From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Sources and Search Strategy
2.3. Search Terms
2.4. Inclusion and Exclusion Criteria
2.5. Study Selection and Data Extraction
3. Understanding the Role of Urologists in Male Infertility Management amid the Advancements in Assisted Reproductive Technology
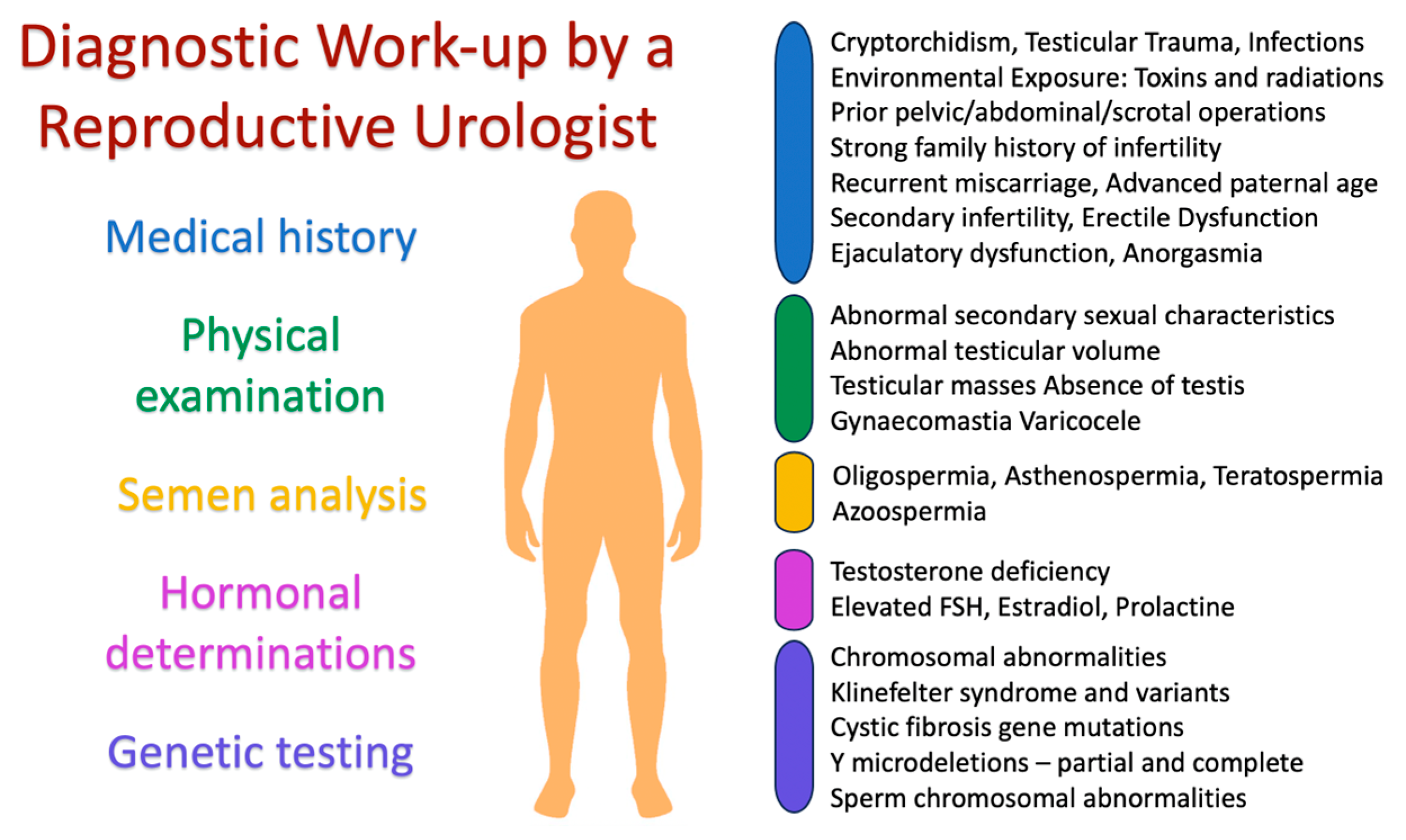
4. The Urologist’s Role in Evaluation and Diagnosis
4.1. Evaluation and Diagnosis of Male Infertility
4.2. Identification of Patients Who Would Not Require Anything Other than ART
4.2.1. Oligoasthenoteratozoospermia
4.2.2. Isolated Teratozoospermia
4.2.3. Absolute Asthenozoospermia
4.2.4. Antisperm Antibodies
4.2.5. Globozoospermia
4.2.6. Female Aging
4.3. Identifications of Situations Where Donor Insemination or Adoption Is the Only Solution
4.3.1. Y Microdeletions
4.3.2. XX Male Syndrome
4.3.3. Testis-Expressed 11 Gene
5. The Pivotal Role of the Reproductive Urologist in Addressing Male Infertility and Enhancing ART Outcomes
5.1. Implementing Surgical Interventions in the Management of Male Infertility
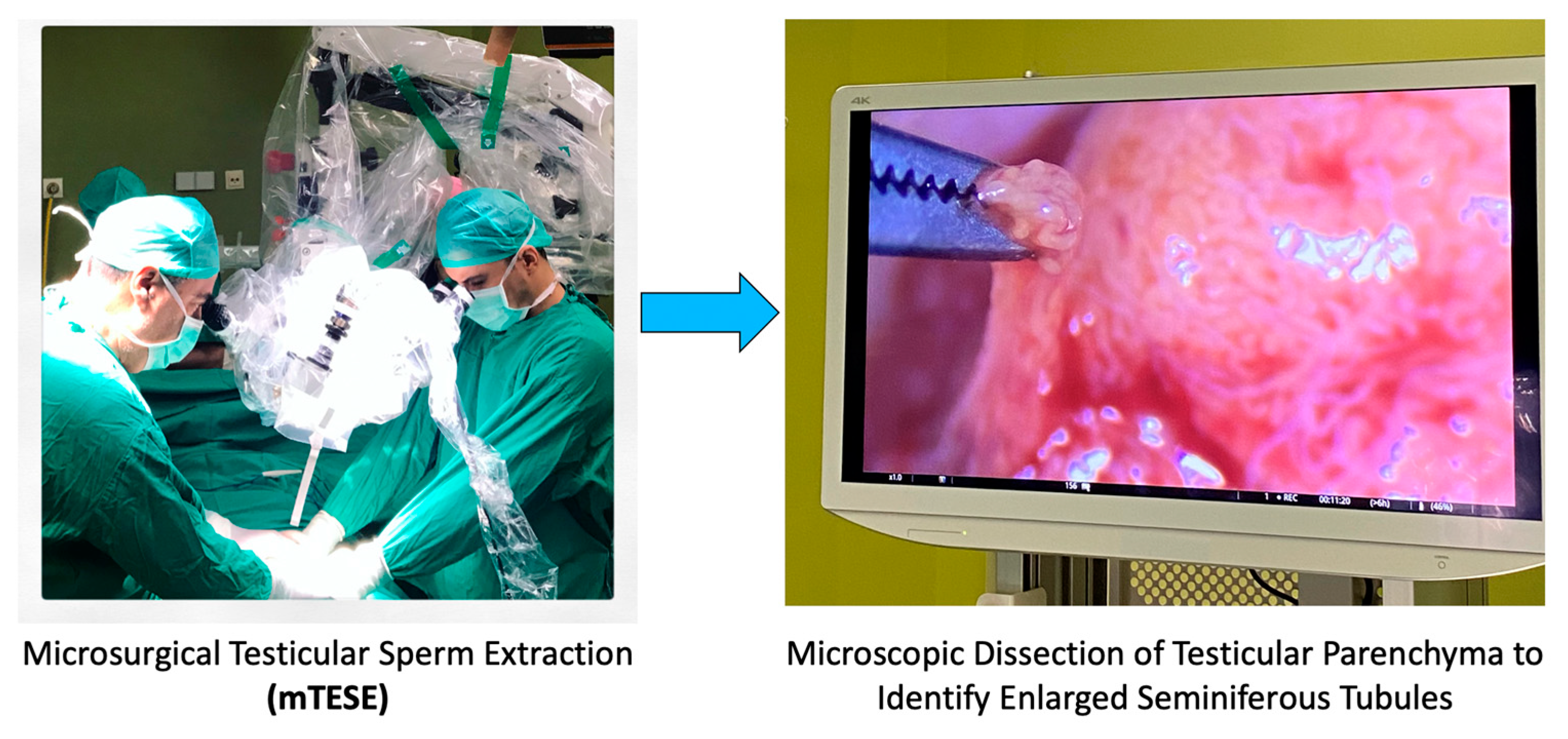
5.1.1. Addressing Abnormal Sperm Production: A Focus on Non-Obstructive Azoospermia
5.1.2. The Interplay of Varicocele and Assisted Reproductive Technologies
5.1.3. Addressing Post-Testicular Obstructions: Surgical and ART Approaches
5.2. Pharmacological Approaches in the Management of Male Infertility
5.2.1. Genital Tract Infections
5.2.2. Hypogonadotropic Hypogonadism
5.2.3. Endocrine Disorders
5.2.4. Addressing Erectile Dysfunction and Premature Ejaculation for Fertility Enhancement
5.3. Role of Urologists in Managing the Impact of Lifestyle Factors on Male Fertility and Overall Health
5.4. The Reproductive Urologist’s Role in Special Circumstances
5.4.1. Addressing Idiopathic Recurrent Pregnancy Loss
5.4.2. Overcoming Recurrent Failures in Assisted Reproductive Technologies
5.4.3. Implications of DNA Fragmentation on ART Outcomes: Strategies for Sperm Isolation and Recovery
6. Uncovering Conditions That Might Affect the Offspring’s Health
6.1. Advanced Paternal Age
6.2. Effects of Klinefelter Syndrome on the Health of the Offspring
6.3. Consequences of Y-Chromosome Microdeletions
6.4. Impact of ICSI on the Offspring’s Health
7. Barriers to Urologist Involvement in the Era of Assisted Reproductive Technology
- Limited availability and distribution: One of the primary challenges is the limited number and distribution of specialized male reproductive urologists. Certain regions, especially rural or underserved areas, lack access to these experts, making it challenging for patients to seek specialized care [16].
