Cranial and Odontological Methods for Sex Estimation—A Scoping Review
Abstract
1. Introduction
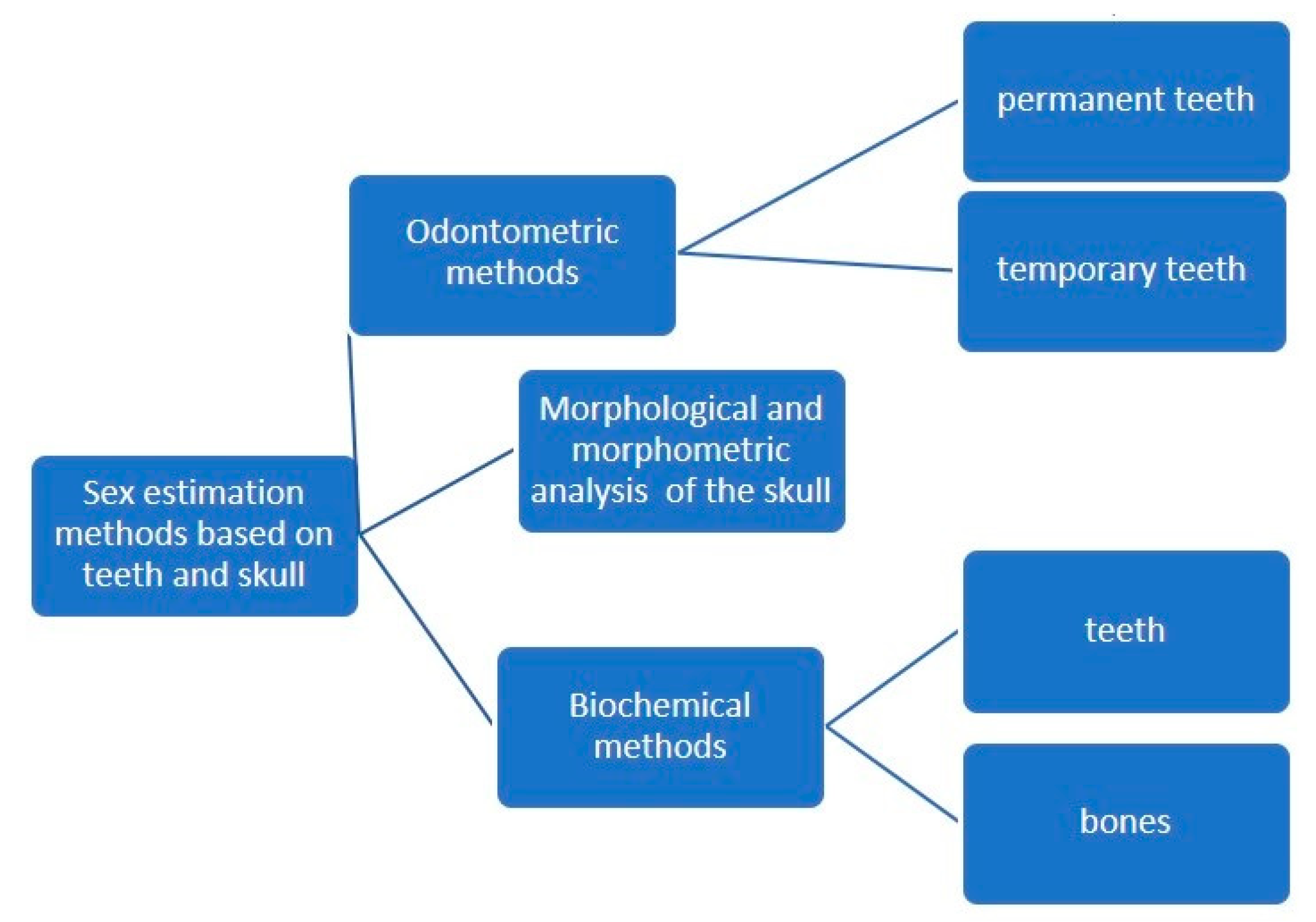
1.1. Morphological and Morphometric Methods
1.2. Biochemical Methods
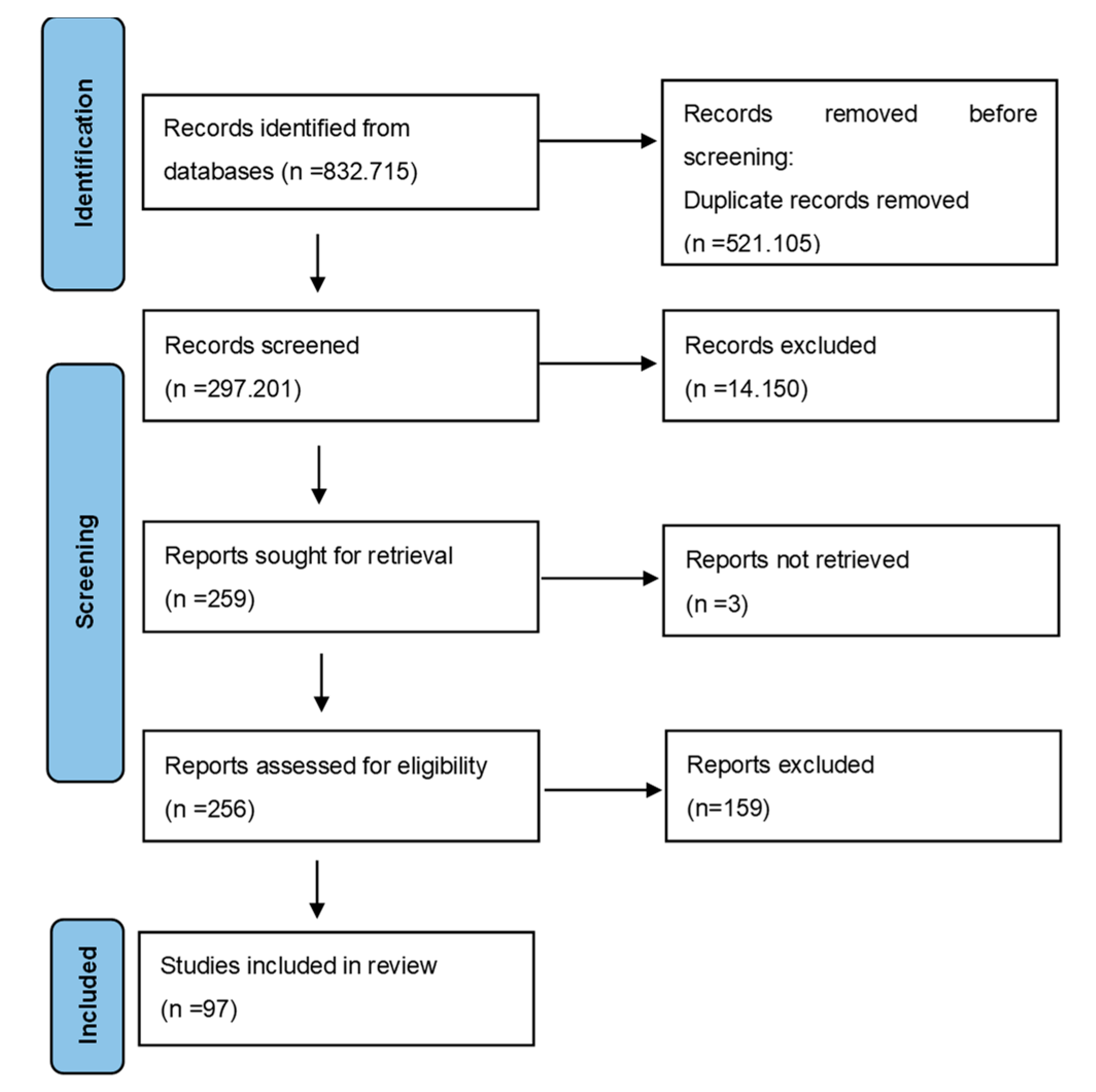
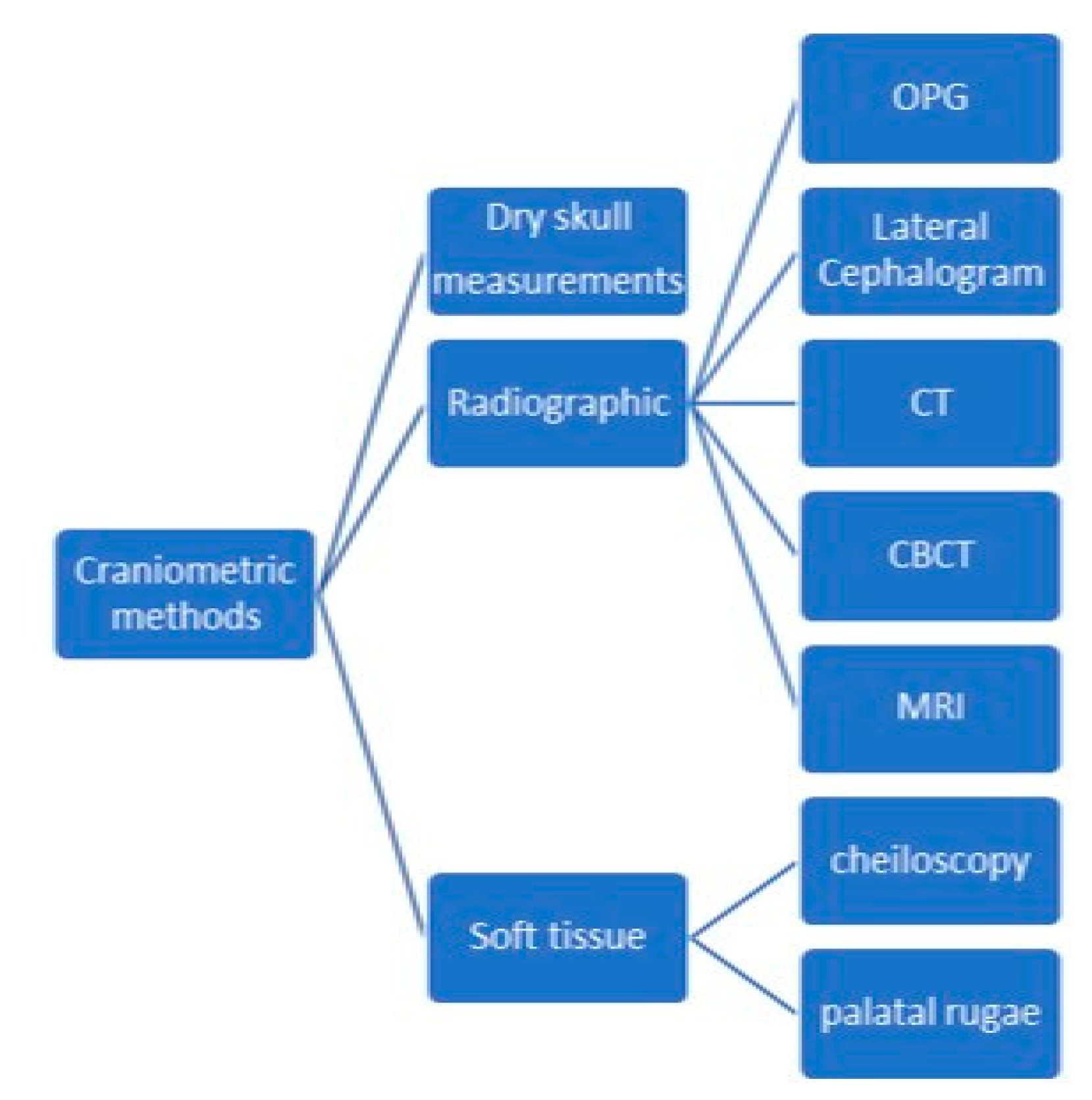
2. Materials and Methods
- Population: all ages, genders and ethnicities included;
- Intervention: cranial and odontological methods for sex estimation;
- Comparison: age range, sample size, method used, sex estimation accuracy;
- Outcome: to determine the methods and find out which ones have the highest sex estimation accuracy.
- Population included: all ethnic groups;
- Patients, autopsy cases and skeletons from archaeological records;
- Original articles;
- With or without abstracts;
- Articles written in English;
- Methodologies based on both skull and teeth assessments;
- Both metric and non-metric methods;
- Both temporary and permanent teeth;
- Study focus relevant to our search question;
- No minimum number of individuals required.
- Studies covering non-human subjects;
- Studies published before 2015;
- Abstracts without full reports;
- Review articles.
- Methodology used;
- Population /ethnicity;
- Sample size;
- Main conclusions;
- Accuracy of the method applied, where available.
3. Results
4. Discussion
4.1. Populations
4.2. Sample Size
4.3. Sex Estimation in Children
4.4. Odontometric Studies
4.4.1. Mandibular Canine Index
4.4.2. Other Teeth Measurements
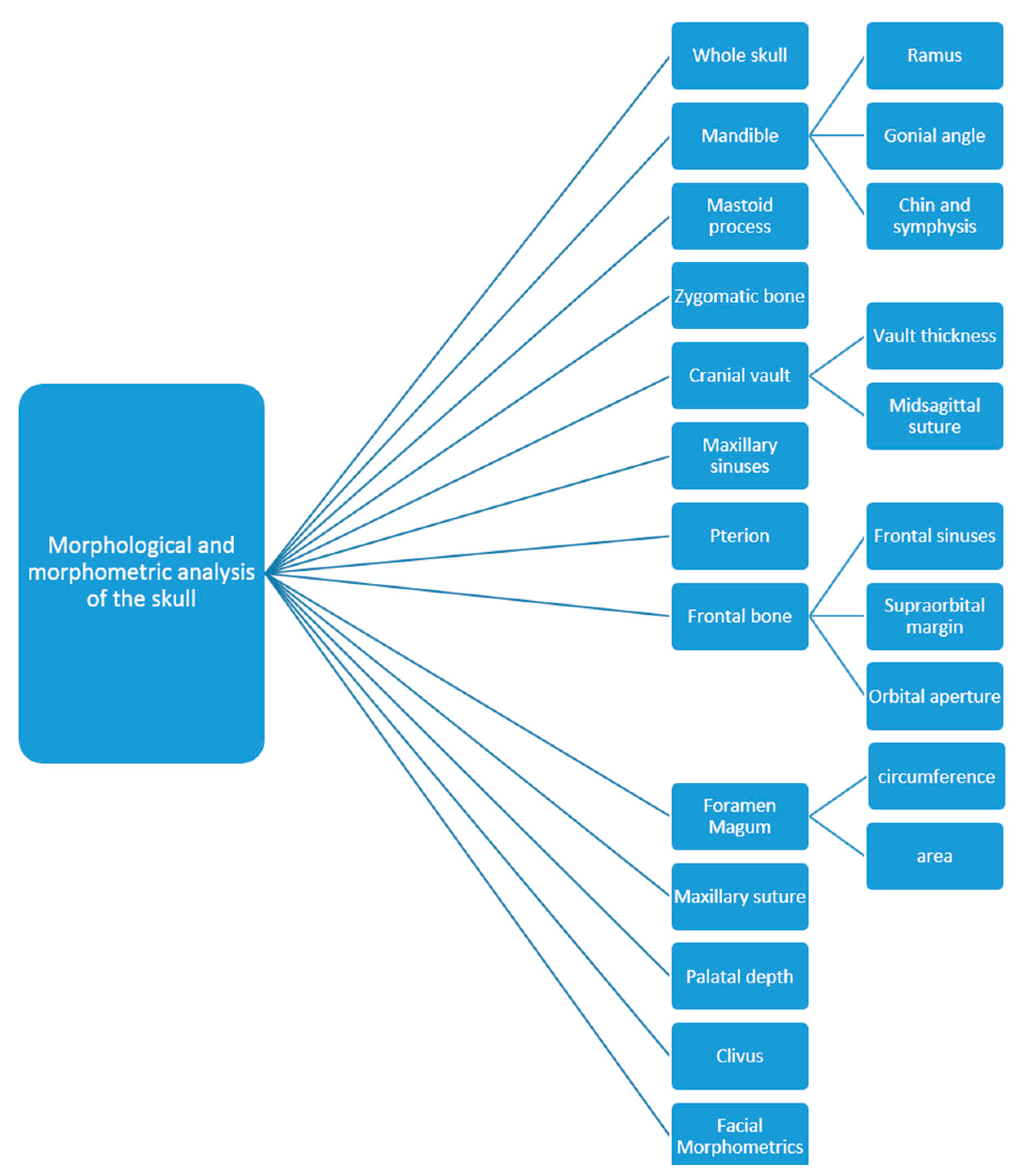
4.5. Morphometrics of the Skull
4.5.1. Dimorphism of the Gonial Angle
4.5.2. Dimorphism of the Ramus Height
4.5.3. Dimorphism of the Chin and Mandibular Symphysis
4.5.4. Dimorphism of the Foramen Magnum
4.5.5. Dimorphism of the Maxillary Sinuses
4.5.6. Dimorphism of the Left Side versus the Right Side of the Skull
4.5.7. Dimorphism of the Mastoid
4.5.8. Dimorphism of the Palate, the Pterion and the Orbital Aperture of the Frontal Bone
4.6. High Sex Prediction Accuracy
4.7. Machine Learning
4.8. Biochemical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Krishan, K.; Chatterjee, P.M.; Kanchan, T.; Kaur, S.; Baryah, N.; Singh, R.K. A review of sex estimation techniques during examination of skeletal remains in forensic anthropology casework. Forensic Sci. Int. 2016, 261, e1–e165. [Google Scholar] [CrossRef] [PubMed]
- Bedalov, A.; Bašić, Ž.; Marelja, I.; Dolić, K.; Bukarica, K.; Missoni, S.; Šlaus, M.; Primorac, D.; Andjelinović, Š.; Kružić, I. Sex estimation of the sternum by automatic image processing of multi-slice computed tomography images in a Croatian population sample: A retrospective study. Croat. Med. J. 2019, 60, 237–245. [Google Scholar] [CrossRef]
- Bubalo, P.; Baković, M.; Tkalčić, M.; Petrovečki, V.; Mayer, D. Acetabular osteometrie standards for sex estimation in contemporary Croatian population. Croat. Med. J. 2019, 60, 221–226. [Google Scholar] [CrossRef]
- Bašić, Ž.; Kružić, I.; Jerković, I.; Andelinovic, D.; Andelinovic, Š. Sex estimation standards for medieval and contemporary Croats. Croat. Med. J. 2017, 58, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Mishra, P.; Shrivastava, K.; Singh, N. An Overview of age, sex and race determination from teeth and skull. Adv. Hum. Biol. 2015, 5, 20–31. [Google Scholar]
- Buonasera, T.; Eerkens, J.; de Flamingh, A.; Engbring, L.; Yip, J.; Li, H.; Haas, R.; DiGiuseppe, D.; Grant, D.; Salemi, M.; et al. A comparison of proteomic, genomic, and osteological methods of archaeological sex estimation. Sci. Rep. 2020, 10, 11897. [Google Scholar] [CrossRef] [PubMed]
- Loreille, O.; Ratnayake, S.; Bazinet, A.L.; Stockwell, T.B.; Sommer, D.D.; Rohland, N.; Mallick, S.; Johnson, P.L.F.; Skoglund, P.; Onorato, A.J.; et al. Biological sexing of a 4000-year-old egyptian mummy head to assess the potential of nuclear DNA recovery from the most damaged and limited forensic specimens. Genes 2018, 9, 135. [Google Scholar] [CrossRef]
- Arigbabu, O.A.; Liao, I.Y.; Abdullah, N.; Mohamad Noor, M.H. Computer vision methods for cranial sex estimation. IPSJ Trans. Comput. Vis. Appl. 2017, 9, 19. [Google Scholar] [CrossRef]
- Gao, H.; Geng, G.; Yang, W. Sex Determination of 3D skull based on a novel unsupervised learning method. Comput. Math. Methods Med. 2018, 2018, 4567267. [Google Scholar] [CrossRef]
- Chovalopoulou, M.E.; Valakos, E.D.; Manolis, S.K. Sex determination by three-dimensional geometric morphometrics of craniofacial form. Anthropol. Anzeiger. 2016, 73, 195–206. [Google Scholar] [CrossRef]
- Agbolade, O.; Nazri, A.; Yaakob, R.; Ghani, A.A.; Cheah, Y.K. Morphometric approach to 3D soft-tissue craniofacial analysis and classification of ethnicity, sex, and age. PLoS ONE 2020, 15, e0228402. [Google Scholar] [CrossRef] [PubMed]
- Cole, C.; Eliopoulos, C.; Zorba, E.; Borrini, M. An anthropometric method for sex determination from the mandible: Test on British medieval skeletal collections. J. Biol. Res. 2017, 90, 30–35. [Google Scholar] [CrossRef][Green Version]
- Tise, M.L.; Spradley, M.K.; Anderson, B.E. Postcranial sex estimation of individuals considered Hispanic. J. Forensic Sci. 2013, 58, S9–S14. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zhou, M.; Zhang, P.; Geng, G.; Liu, X.; Zhang, H. skull sex estimation based on wavelet transform and Fourier transform. Biomed. Res. Int. 2020, 2020, 8608209. [Google Scholar] [CrossRef] [PubMed]
- Nourbakhsh, R.; Razi, S.; Razi, T. Evaluation of relation of dimensional measurement of different anatomic skull structures to determine sexual dimorphism in cone beam CT images of an Iranian population. J. Res. Med. Dent. Sci. 2018, 6, 33–38. [Google Scholar] [CrossRef]
- Sarač-Hadžihalilović, A.; Hojkurić, E.; Musić, M.; Hasanbegović, I.; Ajanović, Z.; Dervišević, L.; Brkić, S. Model “P” in gender prediction based on the mastoid process. Med. Glas. 2020, 17, 279–6284. [Google Scholar] [CrossRef]
- Behl, A.; Grewal, S.; Bajaj, K.; Baweja, P.; Kaur, G.; Kataria, P. Mandibular ramus and gonial angle—Identification tool in age estimation and sex determination: A digital panoramic radiographic study in north indian population. J. Indian Acad. Oral. Med. Radiol. 2020, 32, 31–36. [Google Scholar] [CrossRef]
