Contamination by Antibiotic-Resistant Bacteria on Cell Phones of Vendors in a Peruvian Market
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
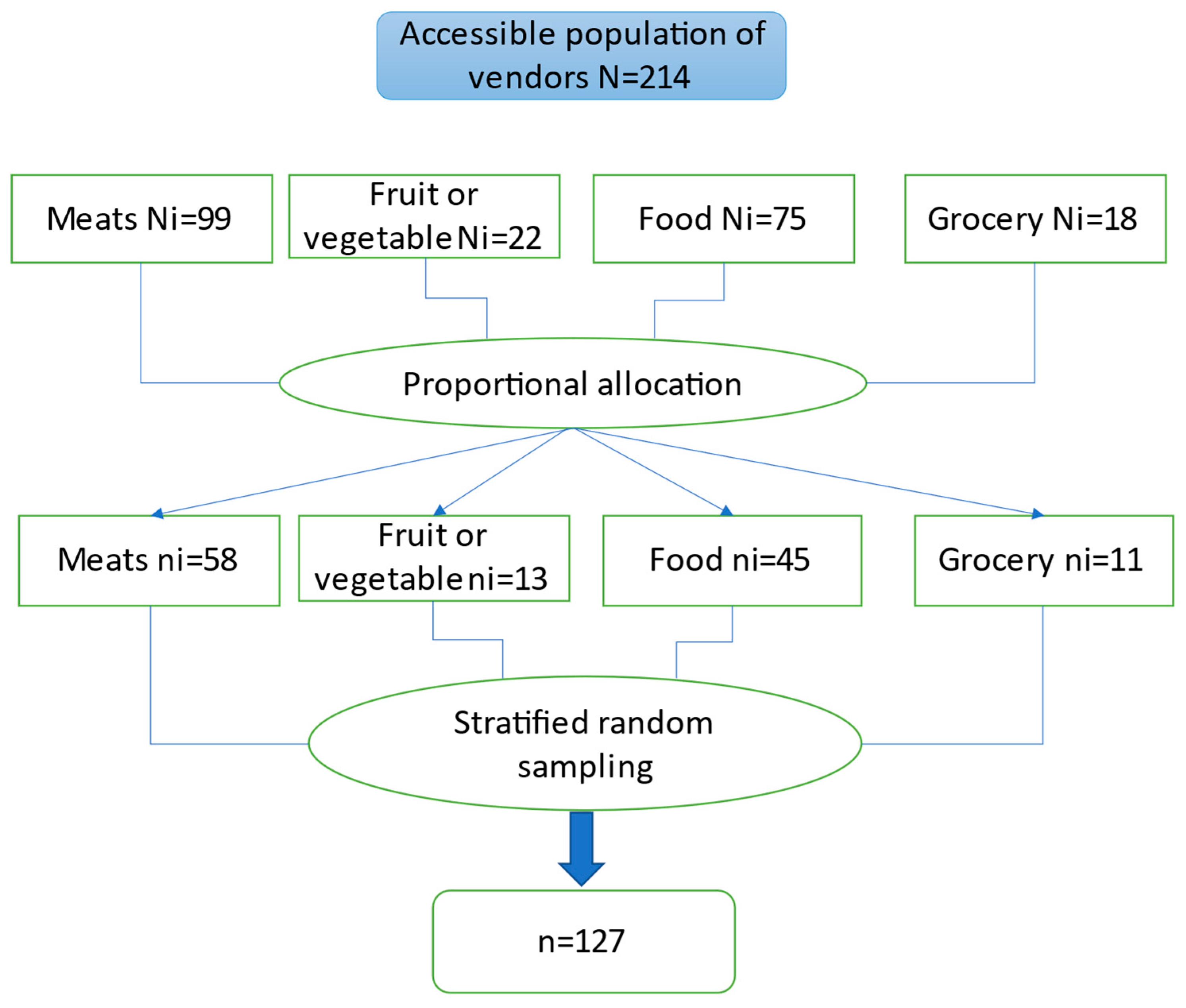
2.2. Population and Sample
2.3. Variables and Instruments
2.4. Isolation and Sensitivity of Bacteria
2.5. Ethical Aspects
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jansen, A.S.; Balbinot, G.C.; Daur, A.V.; Da Silva, A.C.F.; Nogueira, K.S.; Fernandes, T.; Marconi, C. Detection of potentially pathogenic bacteria on cell phones of hospital and university-based populations in Curitiba, southern Brazil. A cross-sectional study. Sao Paulo Med. J. 2019, 137, 343–348. [Google Scholar] [CrossRef]
- Kanayama, A.K.; Takahashi, H.; Yoshizawa, S.; Tateda, K.; Kaneko, A.; Kobayashi, I. Staphylococcus aureus surface contamination of mobile phones and presence of genetically identical strains on the hands of nursing personnel. Am. J. Infect. Control 2017, 45, 929–931. [Google Scholar] [CrossRef]
- Nour, M.O.; Natto, H.A.; Sinky, T.H.; Alfaifi, M.S.; Alharbi, M.F.J.; Allahyani, A.M.K.; Bawahhab, W.A.; Flemban, A.Y.; Al-Zahrani, A.A.D. Infection transmission via mobile phones in a mass gathering setting: Public knowledge, attitudes and practices in Makkah city, Saudi Arabia. Heliyon 2023, 9, e13894. [Google Scholar] [CrossRef]
- Rahman, H.; Rozario, S.R.; Fakhruddin, A.N.M.; Rabbani, K.A. Prevalence of multidrug-Resistant bacteria on mobile phone surface. J. Microsc. Ultrastruct. 2020, 8, 14–19. [Google Scholar] [CrossRef]
- Egert, M.; Späth, K.; Weik, K.; Kunzelmann, H.; Horn, C.; Kohl, M.; Blessing, F. Bacteria on smartphone touchscreens in a German university setting and evaluation of two popular cleaning methods using commercially available cleaning products. Folia Microbiol. 2015, 60, 159–164. [Google Scholar] [CrossRef]
- Wißmann, J.; Kirchhoff, L.; Brüggemann, Y.; Todt, D.; Steinmann, J.; Steinmann, E. Persistence of Pathogens on Inanimate Surfaces: A Narrative Review. Microorganisms 2021, 9, 343. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, V.M.; dos Santos, L.A.M.; Campos-Modesto, L.; Campos-Modesto, J. Hospital microbiota vectored by ants. Microbes Infect. Chemother. 2021, 1, e1199. [Google Scholar] [CrossRef]
- Juyal, D.; Adekhandi, S.; Sharma, M.; Prakash, R.; Sharma, N.; Rana, A.; Parihar, A.; Pal, S. Mobile phones: Reservoirs for the transmission of nosocomial pathogens. Adv. Biomed. Res. 2015, 4, 144. [Google Scholar] [CrossRef] [PubMed]
- Acevedo-Osorio, G.O.; Gómez-Fernández, A.M.; Oyola-Leiva, N.; Arboleda-Angulo, L.D.; Orozco-Cardona, L.M. Evaluación microbiológica de dispositivos móviles en personal quirúrgico de una institución de salud, Pereira, Colombia, 2018. Univ. Y Salud 2020, 22, 77–83. [Google Scholar] [CrossRef]