- Lack of awareness and education: There is a significant knowledge gap among healthcare professionals and the general public regarding the role of urologists in male infertility. This lack of awareness leads to under-referral and underutilization of urologist services, with many cases of male infertility going undiagnosed or mismanaged [16].
- Financial constraints: The high costs associated with infertility treatments coupled with the absence of comprehensive health insurance coverage for such services in many regions deter many from seeking specialized care. This financial barrier is further exacerbated by the often-prohibitive costs of ART procedures [221].
- Misconceptions about infertility: A prevailing misconception exists that infertility is primarily a female-centric issue. This skewed perspective often results in a disproportionate focus on female infertility, sidelining male factors that, in reality, contribute to almost half of all infertility cases [16].
- Lifestyle and environmental factors: The modern era has seen a decline in male fertility attributed to various lifestyle and environmental factors. However, the role of urologists in addressing these factors and guiding patients toward healthier lifestyles remains underemphasized [12].
- Rapid advancements in ART: The swift advancements in ART techniques have sometimes overshadowed traditional diagnostic and therapeutic approaches. As a result, the emphasis on understanding and treating the root causes of male infertility, where urologists play a pivotal role, has been diminished [222].
8. The Future of Urologists: What Place Do They Have in the Era of ART?
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agarwal, A.; Mulgund, A.; Hamada, A.; Chyatte, M.R. A unique view on male infertility around the globe. Reprod. Biol. Endocrinol. 2015, 13, 37. [Google Scholar] [CrossRef]
- Olisa, N.P.; Campo-Engelstein, L.; Martins da Silva, S. Male infertility: What on earth is going on? Pilot international questionnaire study regarding clinical evaluation and fertility treatment for men. Reprod. Fertil. 2022, 3, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Palermo, G.D.; O’Neill, C.L.; Chow, S.; Cheung, S.; Parrella, A.; Pereira, N.; Rosenwaks, Z. Intracytoplasmic sperm injection: State of the art in humans. Reproduction 2017, 154, F93–F110. [Google Scholar] [CrossRef]
- Marinaro, J.; Goldstein, M. Microsurgical Management of Male Infertility: Compelling Evidence That Collaboration with Qualified Male Reproductive Urologists Enhances Assisted Reproductive Technology (ART) Outcomes. J. Clin. Med. 2022, 11, 4593. [Google Scholar] [CrossRef]
- Sunderam, S.; Kissin, D.M.; Zhang, Y.; Jewett, A.; Boulet, S.L.; Warner, L.; Kroelinger, C.D.; Barfield, W.D. Assisted Reproductive Technology Surveillance—United States, 2017. MMWR Surveill. Summ. 2020, 69, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Singh, A.K. Trends of male factor infertility, an important cause of infertility: A review of literature. J. Hum. Reprod. Sci. 2015, 8, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Auger, J.; Kunstmann, J.M.; Czyglik, F.; Jouannet, P. Decline in semen quality among fertile men in Paris during the past 20 years. N. Engl. J. Med. 1995, 332, 281–285. [Google Scholar] [CrossRef]
- Sripada, S.; Fonseca, S.; Lee, A.; Harrild, K.; Giannaris, D.; Mathers, E.; Bhattacharya, S. Trends in semen parameters in the northeast of Scotland. J. Androl. 2007, 28, 313–319. [Google Scholar] [CrossRef]
- Levitas, E.; Lunenfeld, E.; Weisz, N.; Friger, M.; Potashnik, G. Relationship between age and semen parameters in men with normal sperm concentration: Analysis of 6022 semen samples. Andrologia 2007, 39, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Feki, N.C.; Abid, N.; Rebai, A.; Sellami, A.; Ayed, B.B.; Guermazi, M.; Bahloul, A.; Rebai, T.; Ammar, L.K. Semen quality decline among men in infertile relationships: Experience over 12 years in the South of Tunisia. J. Androl. 2009, 30, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Rodprasert, W.; Virtanen, H.E.; Sadov, S.; Perheentupa, A.; Skakkebaek, N.E.; Jorgensen, N.; Toppari, J. An update on semen quality among young Finnish men and comparison with Danish data. Andrology 2019, 7, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Durairajanayagam, D. Lifestyle causes of male infertility. Arab J. Urol. 2018, 16, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Balawender, K.; Orkisz, S. The impact of selected modifiable lifestyle factors on male fertility in the modern world. Cent. Eur. J. Urol. 2020, 73, 563–568. [Google Scholar] [CrossRef]
- Minhas, S.; Bettocchi, C.; Boeri, L.; Capogrosso, P.; Carvalho, J.; Cilesiz, N.C.; Cocci, A.; Corona, G.; Dimitropoulos, K.; Gul, M.; et al. European Association of Urology Guidelines on Male Sexual and Reproductive Health: 2021 Update on Male Infertility. Eur. Urol. 2021, 80, 603–620. [Google Scholar] [CrossRef]
- Ilacqua, A.; Izzo, G.; Emerenziani, G.P.; Baldari, C.; Aversa, A. Lifestyle and fertility: The influence of stress and quality of life on male fertility. Reprod. Biol. Endocrinol. 2018, 16, 115. [Google Scholar] [CrossRef]
- Mehta, A.; Nangia, A.K.; Dupree, J.M.; Smith, J.F. Limitations and barriers in access to care for male factor infertility. Fertil. Steril. 2016, 105, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, P.N.; Sigman, M.; Collura, B.; De Jonge, C.J.; Eisenberg, M.L.; Lamb, D.J.; Mulhall, J.P.; Niederberger, C.; Sandlow, J.I.; Sokol, R.Z.; et al. Diagnosis and Treatment of Infertility in Men: AUA/ASRM Guideline Part I. J. Urol. 2021, 205, 36–43. [Google Scholar] [CrossRef]
- Abdel Raheem, A.; Garaffa, G.; Rushwan, N.; De Luca, F.; Zacharakis, E.; Abdel Raheem, T.; Freeman, A.; Serhal, P.; Harper, J.C.; Ralph, D. Testicular histopathology as a predictor of a positive sperm retrieval in men with non-obstructive azoospermia. BJU Int. 2013, 111, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Caroppo, E.; Colpi, E.M.; Gazzano, G.; Vaccalluzzo, L.; Scroppo, F.I.; D’Amato, G.; Colpi, G.M. Testicular histology may predict the successful sperm retrieval in patients with non-obstructive azoospermia undergoing conventional TESE: A diagnostic accuracy study. J. Assist. Reprod. Genet. 2017, 34, 149–154. [Google Scholar] [CrossRef]
- Cetinkaya, M.; Onem, K.; Zorba, O.U.; Ozkara, H.; Alici, B. Evaluation of Microdissection Testicular Sperm Extraction Results in Patients with Non-Obstructive Azoospermia: Independent Predictive Factors and Best Cutoff Values for Sperm Retrieval. Urol. J. 2015, 12, 2436–2443. [Google Scholar]
- Kaltsas, A.; Markou, E.; Zachariou, A.; Dimitriadis, F.; Symeonidis, E.N.; Zikopoulos, A.; Mamoulakis, C.; Tien, D.M.B.; Takenaka, A.; Sofikitis, N. Evaluating the Predictive Value of Diagnostic Testicular Biopsy for Sperm Retrieval Outcomes in Men with Non-Obstructive Azoospermia. J. Pers. Med. 2023, 13, 1362. [Google Scholar] [CrossRef]
- Kalsi, J.; Thum, M.Y.; Muneer, A.; Abdullah, H.; Minhas, S. In the era of micro-dissection sperm retrieval (m-TESE) is an isolated testicular biopsy necessary in the management of men with non-obstructive azoospermia? BJU Int. 2012, 109, 418–424. [Google Scholar] [CrossRef]
- Kalsi, J.S.; Shah, P.; Thum, Y.; Muneer, A.; Ralph, D.J.; Minhas, S. Salvage micro-dissection testicular sperm extraction; outcome in men with non-obstructive azoospermia with previous failed sperm retrievals. BJU Int. 2015, 116, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Carrell, D.T. The clinical implementation of sperm chromosome aneuploidy testing: Pitfalls and promises. J. Androl. 2008, 29, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Aran, B.; Blanco, J.; Vidal, F.; Vendrell, J.M.; Egozcue, S.; Barri, P.N.; Egozcue, J.; Veiga, A. Screening for abnormalities of chromosomes X, Y, and 18 and for diploidy in spermatozoa from infertile men participating in an in vitro fertilization-intracytoplasmic sperm injection program. Fertil. Steril. 1999, 72, 696–701. [Google Scholar] [CrossRef]
- Kohn, T.P.; Kohn, J.R.; Darilek, S.; Ramasamy, R.; Lipshultz, L. Genetic counseling for men with recurrent pregnancy loss or recurrent implantation failure due to abnormal sperm chromosomal aneuploidy. J. Assist. Reprod. Genet. 2016, 33, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Zhang, Y.; Deng, H.; Feng, Y.; Chong, W.; Hai, Y.; Hao, P.; He, J.; Li, T.; Peng, L.; et al. Preimplantation Genetic Testing for Aneuploidy with Comprehensive Chromosome Screening in Patients Undergoing In Vitro Fertilization: A Systematic Review and Meta-analysis. Obs. Gynecol. 2022, 140, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Yang, C.; Yang, S.; Sun, S.; Mu, M.; Rao, M.; Zu, R.; Yan, J.; Ren, B.; Yang, R.; et al. Obstetric and neonatal outcomes of pregnancies resulting from preimplantation genetic testing: A systematic review and meta-analysis. Hum. Reprod. Update 2021, 27, 989–1012. [Google Scholar] [CrossRef]