- Kartheeki, B.; Nayyar, A.S.; Sindhu, Y.U.; Lakshmana, N. Accuracy of mandibular rami measurements in prediction of sex. Arch. Med. Health Sci. 2017, 5, 50–54. [Google Scholar] [CrossRef]
- Sella Tunis, T.; Hershkovitz, I.; May, H.; Vardimon, A.D.; Sarig, R.; Shpack, N. Variation in chin and mandibular symphysis size and shape in males and females: A CT-Based study. Int. J. Environ. Res. Public Health 2020, 17, 4249. [Google Scholar] [CrossRef]
- Veeraraghavan, G.; Lingappa, A.; Shankara, S.P.; Mamatha, G.P.; Sebastian, B.T.; Mujib, A. Determination of sex from tooth pulp tissue. Libyan J. Med. 2010, 5, 5084. [Google Scholar] [CrossRef]
- Zagga, A.; Ahmed, H.; Ismail, S.; Tadros, A. Molecular sex identification of dry human teeth specimens from Sokoto, Northwestern Nigeria. J. Forensic Dent. Sci. 2014, 6, 132–138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Capitaneanu, C.; Willems, G.; Thevissen, P. A systematic review of odontological sex estimation methods. J. Forensic Odontostomatol. 2017, 35, 1–19. [Google Scholar]
- Chowdhury, R.; Singhvi, A.; Bagul, N.; Bhatia, S.; Singh, G.; Goswami, S. Sex determination by amplification of amelogenin gene from dental pulp tissue by polymerase chain reaction. Indian J. Dent. Res. 2018, 29, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Dutta, P.; Bhosale, S.; Singh, R.; Gubrellay, P.; Patil, J.; Sehdev, B.; Bhagat, S.; Bansal, T. Amelogenin gene—The pioneer in gender determination from forensic dental samples. J. Clin. Diagn. Res. 2017, 11, ZC56–ZC59. [Google Scholar] [CrossRef] [PubMed]
- Dash, H.R.; Rawat, N.; Das, S. Alternatives to amelogenin markers for sex determination in humans and their forensic relevance. Mol. Biol. Rep. 2020, 47, 2347–2360. [Google Scholar] [CrossRef]
- Higuchi, R.; Bowman, B.; Freiberger, M.; Ryder, O.A.; Wilson, A.C. DNA sequences from the quagga, an extinct member of the horse family. Nature 1984, 312, 282–284. [Google Scholar] [CrossRef]
- Pääbo, S. Molecular cloning of ancient Egyptian mummy DNA. Nature 1985, 314, 644–645. [Google Scholar] [CrossRef]
- Krings, M.; Stone, A.; Schmitz, R.W.; Krainitzki, H.; Stoneking, M.; Pääbo, S. Neandertal DNA sequences and the origin of modern humans. Cell 1997, 90, 19–30. [Google Scholar] [CrossRef]
- Nesheva, D.V. Aspects of ancient mitochondrial dna analysis in different populations for understanding human evolution. Balk. J. Med. Genet. 2014, 17, 5–14. [Google Scholar] [CrossRef]
- Pilli, E.; Vai, S.; Caruso, M.G.; D’Errico, G.; Berti, A.; Caramelli, D. Neither femur nor tooth: Petrous bone for identifying archaeological bone samples via forensic approach. Forensic Sci. Int. 2018, 283, 144–149. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Aki, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Tabasum, Q.; Sehrawat, J.S.; Talwar, M.K.; Pathak, R.K. Odontometric sex estimation from clinically extracted molar teeth in a North Indian population sample. J. Forensic Dent. Sci. 2017, 9, 176. [Google Scholar] [CrossRef] [PubMed]
- Grewal, D.S.; Khangura, R.K.; Sircar, K.; Tyagi, K.K.; Kaur, G.; David, S. Morphometric analysis of odontometric parameters for gender determination. J. Clin. Diagn. Res. 2017, 11, ZC09–ZC13. [Google Scholar] [CrossRef] [PubMed]
- Girish, H.C.; Murgod, S.; Savita, J.K. Gender determination by odontometric method. J. Forensic Dent. Sci. 2017, 9, 44. [Google Scholar] [CrossRef]
- Priyadharshini, K.; Ambika, M.; Sekar, B.; Mohanbabu, V.; Sabarinath, B.; Pavithra, I. Comparison of cheiloscopy, odontometric, and facial index for sex determination in forensic dentistry. J. Forensic Dent. Sci. 2018, 10, 88–91. [Google Scholar] [CrossRef]
- Pereira, T.; Shetty, S.; Surve, R.; Gotmare, S.; Kamath, P.; Kumar, S. Palatoscopy and odontometrics for sex identification and hereditary pattern analysis in a Navi Mumbai population: A cross-sectional study. J. Oral. Maxillofac. Pathol. 2018, 22, 271–278. [Google Scholar] [CrossRef]
- Dash, K.; Panda, A.; Behura, S.; Ramachandra, S.; Bhuyan, L.; Bandopadhyay, A. Employing dimensional disparity of teeth to establish the gender in Odisha population: A dimorphic study. J. Int. Soc. Prev. Community Dent. 2018, 8, 174–178. [Google Scholar] [CrossRef]
- Phulari, R.S.; Rathore, R.; Talegaon, T.; Jariwala, P. Comparative assessment of maxillary canine index and maxillary first molar dimensions for sex determination in forensic odontology. J. Forensic Dent. Sci. 2017, 9, 110. [Google Scholar] [CrossRef]
- Singh, A.; Bhatia, H.P.; Sood, S.; Sharma, N. Demystifying the mysteries: Sexual dimorphism in primary teeth. J. Clin. Diagn. Res. 2017, 11, ZC110–ZC114. [Google Scholar] [CrossRef]
- Satish, B.N.V.S.; Moolrajani, C.; Basnaker, M.; Kumar, P. Dental sex dimorphism: Using odontometrics and digital jaw radiography. J. Forensic Dent. Sci. 2017, 9, 43. [Google Scholar] [CrossRef]
- Chennoju, S.K.; Ramaswamy, P.; Swathi, E.; Smitha, B.; Sankaran, S. Discriminant canine index—A novel approach in sex determination. Ann. Stomatol. 2015, 6, 43–46. [Google Scholar] [CrossRef]
- Alam, M.K.; Alzarea, B.K.; Ganji, K.K.; Kundi, I.; Patil, S. 3D CBCT human adult odontometrics: Comparative assessment in Saudi, Jordan and Egypt population. Saudi Dent. J. 2019, 31, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Shireen, A.; Ara, S. Odontometric analysis of permanent maxillary first molar in gender determination. J. Forensic Dent. Sci. 2016, 8, 145–149. [Google Scholar] [CrossRef]
- Govindaram, D.; Bharanidharan, R.; Ramya, R.; Rameshkumar, A.; Priyadharsini, N.; Rajkumar, K. Root Length: As a determinant tool of sexual dimorphism in an ethnic Tamil population. J. Forensic Dent. Sci. 2018, 10, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Rajarathnam, B.; David, M.; Indira, A. Mandibular canine dimensions as an aid in gender estimation. J. Forensic Dent. Sci. 2016, 8, 83–89. [Google Scholar] [CrossRef]
- Yadav, A.B.; Angadi, P.V.; Yadav, S.K. Sex assessment efficacy of permanent maxillary first molar cusp dimensions in Indians. Contemp. Clin. Dent. 2015, 6, 489–495. [Google Scholar] [CrossRef]
- Gupta, J.; Daniel, M.J. Crown size and arch width dimension as an indicator in gender determination for a Puducherry population. J. Forensic Dent. Sci. 2016, 8, 120–125. [Google Scholar] [CrossRef]
