Cardiovascular Safety of Hydroxychloroquine–Azithromycin in 424 COVID-19 Patients
Abstract
1. Introduction
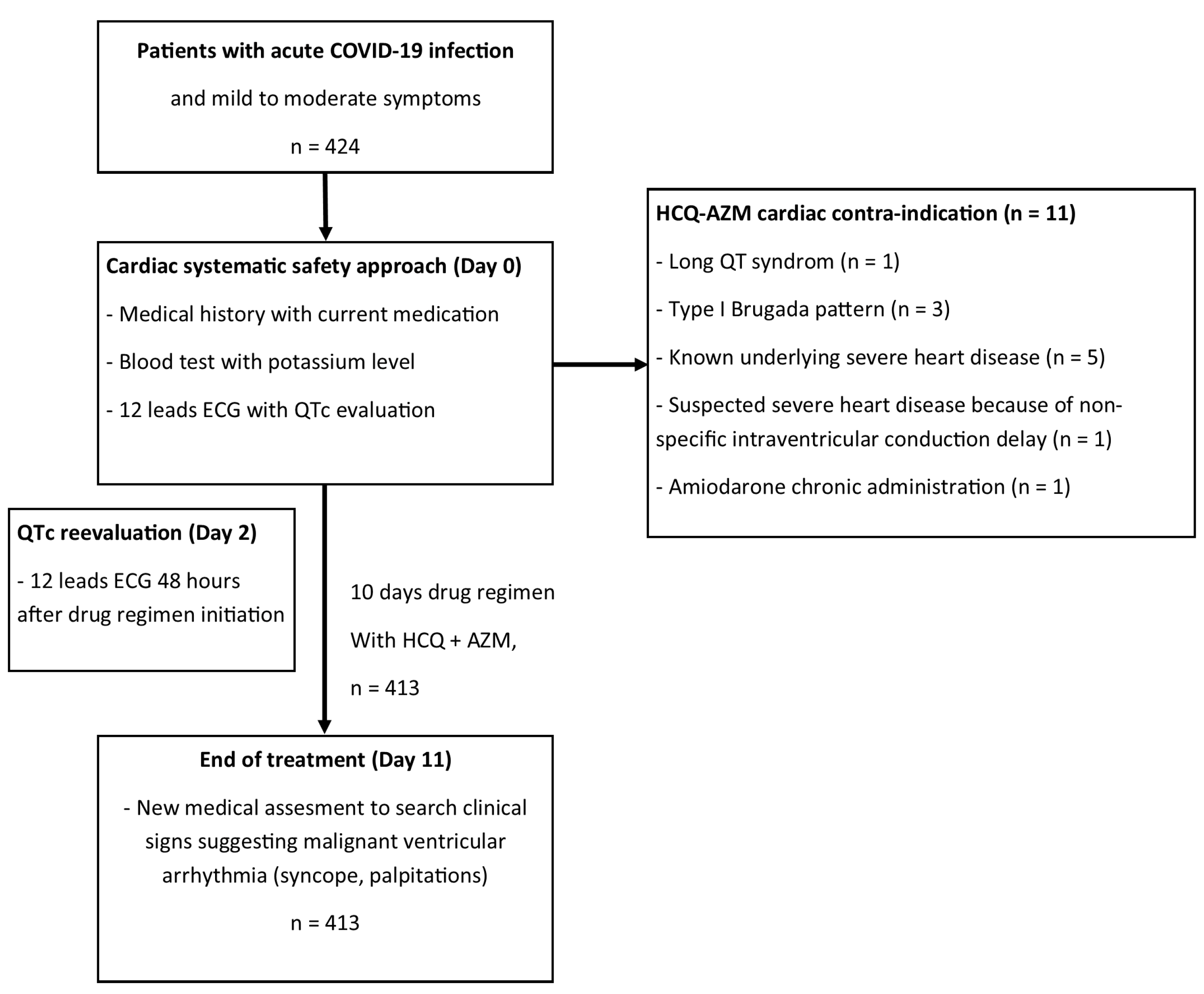
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coronavirus Resource Center. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Available online: https://coronavirus.jhu.edu/map.html (accessed on 31 March 2022).