- Dviri, M.; Madjunkova, S.; Koziarz, A.; Madjunkov, M.; Mashiach, J.; Nekolaichuk, E.; Trivodaliev, K.; Al-Asmar, N.; Moskovtsev, S.I.; Librach, C. Is there an association between paternal age and aneuploidy? Evidence from young donor oocyte-derived embryos: A systematic review and individual patient data meta-analysis. Hum. Reprod. Update 2021, 27, 486–500. [Google Scholar] [CrossRef]
- Cornelisse, S.; Zagers, M.; Kostova, E.; Fleischer, K.; van Wely, M.; Mastenbroek, S. Preimplantation genetic testing for aneuploidies (abnormal number of chromosomes) in in vitro fertilisation. Cochrane Database Syst. Rev. 2020, 9, CD005291. [Google Scholar] [CrossRef] [PubMed]
- Mulawkar, P.M.; Maheshwari, P.N.; Agrawal, S.G. Clinical Andrologists: Do We Really Need Them in the Era of ART? J. Hum. Reprod. Sci. 2021, 14, 105–112. [Google Scholar] [CrossRef]
- Esteves, S.C.; Zini, A.; Aziz, N.; Alvarez, J.G.; Sabanegh, E.S., Jr.; Agarwal, A. Critical appraisal of World Health Organization’s new reference values for human semen characteristics and effect on diagnosis and treatment of subfertile men. Urology 2012, 79, 16–22. [Google Scholar] [CrossRef]
- Babayev, S.N.; Park, C.W.; Bukulmez, O. Intracytoplasmic sperm injection indications: How rigorous? Semin. Reprod. Med. 2014, 32, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Cissen, M.; Bensdorp, A.; Cohlen, B.J.; Repping, S.; de Bruin, J.P.; van Wely, M. Assisted reproductive technologies for male subfertility. Cochrane Database Syst. Rev. 2016, 2, CD000360. [Google Scholar] [CrossRef] [PubMed]
- Tournaye, H.; Verheyen, G.; Albano, C.; Camus, M.; Van Landuyt, L.; Devroey, P.; Van Steirteghem, A. Intracytoplasmic sperm injection versus in vitro fertilization: A randomized controlled trial and a meta-analysis of the literature. Fertil. Steril. 2002, 78, 1030–1037. [Google Scholar] [CrossRef]
- Shuai, H.L.; Ye, Q.; Huang, Y.H.; Xie, B.G. Comparison of conventional in vitro fertilisation and intracytoplasmic sperm injection outcomes in patients with moderate oligoasthenozoospermia. Andrologia 2015, 47, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Kruger, T.F.; Menkveld, R.; Stander, F.S.; Lombard, C.J.; Van der Merwe, J.P.; van Zyl, J.A.; Smith, K. Sperm morphologic features as a prognostic factor in in vitro fertilization. Fertil. Steril. 1986, 46, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- Kruger, T.F.; Acosta, A.A.; Simmons, K.F.; Swanson, R.J.; Matta, J.F.; Oehninger, S. Predictive value of abnormal sperm morphology in in vitro fertilization. Fertil. Steril. 1988, 49, 112–117. [Google Scholar] [CrossRef]
- Gunalp, S.; Onculoglu, C.; Gurgan, T.; Kruger, T.F.; Lombard, C.J. A study of semen parameters with emphasis on sperm morphology in a fertile population: An attempt to develop clinical thresholds. Hum. Reprod. 2001, 16, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Menkveld, R.; Wong, W.Y.; Lombard, C.J.; Wetzels, A.M.; Thomas, C.M.; Merkus, H.M.; Steegers-Theunissen, R.P. Semen parameters, including WHO and strict criteria morphology, in a fertile and subfertile population: An effort towards standardization of in-vivo thresholds. Hum. Reprod. 2001, 16, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- Plachot, M.; Belaisch-Allart, J.; Mayenga, J.M.; Chouraqui, A.; Tesquier, L.; Serkine, A.M. Outcome of conventional IVF and ICSI on sibling oocytes in mild male factor infertility. Hum. Reprod. 2002, 17, 362–369. [Google Scholar] [CrossRef]
- Hotaling, J.M.; Smith, J.F.; Rosen, M.; Muller, C.H.; Walsh, T.J. The relationship between isolated teratozoospermia and clinical pregnancy after in vitro fertilization with or without intracytoplasmic sperm injection: A systematic review and meta-analysis. Fertil. Steril. 2011, 95, 1141–1145. [Google Scholar] [CrossRef] [PubMed]
- Tournaye, H. Male factor infertility and ART. Asian J. Androl. 2012, 14, 103–108. [Google Scholar] [CrossRef]
- Esteves, S.C.; Varghese, A.C. Laboratory handling of epididymal and testicular spermatozoa: What can be done to improve sperm injections outcome. J. Hum. Reprod. Sci. 2012, 5, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Povlsen, B.B.; Aw, L.D.; Laursen, R.J.; Esteves, S.C.; Humaidan, P. Pregnancy and birth after intracytoplasmic sperm injection with normal testicular spermatozoa in a patient with azoospermia and tail stump epididymal sperm. Int. Braz. J. Urol. 2015, 41, 1220–1225. [Google Scholar] [CrossRef] [PubMed]
- Esteves, S.C.; Schneider, D.T.; Verza, S., Jr. Influence of antisperm antibodies in the semen on intracytoplasmic sperm injection outcome. Int. Braz. J. Urol. 2007, 33, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Chamley, L.W.; Clarke, G.N. Antisperm antibodies and conception. Semin. Immunopathol. 2007, 29, 169–184. [Google Scholar] [CrossRef]
- Zini, A.; Fahmy, N.; Belzile, E.; Ciampi, A.; Al-Hathal, N.; Kotb, A. Antisperm antibodies are not associated with pregnancy rates after IVF and ICSI: Systematic review and meta-analysis. Hum. Reprod. 2011, 26, 1288–1295. [Google Scholar] [CrossRef]
- Dam, A.H.; Feenstra, I.; Westphal, J.R.; Ramos, L.; van Golde, R.J.; Kremer, J.A. Globozoospermia revisited. Hum. Reprod. Update 2007, 13, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Alviggi, C.; Humaidan, P.; Howles, C.M.; Tredway, D.; Hillier, S.G. Biological versus chronological ovarian age: Implications for assisted reproductive technology. Reprod. Biol. Endocrinol. 2009, 7, 101. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Y.; Mensah, V.; Huber, W.J., 3rd; Huang, Y.T.; Alvero, R. Discordant anti-mullerian hormone (AMH) and follicle stimulating hormone (FSH) among women undergoing in vitro fertilization (IVF): Which one is the better predictor for live birth? J. Ovarian. Res. 2018, 11, 60. [Google Scholar] [CrossRef]
- Krausz, C.; Casamonti, E. Spermatogenic failure and the Y chromosome. Hum. Genet. 2017, 136, 637–655. [Google Scholar] [CrossRef]
- Vogt, P.H.; Edelmann, A.; Kirsch, S.; Henegariu, O.; Hirschmann, P.; Kiesewetter, F.; Kohn, F.M.; Schill, W.B.; Farah, S.; Ramos, C.; et al. Human Y chromosome azoospermia factors (AZF) mapped to different subregions in Yq11. Hum. Mol. Genet. 1996, 5, 933–943. [Google Scholar] [CrossRef]
- Krausz, C.; Forti, G.; McElreavey, K. The Y chromosome and male fertility and infertility. Int. J. Androl. 2003, 26, 70–75. [Google Scholar] [CrossRef]
- Hinch, A.G.; Altemose, N.; Noor, N.; Donnelly, P.; Myers, S.R. Recombination in the human Pseudoautosomal region PAR1. PLoS Genet. 2014, 10, e1004503. [Google Scholar] [CrossRef] [PubMed]
- Colaco, S.; Modi, D. Genetics of the human Y chromosome and its association with male infertility. Reprod. Biol. Endocrinol. 2018, 16, 14. [Google Scholar] [CrossRef] [PubMed]
- Hopps, C.V.; Mielnik, A.; Goldstein, M.; Palermo, G.D.; Rosenwaks, Z.; Schlegel, P.N. Detection of sperm in men with Y chromosome microdeletions of the AZFa, AZFb and AZFc regions. Hum. Reprod. 2003, 18, 1660–1665. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Lee, H.S.; Choe, J.H.; Lee, J.S.; Seo, J.T. Success rate of microsurgical multiple testicular sperm extraction and sperm presence in the ejaculate in korean men with y chromosome microdeletions. Korean J. Urol. 2013, 54, 536–540. [Google Scholar] [CrossRef]
- Abur, U.; Gunes, S.; Asci, R.; Altundag, E.; Akar, O.S.; Ayas, B.; Karadag Alpaslan, M.; Ogur, G. Chromosomal and Y-chromosome microdeletion analysis in 1,300 infertile males and the fertility outcome of patients with AZFc microdeletions. Andrologia 2019, 51, e13402. [Google Scholar] [CrossRef]
- de la Chapelle, A. The etiology of maleness in XX men. Hum. Genet. 1981, 58, 105–116. [Google Scholar] [CrossRef]
- Vorona, E.; Zitzmann, M.; Gromoll, J.; Schuring, A.N.; Nieschlag, E. Clinical, endocrinological, and epigenetic features of the 46,XX male syndrome, compared with 47,XXY Klinefelter patients. J. Clin. Endocrinol. Metab. 2007, 92, 3458–3465. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.; Page, D.C.; de la Chapelle, A. Chromosome Y-specific DNA is transferred to the short arm of X chromosome in human XX males. Science 1986, 233, 786–788. [Google Scholar] [CrossRef] [PubMed]