- Patel, R.A.; Chaudhary, A.R.; Dudhia, B.B.; Macwan, Z.S.; Patel, P.S.; Jani, Y.V. Mandibular canine index: A study for gender determination in Gandhinagar population. J. Forensic Dent. Sci. 2017, 9, 135–143. [Google Scholar] [CrossRef]
- Agrawal, A.; Manjunatha, B.; Dholia, B.; Althomali, Y. Comparison of sexual dimorphism of permanent mandibular canine with mandibular first molar by odontometrics. J. Forensic Dent. Sci. 2015, 7, 238–243. [Google Scholar] [CrossRef]
- Krishnan, R.P.; Thangavelu, R.; Rathnavelu, V.; Narasimhan, M. Gender determination: Role of lip prints, finger prints and mandibular canine index. Exp. Ther. Med. 2016, 11, 2329–2332. [Google Scholar] [CrossRef]
- Couto, D.M.S.E.; Gallassi, N.C.D.; Gomes, S.L.; Ulbricht, V.; Pereira Neto, J.S.; Daruge, E., Jr.; Francesquini, L., Jr. Brazilian’s dental anthropometry: Human identification. J. Forensic Dent. Sci. 2019, 11, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, M.F.; Oliveira Santos, I.; Santos, A.L.; Gonçalves, D. Sample-specific odontometric sex estimation: A method with potential application to burned remains. Sci. Justice 2017, 57, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.; Pereira, M.L.; Gouveia, S.; Tavares, J.N.; Azevedo, Á.; Caldas, I.M. A new approach to sex estimation using the mandibular canine index. Med. Sci. Law. 2016, 56, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Daniele, G.; Matilde, S.S.A.; María, M.; Rafael, R.V.; Milagros, A.M. Sex estimation by tooth dimension in a contemporary Spanish population. Forensic Sci. Int. 2020, 317, 110549. [Google Scholar] [CrossRef] [PubMed]
- Atreya, A.; Shrestha, R.; Tuladhar, L.R.; Nepal, S.; Shrestha, R.; Sah, S.K. Sex Predictability by using mandibular canine index. J. Nepal Health Res. Counc. 2020, 17, 501–505. [Google Scholar] [CrossRef]
- Gamulin, O.; Škrabić, M.; Serec, K.; Par, M.; Baković, M.; Krajačić, M.; Babić, S.D.; Šegedin, N.; Osmani, A.; Vodanović, M. Possibility of human gender recognition using Raman spectra of teeth. Molecules 2021, 26, 3983. [Google Scholar] [CrossRef]
- Rakhshan, V.; Ghorbanyjavadpour, F.; Ashoori, N. Buccolingual and mesiodistal dimensions of the permanent teeth, their diagnostic value for sex identification, and Bolton indices. Biomed. Res. Int. 2022, 2022, 8381436. [Google Scholar] [CrossRef]
- Missier, M.; Samuel, S.; George, A. Facial indices in lateral cephalogram for sex prediction in Chennai population—A semi-novel study. J. Forensic Dent. Sci. 2018, 10, 151–157. [Google Scholar] [CrossRef]
- Perez, I.; Chavez, A.; Ponce, D. Applicability of the Ricketts’ posteroanterior cephalometry for sex determination using logistic regression analysis in Hispano American Peruvians. J. Forensic Dent. Sci. 2016, 8, 111. [Google Scholar] [CrossRef][Green Version]
- Albalawi, A.; Alam, M.; Vundavalli, S.; Ganji, K.; Patil, S. Mandible: An indicator for sex determination—A three-dimensional cone-beam computed tomography study. Contemp. Clin. Dent. 2019, 10, 69–73. [Google Scholar] [CrossRef]
- Vinutha, S.P.; Suresh, V.; Shubha, R. Discriminant function analysis of Foramen Magnum variables in South Indian population: A study of computerised tomographic images. Anat. Res. Int. 2018, 2018, 2056291. [Google Scholar] [CrossRef] [PubMed]
- Nuzzolese, E.; Randolph-Quinney, P.; Randolph-Quinney, J.; Di Vella, G. Geometric morphometric analysis of sexual dimorphism in the mandible from panoramic x-ray images. J. Forensic Odontostomatol. 2019, 37, 35–44. [Google Scholar] [PubMed]
- More, C.B.; Vijayvargiya, R.; Saha, N. Morphometric analysis of mandibular ramus for sex determination on digital orthopantomogram. J. Forensic Dent. Sci. 2017, 9, 1–5. [Google Scholar] [CrossRef]
- Raikar, N.; Meundi, M.; David, C.; Rao, M.; Jogigowda, S. Sexual dimorphism in Foramen Magnum dimensions in the South Indian population: A digital submentovertex radiographic study. J. Forensic Dent. Sci. 2016, 8, 180. [Google Scholar] [CrossRef] [PubMed]
- Rajkumari, S.; Nikitha, K.; Monisha, S.; Nishagrade, S.; Thayumanavan, B.; Murali, B. Role of orthopantamograph in forensic identification: A retrospective study among Chennai Population. J. Pharm. Bioallied. Sci. 2019, 11, S393–S396. [Google Scholar] [CrossRef]
- Poongodi, V.; Kanmani, R.; Anandi, M.S.; Krithika, C.L.; Kannan, A.; Raghuram, P.H. Prediction of age and gender using digital radiographic method: A retrospective study. J. Pharm. Bioallied. Sci. 2015, 7, S504–S508. [Google Scholar] [CrossRef]
- de Queiroz, C.L.; Terada, A.S.S.D.; Dezem, T.U.; Gomes de Araújo, L.; Galo, R.; Oliveira-Santos, C.; Alves da Silva, R.H. Sex determination of adult human maxillary sinuses on panoramic radiographs. Acta Stomatol. Croat. 2016, 50, 215–221. [Google Scholar] [CrossRef]
- Rani, S.U.; Rao, G.V.; Kumar, D.R.; Sravya, T.; Sivaranjani, Y.; Kumar, M.P. Age and gender assessment through three-dimensional morphometric analysis of maxillary sinus using magnetic resonance imaging. J. Forensic Dent. Sci. 2017, 9, 46. [Google Scholar] [CrossRef]
- Ibrahim, A.; Alias, A.; Nor, F.M.; Swarhib, M.; Abu Bakar, S.N.; Das, S.; Abdullah, N.; Noor, M.H.M. Study of sexual dimorphism of Malaysian crania: An important step in identification of the skeletal remains. Anat. Cell Biol. 2017, 50, 86–92. [Google Scholar] [CrossRef]
- Kanjani, V.; Rani, A.; Kanjani, D. Morphometric analysis of the orbital aperture in North Indian Population: A retrospective digital forensic study. Int. J. Appl. Basic Med. Res. 2019, 9, 85–88. [Google Scholar] [CrossRef]
- Bangi, B.B.; Ginjupally, U.; Nadendla, L.K.; Vadla, B. 3D evaluation of maxillary sinus using computed tomography: A sexual dimorphic study. Int. J. Dent. 2017, 2017, 9017078. [Google Scholar] [CrossRef] [PubMed]
- Soman, B.; Sujatha, G.; Lingappa, A. Morphometric evaluation of the frontal sinus in relation to age and gender in subjects residing in Davangere, Karnataka. J. Forensic Dent. Sci. 2016, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Bulut, O.; Freudenstein, N.; Hekimoglu, B.; Gurcan, S. Dilemma of gonial angle in sex determination: Sexually dimorphic or not? Am. J. Forensic Med. Pathol. 2019, 40, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Gamba, T.D.O.; Alves, M.C.; Haiter-Neto, F. Mandibular sexual dimorphism analysis in CBCT scans. J. Forensic Leg. Med. 2016, 38, 106–110. [Google Scholar] [CrossRef]
