Does Sarcopenia Accompanying End-Stage Knee Osteoarthritis Affect the Outcomes following Total Knee Arthroplasty?
Abstract
1. Introduction
- What is the incidence of sarcopenia in patients undergoing TKA for advanced knee OA?
- Are the PROMs in patients with sarcopenia associated with knee OA inferior to those in patients without sarcopenia?
- If yes, what are the predisposing factors for sarcopenia in patients with advanced knee OA?
2. Materials and Methods
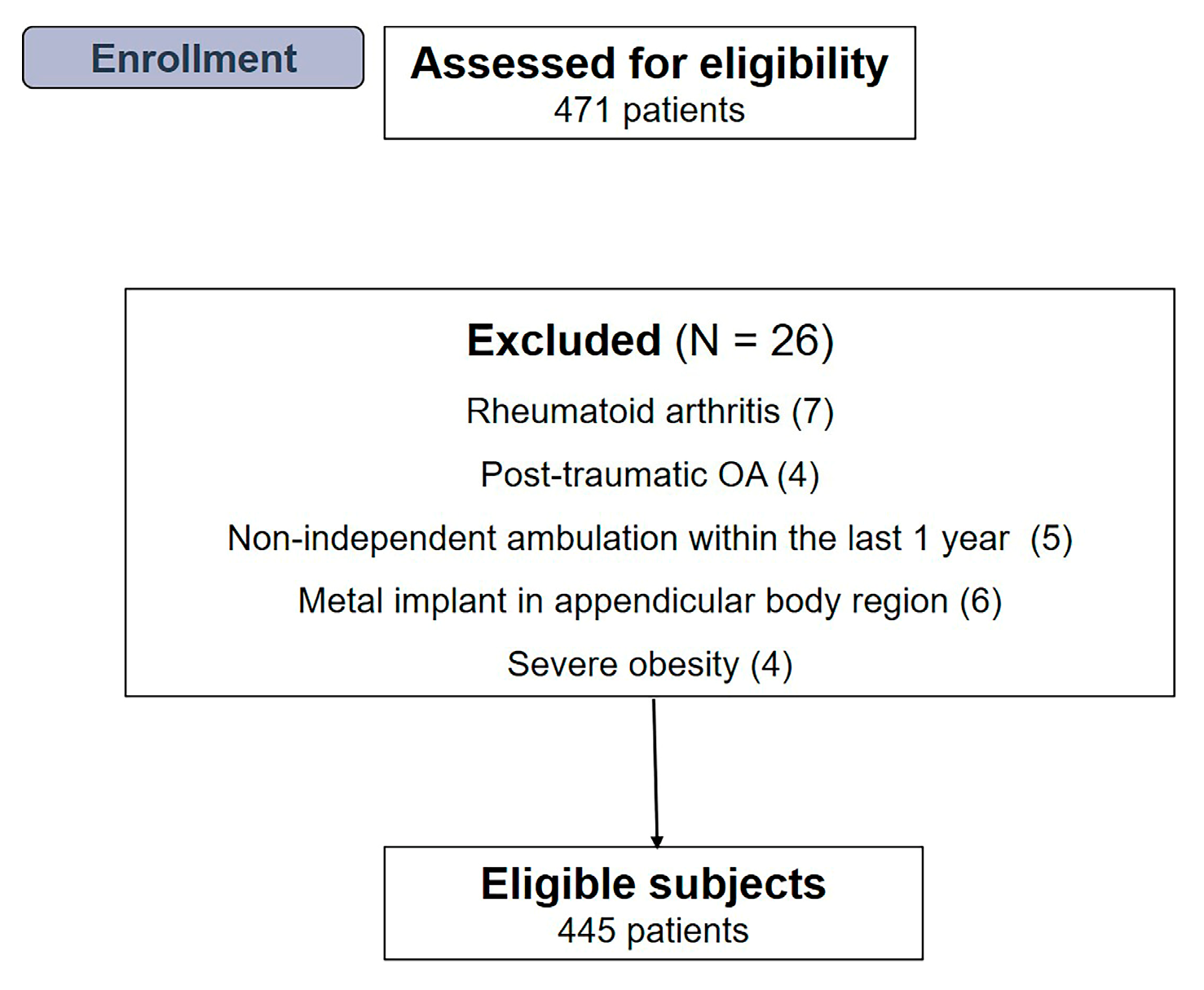
2.1. Participants
2.2. Anthropometric Measurements
2.3. Definition of Sarcopenia
2.4. Outcome Assessments
2.5. Postoperative Protocols
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S. Disability-adjusted life years (dalys) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef] [PubMed]
- Stenquist, D.S.; Elman, S.A.; Davis, A.M.; Bogart, L.M.; Brownlee, S.A.; Sanchez, E.S.; Santiago, A.; Ghazinouri, R.; Katz, J.N. Physical activity and experience of total knee replacement in patients one to four years postsurgery in the dominican republic: A qualitative study. Arthritis Care Res. 2015, 67, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Canovas, F.; Dagneaux, L. Quality of life after total knee arthroplasty. Orthop. Traumatol. Surg. Res. 2018, 104, S41–S46. [Google Scholar] [CrossRef]
- Chang, M.J.; Kim, S.H.; Kang, Y.G.; Chang, C.B.; Kim, T.K. Activity levels and participation in physical activities by korean patients following total knee arthroplasty. BMC Musculoskelet. Disord. 2014, 15, 240. [Google Scholar] [CrossRef]
- Inacio, M.; Paxton, E.; Graves, S.; Namba, R.; Nemes, S. Projected increase in total knee arthroplasty in the united states—An alternative projection model. Osteoarthr. Cart. 2017, 25, 1797–1803. [Google Scholar] [CrossRef]
- Kim, T.W.; Kang, S.-B.; Chang, C.B.; Moon, S.-Y.; Lee, Y.-K.; Koo, K.-H. Current trends and projected burden of primary and revision total knee arthroplasty in korea between 2010 and 2030. J. Arthroplast. 2021, 36, 93–101. [Google Scholar] [CrossRef]
