Micropulse Laser Therapy as an Integral Part of Eye Disease Management
Abstract
1. Introduction
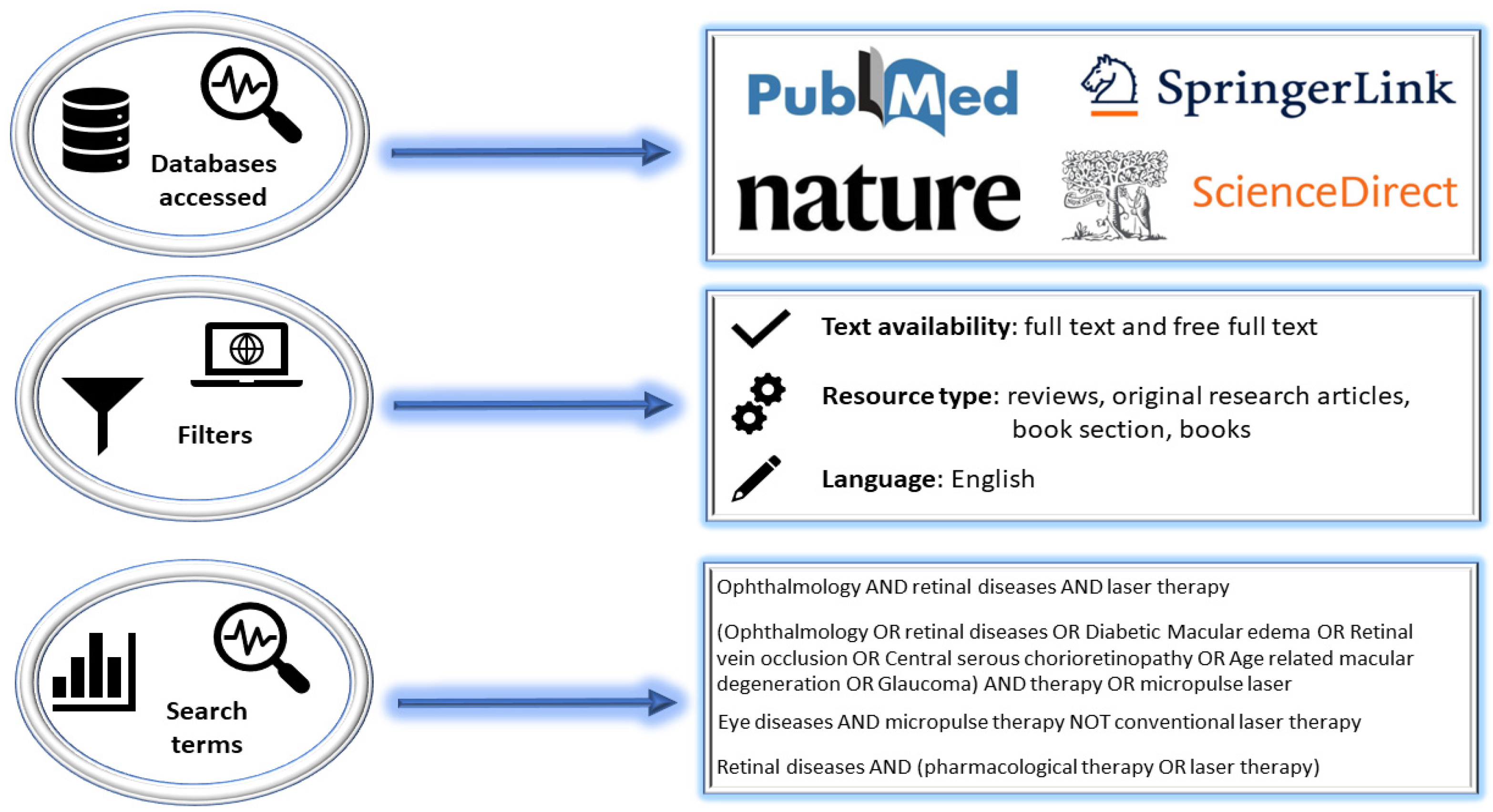
2. Research Methodology
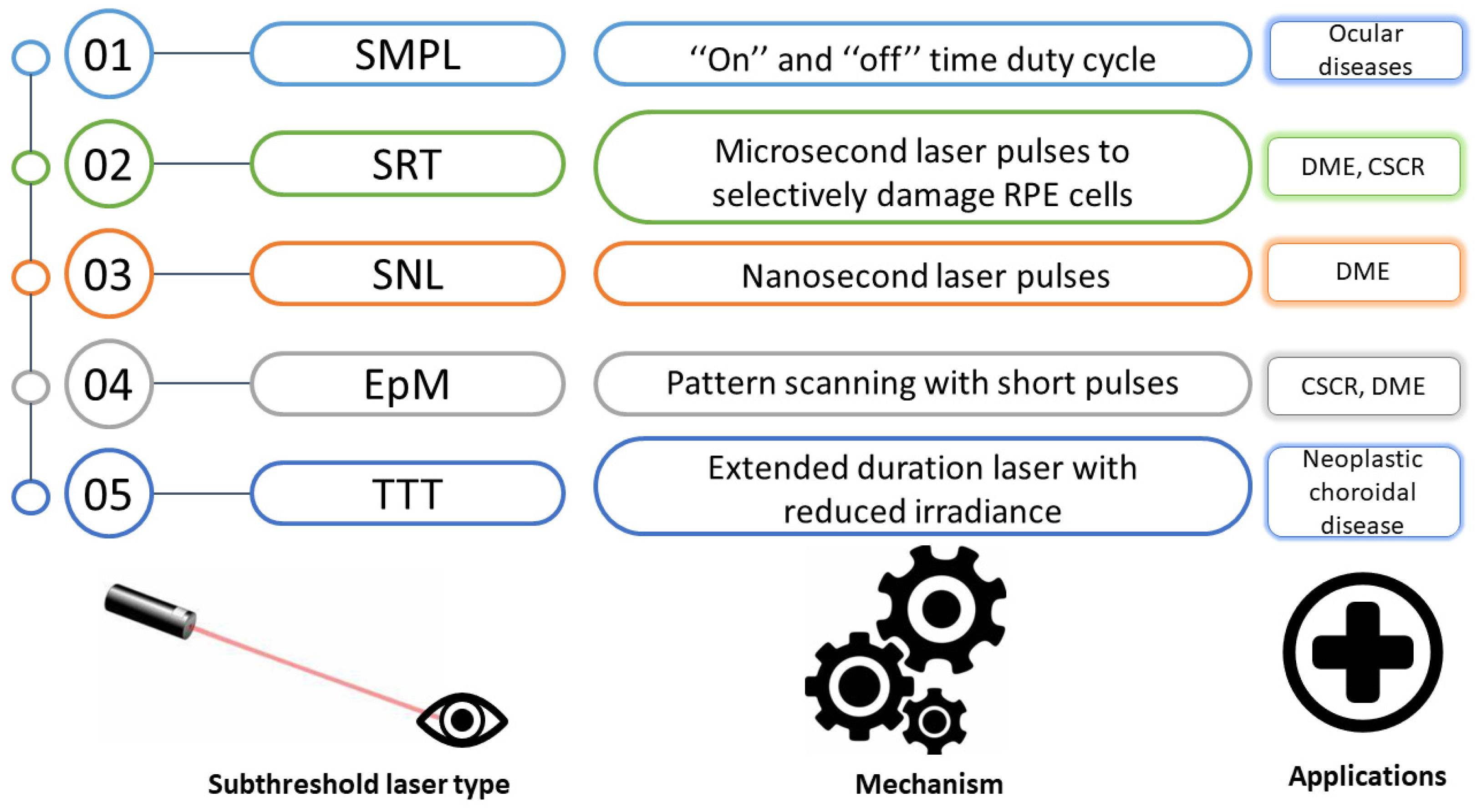
3. Types and Mechanisms of Micropulse Laser Strategies
4. Diabetic Macular Edema
5. Retinal Vein Occlusion
6. Central Serous Chorioretinopathy
7. Micropulse Laser Therapy for Age-Related Macular Degeneration
8. Glaucoma
8.1. Primary Open Angle Glaucoma
8.2. Secondary Glaucoma: Pseudoexfoliative Glaucoma
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marchesi, N.; Fahmideh, F.; Boschi, F.; Pascale, A.; Barbieri, A. Ocular Neurodegenerative Diseases: Interconnection between Retina and Cortical Areas. Cells 2021, 10, 2394. [Google Scholar] [CrossRef]
- Daruich, A.; Matet, A.; Moulin, A.; Kowalczuk, L.; Nicolas, M.; Sellam, A.; Rothschild, P.-R.; Omri, S.; Gélizé, E.; Jonet, L.; et al. Mechanisms of macular edema: Beyond the surface. Prog. Retin. Eye Res. 2018, 63, 20–68. [Google Scholar] [CrossRef]
- Ariga, M.; Nivean, M.; Utkarsha, P. Pseudoexfoliation Syndrome and Glaucoma. J. Curr. Glaucoma Pract. 2023, 7, 118–120. [Google Scholar] [CrossRef]
- Yanai, R.; Okunuki, Y.; Park, D.H.; Zunaina, E. Next Therapeutic Targets in Ocular Diseases. Front. Med. 2022, 9, 953377. [Google Scholar] [CrossRef]
- Mikalauskiene, L.; Grzybowski, A.; Zemaitiene, R. Ocular Surface Changes Associated with Ophthalmic Surgery. J. Clin. Med. 2021, 10, 1642. [Google Scholar] [CrossRef]
- Zhang, A.Y.; Kumar, D.; Tripathy, K. Laser Principles in Ophthalmology; StatPearls Publishing: Treasure Island, FL, USA; University of North Carolina: Chapel Hill, CA, USA, 2022. [Google Scholar]
- Pašta, J. Laser therapy in ophthalmology. In Lasers for Medical Applications; Woodhead Publishing Series in Electronic and Optical, Materials; Jelínková, H.B.T.-L., Ed.; Woodhead Publishing: Cambridge, UK, 2013; pp. 395–458. ISBN 978-0-85709-237-3. [Google Scholar]