- Chandra, S.; Chaturvedi, N.; Sah, K.; Sinha, S.; Hamza, A. Craniometric measurement by jug handle view: An aid for gender determination. J. Indian Acad. Oral. Med. Radiol. 2018, 30, 398–401. [Google Scholar] [CrossRef]
- Chaurasia, A.; Patil, R.; Katheriya, G. Radiomorphometeric evaluation of clivus in indian paediatric population visiting a tertiary dental hospital-a cone beam computed tomography study. J. Clin. Diagn. Res. 2018, 12, ZC05–ZC08. [Google Scholar] [CrossRef]
- Prabhat, M.; Rai, S.; Kaur, M.; Prabhat, K.; Bhatnagar, P.; Panjwani, S. Computed tomography based forensic gender determination by measuring the size and volume of the maxillary sinuses. J. Forensic Dent. Sci. 2016, 8, 40–46. [Google Scholar] [CrossRef]
- Belaldavar, C.; Acharya, A.B.; Angadi, P. Sex estimation in Indians by digital analysis of the gonial angle on lateral cephalographs. J. Forensic Odontostomatol. 2019, 37, 45–50. [Google Scholar]
- Shireen, A.; Goel, S.; Ahmed, I.; Sabeh, A.; Mahmoud, W. Radiomorphometric evaluation of the frontal sinus in relation to age and gender in Saudi population. J. Int. Soc. Prev. Community Dent. 2019, 9, 584–596. [Google Scholar] [CrossRef]
- Sambhana, S.; Sanghvi, P.; Mohammed, R.; Shanta, P.; Thetay, A.R.; Chaudhary, V. Assessment of sexual dimorphism using digital orthopantomographs in South Indians. J. Forensic Dent. Sci. 2016, 8, 180. [Google Scholar] [CrossRef]
- Damera, A.; Mohanalakhsmi, J.; Yellarthi, P.; Rezwana, B. Radiographic evaluation of mandibular ramus for gender estimation: Retrospective study. J. Forensic Dent. Sci. 2016, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Esmaeilyfard, R.; Paknahad, M.; Dokohaki, S. Sex classification of first molar teeth in cone beam computed tomography images using data mining. Forensic Sci. Int. 2021, 318, 110633. [Google Scholar] [CrossRef] [PubMed]
- Paknahad, M.; Dokohaki, S.; Khojastepour, L.; Shahidi, S.; Haghnegahdar, A. A Radio-Odontometric analysis of sexual dimorphism in first molars using cone-beam computed tomography. Am. J. Forensic Med. Pathol. 2022, 43, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.; Singh, S.; Thomas, A.; Chauhan, N. Geometric morphometric analysis for sex determination using lateral cephalograms in Indian population: A preliminary study. J. Oral Maxillofac. Pathol. 2021, 25, 364. [Google Scholar] [CrossRef]
- Suzuki, K.; Nakano, H.; Inoue, K.; Nakajima, Y.; Mizobuchi, S.; Omori, M.; Kato-Kogoe, N.; Mishima, K.; Ueno, T. Examination of new parameters for sex determination of mandible using Japanese computer tomography data. Dentomaxillofac. Radiol. 2020, 49, 20190282. [Google Scholar] [CrossRef]
- Sinthubua, A.; Ruengdit, S.; Das, S.; Mahakkanukrauh, P. A new method for sex estimation from maxillary suture length in a Thai population. Anat. Cell Biol. 2017, 50, 261–264. [Google Scholar] [CrossRef]
- De Boer, H.H.; Van der Merwe, A.E.; Soerdjbalie-Maikoe, V.V. Human cranial vault thickness in a contemporary sample of 1097 autopsy cases: Relation to body weight, stature, age, sex and ancestry. Int. J. Legal Med. 2016, 130, 1371–1377. [Google Scholar] [CrossRef]
- Mahakkanukrauh, P.; Sinthubua, A.; Prasitwattanaseree, S.; Ruengdit, S.; Singsuwan, P.; Praneatpolgrang, S.; Duangto, P. Craniometric study for sex determination in a Thai population. Anat. Cell Biol. 2015, 48, 275–283. [Google Scholar] [CrossRef]
- Mankapure, P.K.; Barpande, S.R.; Bhavthankar, J.D. Evaluation of sexual dimorphism in arch depth and palatal depth in 500 young adults of Marathwada region, India. J. Forensic Dent. Sci. 2017, 9, 153–156. [Google Scholar] [CrossRef]
- Singh, A.; Sreedhar, G.; George, J.; Shukla, A.; Vashishta, V.; Negi, M.P.S. Anthropometric study using craniofacial features to determine gender in Lucknow population. J. Forensic Dent. Sci. 2017, 9, 120–124. [Google Scholar] [CrossRef]
- Kamath, V.G.; Asif, M.; Shetty, R.; Avadhani, R. Binary logistic regression analysis of Foramen Magnum dimensions for sex determination. Anat. Res. Int. 2015, 2015, 459428. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, A.G.; Tashtoush, A.A.; Alshboul, O.A.; Allouh, M.Z.; Altarifi, A.A. Morphometric study of the hard palate and its relevance to dental and forensic sciences. Int. J. Dent. 2019, 2019, 1687345. [Google Scholar] [CrossRef] [PubMed]
- Bertsatos, A.; Papageorgopoulou, C.; Valakos, E.; Chovalopoulou, M.E. Investigating the sex-related geometric variation of the human cranium. Int. J. Legal Med. 2018, 132, 1505–1514. [Google Scholar] [CrossRef]
- Chovalopoulou, M.E.; Valakos, E.D.; Manolis, S.K. Sex determination by three-dimensional geometric morphometrics of the vault and midsagittal curve of the neurocranium in a modern Greek population sample. HOMO—J. Comp. Hum. Biol. 2016, 67, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Small, C.; Schepartz, L.; Hemingway, J.; Brits, D. Three-dimensionally derived interlandmark distances for sex estimation in intact and fragmentary crania. Forensic Sci. Int. 2018, 287, 127–135. [Google Scholar] [CrossRef]
- Bhayya, H.; Avinash Tejasvi, M.; Jayalakshmi, B.; Reddy, M.M. Craniometric assessment of gender using mastoid process. J. Indian Acad. Oral Med. Radiol. 2018, 30, 52–57. [Google Scholar] [CrossRef]
- Musilová, B.; Dupej, J.; Brůžek, J.; Bejdová, Š.; Velemínská, J. Sex and ancestry related differences between two Central European populations determined using exocranial meshes. Forensic Sci. Int. 2019, 297, 364–369. [Google Scholar] [CrossRef]
- Uabundit, N.; Chaiyamoon, A.; Iamsaard, S.; Yurasakpong, L.; Nantasenamat, C.; Suwannakhan, A.; Phunchago, N. Classification and morphometric features of pterion in Thai population with potential sex prediction. Medicina 2021, 57, 1282. [Google Scholar] [CrossRef]
- Bertsatos, A.; Chovalopoulou, M.E.; Brůžek, J.; Bejdová, Š. Advanced procedures for skull sex estimation using sexually dimorphic morphometric features. Int. J. Legal Med. 2020, 134, 1927–1937. [Google Scholar] [CrossRef]