- Koh, I.J.; Kim, T.K.; Chang, C.B.; Cho, H.J.; In, Y. Trends in use of total knee arthroplasty in korea from 2001 to 2010. Clin. Orthop. Relat. Res. 2013, 471, 1441–1450. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M. Sarcopenia: European consensus on definition and diagnosisreport of the european working group on sarcopenia in older peoplea. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Bokshan, S.L.; Han, A.L.; DePasse, J.M.; Eltorai, A.E.; Marcaccio, S.E.; Palumbo, M.A.; Daniels, A.H. Effect of sarcopenia on postoperative morbidity and mortality after thoracolumbar spine surgery. Orthopedics 2016, 39, e1159–e1164. [Google Scholar] [CrossRef]
- Evans, W.J. Sarcopenia should reflect the contribution of age-associated changes in skeletal muscle to risk of morbidity and mortality in elderly people. J. Am. Med. Dir. Assoc. 2015, 16, 546–547. [Google Scholar] [CrossRef]
- Volpi, E.; Nazemi, R.; Fujita, S. Muscle tissue changes with aging. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 405. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A. Sarcopenia: Revised european consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Bennell, K.L.; Wrigley, T.V.; Hunt, M.A.; Lim, B.-W.; Hinman, R.S. Update on the role of muscle in the genesis and management of knee osteoarthritis. Rheum. Dis. Clin. North. Am. 2013, 39, 145–176. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, A.; Mills, P.M.; Barrett, R.S. Muscle weakness in hip osteoarthritis: A systematic review. Arthritis Care Res. 2013, 65, 340–352. [Google Scholar] [CrossRef]
- Baker, K.R.; Xu, L.; Zhang, Y.; Nevitt, M.; Niu, J.; Aliabadi, P.; Yu, W.; Felson, D. Quadriceps weakness and its relationship to tibiofemoral and patellofemoral knee osteoarthritis in chinese: The beijing osteoarthritis study. Arthritis Rheum. 2004, 50, 1815–1821. [Google Scholar] [CrossRef]
- Zhai, G.; Blizzard, L.; Srikanth, V.; Ding, C.; Cooley, H.; Cicuttini, F.; Jones, G. Correlates of knee pain in older adults: Tasmanian older adult cohort study. Arthritis Care Res. 2006, 55, 264–271. [Google Scholar] [CrossRef]
