Acrometastases to the Hand: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Selection
2.2. Data Extraction
3. Results
4. Discussion
4.1. Pathophysiology
4.2. Imaging
4.3. Patient Population
4.4. Therapeutic Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mavrogenis, A.F.; Mimidis, G.; Kokkalis, Z.T.; Karampi, E.-S.; Karampela, I.; Papagelopoulos, P.J.; Armaganidis, A. Acrometastases. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 279–283. [Google Scholar] [CrossRef]
- Kerin, R. Metastatic tumors of the hand. A review of the literature. J. Bone Jt. Surg. Am. 1983, 65, 1331–1335. [Google Scholar] [CrossRef] [PubMed]
- Spiteri, V.; Bibra, A.; Ashwood, N.; Cobb, J. Managing acrometastases treatment strategy with a case illustration. Ann. R. Coll. Surg. Engl. 2008, 90, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, T. Occult malignancy presenting as metastatic disease to the hand and wrist. Skeletal Radiol. 1995, 24, 135–137. [Google Scholar] [CrossRef]
- Stomeo, D.; Tulli, A.; Ziranu, A.; Perisano, C.; De Santis, V.; Maccauro, G. Acrometastasis: A literature review. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 2906–2915. [Google Scholar]
- Henderson, J.J. Metastatic carcinoma in the hand presenting as an acute paronychia. Br. J. Clin. Pract. 1987, 41, 805–806. [Google Scholar] [PubMed]
- Yasaka, N.; Ando, I.; Kukita, A. An acral “inflammatory” cutaneous metastasis of oesophageal carcinoma. Br. J. Dermatol. 1999, 141, 938–939. [Google Scholar] [CrossRef] [PubMed]
- Henderson, M.; Jehangir, S. A swollen finger and abdominal pain. Lancet 2001, 358, 636. [Google Scholar] [CrossRef]
- Parungao, A.J.; Milner, S. Photo quiz A swollen, draining thumb. Am. Fam. Physician 2002, 65, 105–106. [Google Scholar]
- Theunissen, C.C.W.; Engelkens, H.J.H.; Mulder, L.J.M.M.; Dees, A. Diagnostic image (107). A man with weight loss and skin nodes. Ned. Tijdschr. Geneeskd. 2002, 146, 1832. [Google Scholar]
- Blanes, M.; Bañuls, J.; Guijarro, J.; Betlloch, I.; Payá, A. Metástasis acral digital ósea por adenocarcinoma renal con afectación cutánea. Actas Dermosifiliogr. 2003, 94, 555–558. [Google Scholar] [CrossRef]
- Chou, Y.C.; Lin, J.Y.; Chen, H.H.; Ho, H.C. A patient of esophageal carcinoma presenting with fever and multiple nodules on fingers. Dermatol. Sin. 2004, 22, 199–200. [Google Scholar]
- Gawley, B.; Motykie, G.; Piazza, R.C.; Holding, J.; Gould, L.J. Rapid progression of metastatic bronchogenic carcinoma after felon drainage. Orthopedics 2006, 29, 1035–1036. [Google Scholar] [CrossRef]
- Park, K.-H.; Rho, Y.-H.; Choi, S.-J.; Hong, S.-J.; Lee, J.-H.; Choi, I.-K.; Kim, S.-J.; Seo, J.-H.; Choi, C.-W.; Kim, B.-S.; et al. Acute arthritis of carpal bones secondary to metastatic gastric cancer. Clin. Rheumatol. 2006, 25, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Kanatani, T.; Fujioka, H.; Yamasaki, K. Re: Metastatic oesophageal carcinoma presenting in a finger. J. Hand Surg. 2008, 33, 537–538. [Google Scholar] [CrossRef] [PubMed]
- Anandan, S.M.; Iyer, S.; Uppal, R. Metastases to the finger masquerading as flexor tenosynovitis. J. Hand Surg. 2010, 35, 597–598. [Google Scholar] [CrossRef]
- Sim, E. Misinterpretation of a metastatic process within the scope of Sudeck’s dystrophy. A case report. Aktuelle Traumatol. 1989, 19, 85–89. [Google Scholar] [PubMed]
- Bowles, F.; Wells, H. Paronychia: Not always what it seems. Emerg. Med. J. 2011, 28, 342. [Google Scholar] [CrossRef]