- Inskip, S.; Scheib, C.L.; Wohns, A.W.; Ge, X.; Kivisild, T.; Robb, J. Evaluating macroscopic sex estimation methods using genetically sexed archaeological material: The medieval skeletal collection from St John’s Divinity School, Cambridge. Am. J. Phys. Anthropol. 2019, 168, 340–351. [Google Scholar] [CrossRef]
- Srivastava, M.; Tripathi, S.; Astekar, M.; Singal, D.; Srivastava, A.; Vashisth, P. Sex determination from mesiodens of Indian children by amelogenin gene. J. Forensic Dent. Sci. 2017, 9, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Kulstein, G.; Hadrys, T.; Wiegand, P. As solid as a rock—Comparison of CE- and MPS-based analyses of the petrosal bone as a source of DNA for forensic identification of challenging cranial bones. Int. J. Legal Med. 2018, 132, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.; Cannet, C.; Zvénigorosky, V.; Geraut, A.; Koch, G.; Delabarde, T.; Ludes, B.; Raul, J.S.; Keyser, C. The petrous bone: Ideal substrate in legal medicine? Forensic Sci. Int. Genet. 2020, 47, 102305. [Google Scholar] [CrossRef] [PubMed]
- Froment, C.; Hourset, M.; Sáenz-Oyhéréguy, N.; Mouton-Barbosa, E.; Willmann, C.; Zanolli, C.; Esclassan, R.; Donat, R.; Thèves, C.; Burlet-Schiltz, O.; et al. Analysis of 5000 year-old human teeth using optimized large-scale and targeted proteomics approaches for detection of sex-specific peptides. J. Proteom. 2020, 211, 103548. [Google Scholar] [CrossRef] [PubMed]
- Ziganshin, R.H.; Berezina, N.Y.; Alexandrov, P.L.; Ryabinin, V.V.; Buzhilova, A.P. Optimization of method for human sex determination using peptidome analysis of teeth enamel from teeth of different biological generation, archeological age, and degrees of taphonomic preservation. Biochemistry 2020, 85, 614–622. [Google Scholar] [CrossRef]
- Gowland, R.; Stewart, N.A.; Crowder, K.D.; Hodson, C.; Shaw, H.; Gron, K.J.; Montgomery, J. Sex estimation of teeth at different developmental stages using dimorphic enamel peptide analysis. Am. J. Phys. Anthropol. 2021, 174, 859–869. [Google Scholar] [CrossRef]
- Noble, J.; Cardini, A.; Flavel, A.; Franklin, D. Geometric morphometrics on juvenile crania: Exploring age and sex variation in an Australian population. Forensic Sci. Int. 2019, 294, 57–68. [Google Scholar] [CrossRef]
| No. | Reference | Methodology | Population | No. of Cases/Age | Main Conclusions | Sex Estimation Accuracy |
|---|---|---|---|---|---|---|
| 1 | [32] | Linear and diagonal dimensions recorded at both crown and cementoenamel junction levels of extracted molars | Northwest Indian | 73 males 57 females | The calculated index of sexual dimorphism was higher in lower molars than in the upper molars | Max 70% |
| 2 | [33] | Four odontometric parameters: ICW, IPW, AL and CW, measured directly with the subject | Indian | 100 males 100 females | Maxillary parameters exhibited higher mean values in males compared to females | |
| 3 | [34] | MD and BL dimensions of all upper teeth | Indian | 250 males 250 females | The MD and BL dimensions were statistically significant different between males and females | 99.8% using stepwise discriminant functions |
| 4 | [35] | Lip prints; Mandibular Canine Index; Facial Index | Indian | 50 males 50 females | Type II pattern in lips most common No significant difference in odontometric analysis | |
| 5 | [36] | Maxillary impressions; palatine rugae; MD canines; ICW; MD and BL of upper molars | Indian | 60 males 60 females 20 families of 4 members | Females—more wavy rugaes Males—all measured indexes were higher than in females | |
| 6 | [37] | MD, BL measurements of 28 teeth | Indian | 100 males 100 females 18–25 years old | Larger dimensions of teeth in males when compared to females | |
| 7 | [38] | Maxillary Canine Index and maxillary first molar dimensions | Indian | 100 males 100 females 15–25 years old | BL dimension of maxillary first molar is a more reliable indicator for gender determination | |
| 8 | [39] | MD and BL dimensions of upper and lower temporary teeth | Indian | 250 males 250 females 3–5 years old | Boys generally had larger crown diameters than girls | |
| 9 | [40] | Maximum ramus height, bigonion width and bicondylar breadth in OPG MD of upper central incisors, canines | Indian | 100 males 100 females 18–30 years old | Ramus height—most dimorphic Permanent maxillary central incisor—more dimorphic than the maxillary canines | |
| 10 | [41] | MD—left mandibular canine | Indian | 60 males 60 females 15–40 years old | Increased MD diameter in males | 72.5% |
| 11 | [42] | CBCT and odontometrics of 28 teeth | Jordanian, Saudi, Egyptian | 159 males 93 females 20–45 years old | Odontometric differences of 28 teeth between gender and among Saudi, Jordanian and Egyptian populations were insignificant (p > 0.05) | |
| 12 | [43] | MD and BL of permanent upper first molar | Indian | 300 males 300 females 17–25 years old | The differences between males and females in MD and BL dimensions measured were statistically significant (p < 0.05) | |
| 13 | [44] | OPG—root length observed in all permanent teeth | Indian | 500 males 500 females 21–60 years old | Sexual dimorphism in root length was observed in 13, 14, 15, 16, 23, 26, 33, 36, 43 and 46 (mesial) Most dimorphic teeth were canines | |
| 14 | [45] | MD of left and right canine, intercanine distance, MCI | Indian | 100 males 100 females 18–25 years old | Significant sexual dimorphism of mandibular canines | 73% |
| 15 | [46] | MD and BL of upper first molar | Indian | 149 males 151 females 18–30 years old | BL crown dimension and the hypocone (distolingual) cusp showed the highest sexual dimorphism | 64.3% |
| 16 | [47] | MCI and Pont Index | Indian | 53 males 53 females 18–25 years old | MCI and Pont’s Index showed significant sexual dimorphism | Standard right MCI could predict sex accurately at 75.4% Standard left MCI could predict sex accurately at 66.9% |
| 17 | [48] | MD diameter of permanent mandibular right and left canines, as well as mandibular intercanine distance | Indian | 200 males 200 females 20–40 years old | The MD crown width of the permanent mandibular right and left canines, as well as the mandibular intercanine distance of the males, was found to be larger in size | 78.8% |