- Yao, N.; Yang, X.-F.; Zhu, B.; Liao, C.-Y.; He, Y.-M.; Du, J.; Liu, N.; Zhou, C.-B. Bacterial Colonization on Healthcare Workers’ Mobile Phones and Hands in Municipal Hospitals of Chongqing, China: Cross-contamination and Associated Factors. J. Epidemiol. Glob. Health 2022, 12, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Loyola, S.; Gutierrez, L.; Avendaño, E.; Severino, N.; Tamariz, J. Multidrug-resistant bacteria isolated from cell phones in five intensive care units: Exploratory dispersion analysis. Germs 2018, 8, 85–91. [Google Scholar] [CrossRef]
- Amar, A.K.; Ramakrishnan, K.; Sawant, A.R.; Karamveer, K.; Menon, J.; Tiwary, B.K.; Prashanth, K. Investigations on microbiome of the used clinical device revealed many uncultivable newer bacterial species associated with persistent chronic infections. Microbes Infect. Chemother. 2022, 2, e1542. [Google Scholar] [CrossRef]
- Boucherabine, S.; Nassar, R.; Zaher, S.; Mohamed, L.; Olsen, M.; Alqutami, F.; Hachim, M.; Alkhaja, A.; Campos, M.; Jones, P.; et al. Metagenomic Sequencing and Reverse Transcriptase PCR Reveal That Mobile Phones and Environmental Surfaces Are Reservoirs of Multidrug-Resistant Superbugs and SARS-CoV-2. Front. Cell Infect. Microbiol. 2022, 12, 806077. [Google Scholar] [CrossRef] [PubMed]
- Kotris, I.; Drenjančević, D.; Talapko, J.; Bukovski, S. Identification of microorganisms on mobile phones of intensive care unit health care workers and medical students in the tertiary hospital. Med. Glas. 2017, 14, 85–90. [Google Scholar] [CrossRef]
- De Groote, P.; Blot, K.; Conoscenti, E.; Labeau, S.; Blot, S. Mobile phones as a vector for Healthcare-Associated Infection: A systematic review. Intensiv. Crit. Care Nurs. 2022, 72, 103266. [Google Scholar] [CrossRef]
- Kopp, J.; Cawcutt, K.A.; Musil, L.; Huang, X.; Minard, C.G.; Hetland, B. Efficacy of a bioburden reduction intervention on mobile phones of critical care nurses. Am. J. Infect. Control 2023, 51, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Raraz-Vidal, J.; Raraz-Vidal, O. Pie diabético y resistencia a los antibióticos durante la pandemia de COVID-19. Rev. Peru. Investig. Salud 2022, 6, 77–78. [Google Scholar] [CrossRef]
- Akinyemi, K.O.; Atapu, A.D.; Adetona, O.O.; Coker, A.O. The potential role of mobile phones in the spread of bacterial infections. J. Infect. Dev. Ctries. 2009, 3, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Peros, C.S.; Dasgupta, R.; Kumar, P.; Johnson, B.A. Bushmeat, wet markets, and the risks of pandemics: Exploring the nexus through systematic review of scientific disclosures. Environ. Sci. Policy 2021, 124, 169–178. [Google Scholar] [CrossRef]