- Ho, K.K.-W.; Lau, L.C.-M.; Chau, W.-W.; Poon, Q.; Chung, K.-Y.; Wong, R.M.-Y. End-stage knee osteoarthritis with and without sarcopenia and the effect of knee arthroplasty—A prospective cohort study. BMC Geriatr. 2021, 21, 2. [Google Scholar] [CrossRef]
- McIsaac, D.; Beaule, P.; Bryson, G.; Van Walraven, C. The impact of frailty on outcomes and healthcare resource usage after total joint arthroplasty: A population-based cohort study. Bone Jt. J. 2016, 98, 799–805. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Smith, R.; Aulet, M.; Bensen, B.; Lichtman, S.; Wang, J.; Pierson, R., Jr. Appendicular skeletal muscle mass: Measurement by dual-photon absorptiometry. Am. J. Clin. Nutr. 1990, 52, 214–218. [Google Scholar] [CrossRef]
- Chen, L.-K.; Woo, J.; Assantachai, P.; Auyeung, T.-W.; Chou, M.-Y.; Iijima, K.; Jang, H.C.; Kang, L.; Kim, M.; Kim, S. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307.e302. [Google Scholar] [CrossRef]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Lakomkin, N.; Zuckerman, S.L.; Stannard, B.; Montejo, J.; Sussman, E.S.; Virojanapa, J.; Kuzmik, G.; Goz, V.; Hadjipanayis, C.G.; Cheng, J.S. Preoperative risk stratification in spine tumor surgery: A comparison of the modified charlson index, frailty index, and asa score. Spine 2019, 44, E782–E787. [Google Scholar] [CrossRef] [PubMed]
- Roos, E.M.; Lohmander, L.S. The knee injury and osteoarthritis outcome score (koos): From joint injury to osteoarthritis. Health Qual. Life Outcomes 2003, 1, 64. [Google Scholar] [CrossRef]
- Bellamy, N.; Buchanan, W.W.; Goldsmith, C.H.; Campbell, J.; Stitt, L.W. Validation study of womac: A health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 1988, 15, 1833–1840. [Google Scholar] [PubMed]
- Norkin, C.C.; White, D.J. Measurement of Joint Motion: A Guide to Goniometry; FA Davis: Philadelphia, PA, USA, 2016. [Google Scholar]
- Jo, C.; Ko, S.; Shin, W.C.; Han, H.-S.; Lee, M.C.; Ko, T.; Ro, D.H. Transfusion after total knee arthroplasty can be predicted using the machine learning algorithm. Knee Surg. Sport. Traumatol. Arthrosc. 2020, 28, 1757–1764. [Google Scholar] [CrossRef]