- Gawęcki, M. Micropulse Laser Treatment of Retinal Diseases. J. Clin. Med. 2019, 8, 242. [Google Scholar] [CrossRef]
- Ulbig, M.R.; Arden, G.B.; Hamilton, A.M. Color contrast sensitivity and pattern electroretinographic findings after diode and argon laser photocoagulation in diabetic retinopathy. Am. J. Ophthalmol. 1994, 117, 583–588. [Google Scholar] [CrossRef]
- Early Photocoagulation for Diabetic Retinopathy. ETDRS report number 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1991, 98, 766–785. [Google Scholar]
- Friberg, T.R.; Karatza, E.C. The treatment of macular disease using a micropulsed and continuous wave 810-nm diode laser. Ophthalmology 1997, 104, 2030–2038. [Google Scholar] [CrossRef]
- Scholz, P.; Altay, L.; Fauser, S. A Review of Subthreshold Micropulse Laser for Treatment of Macular Disorders. Adv. Ther. 2017, 34, 1528–1555. [Google Scholar] [CrossRef]
- Wells-Gray, E.M.; Doble, N.; Ohr, M.P.; Choi, S.S. Structural Integrity of Individual Cone Photoreceptors after Short-Wavelength Subthreshold Micropulse Laser Therapy for Diabetic Macular Edema. Ophthalmic. Surg. Lasers Imaging Retin. 2018, 49, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Al Habash, A.; AlAhmadi, A.S. Outcome of MicroPulse® Transscleral Photocoagulation in Different Types of Glaucoma. Clin. Ophthalmol. 2019, 13, 2353–2360. [Google Scholar] [CrossRef]
- National Library of Medicine. National Center for Biotechnology Information. PubMed Database. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 15 May 2023).
- Nature Database. Available online: https://www.nature.com/ (accessed on 16 May 2023).
- SpringerLink Database. Available online: https://link.springer.com/search?query=textile+waste+from+cutting+processes+AND+sustainable+management+of+textile+reuse (accessed on 15 June 2023).
- ScienceDirect Database. Available online: https://www.sciencedirect.com/ (accessed on 18 May 2023).