- Barnett, T.; Fournié, G. Zoonoses and wet markets: Beyond technical interventions. Lancet Planet. Health 2021, 5, e2–e3. [Google Scholar] [CrossRef]
- Ruiz-Roldán, L.; Martinez-Puchol, S.; Gomes, C.; Palma, N.; Riveros, M.; Ocampo, K.; Durand, D.; Ochoa, T.J.; Ruiz, J.; Pons, M.J. Presencia de Enterobacteriaceae y Escherichia coli multirresistente a antimicrobianos en carne adquirida en mercados tradicionales en Lima. Rev. Peru. Med. Exp. Salud Publica 2018, 35, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Cortez-Sandoval, V.; González, R.; Ramos, D. Detección de enterobacterias productoras de β-lactamasas de espectro extendido (BLEE) aisladas en carne de pollo de mercados de abasto de un distrito de Lima, Peru. Rev. Investig. Vet. Peru 2022, 33, e22899. [Google Scholar] [CrossRef]
- Stoneham, G.; Hester, S.M.; Li, J.S.; Zhou, R.; Chaudhry, A. The Boundary of the Market for Biosecurity Risk. Risk Anal. 2021, 41, 1447–1462. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M.; Campos, M.; Lohning, A.; Jones, P.; Legget, J.; Bannach-Brown, A.; McKirdy, S.; Alghafri, R.; Tajouri, L. Mobile phones represent a pathway for microbial transmission: A scoping review. Travel Med. Infect. Dis. 2020, 35, 101704. [Google Scholar] [CrossRef]
- Bayraktar, M.; Kaya, E.; Ozturk, A.; Ibahim, B.M.S. Antimicrobial susceptibility of bacterial pathogens isolated from healthcare workers’ cellphones. Infect. Dis. Now 2021, 51, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Bodena, D.; Teklemariam, Z.; Balakrishnan, S.; Tesfa, T. Bacterial contamination of mobile phones of health professionals in Eastern Ethiopia: Antimicrobial susceptibility and associated factors. Trop. Med. Health 2019, 47, 15. [Google Scholar] [CrossRef]
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol. Am. Soc. Microbiol. 2009, 15, 55–63. [Google Scholar]
- Pérez-Cano, H.; Santos, M.R.; Moreno, B.C. Microbiota en teléfonos móviles de médicos oftalmólogos. Arch. Soc. Esp. Oftalmol. 2019, 94, 55–59. [Google Scholar] [CrossRef]
- Meadow, J.F.; Altrichter, A.E.; Green, J.L. Mobile phones carry the personal microbiome of their owners. PeerJ 2014, 2, e447. [Google Scholar] [CrossRef]
- Arteaga-Livias, K.; Pinzas-Acosta, K.; Perez-Abad, L.; Panduro-Correa, V.; Rabaan, A.A.; Pecho-Silva, S.; Dámaso-Mata, B. A multidrug-resistant Klebsiella pneumoniae outbreak in a Peruvian hospital: Another threat from the COVID-19 pandemic. Infect. Control Hosp. Epidemiol. 2021, 43, 267–268. [Google Scholar] [CrossRef]
