Evaluating the Nephrotoxicity of Area-under-the-Curve-Based Dosing of Vancomycin with Concomitant Antipseudomonal Beta-Lactam Antibiotics: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. General Guidelines
2.2. Database Searches and Identification of Eligible Papers
2.3. Inclusion and Exclusion Criteria
2.4. Methodological Quality Appraisal
2.5. Data Extraction and Management
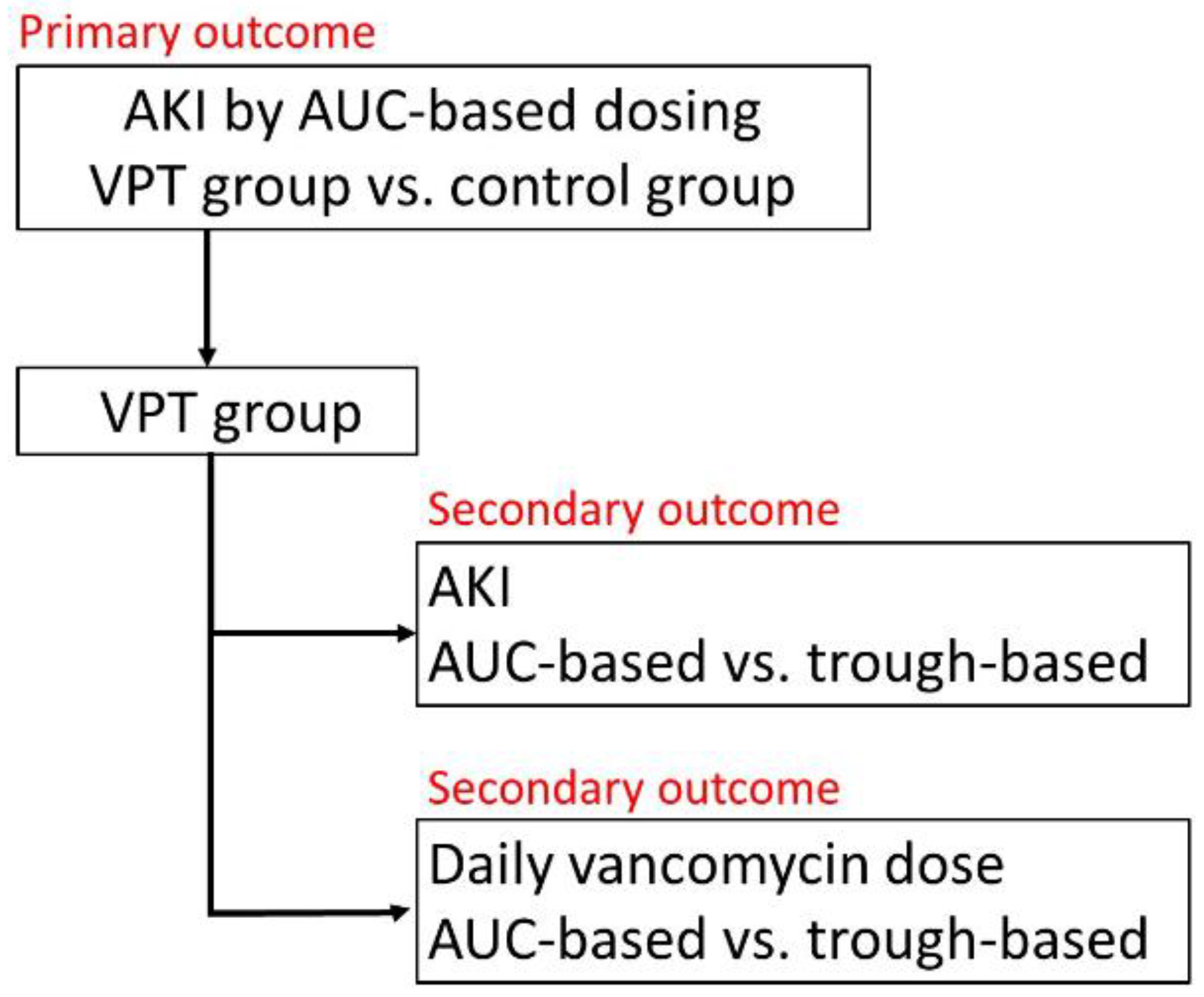
2.6. Primary and Secondary Outcomes
2.7. Statistical Analysis
3. Results
3.1. Study Selection
3.2. Methodological Quality of the Included Studies
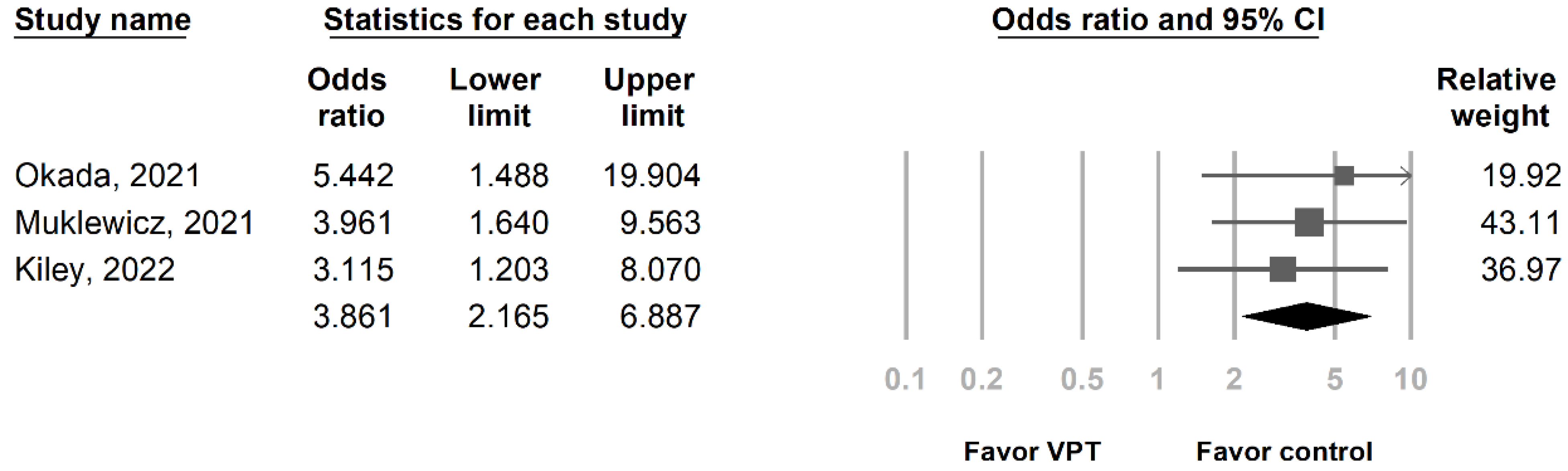
3.3. Primary Outcome: Risk of AKI in Patients Who Received Vancomycin + Piperacillin/Tazobactam under AUC-Based Dosing
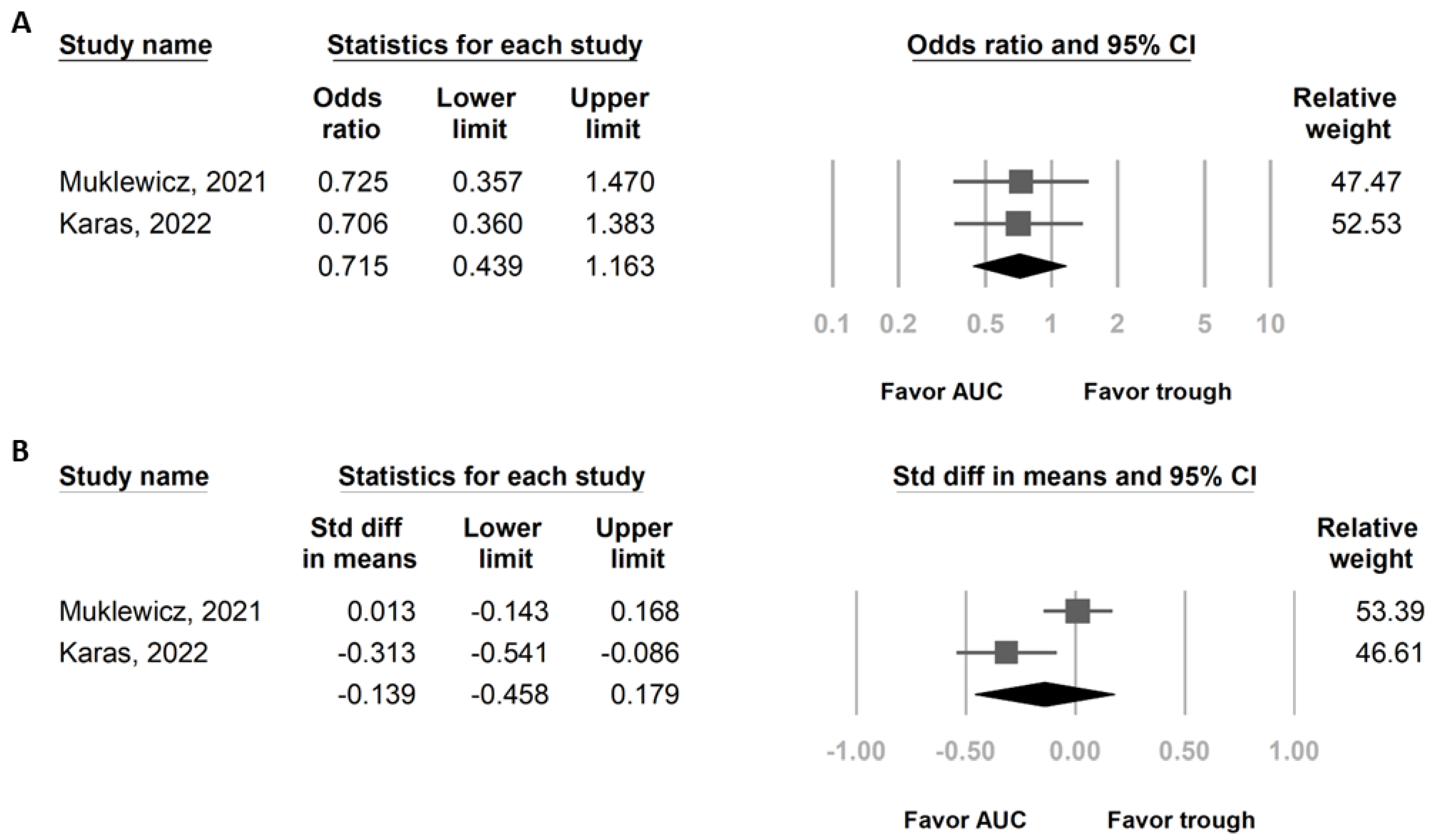
3.4. Secondary Outcomes: Risk of AKI and Daily Vancomycin Dose in Patients Who Received Vancomycin + Piperacillin/Tazobactam under Different Vancomycin Dosing