- Mansouri, A.; Sampat, K.M.; Malik, K.J.; Steiner, J.N.; Glaser, B.M. Efficacy of subthreshold micropulse laser in the treatment of diabetic macular edema is influenced by pre-treatment central foveal thickness. Eye 2014, 28, 1418–1424. [Google Scholar] [CrossRef]
- Kozak, I.; Luttrull, J.K. Modern retinal laser therapy. Saudi J. Ophthalmol. Off. J. Saudi Ophthalmol. Soc. 2015, 29, 137–146. [Google Scholar] [CrossRef]
- Pankratov, M.M. Pulsed delivery of laser energy in experimental thermal retinal photocoagulation. In Laser-Tissue Interaction; SPIE: Philadelphia, PA, USA, 1990; Volume 1202, pp. 205–213. [Google Scholar]
- Moorman, C.M.; Hamilton, A.M. Clinical applications of the MicroPulse diode laser. Eye 1999, 13 Pt 2, 145–150. [Google Scholar] [CrossRef]
- Sivaprasad, S.; Elagouz, M.; McHugh, D.; Shona, O.; Dorin, G. Micropulsed diode laser therapy: Evolution and clinical applications. Surv. Ophthalmol. 2010, 55, 516–530. [Google Scholar] [CrossRef]
- Aquino, M.C.D.; Barton, K.; Tan, A.M.W.T.; Sng, C.; Li, X.; Loon, S.C.; Chew, P.T.K. Micropulse versus continuous wave transscleral diode cyclophotocoagulation in refractory glaucoma: A randomized exploratory study. Clin. Experiment. Ophthalmol. 2015, 43, 40–46. [Google Scholar] [CrossRef]
- Brinkmann, R.; Roider, J.; Birngruber, R. Selective retina therapy (SRT): A review on methods, techniques, preclinical and first clinical results. Bull. Soc. Belge Ophtalmol. 2006, 302, 51–69. [Google Scholar]
- Wood, J.P.M.; Plunkett, M.; Previn, V.; Chidlow, G.; Casson, R.J. Nanosecond pulse lasers for retinal applications. Lasers Surg. Med. 2011, 43, 499–510. [Google Scholar] [CrossRef]
- Brinkmann, R.; Hüttmann, G.; Rögener, J.; Roider, J.; Birngruber, R.; Lin, C.P. Origin of retinal pigment epithelium cell damage by pulsed laser irradiance in the nanosecond to microsecond time regimen. Lasers Surg. Med. 2000, 27, 451–464. [Google Scholar] [CrossRef]
- Ong, J.; Selvam, A.; Maltsev, D.S.; Zhang, X.; Wu, L.; Chhablani, J. Subthreshold laser systems: A narrative review of the current status and advancements for retinal diseases. Ann. Eye Sci. 2022, 7, 15. [Google Scholar] [CrossRef]
- Korobelnik, J.-F.; Do, D.V.; Schmidt-Erfurth, U.; Boyer, D.S.; Holz, F.G.; Heier, J.S.; Midena, E.; Kaiser, P.K.; Terasaki, H.; Marcus, D.M.; et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology 2014, 121, 2247–2254. [Google Scholar] [CrossRef] [PubMed]
- Everett, L.A.; Paulus, Y.M. Laser Therapy in the Treatment of Diabetic Retinopathy and Diabetic Macular Edema. Curr. Diab. Rep. 2021, 21, 35. [Google Scholar] [CrossRef] [PubMed]
- Photocoagulation for Diabetic Macular Edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch. Ophthalmol. 1985, 103, 1796–1806. [Google Scholar] [CrossRef]
- Koushan, K.; Eshtiaghi, A.; Fung, P.; Berger, A.R.; Chow, D.R. Treatment of Diabetic Macular Edema with Aflibercept and Micropulse Laser (DAM Study). Clin. Ophthalmol. 2022, 16, 1109–1115. [Google Scholar] [CrossRef]
- Lavinsky, D.; Cardillo, J.A.; Melo, L.A.S.J.; Dare, A.; Farah, M.E.; Belfort, R.J. Randomized clinical trial evaluating mETDRS versus normal or high-density micropulse photocoagulation for diabetic macular edema. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4314–4323. [Google Scholar] [CrossRef]
- Bougatsou, P.; Panagiotopoulou, E.-K.; Gkika, M.; Dardabounis, D.; Konstantinidis, A.; Sideroudi, H.; Perente, I.; Labiris, G. Comparison of Subthreshold 532 nm Diode Micropulse Laser with Conventional Laser Photocoagulation in the Treatment of Non-Centre Involved Clinically Significant Diabetic Macular Edema. Acta Medica 2020, 63, 25–30. [Google Scholar] [CrossRef]
- Al-Barki, A.; Al-Hijji, L.; High, R.; Schatz, P.; Do, D.; Nguyen, Q.D.; Luttrull, J.K.; Kozak, I. Comparison of short-pulse subthreshold (532 nm) and infrared micropulse (810 nm) macular laser for diabetic macular edema. Sci. Rep. 2021, 11, 14. [Google Scholar] [CrossRef]
- Lois, N.; Gardner, E.; Waugh, N.; Azuara-Blanco, A.; Mistry, H.; McAuley, D.; Acharya, N.; Aslam, T.M.; Bailey, C.; Chong, V.; et al. Diabetic macular oedema and diode subthreshold micropulse laser (DIAMONDS): Study protocol for a randomised controlled trial. Trials 2019, 20, 122. [Google Scholar] [CrossRef]
- Lois, N.; Campbell, C.; Waugh, N.; Azuara-Blanco, A.; Maredza, M.; Mistry, H.; McAuley, D.; Acharya, N.; Aslam, T.M.; Bailey, C.; et al. Diabetic Macular Edema and Diode Subthreshold Micropulse Laser: A Randomized Double-Masked Noninferiority Clinical Trial. Ophthalmology 2023, 130, 14–27. [Google Scholar] [CrossRef]
- Moisseiev, E.; Abbassi, S.; Thinda, S.; Yoon, J.; Yiu, G.; Morse, L.S. Subthreshold micropulse laser reduces anti-VEGF injection burden in patients with diabetic macular edema. Eur. J. Ophthalmol. 2018, 28, 68–73. [Google Scholar] [CrossRef]
- Khattab, A.M.; Hagras, S.M.; AbdElhamid, A.; Torky, M.A.; Awad, E.A.; Abdelhameed, A.G. Aflibercept with adjuvant micropulsed yellow laser versus aflibercept monotherapy in diabetic macular edema. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 257, 1373–1380. [Google Scholar] [CrossRef] [PubMed]
- Abouhussein, M.A.; Gomaa, A.R. Aflibercept plus micropulse laser versus aflibercept monotherapy for diabetic macular edema: 1-year results of a randomized clinical trial. Int. Ophthalmol. 2020, 40, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Gawęcki, M. Subthreshold Diode Micropulse Laser Combined with Intravitreal Therapy for Macular Edema-A Systematized Review and Critical Approach. J. Clin. Med. 2021, 10, 1394. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, K.; Hamada, M.; Ohkoshi, K. Minimally invasive laser treatment combined with intravitreal injection of anti-vascular endothelial growth factor for diabetic macular oedema. Sci. Rep. 2019, 9, 7585. [Google Scholar] [CrossRef]
- Frizziero, L.; Calciati, A.; Torresin, T.; Midena, G.; Parrozzani, R.; Pilotto, E.; Midena, E. Diabetic Macular Edema Treated with 577-nm Subthreshold Micropulse Laser: A Real-Life, Long-Term Study. J. Pers. Med. 2021, 11, 405. [Google Scholar] [CrossRef]
- Çıtırık, M. Nondamaging retinal laser therapy in recurrent diabetic macular edema after antiVEGF injections. Turk. J. Med. Sci. 2021, 51, 2616–2620. [Google Scholar] [CrossRef]
- Passos, R.M.; Malerbi, F.K.; Rocha, M.; Maia, M.; Farah, M.E. Real-life outcomes of subthreshold laser therapy for diabetic macular edema. Int. J. Retin. Vitr. 2021, 7, 4. [Google Scholar] [CrossRef]
