Protective Effect of Irsogladine against Aspirin-Induced Mucosal Injury in Human Induced Pluripotent Stem Cell-Derived Small Intestine
Abstract
1. Introduction
2. Materials and Methods
2.1. 2D-hiPSC-SI
2.2. ASA-Induced Small Intestinal Injury Model
2.3. Evaluation of the Effect of Irsogladine on ASA-Induced Small Intestinal Injury
2.4. RNA Extraction, Reverse Transcription, and Real-Time PCR
2.5. Histology and Histochemistry
2.6. Immunohistochemical Staining
2.7. Immunofluorescence Staining
2.8. RNA In Situ Hybridization
2.9. Lucifer Yellow Permeability
2.10. Statistical Analysis
3. Results
3.1. 2D-hiPSC-SI Cultured at the ALI
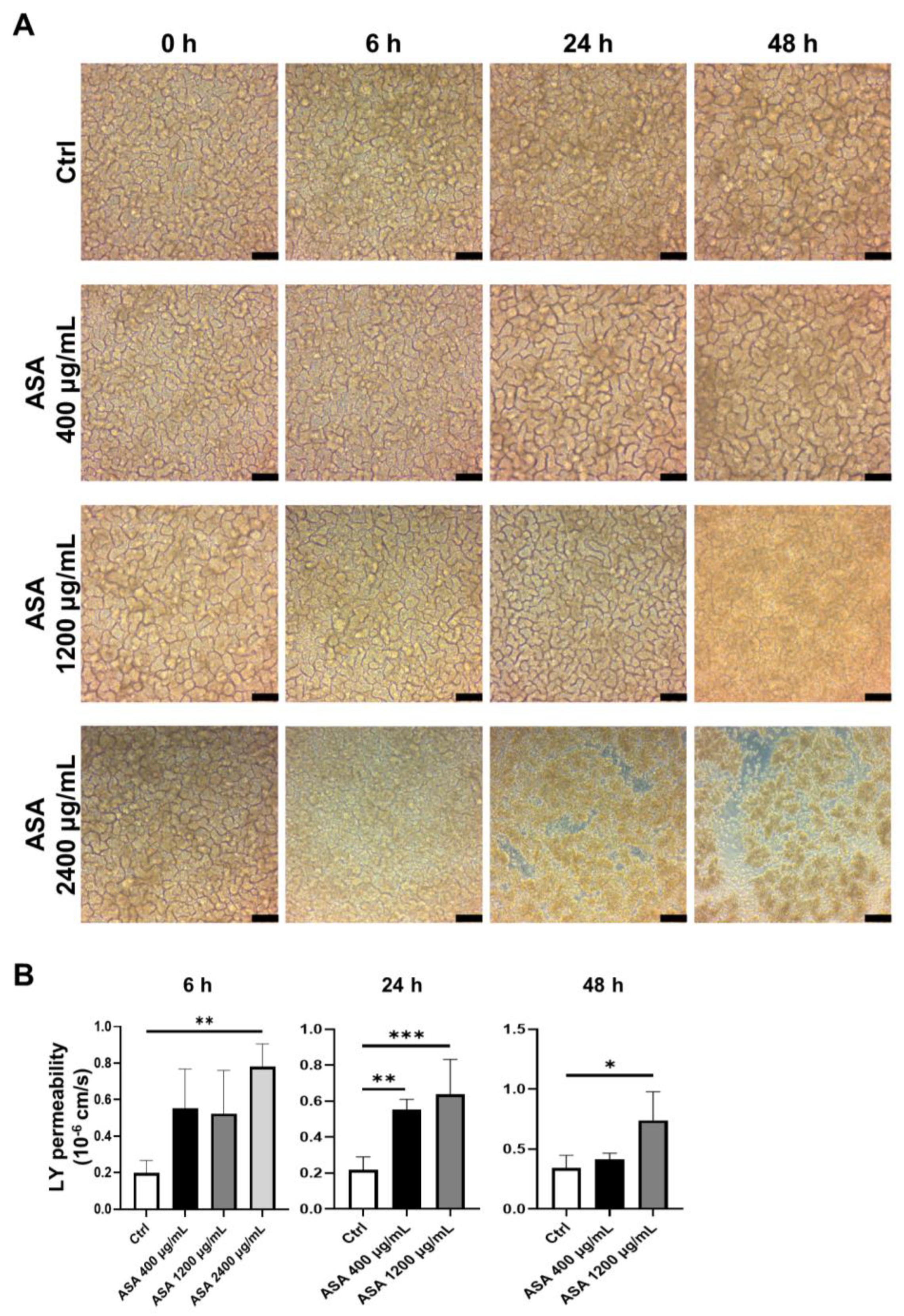
3.2. ASA-Induced Small Intestinal Injury in 2D-hiPSC-SI
3.3. Changes in mRNA Expression of Intestinal Epithelial Cell Markers after ASA-Induced Small Intestinal Injury
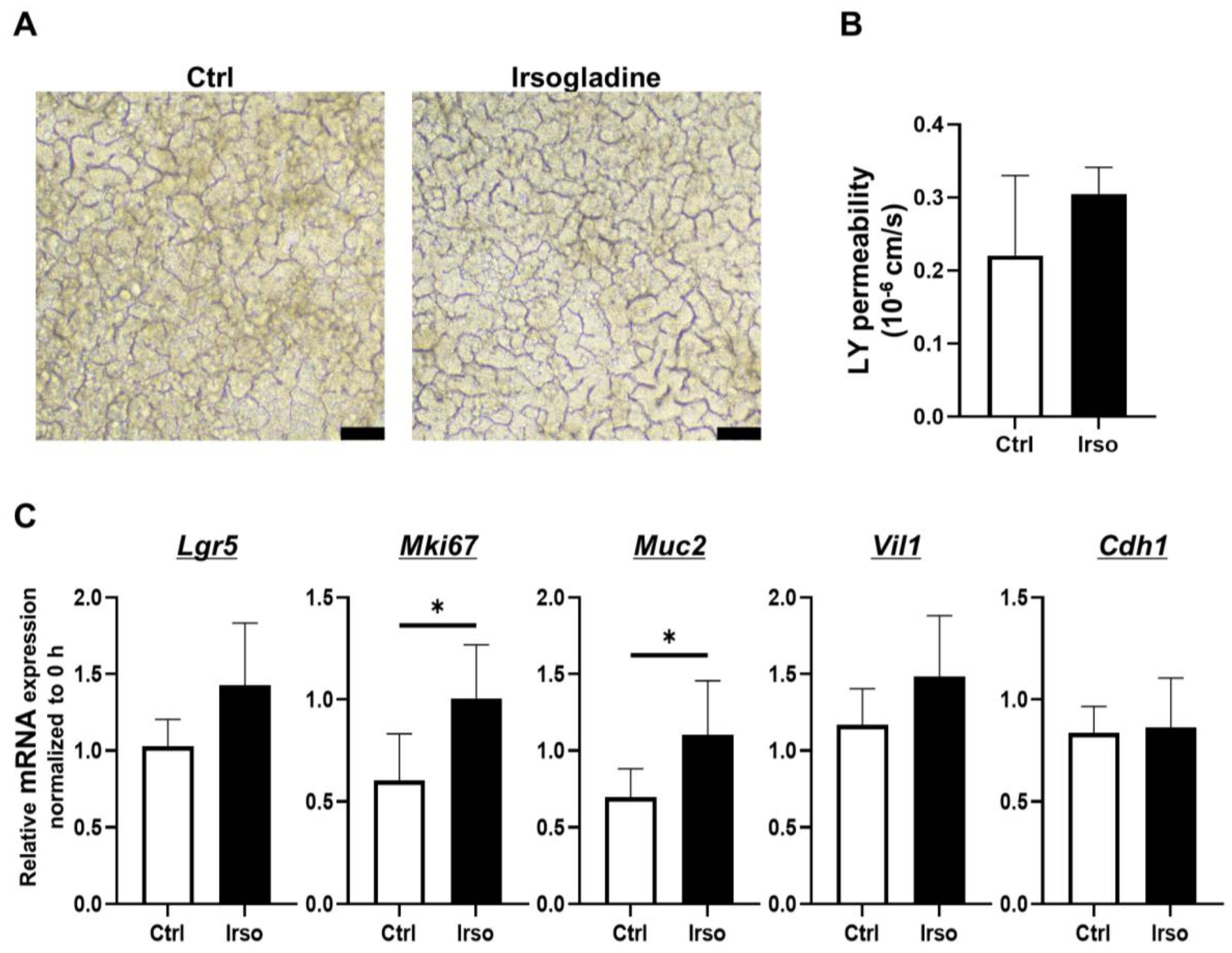
3.4. Effects of Irsogladine on 2D-hiPSC-SI under Normal Conditions
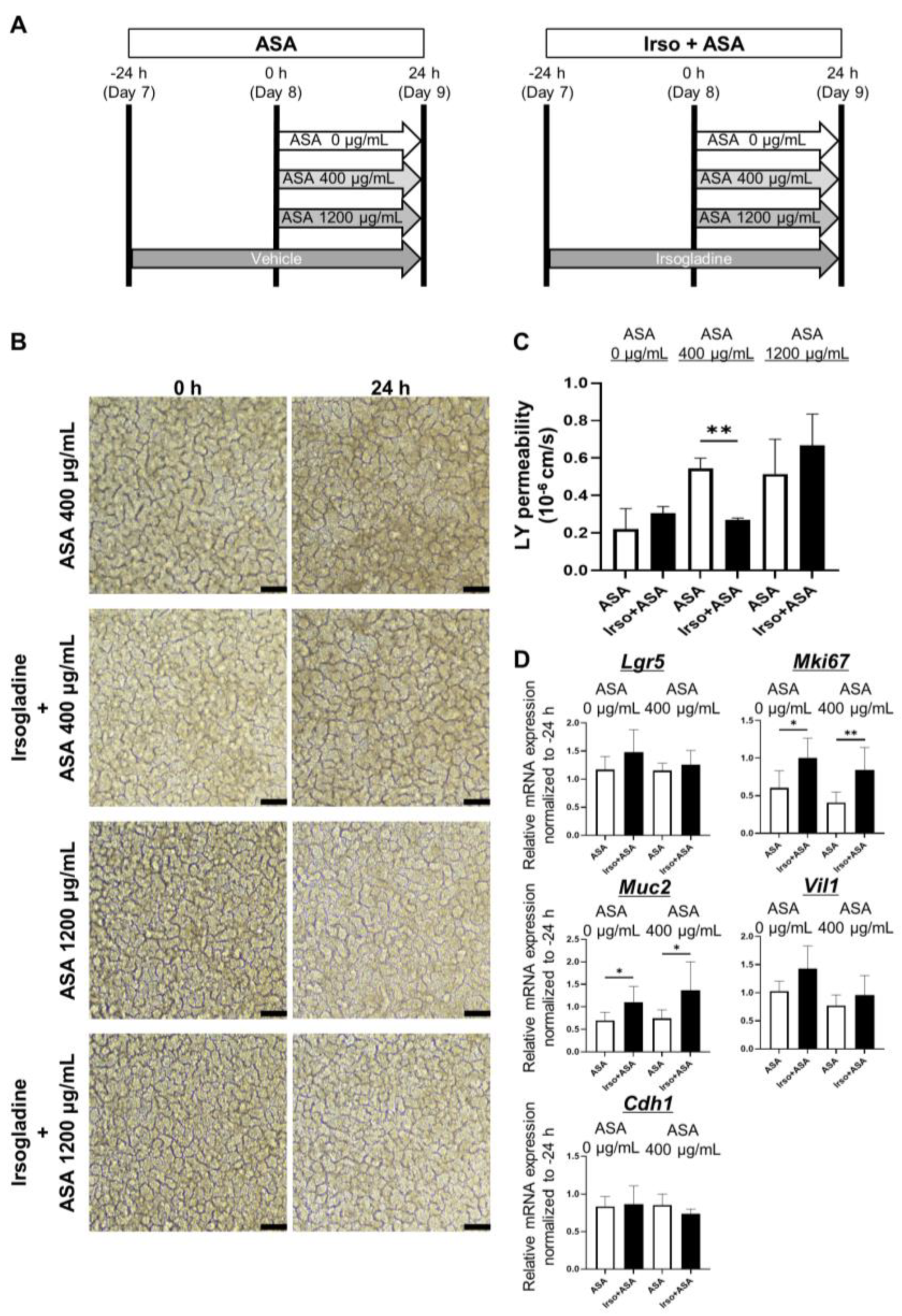
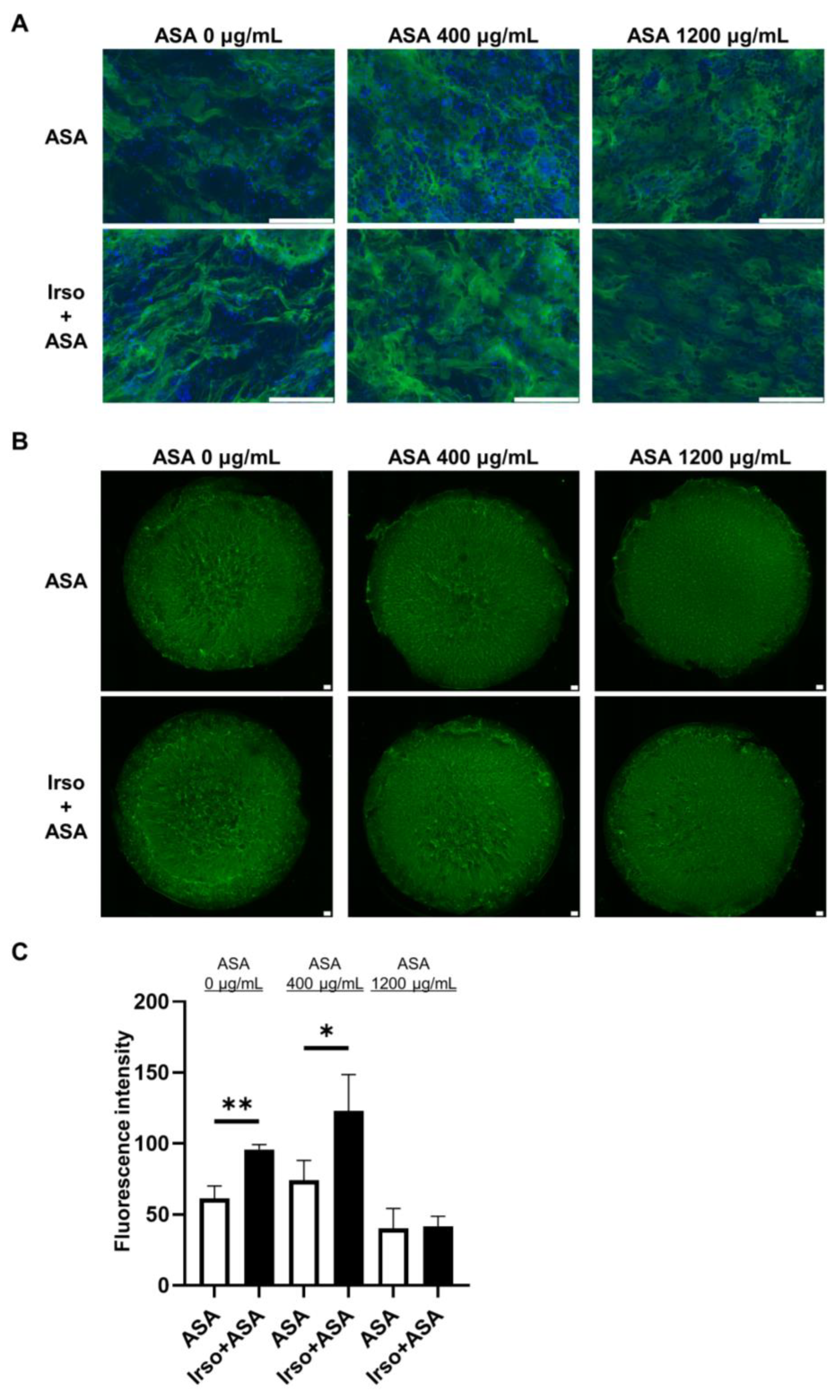
3.5. Irsogladine Plays a Protective Role against ASA-Induced Injury in 2D-hiPSC-SI
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weisman, S.M.; Graham, D.Y. Evaluation of the benefits and risks of low-dose aspirin in the secondary prevention of cardiovascular and cerebrovascular events. Arch. Intern. Med. 2002, 162, 2197–2202. [Google Scholar] [CrossRef]
- McQuaid, K.R.; Laine, L. Systematic review and meta-analysis of adverse events of low-dose aspirin and clopidogrel in randomized controlled trials. Am. J. Med. 2006, 119, 624–638. [Google Scholar] [CrossRef]
- Hayden, M.; Pignone, M.; Phillips, C.; Mulrow, C. Aspirin for the primary prevention of cardiovascular events: A summary of the evidence for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2002, 136, 161–172. [Google Scholar] [CrossRef]