- Gautret, P.; Million, M.; Jarrot, P.A.; Camoin-Jau, L.; Colson, P.; Fenollar, F.; Leone, M.; La Scola, B.; Devaux, C.; Gaubert, J.Y.; et al. Natural history of COVID-19 and therapeutic options. Expert Rev. Clin. Immunol. 2020, 16, 1159–1184. [Google Scholar] [CrossRef]
- World Health Organization. Nirmatrelvir-Ritonavir for COVID-19; World Health Organization: Geneva, Switzerland, 2022; Available online: https://apps.who.int/iris/handle/10665/359751 (accessed on 31 March 2023).
- Global HCQ/CQ Studies. Available online: https://c19study.com (accessed on 31 March 2022).
- Lagier, J.C.; Million, M.; Cortaredona, S.; Delorme, L.; Colson, P.; Fournier, P.E.; Brouqui, P.; Raoult, D.; Parola, P.; IHU Task Force. Outcomes of 2111 COVID-19 Hospitalized Patients Treated with Hydroxychloroquine/Azithromycin and Other Regimens in Marseille, France, 2020: A Monocentric Retrospective Analysis. Ther. Clin. Risk Manag. 2022, 18, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Million, M.; Lagier, J.C.; Tissot-Dupont, H.; Ravaux, I.; Dhiver, C.; Tomei, C.; Cassir, N.; Delorme, L.; Cortaredona, S.; Amrane, S.; et al. Early combination therapy with hydroxychloroquine and azithromycin reduces mortality in 10,429 COVID-19 outpatients. Rev. Cardiovasc. Med. 2021, 22, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Cao, Z.; Han, M.; Wang, Z.; Chen, J.; Sun, W.; Wu, Y.; Xiao, W.; Liu, S.; Chen, E.; et al. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: Open label, randomised controlled trial. BMJ 2020, 369, m1849. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, A.B.; Zampieri, F.G.; Rosa, R.G.; Azevedo, L.C.P.; Veiga, V.C.; Avezum, A.; Damiani, L.P.; Marcadenti, A.; Kawano-Dourado, L.; Lisboa, T.; et al. Hydroxychloroquine with or without Azithromycin in Mild-to-Moderate COVID-19. N. Engl. J. Med. 2020, 383, 2041. [Google Scholar] [CrossRef] [PubMed]
- Self, W.H.; Semler, M.W.; Leither, L.M.; Casey, J.D.; Angus, D.C.; Brower, R.G.; Chang, S.Y.; Collins, S.P.; Eppensteiner, J.C.; Filbin, M.R.; et al. Effect of Hydroxychloroquine on Clinical Status at 14 Days in Hospitalized Patients with COVID-19: A Randomized Clinical Trial. JAMA 2020, 324, 2165. [Google Scholar] [CrossRef]
- Geleris, J.; Sun, Y.; Platt, J.; Zucker, J.; Baldwin, M.; Hripcsak, G.; Labella, A.; Manson, D.K.; Kubin, C.; Barr, R.G.; et al. Observational Study of Hydroxychloroquine in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2020, 382, 2411–2418. [Google Scholar] [CrossRef]
- Chatre, C.; Roubille, F.; Vernhet, H.; Jorgensen, C.; Pers, Y.-M. Cardiac Complications Attributed to Chloroquine and Hydroxychloroquine: A Systematic Review of the Literature. Drug Saf. 2018, 41, 919–931. [Google Scholar] [CrossRef]
- Ray, W.A.; Murray, K.T.; Hall, K.; Arbogast, P.G.; Stein, C.M. Azithromycin and the risk of cardiovascular death. N. Engl. J. Med. 2012, 366, 1881–1890. [Google Scholar] [CrossRef]
- Chorin, E.; Dai, M.; Shulman, E.; Wadhwani, L.; Bar-Cohen, R.; Barbhaiya, C.; Aizer, A.; Holmes, D.; Bernstein, S.; Spinelli, M.; et al. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat. Med. 2020, 26, 808–809. [Google Scholar] [CrossRef]