- Citirik, M. The impact of central foveal thickness on the efficacy of subthreshold micropulse yellow laser photocoagulation in diabetic macular edema. Lasers Med. Sci. 2019, 34, 907–912. [Google Scholar] [CrossRef]
- Değirmenci, M.F.K.; Demirel, S.; Batıoğlu, F.; Özmert, E. Short-term Efficacy of Micropulse Yellow Laser in Non-center-involving Diabetic Macular Edema: Preliminary Results. Turk. J. Ophthalmol. 2018, 48, 245–249. [Google Scholar] [CrossRef]
- Vujosevic, S.; Gatti, V.; Muraca, A.; Brambilla, M.; Villani, E.; Nucci, P.; Rossetti, L.; De Cilla’, S. Optical coherence tomography angiography changes after subthreshold micropulse yellow laser in diabetic macular edema. Retina 2020, 40, 312–321. [Google Scholar] [CrossRef]
- Vujosevic, S.; Toma, C.; Villani, E.; Brambilla, M.; Torti, E.; Leporati, F.; Muraca, A.; Nucci, P.; De Cilla, S. Subthreshold Micropulse Laser in Diabetic Macular Edema: 1-Year Improvement in OCT/OCT-Angiography Biomarkers. Transl. Vis. Sci. Technol. 2020, 9, 31. [Google Scholar] [CrossRef]
- Latalska, M.; Prokopiuk, A.; Wróbel-Dudzińska, D.; Mackiewicz, J. Subthreshold micropulse yellow 577 nm laser therapy of diabetic macular oedema in rural and urban patients of south-eastern Poland. Ann. Agric. Environ. Med. 2017, 24, 96–99. [Google Scholar] [CrossRef]
- Abouhussein, M.A. Micropulse laser for diabetic macular edema. Delta J. Ophthalmol. 2016, 17, 167–171. [Google Scholar] [CrossRef]
- Inagaki, K.; Ohkoshi, K.; Ohde, S.; Deshpande, G.A.; Ebihara, N.; Murakami, A. Subthreshold Micropulse Photocoagulation for Persistent Macular Edema Secondary to Branch Retinal Vein Occlusion including Best-Corrected Visual Acuity Greater than 20/40. J. Ophthalmol. 2014, 2014, 251257. [Google Scholar] [CrossRef]
- Li, L.; Ren, Q.; Sun, Z.; Yu, H. Clinical efficacy of conbercept plus micropulse laser (577 nm) treatment in macular edema secondary to non-ischemic central retinal vein occlusion. Pak. J. Med. Sci. 2022, 38, 1366–1370. [Google Scholar] [CrossRef]
- Buyru Özkurt, Y.; Akkaya, S.; Aksoy, S.; Şimşek, M.H. Comparison of ranibizumab and subthreshold micropulse laser in treatment of macular edema secondary to branch retinal vein occlusion. Eur. J. Ophthalmol. 2018, 28, 690–696. [Google Scholar] [CrossRef]
- Parodi, M.B.; Iacono, P.; Bandello, F. Subthreshold grid laser versus intravitreal bevacizumab as second-line therapy for macular edema in branch retinal vein occlusion recurring after conventional grid laser treatment. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 253, 1647–1651. [Google Scholar] [CrossRef]
- Parodi, M.B.; Spasse, S.; Iacono, P.; Di Stefano, G.; Canziani, T.; Ravalico, G. Subthreshold grid laser treatment of macular edema secondary to branch retinal vein occlusion with micropulse infrared (810 nanometer) diode laser. Ophthalmology 2006, 113, 2237–2242. [Google Scholar] [CrossRef]
- Parodi, M.B.; Iacono, P.; Ravalico, G. Intravitreal triamcinolone acetonide combined with subthreshold grid laser treatment for macular oedema in branch retinal vein occlusion: A pilot study. Br. J. Ophthalmol. 2008, 92, 1046–1050. [Google Scholar] [CrossRef]
- Terashima, H.; Hasebe, H.; Okamoto, F.; Matsuoka, N.; Sato, Y.; Fukuchi, T. Combination therapy of intravitreal ranibizumab and subthreshold micropulse photocoagulation for macular edema secondary to branch retinal vein occlusion: 6-month result. Retina 2019, 39, 1377–1384. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.W.; Kang, S.W.; Kim, Y.T.; Chung, S.E.; Lee, S.W. Comparative study of patients with central serous chorioretinopathy undergoing focal laser photocoagulation or photodynamic therapy. Br. J. Ophthalmol. 2011, 95, 514–517. [Google Scholar] [CrossRef]
- Lee, P.Y.; Kim, K.S.; Lee, W.K. Severe choroidal ischemia following photodynamic therapy for pigment epithelial detachment and chronic central serous chorioretinopathy. Jpn. J. Ophthalmol. 2009, 53, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.-M.; Lai, T.Y.Y.; Lai, R.Y.K.; Liu, D.T.L.; Lam, D.S.C. Half-dose verteporfin photodynamic therapy for acute central serous chorioretinopathy: One-year results of a randomized controlled trial. Ophthalmology 2008, 115, 1756–1765. [Google Scholar] [CrossRef]
- Chen, S.-N.; Hwang, J.-F.; Tseng, L.-F.; Lin, C.-J. Subthreshold diode micropulse photocoagulation for the treatment of chronic central serous chorioretinopathy with juxtafoveal leakage. Ophthalmology 2008, 115, 2229–2234. [Google Scholar] [CrossRef]
- Zeng, M.; Chen, X.; Song, Y.; Cai, C. Subthreshold micropulse laser photocoagulation versus half-dose photodynamic therapy for acute central serous chorioretinopathy. BMC Ophthalmol. 2022, 22, 110. [Google Scholar] [CrossRef] [PubMed]
- Daruich, A.; Matet, A.; Dirani, A.; Bousquet, E.; Zhao, M.; Farman, N.; Jaisser, F.; Behar-Cohen, F. Central serous chorioretinopathy: Recent findings and new physiopathology hypothesis. Prog. Retin. Eye Res. 2015, 48, 82–118. [Google Scholar] [CrossRef]
- Brancato, R.; Bandello, F. Treatment of central serous chorioretinopathy with beta-blockers and calcium antagonists. Macula Soc. 1994, 3, 114. [Google Scholar]
- Ficker, L.; Vafidis, G.; While, A.; Leaver, P. Long-term follow-up of a prospective trial of argon laser photocoagulation in the treatment of central serous retinopathy. Br. J. Ophthalmol. 1988, 72, 829–834. [Google Scholar] [CrossRef]
- Verma, L.; Sinha, R.; Venkatesh, P.; Tewari, H.K. Comparative evaluation of diode laser versus argon laser photocoagulation in patients with central serous retinopathy: A pilot, randomized controlled trial [ISRCTN84128484]. BMC Ophthalmol. 2004, 4, 15. [Google Scholar] [CrossRef]
- Kiraly, P.; Smrekar, J.; Jaki Mekjavić, P. Morphological parameters predicting subthreshold micropulse laser effectiveness in central serous chorioretinopathy. Lasers Med. Sci. 2022, 37, 3129–3136. [Google Scholar] [CrossRef]
- van Dijk, E.H.C.; Fauser, S.; Breukink, M.B.; Blanco-Garavito, R.; Groenewoud, J.M.M.; Keunen, J.E.E.; Peters, P.J.H.; Dijkman, G.; Souied, E.H.; MacLaren, R.E.; et al. Half-Dose Photodynamic Therapy versus High-Density Subthreshold Micropulse Laser Treatment in Patients with Chronic Central Serous Chorioretinopathy: The PLACE Trial. Ophthalmology 2018, 125, 1547–1555. [Google Scholar] [CrossRef]