- Zenteno-Ruiz, J.C.; Kofman-Alfaro, S.; Mendez, J.P. 46,XX sex reversal. Arch. Med. Res. 2001, 32, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Harley, V.R. Disorders of sex development: New genes, new concepts. Nat. Rev. Endocrinol. 2013, 9, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Rajender, S.; Rajani, V.; Gupta, N.J.; Chakravarty, B.; Singh, L.; Thangaraj, K. SRY-negative 46,XX male with normal genitals, complete masculinization and infertility. Mol. Hum. Reprod. 2006, 12, 341–346. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fechner, P.Y.; Marcantonio, S.M.; Jaswaney, V.; Stetten, G.; Goodfellow, P.N.; Migeon, C.J.; Smith, K.D.; Berkovitz, G.D.; Amrhein, J.A.; Bard, P.A.; et al. The role of the sex-determining region Y gene in the etiology of 46,XX maleness. J. Clin. Endocrinol. Metab. 1993, 76, 690–695. [Google Scholar] [CrossRef]
- Ogata, T.; Hawkins, J.R.; Taylor, A.; Matsuo, N.; Hata, J.; Goodfellow, P.N. Sex reversal in a child with a 46,X,Yp+ karyotype: Support for the existence of a gene(s), located in distal Xp, involved in testis formation. J. Med. Genet. 1992, 29, 226–230. [Google Scholar] [CrossRef]
- Boucekkine, C.; Toublanc, J.E.; Abbas, N.; Chaabouni, S.; Ouahid, S.; Semrouni, M.; Jaubert, F.; Toublanc, M.; McElreavey, K.; Vilain, E.; et al. Clinical and anatomical spectrum in XX sex reversed patients. Relationship to the presence of Y specific DNA-sequences. Clin. Endocrinol. 1994, 40, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.A. 46, XX man with SRY gene translocation: Cytogenetic characteristics, clinical features and management. Am. J. Med. Sci. 2008, 335, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Majzoub, A.; Arafa, M.; Starks, C.; Elbardisi, H.; Al Said, S.; Sabanegh, E. 46 XX karyotype during male fertility evaluation; case series and literature review. Asian J. Androl. 2017, 19, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Yatsenko, A.N.; Georgiadis, A.P.; Ropke, A.; Berman, A.J.; Jaffe, T.; Olszewska, M.; Westernstroer, B.; Sanfilippo, J.; Kurpisz, M.; Rajkovic, A.; et al. X-linked TEX11 mutations, meiotic arrest, and azoospermia in infertile men. N. Engl. J. Med. 2015, 372, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Silber, S.; Leu, N.A.; Oates, R.D.; Marszalek, J.D.; Skaletsky, H.; Brown, L.G.; Rozen, S.; Page, D.C.; Wang, P.J. TEX11 is mutated in infertile men with azoospermia and regulates genome-wide recombination rates in mouse. EMBO Mol. Med. 2015, 7, 1198–1210. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.; Zheng, L.; Ji, Z.; Mei, L.; Ding, L.; Lin, S.; Wang, X.; Yang, X.; Li, P. A novel TEX11 mutation induces azoospermia: A case report of infertile brothers and literature review. BMC Med. Genet. 2018, 19, 63. [Google Scholar] [CrossRef]
- Krausz, C.; Riera-Escamilla, A.; Moreno-Mendoza, D.; Holleman, K.; Cioppi, F.; Algaba, F.; Pybus, M.; Friedrich, C.; Wyrwoll, M.J.; Casamonti, E.; et al. Genetic dissection of spermatogenic arrest through exome analysis: Clinical implications for the management of azoospermic men. Genet. Med. 2020, 22, 1956–1966. [Google Scholar] [CrossRef]
- Krausz, C.; Cioppi, F. Genetic Factors of Non-Obstructive Azoospermia: Consequences on Patients’ and Offspring Health. J. Clin. Med. 2021, 10, 4009. [Google Scholar] [CrossRef]
- Björndahl, L.; Kirkman Brown, J.; other Editorial Board Members of the WHO Laboratory Manual for the Examination and Processing of Human Semen. The sixth edition of the WHO Laboratory Manual for the Examination and Processing of Human Semen: Ensuring quality and standardization in basic examination of human ejaculates. Fertil. Steril. 2022, 117, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Wosnitzer, M.S. Genetic evaluation of male infertility. Transl. Androl. Urol. 2014, 3, 17–26. [Google Scholar] [CrossRef]
- Ferlin, A.; Schipilliti, M.; Di Mambro, A.; Vinanzi, C.; Foresta, C. Osteoporosis in Klinefelter’s syndrome. Mol. Hum. Reprod. 2010, 16, 402–410. [Google Scholar] [CrossRef]
- Calogero, A.E.; Giagulli, V.A.; Mongioi, L.M.; Triggiani, V.; Radicioni, A.F.; Jannini, E.A.; Pasquali, D.; Klinefelter Italia, N.G. Klinefelter syndrome: Cardiovascular abnormalities and metabolic disorders. J. Endocrinol. Investig. 2017, 40, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Jetty, V.; Goldenberg, N.; Shah, P.; Wang, P. Thrombophilia in Klinefelter Syndrome with Deep Venous Thrombosis, Pulmonary Embolism, and Mesenteric Artery Thrombosis on Testosterone Therapy: A Pilot Study. Clin. Appl. Thromb. Hemost. 2017, 23, 973–979. [Google Scholar] [CrossRef]
- Brinton, L.A. Breast cancer risk among patients with Klinefelter syndrome. Acta Paediatr. 2011, 100, 814–818. [Google Scholar] [CrossRef]
- Bonouvrie, K.; van der Werff Ten Bosch, J.; van den Akker, M. Klinefelter syndrome and germ cell tumors: Review of the literature. Int. J. Pediatr. Endocrinol. 2020, 2020, 18. [Google Scholar] [CrossRef]
- Husain, F.A. Reproductive issues from the Islamic perspective. Hum. Fertil. 2000, 3, 124–128. [Google Scholar] [CrossRef]
- Deruyver, Y.; Vanderschueren, D.; Van der Aa, F. Outcome of microdissection TESE compared with conventional TESE in non-obstructive azoospermia: A systematic review. Andrology 2014, 2, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Achermann, A.P.P.; Pereira, T.A.; Esteves, S.C. Microdissection testicular sperm extraction (micro-TESE) in men with infertility due to nonobstructive azoospermia: Summary of current literature. Int. Urol. Nephrol. 2021, 53, 2193–2210. [Google Scholar] [CrossRef]
- Kavoussi, P.K.; West, B.T.; Chen, S.H.; Hunn, C.; Gilkey, M.S.; Machen, G.L.; Kavoussi, K.M.; Esqueda, A.; Wininger, J.D.; Kavoussi, S.K. A comprehensive assessment of predictors of fertility outcomes in men with non-obstructive azoospermia undergoing microdissection testicular sperm extraction. Reprod. Biol. Endocrinol. 2020, 18, 90. [Google Scholar] [CrossRef]
- Corona, G.; Minhas, S.; Giwercman, A.; Bettocchi, C.; Dinkelman-Smit, M.; Dohle, G.; Fusco, F.; Kadioglou, A.; Kliesch, S.; Kopa, Z.; et al. Sperm recovery and ICSI outcomes in men with non-obstructive azoospermia: A systematic review and meta-analysis. Hum. Reprod. Update 2019, 25, 733–757. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, K. Hormonal therapy for non-obstructive azoospermia: Basic and clinical perspectives. Reprod. Med. Biol. 2015, 14, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kaltsas, A.; Markou, E.; Zachariou, A.; Dimitriadis, F.; Mamoulakis, C.; Andreadakis, S.; Giannakis, I.; Tsounapi, P.; Takenaka, A.; Sofikitis, N. Varicoceles in Men with Non-obstructive Azoospermia: The Dilemma to Operate or Not. Front. Reprod. Health 2022, 4, 811487. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.; Agarwal, A.; Kavoussi, P.; Rambhatla, A.; Saleh, R.; Cannarella, R.; Harraz, A.M.; Boitrelle, F.; Kuroda, S.; Hamoda, T.A.A.; et al. Consensus and Diversity in the Management of Varicocele for Male Infertility: Results of a Global Practice Survey and Comparison with Guidelines and Recommendations. World J. Mens Health 2023, 41, 164–197. [Google Scholar] [CrossRef] [PubMed]
- Sajadi, H.; Hosseini, J.; Farrahi, F.; Dadkhah, F.; Sepidarkish, M.; Sabbaghian, M.; Eftekhari-Yazdi, P.; Sadighi Gilani, M.A. Varicocelectomy May Improve Results for Sperm Retrieval and Pregnancy Rate in Non-Obstructive Azoospermic Men. Int. J. Fertil. Steril. 2019, 12, 303–305. [Google Scholar] [CrossRef]
- Esteves, S.C.; Miyaoka, R.; Roque, M.; Agarwal, A. Outcome of varicocele repair in men with nonobstructive azoospermia: Systematic review and meta-analysis. Asian J. Androl. 2016, 18, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.F.S.; Ostergren, P.; Dupree, J.M.; Ohl, D.A.; Sonksen, J.; Fode, M. Varicocele and male infertility. Nat. Rev. Urol. 2017, 14, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Prabakaran, S.; Allamaneni, S.S. Relationship between oxidative stress, varicocele and infertility: A meta-analysis. Reprod. BioMed. Online 2006, 12, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Sofikitis, N.; Stavrou, S.; Skouros, S.; Dimitriadis, F.; Tsounapi, P.; Takenaka, A. Mysteries, Facts, and Fiction in Varicocele Pathophysiology and Treatment. Eur. Urol. Suppl. 2014, 13, 89–99. [Google Scholar] [CrossRef]