- Kontogeorgakos, V.; Korompilias, A.; Georgousis, M.; Papachristou, D. Digital Metastasis Presenting as Infection. J. Hand Microsurg. 2016, 3, 25–27. [Google Scholar] [CrossRef]
- Roohi, S.A.; Samsudin, O.C.; Shukur, M.H.; Ibrahim, S. Metastatic adenocarcinoma to the wrist presenting as carpal tunnel syndrome: A report of an unusual case. Malaysian Orthop. J. 2011, 5, 75–77. [Google Scholar] [CrossRef]
- Verardino, G.C.; da Silva, R.S.; Obadia, D.L.; Gripp, A.C.; Alves, M.D.F.G.S. Rare cutaneous metastasis from a probable basaloid carcinoma of the colon mimicking pyogenic granuloma. An. Bras. Dermatol. 2011, 86, 537–540. [Google Scholar] [CrossRef]
- Borgohain, B.; Borgohain, N.; Khonglah, T.; Bareh, J. Occult renal cell carcinoma with acrometastasis and ipsilateral juxta-articular knee lesions mimicking acute inflammation. Adv. Biomed. Res. 2012, 1, 48. [Google Scholar] [CrossRef] [PubMed]
- Ragois, P.; Didailler, P.; Rizzi, P. Métastase cutanée d’un cancer pulmonaire simulant une maladie de Dupuytren. Chir. Main 2012, 31, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.S.S.; Yip, L.Y.; Nguyen, M.; Wijesinghe, W.; Sahu, A. A Painful Finger: An Unusual Presentation of Von Hippel-Lindau-Associated Advanced Renal Cell Carcinoma. Case Rep. Oncol. 2020, 13, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Farouk, A.R.; McBride, D.J.; Bingham, J. A case of tumour simulating pulp space infection. Br. J. Clin. Pract. 1990, 44, 737–738. [Google Scholar]
- Kolomiets, S.A.; Lytkin, A.S. Metastasis of small-cell lung cancer to the bone of the finger simulating osseous paronychia. Vestn. Khir. Im. I. I. Grek. 1991, 147, 55–56. [Google Scholar]
- Lambert, D.; Escallier, F.; Collet, E.; Dallac, S.; Maingon, P.; Mayer, F.; Bastien, H. Distal phalangeal metastasis of a chondrosarcoma presenting initially as bilateral onycholysis. Clin. Exp. Dermatol. 1992, 17, 463–465. [Google Scholar] [CrossRef] [PubMed]
- Moens, P.; De Smet, L.; Fabry, G.; Van Damme, B. Acrometastasis of the hand presenting as a whitlow. General review apropos of a case. Rev. Chir. Orthop. Reparatrice Appar. Mot. 1993, 79, 66–69. [Google Scholar]
- Javed, S.; Fenyar, B.; Cilursu, A.M. Finger Metastasis Mimicking Osteomyelitis Case Report and Review of Literature. JCR J. Clin. Rheumatol. 1997, 3, 168–170. [Google Scholar] [CrossRef]
- Chin, K.R.; Pess, G.M.; Jupiter, J.B. Chronic lymphocytic leukemia presenting as pyogenic arthritis of the proximal interphalangeal joint. J. Hand Surg. Am. 1998, 23, 545–550. [Google Scholar] [CrossRef]
- Umebayashi, Y. Metastasis of Esophageal Carcinoma Manifesting as Whitlow-like Lesions. J. Dermatol. 1998, 25, 256–259. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372. [Google Scholar] [CrossRef]
- Filloux, J.F.; Fontaine, C. Tumeurs métastatiques des parties molles des doigts. À propos de six cas. Chir. Main 2000, 19, 63–66. [Google Scholar] [CrossRef]
- Houston, J.D.; Telepak, R.J. An Isolated Digital Metastasis of Esophageal Basaloid Squamous Cell Carcinoma. Clin. Nucl. Med. 2000, 25, 557–558. [Google Scholar] [CrossRef]
- Ryu, J.S.; Cho, J.W.; Moon, T.H.; Lee, H.L.; Han, H.S.; Choi, G.S. Squamous cell lung cancer with solitary subungual metastasis. Yonsei Med. J. 2000, 41, 666. [Google Scholar] [CrossRef]