- Bessière, F.; Roccia, H.; Delinière, A.; Charrière, R.; Chevalier, P.; Argaud, L.; Cour, M. Assessment of QT intervals in a case series of patients with coronavirus disease 2019 (COVID-19) infection treated with hydroxychloroquine alone or in combination with azithromycin in an intensive care unit. JAMA Cardiol. 2020, 5, 1067–1069. [Google Scholar] [CrossRef]
- Gasperetti, A.; Biffi, M.; Duru, F.; Schiavone, M.; Ziacchi, M.; Mitacchione, G.; Lavalle, C.; Saguner, A.; Lanfranchi, A.; Casalini, G.; et al. Arrhythmic safety of hydroxychloroquine in COVID-19 patients from different clinical settings. Europace 2020, 22, 1855–1863. [Google Scholar] [CrossRef] [PubMed]
- Hancox, J.C.; Hasnain, M.; Vieweg, W.V.R.; Crouse, E.L.B.; Baranchuk, A. Azithromycin, cardiovascular risks, QTc interval prolongation, torsade de pointes, and regulatory issues: A narrative review based on the study of case reports. Ther. Adv. Infect. Dis. 2013, 1, 155–165. [Google Scholar] [CrossRef]
- Fossa, A.A.; Wisialowski, T.; Duncan, J.N.; Deng, S.; Dunne, M. Azithromycin/Chloroquine combination does not increase cardiac instability despite an increase in monophasic action potential duration in the anesthetized guinea pig. Am. J. Trop. Med. Hyg. 2007, 77, 929–938. [Google Scholar] [CrossRef]
- Brouqui, P.; Drancourt, M.; Raoult, D.; On behalf of the IHU Task Force. COVID-19 Management at IHU Méditerranée Infection: A One-Year Experience. J. Clin. Med. 2021, 10, 2881. [Google Scholar] [CrossRef] [PubMed]
- Million, M.; Lagier, J.C.; Gautret, P.; Colson, P.; Fournier, P.E.; Amrane, S.; Hocquart, M.; Mailhe, M.; Esteves-Vieira, V.; Doudier, B.; et al. Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: A retrospective analysis of 1061 cases in Marseille, France. Travel Med. Infect. Dis. 2020, 35, 101738. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Wang, B.; Kang, Y. Novel coronavirus infection during the 2019–2020 epidemic: Preparing intensive care units-The experience in Sichuan Province, China. Intensive Care Med. 2020, 46, 357–360. [Google Scholar] [CrossRef]
- Haeusler, I.L.; Chan, X.H.S.; Guérin, P.J.; White, N.J. The arrhythmogenic cardiotoxicity of the quinoline and structurally related antimalarial drugs: A systematic review. BMC Med. 2018, 16, 200. [Google Scholar] [CrossRef] [PubMed]
- Costedoat-Chalumeau, N.; Hulot, J.-S.; Amoura, Z.; Leroux, G.; Lechat, P.; Funck-Brentano, C.; Piette, J.C. Heart conduction disorders related to antimalarials toxicity: An analysis of electrocardiograms in 85 patients treated with hydroxychloroquine for connective tissue diseases. Rheumatology 2007, 46, 808–810. [Google Scholar] [CrossRef]
- American College of Cardiology. Ventricular Arrhythmia Risk Due to Hydroxychloroquine-Azithromycin Treatment for COVID-19. Available online: https://www.acc.org/latest-in-cardiology/articles/2020/03/27/14/00/ventricular-arrhythmia-risk-due-to-hydroxychloroquine-azithromycin-treatment-for-covid-19 (accessed on 26 December 2022).