- Gawęcki, M.; Jaszczuk-Maciejewska, A.; Jurska-Jaśko, A.; Kneba, M.; Grzybowski, A. Transfoveal Micropulse Laser Treatment of Central Serous Chorioretinopathy within Six Months of Disease Onset. J. Clin. Med. 2019, 8, 1398. [Google Scholar] [CrossRef]
- Sousa, K.; Calvão-Santos, G.; João, M.; Gomes, N.; Falcão, M. 532-nm Subthreshold Micropulse Laser for the Treatment of Chronic Central Serous Retinopathy. Clin. Ophthalmol. 2020, 14, 525–531. [Google Scholar] [CrossRef]
- Long, H.; Liu, M.; Hu, Q.; Li, X. 577 nm subthreshold micropulse laser treatment for acute central serous chorioretinopathy: A comparative study. BMC Ophthalmol. 2022, 22, 105. [Google Scholar] [CrossRef]
- Lanzetta, P.; Furlan, F.; Morgante, L.; Veritti, D.; Bandello, F. Nonvisible subthreshold micropulse diode laser (810 nm) treatment of central serous chorioretinopathy. A pilot study. Eur. J. Ophthalmol. 2008, 18, 934–940. [Google Scholar] [CrossRef]
- Abd Elhamid, A.H. Subthreshold micropulse yellow laser treatment for nonresolving central serous chorioretinopathy. Clin. Ophthalmol. 2015, 9, 2277–2283. [Google Scholar] [CrossRef]
- Ambiya, V.; Goud, A.; Mathai, A.; Rani, P.K.; Chhablani, J. Microsecond yellow laser for subfoveal leaks in central serous chorioretinopathy. Clin. Ophthalmol. 2016, 10, 1513–1519. [Google Scholar]
- Arsan, A.; Kanar, H.S.; Sonmez, A. Visual outcomes and anatomic changes after sub-threshold micropulse yellow laser (577-nm) treatment for chronic central serous chorioretinopathy: Long-term follow-up. Eye 2018, 32, 726–733. [Google Scholar] [CrossRef]
- Koss, M.J.; Beger, I.; Koch, F.H. Subthreshold diode laser micropulse photocoagulation versus intravitreal injections of bevacizumab in the treatment of central serous chorioretinopathy. Eye 2012, 26, 307–314. [Google Scholar] [CrossRef]
- Roisman, L.; Magalhães, F.P.; Lavinsky, D.; Moraes, N.; Hirai, F.E.; Cardillo, J.A.; Farah, M.E. Micropulse diode laser treatment for chronic central serous chorioretinopathy: A randomized pilot trial. Ophthalmic Surg. Lasers Imaging Retin. 2013, 44, 465–470. [Google Scholar] [CrossRef]
- Malik, K.J.; Sampat, K.M.; Mansouri, A.; Steiner, J.N.; Glaser, B.M. Low-intensity/high-density subthreshold microPulse diode laser for chronic central serous chorioretinopathy. Retina 2015, 35, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.K.; Jayadev, C.; Mohan, A.; Vijayan, P.; Battu, R.; Dabir, S.; Shetty, B.; Shetty, R. Subthreshold micropulse yellow laser (577 nm) in chronic central serous chorioretinopathy: Safety profile and treatment outcome. Eye 2015, 29, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Park, H.S.; Kim, S.Y. Short-term efficacy of subthreshold micropulse yellow laser (577-nm) photocoagulation for chronic central serous chorioretinopathy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 253, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Scholz, P.; Ersoy, L.; Boon, C.J.F.; Fauser, S. Subthreshold Micropulse Laser (577 nm) Treatment in Chronic Central Serous Chorioretinopathy. Ophthalmologica 2015, 234, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Luttrull, J.K. Low-intensity/high-density subthreshold diode micropulse laser for central serous chorioretinopathy. Retina 2016, 36, 1658–1663. [Google Scholar] [CrossRef] [PubMed]
- Gawęcki, M.; Jaszczuk-Maciejewska, A.; Jurska-Jaśko, A.; Grzybowski, A. Functional and morphological outcome in patients with chronic central serous chorioretinopathy treated by subthreshold micropulse laser. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 2299–2306. [Google Scholar] [CrossRef]
- Maruko, I.; Koizumi, H.; Hasegawa, T.; Arakawa, H.; Iida, T. Subthreshold 577 nm micropulse laser treatment for central serous chorioretinopathy. PLoS ONE 2017, 12, e0184112. [Google Scholar] [CrossRef]
- Luttrull, J.K.; Sinclair, S.H.; Elmann, S.; Chang, D.B.; Kent, D. Slowed Progression of Age-Related Geographic Atrophy Following Subthreshold Laser. Clin. Ophthalmol. 2020, 14, 2983–2993. [Google Scholar] [CrossRef]
- Querques, G.; Sacconi, R.; Gelormini, F.; Borrelli, E.; Prascina, F.; Zucchiatti, I.; Querques, L.; Bandello, F. Subthreshold laser treatment for reticular pseudodrusen secondary to age-related macular degeneration. Sci. Rep. 2021, 11, 2193. [Google Scholar] [CrossRef]
- Huang, Z.; Deng, K.-Y.; Deng, Y.-M.; Hui, Y.-N.; Song, Y.-P. Long-term outcomes of drusenoid pigment epithelium detachment in intermediate AMD treated with 577 nm subthreshold micropulse laser: A preliminary clinical study. Int. J. Ophthalmol. 2022, 15, 474–482. [Google Scholar] [CrossRef]
- Johnson, T.M.; Glaser, B.M. Micropulse laser treatment of retinal-choroidal anastomoses in age-related macular degeneration. Graefe’s Arch. Clin. Exp. Ophthalmol. 2005, 243, 570–575. [Google Scholar] [CrossRef]
- Luttrull, J.K.; Chang, D.B.; Margolis, B.W.L.; Dorin, G.; Luttrull, D.K. Laser resensitization of medically unresponsive neovascular age-related macular degeneration: Efficacy and implications. Retina 2015, 35, 1184–1194. [Google Scholar] [CrossRef][Green Version]
- Grippo, T.M.; de Crom, R.M.P.C.; Giovingo, M.; Töteberg-Harms, M.; Francis, B.A.; Jerkins, B.; Brubaker, J.W.; Radcliffe, N.; An, J.; Noecker, R. Evidence-Based Consensus Guidelines Series for MicroPulse Transscleral Laser Therapy: Dosimetry and Patient Selection. Clin. Ophthalmol. 2022, 16, 1837–1846. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Song, S.-J.; Liu, B.; Hassanpour, K.; Zhang, C.; Loewen, N. Efficacy and safety of micropulse laser trabeculoplasty for primary open angle glaucoma. Int. J. Ophthalmol. 2019, 12, 784–788. [Google Scholar] [PubMed]
- Wise, J.B.; Witter, S.L. Argon laser therapy for open-angle glaucoma. A pilot study. Arch. Ophthalmol. 1979, 97, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Glaucoma Laser Trial Research Group. The Glaucoma Laser Trial (GLT) and glaucoma laser trial follow-up study: 7. Results. Am. J. Ophthalmol. 1995, 120, 718–731. [Google Scholar] [CrossRef]
- Latina, M.A.; Park, C. Selective targeting of trabecular meshwork cells: In vitro studies of pulsed and CW laser interactions. Exp. Eye Res. 1995, 60, 359–371. [Google Scholar] [CrossRef]
- Latina, M.A.; Sibayan, S.A.; Shin, D.H.; Noecker, R.J.; Marcellino, G. Q-switched 532-nm Nd:YAG laser trabeculoplasty (selective laser trabeculoplasty): A multicenter, pilot, clinical study. Ophthalmology 1998, 105, 2082–2090. [Google Scholar] [CrossRef]
