Pulmonary Coccidioidomycosis: A Case Report and Literature Review
Abstract
:1. Introduction
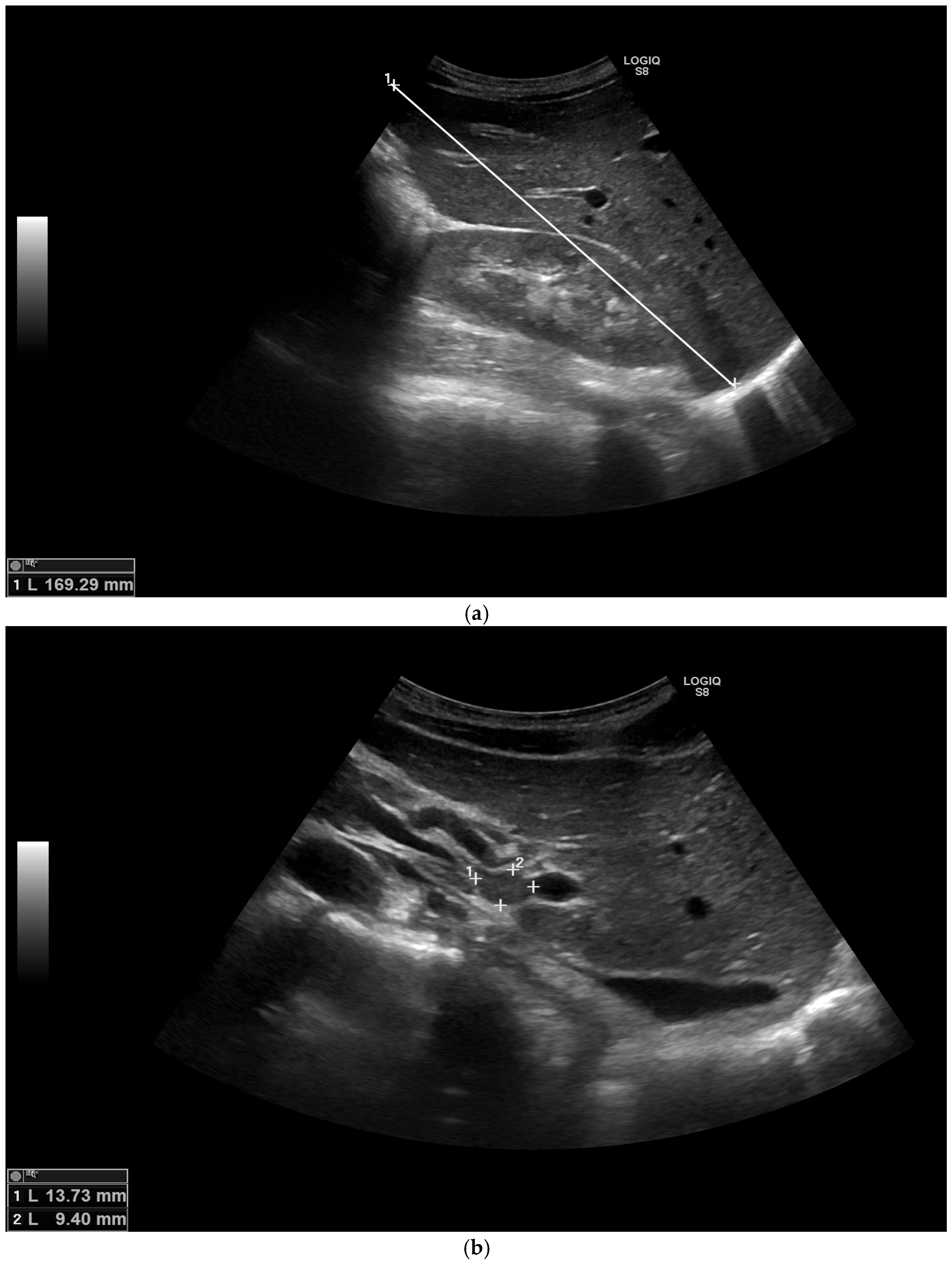
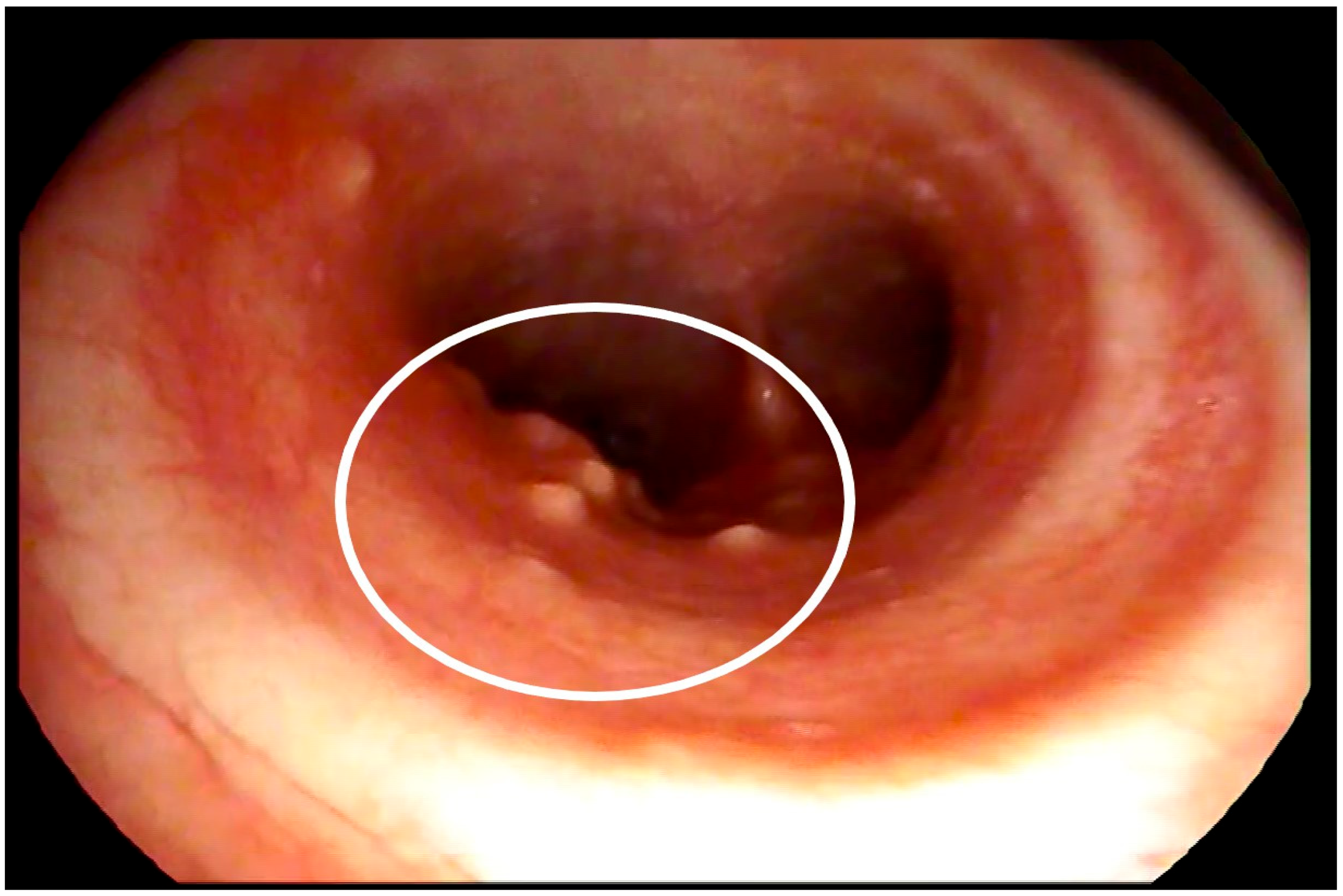
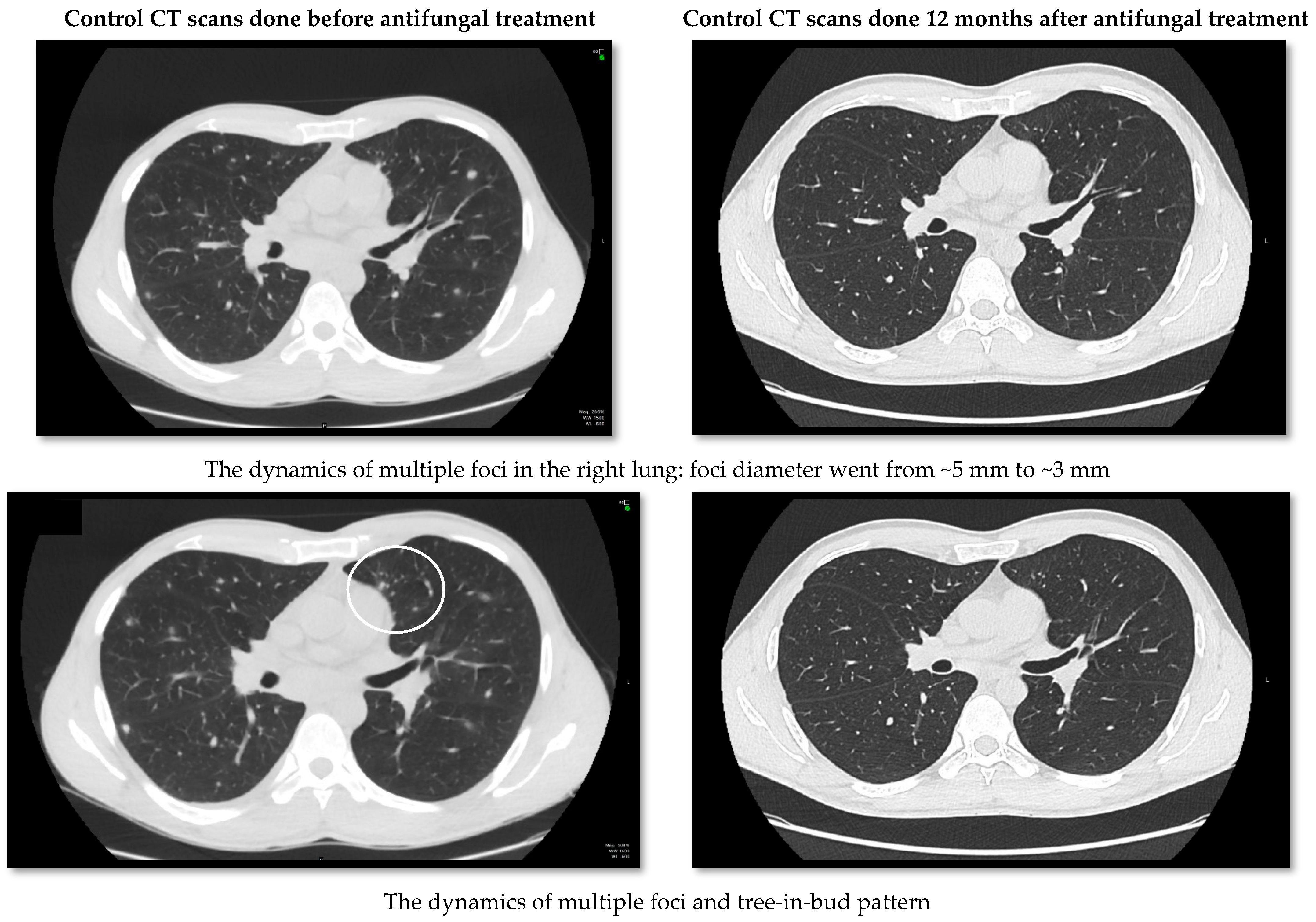
2. Case Report
3. Literature Review
3.1. Clinical Features and Diagnostics
3.2. Treatment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thompson, G.; Brown, J.; Benedict, K.; Park, B.J. Coccidioidomycosis: Epidemiology. Clin. Epidemiol. 2013, 5, 185–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stockamp, N.W.; Thompson, G.R., 3rd. Coccidioidomycosis. Infect. Dis. Clin. N. Am. 2016, 30, 229–246. [Google Scholar] [CrossRef] [PubMed]
- Matlock, M.; Hopfer, S.; Ogunseitan, O.A. Communicating Risk for a Climate-Sensitive Disease: A Case Study of Valley Fever in Central California. Int. J. Environ. Res. Public Health 2019, 16, 3254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sondermeyer, G.; Lee, L.; Gilliss, D.; Tabnak, F.; Vugia, D. Coccidioidomycosis-associated Hospitalizations, California, USA, 2000–2011. Emerg. Infect. Dis. 2013, 19, 1590–1597. [Google Scholar] [CrossRef] [PubMed]
- Donovan, F.M.; Shubitz, L.; Powell, D.; Orbach, M.; Frelinger, J.; Galgiani, J.N. Early Events in Coccidioidomycosis. Clin. Microbiol. Rev. 2019, 33, e00112-19. [Google Scholar] [CrossRef]
- Nguyen, C.; Barker, B.; Hoover, S.; Nix, D.E.; Ampel, N.M.; Frelinger, J.A.; Orbach, M.J.; Galgiani, J.N. Recent Advances in Our Understanding of the Environmental, Epidemiological, Immunological, and Clinical Dimensions of Coccidioidomycosis. Clin. Microbiol. Rev. 2013, 26, 505–525. [Google Scholar] [CrossRef] [Green Version]
