Experimental and Emerging Free Fatty Acid Receptor Agonists for the Treatment of Type 2 Diabetes
Abstract
:1. Introduction
2. Free Fatty Acids, Glucose Metabolism, and Type 2 Diabetes
2.1. Free Fatty Acids 1 (FFA1)
2.2. Free Fatty Acids 4 (FFA4)
3. The Relationship between Free Fatty Acids and Incretins
4. Therapeutic Avenues
5. Conclusions
Funding
Conflicts of Interest
References
- Watterson, K.R.; Hudson, B.D.; Ulven, T.; Milligan, G. Treatment of type 2 diabetes by free Fatty Acid receptor agonists. Front. Endocrinol. 2014, 5, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Offermanns, S. Free fatty acid (FFA) and hydroxycarboxylic acid (HCA) receptors. Annu. Rev. Pharmacol. Toxicol. 2014, 54, 407–434. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, M.; Bender, E.; Schamberger, J.; Eitner, F. Pharmacology of Free Fatty Acid Receptors and Their Allosteric Modulators. Int. J. Mol. Sci. 2021, 22, 1763. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; Ichimura, A.; Ohue-Kitano, R.; Igarashi, M. Free Fatty Acid Receptors in Health and Disease. Physiol. Rev. 2020, 100, 171–210. [Google Scholar] [CrossRef]
- Stoian, A.P.; Banerjee, Y.; Rizvi, A.A.; Rizzo, M. Diabetes and the COVID-19 Pandemic: How Insights from Recent Experience Might Guide Future Management. Metab. Syndr. Relat. Disord. 2020, 18, 173–175. [Google Scholar] [CrossRef]
- Lim, S.; Bae, J.H.; Kwon, H.S.; Nauck, M.A. COVID-19 and diabetes mellitus: From pathophysiology to clinical management. Nat. Rev. Endocrinol. 2021, 17, 11–30. [Google Scholar] [CrossRef]
- Ceriello, A.; Stoian, A.P.; Rizzo, M. COVID-19 and diabetes management: What should be considered? Diabetes Res. Clin. Pract. 2020, 163, 108151. [Google Scholar] [CrossRef]
- Al Mahmeed, W.; Al-Rasadi, K.; Banerjee, Y.; Ceriello, A.; Cosentino, F.; Galia, M.; Goh, S.Y.; Kempler, P.; Lessan, N.; Papanas, N.; et al. CArdiometabolic Panel of International experts on Syndemic COvid-19 (CAPISCO). Promoting a Syndemic Approach for Cardiometabolic Disease Management During COVID-19: The CAPISCO International Expert Panel. Front. Cardiovasc. Med. 2021, 8, 787761. [Google Scholar] [CrossRef]
- Rizvi, A.A.; Janez, A.; Al Mahmeed, W.; Rizzo, M. Diabetes and COVID-19: A Tale of 2 Pandemics. J. Cardiovasc. Pharmacol. 2021, 78, e1–e2. [Google Scholar] [CrossRef] [PubMed]
- Nogal, A.; Valdes, A.M.; Menni, C. The role of short-chain fatty acids in the interplay be-tween gut microbiota and diet in cardio-metabolic health. Gut Microbes 2021, 13, 1897212. [Google Scholar] [CrossRef]
- Chen, C.; Li, H.; Long, Y.Q. GPR40 agonists for the treatment of type 2 diabetes mellitus: The biological characteristics and the chemical space. Bioorg. Med. Chem. Lett. 2016, 26, 5603–5612. [Google Scholar] [CrossRef] [PubMed]
