Medicina 2023, 59, 1530. https://doi.org/10.3390/medicina59091530
Simion L, Ionescu S, Chitoran E, Rotaru V, Cirimbei C, Madge O-L, Nicolescu AC, Tanase B, Dicu-Andreescu I-G, Dinu DM, et al. Indocyanine Green (ICG) and Colorectal Surgery: A Literature Review on Qualitative and Quantitative Methods of Usage. Medicina. 2023; 59(9):1530. https://doi.org/10.3390/medicina59091530
Chicago/Turabian StyleSimion, Laurentiu, Sinziana Ionescu, Elena Chitoran, Vlad Rotaru, Ciprian Cirimbei, Octavia-Luciana Madge, Alin Codrut Nicolescu, Bogdan Tanase, Irinel-Gabriel Dicu-Andreescu, Denisa Mihaela Dinu, and et al. 2023. "Indocyanine Green (ICG) and Colorectal Surgery: A Literature Review on Qualitative and Quantitative Methods of Usage" Medicina 59, no. 9: 1530. https://doi.org/10.3390/medicina59091530
APA StyleSimion, L., Ionescu, S., Chitoran, E., Rotaru, V., Cirimbei, C., Madge, O.-L., Nicolescu, A. C., Tanase, B., Dicu-Andreescu, I.-G., Dinu, D. M., Luca, D. C., Stanculeanu, D. L., Gheorghe, A. S., Zob, D., & Marincas, M. (2023). Indocyanine Green (ICG) and Colorectal Surgery: A Literature Review on Qualitative and Quantitative Methods of Usage. Medicina, 59(9), 1530. https://doi.org/10.3390/medicina59091530