- Maille, B.; Wilkin, M.; Million, M.; Rességuier, N.; Franceschi, F.; Koutbi-Franceschi, L.; Hourdain, J.; Martinez, E.; Zabern, M.; Gardella, C.; et al. Smartwatch electrocardiogram and artificial intelligence for assessing cardiac-rhythm safety of drug therapy in the COVID-19 pandemic. The QT-logs study. Int. J. Cardiol. 2021, 331, 333–339. [Google Scholar] [CrossRef]
- Roden Dan, M.; Harrington Robert, A.; Poppas Athena Russo Andrea, M. Considerations for drug interactions on QTc in exploratory COVID-19 treatment. Circulation 2020, 141, e906–e907. [Google Scholar] [PubMed]
- Chen, D.; Li, X.; Song, Q.; Hu, C.; Su, F.; Dai, J. Hypokalemia and clinical implications in patients with coronavirus disease 2019 (COVID-19). MedRxiv 2020. [Google Scholar] [CrossRef]
- Vandenberk, B.; Vandael, E.; Robyns, T.; Vandenberghe, J.; Garweg, C.; Foulon, V.; Ector, J.; Willems, R. Which QT correction formulae to use for QT monitoring? J. Am. Heart Assoc. 2016, 5, e003264. [Google Scholar] [CrossRef]
- O’Connell, T.F.; Bradley, C.J.; Abbas, A.E.; Williamson, B.D.; Rusia, A.; Tawney, A.M.; Gaines, R.; Schott, J.; Dmitrienko, A.; Haines, D.E. Hydroxychloroquine/Azithromycin therapy and QT prolongation in hospitalized patients with COVID-19. JACC Clin. Electrophysiol. 2021, 7, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Million, M.; Cortaredona, S.; Delorme, L.; Colson, P.; Levasseur, A.; Tissot-Dupont, H.; Bendamardji, K.; Lahouel, S.; La Scola, B.; Camoin, L.; et al. Early Treatment with Hydroxychloroquine and Azithromycin: A ‘Real-Life’ Monocentric Retrospective Cohort Study of 30,423 COVID-19 Patients. medRxiv 2023. [Google Scholar] [CrossRef]
| Molecules | Indications in Infectious Diseases |
|---|---|
| Polyenes Amphotericin B | Fungal infections |
| Antimalarial combinations Artemether/lumefantrine Artenimol/piperaquine | Malaria |
| HIV Protease inhibitors Atazanavir Lopinavir/ritonavir Nelfinavir Saquinavir | HIV |
| Macrolides Azithromycin b Clarithromycin Erythromycin Roxithromycin Telithromycin | Bacterial infections |
| Diarylquinolines Bedaquiline | Tuberculosis |
| Antimalarial quinolines Chloroquine Hydroxychloroquine b Primaquine Quinine | Malaria, Q fever |
| Quinolones Ciprofloxacin Levofloxacin Moxifloxacin Norfloxacin Ofloxacin | Bacterial infections |