- Austwick, P.K.C. Ecological Bulletins No. 31, Environmental Protection and Biological Forms of Control of Pest Organisms 1980. pp. 91–102. Available online: https://www.jstor.org/stable/i20112779 (accessed on 11 April 2021).
- Blair, J.E.; Mayer, A.P.; Currier, J.; Files, J.A.; Wu, Q. Coccidioidomycosis in Elderly Persons. Clin. Infect. Dis. 2008, 47, 1513–1518. [Google Scholar] [CrossRef] [Green Version]
- The Department of Statistics of Lithuania. Incoming, Outcoming and Local Tourism, When Services Are Provided by Tourism Companies. 2019. Available online: https://osp.stat.gov.lt/turizmas (accessed on 11 April 2021).
- Valdivia, L.; Nix, D.; Wright, M.; Lindberg, E.; Fagan, T.; Lieberman, D.; Stoffer, T.; Ampel, N.M.; Galgiani, J.N. Coccidioidomycosis as a Common Cause of Community-acquired Pneumonia. Emerg. Infect. Dis. 2006, 12, 958–962. [Google Scholar] [CrossRef]
- Eldridge, M.L.; Chambers, C.J.; Sharon, V.R.; Thompson, G.R. Fungal infections of the skin and nail: New treatment options. Expert Rev. Anti-Infect. Ther. 2014, 12, 1389–1405. [Google Scholar] [CrossRef]
- Merchant, M.; Romero, A.O.; Libke, R.D.; Joseph, J. Pleural effusion in hospitalized patients with Coccidioidomycosis. Respir. Med. 2008, 102, 537–540. [Google Scholar] [CrossRef] [Green Version]
- Thompson, G.R. Pulmonary Coccidioidomycosis. Semin. Respir. Crit. Care Med. 2011, 32, 754–763. [Google Scholar] [CrossRef]