- Metan, G.; Demir Çuha, M.; Hazirolan, G.; Telli Dizman, G.; Tanriverdi, E.S.; Otlu, B.; Tas, Z.; Zarakolu, P.; Arik, Z.; Topeli, A.; et al. The Impact of COVID-19 Pandemic on Nosocomial Multidrug-Resistant Bacterial Bloodstream Infections and Antibiotic Consumption in a Tertiary Care Hospital. GMS Hyg. Infect. Control 2022, 17, Doc15. [Google Scholar] [CrossRef] [PubMed]
- Nakitanda, A.O.; Karlsson, P.; Löfling, L.; Cesta, C.E.; Odsbu, I. Antimicrobial use in Sweden during the COVID-19 pandemic: Prescription fill and inpatient care requisition patterns. BMC Infect. Dis. 2022, 22, 492. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, A.; Fujii, H.; Hotta, A.; Asanuma, R.; Irie, H. Prevalence of bacterial contamination of touchscreens and posterior surfaces of smartphones owned by healthcare workers: A cross-sectional study. BMC Infect. Dis. 2021, 21, 681. [Google Scholar] [CrossRef]
- Dhama, K.; Khan, S.; Tiwari, R.; Sircar, S.; Bhat, S.; Malik, Y.S.; Singh, K.P.; Chaicumpa, W.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. Coronavirus Disease 2019–COVID-19. Clin. Microbiol. Rev. 2020, 33, e00028-20. [Google Scholar] [CrossRef]
- Salmanov, A.G.; Ushkalov, V.O.; Shunko, Y.Y.; Piven, N.; Vygovska, L.M.; Verner, O.M.; Kushnirenko, S. One Health: Antibiotic-Resistant bacteria Contamination in Fresh Vegetables Sold at a Retail Markets in Kyiv, Ukraine. Wiadomości Lek. 2021, 74, 83–89. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Khong, D.T.; Le, H.V.; Tran, H.T.; Phan, Q.N.; Le, H.T.T.; Kawahara, R.; Yamamoto, Y. Quantitative Analysis of Colistin-Resistant Escherichia coli in Retail Meat from Local Vietnamese Markets. BioMed Res. Int. 2021, 2021, 6678901. [Google Scholar] [CrossRef]
- Adebowale, O.; Makanjuola, M.; Bankole, N.; Olasoju, M.; Alamu, A.; Kperegbeyi, E.; Oladejo, O.; Fasanmi, O.; Adeyemo, O.; Fasina, F.O. Multi-Drug Resistant Escherichia coli, Biosecurity and Anti-Microbial Use in Live Bird Markets, Abeokuta, Nigeria. Antibiotics 2022, 11, 253. [Google Scholar] [CrossRef] [PubMed]
- Gashaw, M.; Abtew, D.; Addis, Z. Prevalence and Antimicrobial Susceptibility Pattern of Bacteria Isolated from Mobile Phones of Health Care Professionals Working in Gondar Town Health Centers. ISRN Public Health 2014, 2014, e205074. [Google Scholar] [CrossRef]
- Chowdhury, M.M.H.; Debnath, T.; Bhowmik, S.; Islam, T. Presence of multidrug-resistant bacteria on mobile phones of healthcare workers accelerates the spread of nosocomial infection and Regarded as a Threat to Public Health in Bangladesh. J. Microsc. Ultrastruct. 2018, 6, 165–169. [Google Scholar] [CrossRef]
- Smibert, O.; Aung, A.; Woolnough, E.; Carter, G.; Schultz, M.; Howden, B.; Seemann, T.; Spelman, D.; McGloughlin, S.; Peleg, A. Mobile phones and computer keyboards: Unlikely reservoirs of multidrug-resistant organisms in the tertiary intensive care unit. J. Hosp. Infect. 2018, 99, 295–298. [Google Scholar] [CrossRef]