- Garcia Rodriguez, L.A.; Martin-Perez, M.; Hennekens, C.H.; Rothwell, P.M.; Lanas, A. Bleeding Risk with Long-Term Low-Dose Aspirin: A Systematic Review of Observational Studies. PLoS ONE 2016, 11, e0160046. [Google Scholar] [CrossRef]
- Serrano, P.; Lanas, A.; Arroyo, M.T.; Ferreira, I.J. Risk of upper gastrointestinal bleeding in patients taking low-dose aspirin for the prevention of cardiovascular diseases. Aliment. Pharmacol. Ther. 2002, 16, 1945–1953. [Google Scholar] [CrossRef]
- Hearnshaw, S.A.; Logan, R.F.; Lowe, D.; Travis, S.P.; Murphy, M.F.; Palmer, K.R. Acute upper gastrointestinal bleeding in the UK: Patient characteristics, diagnoses and outcomes in the 2007 UK audit. Gut 2011, 60, 1327–1335. [Google Scholar] [CrossRef]
- Oakland, K.; Guy, R.; Uberoi, R.; Hogg, R.; Mortensen, N.; Murphy, M.F.; Jairath, V.; Collaborative, U.K.L.G.B. Acute lower GI bleeding in the UK: Patient characteristics, interventions and outcomes in the first nationwide audit. Gut 2018, 67, 654–662. [Google Scholar] [CrossRef]
- Lemesle, G.; Ninni, S.; de Groote, P.; Schurtz, G.; Lamblin, N.; Bauters, C. Relative impact of bleedings over ischaemic events in patients with heart failure: Insights from the CARDIONOR registry. ESC Heart Fail. 2020, 7, 3821–3829. [Google Scholar] [CrossRef]
- Kawai, T.; Oda, K.; Funao, N.; Nishimura, A.; Matsumoto, Y.; Mizokami, Y.; Ashida, K.; Sugano, K. Vonoprazan prevents low-dose aspirin-associated ulcer recurrence: Randomised phase 3 study. Gut 2018, 67, 1033–1041. [Google Scholar] [CrossRef]
- Sugano, K.; Matsumoto, Y.; Itabashi, T.; Abe, S.; Sakaki, N.; Ashida, K.; Mizokami, Y.; Chiba, T.; Matsui, S.; Kanto, T.; et al. Lansoprazole for secondary prevention of gastric or duodenal ulcers associated with long-term low-dose aspirin therapy: Results of a prospective, multicenter, double-blind, randomized, double-dummy, active-controlled trial. J. Gastroenterol. 2011, 46, 724–735. [Google Scholar] [CrossRef]