- Birowo, P.; Rahendra Wijaya, J.; Atmoko, W.; Rasyid, N. The effects of varicocelectomy on the DNA fragmentation index and other sperm parameters: A meta-analysis. Basic Clin. Androl. 2020, 30, 15. [Google Scholar] [CrossRef]
- Baazeem, A.; Belzile, E.; Ciampi, A.; Dohle, G.; Jarvi, K.; Salonia, A.; Weidner, W.; Zini, A. Varicocele and male factor infertility treatment: A new meta-analysis and review of the role of varicocele repair. Eur. Urol. 2011, 60, 796–808. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, J.Y.; Kang, D.H.; Lee, H.; Seo, J.T.; Cho, K.S. Impact of surgical varicocele repair on pregnancy rate in subfertile men with clinical varicocele and impaired semen quality: A meta-analysis of randomized clinical trials. Korean J. Urol. 2013, 54, 703–709. [Google Scholar] [CrossRef]
- Elzanaty, S. Varicocele repair in non-obstructive azoospermic men: Diagnostic value of testicular biopsy—A meta-analysis. Scand. J. Urol. 2014, 48, 494–498. [Google Scholar] [CrossRef]
- Kim, H.J.; Seo, J.T.; Kim, K.J.; Ahn, H.; Jeong, J.Y.; Kim, J.H.; Song, S.H.; Jung, J.H. Clinical significance of subclinical varicocelectomy in male infertility: Systematic review and meta-analysis. Andrologia 2016, 48, 654–661. [Google Scholar] [CrossRef]
- Schauer, I.; Madersbacher, S.; Jost, R.; Hubner, W.A.; Imhof, M. The impact of varicocelectomy on sperm parameters: A meta-analysis. J. Urol. 2012, 187, 1540–1547. [Google Scholar] [CrossRef] [PubMed]
- Chiles, K.A.; Schlegel, P.N. Cost-effectiveness of varicocele surgery in the era of assisted reproductive technology. Asian J. Androl. 2016, 18, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yue, H.; Yamaguchi, K.; Okada, K.; Matsushita, K.; Ando, M.; Chiba, K.; Fujisawa, M. Effect of surgical repair on testosterone production in infertile men with varicocele: A meta-analysis. Int. J. Urol. 2012, 19, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, D.; Lin, G.; Bao, J.; Wang, J.; Tan, W. Efficacy of varicocelectomy in the treatment of hypogonadism in subfertile males with clinical varicocele: A meta-analysis. Andrologia 2017, 49, e12778. [Google Scholar] [CrossRef]
- WHO. WHO Manual for the Standardized Investigation, Diagnosis and Management of the Infertile Male; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Wosnitzer, M.S.; Goldstein, M. Obstructive azoospermia. Urol. Clin. N. Am. 2014, 41, 83–95. [Google Scholar] [CrossRef]
- Penzias, A.; Bendikson, K.; Butts, S.; Coutifaris, C.; Falcone, T.; Gitlin, S.; Gracia, C.; Hansen, K.P.; Jindal, S.; Kalra, S.; et al. The management of obstructive azoospermia: A committee opinion. Fertil. Steril. 2019, 111, 873–880. [Google Scholar] [CrossRef]
- Schoor, R.A.; Elhanbly, S.; Niederberger, C.S.; Ross, L.S. The role of testicular biopsy in the modern management of male infertility. J. Urol. 2002, 167, 197–200. [Google Scholar] [CrossRef]
- de Souza, D.A.S.; Faucz, F.R.; Pereira-Ferrari, L.; Sotomaior, V.S.; Raskin, S. Congenital bilateral absence of the vas deferens as an atypical form of cystic fibrosis: Reproductive implications and genetic counseling. Andrology 2018, 6, 127–135. [Google Scholar] [CrossRef]
- Esteves, S.C.; Lee, W.; Benjamin, D.J.; Seol, B.; Verza, S., Jr.; Agarwal, A. Reproductive potential of men with obstructive azoospermia undergoing percutaneous sperm retrieval and intracytoplasmic sperm injection according to the cause of obstruction. J. Urol. 2013, 189, 232–237. [Google Scholar] [CrossRef]
- Coward, R.M.; Mills, J.N. A step-by-step guide to office-based sperm retrieval for obstructive azoospermia. Transl. Androl. Urol. 2017, 6, 730–744. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, A.A.; Anthony, M.; Martinez Acevedo, A.; Fuchs, E.F.; Hedges, J.C.; Ostrowski, K.A. Reconsidering vasectomy reversal over assisted reproduction in older couples. Fertil. Steril. 2018, 109, 1020–1024. [Google Scholar] [CrossRef]
- Dubin, J.M.; White, J.; Ory, J.; Ramasamy, R. Vasectomy reversal vs. sperm retrieval with in vitro fertilization: A contemporary, comparative analysis. Fertil. Steril. 2021, 115, 1377–1383. [Google Scholar] [CrossRef]
- Grober, E.D.; Karpman, E.; Fanipour, M. Vasectomy reversal outcomes among patients with vasal obstructive intervals greater than 10 years. Urology 2014, 83, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Gimenes, F.; Souza, R.P.; Bento, J.C.; Teixeira, J.J.; Maria-Engler, S.S.; Bonini, M.G.; Consolaro, M.E. Male infertility: A public health issue caused by sexually transmitted pathogens. Nat. Rev. Urol. 2014, 11, 672–687. [Google Scholar] [CrossRef] [PubMed]
- Fode, M.; Fusco, F.; Lipshultz, L.; Weidner, W. Sexually Transmitted Disease and Male Infertility: A Systematic Review. Eur. Urol. Focus 2016, 2, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Boeri, L.; Capogrosso, P.; Ventimiglia, E.; Pederzoli, F.; Cazzaniga, W.; Chierigo, F.; Pozzi, E.; Clementi, M.; Vigano, P.; Montanari, E.; et al. High-risk human papillomavirus in semen is associated with poor sperm progressive motility and a high sperm DNA fragmentation index in infertile men. Hum. Reprod. 2019, 34, 209–217. [Google Scholar] [CrossRef]
- Foresta, C.; Noventa, M.; De Toni, L.; Gizzo, S.; Garolla, A. HPV-DNA sperm infection and infertility: From a systematic literature review to a possible clinical management proposal. Andrology 2015, 3, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Z.; Feng, X.; Li, N.; Zhao, W.; Wei, L.; Chen, Y.; Yang, W.; Ma, H.; Yao, B.; Zhang, K.; et al. Human papillomavirus in semen and the risk for male infertility: A systematic review and meta-analysis. BMC Infect. Dis. 2017, 17, 714. [Google Scholar] [CrossRef]
- Xiong, Y.Q.; Chen, Y.X.; Cheng, M.J.; He, W.Q.; Chen, Q. The risk of human papillomavirus infection for male fertility abnormality: A meta-analysis. Asian J. Androl. 2018, 20, 493–497. [Google Scholar] [CrossRef]
- Depuydt, C.E.; Donders, G.G.G.; Verstraete, L.; Vanden Broeck, D.; Beert, J.F.A.; Salembier, G.; Bosmans, E.; Ombelet, W. Infectious human papillomavirus virions in semen reduce clinical pregnancy rates in women undergoing intrauterine insemination. Fertil. Steril. 2019, 111, 1135–1144. [Google Scholar] [CrossRef]
- Condorelli, R.A.; Russo, G.I.; Calogero, A.E.; Morgia, G.; La Vignera, S. Chronic prostatitis and its detrimental impact on sperm parameters: A systematic review and meta-analysis. J. Endocrinol. Investig. 2017, 40, 1209–1218. [Google Scholar] [CrossRef]
- Boeri, L.; Pederzoli, F.; Capogrosso, P.; Abbate, C.; Alfano, M.; Mancini, N.; Clementi, M.; Montanari, E.; Montorsi, F.; Salonia, A. Semen infections in men with primary infertility in the real-life setting. Fertil. Steril. 2020, 113, 1174–1182. [Google Scholar] [CrossRef]
- Berger, R.E.; Alexander, E.R.; Harnisch, J.P.; Paulsen, C.A.; Monda, G.D.; Ansell, J.; Holmes, K.K. Etiology, manifestations and therapy of acute epididymitis: Prospective study of 50 cases. J. Urol. 1979, 121, 750–754. [Google Scholar] [CrossRef]
- Haidl, G.; Haidl, F.; Allam, J.P.; Schuppe, H.C. Therapeutic options in male genital tract inflammation. Andrologia 2019, 51, e13207. [Google Scholar] [CrossRef]
- Salonia, A.; Rastrelli, G.; Hackett, G.; Seminara, S.B.; Huhtaniemi, I.T.; Rey, R.A.; Hellstrom, W.J.G.; Palmert, M.R.; Corona, G.; Dohle, G.R.; et al. Paediatric and adult-onset male hypogonadism. Nat. Rev. Dis. Primers 2019, 5, 38. [Google Scholar] [CrossRef]
- Martin Martins, J.; de Pina Jorge, M.; Martins Maia, C.; Roque, J.; Lemos, C.; Nunes, D.; Reis, D.; Mota, C. Primary and Secondary Hypogonadism in Male Persons with Diabetes Mellitus. Int. J. Endocrinol. 2021, 2021, 8799537. [Google Scholar] [CrossRef]
- Grossmann, M.; Matsumoto, A.M. A Perspective on Middle-Aged and Older Men with Functional Hypogonadism: Focus on Holistic Management. J. Clin. Endocrinol. Metab. 2017, 102, 1067–1075. [Google Scholar] [CrossRef]
- Ding, Y.M.; Zhang, X.J.; Li, J.P.; Chen, S.S.; Zhang, R.T.; Tan, W.L.; Shi, X.J. Treatment of idiopathic oligozoospermia with recombinant human follicle-stimulating hormone: A prospective, randomized, double-blind, placebo-controlled clinical study in Chinese population. Clin. Endocrinol. 2015, 83, 866–871. [Google Scholar] [CrossRef]
- Oka, S.; Shiraishi, K.; Matsuyama, H. Effects of human chorionic gonadotropin on testicular interstitial tissues in men with non-obstructive azoospermia. Andrology 2017, 5, 232–239. [Google Scholar] [CrossRef]
- Attia, A.M.; Abou-Setta, A.M.; Al-Inany, H.G. Gonadotrophins for idiopathic male factor subfertility. Cochrane Database Syst. Rev. 2013, CD005071. [Google Scholar] [CrossRef]