- Moniri, N.H.; Farah, Q. Short-chain free-fatty acid G protein-coupled receptors in colon cancer. Biochem. Pharmacol. 2021, 186, 114483. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Gu, Y.; Wu, C.; Yu, F.; Chen, Y.; Zhu, J.; Yao, X.; Bei, C.; Zhu, Q. Agonist-induced activation of human FFA1 receptor signals to extracellular signal-regulated kinase 1 and 2 through Gq- and Gi-coupled signaling cascades. Cell. Mol. Biol. Lett. 2017, 22, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, Y.; Swaminath, G.; Cao, Q.; Yang, L.; Guo, Q.; Salomonis, H.; Lu, J.; Houze, J.B.; Dransfield, P.J.; Wang, Y.; et al. Activation of FFA1 mediates GLP-1 secretion in mice. Evidence for allosterism at FFA1. Mol. Cell. Endocrinol. 2013, 369, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Gribble, F.M.; Diakogiannaki, E.; Reimann, F. Gut Hormone Regulation and Secretion via FFA1 and FFA4. Handb. Exp. Pharmacol. 2017, 236, 181–203. [Google Scholar] [CrossRef] [PubMed]
- Ježek, P.; Jabůrek, M.; Holendová, B.; Plecitá-Hlavatá, L. Fatty Acid-Stimulated Insulin Secretion vs. Lipotoxicity. Molecules 2018, 23, 1483. [Google Scholar] [CrossRef] [Green Version]
- Goldspink, D.A.; Lu, V.B.; Billing, L.J.; Larraufie, P.; Tolhurst, G.; Gribble, F.M.; Reimann, F. Mechanistic insights into the detection of free fatty and bile acids by ileal glucagon-like peptide-1 secreting cells. Mol. Metab. 2018, 7, 90–101. [Google Scholar] [CrossRef]
- Marcinak, J.; Cao, C.; Lee, D.; Ye, Z. Fasiglifam for glycaemic control in people with type 2 diabetes: A phase III.; placebo-controlled study. Diabetes Obes. Metab. 2017, 19, 1714–1721. [Google Scholar] [CrossRef]
- Mancini, A.D.; Poitout, V. The fatty acid receptor FFA1/GPR40 a decade later: How much do we know? Trends Endocrinol. Metab. 2013, 24, 398–407. [Google Scholar] [CrossRef]
- Burant, C.F. Activation of GPR40 as a therapeutic target for the treatment of type 2 diabtes. Diabetes Care 2013, 36, 175–179. [Google Scholar] [CrossRef] [Green Version]
- Poitout, V.; Lin, D.C. Modulating GPR40: Therapeutic promise and potential in diabetes. Drug Discov. Today 2013, 18, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Cornall, L.M.; Mathai, M.L.; Hryciw, D.H.; McAinch, A.J. GPR120 agonism as a counter measure against metabolic diseases. Drug Discov. Today 2014, 19, 670–679. [Google Scholar] [CrossRef]
- Ichimura, A.; Hirasawa, A.; Poulain-Godefroy, O.; Bonnefond, A.; Hara, T.; Yengo, L.; Kimura, I.; Leloire, A.; Liu, N.; Iida, K.; et al. Dysfunction of lipid sensor GPR120 leads to obesity in both mouse and human. Nature 2012, 483, 350–354. [Google Scholar] [CrossRef] [Green Version]
- Gong, Z.; Yoshimura, M.; Aizawa, S.; Kurotani, R.; Zigman, J.M.; Sakai, T.; Sakata, I. G protein coupled receptor 120 signaling regulates ghrelin secretion in vivo and in vitro. Am. J. Physiol. Endocrinol. Metab. 2014, 306, 28–35. [Google Scholar] [CrossRef] [Green Version]