- Yeomans, N.; Lanas, A.; Labenz, J.; van Zanten, S.V.; van Rensburg, C.; Racz, I.; Tchernev, K.; Karamanolis, D.; Roda, E.; Hawkey, C.; et al. Efficacy of esomeprazole (20 mg once daily) for reducing the risk of gastroduodenal ulcers associated with continuous use of low-dose aspirin. Am. J. Gastroenterol. 2008, 103, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- Kamada, T.; Satoh, K.; Itoh, T.; Ito, M.; Iwamoto, J.; Okimoto, T.; Kanno, T.; Sugimoto, M.; Chiba, T.; Nomura, S.; et al. Evidence-based clinical practice guidelines for peptic ulcer disease 2020. J. Gastroenterol. 2021, 56, 303–322. [Google Scholar] [CrossRef] [PubMed]
- Morris, A.J.; Wasson, L.A.; MacKenzie, J.F. Small bowel enteroscopy in undiagnosed gastrointestinal blood loss. Gut 1992, 33, 887–889. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Takeuchi, T.; Handa, O.; Sakata, Y.; Tanigawa, T.; Shiba, M.; Naito, Y.; Higuchi, K.; Fujimoto, K.; Yoshikawa, T.; et al. A multicenter, randomized, double-blind, placebo-controlled trial of high-dose rebamipide treatment for low-dose aspirin-induced moderate-to-severe small intestinal damage. PLoS ONE 2015, 10, e0122330. [Google Scholar] [CrossRef] [PubMed]
- Kuramoto, T.; Umegaki, E.; Nouda, S.; Narabayashi, K.; Kojima, Y.; Yoda, Y.; Ishida, K.; Kawakami, K.; Abe, Y.; Takeuchi, T.; et al. Preventive effect of irsogladine or omeprazole on non-steroidal anti-inflammatory drug-induced esophagitis, peptic ulcers, and small intestinal lesions in humans, a prospective randomized controlled study. BMC Gastroenterol. 2013, 13, 85. [Google Scholar] [CrossRef]
- Yoshihara, T.; Oikawa, Y.; Kato, T.; Kessoku, T.; Kobayashi, T.; Kato, S.; Misawa, N.; Ashikari, K.; Fuyuki, A.; Ohkubo, H.; et al. The protective effect of Bifidobacterium bifidum G9-1 against mucus degradation by Akkermansia muciniphila following small intestine injury caused by a proton pump inhibitor and aspirin. Gut Microbes 2020, 11, 1385–1404. [Google Scholar] [CrossRef]