- Martina, P.; Martinez, M.; Centeno, C.; Von Specht, M.; Ferreras, J. Dangerous passengers: Multidrug-resistant bacteria on hands and mobile phones. J. Prev. Med. Hyg. 2019, 60, E293–E299. [Google Scholar] [CrossRef]
- Modica, D.C.; Maurici, M.; D’Alò, G.L.; Mozzetti, C.; Messina, A.; Distefano, A.; Pica, F.; De Filippis, P. Taking Screenshots of the Invisible: A Study on Bacterial Contamination of Mobile Phones from University Students of Healthcare Professions in Rome, Italy. Microorganisms 2020, 8, E1075. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Carhuamaca, A.; Castañeda-Japan, J.; Dámaso-Mata, B.; Panduro-Correa, V.; Arteaga-Livias, K. Bacterial resistance in cell phone cultures of medical students. Infez Med. 2019, 27, 374–379. [Google Scholar] [PubMed]
- Albastaki, A.; Olsen, M.; Almulla, H.; Nassar, R.; Boucherabine, S.; Mohamed, L.; Almheiri, R.; Kachigunda, B.; McKirdy, S.; Alghafri, R.; et al. Mobile phones as fomites for pathogenic microbes: A cross-sectional survey of perceptions and sanitization habits of health care workers in Dubai, United Arab Emirates. Infect. Dis. Health 2022, 28, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Rosa, D.A.C.; de Sousa, S.S.; da Silva, M.N.R.; Gamboge, L.R.R.; Deusdará, R.; Lapa, J. Knowledge about COVID-19 and Associated Factors Early in the Outbreak among the Brazilian Population. Int. J. Environ. Res. Public Health 2022, 19, 13824. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.; Akl, E.A.; Duda, S.; Solo, K.; Yaacoub, S.; Schünemann, H.J.; Chu, D.K.; Akl, E.A.; El-harakeh, A.; Bognanni, A.; et al. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: A systematic review and meta-analysis. Lancet 2020, 395, 1973–1987. [Google Scholar] [CrossRef]
- Villena-Prado, J.J.; Villena-Prado, J.J. Medidas preventivas contra el SARS-CoV-2 en la comunidad: ¿Qué dice la evidencia? Rev. Fac. Med. Hum. 2021, 21, 237–239. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Chen, C.-J.; Wu, H.-Y.; Chen, I.; Chang, Y.-H.; Yang, P.-H.; Wang, T.-Y.; Chen, L.-C.; Liu, K.-T.; Yeh, I.-J.; et al. Plastic wrap combined with alcohol wiping is an effective method of preventing bacterial colonization on mobile phones. Medicine 2020, 99, e22910. [Google Scholar] [CrossRef]
- Bhoonderowa, A.; Gookool, S.; Biranjia-Hurdoyal, S.D. The Importance of Mobile Phones in the Possible Transmission of Bacterial Infections in the Community. J. Community Health 2014, 39, 965–967. [Google Scholar] [CrossRef]