- Corona, G.; Pizzocaro, A.; Vena, W.; Rastrelli, G.; Semeraro, F.; Isidori, A.M.; Pivonello, R.; Salonia, A.; Sforza, A.; Maggi, M. Diabetes is most important cause for mortality in COVID-19 hospitalized patients: Systematic review and meta-analysis. Rev. Endocr. Metab. Disord. 2021, 22, 275–296. [Google Scholar] [CrossRef] [PubMed]
- Salonia, A.; Corona, G.; Giwercman, A.; Maggi, M.; Minhas, S.; Nappi, R.E.; Sofikitis, N.; Vignozzi, L. SARS-CoV-2, testosterone and frailty in males (PROTEGGIMI): A multidimensional research project. Andrology 2021, 9, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Pivonello, R.; Auriemma, R.S.; Pivonello, C.; Isidori, A.M.; Corona, G.; Colao, A.; Millar, R.P. Sex Disparities in COVID-19 Severity and Outcome: Are Men Weaker or Women Stronger? Neuroendocrinology 2021, 111, 1066–1085. [Google Scholar] [CrossRef]
- Kalfas, T.; Kaltsas, A.; Symeonidis, E.N.; Symeonidis, A.; Zikopoulos, A.; Moustakli, E.; Tsiampali, C.; Tsampoukas, G.; Palapela, N.; Zachariou, A.; et al. COVID-19 and Male Infertility: Is There a Role for Antioxidants? Antioxidants 2023, 12, 1483. [Google Scholar] [CrossRef]
- Rastrelli, G.; Di Stasi, V.; Inglese, F.; Beccaria, M.; Garuti, M.; Di Costanzo, D.; Spreafico, F.; Greco, G.F.; Cervi, G.; Pecoriello, A.; et al. Low testosterone levels predict clinical adverse outcomes in SARS-CoV-2 pneumonia patients. Andrology 2021, 9, 88–98. [Google Scholar] [CrossRef]
- Salciccia, S.; Del Giudice, F.; Gentile, V.; Mastroianni, C.M.; Pasculli, P.; Di Lascio, G.; Ciardi, M.R.; Sperduti, I.; Maggi, M.; De Berardinis, E.; et al. Interplay between male testosterone levels and the risk for subsequent invasive respiratory assistance among COVID-19 patients at hospital admission. Endocrine 2020, 70, 206–210. [Google Scholar] [CrossRef]
- Cinislioglu, A.E.; Cinislioglu, N.; Demirdogen, S.O.; Sam, E.; Akkas, F.; Altay, M.S.; Utlu, M.; Sen, I.A.; Yildirim, F.; Kartal, S.; et al. The relationship of serum testosterone levels with the clinical course and prognosis of COVID-19 disease in male patients: A prospective study. Andrology 2022, 10, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Kadihasanoglu, M.; Aktas, S.; Yardimci, E.; Aral, H.; Kadioglu, A. SARS-CoV-2 Pneumonia Affects Male Reproductive Hormone Levels: A Prospective, Cohort Study. J. Sex. Med. 2021, 18, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Salonia, A.; Pontillo, M.; Capogrosso, P.; Gregori, S.; Tassara, M.; Boeri, L.; Carenzi, C.; Abbate, C.; Cignoli, D.; Ferrara, A.M.; et al. Severely low testosterone in males with COVID-19: A case-control study. Andrology 2021, 9, 1043–1052. [Google Scholar] [CrossRef]
- Salonia, A.; Pontillo, M.; Capogrosso, P.; Gregori, S.; Carenzi, C.; Ferrara, A.M.; Rowe, I.; Boeri, L.; Larcher, A.; Ramirez, G.A.; et al. Testosterone in males with COVID-19: A 7-month cohort study. Andrology 2022, 10, 34–41. [Google Scholar] [CrossRef]
- Patel, N.; Kashanian, J.A. Thyroid Dysfunction and Male Reproductive Physiology. Semin. Reprod. Med. 2016, 34, 356–360. [Google Scholar] [CrossRef]
- Krassas, G.E.; Pontikides, N.; Deligianni, V.; Miras, K. A prospective controlled study of the impact of hyperthyroidism on reproductive function in males. J. Clin. Endocrinol. Metab. 2002, 87, 3667–3671. [Google Scholar] [CrossRef]
- Krajewska-Kulak, E.; Sengupta, P. Thyroid function in male infertility. Front. Endocrinol. 2013, 4, 174. [Google Scholar] [CrossRef]
- Singh, P.; Singh, M.; Cugati, G.; Singh, A.K. Hyperprolactinemia: An often missed cause of male infertility. J. Hum. Reprod. Sci. 2011, 4, 102–103. [Google Scholar] [CrossRef] [PubMed]
- Kasturi, S.S.; Tannir, J.; Brannigan, R.E. The metabolic syndrome and male infertility. J. Androl. 2008, 29, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Glazer, C.H.; Bonde, J.P.; Giwercman, A.; Vassard, D.; Pinborg, A.; Schmidt, L.; Vaclavik Brauner, E. Risk of diabetes according to male factor infertility: A register-based cohort study. Hum. Reprod. 2017, 32, 1474–1481. [Google Scholar] [CrossRef]
- Kim, H.H.; Schlegel, P.N. Endocrine manipulation in male infertility. Urol. Clin. N. Am. 2008, 35, 303–318. [Google Scholar] [CrossRef]
- Sofikitis, N.; Kaltsas, A.; Dimitriadis, F.; Rassweiler, J.; Grivas, N.; Zachariou, A.; Kaponis, A.; Tsounapi, P.; Paterakis, N.; Karagiannis, A.; et al. The Effect of PDE5 Inhibitors on the Male Reproductive Tract. Curr. Pharm. Des. 2021, 27, 2697–2713. [Google Scholar] [CrossRef]
- Dimitriadis, F.; Kaltsas, A.; Zachariou, A.; Mamoulakis, C.; Tsiampali, C.; Giannakis, I.; Paschopoulos, M.; Papatsoris, A.; Loutradis, D.; Tsounapi, P.; et al. PDE5 inhibitors and male reproduction: Is there a place for PDE5 inhibitors in infertility clinics or andrology laboratories? Int. J. Urol. 2022, 29, 1405–1418. [Google Scholar] [CrossRef]
- Dong, L.; Zhang, X.; Yan, X.; Shen, Y.; Li, Y.; Yu, X. Effect of Phosphodiesterase-5 Inhibitors on the Treatment of Male Infertility: A Systematic Review and Meta-Analysis. World J. Mens Health 2021, 39, 776–796. [Google Scholar] [CrossRef] [PubMed]
- ElMazoudy, R.; AbdelHameed, N.; ElMasry, A. Paternal dapoxetine administration induced deterioration in reproductive performance, fetal outcome, sexual behavior and biochemistry of male rats. Int. J. Impot. Res. 2015, 27, 206–214. [Google Scholar] [CrossRef]
- Jannini, E.A.; Ciocca, G.; Limoncin, E.; Mollaioli, D.; Di Sante, S.; Gianfrilli, D.; Lombardo, F.; Lenzi, A. Premature ejaculation: Old story, new insights. Fertil. Steril. 2015, 104, 1061–1073. [Google Scholar] [CrossRef] [PubMed]
- Colpi, G.M.; Francavilla, S.; Haidl, G.; Link, K.; Behre, H.M.; Goulis, D.G.; Krausz, C.; Giwercman, A. European Academy of Andrology guideline Management of oligo-astheno-teratozoospermia. Andrology 2018, 6, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Tournaye, H.; Krausz, C.; Oates, R.D. Novel concepts in the aetiology of male reproductive impairment. Lancet Diabetes Endocrinol. 2017, 5, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Morrison, C.D.; Brannigan, R.E. Metabolic syndrome and infertility in men. Best Pract. Res. Clin. Obs. Gynaecol. 2015, 29, 507–515. [Google Scholar] [CrossRef]
- Rastrelli, G.; Lotti, F.; Reisman, Y.; Sforza, A.; Maggi, M.; Corona, G. Metabolically healthy and unhealthy obesity in erectile dysfunction and male infertility. Expert Rev. Endocrinol. Metab. 2019, 14, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Dang, J.T.; Switzer, N.; Yu, J.; Tian, C.; Birch, D.W.; Karmali, S. Impact of Bariatric Surgery on Male Sex Hormones and Sperm Quality: A Systematic Review and Meta-Analysis. Obes. Surg. 2019, 29, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Harlev, A.; Agarwal, A.; Esteves, S.C. Cigarette Smoking and Semen Quality: A New Meta-analysis Examining the Effect of the 2010 World Health Organization Laboratory Methods for the Examination of Human Semen. Eur. Urol. 2016, 70, 635–645. [Google Scholar] [CrossRef]
- Prentki Santos, E.; Lopez-Costa, S.; Chenlo, P.; Pugliese, M.N.; Curi, S.; Ariagno, J.; Repetto, H.; Sardi, M.; Palaoro, L.; Mendeluk, G. Impact of spontaneous smoking cessation on sperm quality: Case report. Andrologia 2011, 43, 431–435. [Google Scholar] [CrossRef]
- Ricci, E.; Al Beitawi, S.; Cipriani, S.; Candiani, M.; Chiaffarino, F.; Vigano, P.; Noli, S.; Parazzini, F. Semen quality and alcohol intake: A systematic review and meta-analysis. Reprod. BioMed. Online 2017, 34, 38–47. [Google Scholar] [CrossRef]
- National Institute on Alcohol Abuse. The Physicians’ Guide to Helping Patients with Alcohol Problems; National Institute on Alcohol Abuse: Bethesda, MD, USA; Indiana University: Bloomington, IL, USA, 1995. [Google Scholar]
- Muthusami, K.R.; Chinnaswamy, P. Effect of chronic alcoholism on male fertility hormones and semen quality. Fertil. Steril. 2005, 84, 919–924. [Google Scholar] [CrossRef]