- Mestas, J.; Hughes, C.C. Of mice and not men: Differences between mouse and human immunology. J. Immunol. 2004, 172, 2731–2738. [Google Scholar] [CrossRef]
- Nelson, D.R.; Zeldin, D.C.; Hoffman, S.M.; Maltais, L.J.; Wain, H.M.; Nebert, D.W. Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants. Pharmacogenetics 2004, 14, 1–18. [Google Scholar] [CrossRef]
- Nakamura, T.; Sakaeda, T.; Ohmoto, N.; Tamura, T.; Aoyama, N.; Shirakawa, T.; Kamigaki, T.; Nakamura, T.; Kim, K.I.; Kim, S.R.; et al. Real-time quantitative polymerase chain reaction for MDR1, MRP1, MRP2, and CYP3A-mRNA levels in Caco-2 cell lines, human duodenal enterocytes, normal colorectal tissues, and colorectal adenocarcinomas. Drug Metab. Dispos. 2002, 30, 4–6. [Google Scholar] [CrossRef]
- Sato, T.; Vries, R.G.; Snippert, H.J.; van de Wetering, M.; Barker, N.; Stange, D.E.; van Es, J.H.; Abo, A.; Kujala, P.; Peters, P.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009, 459, 262–265. [Google Scholar] [CrossRef]
- Fujii, M.; Clevers, H.; Sato, T. Modeling Human Digestive Diseases With CRISPR-Cas9-Modified Organoids. Gastroenterology 2019, 156, 562–576. [Google Scholar] [CrossRef]
- Spence, J.R.; Mayhew, C.N.; Rankin, S.A.; Kuhar, M.F.; Vallance, J.E.; Tolle, K.; Hoskins, E.E.; Kalinichenko, V.V.; Wells, S.I.; Zorn, A.M.; et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 2011, 470, 105–109. [Google Scholar] [CrossRef]
- Finkbeiner, S.R.; Freeman, J.J.; Wieck, M.M.; El-Nachef, W.; Altheim, C.H.; Tsai, Y.H.; Huang, S.; Dyal, R.; White, E.S.; Grikscheit, T.C.; et al. Generation of tissue-engineered small intestine using embryonic stem cell-derived human intestinal organoids. Biol. Open 2015, 4, 1462–1472. [Google Scholar] [CrossRef]