- Abramowitz, B.; Chadha, N.; Kouchouk, A.; Alhabshan, R.; Belyea, D.A.; Lamba, T. Selective laser trabeculoplasty vs micropulse laser trabeculoplasty in open-angle glaucoma. Clin. Ophthalmol. 2018, 12, 1599–1604. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Q.; Chen, T.A.; Deiner, M.S.; Ou, Y. Clinical Outcomes of Micropulse Laser Trabeculoplasty Compared to Selective Laser Trabeculoplasty at One Year in Open-Angle Glaucoma. Clin. Ophthalmol. 2021, 15, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Emanuel, M.E.; Grover, D.S.; Fellman, R.L.; Godfrey, D.G.; Smith, O.; Butler, M.R.; Kornmann, H.L.; Feuer, W.J.; Goyal, S. Micropulse Cyclophotocoagulation: Initial Results in Refractory Glaucoma. J. Glaucoma 2017, 26, 726–729. [Google Scholar] [CrossRef]
- Varikuti, V.N.V.; Shah, P.; Rai, O.; Chaves, A.C.; Miranda, A.; Lim, B.-A.; Dorairaj, S.K.; Sieminski, S.F. Outcomes of Micropulse Transscleral Cyclophotocoagulation in Eyes with Good Central Vision. J. Glaucoma 2019, 28, 901–905. [Google Scholar] [CrossRef]
- Konstas, A.G.; Mantziris, D.A.; Stewart, W.C. Diurnal intraocular pressure in untreated exfoliation and primary open-angle glaucoma. Arch. Ophthalmol. 1997, 115, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Makri, O.E.; Pagoulatos, D.; Kagkelaris, K.; Plotas, P.; Georgakopoulos, C.D. Evaluation of intraocular pressure in the first 24hours after micropulse laser trabeculoplasty in eyes with pseudoexfoliation glaucoma. J. Fr. Ophtalmol. 2019, 42, 983–986. [Google Scholar] [CrossRef] [PubMed]
- Makri, O.E.; Plotas, P.; Christopoulou, E.; Georgakopoulos, C.D. Effect of a single session of micropulse laser trabeculoplasty on corneal endothelial parameters. Clin. Exp. Optom. 2020, 103, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Tekeli, O.; Köse, H.C. Outcomes of micropulse transscleral cyclophotocoagulation in primary open-angle glaucoma, pseudoexfoliation glaucoma, and secondary glaucoma. Eur. J. Ophthalmol. 2021, 31, 1113–1121. [Google Scholar] [CrossRef]
- Aydin Kurna, S.; Sonmez, A.D.; Yamic, M.; Altun, A. Long-term results of micropulse laser trabeculoplasty with 577-nm yellow wavelength in patients with uncontrolled primary open-angle glaucoma and pseudoexfoliation glaucoma. Lasers Med. Sci. 2022, 37, 2745–2752. [Google Scholar] [CrossRef]
- Abouhussein, M.A. Micropulse laser trabeculoplasty for open-angle glaucoma. Delta J. Ophthalmol. 2016, 17, 80–84. [Google Scholar] [CrossRef]
- Detry-Morel, M.; Muschart, F.; Pourjavan, S. Micropulse diode laser (810 nm) versus argon laser trabeculoplasty in the treatment of open-angle glaucoma: Comparative short-term safety and efficacy profile. Bull. Soc. Belge Ophtalmol. 2008, 308, 21–28. [Google Scholar]
- Fea, A.M.; Bosone, A.; Rolle, T.; Brogliatti, B.; Grignolo, F.M. Micropulse diode laser trabeculoplasty (MDLT): A phase II clinical study with 12 months follow-up. Clin. Ophthalmol. 2008, 2, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.Y.; Yau, G.S.K.; Yick, D.W.F.; Yuen, C.Y.F. MicroPulse Laser Trabeculoplasty for the Treatment of Open-Angle Glaucoma. Medicine 2015, 94, e2075. [Google Scholar] [CrossRef] [PubMed]
- Marcela, D.R.; De León De León, I.; José, D.R.; Ortega Santana, F.; Alfonso, D.R.; López, G. Selective Laser Trabeculoplasty vs. Micropulse Laser Trabeculoplasty for the Treatment of Open Angle Glaucoma and Ocular Hypertension. Int. J. Res. Med. Heal. Sci. 2017, 14, 30–34. [Google Scholar]
- Rantala, E.; Välimäki, J. Micropulse diode laser trabeculoplasty—180-degree treatment. Acta Ophthalmol. 2012, 90, 441–444. [Google Scholar] [CrossRef]
- Babalola, O.E. Micropulse diode laser trabeculoplasty in Nigerian patients. Clin. Ophthalmol. 2015, 9, 1347–1351. [Google Scholar] [CrossRef]
| Type of Study | Number of Treated Eyes/ Follow-up Period (Months) | Evaluation | Results | Refs. |
|---|---|---|---|---|
| Prospective, randomized, double-masked | 123/12 | Comparison between the effectiveness of different laser treatments for DME, specifically comparing two types of laser treatments (810 nm ND-MPLT and 810 nm HD-MPLT) with a traditional photocoagulation method (mETDRS) | HD-MPLT and mETDRS photocoagulation are similarly effective in terms of BCVA and CRT outcomes for patients with CS-DME, while ND-MPLT demonstrated stable BCVA without significant retreatment needs | [33] |
| Prospective and randomized | 60/6 | Comparison of the effectiveness of two different laser treatments for DME, specifically comparing 532 nm MPLT with 532 nm focal photocoagulation | The reduction in CRT was significantly better in the MPLT group compared to the focal photocoagulation group | [34] |
| Prospective and randomized | 116/6 | Comparison of the effectiveness of two different laser treatments for DME, specifically comparing short-pulse subthreshold 532 nm laser treatment with micropulse 810 nm laser treatment | Visual acuity significantly improved in the micropulse 810 nm laser treatment group | [35] |
| Prospective, randomized, double-masked | 266/24 | Comparison of the effectiveness of two different laser treatments for CI-DME. The study compared 577 nm MPLT with the traditional modified mETDRS photocoagulation | No significant differences between the MPLT and mETDRS groups in terms of BCVA and CRT | [36,37] |
| Type of Study | Number of Treated Eyes/ Follow-up Period (Months) | Evaluation | Results | Refs. |
|---|---|---|---|---|
| Retrospective | 134/16 | Retrospective assessment of 134 eyes with previously untreated center-involving mild DME treated with 577 nm SMPL | A decrease in the CRT and average thickness of the nine ETDRS sectors was found in comparison to baseline; no side effects were observed | [43] |
| 70/2 | Retrospective assessment of the correlation between the severity of DME and the efficacy of subthreshold yellow NRT | NRT provided an improvement in BCVA and CRT | [44] | |
| 56/3 | Treatment with 577 nm MPLT | Significant improvement in BCVA | [45] | |
| 80/6 | Significant decrease in CRT and stabilization of BCVA | [46] | ||
| 9/3 | Significant decrease in CRT and stabilization of BCVA | [47] | ||
| Prospective | 52/12 | Significant improvement in BCVA and ETDRS and a significant decrease in hyper-reflective retinal spots and microaneurysms | [48,49] | |
| 75/6 | Significant reduction in CRT and a stabilization of BCVA | [50] | ||