- Pal, P.; Roy, A.; Moore, G.; Muzslay, M.; Lee, E.; Alder, S.; Wilson, P.; Powles, T.; Kelly, J. Keypad mobile phones are associated with a significant increased risk of microbial contamination compared to touch screen phones. J. Infect. Prev. 2013, 14, 65–68. [Google Scholar] [CrossRef]
- Shakir, I.A.; Patel, N.H.; Chamberland, R.R.; Kaar, S.G. Investigation of Cell Phones as a Potential Source of Bacterial Contamination in the Operating Room. J. Bone Jt. Surg. 2015, 97, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Dorost, A.; Safari, Y.; Akhlaghi, M.; Soleimani, M.; Yoosefpour, N. Microbial contamination data of keypad and touch screen of cell phones among hospital and non-hospital staffs—A case study: Iran. Data Brief 2018, 20, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Yoo, C.; Lee, C.; Chung, H.S.; Kim, Y.W.; Han, S.K.; Yim, J. Contamination rates between smart cell phones and non-smart cell phones of healthcare workers. J. Hosp. Med. 2013, 8, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Al Momani, W.; Khatatbeh, M.; Altaany, Z. Antibiotic susceptibility of bacterial pathogens recovered from the hand and mobile phones of university students. Germs 2019, 9, 9–16. [Google Scholar] [CrossRef]
| Variable | Frequency | Percentage |
|---|---|---|
| Sex | ||
| Female | 94 | 74.1 |
| Male | 33 | 25.9 |
| Age | ||
| (X + SD) | 43.4 + 12.9 | |
| Sector | ||
| Foods | 45 | 35.4 |
| Fruits and vegetables | 13 | 10.2 |
| Grocery | 11 | 8.6 |
| Meats | 58 | 45.7 |
| Clients in 1 h | ||
| (Median: IQR) | 5: (4–10) | |
| Client permanence (minutes) | ||
| (Median: IQR) | 5: (4–10) | |
| Client distance (meters) | ||
| (X + SD) | 1.3 + 0.3 | |
| Knowledge of bacteria on cell phones | ||
| Yes | 75 | 59.1 |
| No | 52 | 40.9 |
| Share cell phone with others | ||
| No | 115 | 90.6 |
| Yes | 12 | 9.4 |
| Use of cell phone in the bathroom | ||
| No | 94 | 74.1 |
| Yes | 33 | 25.9 |
| Cell phone disinfection | ||
| No | 14 | 11.1 |
| Yes | 113 | 88.9 |
| Use of screen protector | ||
| No | 32 | 25.2 |
| Yes | 95 | 74.8 |
| Screen protector type | ||
| Plastic | 13 | 13.7 |
| Fiberglass | 82 | 86.3 |
| Use of equipment protector | ||
| No | 49 | 38.6 |
| Yes | 78 | 61.4 |
| Equipment protector type | ||
| Plastic | 68 | 87.2 |
| Others | 10 | 12.8 |
| Cell phone type | ||
| Touchscreen | 113 | 88.9 |
| Keys | 14 | 11.1 |
| Cracked screen | ||
| No | 76 | 59.8 |
| Yes | 51 | 40.2 |
| Characteristics | Antibiotic Resistance | p # | |||
|---|---|---|---|---|---|
| Negative | % | Positive | % | ||
| Sex | |||||
| Female | 30 | 34.9 | 56 | 65.1 | 0.792 |
| Male | 10 | 32.3 | 21 | 67.7 | |
| Age | |||||
| (X + SD) | 43.8 + 12.7 | 42.7 + 13.1 | 0.709 * | ||
| Sector | 0.148 | ||||
| Foods | 17 | 41.5 | 24 | 58.5 | |