- Taneera, J.; Lang, S.; Sharma, A.; Fadista, J.; Zhou, Y.; Ahlqvist, E.; Jonsson, A.; Lyssenko, V.; Vikman, P.; Hansson, O.; et al. A systems genetics approach identifies genes and pathways for type 2 diabetes in human islets. Cell Metab. 2012, 16, 122–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stone, V.M.; Dhayal, S.; Brocklehurst, K.J.; Lenaghan, C.; Sörhede Winzell, M.; Hammar, M.; Xu, X.; Smith, D.M.; Morgan, N.G. GPR120 (FFgAR4) is preferentially expressed in pancreatic delta cells and regulates somatostatin secretion from murine islets of Langerhans. Diabetologia 2014, 57, 1182–1891. [Google Scholar] [CrossRef] [Green Version]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wellhauser, L.; Belsham, D.D. Activation of the omega-3 fatty acid receptor GPR120 medi-ates anti-inflammatory actions in immortalized hypothalamic neurons. J. Neuroinflamm. 2014, 11, 60. [Google Scholar] [CrossRef] [Green Version]
- Halder, S.; Kumar, S.; Sharma, R. The therapeutic potential of GPR120: A patent review. Exp. Opin. Ther. Pat. 2013, 23, 1581–1590. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Pfeiffer, A.F.H. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: A pathophysiological update. Diabetes Obes. Metab. 2021, 23, 5–29. [Google Scholar] [CrossRef]
- Sachinidis, A.; Nikolic, D.; Stoian, A.P.; Papanas, N.; Tarar, O.; Rizvi, A.A.; Rizzo, M. Cardiovascular outcomes trials with incretin-based medications: A critical review of data available on GLP-1 receptor agonists and DPP-4 inhibitors. Metabolism 2020, 111, 154343. [Google Scholar] [CrossRef] [PubMed]
- Stoian, A.P.; Papanas, N.; Prazny, M.; Rizvi, A.A.; Rizzo, M. Incretin-Based Therapies Role in COVID-19 Era: Evolving Insights. J. Cardiovasc. Pharmacol. Ther. 2020, 25, 494–496. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Qiu, Q.; Geng, X.; Yang, J.; Huang, W.; Qian, H. Free fatty acid receptor agonists for the treatment of type 2 diabetes: Drugs in preclinical to phase II clinical development. Exp. Opin. Investig. Drugs 2016, 25, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Ghislain, J.; Poitout, V. Targeting lipid GPCRs to treat type 2 diabetes mellitus—Progress and challenges. Nat. Rev. Endocrinol. 2021, 17, 162–175. [Google Scholar] [CrossRef]
- Cani, P.D.; Everard, A.; Duparc, T. Gut microbiota, enteroendocrine functions and metabolism. Curr. Opin. Pharmacol. 2013, 13, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Ulven, T. Short-chain free fatty acid receptors FFA2/GPR43 and FFA3/GPR41 as new potential therapeutic targets. Front. Endocrinol. 2012, 3, 111. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Jiao, X.; Kayser, F.; Liu, J.; Wang, Z.; Wanska, M.; Greenberg, J.; Weiszmann, J.; Ge, H.; Tian, H.; et al. The first synthetic agonists of FFA2: Discovery and Sar of phenylacetamide as allosteric modulators. Bioorg. Med. Chem. Lett. 2010, 20, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Leifke, E.; Naik, H.; Wu, J.; Viswanathan, P.; Demanno, D.; Kipnes, M.; Vakilynejad, M. A Multiple-Ascending-Dose Study to Evaluate Safety, Pharmacokinetics, and Pharmacodynamics of a Novel GPR40 Agonist, TAK-875, in Subjects with Type 2 Diabetes. Clin. Pharmacol. Ther. 2012, 92, 29–39. [Google Scholar] [CrossRef] [Green Version]