| 20/6 | Significant improvement in BCVA and an important reduction in CRT | [51] |
| Type of Study | Number of Treated Eyes/ Follow-up Period (Months) | Evaluation | Results | Ref. |
|---|---|---|---|---|
| Prospective randomized | 35: 18- SGLT group; 17-IVB group/12 | Comparison of the effectiveness of SMPLT vs. IVB in recurrent ME secondary to BRVO | The IVB group showed a significant reduction in CRT, from 484 µm to 271 µm, as well as an improvement in BCVA; no significant effects were observed in the SMPLT group | [55] |
| 36: 17 SMPLT group; 19 LPC group/24 | Comparison of the effectiveness of different laser treatments for macular edema secondary to BRVO occurring three to eighteen months prior | At the 12-month follow-up, there were no significant differences between the groups in terms of BCVA, CRT, or MV; at the 24-month follow-up, 65% of patients who underwent SMPLT treatment and 58% of patients who received LPC treatment showed an improvement of at least 10 letters in visual acuity | [56] | |
| 24: 13 SMPLT group; 1 SMPLT + IVT group/12 | Comparison of the effectiveness of SMPLT and SMPLT + IVT | After 12 months, the combined treatment of SMPLT and IVT resulted in a significant improvement of at least 10 letters in visual acuity for 91% of patients, whereas the SMPLT-alone group showed a 62% improvement with a statistically significant difference between the two groups | [57] | |
| Retrospective, consecutive, case–control study | 46: 22 IVR + SMPLT group 24—IVR group/6 | Comparison of the effectiveness of IVR + SMPLT vs. IVR in eyes with ME secondary to BRVO, treatment-naïve; assessment of CRT, BCVA | Both groups showed a significant improvement in BCVA and CME, with no significant difference between the two groups; the number of injections in the group receiving IVR was statistically higher (2.3) compared to the group receiving IVR plus SMPLT | [58] |
| Retrospective | 51: 27 IVR group 24 SMPLT group/12 | Comparison of the effectiveness of SMPLT vs. IVR in patients with ME secondary to BRVO, BCVA, CRT; the frequency of treatments was assessed after a minimum of 3 months from the occlusive event | There were no significant differences in the final BCVA or CRT between the two groups; the IVR group received an average of 3.81 treatments, while the SMPLT group received 1.5 treatments | [54] |
| Retrospective, interventional case series | 32: BCVA ≤ 20/40 group BCVA > 20/40 group/12 | Comparison of the effectiveness of SMPLT in eyes with longstanding ME after BRVO (at least 6 months) | Both groups showed a significant decrease in CRT, with no notable difference between them; there was no significant improvement in BCVA in either group | [52] |
| Study | No of Eyes/Duration of CSCR (Months) | Evaluation | Outcomes | Ref. |
|---|---|---|---|---|
| Prospective, Interventional, noncomparative | 26/>4 | Provide information on the visual and clinical effects in chronic idiopathic CSCR with juxta foveal leaking of subthreshold diode micropulse laser photocoagulation | BCVA improved, in a statistically relevant manner, in 100% of patients, increasing in 57.7% of cases by at least three lines and in 23.1% of cases by one to three lines | [62] |
| 24/>3 | For treating CSCR, it is important to confirm the effectiveness of nonvisible micropulse diode laser irradiation | BCVA enhancement | [73] | |
| 15/≥3 | Provide information on the safety and effectiveness of micropulse 577 nm yellow laser treatment in the therapeutic management of persistent conditions of CSCR | A 100% decrease in SRF; after three months and six months, respectively, total SRF absorption was 73% and 86%; BCVA increased from 0.67 Snellen to 0.85 Snellen | [74] | |
| 10/≥3 | Analyze the effectiveness of guided yellow microsecond laser in the treatment of persistent CSCR sub-foveal leaks | SRF decrease was 100%; total SRF resorption at six months was 60%, CRT decreased from a mean of 298 to 215 µm, and the variation in BCVA from 73.3 to 76.9 letters ETDRS at six months was not statistically significant | [75] | |
| 39/>3 | The anatomic result, visual outcomes, and the safety profile of the treatment for chronic CSCR and the evaluation of the long-term effectiveness of 577 nm sub-threshold micropulse yellow laser treatment; 17.82 months average follow-up (13–24 months) | A decrease in the mean CRT from 369 μm to 250 μm; an enhancement in BCVA in 89.7% of cases | [76] | |
| Prospective, comparative, controlled | 52: 16 SDM group; 10 BCZ group; 26 observation group/>3 | Comparison of the effectiveness in CSCR of SDM vs. BCZ | When treating CSCR, SDM photocoagulation was more effective than intravitreal injections with 1.25 mg BCZ, which determined an improvement in macular perimetry and visual acuity | [77] |
| Prospective, randomized, double-blind, sham-controlled pilot trial | 15: 5 sham group; 10 SDM group/≥6 | In individuals with chronic CSCR, evaluation of 810 nm SDM laser | When compared to the sham group at 3 months, BCVA was considerably improved in the intervention group | [78] |
| Retrospective | 11/≥113 | The macular thickness and visual results in individuals with symptomatic chronic CSCR modification after therapy with an 810 nm subthreshold micropulse diode laser | Following SMLT, the maximum macular thickness reduced by values between 20 μm and 338 μm; the median BCVA boost was six letters ETDRS | [79] |
| 15/>3 | Analyze the safety and effectiveness of one session of chronic CSCR therapy using a subthreshold micropulse yellow laser (577 nm) | A 100% decrease in SRF, 40 percent in SRF overall resorption, anda statistically significant enhancement in BCVA from 20/40 Snellen to 20/30 Snellen | [80] | |
| 10/≥6 | Evaluate the effectiveness of subthreshold micropulse yellow laser photocoagulation on the short term for treating chronic CSCR | BCVA enhanced from 0.21 logMar to 0.035 logMar, while CRT decreased from 349.2 m to 261.2 m | [81] | |
| 38/>1.5 | For evaluating the effects of treatment for individuals with chronic CSCR, a 577 nm SMLT was used | A 74% decrease in SRF; CRT was typically reduced by 115 μm, while BCVA was improved by 0.06 logMAR | [82] | |
| 11/1–7 (3.6 on average) | Evaluate the outcomes of the CSCR therapy with low-intensity/high-density SDM | Improvement in BCVA from an average 20/37 to an average of 20/24, with a CRT decrease from mean 508 µm to average 250 µm | [83] | |