| 18 | [49] | MD and BL diameter of mandibular canine and mandibular first molar—study casts | Indian | 50 males 50 females 17–25 years old | Sexual dimorphism can be predicted by measuring mesiodistal dimension of mandibular canine and mandibular first molar | |
| 19 | [50] | Lip prints Finger prints MCI | Indian | 25 males 25 females 18–25 years old | MCI was not found to be a significant indicator of gender Lip prints exhibited sexual dimorphism | |
| 20 | [51] | Dental measurements on upper right teeth | Brazilian | 100 males 100 females 18–30 years old | Dental measurements are useful tools for sex determination, and the canine measurements showed a proportional correlation with stature | 70.5% |
| 21 | [52] | Experimentally burned teeth at 400 °C, 700 °C and 900 °C | Portuguese | The perimeter at the CEJ and the combined measurements of the MD and BL diameters, at the same level, were quite promising in the post-burning analysis | ˃80% | |
| 22 | [53] | MCI measured from dental casts | Portuguese | 50 males 70 females 16–30 years old | MCI may not be particularly useful in sex prediction | 64.2% |
| 23 | [54] | MD dimension of teeth from study castsPCA from the logarithm of the dental widths | Spanish | 120 patients mean age: 14.48 ± 2.78 (males) mean age: 14.71 ± 2.69 (females) | Tooth dimension can be a considered a valuable complementary tool in sex determination for Spanish population | 76.2% |
| 24 | [55] | MD widths of mandibular canines ICW From casts | Nepal | 40 male 40 female | Sex predictability by using MCI showed poor sex predictability and should be used cautiously in Nepalese population | 57.5–62.5% |
| 25 | [56] | Raman spectroscopy of teeth PCA of teeth from anthropological collection | Croatian | 55 teeth 11–76 years old | The accuracy of classification models depends both on the tooth type (molar and premolar) and recording site (anatomical neck and apex) on the tooth | ˃90% |
| 26 | [57] | MD and BL dimensions of permanent teeth measured from dental casts and radiographs | Iranian | 74 male 257 female 12–35 years old | Sex dimorphism is very strong in the dentition Ageing significantly reduces measurements Mandibular canines were the most dimorphic teethBolton ratio was not affected by sex |
| No. | Reference | Methodology | Population | No. of Cases/Age | Main Conclusions | Sex Estimation Accuracy |
|---|---|---|---|---|---|---|
| 1 | [42] | CBCT and odontometrics of 28 teeth | Jordanian, Saudi, Egyptian | 159 males 93 females, 20–45 years old | Odontometric differences of 28 teeth between gender and among Saudi, Jordanian and Egyptian populations were insignificant (p > 0.05) | |
| 2 | [58] | A total of 99 cephalometric variables were compared, subjected to statistical analysis and tested for significance using the t-test | Dravidian | 125 males 125 females 25–40 years old | Twenty-four variables showed statistical significance | 52—78% |
| 3 | [59] | PA cephalometric analysis | Hispano-American Peruvians | 1525 patients 5–44 years old | Significant differences between sexes Males, on average, are larger and have increased muscle attachment in their skeletons than females | 63–75% |
| 4 | [60] | Mandible morphometry on CBCT scans | Korean | 96 males 104 females 18–60 years old | Gender can be accurately predicted using this technique | 67% |
| 5 | [61] | CT scans of FM | Indian | 110 males 90 females | Shape and dimensions of FM should be taken into consideration during surgery involving the craniovertebral junction and in forensic and anthropological investigations | 65% |
| 6 | [62] | Morphometric analysis of the mandible with OPG | Italian | 50 males 20–68 years 50 females 21–62 years old | Mandible exhibits great sexual dimorphism | 92.5% |
| 7 | [63] | Morphometric analysis with OPG | Indian | 500 males 500 females 21–60 years old | 69% | |
| 8 | [64] | Submentovertex radiography | South Indian | 75 males 75 females | Circumference in FM was the best sex indicator | 67.3% |
| 9 | [65] | OPG measurements of the mandible | Chennai | 150 OPGs 3–70 years old, divided into seven groups | Highly statistically significant differences between genders | |
| 10 | [66] | OPG measurements of the mandible | Indian | 113 males 87 females 4–75 years old | Significant differences between all the parameters: gonial angle, height and width of the ramus of mandible | |
| 11 | [67] | Maxillary sinuses measured with OPG | Brazilian | 32 males 32 females ˃20 years old | There were differences between the mean values of the maxillary sinus dimensions evaluated for both sexes However, when the values were between 27 mm and 31 mm for height, and 44 mm and 48 mm for width, it was impossible to determine the sex | |
| 12 | [68] | Maxillary sinus measurements with MRI scan | Indian | 30 males 30 females 21–73 years old | Sexual dimorphism was shown by the volume of the maxillary sinuses on the left side | |
| 13 | [69] | CT scans of skulls | Malaysian | 45 males 42 females 18–75 years old | Males showed higher values for all the parameters than females, except for the left orbital height | 85.1% |
| 14 | [70] | Orbital aperture dimension with PA cephalogram | North Indian | 250 males 250 females 20–50 years old | All the linear measurements, such as orbital height, orbital width and interorbital distance, were significantly greater in males than females | 84.8% |
| 15 | [71] | Maxillary sinuses measured with CT scans | Indian | 50 males 50 females ˃20 years old | Volume of left maxillary sinus of males is larger than that of females | 84% in males 92% in females |
| 16 | [19] | Chin and mandibular symphysis measurements with CT scans | Caucasian | 203 males 216 females ˃18 years old Age-matched samples | Chin width (the frontal view) was found to be a sexually selected trait; it can be considered as a parameter for sex determination The chin was found to be a more heterogeneous anatomical structure than symphysis and it was sexually more dismorphic | |
| 17 | [72] | Frontal sinus measured with PA cephalograms | Indian | 100 males 100 females ≥14 years old | It was found that the left width and area are most suitable for gender determination | |
| 18 | [73] | CT scans of the gonial angle | Turkish | 150 males 150 females Three age groups 20–80 years old | Males showed slightly smaller gonial angle values than those of females in all age groups Gonial angle is not a particularly good indicator to identify the sex from the cranium | |