| Leprostatics Clofazimine | Mycobacterial infection |
| NNRTIs Efavirenz | HIV |
| Azole antifungals Fluconazole Posaconazole Voriconazole | Fungal infections |
| Meglumine antimoniate | Leishmaniosis |
| Amebicides and miscellaneous antibiotics Metronidazole | Amoebiasis and anaerobic bacterial infections |
| Antipseudomonal penicillins Piperacillin/tazobactam | Bacterial infections |
| Purine nucleosides Remdesivir | COVID-19 |
| Sulfonamides Sulfamethoxazole and trimethoprime | Bacterial and parasitic infections |
| Item | Contraindication a | Safe Prescription of HCQ-AZM |
|---|---|---|
| Patient interview: cardiac history | Severe structural or electrical heart disease a | - Absence of history of severe cardiopathy associated with increased risk of torsade de pointe |
| Patient interview: co-medications | Co-medication of HCQ-AZ with QTc-prolonging drugs b | - Patients without QTc-prolonging drugs- Patients for whom QT-prolonging therapy could be discontinued for 10 days |
| Initial 12-lead ECG | Baseline corrected QT interval > 500 ms, channelopathy, Brugada syndrome, pathological Q waves, left ventricular hypertrophy, and left bundle branch block.Any abnormal ECG after cardiological advice c | - Patients with normal ECG- Patients with abnormal ECG but no contraindication to HCQ-AZ after cardiological advice c |
| Kalemia | Dyskalemia (K+ <3.6 mmol/L or K+ >5 mmol/L) d | - Patients without dyskalemia- Patients with corrected dyskalemia |
| Characteristic | Value (n = 424) |
|---|---|
| Male sex—no. (%) | 208 (49.5) |
| Mean age ± SD—year | 46.3 ± 16.1 |
| ≥65 years—no. (%) | 47 (11.1) |
| Clinical setting—no. (%) | |
| Day-care patients | 333 (78.5) |
| Inpatients | 91 (21.5) |
| Cardiovascular treatment—no. (%) | |
| ACE inhibitors/ARBs | 34 (8.0) |
| Beta-blockers | 15 (3.5) |
| Diuretics | 17 (4.0) |
| Calcium channel blockers | 1 (0.2) |
| Digoxin | 1 (0.2) |
| Flecainide | 4 (0,9) |
| Amiodarone | 1(0.2) |
| Baseline ECG | |
| Mean heart rate ± SD—beats/min | 74.6 ± 13.6 |
| Mean QRS duration ± SD—ms | 82.3 ± 1646 |
| Mean QTc duration ± SD—ms | 396.8 ± 29.4 |
| Initial ECG patterns suggesting: | |
| Long QT interval—no. (%) | 1 (0.2) |
| Type I Brugada syndrome—no. (%) | 3 (0.7) |
| Bundle branch block—no. (%) | 40 (9.4) |
| Left ventricular hypertrophy—no. (%) | 4 (0.9) |