- Parvizi, J.; Tan, T.L.; Goswami, K.; Higuera, C.; Della Valle, C.; Chen, A.F.; Shohat, N. The 2018 definition of periprosthetic hip and knee infection: An evidence-based and validated criteria. J. Arthroplast. 2018, 33, 1309–1314. [Google Scholar] [CrossRef]
- Song, S.J.; Kim, K.I.; Bae, D.K.; Park, C.H. Mid-term lifetime survivals of octogenarians following primary and revision total knee arthroplasties were satisfactory: A retrospective single center study in contemporary period. Knee Surg. Relat. Res. 2020, 32, 50. [Google Scholar] [CrossRef]
- Misra, D.; Fielding, R.A.; Felson, D.T.; Niu, J.; Brown, C.; Nevitt, M.; Lewis, C.E.; Torner, J.; Neogi, T.; MOST study. Risk of knee osteoarthritis with obesity, sarcopenic obesity, and sarcopenia. Arthritis Rheumatol. 2019, 71, 232–237. [Google Scholar] [CrossRef]
- Papalia, R.; Zampogna, B.; Torre, G.; Lanotte, A.; Vasta, S.; Albo, E.; Tecame, A.; Denaro, V. Sarcopenia and its relationship with osteoarthritis: Risk factor or direct consequence? Musculoskelet. Surg. 2014, 98, 9–14. [Google Scholar] [CrossRef]
- Von Haehling, S.; Morley, J.E.; Anker, S.D. An overview of sarcopenia: Facts and numbers on prevalence and clinical impact. J. Cachexia Sarcopenia Muscle 2010, 1, 129–133. [Google Scholar] [CrossRef]
- Babu, J.M.; Kalagara, S.; Durand, W.; Antoci, V.; Deren, M.E.; Cohen, E. Sarcopenia as a risk factor for prosthetic infection after total hip or knee arthroplasty. J. Arthroplast. 2019, 34, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Park, C.J.; Jung, M.-G.; Choi, Y.H.; Park, K.-S.; Koh, H.S. High prevalence of sarcopenia in asian female patients awaiting primary total knee arthroplasty: Application of updated diagnostic tools from the asian working group for sarcopenia. J. Orthop. Surg. 2022, 30, 10225536221113034. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Granic, A.; Davies, K.; Kirkwood, T.B.; Jagger, C.; Sayer, A.A. Prevalence and incidence of sarcopenia in the very old: Findings from the newcastle 85+ study. J. Cache Sarcop Muscle 2017, 8, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Wong, M.; Leung, J.; Lee, J.; Auyeung, T.W.; Woo, J. Incidence, reversibility, risk factors and the protective effect of high body mass index against sarcopenia in community-dwelling older c hinese adults. Geriatr. Gerontol. Int. 2014, 14, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-R.; Cho, S.