- Ronaghi, R.; Rashidian, A.; Peterson, M.; Mills, P. Central Valley Lung Nodule Calculator: Development and Prospective Analysis of a Newly Developed Calculator in a Coccidiodomycosis Endemic Area. In Proceedings of the 59th Annual Coccidioidomycosis Study Group, San Diego, CA, USA, 11 April 2015. [Google Scholar]
- Thompson, G.R.; Le, T.; Chindamporn, A.; A Kauffman, C.; Alastruey-Izquierdo, A.; Ampel, N.M.; Andes, D.R.; Armstrong-James, D.; Ayanlowo, O.; Baddley, J.W.; et al. Global guideline for the diagnosis and management of the endemic mycoses: An initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology. Lancet Infect. Dis. 2021, 21, e364–e374. [Google Scholar] [CrossRef]
- Sutton, D.A. Diagnosis of Coccidioidomycosis by Culture: Safety Considerations, Traditional Methods, and Susceptibility Testing. Ann. N. Y. Acad. Sci. 2007, 1111, 315–325. [Google Scholar] [CrossRef]
- Kaufman, L.; Valero, G.; Padhye, A.A. Misleading Manifestations of Coccidioides immitis In Vivo. J. Clin. Microbiol. 1998, 36, 3721–3723. [Google Scholar] [CrossRef] [Green Version]
- Schuetz, A.N.; Pisapia, D.; Yan, J.; Hoda, R.S. An atypical morphologic presentation of coccidioides spp. in fine-needle aspiration of lung. Diagn. Cytopathol. 2011, 40, 163–167. [Google Scholar] [CrossRef]
- Pappagianis, D. Serologic studies in coccidioidomycosis. Semin. Respir. Infect. 2001, 16, 242–250. [Google Scholar] [CrossRef]
- Mitchell, M.; Dizon, D.; Libke, R.; Peterson, M.; Slater, D.; Dhillon, A. Development of a Real-Time PCR Assay for Identification of Coccidioides immitis by Use of the BD Max System. J. Clin. Microbiol. 2015, 53, 926–929. [Google Scholar] [CrossRef] [Green Version]