| Fruits and vegetables | 5 | 35.5 | 8 | 61.5 | |
| Grocery | 13 | 24.1 | 41 | 75.9 | |
| Meats | 5 | 55.6 | 4 | 44.4 | |
| Clients in 1 h | 0.921 * | ||||
| (Median: IQR) | 5: (4–10) | 6: (4–10) | |||
| Client permanence (minutes) | 0.998 * | ||||
| (Median: IQR) | 5: (4–10) | 5: (4–10) | |||
| Client distance (meters) | 0.023 * | ||||
| (X + SD) | 1.45 + 0.27 | 1.29 + 0.39 | |||
| Knowledge of bacteria in cell phones | 0.666 | ||||
| Yes | 16 | 32 | 34 | 68 | |
| No | 24 | 35.8 | 43 | 64.2 | |
| Share cell phones with others | 0.909 | ||||
| No | 30 | 34.5 | 57 | 65.5 | |
| Yes | 10 | 33.3 | 20 | 66.7 | |
| Use of cell phone in the bathroom | 0.503 | ||||
| No | 16 | 20 | 64 | 80 | |
| Yes | 4 | 14.3 | 24 | 85.7 | |
| Cell phone disinfection | 0.13 | ||||
| No | 2 | 15.4 | 11 | 84.6 | |
| Yes | 38 | 36.5 | 66 | 63.5 | |
| Use of screen protector | 0.133 | ||||
| No | 14 | 45.2 | 17 | 54.8 | |
| Yes | 26 | 30.2 | 60 | 69.8 | |
| Screen protector type | 0.351 | ||||
| Plastic | 2 | 18.2 | 9 | 81.8 | |
| Fiberglass | 24 | 32 | 51 | 68 | |
| Use of equipment protector | 0.012 | ||||
| No | 22 | 47.8 | 24 | 52.2 | |
| Yes | 18 | 25.4 | 53 | 74.6 | |
| Equipment protector type | 0.026 | ||||
| Plastic | 13 | 20.9 | 49 | 79.1 | |
| Others | 5 | 55.6 | 4 | 44.4 | |
| Cell phone type | 0.005 | ||||
| Touchscreen | 31 | 29.8 | 73 | 70.2 | |
| Keys | 9 | 69.2 | 4 | 30.7 | |
| Cracked screen | 0.223 | ||||
| No | 26 | 38.8 | 41 | 61.2 | |
| Yes | 14 | 28 | 36 | 72 | |
| Bacteria\Antibiotics | CLI | VAN | AZM | ERI | FOS | FOX | IMP | MEM | SAM | CFZ | LVX | COL | CTX | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | R | I | S | |
| S. aureus | 4 | 3 | 11 | 5 | 2 | 11 | 2 | 10 | 6 | 4 | 12 | 2 | 5 | 2 | 11 | 2 | 10 | 6 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| (total = 18) | |||||||||||||||||||||||||||||||||||||||
| Coagulase-negative staphylococci | 17 | 21 | 23 | 12 | 9 | 40 | 15 | 23 | 23 | 10 | 37 | 14 | 23 | 16 | 22 | 13 | 44 | 4 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| (total = 61) | |||||||||||||||||||||||||||||||||||||||
| E. coli | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 0 | 3 | 11 | 3 | 5 | 6 | 2 | 7 | 5 | 4 | 10 | 0 | 4 | 5 | 5 | 4 | 1 | 9 | 4 | 9 | 1 |
| (total = 14) | |||||||||||||||||||||||||||||||||||||||
| E. faecalis | NR | NR | NR | 4 | 9 | 4 | 3 | 7 | 7 | 3 | 13 | 1 | NR | NR | NR | 2 | 8 | 7 | NR | NR | NR | NR | NR | NR | 1 | 2 | 14 | NR | NR | NR | 2 | 6 | 9 | NR | NR | NR | NR | NR | NR |
| (total = 17) | |||||||||||||||||||||||||||||||||||||||
| E. aerogenes | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 0 | 0 | 4 | 0 | 0 | 4 | 1 | 2 | 1 | 2 | 2 | 0 | 0 | 0 | 4 | 0 | 2 | 2 | 0 | 4 | 0 |