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, C.; Bayer, A.; Cosgrove, S.E.; Daum, R.S.; Fridkin, S.K.; Gorwitz, R.J.; Kaplan, S.L.; Karchmer, A.W.; Levine, D.P.; Murray, B.E.; et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin. Infect. Dis. 2011, 52, e18–e55. [Google Scholar] [CrossRef]
- Sinha Ray, A.; Haikal, A.; Hammoud, K.A.; Yu, A.S. Vancomycin and the Risk of AKI: A Systematic Review and Meta-Analysis. Clin. J. Am. Soc. Nephrol. 2016, 11, 2132–2140. [Google Scholar] [CrossRef]
- Bellos, I.; Karageorgiou, V.; Pergialiotis, V.; Perrea, D.N. Acute kidney injury following the concurrent administration of antipseudomonal β-lactams and vancomycin: A network meta-analysis. Clin. Microbiol. Infect. 2020, 26, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Luther, M.K.; Timbrook, T.T.; Caffrey, A.R.; Dosa, D.; Lodise, T.P.; LaPlante, K.L. Vancomycin Plus Piperacillin-Tazobactam and Acute Kidney Injury in Adults: A Systematic Review and Meta-Analysis. Crit. Care Med. 2018, 46, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Rybak, M.; Lomaestro, B.; Rotschafer, J.C.; Moellering, R., Jr.; Craig, W.; Billeter, M.; Dalovisio, J.R.; Levine, D.P. Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am. J. Health Syst. Pharm. 2009, 66, 82–98. [Google Scholar] [CrossRef] [PubMed]
- Rybak, M.J.; Le, J.; Lodise, T.P.; Levine, D.P.; Bradley, J.S.; Liu, C.; Mueller, B.A.; Pai, M.P.; Wong-Beringer, A.; Rotschafer, J.C.; et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: A revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Am. J. Health Syst. Pharm. 2020, 77, 835–864. [Google Scholar] [CrossRef] [PubMed]
- Lodise, T.P.; Drusano, G. Vancomycin Area Under the Curve-Guided Dosing and Monitoring for Adult and Pediatric Patients With Suspected or Documented Serious Methicillin-Resistant Staphylococcus aureus Infections: Putting the Safety of Our Patients First. Clin. Infect. Dis. 2021, 72, 1497–1501. [Google Scholar] [CrossRef]
- Aljefri, D.M.; Avedissian, S.N.; Rhodes, N.J.; Postelnick, M.J.; Nguyen, K.; Scheetz, M.H. Vancomycin Area Under the Curve and Acute Kidney Injury: A Meta-analysis. Clin. Infect. Dis. 2019, 69, 1881–1887. [Google Scholar] [CrossRef]
- Finch, N.A.; Zasowski, E.J.; Murray, K.P.; Mynatt, R.P.; Zhao, J.J.; Yost, R.; Pogue, J.M.; Rybak, M.J. A Quasi-Experiment To Study the Impact of Vancomycin Area under the Concentration-Time Curve-Guided Dosing on Vancomycin-Associated Nephrotoxicity. Antimicrob. Agents Chemother. 2017, 61, e01293-17. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ott. Ott. Hosp. Res. Inst. 2011, 2, 1–12. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Fixed-effect versus random-effects models. Introd. Meta-Anal. 2009, 77, 85. [Google Scholar]
- Page, M.J.; Higgins, J.P.; Sterne, J.A. Chapter 13: Assessing risk of Bias Due to Missing Results in a Synthesis. In Cochrane Handbook for Systematic Reviews of Interventions; Cochrane: London, UK, 2019; pp. 349–374. Available online: https://training.cochrane.org/handbook/current/chapter-13 (accessed on 1 March 2023).