| 19 | [74] | Mandibular CBCT scans | Brazilian | 74 males 86 females 18–60 years old | 95.1% | |
| 20 | [75] | Bi-zygomatic distance and intervolt distance measured with”jug handle” radiograph | Indian | 30 males 30 females 18–25 years old | Bizygomatic distance is a more reliable parameter to determine gender as compared to intervault distance | |
| 21 | [17] | Mandibular ramus and gonial angle measurements with OPG | North Indian | 200 males 200 females 10–40 years old | The mandibular ramus showed a high sexual dimorphism, with condylar and coronoid ramus heights as the most significant predictor for age and sex estimation Gonial angle can only be used as an additional tool | |
| 22 | [18] | Mandibular rami measurements with OPG | South Indian | 229 males 271 females 20–60 years old | Condylar height/maximum ramus height was found to be the best sex predictor | 80.4% |
| 23 | [15] | CBCT measurements of the skull | Iranian | 51 males 51 females 46.65 ± 12.72 years old | Highest accuracy related to mandible bone—89% Lowest accuracy related to FM—71% | |
| 24 | [76] | Clivus measurements with CBCT scan | Indian | 76 males 74 females 6–17 years old | The clivus length was statistically significant The clivus length was greater in male population | |
| 25 | [44] | OPG—root length observed in all permanent teeth | Indian | 500 males 500 females 21–60 years old | Sexual dimorphism in root length was observed in 13, 14, 15, 16, 23, 26, 33, 36, 43 and 46 (mesial); The most dimorphic teeth are canines | |
| 26 | [77] | CT images used to measure the mediolateral, superoinferior and anteroposterior dimensions and the volume of the maxillary sinuses | Indian | 15 males 15 females | 83.3% | |
| 27 | [78] | Lateral cephalograms—gonial angle | Indian | 149 males 155 females 18–30 years old | 56.3% | |
| 28 | [79] | Morphometric evaluation of frontal sinus with PA radiographs | Saudi Arabian | 200 males 200 females 14–70 years old | Right width and left width are most suited regressors for sex determination | 67.70–95.90% |
| 29 | [80] | OPG—ten mandibular variables were measured | South Indian | 192 males 192 females | Coronoid height was the single best parameter, providing an accuracy of 74.1% | Overall accuracy: 75.8% |
| 30 | [81] | Measurements of the mandibular ramus: maximum ramus breadth, maximum ramus height and coronoid height using Planmeca ProMax | Indian | 80 OPGs | Greatest sexual dimorphism was noticed in the maximum ramus height | Prediction rate using all five variables: 83.8% |
| 31 | [82] | Linear tooth measurements with CBCT machine learning: naive Bayesian, random forest, support vector machine | Iranian | 245 males 240 females | Naive Bayesian—highest accuracy for sex classification | Average accuracy: 92.31% |
| 32 | [83] | Roof, height and floor of pulp chamber Marginal enamel/dentine thickness Tooth width and crown length CBCT | Iranian | 100 males 100 females Mean age: 21.28 ± 2.47 | Maxillary first molars were more dimorphic than mandibular teeth Mesio-distal variables were more dimorphic than bucco-lingual ones | Highest accuracy: 84% |
| 33 | [84] | PCA with lateral cephalograms | Indian | 54 males 51 females | Sex was clearly associated with occlusion | Over 96% variation between male and female |
| 34 | [85] | PCA of mandible surface CT scans | Japanese | 23 males 22 females Mean age: 43.1 ± 14.6 | Significant differences between male and female, the mandibular branch of males was larger than that of females, and the mandible angle was overhanging outside |
| No. | Reference | Methodology | Population | No. of Cases/Age | Main Conclusions | Sex Estimation Accuracy |
|---|---|---|---|---|---|---|
| 1 | [9] | Morphological features from the 3D skull MKDSIF-FCM algorithm | Han Chinese | Accuracy improvements of nearly 8.6%, 3.5% and 2.2% compared to other algorithms | ||
| 2 | [14] | Supraorbital margin and frontal bone quantified by wavelet transform and Fourier transform | Han Chinese | 73 males 60 females 22–28 years old | Compared with the traditional methods, the correct rate is higher | 90.9% for males 94.4% for females |
| 3 | [86] | Photographs of maxillary sutures—dry skulls | Thai | 96 males 94 females | Maxillary suture length can be applied for sex estimation | 79.47% |
| 4 | [87] | Cranial vault thickness—autopsy cases | Caucasion Negroid Mongoloid | 1097 cases 103 ˂ 19 years old 994 ˃ 19 years old | Females appear to have a larger frontal cranial thickness Cranial vault thickness generally cannot be used as an indicator for sex | |
| 5 | [88] | Various craniometric measurements on dry skulls | Thai | 100 males 36–96 years old 100 females 15–93 years old | Mastoid length (right and left), nasal height, FM length, cranial base length, bizygomatic breadth, FM breadth, biauricular breadth, upper facial breadth, basion-nasospinale length, maximum cranial length and biorbital breadth expressed significant sexual dimorphism | 88–92.2% |
| 6 | [89] | Maxillary arch depth and palatal depth measured from dental casts | Indian | 250 males 250 females 17–25 years | Only mean maxillary arch depth values were found to be statistically significantly different | |
| 7 | [90] | Anthropometric measurements of patients | Indian | 50 males 50 females 30–40 years old | Significantly higher facial height, pronasale-to-menton distance and interzygomatic width in males as compared to females | |
| 8 | [91] | Measurements of FM in skulls | Indian | 41 males 31 females ˃18 years old | Predictability of area was the highest:70.3% | |
| 9 | [92] | Palate measurements from dental casts | Jordanian | 66 males 84 females 18–50 years old 75 males 75 females 6–12 years old | The palatal dimensions that reflect the palatal size were significantly higher in males than in females | |
| 10 | [11] | 3D soft tissue craniofacial analysis | British and Irish white Europeans | 102 British males 27 Irish males 132 British females 31 Irish females Below 13–over 50 years old | The magnitude of dimorphism in sex is revealed in facial, nasal and crania measurements Males are relatively larger than females, especially in the mouth and nasal regions | |