- Burant, C.F.; Viswanathan, P.; Marcinak, J.; Cao, C.; Vakilynejad, M.; Xie, B.; Leifke, E. TAK-875 versus placebo or glimepiride in type 2 diabetes mellitus: A phase 2, randomised, double-blind, placebo-controlled trial. Lancet 2012, 379, 1403–1411. [Google Scholar] [CrossRef]
- Kaku, K.; Enya, K.; Nakaya, R.; Ohira, T.; Matsuno, R. Efficacy and safety of fasiglifam (TAK-875), a G protein-coupled receptor 40 agonist, in Japanese patients with type 2 diabetes inadequately controlled by diet and exercise: A randomized, double-blind, placebo-controlled, phase III trial. Diabetes Obes. Metab. 2015, 17, 675–681. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, H.; Yoshida, S.; Minoura, H.; Negoro, K.; Shimaya, A.; Shimokawa, T.; Shibasaki, M. Novel GPR40 agonist AS2575959 exhibits glucose metabolism improve-ment and synergistic effect with sitagliptin on insulin and incretin secretion. Life Sci. 2014, 94, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ito, R.; Tsujihata, Y.; Matsuda-Nagasumi, K.; Mori, I.; Negoro, N.; Takeuchi, K. Tak-875, a GPR40/FFAR1 agonist, in combination with metformin prevents progression of diabetes and beta-cell dysfunction in Zucker diabetic fatty rats. Br. J. Pharmacol. 2013, 170, 568–580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakashima, R.; Yano, T.; Ogawa, J.; Tanaka, N.; Toda, N.; Yoshida, M.; Takano, R.; Inoue, M.; Honda, T.; Kume, S.; et al. Potentiation of insulin secretion and improvement of glucose intolerance by combining a novel G protein-coupled receptor 40 agonist DS-1558 with glucagon-like peptide-1receptor agonists. Eur. J. Pharmacol. 2014, 737, 194–201. [Google Scholar] [CrossRef]
- Pereira, M.J.; Lundkvist, P.; Kamble, P.G.; Lau, J.; Martins, J.G.; Sjöström, C.D.; Schnecke, V.; Walentinsson, A.; Johnsson, E.; Eriksson, J.W. A randomized controlled trial of Dapagliflozin Plus Exenatide once weekly versus placebo in individuals with obesity and without diabetes: Metabolic and marker ef-fects associated with body weight loss. Diabetes Ther. 2018, 9, 1511–1532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.G.; Luo, Y.; Wang, H.; Li, J.Y.; Yang, J.; Liu, Y.X.; Qu, H.Q.; Wang, B.L.; Zhu, M. Liraglutide Improves lipotoxicity-induced oxidative stress by activating the NRF2 pathway in HepG2 cells. Horm. Metab. Res. 2020, 52, 532–539. [Google Scholar] [CrossRef]
- Rizzo, M.; Nikolic, D.; Patti, A.M.; Mannina, C.; Montalto, G.; McAdams, B.S.; Rizvi, A.A.; Cosentino, F. GLP-1 receptor agonists and reduction of cardiometabolic risk: Potential underlying mechanisms. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 2814–2821. [Google Scholar] [CrossRef]