- Okada, N.; Izumi, Y.; Nakamoto, A.; Chuma, M.; Goda, M.; Yagi, K.; Aizawa, F.; Hamano, H.; Zamami, Y.; Azuma, M.; et al. Impact of Area under the Concentration-Time Curve on the Prevalence of Vancomycin-Induced Nephrotoxicity in Combination with Tazobactam/Piperacillin or Cefepime: A Single-Institution Retrospective Study. Clin. Ther. 2021, 43, 1910–1920.e3. [Google Scholar] [CrossRef] [PubMed]
- Muklewicz, J.D.; Steuber, T.D.; Edwards, J.D. Evaluation of area under the concentration-time curve-guided vancomycin dosing with or without piperacillin-tazobactam on the incidence of acute kidney injury. Int. J. Antimicrob. Agents 2021, 57, 106234. [Google Scholar] [CrossRef] [PubMed]
- Kiley, P.S.; Pearston, A.P.; Hodge, L.A.; Kaplan, M.C.; Baczek, S.M.; Stanley, J.S.; Wilson, T.J.; Soriano, K.M.; Yao, A.; Shaeffer, Z.A.; et al. Retrospective Cohort Study of the Incidence of Acute Kidney Injury with Vancomycin Area under the Curve-Based Dosing with Concomitant Piperacillin-Tazobactam Compared to Meropenem or Cefepime. Antimicrob. Agents Chemother. 2022, 66, e0004022. [Google Scholar] [CrossRef] [PubMed]
- Karas, C.; Manning, K.; Childress, D.T.; Covington, E.W.; Manis, M.M. Evaluating the Safety of Trough Versus Area Under the Curve (AUC)-Based Dosing Method of Vancomycin With Concomitant Piperacillin-Tazobactam. J. Pharm. Technol. 2022, 38, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Wong, T.; Huang, S.; Mui, E.; Nguyen, V.; Espinosa, G.; Desai, J.; Holubar, M.; Deresinski, S. Conversion from Vancomycin Trough Concentration-Guided Dosing to Area Under the Curve-Guided Dosing Using Two Sample Measurements in Adults: Implementation at an Academic Medical Center. Pharmacotherapy 2019, 39, 433–442. [Google Scholar] [CrossRef]
- Neely, M.N.; Kato, L.; Youn, G.; Kraler, L.; Bayard, D.; van Guilder, M.; Schumitzky, A.; Yamada, W.; Jones, B.; Minejima, E. Prospective Trial on the Use of Trough Concentration versus Area under the Curve To Determine Therapeutic Vancomycin Dosing. Antimicrob. Agents Chemother. 2018, 62, e02042-17. [Google Scholar] [CrossRef]
- Lodise, T.P.; Rosenkranz, S.L.; Finnemeyer, M.; Evans, S.; Sims, M.; Zervos, M.J.; Creech, C.B.; Patel, P.C.; Keefer, M.; Riska, P.; et al. The Emperor’s New Clothes: PRospective Observational Evaluation of the Association Between Initial VancomycIn Exposure and Failure Rates Among ADult HospitalizEd Patients With Methicillin-resistant Staphylococcus aureus Bloodstream Infections (PROVIDE). Clin. Infect. Dis. 2020, 70, 1536–1545. [Google Scholar] [CrossRef]
- Liang, S.Y.; Kumar, A. Empiric antimicrobial therapy in severe sepsis and septic shock: Optimizing pathogen clearance. Curr. Infect. Dis. Rep. 2015, 17, 493. [Google Scholar] [CrossRef]
- Hashimoto, N.; Kimura, T.; Hamada, Y.; Niwa, T.; Hanai, Y.; Chuma, M.; Fujii, S.; Matsumoto, K.; Shigemi, A.; Kawamura, H.; et al. Candidates for area under the concentration-time curve (AUC)-guided dosing and risk reduction based on analyses of risk factors associated with nephrotoxicity in vancomycin-treated patients. J. Glob. Antimicrob. Resist. 2021, 27, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Hammond, D.A.; Smith, M.N.; Li, C.; Hayes, S.M.; Lusardi, K.; Bookstaver, P.B. Systematic Review and Meta-Analysis of Acute Kidney Injury Associated with Concomitant Vancomycin and Piperacillin/tazobactam. Clin. Infect. Dis. 2017, 64, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.V.; Fong, G.; Bolaris, M.; Neely, M.; Minejima, E.; Kang, A.; Lee, G.; Gong, C.L. Cost-benefit analysis comparing trough, two-level AUC and Bayesian AUC dosing for vancomycin. Clin. Microbiol. Infect. 2021, 27, 1346.e1–1346.e7. [Google Scholar] [CrossRef] [PubMed]