- Gabrielsen, J.S.; Tanrikut, C. Chronic exposures and male fertility: The impacts of environment, diet, and drug use on spermatogenesis. Andrology 2016, 4, 648–661. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Desai, N.R.; Makker, K.; Varghese, A.; Mouradi, R.; Sabanegh, E.; Sharma, R. Effects of radiofrequency electromagnetic waves (RF-EMW) from cellular phones on human ejaculated semen: An in vitro pilot study. Fertil. Steril. 2009, 92, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Mafra, F.A.; Christofolini, D.M.; Bianco, B.; Gava, M.M.; Glina, S.; Belangero, S.I.; Barbosa, C.P. Chromosomal and molecular abnormalities in a group of Brazilian infertile men with severe oligozoospermia or non-obstructive azoospermia attending an infertility service. Int. Braz. J. Urol. 2011, 37, 244–250; discussion 250–251. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Raman, J.D.; Nobert, C.F.; Goldstein, M. Increased incidence of testicular cancer in men presenting with infertility and abnormal semen analysis. J. Urol. 2005, 174, 1819–1822; discussion 1822. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, M.L.; Li, S.; Behr, B.; Cullen, M.R.; Galusha, D.; Lamb, D.J.; Lipshultz, L.I. Semen quality, infertility and mortality in the USA. Hum. Reprod. 2014, 29, 1567–1574. [Google Scholar] [CrossRef]
- Jensen, T.K.; Jacobsen, R.; Christensen, K.; Nielsen, N.C.; Bostofte, E. Good semen quality and life expectancy: A cohort study of 43,277 men. Am. J. Epidemiol. 2009, 170, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Practice Committee of the American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2013, 99, 63. [Google Scholar] [CrossRef]
- Tan, J.; Taskin, O.; Albert, A.; Bedaiwy, M.A. Association between sperm DNA fragmentation and idiopathic recurrent pregnancy loss: A systematic review and meta-analysis. Reprod. BioMed. Online 2019, 38, 951–960. [Google Scholar] [CrossRef]
- McQueen, D.B.; Zhang, J.; Robins, J.C. Sperm DNA fragmentation and recurrent pregnancy loss: A systematic review and meta-analysis. Fertil. Steril. 2019, 112, 54–60. [Google Scholar] [CrossRef]
- Smits, R.M.; Mackenzie-Proctor, R.; Yazdani, A.; Stankiewicz, M.T.; Jordan, V.; Showell, M.G. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2019, 3, CD007411. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, R.; Besada, S.; Lamb, D.J. Fluorescent in situ hybridization of human sperm: Diagnostics, indications, and therapeutic implications. Fertil. Steril. 2014, 102, 1534–1539. [Google Scholar] [CrossRef]
- Ly, K.D.; Agarwal, A.; Nagy, Z.P. Preimplantation genetic screening: Does it help or hinder IVF treatment and what is the role of the embryo? J. Assist. Reprod. Genet. 2011, 28, 833–849. [Google Scholar] [CrossRef]
- Tharakan, T.; Bettocchi, C.; Carvalho, J.; Corona, G.; Jones, T.H.; Kadioglu, A.; Salamanca, J.I.M.; Serefoglu, E.C.; Verze, P.; Salonia, A.; et al. European Association of Urology Guidelines Panel on Male Sexual and Reproductive Health: A Clinical Consultation Guide on the Indications for Performing Sperm DNA Fragmentation Testing in Men with Infertility and Testicular Sperm Extraction in Nonazoospermic Men. Eur. Urol. Focus 2022, 8, 339–350. [Google Scholar] [CrossRef]
- Vandekerckhove, F.W.; De Croo, I.; Gerris, J.; Vanden Abbeel, E.; De Sutter, P. Sperm Chromatin Dispersion Test before Sperm Preparation Is Predictive of Clinical Pregnancy in Cases of Unexplained Infertility Treated with Intrauterine Insemination and Induction with Clomiphene Citrate. Front. Med. 2016, 3, 63. [Google Scholar] [CrossRef]
- Bungum, M.; Humaidan, P.; Axmon, A.; Spano, M.; Bungum, L.; Erenpreiss, J.; Giwercman, A. Sperm DNA integrity assessment in prediction of assisted reproduction technology outcome. Hum. Reprod. 2007, 22, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Duran, E.H.; Morshedi, M.; Taylor, S.; Oehninger, S. Sperm DNA quality predicts intrauterine insemination outcome: A prospective cohort study. Hum. Reprod. 2002, 17, 3122–3128. [Google Scholar] [CrossRef]
- Chen, Q.; Zhao, J.Y.; Xue, X.; Zhu, G.X. The association between sperm DNA fragmentation and reproductive outcomes following intrauterine insemination, a meta analysis. Reprod. Toxicol. 2019, 86, 50–55. [Google Scholar] [CrossRef]
- Muriel, L.; Meseguer, M.; Fernandez, J.L.; Alvarez, J.; Remohi, J.; Pellicer, A.; Garrido, N. Value of the sperm chromatin dispersion test in predicting pregnancy outcome in intrauterine insemination: A blind prospective study. Hum. Reprod. 2006, 21, 738–744. [Google Scholar] [CrossRef]
- Ramasamy, R.; Scovell, J.M.; Kovac, J.R.; Cook, P.J.; Lamb, D.J.; Lipshultz, L.I. Fluorescence in situ hybridization detects increased sperm aneuploidy in men with recurrent pregnancy loss. Fertil. Steril. 2015, 103, 906–909.e1. [Google Scholar] [CrossRef]
- Borges, E., Jr.; Zanetti, B.F.; Setti, A.S.; Braga, D.; Provenza, R.R.; Iaconelli, A., Jr. Sperm DNA fragmentation is correlated with poor embryo development, lower implantation rate, and higher miscarriage rate in reproductive cycles of non-male factor infertility. Fertil. Steril. 2019, 112, 483–490. [Google Scholar] [CrossRef]
- Pabuccu, E.G.; Caglar, G.S.; Tangal, S.; Haliloglu, A.H.; Pabuccu, R. Testicular versus ejaculated spermatozoa in ICSI cycles of normozoospermic men with high sperm DNA fragmentation and previous ART failures. Andrologia 2017, 49, e12609. [Google Scholar] [CrossRef]
- Esteves, S.C. Novel concepts in male factor infertility: Clinical and laboratory perspectives. J. Assist. Reprod. Genet. 2016, 33, 1319–1335. [Google Scholar] [CrossRef]
- Cissen, M.; Wely, M.V.; Scholten, I.; Mansell, S.; Bruin, J.P.; Mol, B.W.; Braat, D.; Repping, S.; Hamer, G. Measuring Sperm DNA Fragmentation and Clinical Outcomes of Medically Assisted Reproduction: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0165125. [Google Scholar] [CrossRef]
- Simon, L.; Zini, A.; Dyachenko, A.; Ciampi, A.; Carrell, D.T. A systematic review and meta-analysis to determine the effect of sperm DNA damage on in vitro fertilization and intracytoplasmic sperm injection outcome. Asian J. Androl. 2017, 19, 80–90. [Google Scholar] [CrossRef]
- Agarwal, A.; Cho, C.L.; Esteves, S.C. Should we evaluate and treat sperm DNA fragmentation? Curr. Opin. Obs. Gynecol. 2016, 28, 164–171. [Google Scholar] [CrossRef]
- Esteves, S.C.; Roque, M.; Bedoschi, G.; Haahr, T.; Humaidan, P. Intracytoplasmic sperm injection for male infertility and consequences for offspring. Nat. Rev. Urol. 2018, 15, 535–562. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.; Alsomait, H.; Seshadri, S.; El-Toukhy, T.; Khalaf, Y. The effect of sperm DNA fragmentation on live birth rate after IVF or ICSI: A systematic review and meta-analysis. Reprod. BioMed. Online 2015, 30, 120–127. [Google Scholar] [CrossRef]
- Zini, A.; Boman, J.M.; Belzile, E.; Ciampi, A. Sperm DNA damage is associated with an increased risk of pregnancy loss after IVF and ICSI: Systematic review and meta-analysis. Hum. Reprod. 2008, 23, 2663–2668. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.; Gallos, I.D.; Conner, S.J.; Rajkhowa, M.; Miller, D.; Lewis, S.; Kirkman-Brown, J.; Coomarasamy, A. The effect of sperm DNA fragmentation on miscarriage rates: A systematic review and meta-analysis. Hum. Reprod. 2012, 27, 2908–2917. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Q.; Wang, Y.; Li, Y. Whether sperm deoxyribonucleic acid fragmentation has an effect on pregnancy and miscarriage after in vitro fertilization/intracytoplasmic sperm injection: A systematic review and meta-analysis. Fertil. Steril. 2014, 102, 998–1005.e8. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Majzoub, A.; Esteves, S.C.; Ko, E.; Ramasamy, R.; Zini, A. Clinical utility of sperm DNA fragmentation testing: Practice recommendations based on clinical scenarios. Transl. Androl. Urol. 2016, 5, 935–950. [Google Scholar] [CrossRef] [PubMed]
- Esteves, S.C.; Roque, M.; Garrido, N. Use of testicular sperm for intracytoplasmic sperm injection in men with high sperm DNA fragmentation: A SWOT analysis. Asian J. Androl. 2018, 20, 1. [Google Scholar] [CrossRef]
- Ambar, R.F.; Agarwal, A.; Majzoub, A.; Vij, S.; Tadros, N.N.; Cho, C.L.; Parekh, N.; Borges, E.; Glina, S. The Use of Testicular Sperm for Intracytoplasmic Sperm Injection in Patients with High Sperm DNA Damage: A Systematic Review. World J. Mens Health 2021, 39, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Khandwala, Y.S.; Zhang, C.A.; Lu, Y.; Eisenberg, M.L. The age of fathers in the USA is rising: An analysis of 168 867 480 births from 1972 to 2015. Hum. Reprod. 2017, 32, 2110–2116. [Google Scholar] [CrossRef]
- Gunes, S.; Hekim, G.N.; Arslan, M.A.; Asci, R. Effects of aging on the male reproductive system. J. Assist. Reprod. Genet. 2016, 33, 441–454. [Google Scholar] [CrossRef] [PubMed]
- McSwiggin, H.M.; O’Doherty, A.M. Epigenetic reprogramming during spermatogenesis and male factor infertility. Reproduction 2018, 156, R9–R21. [Google Scholar] [CrossRef]
- Halvaei, I.; Litzky, J.; Esfandiari, N. Advanced paternal age: Effects on sperm parameters, assisted reproduction outcomes and offspring health. Reprod. Biol. Endocrinol. 2020, 18, 110. [Google Scholar] [CrossRef]