- Nikolic, D.; Giglio, R.V.; Rizvi, A.A.; Patti, A.M.; Montalto, G.; Maranta, F.; Cianflone, D.; Stoian, A.P.; Rizzo, M. Liraglutide reduces carotid intima-media thickness by reducing small dense low-density lipoproteins in a real-world setting of patients with type 2 diabetes: A novel anti-atherogenic effect. Diabetes Ther. 2021, 12, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Anholm, C.; Kumarathurai, P.; Pedersen, L.R.; Samkani, A.; Walzem, R.L.; Nielsen, O.W.; Kristiansen, O.P.; Fenger, M.; Madsbad, S.; Sajadieh, A.; et al. Liraglutide in combination with metformin may improve the atherogenic lipid profile and decrease C-reactive protein level in statin treated obese patients with coronary artery disease and newly diagnosed type 2 diabetes: A randomized trial. Atherosclerosis 2019, 288, 60–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berneis, K.; Rizzo, M.; Stettler, C.; Chappuis, B.; Braun, M.; Diem, P.; Christ, E.R. Comparative effects of rosiglitazone and pioglitazone on fasting and postprandial low-density lipoprotein size and subclasses in patients with Type 2 diabetes. Expert Opin. Pharmacother. 2008, 9, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Berneis, K. The clinical relevance of low-density-lipoproteins size modulation by statins. Cardiovasc. Drugs Ther. 2006, 20, 205–217. [Google Scholar] [CrossRef]
- Nikolic, D.; Banach, M.; Nikfar, S.; Salari, P.; Mikhailidis, D.P.; Toth, P.P.; Abdollahi, M.; Ray, K.K.; Pencina, M.J.; Malyszko, J.; et al. Lipid and Blood Pressure Meta-Analysis Collaboration Group. A meta-analysis of the role of statins on renal outcomes in patients with chronic kidney disease. Is the duration of therapy important? Int. J. Cardiol. 2013, 168, 5437–5447. [Google Scholar] [CrossRef] [PubMed]
- Crismaru, I.; Pantea Stoian, A.; Bratu, O.G.; Gaman, M.A.; Stanescu, A.M.A.; Bacalbasa, N.; Diaconu, C.C. Low-density lipoprotein cholesterol lowering treatment: The current approach. Lipids Health Dis. 2020, 19, 85. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef] [Green Version]
- Kutoh, E.; Kuto, A.N.; Wada, A.; Kurihara, R.; Kojima, R. Regulations of free fatty acids and diabetic parameters in drug naïve sub-jects with type 2 diabetes treated with canagliflozin monotherapy. Drug Res. 2021. [Google Scholar] [CrossRef]
- Rizzo, M.; Berneis, K. Should we measure routinely the LDL peak particle size? Int. J. Cardiol. 2006, 107, 166–170. [Google Scholar] [CrossRef]
- Berneis, K.; Rizzo, M.; Hersberger, M.; Rini, G.B.; Di Fede, G.; Pepe, I.; Spinas, G.A.; Carmina, E. Atherogenic forms of dyslipidaemia in women with polycystic ovary syndrome. Int. J. Clin. Pract. 2009, 63, 56–62. [Google Scholar] [CrossRef]
- Rizvi, A.A.; Stoian, A.P.; Janez, A.; Rizzo, M. Lipoproteins and Cardiovascular Disease: An, Update on the Clinical Significance of Atherogenic Small, Dense LDL and New Therapeutical Options. Biomedicines 2021, 9, 1579. [Google Scholar] [CrossRef]
- Bayram, F.; Kocer, D.; Gundogan, K.; Kaya, A.; Demir, O.; Coskun, R.; Sabuncu, T.; Ka-raman, A.; Cesur, M.; Rizzo, M.; et al. Prevalence of dyslipidemia and associated risk factors in Turkish adults. J. Clin. Lipidol. 2014, 8, 206–216. [Google Scholar] [CrossRef]