-K.; Im, S.G.; Jung, S.-Y.; Kim, D.; Jang, E.J.; Sung, Y.-K. Treatment patterns of knee osteoarthritis patients in Korea. Korean J. Intern. Med. 2019, 34, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total |
|---|---|
| Peripheral vascular disease or pain at rest | 1 |
| Congestive heart failure | 1 |
| Prior MI | 1 |
| DM | 1 |
| Prior transient ischemic attack or stroke | 1 |
| COPD | 1 |
| Renal failure | 2 |
| Hemiplegia | 2 |
| Ascites or esophageal varices | 3 |
| Disseminated cancer | 6 |
| ≤40 yrs old | 0 |
| 41–51 years old | 1 |
| 51–60 years old | 2 |
| 61–70 years old | 3 |
| ≥70 years old | 4 |
| Variables | Total N = 445 (100%) | Group S N = 42 (9.4%) | Group NS N = 403 (90.6%) | p Value |
|---|---|---|---|---|
| Age, years 1 | 71.8 (60–88) | 76.2 (69–88) | 67.8 (60–76) | <0.001 4 |
| Sex, n 2 Female, n Male, n | ||||
| 393 (88.3) | 34 (83.3) | 359 (88.8) | 0.129 5 | |
| 52 (11.7) | 8 (16.7) | 44 (11.2) | ||
| BMI, kg/m2 3 | 26.9 ± 3.5 | 23.2 ± 3.2 | 27.6 ± 3.1 | <0.001 4 |
| F/U period, months 1 | 14.5 (12–27) | 12.4 (12–27) | 12.3 (12–27) | 0.871 4 |
| Bilaterality, n 2 | 190 (42.7) | 17 (40.5) | 173 (42.9) | 0.870 5 |
| Preop K-L grade, n 2 Grade III, n Grade IV, n | ||||
| 112 (25.2) | 11 (26.2) | 101 (25.1) | 0.853 5 | |
| 333 (74.8) | 31 (73.8) | 302 (74.9) | ||
| mCCI, n 2 | ||||
| 2 | 18 (4.0) | - | 18 (4.5) | |
| 3 and 4 | 210 (47.2) | 3 (7.1) | 207 (51.4) | |
| 5–8 | 156 (35.1) | 20 (47.6) | 136 (33.7) | |
| ≥9 | 61 (13.7) | 19 (45.2) | 42 (10.4) | <0.001 5 |
| Hb level, (g/dL) 3 | 12.6 ± 1.9 | 11.8 ± 2.5 | 12.9 ± 1.6 | 0.021 4 |
| Total protein, (g/dL) 3 | 6.7 ± 1.2 | 6.1 ± 1.6 | 6.9 ± 1.0 | 0.037 4 |
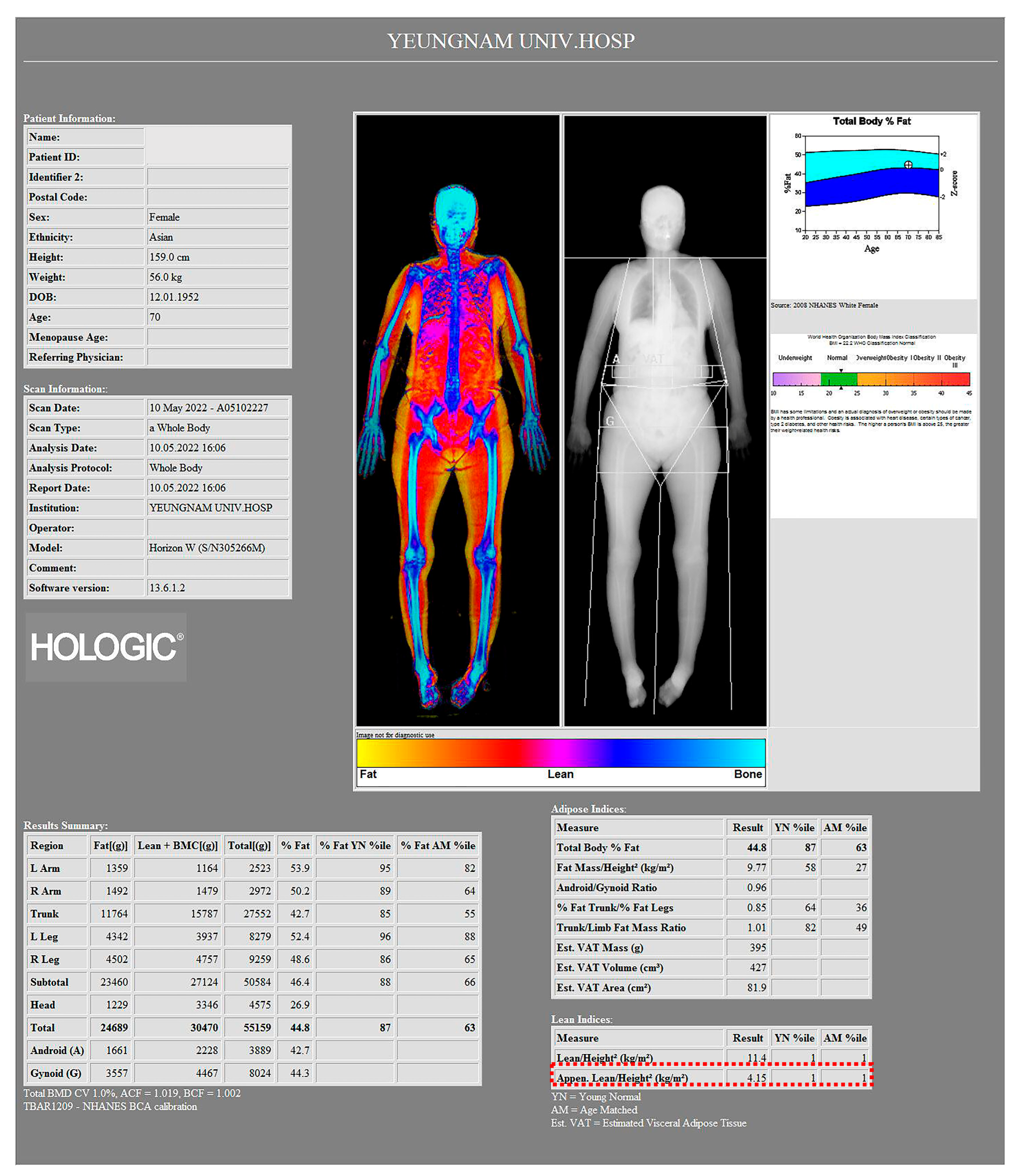
| ASMI (ASM/height2), kg/m2 3 | 5.7 ± 0.6 | 5.2 ± 0.4 | 6.1 ± 0.6 | <0.001 4 |
| Grip Strength, kg 3 | 17.9 ± 3.2 | 16.2 ± 3.2 | 19.0 ± 3.2 | <0.001 4 |
| 6 m walking speed, m/s 3 | 1.1 ± 0.2 | 0.8 ± 0.2 | 1.3 ± 0.2 | 0.018 4 |