| 51/>4 | Analyze the functional and morphological consequences, as well as the variables affecting the visual outcome, in individuals with chronic CSCR who have had SMLT | SRF was completely absorbed in 70.6%; CRT was reduced on average from 337.6 to 260 µm; a statistically relevant mean BCVA +0.08 logMAR improvement | [84] | |
| 29: 15 conventional laser group; 14 SML group />3 | Comparatively assess the effectiveness of conventional lasers vs. SML in treating focal retinal pigment epithelium leakages in individuals suffering from CSCR | SML had therapeutic results comparable to conventional lasers, although retinal pigment epithelium damage was avoided in eyes treated with CSCR. Total SRF resorption was reported in 64.3% of the SML group; CRT decreased from 328 μm to 192 μm, on average. Statistically negligible BCVA fluctuation from 0.96 to 0.94 Snellen | [85] |
| Type of Study | Number of Treated Eyes/ Follow-up Period (Months) | Evaluation | Results | Ref. |
|---|---|---|---|---|
| Retrospective | 13/3–7 (average 5) | Evaluating the efficacy of subthreshold diode micropulse laser therapy in eyes with a lack of response to all anti-vascular endothelial growth factor drugs, encompassing a history of at least three consecutive ineffective aflibercept injections | Out of the total eyes studied, 92% demonstrated improvement, with 69% (9 out of 13) achieving complete resolution of macular exudation; visual acuity remained unaffected, while notable improvements were observed in both central and maximum macular thicknesses | [90] |
| Prospective non-randomized | 20/3 | Assessment of the safety and short-term effectiveness of subthreshold laser treatment in patients with reticular pseudo drusen secondary to dry age-related macular degeneration | During the follow-up, there was a noteworthy rise in the number of Stage 1 reticular pseudo drusen, accompanied by a substantial decrease in Stage 3 reticular pseudo drusen; analysis of the outer nuclear layer thickness revealed a significant increase after the treatment, which was linked to reticular pseudo drusen regression; the findings suggest that subthreshold laser treatment as an end-point management approach appears to be safe for reticular pseudo drusen secondary to dry age-related macular degeneration, with positive short-term safety outcomes | [87] |
| Type of Study | Number of treated Eyes/ Follow-up period (Months) | Evaluation | Results | Ref. |
|---|---|---|---|---|
| Prospective interventional case series | 30/6 | Assessing the effectiveness of laser treatments for primary open-angle glaucoma, with 77 nm laser equipment with defined therapy parameters | Baseline IOP was 18.07 ± 1.91 mmHg, on average; the IOP dropped rapidly and significantly to 14.17 ± 1.56 mmHg at six months after baseline | [106] |
| Prospective, comparative, randomized | 31: 16 MDLT group; 15 ALT group/3 | Comparison of the effectiveness of two different laser treatments for evaluating the IOP decreasing benefits and safety in open-angle glaucoma subjects treated with ALT and MDLT (810 nm) | At three months, ALT dramatically reduced IOP, while MDLT did not; diode laser trabeculoplasty dramatically reduced the proportion of eyes having an IOP decrease of 20% or more compared to argon laser trabeculoplasty; the inflammation of the anterior segment caused by MDLT was not severe, and its safety profile appeared to be favorable | [107] |
| Prospective | 20/12 | Assessment of individuals with OAG that was medically unsupervised, the pressure-lowering advantages of subthreshold MDLT | In 75% of OAG eyes with inadequate medical management, MDLT successfully decreased IOP without any major side effects | [108] |
| 48/6 | Assessing the effectiveness of laser treatments with one session with unilateral MLT therapy that used a 577 nm diode laser to treat the trabecular meshwork across 360 degrees for lowering medication burden or IOP | At six months after the laser treatment, MLT had a low failure rate, negligible post-laser inflammation, and was successful in lowering IOP and prescription drugs in OAG | [109] | |
| 69: 38 MLT group; 31 SLT group/3 | Examining the effectiveness, acceptability, and safety of SLT against MLT in lowering IOP in unmanaged open-angle glaucoma | With less pain experienced both before and after the surgery, micropulse trabeculoplasty showed similar effectiveness to SLT in a 52-week monitoring interval | [110] | |
| Retrospective | 40/12 | Analyzing the results of 180-degree MDLT in glaucoma patients who have secondary open-angle glaucoma | In individuals with open-angle glaucoma, 180° MDLT is a safe but unsuccessful procedure | [111] |
| 30/5.33 | Assessing the effectiveness of the 810 diode Optos FastPulse laser (34 cycles of treatment) for primary open-angle glaucoma in patients with medical greatest intervention failing; it was evaluated how the pressure changed one hour after the therapy | IOP decreased by 17.2% from the starting point pre-laser at a mean post-laser reduction of 3.2 mmHg; over varied time intervals, ranging from several weeks to a few months, the IOP dropped steadily | [112] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bodea, F.; Bungau, S.G.; Bogdan, M.A.; Vesa, C.M.; Radu, A.; Tarce, A.G.; Purza, A.L.; Tit, D.M.; Bustea, C.; Radu, A.-F. Micropulse Laser Therapy as an Integral Part of Eye Disease Management. Medicina 2023, 59, 1388. https://doi.org/10.3390/medicina59081388
Bodea F, Bungau SG, Bogdan MA, Vesa CM, Radu A, Tarce AG, Purza AL, Tit DM, Bustea C, Radu A-F. Micropulse Laser Therapy as an Integral Part of Eye Disease Management. Medicina. 2023; 59(8):1388. https://doi.org/10.3390/medicina59081388
Chicago/Turabian StyleBodea, Flaviu, Simona Gabriela Bungau, Mihaela Alexandra Bogdan, Cosmin Mihai Vesa, Ada Radu, Alexandra Georgiana Tarce, Anamaria Lavinia Purza, Delia Mirela Tit, Cristian Bustea, and Andrei-Flavius Radu. 2023. "Micropulse Laser Therapy as an Integral Part of Eye Disease Management" Medicina 59, no. 8: 1388. https://doi.org/10.3390/medicina59081388
APA StyleBodea, F., Bungau, S. G., Bogdan, M. A., Vesa, C. M., Radu, A., Tarce, A. G., Purza, A. L., Tit, D. M., Bustea, C., & Radu, A.-F. (2023). Micropulse Laser Therapy as an Integral Part of Eye Disease Management. Medicina, 59(8), 1388. https://doi.org/10.3390/medicina59081388