- Goedecke, J.H.; Utzschneider, K.; Faulenbach, M.V.; Rizzo, M.; Berneis, K.; Spinas, G.A.; Dave, J.; Levitt, N.S.; Lambert, E.V.; Olsson, T.; et al. Ethnic differences in serum lipoproteins and their determinants in South African women. Metabolism 2010, 59, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Toth, P.P.; Barylski, M.; Nikolic, D.; Rizzo, M.; Montalto, G.; Banach, M. Should low high-density lipoprotein cholesterol (HDL-C) be treated? Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 353–368. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Berneis, K. Who needs to care about small, dense low density lipoproteins? Int. J. Clin. Pract. 2007, 61, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.A. Cytokine biomarkers, endothelial inflammation, and atherosclerosis in the met-abolic syndrome: Emerging concepts. Am. J. Med. Sci. 2009, 338, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Suceveanu, A.I.; Mazilu, L.; Katsiki, N.; Parepa, I.; Voinea, F.; Pantea-Stoian, A.; Rizzo, M.; Botea, F.; Herlea, V.; Serban, D.; et al. NLRP3 Inflammasome Biomarker-Could Be the New Tool for Improved Cardiometabolic Syndrome Outcome. Metabolites 2020, 10, 448. [Google Scholar] [CrossRef]
- Abate, N.; Sallam, H.S.; Rizzo, M.; Nikolic, D.; Obradovic, M.; Bjelogrlic, P.; Isenovic, E.R. Resistin: An inflammatory cytokine. Role in cardiovascular diseases, diabe-tes and the metabolic syndrome. Curr. Pharm. Des. 2014, 20, 4961–4969. [Google Scholar] [CrossRef] [PubMed]
- Ha, X.; Cai, X.; Cao, H.; Li, J.; Yang, B.; Jiang, R.; Li, X.; Li, B.; Xin, Y. Docking protein 1 and free fatty acids are associated with insulin resistance in patients with type 2 diabetes mellitus. J. Int. Med. Res. 2021, 49, 3000605211048293. [Google Scholar] [CrossRef]
- Menon, V.; Lincoff, A.M.; Nicholls, S.J.; Jasper, S.; Wolski, K.; McGuire, D.K.; Mehta, C.R.; Rosenstock, J.; Lopez, C.; Marcinak, J.; et al. Fasiglifam-Induced Liver Injury in Patients with Type 2 Diabetes: Results of a Randomized Controlled Cardiovascular Outcomes Safety Trial. Diabetes Care 2018, 41, 2603–2609. [Google Scholar] [CrossRef] [Green Version]
- Shavadia, J.S.; Sharma, A.; Gu, X.; Neaton, J.; DeLeve, L.; Holmes, D.; Home, P.; Eckel, R.H.; Watkins, P.B.; Granger, C.B. Determination of fasiglifam-induced liver toxicity: Insights from the data monitoring committee of the fasiglifam clinical trials program. Clin. Trials 2019, 16, 253–262. [Google Scholar] [CrossRef] [PubMed]
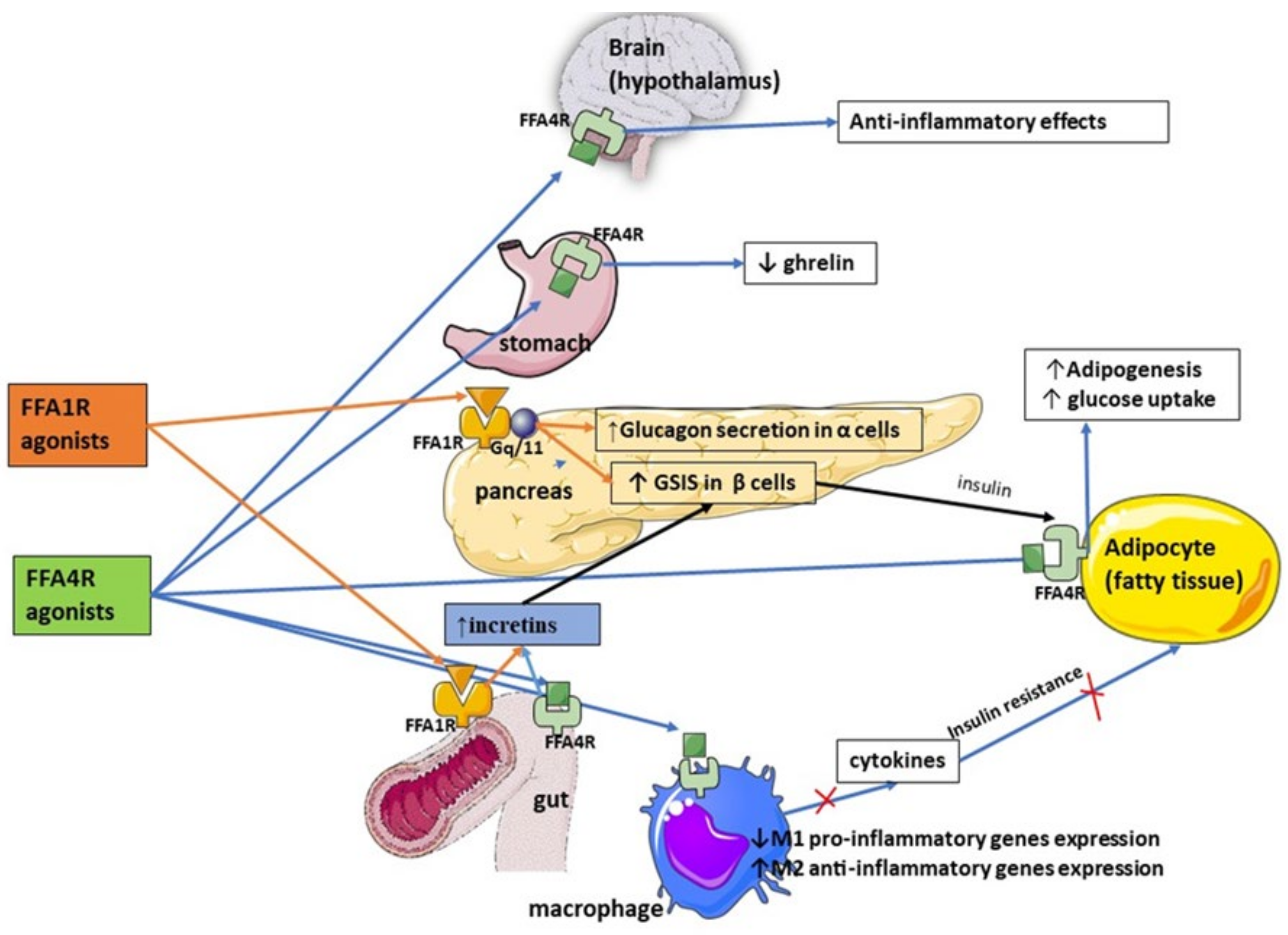
| FFA1 expressed by enteroendocrine cells regulates release of incretin hormones, such as GLP-1 and Cholecystokinin-5, which in turn enhance pancreatic insulin secretion and promote satiety |
| FFA1 agonists (Fasiglifam) have been shown to improve glycemic control, increase insulin sensitivity, induce body mass loss, and reduce inflammation |
| FFA2 and FFA3 receptors are linked to enhanced incretin secretion from enteroendocrine cells |
| There is still a paucity of research on inter-relationship between FFAs and incretins, research on FFA2/FFA3 agonists, and the role of FFARs in improving glucose uptake and increasing GLP-1 secretion |