- Wack, E.E.; Ampel, N.M.; Sunenshine, R.H.; Galgiani, J.N. The Return of Delayed-Type Hypersensitivity Skin Testing for Coccidioidomycosis. Clin. Infect. Dis. 2015, 61, 787–791. [Google Scholar] [CrossRef]
- Szeyko, L.A.; Taljanovic, M.S.; Dzioba, R.B.; Rapiejko, J.L.; Adam, R.D. Vertebral Coccidioidomycosis: Presentation and Multidisciplinary Management. Am. J. Med. 2012, 125, 304–314. [Google Scholar] [CrossRef]
- Nelson, E.C.; Thompson, G.R.; Vidovszky, T.J. Image of the month. Disseminated coccidioidomycosis. Arch. Surg. 2012, 147, 95–96. [Google Scholar] [CrossRef]
- Narasimhan, A.; Rashidian, A.; Faiad, G.; Libke, R.; Paul, S.; Nassar, N.; Sharma, S. Study of prevalence, risk factors and outcomes of abdominal pseudocyst (APC) in patients with coccidioidal meningitis (CM) and ventriculoperitoneal shunts (VPS). In Proceedings of the Infectious Diseases of America 49th Annual Meeting, Boston, MA, USA, 22 October 2011. [Google Scholar]
- Phelps, R.R.L.; Lu, A.Y.; Lee, A.T.; Yue, J.K.; Winkler, E.A.; Raygor, K.P.; Oh, T.; Barkovich, M.J.; Hollander, H. Cerebrovascular complications of coccidioidomycosis meningitis: Case report and systematic review. J. Clin. Neurosci. 2020, 80, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Catanzaro, A.; Galgiani, J.N.; Levine, B.E.; Sharkey-Mathis, P.K.; Fierer, J.; Stevens, D.A.; Chapman, S.W.; Cloud, G. Fluconazole in the treatment of chronic pulmonary and nonmeningeal disseminated coccidioidomycosis. Am. J. Med. 1995, 98, 249–256. [Google Scholar] [CrossRef]
- Gadour, E.; Kotb, A. Systematic Review of Antifungal-Induced Acute Liver Failure. Cureus 2021, 13, e18940. [Google Scholar] [CrossRef] [PubMed]