- Onozato, D.; Yamashita, M.; Nakanishi, A.; Akagawa, T.; Kida, Y.; Ogawa, I.; Hashita, T.; Iwao, T.; Matsunaga, T. Generation of Intestinal Organoids Suitable for Pharmacokinetic Studies from Human Induced Pluripotent Stem Cells. Drug Metab. Dispos. 2018, 46, 1572–1580. [Google Scholar] [CrossRef]
- Onozato, D.; Ogawa, I.; Kida, Y.; Mizuno, S.; Hashita, T.; Iwao, T.; Matsunaga, T. Generation of Budding-Like Intestinal Organoids from Human Induced Pluripotent Stem Cells. J. Pharm. Sci. 2021, 110, 2637–2650. [Google Scholar] [CrossRef]
- Qiu, S.; Kabeya, T.; Ogawa, I.; Anno, S.; Hayashi, H.; Kanaki, T.; Hashita, T.; Iwao, T.; Matsunaga, T. Gellan Gum Promotes the Differentiation of Enterocytes from Human Induced Pluripotent Stem Cells. Pharmaceutics 2020, 12, 951. [Google Scholar] [CrossRef]
- Katano, T.; Ootani, A.; Mizoshita, T.; Tanida, S.; Tsukamoto, H.; Ozeki, K.; Kataoka, H.; Joh, T. Gastric mesenchymal myofibroblasts maintain stem cell activity and proliferation of murine gastric epithelium in vitro. Am. J. Pathol. 2015, 185, 798–807. [Google Scholar] [CrossRef]
- Chi, T.; Zhao, Q.; Wang, P. Fecal 16S rRNA Gene Sequencing Analysis of Changes in the Gut Microbiota of Rats with Low-Dose Aspirin-Related Intestinal Injury. Biomed Res. Int. 2021, 2021, 8848686. [Google Scholar] [CrossRef]
- Kawashima, R.; Tamaki, S.; Kawakami, F.; Maekawa, T.; Ichikawa, T. Histamine H2-Receptor Antagonists Improve Non-Steroidal Anti-Inflammatory Drug-Induced Intestinal Dysbiosis. Int. J. Mol. Sci. 2020, 21, 8166. [Google Scholar] [CrossRef]
- Chen, S.; Jiang, J.; Chao, G.; Hong, X.; Cao, H.; Zhang, S. Pure Total Flavonoids From Citrus Protect Against Nonsteroidal Anti-inflammatory Drug-Induced Small Intestine Injury by Promoting Autophagy in vivo and in vitro. Front. Pharmacol. 2021, 12, 622744. [Google Scholar] [CrossRef]