- Keil, E.; Wrenn, R.H.; Deri, C.R.; Slaton, C.N.; Shroba, J.; Parish, A.; Erkanli, A.; Spivey, J. Comparison of open-access, trough-only online calculators versus trapezoidal method for calculation of vancomycin area under the curve (AUC). Ann. Pharmacother. 2022, 10600280221138867. [Google Scholar] [CrossRef] [PubMed]
- Miano, T.A.; Hennessy, S.; Yang, W.; Dunn, T.G.; Weisman, A.R.; Oniyide, O.; Agyekum, R.S.; Turner, A.P.; Ittner, C.A.G.; Anderson, B.J.; et al. Association of vancomycin plus piperacillin-tazobactam with early changes in creatinine versus cystatin C in critically ill adults: A prospective cohort study. Intensive Care Med. 2022, 48, 1144–1155. [Google Scholar] [CrossRef]

| Author, Year, Country, Reference | Okada, 2021, Japan, [14] | Muklewicz, 2021, USA, [15] | Kiley, 2022, USA, [16] | Karas, 2022, USA, [17] |
|---|---|---|---|---|
| Study Design | Retrospective, single hospital | Retrospective, 3 hospitals | Retrospective, single hospital | Retrospective, 3 hospitals |
| Period | 04/2010–03/2020 | case: 04/2019–07/2019; control: 08/2019–03/2020 | 10/2019–09/2020 | 01/2015–06/2021 |
| Sample size | VPT: 61, C a: 42 | VPT: 118, C b: 210; AUC-based: 118, trough-based: 118 | VPT: 331, C c: 104 | AUC-based: 150, trough-based: 150 |
| AUC calculation method | Bayesian model | Bayesian model | Bayesian model or Sawchuk–Zaske method | Two-sample AUC or Bayesian model |
| Actual AUC level | VPT: median 452; C a: median 427.7 | AUC mean 445.7 | VPT: mean 503; C c: mean 495, | NA |
| Duration of combination therapy | VPT: median 5 days, C a: median 4 days | VPT: mean 6.2 days, C b: mean 4.8 days; AUC-based: mean 4.0 days, trough-based: mean 4.9 days | VPT: median 4 days, C c: median 4 days | AUC-based: mean 5.18 days; trough-based: mean 5.11 days |
| AKI definition | KDIGO | IDSA/AKIN/RIFLE | RIFLE | IDSA/KDIGO/RIFLE |
| Other outcomes examined | AUC cutoff: 600 | 30 days readmission, in-hospital mortality | AUC cutoff: 550 | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, C.-Y.; Sarwal, A. Evaluating the Nephrotoxicity of Area-under-the-Curve-Based Dosing of Vancomycin with Concomitant Antipseudomonal Beta-Lactam Antibiotics: A Systematic Review and Meta-Analysis. Medicina 2023, 59, 691. https://doi.org/10.3390/medicina59040691
Chiu C-Y, Sarwal A. Evaluating the Nephrotoxicity of Area-under-the-Curve-Based Dosing of Vancomycin with Concomitant Antipseudomonal Beta-Lactam Antibiotics: A Systematic Review and Meta-Analysis. Medicina. 2023; 59(4):691. https://doi.org/10.3390/medicina59040691
Chicago/Turabian StyleChiu, Chia-Yu, and Amara Sarwal. 2023. "Evaluating the Nephrotoxicity of Area-under-the-Curve-Based Dosing of Vancomycin with Concomitant Antipseudomonal Beta-Lactam Antibiotics: A Systematic Review and Meta-Analysis" Medicina 59, no. 4: 691. https://doi.org/10.3390/medicina59040691
APA StyleChiu, C.-Y., & Sarwal, A. (2023). Evaluating the Nephrotoxicity of Area-under-the-Curve-Based Dosing of Vancomycin with Concomitant Antipseudomonal Beta-Lactam Antibiotics: A Systematic Review and Meta-Analysis. Medicina, 59(4), 691. https://doi.org/10.3390/medicina59040691