| FFA4 agonism is associated with improved sensitivity to insulin via anti-inflammatory effect on macrophages through recruitment of β-arrestin 2, increased expression of M2 anti-inflammatory genes, and reduced expression of pro-inflammatory in M1 macrophages |
| FFA4 inhibits secretion of ghrelin, thus stimulating satiety, promoting incretin release leading to a glucoprotective effect in diabetes, and regulating glucagon secretion |
| FFAs couple with FFARs to regulate inflammation and peptide hormone secretion |
| Trial | Ref. | Year Published | Agent Studied | Study Design | Results |
|---|---|---|---|---|---|
| Leifke et al. | [38] | 2012 | TAK-875 | Phase 1, randomized, double-blind, multiple ascending-dose | Significantly lower fasting and post-challenge glucose |
| Burant et al. | [39] | 2012 | TAK-875 | Phase 2, randomized, double-blind with placebo and glimepiride comparator groups | HbA1c decreased by 1% with study agent |
| Ito et al. | [42] | 2013 | TAK-875 | Combination with metformin in Zucker diabetic rats | Prevention of diabetes progression and beta-cell dysfunction |
| Tanaka et al. | [41] | 2014 | AS2575959 | Combination with sitagliptin | Improved glucose homeostasis |
| Nakashima et al. | [43] | 2014 | DS-1558 | Combination with GLP-1 receptor agonists | Improved glucose tolerance and insulin secretion |
| Kaku et al. | [40] | 2015 | TAK-875 | Phase 3, randomized, double-blind, placebo-controlled, multiple-dose | Dose-related reduction in HbA1c compared with placebo |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patti, A.M.; Giglio, R.V.; Papanas, N.; Serban, D.; Stoian, A.P.; Pafili, K.; Al Rasadi, K.; Rajagopalan, K.; Rizvi, A.A.; Ciaccio, M.; et al. Experimental and Emerging Free Fatty Acid Receptor Agonists for the Treatment of Type 2 Diabetes. Medicina 2022, 58, 109. https://doi.org/10.3390/medicina58010109
Patti AM, Giglio RV, Papanas N, Serban D, Stoian AP, Pafili K, Al Rasadi K, Rajagopalan K, Rizvi AA, Ciaccio M, et al. Experimental and Emerging Free Fatty Acid Receptor Agonists for the Treatment of Type 2 Diabetes. Medicina. 2022; 58(1):109. https://doi.org/10.3390/medicina58010109
Chicago/Turabian StylePatti, Angelo Maria, Rosaria Vincenza Giglio, Nikolaos Papanas, Dragos Serban, Anca Pantea Stoian, Kalliopi Pafili, Khalid Al Rasadi, Kanya Rajagopalan, Ali A. Rizvi, Marcello Ciaccio, and et al. 2022. "Experimental and Emerging Free Fatty Acid Receptor Agonists for the Treatment of Type 2 Diabetes" Medicina 58, no. 1: 109. https://doi.org/10.3390/medicina58010109
APA StylePatti, A. M., Giglio, R. V., Papanas, N., Serban, D., Stoian, A. P., Pafili, K., Al Rasadi, K., Rajagopalan, K., Rizvi, A. A., Ciaccio, M., & Rizzo, M. (2022). Experimental and Emerging Free Fatty Acid Receptor Agonists for the Treatment of Type 2 Diabetes. Medicina, 58(1), 109. https://doi.org/10.3390/medicina58010109