| Pathological Q waves—no. (%) | 9 (2.1) |
| Early repolarization pattern—no. (%) | 37 (8.7) |
| QTc risk score Tisdale score (points), median (IQR) | 7 (6–7) |
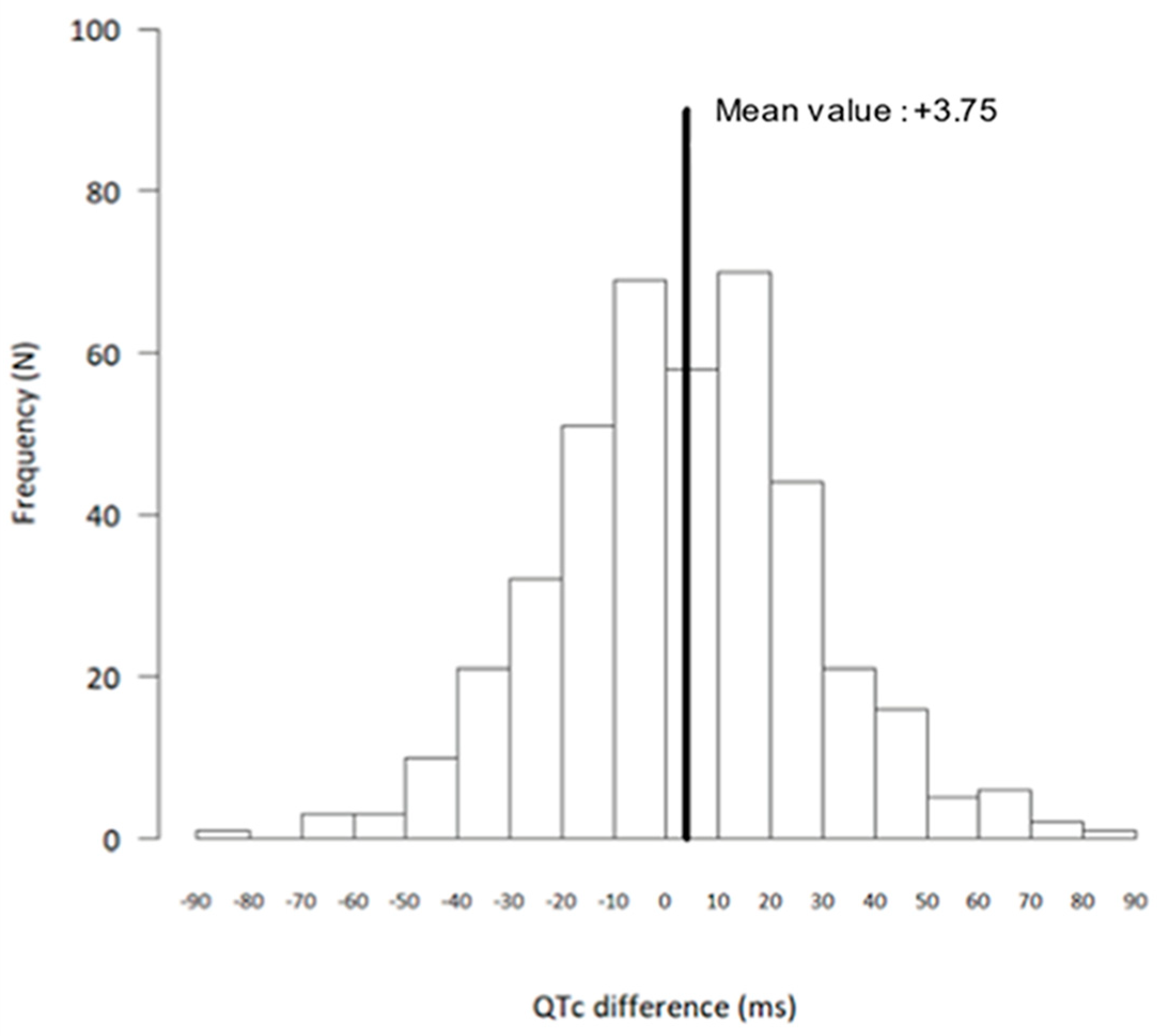
| Variable | Mean Baseline QTc ± SD—ms | Mean Day 2 QTc ± SD—ms | Mean Absolute Difference in QTc (Day 2 vs. Baseline) ± SD—ms | p-Value for Comparison of QTc between Baseline and Day 2 |
|---|---|---|---|---|
| General population (n = 413) | 396.0 ± 28.7 | 399.7 ± 28.7 | +3.75 ± 25.4 | 0.003 |
| Sex | ||||
| Female (n = 214) | 401.1 ± 27.4 | 407.0 ± 25.6 | +5.61 ± 25.3 | 0.001 |
| Male (n = 199) | 390.2 ± 29 | 392.0 ± 29.8 | +1.73 ± 25.5 | 0.31 |
| Age | ||||
| <65 years (n = 366) | 393.7 ± 27.4 | 397.2 ± 27.8 | +3.56 ± 25.3 | 0.007 |
| ≥65 years (n = 47) | 413.9 ± 32.2 | 419.1 ± 27.8 | +4.62 ± 26.6 | 0.19 |
| Cardiovascular disease | ||||
| Absent (n = 350) | 392.2 ± 27.2 | 396.3 ± 28.0 | +4.04 ± 25.9 | 0.004 |
| Present (n = 63) | 416.8 ± 27.9 | 418.9 ± 24.5 | +2.11 ± 22.7 | 0.47 |
| Patient setting | ||||
| Day-care (n = 328) | 391.8 ± 27.8 | 395.9 ± 27.8 | +4.11 ± 26.2 | 0.005 |
| Inpatient (n = 85) | 412.3 ± 26.3 | 414.6 ± 27.4 | +2.33 ± 22.1 | 0.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Million, M.; Lagier, J.-C.; Hourdain, J.; Franceschi, F.; Deharo, J.-C.; Parola, P.; Brouqui, P. Cardiovascular Safety of Hydroxychloroquine–Azithromycin in 424 COVID-19 Patients. Medicina 2023, 59, 863. https://doi.org/10.3390/medicina59050863
Million M, Lagier J-C, Hourdain J, Franceschi F, Deharo J-C, Parola P, Brouqui P. Cardiovascular Safety of Hydroxychloroquine–Azithromycin in 424 COVID-19 Patients. Medicina. 2023; 59(5):863. https://doi.org/10.3390/medicina59050863
Chicago/Turabian StyleMillion, Matthieu, Jean-Christophe Lagier, Jérôme Hourdain, Frédéric Franceschi, Jean-Claude Deharo, Philippe Parola, and Philippe Brouqui. 2023. "Cardiovascular Safety of Hydroxychloroquine–Azithromycin in 424 COVID-19 Patients" Medicina 59, no. 5: 863. https://doi.org/10.3390/medicina59050863
APA StyleMillion, M., Lagier, J.-C., Hourdain, J., Franceschi, F., Deharo, J.-C., Parola, P., & Brouqui, P. (2023). Cardiovascular Safety of Hydroxychloroquine–Azithromycin in 424 COVID-19 Patients. Medicina, 59(5), 863. https://doi.org/10.3390/medicina59050863