| Total | 60–69 Years | 70–79 Years | ≥80 Years | p Value | |
|---|---|---|---|---|---|
| Female | 34/393 (8.7) | 7/154 (4.5) | 16/195 (8.2) | 11/44 (25.0) | |
| Male | 8/52 (15.4) | 0/11 | 2/14 (14.3) | 6/27 (22.2) | |
| Total | 42/445 (9.4) | 7/165 (5.5) | 19/209 (9.1) | 16/71 (22.5) | <0.001 2 |
| Variables | Group S N = 42 | Group NS N = 403 | p Value 2 |
|---|---|---|---|
| KOOS Pain | |||
| Preop | 43.8 ± 8.1 | 44.3 ± 8.5 | 0.738 |
| PO at 3 months | 80.7 ± 6.0 | 81.0 ± 7.9 | 0.681 |
| PO at 6 months | 82.1 ± 6.1 | 82.0 ± 6.5 | 0.731 |
| PO at 12 months | 84.1 ± 4.9 | 84.2 ± 5.1 | 0.542 |
| KOOS Symptom | |||
| Preop | 47.2 ± 9.2 | 46.8 ± 8.1 | 0.814 |
| PO at 3 months | 60.1 ± 6.2 | 65.4 ± 6.1 | 0.047 |
| PO at 6 months | 69.4 ± 4.2 | 76.1 ± 4.9 | 0.026 |
| PO at 12 months | 71.4 ± 2.1 | 78.1 ± 3.9 | 0.061 |
| KOOS ADL | |||
| Preop | 42.1 ± 6.8 | 42.8 ± 5.3 | 0.713 |
| PO at 3 months | 65.1 ± 8.2 | 75.4 ± 6.1 | <0.001 |
| PO at 6 months | 73.8 ± 2.2 | 80.1 ± 2.7 | <0.001 |
| PO at 12 months | 81.8 ± 2.2 | 83.1 ± 1.7 | 0.106 |
| KOOS Sport/Rec | |||
| Preop | 33.2 ± 5.8 | 32.9 ± 3.9 | 0.523 |
| PO at 3 months | 45.1 ± 8.2 | 55.4 ± 5.1 | <0.001 |
| PO at 6 months | 67.8 ± 3.2 | 77.1 ± 4.7 | <0.001 |
| PO at 12 months | 75.1 ± 3.7 | 77.7 ± 4.2 | 0.720 |
| KOOS QOL | |||
| Preop | 40.1 ± 7.1 | 40.7 ± 4.8 | 0.832 |
| PO at 3 months | 72.1 ± 9.1 | 78.7 ± 8.8 | <0.001 1 |
| PO at 6 months | 78.9 ± 5.0 | 84.1 ± 3.2 | <0.001 1 |
| PO at 12 months | 82.9 ± 1.9 | 82.1 ± 2.3 | 0.638 |
| NOTE. 1 Data are presented as number ± standard deviation. 2 Independent samples t-test was used to compare the difference in clinical outcomes between the groups. The level of statistical significance was set at p < 0.05. KOOS, Knee Injury and Osteoarthritis Outcome Score; Preop, preoperatively; PO, postoperatively; ADL, activities of daily living; Rec, recreation; QOL, quality of life. | |||
| (B) WOMAC 1 | |||
| Variables | Group S N = 42 | Group NS N = 403 | p Value 2 |
| WOMAC Pain | |||
| Preop | 9.6 ± 7.2 | 9.7 ± 4.1 | 0.614 |
| PO at 3 months | 6.6 ± 5.2 | 4.7 ± 4.1 | 0.031 |
| PO at 6 months | 3.7 ± 4.3 | 2.8 ± 2.9 | 0.017 |