- Malaspina, D. Paternal factors and schizophrenia risk: De novo mutations and imprinting. Schizophr. Bull. 2001, 27, 379–393. [Google Scholar] [CrossRef]
- de Kluiver, H.; Buizer-Voskamp, J.E.; Dolan, C.V.; Boomsma, D.I. Paternal age and psychiatric disorders: A review. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2017, 174, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Malaspina, D.; Gilman, C.; Kranz, T.M. Paternal age and mental health of offspring. Fertil. Steril. 2015, 103, 1392–1396. [Google Scholar] [CrossRef]
- Kaltsas, A.; Moustakli, E.; Zikopoulos, A.; Georgiou, I.; Dimitriadis, F.; Symeonidis, E.N.; Markou, E.; Michaelidis, T.M.; Tien, D.M.B.; Giannakis, I.; et al. Impact of Advanced Paternal Age on Fertility and Risks of Genetic Disorders in Offspring. Genes 2023, 14, 486. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.; Scarselli, F.; Minasi, M.G.; Casciani, V.; Zavaglia, D.; Dente, D.; Tesarik, J.; Franco, G. Birth of 16 healthy children after ICSI in cases of nonmosaic Klinefelter syndrome. Hum. Reprod. 2013, 28, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Denschlag, D.; Tempfer, C.; Kunze, M.; Wolff, G.; Keck, C. Assisted reproductive techniques in patients with Klinefelter syndrome: A critical review. Fertil. Steril. 2004, 82, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Fullerton, G.; Hamilton, M.; Maheshwari, A. Should non-mosaic Klinefelter syndrome men be labelled as infertile in 2009? Hum. Reprod. 2010, 25, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Brilli, S.; Forti, G. Managing infertility in patients with Klinefelter syndrome. Expert Rev. Endocrinol. Metab. 2014, 9, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Zitzmann, M.; Aksglaede, L.; Corona, G.; Isidori, A.M.; Juul, A.; T’Sjoen, G.; Kliesch, S.; D’Hauwers, K.; Toppari, J.; Slowikowska-Hilczer, J.; et al. European academy of andrology guidelines on Klinefelter Syndrome Endorsing Organization: European Society of Endocrinology. Andrology 2021, 9, 145–167. [Google Scholar] [CrossRef]
- Schulster, M.; Bernie, A.M.; Ramasamy, R. The role of estradiol in male reproductive function. Asian J. Androl. 2016, 18, 435–440. [Google Scholar] [CrossRef]
- Ring, J.D.; Lwin, A.A.; Kohler, T.S. Current medical management of endocrine-related male infertility. Asian J. Androl. 2016, 18, 357–363. [Google Scholar] [CrossRef]
- Xu, X.; Sun, M.; Ye, J.; Luo, D.; Su, X.; Zheng, D.; Feng, L.; Gao, L.; Yu, C.; Guan, Q. The Effect of Aromatase on the Reproductive Function of Obese Males. Horm. Metab. Res. 2017, 49, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, F.; Busetto, G.M.; De Berardinis, E.; Sperduti, I.; Ferro, M.; Maggi, M.; Gross, M.S.; Sciarra, A.; Eisenberg, M.L. A systematic review and meta-analysis of clinical trials implementing aromatase inhibitors to treat male infertility. Asian J. Androl. 2020, 22, 360–367. [Google Scholar] [CrossRef]
- El Meliegy, A.; Motawi, A.; El Salam, M.A.A. Systematic review of hormone replacement therapy in the infertile man. Arab J. Urol. 2018, 16, 140–147. [Google Scholar] [CrossRef]
- Davies, M.J.; Moore, V.M.; Willson, K.J.; Van Essen, P.; Priest, K.; Scott, H.; Haan, E.A.; Chan, A. Reproductive technologies and the risk of birth defects. N. Engl. J. Med. 2012, 366, 1803–1813. [Google Scholar] [CrossRef]
- Bonduelle, M.; Wennerholm, U.B.; Loft, A.; Tarlatzis, B.C.; Peters, C.; Henriet, S.; Mau, C.; Victorin-Cederquist, A.; Van Steirteghem, A.; Balaska, A.; et al. A multi-centre cohort study of the physical health of 5-year-old children conceived after intracytoplasmic sperm injection, in vitro fertilization and natural conception. Hum. Reprod. 2005, 20, 413–419. [Google Scholar] [CrossRef]
- Boulet, S.L.; Mehta, A.; Kissin, D.M.; Warner, L.; Kawwass, J.F.; Jamieson, D.J. Trends in use of and reproductive outcomes associated with intracytoplasmic sperm injection. JAMA 2015, 313, 255–263. [Google Scholar] [CrossRef]
- Kissin, D.M.; Zhang, Y.; Boulet, S.L.; Fountain, C.; Bearman, P.; Schieve, L.; Yeargin-Allsopp, M.; Jamieson, D.J. Association of assisted reproductive technology (ART) treatment and parental infertility diagnosis with autism in ART-conceived children. Hum. Reprod. 2015, 30, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Penzias, A.; Azziz, R.; Bendikson, K.; Falcone, T.; Hansen, K.; Hill, M.; Hurd, W.; Jindal, S.; Kalra, S.; Mersereau, J.; et al. Intracytoplasmic sperm injection (ICSI) for non-male factor indications: A committee opinion. Fertil. Steril. 2020, 114, 239–245. [Google Scholar] [CrossRef]
- Ata, B.; Seli, E. Economics of assisted reproductive technologies. Curr. Opin. Obs. Gynecol. 2010, 22, 183–188. [Google Scholar] [CrossRef]
- Rosenbluth, E.M.; BJ, V.A.N.V. Evolving role of assisted reproductive technologies. Clin. Obs. Gynecol. 2011, 54, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jorgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Mindlis, I.; Pinotti, R.; Swan, S.H. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum. Reprod. Update 2017, 23, 646–659. [Google Scholar] [CrossRef]

| Categories/Condition | Description | Assessment/Tests | Treatment/Management |
|---|---|---|---|
| Male infertility evaluation | Evaluation of male infertility | Semen analysis and genetic testing | Individualized based on the findings |
| Semen analysis abnormalities | Any irregularities found during semen analysis such as low sperm count, poor motility, or morphology | Semen analysis | A reproductive urologist can diagnose the underlying cause of the abnormalities and suggest appropriate treatments |
| Azoospermia | Complete absence of sperm in the semen | Semen analysis | A reproductive urologist can help identify and treat the cause |
| Leukocytospermia | Presence of white blood cells in the semen, which may indicate an infection or inflammation | Semen analysis | A reproductive urologist can determine the cause of the inflammation or infection and prescribe appropriate treatments |
| Idiopathic recurrent pregnancy loss | Pregnancy loss due to unknown male factors such as sperm DNA integrity | SDF tests like TUNEL, SCSA, FISH analysis, and the sperm aneuploidy test | Lifestyle changes, antioxidant treatment, and PGT-M with IVF |
| Recurrent ART failure | Repeated failure of ART procedures such as IVF | SDF tests like TUNEL and SCSA | A reproductive urologist can evaluate potential male factor causes for the failure of ART procedures and suggest alternatives or adjustments to the treatment plan |
| ART results and DNA fragmentation | Impact of sperm DNA fragmentation on conception rates in IVF and ICSI | SDF assays | Lifestyle changes, varicocelectomy, and sperm retrieval |
| Klinefelter syndrome | Infertility caused by additional X chromosome in males | Karyotyping | Assisted reproductive techniques and genetic counseling |
| Y-chromosome microdeletions | Infertility due to deletions on the Y chromosome | Y deletion test | Assisted reproductive techniques, genetic counseling, and potential ICSI |
| Advanced paternal age | Infertility associated with advanced paternal age | General health check-up and genetic tests | Counseling and possibly assisted reproductive techniques |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaltsas, A.; Dimitriadis, F.; Zachariou, D.; Zikopoulos, A.; Symeonidis, E.N.; Markou, E.; Tien, D.M.B.; Takenaka, A.; Sofikitis, N.; Zachariou, A. From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology. Medicina 2023, 59, 1835. https://doi.org/10.3390/medicina59101835
Kaltsas A, Dimitriadis F, Zachariou D, Zikopoulos A, Symeonidis EN, Markou E, Tien DMB, Takenaka A, Sofikitis N, Zachariou A. From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology. Medicina. 2023; 59(10):1835. https://doi.org/10.3390/medicina59101835
Chicago/Turabian StyleKaltsas, Aris, Fotios Dimitriadis, Dimitrios Zachariou, Athanasios Zikopoulos, Evangelos N. Symeonidis, Eleftheria Markou, Dung Mai Ba Tien, Atsushi Takenaka, Nikolaos Sofikitis, and Athanasios Zachariou. 2023. "From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology" Medicina 59, no. 10: 1835. https://doi.org/10.3390/medicina59101835
APA StyleKaltsas, A., Dimitriadis, F., Zachariou, D., Zikopoulos, A., Symeonidis, E. N., Markou, E., Tien, D. M. B., Takenaka, A., Sofikitis, N., & Zachariou, A. (2023). From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology. Medicina, 59(10), 1835. https://doi.org/10.3390/medicina59101835