- Gayam, V.; Khalid, M.; Dahal, S.; Garlapati, P.; Gill, A. Hyperacute liver injury following intravenous fluconazole: A rare case of dose-independent hepatotoxicity. J. Fam. Med. Prim. Care 2018, 7, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Spernovasilis, N.; Kofteridis, D.P. Pre-Existing Liver Disease and Toxicity of Antifungals. J. Fungi 2018, 4, 133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Institute of Diabetes and Digestive and Kidney Diseases. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2012. [Google Scholar]
- Galgiani, J.N.; Catanzaro, A.; Cloud, G.A.; Higgs, J.; Friedman, B.A.; Larsen, R.A.; Graybill, J.R. Fluconazole Therapy for Coccidioidal Meningitis. Ann. Intern. Med. 1993, 119, 28–35. [Google Scholar] [CrossRef]
- González, G.M. In vitro activities of isavuconazole against opportunistic filamentous and dimorphic fungi. Med. Mycol. 2009, 47, 71–76. [Google Scholar] [CrossRef] [Green Version]
- Thompson, G.R.; Cadena, J.; Patterson, T.F. Overview of Antifungal Agents. Clin. Chest Med. 2009, 30, 203–215. [Google Scholar] [CrossRef]
- Thompson, G.R.; Bays, D.; Cohen, S.H.; Pappagianis, D. Fluoride Excess in Coccidioidomycosis Patients Receiving Long-Term Antifungal Therapy: An Assessment of Currently Available Triazoles. Antimicrob. Agents Chemother. 2012, 56, 563–564. [Google Scholar] [CrossRef] [Green Version]
- Stevens, D.A.; Rendon, A.; Gaona-Flores, V.; Catanzaro, A.; Anstead, G.M.; Pedicone, L.; Graybill, J.R. Posaconazole Therapy for Chronic Refractory Coccidioidomycosis. Chest 2007, 132, 952–958. [Google Scholar] [CrossRef]