- Lai, Y.; Zhong, W.; Yu, T.; Xia, Z.S.; Li, J.Y.; Ouyang, H.; Shan, T.D.; Yang, H.S.; Chen, Q.K. Rebamipide Promotes the Regeneration of Aspirin-Induced Small-Intestine Mucosal Injury through Accumulation of beta-Catenin. PLoS ONE 2015, 10, e0132031. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kudo, T.; Esaki, M.; Yano, T.; Yamamoto, H.; Sakamoto, C.; Goto, H.; Nakase, H.; Tanaka, S.; Matsui, T.; et al. Prevalence of non-steroidal anti-inflammatory drug-induced enteropathy determined by double-balloon endoscopy: A Japanese multicenter study. Scand. J. Gastroenterol. 2008, 43, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Beura, L.K.; Hamilton, S.E.; Bi, K.; Schenkel, J.M.; Odumade, O.A.; Casey, K.A.; Thompson, E.A.; Fraser, K.A.; Rosato, P.C.; Filali-Mouhim, A.; et al. Normalizing the environment recapitulates adult human immune traits in laboratory mice. Nature 2016, 532, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Perel, P.; Roberts, I.; Sena, E.; Wheble, P.; Briscoe, C.; Sandercock, P.; Macleod, M.; Mignini, L.E.; Jayaram, P.; Khan, K.S. Comparison of treatment effects between animal experiments and clinical trials: Systematic review. BMJ 2007, 334, 197. [Google Scholar] [CrossRef]
- van der Worp, H.B.; Howells, D.W.; Sena, E.S.; Porritt, M.J.; Rewell, S.; O’Collins, V.; Macleod, M.R. Can animal models of disease reliably inform human studies? PLoS Med. 2010, 7, e1000245. [Google Scholar] [CrossRef]
- Bjarnason, I.; Williams, P.; Smethurst, P.; Peters, T.J.; Levi, A.J. Effect of non-steroidal anti-inflammatory drugs and prostaglandins on the permeability of the human small intestine. Gut 1986, 27, 1292–1297. [Google Scholar] [CrossRef]
- Sigthorsson, G.; Tibble, J.; Hayllar, J.; Menzies, I.; Macpherson, A.; Moots, R.; Scott, D.; Gumpel, M.J.; Bjarnason, I. Intestinal permeability and inflammation in patients on NSAIDs. Gut 1998, 43, 506–511. [Google Scholar] [CrossRef]