| PO at 12 months | 2.4 ± 2.3 | 2.3 ± 2.9 | 0.532 |
| WOMAC Stiffness | |||
| Preop | 5.2 ± 2.1 | 5.6 ± 2.7 | 0.729 |
| PO at 3 months | 3.8 ± 6.1 | 2.9 ± 4.7 | 0.032 |
| PO at 6 months | 3.3 ± 1.6 | 2.1 ± 1.9 | 0.017 |
| PO at 12 months | 2.3 ± 1.6 | 2.2 ± 1.9 | 0.817 |
| WOMAC Function | |||
| Preop | 34.1 ± 5.5 | 33.8 ± 6.1 | 0.738 |
| PO at 3 months | 24.1 ± 9.5 | 15.1 ± 5.1 | <0.001 |
| PO at 6 months | 19.2 ± 6.9 | 8.7 ± 2.3 | <0.001 |
| PO at 12 months | 9.2 ± 2.9 | 7.7 ± 2.3 | 0.068 |
| WOMAC Total | |||
| Preop | 48.9 ± 7.1 | 49.1 ± 4.6 | 0.828 |
| PO at 3 months | 34.5 ± 6.8 | 22.7 ± 4.9 | <0.001 |
| PO at 6 months | 26.2 ± 5.8 | 13.6 ± 2.4 | <0.001 |
| PO at 12 months | 11.2 ± 2.5 | 12.2 ± 2.3 | 0.272 |
| NOTE. 1 Data are presented as number ± standard deviation. 2 Independent samples t-test was used to compare the difference in clinical outcomes between the groups. The level of statistical significance was set at p < 0.05. WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index; Preop, preoperatively; PO, postoperatively. | |||
| (C) ROM of the knee joint 1 | |||
| Variables | Group S N = 42 | Group NS N = 403 | p Value 2 |
| FC (°) | |||
| Preop | 7.8 ± 2.9 | 8.3 ± 3.3 | 0.672 |
| PO at 3 months | 1.6 ± 4.2 | 1.7 ± 4.3 | 0.788 |
| PO at 6 months | 1.8 ± 1.9 | 1.9 ± 2.1 | 0.539 |
| PO at 12 months | 1.0 ± 0.9 | 0.8 ± 1.0 | 0.491 |
| FF (°) | |||
| Preop | 117.9 ± 4.4 | 118.2 ± 4.6 | 0.821 |
| PO at 3 months | 129.8 ± 2.6 | 128.1 ± 3.1 | 0.423 |
| PO at 6 months | 135.3 ± 2.8 | 136.4 ± 2.9 | 0.625 |
| PO at 12 months | 139.8 ± 2.6 | 137.1 ± 2.9 | 0.717 |
| Total | Group S N = 42 | Group NS N = 403 | p Value 2 | ||
|---|---|---|---|---|---|
| Systemic | |||||
| Cardiovascular | 8 (1.8) | 1 (2.4) | 7 (1.7) | 0.551 | |
| Pulmonary | 6 (1.3) | 1 (2.4) | 5 (1.2) | 0.450 | |
| Gastrointestinal | - | - | - | - | |
| Hepatic | 44 (9.8) | 4 (9.5) | 40 (9.9) | 0.934 | |
| Nephrotic | - | - | - | - | |
| Endocrinologic | - | - | - | - | |
| Urologic | 87 (19.5) | 8 (19.0) | 79 (19.6) | 0.931 | |
| Cerebral | 7 (1.6) | 1 (2.4) | 6 (1.5) | 0.503 | |