| (total = 4) | |||||||||||||||||||||||||||||||||||||||
| S. marcescens | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 0 | 0 | 2 | 0 | 0 | 2 | 0 | 0 | 2 | 0 | 2 | 0 | 0 | 0 | 2 | 0 | 0 | 2 | 0 | 2 | 0 |
| (total = 2) | |||||||||||||||||||||||||||||||||||||||
| E. cloacae | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 |
| (total = 1) | |||||||||||||||||||||||||||||||||||||||
| Bacteria | Frequency of Antibiotic Resistance | Total | |||||
|---|---|---|---|---|---|---|---|
| Sensitive | Resistant to 1 ATB | Resistant to 2 ATB | Resistant to 3 ATB | Resistant to 4 ATB | Resistant to 5 ATB | ||
| Staphylococcus aureus | 7 (38.9) | 4 (22.2) | 4 (22.2) | 2 (11.1) | 1 (5.6) | 18 | |
| Coagulase-negative staphylococci | 20 (32.8) | 19 (31.2) | 9 (14.8) | 5 (8.2) | 3 (4.9) | 5 (8.2) | 61 |
| Enterococcus faecalis | 5 (29.4) | 9 (52.9) | 3 (17.7) | 17 | |||
| Escherichia coli | 5 (35.7) | 4 (28.6) | 1 (7.1) | 1 (7.1) | 3 (21.4) | 14 | |
| Enterobacter aerogenes | 2 (50) | 1 (25) | 1 (25) | 4 | |||
| Enterobacter cloacae | 1 (100) | 1 | |||||
| Serratia marcescens | 1 (50) | 1 (50) | 2 | ||||
| 40 | 39 | 18 | 8 | 7 | 5 | 117 | |
| ATB = Antibiotics | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Gonzales, W.; Alvarado-Garcia, A.; Suárez-Mamani, M.; Dámaso-Mata, B.; Panduro-Correa, V.; Maguiña, J.L.; Pecho-Silva, S.; Rabaan, A.A.; Rodriguez-Morales, A.J.; Arteaga-Livias, K. Contamination by Antibiotic-Resistant Bacteria on Cell Phones of Vendors in a Peruvian Market. Medicina 2023, 59, 669. https://doi.org/10.3390/medicina59040669
Gómez-Gonzales W, Alvarado-Garcia A, Suárez-Mamani M, Dámaso-Mata B, Panduro-Correa V, Maguiña JL, Pecho-Silva S, Rabaan AA, Rodriguez-Morales AJ, Arteaga-Livias K. Contamination by Antibiotic-Resistant Bacteria on Cell Phones of Vendors in a Peruvian Market. Medicina. 2023; 59(4):669. https://doi.org/10.3390/medicina59040669
Chicago/Turabian StyleGómez-Gonzales, Walter, Anthony Alvarado-Garcia, Marytté Suárez-Mamani, Bernardo Dámaso-Mata, Vicky Panduro-Correa, Jorge L. Maguiña, Samuel Pecho-Silva, Ali A. Rabaan, Alfonso J. Rodriguez-Morales, and Kovy Arteaga-Livias. 2023. "Contamination by Antibiotic-Resistant Bacteria on Cell Phones of Vendors in a Peruvian Market" Medicina 59, no. 4: 669. https://doi.org/10.3390/medicina59040669
APA StyleGómez-Gonzales, W., Alvarado-Garcia, A., Suárez-Mamani, M., Dámaso-Mata, B., Panduro-Correa, V., Maguiña, J. L., Pecho-Silva, S., Rabaan, A. A., Rodriguez-Morales, A. J., & Arteaga-Livias, K. (2023). Contamination by Antibiotic-Resistant Bacteria on Cell Phones of Vendors in a Peruvian Market. Medicina, 59(4), 669. https://doi.org/10.3390/medicina59040669