- Galgiani, J.N.; Ampel, N.M.; Blair, J.E.; Catanzaro, A.; Geertsma, F.; Hoover, S.E.; Johnson, R.H.; Kusne, S.; Lisse, J.; Macdonald, J.D.; et al. 2016 Infectious Diseases Society of America (IDSA) Clinical Practice Guideline for the Treatment of Coccidioidomycosis. Clin. Infect. Dis. 2016, 63, e112–e146. [Google Scholar] [CrossRef]
- Shubitz, L.F.; Trinh, H.T.; Perrill, R.H.; Thompson, C.M.; Hanan, N.; Galgiani, J.N.; Nix, D.E. Modeling Nikkomycin Z Dosing and Pharmacology in Murine Pulmonary Coccidioidomycosis Preparatory to Phase 2 Clinical Trials. J. Infect. Dis. 2014, 209, 1949–1954. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alčauskas, T.; Zablockienė, B.; Zablockis, R.; Svetikas, L.; Bilotaitė, L.; Jančorienė, L. Pulmonary Coccidioidomycosis: A Case Report and Literature Review. Medicina 2022, 58, 655. https://doi.org/10.3390/medicina58050655
Alčauskas T, Zablockienė B, Zablockis R, Svetikas L, Bilotaitė L, Jančorienė L. Pulmonary Coccidioidomycosis: A Case Report and Literature Review. Medicina. 2022; 58(5):655. https://doi.org/10.3390/medicina58050655
Chicago/Turabian StyleAlčauskas, Tadas, Birutė Zablockienė, Rolandas Zablockis, Linas Svetikas, Laura Bilotaitė, and Ligita Jančorienė. 2022. "Pulmonary Coccidioidomycosis: A Case Report and Literature Review" Medicina 58, no. 5: 655. https://doi.org/10.3390/medicina58050655
APA StyleAlčauskas, T., Zablockienė, B., Zablockis, R., Svetikas, L., Bilotaitė, L., & Jančorienė, L. (2022). Pulmonary Coccidioidomycosis: A Case Report and Literature Review. Medicina, 58(5), 655. https://doi.org/10.3390/medicina58050655