| Delirium | 46 (10.3) | 7 (16.7) | 39 (9.7) | 0.179 | |
| Specific | |||||
| Blood transfusion | 31 (7.0) | 12 (28.6) | 19 (4.7) | <0.001 | |
| Venous thromboembolism | |||||
| PTE | - | - | - | - | |
| DVT (proximal) | 7 (1.6) | - | 7 (1.7) | 0.389 | |
| DVT (distal) | 42 (9.4) | 4 (9.5) | 38 (9.4) | 0.984 | |
| Infection | 2 (0.4) | 2 (4.8) | - | 0.009 | |
| Periprosthetic fracture | - | - | - | - |
| Variables | p Value | Odds Ratio (95% CI) | ||
|---|---|---|---|---|
| Univariate Analysis | Multivariate Analysis | Univariate Analysis | Multivariate Analysis | |
| Sex | 0.197 | 1.0 (0.3–1.6) | ||
| Age | <0.001 1 | 0.003 2 | 1.5 (0.8–1.7) | 1.4 (0.9–1.8) |
| BMI | <0.001 1 | 0.019 2 | 0.8 (0.6–0.9) | 0.7 (0.6–0.9) |
| F/U period | 0.965 | 1.0 (0.9–1.1) | ||
| Bilaterality | 0.892 | 1.2 (0.8–1.6) | ||
| (Preop) | ||||
| K-L grade | 0.914 | 1.2 (0.8–1.6) | ||
| mCCI | <0.001 1 | 0.039 2 | 1.1 (0.8–1.4) | 1.2 (0.8–1.5) |
| Hb level | 0.021 1 | 0.371 | 0.8 (0.6–0.9) | 0.7 (0.6–0.9) |
| Total protein | 0.037 1 | 0.247 | 0.7 (0.5–0.8) | 0.6 (0.5–0.8) |
| KOOS | 0.126 | 1.0 (1.0–1.1) | ||
| WOMAC | 0.181 | 1.0 (0.9–1.0) | ||
| ROM | 0.802 | 1.2 (0.8–1.6) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shon, O.-J.; Kim, G.B.; Cho, S.J. Does Sarcopenia Accompanying End-Stage Knee Osteoarthritis Affect the Outcomes following Total Knee Arthroplasty? Medicina 2023, 59, 1078. https://doi.org/10.3390/medicina59061078
Shon O-J, Kim GB, Cho SJ. Does Sarcopenia Accompanying End-Stage Knee Osteoarthritis Affect the Outcomes following Total Knee Arthroplasty? Medicina. 2023; 59(6):1078. https://doi.org/10.3390/medicina59061078
Chicago/Turabian StyleShon, Oog-Jin, Gi Beom Kim, and Seung Jae Cho. 2023. "Does Sarcopenia Accompanying End-Stage Knee Osteoarthritis Affect the Outcomes following Total Knee Arthroplasty?" Medicina 59, no. 6: 1078. https://doi.org/10.3390/medicina59061078
APA StyleShon, O.-J., Kim, G. B., & Cho, S. J. (2023). Does Sarcopenia Accompanying End-Stage Knee Osteoarthritis Affect the Outcomes following Total Knee Arthroplasty? Medicina, 59(6), 1078. https://doi.org/10.3390/medicina59061078