| 11 | [93] | Skull measurements | Greek | 176 individuals | Multivariate combinations: ˃95% | |
| 12 | [94] | Vault and midsagittal curve of the neurocranium measurements | Greek | 94 males 82 females | In contrast to the midsagittal curve of the neurocranium, the shape of the cranial vault can be used as an indicator of sex in the modern Greek population | 89.2% |
| 13 | [95] | Novel interlandmark distance measures across six regions of the cranium (dry skulls) | South Africans of European descent (white) | 114 males 113 females | 74–88.2% | |
| 14 | [10] | 3D geometric morphometric measurements of the cranium (dry skulls) | Greek | 94 males 82 females | There are shape differences between the sexes in the upper-face and the orbits Size is significant for sexual dimorphism in the upper-face region | |
| 15 | [16] | Mastoid process measurements from dry skulls | Bosnian | 50 males 47–71 year old 50 females 43–76 years old | There was a statistically significant difference between the genders on the basis of the mastoid process | |
| 16 | [96] | Mastoid measurements from dry skull | Indian | 25 males 25 females ˃18 years old | The mastoid process is a good indicator for sex determination | 83% |
| 17 | [12] | Mandible measurements from dry skulls | British | 40 males 36 females | Mandibular metrics are good predictors for sex determination | 77.3% |
| 18 | [8] | Computer vision cranial measurements | Malaysian | 54 males 46 females 5–85 years old | CV methods are suitable for sex determination | 78.2–86.2% |
| 19 | [97] | Virtual method—evaluating the exocranial surface | Czech | 208 individuals | Highest accuracy for Czech population—96.2% Highest accuracy for inter-populational differences—92.8% | 91.8% |
| 20 | [98] | Pterion surface evaluated by machine learning | Thai | 100 skulls | PMP and PI distances were significantly longer in males | 80.7% |
| 21 | [99] | Fully automated method with 3D models | CzechGreek | 170 Czech 156 Greek | The method is efficient in estimating sex from cranial remains | Population-specific accuracy: 78.5–96.7% Population generic accuracy: 71.7–90.8% |
| No. | Reference | Methodology | Population | No. of Cases/Age | Main Conclusions | Sex Estimation Accuracy |
|---|---|---|---|---|---|---|
| 1 | [4] | Various anthropological procedures of the skull and skeleton aDNA analysis | Croatian | 84 adult medievalskeletons | For the mandible, the only measurement that showed sexual dimorphism was mandibular body height | Seven multivariate and five univariate discriminant functions for sex estimation with overall accuracy rates above 80% |
| 2 | [100] | Os coxae Skull Os coxae + skull | 66 individuals 13–16th century | The preauricular sulcus, frontal bossing and arc compose should be reconsidered as appropriate traits for sex estimation | The combined estimate (97.7%) outperformed the os coxae-only estimate (95.7%), which outperformed the skull-only estimate (90.4%) |
| No. | Reference | Methodology | No. of Cases | Main Conclusions | Sex Estimation Accuracy |
|---|---|---|---|---|---|
| 1 | [23] | PCR analysis from dental pulp Amelogenin gene analysis Teeth subjected to different conditions | 130 teeth | Teeth buried in soil yielded least amount of DNA over a period of time and no DNA could be obtained at high temperatures | |
| 2 | [101] | PCR analysis | Eight mesiodens teeth | Sex identification through DNA was possible in six out of eight cases | |
| 3 | [24] | DNA—amelogenin analysis | 50 teeth subjected to different conditions, including extreme temperatures of 1050 °C | Pulpal tissue and degenerating odontoblastic processes provided enough DNA for sex identification | 100% retrieval of DNA along with gender determination |
| 4 | [30] | DNA analysis of ancient petrous bone compared to femur and tooth | 39 skeletal element from 13 individuals | Petrous bone is the best skeletal element with regard to DNA conservation in ancient remains | |
| 5 | [102] | Capillary electrophoresis (CE)-and massively parallel sequencing (MPS)-based analysis of petrous bone | Different sections of eight unknown cranial bones and additionally—where available—other skeletal elements | Short tandem repeat (STR) typing from the petrous bones leads to reportable profiles in all individuals | |
| 6 | [103] | DNA extraction from petrous bone and tooth | 50 skeletal remains | More likely to obtain a complete STR profile from petrous bone material | |
| 7 | [104] | MS proteomics on 5000 year old teeth | 11 Neolitic human teeth | The method represents an alternative for sex estimation when DNA is not exploaitable | The targeted proteomics assay allowed the confirmation of the sex in all the samples |
| 8 | [105] | Enamel peptide analysis by liquid chromatography and mass spectrometry without destruction of analyzed teeth | 8 permanent, 15 deciduous teeth from fossil remains | Analysis of teeth enamel peptidome is sutable for sex determination of human fossil remains | |
| 9 | [106] | Enamel peptides | 43 teeth from 29 nonadult individuals 40 gestational weeks to 19 years old from archaeological sites in England | The method enables forensic identification of nonadult human remains, including perinates | 28 out of 29 individuals were identified |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beschiu, L.M.; Ardelean, L.C.; Tigmeanu, C.V.; Rusu, L.-C. Cranial and Odontological Methods for Sex Estimation—A Scoping Review. Medicina 2022, 58, 1273. https://doi.org/10.3390/medicina58091273
Beschiu LM, Ardelean LC, Tigmeanu CV, Rusu L-C. Cranial and Odontological Methods for Sex Estimation—A Scoping Review. Medicina. 2022; 58(9):1273. https://doi.org/10.3390/medicina58091273
Chicago/Turabian StyleBeschiu, Laura Maria, Lavinia Cosmina Ardelean, Codruta Victoria Tigmeanu, and Laura-Cristina Rusu. 2022. "Cranial and Odontological Methods for Sex Estimation—A Scoping Review" Medicina 58, no. 9: 1273. https://doi.org/10.3390/medicina58091273
APA StyleBeschiu, L. M., Ardelean, L. C., Tigmeanu, C. V., & Rusu, L.-C. (2022). Cranial and Odontological Methods for Sex Estimation—A Scoping Review. Medicina, 58(9), 1273. https://doi.org/10.3390/medicina58091273