- Shimada, S.; Tanigawa, T.; Watanabe, T.; Nakata, A.; Sugimura, N.; Itani, S.; Higashimori, A.; Nadatani, Y.; Otani, K.; Taira, K.; et al. Involvement of gliadin, a component of wheat gluten, in increased intestinal permeability leading to non-steroidal anti-inflammatory drug-induced small-intestinal damage. PLoS ONE 2019, 14, e0211436. [Google Scholar] [CrossRef]
- Pelaseyed, T.; Hansson, G.C. Membrane mucins of the intestine at a glance. J. Cell Sci. 2020, 133, jcs240929. [Google Scholar] [CrossRef]
- Otani, T.; Nguyen, T.P.; Tokuda, S.; Sugihara, K.; Sugawara, T.; Furuse, K.; Miura, T.; Ebnet, K.; Furuse, M. Claudins and JAM-A coordinately regulate tight junction formation and epithelial polarity. J. Cell Biol. 2019, 218, 3372–3396. [Google Scholar] [CrossRef]
- Roberton, A.M.; Rabel, B.; Stubbs, L.; Tasman-Jones, C.; Lee, S.P. Aspirin changes the secretion rate and amino acid composition of human small intestinal mucin in subjects with ileal conduits. Glycoconj. J. 1996, 13, 781–789. [Google Scholar] [CrossRef]
- Liu, J.F.; Jamieson, G.G.; Drew, P.A.; Zhu, G.J.; Zhang, S.W.; Zhu, T.N.; Shan, B.E.; Wang, Q.Z. Aspirin induces apoptosis in oesophageal cancer cells by inhibiting the pathway of NF-kappaB downstream regulation of cyclooxygenase-2. ANZ J. Surg. 2005, 75, 1011–1016. [Google Scholar] [CrossRef]
- Wu, J.; Gong, J.; Geng, J.; Song, Y. Deoxycholic acid induces the overexpression of intestinal mucin, MUC2, via NF-kB signaling pathway in human esophageal adenocarcinoma cells. BMC Cancer 2008, 8, 333. [Google Scholar] [CrossRef]
- Bergstrom, K.S.; Xia, L. Mucin-type O-glycans and their roles in intestinal homeostasis. Glycobiology 2013, 23, 1026–1037. [Google Scholar] [CrossRef]
- Johansson, M.E.; Phillipson, M.; Petersson, J.; Velcich, A.; Holm, L.; Hansson, G.C. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 15064–15069. [Google Scholar] [CrossRef]
- Ambort, D.; Johansson, M.E.; Gustafsson, J.K.; Nilsson, H.E.; Ermund, A.; Johansson, B.R.; Koeck, P.J.; Hebert, H.; Hansson, G.C. Calcium and pH-dependent packing and release of the gel-forming MUC2 mucin. Proc. Natl. Acad. Sci. USA 2012, 109, 5645–5650. [Google Scholar] [CrossRef]
- Odenwald, M.A.; Turner, J.R. Intestinal permeability defects: Is it time to treat? Clin. Gastroenterol. Hepatol. 2013, 11, 1075–1083. [Google Scholar] [CrossRef]
- Camilleri, M.; Lasch, K.; Zhou, W. Irritable bowel syndrome: Methods, mechanisms, and pathophysiology. The confluence of increased permeability, inflammation, and pain in irritable bowel syndrome. Am. J. Physiol. -Gastrointest. Liver Physiol. 2012, 303, G775–G785. [Google Scholar] [CrossRef]
- Schoultz, I.; Keita, A.V. The Intestinal Barrier and Current Techniques for the Assessment of Gut Permeability. Cells 2020, 9, 1909. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanno, T.; Katano, T.; Ogawa, I.; Iwao, T.; Matsunaga, T.; Kataoka, H. Protective Effect of Irsogladine against Aspirin-Induced Mucosal Injury in Human Induced Pluripotent Stem Cell-Derived Small Intestine. Medicina 2023, 59, 92. https://doi.org/10.3390/medicina59010092
Kanno T, Katano T, Ogawa I, Iwao T, Matsunaga T, Kataoka H. Protective Effect of Irsogladine against Aspirin-Induced Mucosal Injury in Human Induced Pluripotent Stem Cell-Derived Small Intestine. Medicina. 2023; 59(1):92. https://doi.org/10.3390/medicina59010092
Chicago/Turabian StyleKanno, Takuya, Takahito Katano, Isamu Ogawa, Takahiro Iwao, Tamihide Matsunaga, and Hiromi Kataoka. 2023. "Protective Effect of Irsogladine against Aspirin-Induced Mucosal Injury in Human Induced Pluripotent Stem Cell-Derived Small Intestine" Medicina 59, no. 1: 92. https://doi.org/10.3390/medicina59010092
APA StyleKanno, T., Katano, T., Ogawa, I., Iwao, T., Matsunaga, T., & Kataoka, H. (2023). Protective Effect of Irsogladine against Aspirin-Induced Mucosal Injury in Human Induced Pluripotent Stem Cell-Derived Small Intestine. Medicina, 59(1), 92. https://doi.org/10.3390/medicina59010092