- Tochigi, H.; Nakao, Y.; Horiuchi, Y.; Toyama, Y. Metastatic malignant melanoma in the hand muscle—A case report. Hand Surg. 2000, 5, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Asthana, S.; Deo, S.V.; Shukla, N.K.; Raina, V. Carcinoma breast metastatic to the hand and the foot. Australas. Radiol. 2001, 45, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Caglar, M.; Ceylan, E. Isolated Carpal Bone Metastases from Bronchogenic Cancer Evident on Bone Scintigraphy. Clin. Nucl. Med. 2001, 26, 352–353. [Google Scholar] [CrossRef]
- Chang, H.C.; Lew, K.H.; Low, C.O. Metastasis of an adenocarcinoma of the stomach to the 4th metacarpal bone. Hand Surg. 2001, 6, 239–242. [Google Scholar] [CrossRef]
- Fang, Y.R.; Huang, Y.S.; Wu, J.C.; Chao, Y.; Tsay, S.H.; Chan, C.Y.; Chang, F.Y.; Lee, S.D. An unusual cutaneous metastasis from hepatocellular carcinoma. Zhonghua Yi Xue Za Zhi 2001, 64, 253–257. [Google Scholar]
- Mehta, N.J.; Mehta, R.N.; Jani, K.; Nehra, A. Subungual and ocular metastases from small cell carcinoma of the lung. Hosp. Physician 2001, 37, 38–40. [Google Scholar]
- Silfen, R.; Amir, A.; Tobar, A.; Hauben, D.J. The Digital Pulp as a Presenting Site of Metastatic Esophageal Carcinoma. Ann. Plast. Surg. 2001, 46, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, H.d.A.; Tsai, P.W.C.; Takagaki, T.Y. Thumb metastasis from small cell lung cancer treated with radiation. Rev. Hosp. Clin. Fac. Med. Sao. Paulo. 2002, 57, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Raissouni, Z.; Ismael, F.; Elmrini, A.; Boutayeb, F. Acrometastasis of the hand. Case report. Acta Orthop. Belg. 2002, 68, 297–300. [Google Scholar] [PubMed]
- Vadivelu, R.; Drew, S. Phalangeal metastasis secondary to an occult breast carcinoma. Arch. Orthop. Trauma Surg. 2002, 122, 530–531. [Google Scholar] [CrossRef]
- Aguiar Bujanda, D.; Bohn Sarmiento, U.; Aguiar Morales, J. Some Unusual Paraneoplastic Syndromes. J. Clin. Oncol. 2003, 21, 2620. [Google Scholar] [CrossRef]
- Dimri, K.; Rastogi, N.; Lal, P. Carcinoma of esophagus with unusual metastasis to gingiva and phalanx. Indian J. Cancer 2003, 40, 37–38. [Google Scholar]
- Fusetti, C.; Kurzen, P.; Bonaccio, M.; Büchler, U.; Nagy, L. Hand metastasis in renal cell carcinoma. Urology 2003, 62, 141. [Google Scholar] [CrossRef]
- Oron, A.; Reshef, N.; Siegelmann-Danieli, N.; Lin, E.; Aghasi, M. Colon cancer metastasizing to the proximal phalanx of an index finger—A case report. J. Hand Surg. 2003, 28, 26–27. [Google Scholar] [CrossRef]
- Campa, T.; Fagnoni, E.; Ripamonti, C. Palliative surgery of acrometastases from lung cancer: A case report. Support. Care Cancer 2004, 12, 202–204. [Google Scholar] [CrossRef] [PubMed]
- De Smet, L. Late Appearance and Slow Progression of a Breast Carcinoma Metastasis of the Index Metacarpal. Acta Chir. Belg. 2004, 104, 345–346. [Google Scholar] [CrossRef]
- Fontana, T.; Siciliano, M.; Franceschelli, A.; Annicchiarico, B.E.; Rossi, P.; Bigotti, G.; Bombardieri, G. An atypical bone metastasis of hepatocellular carcinoma: Case report and review of the literature. Clin. Ter. 2004, 155, 447–451. [Google Scholar]
- Riter, H.G.; Ghobrial, I.M. Renal Cell Carcinoma with Acrometastasis and Scalp Metastasis. Mayo Clin. Proc. 2004, 79, 76. [Google Scholar] [CrossRef]
- Sahbaz, S.; Kilinç, O.; Vayvada, H.; Topçu, A.; Yörükoğlu, K.; Uçan, E.S. Distal phalanx metastasis in operated bronchial carcinoma. Tuberk. Toraks 2004, 52, 378–381. [Google Scholar] [PubMed]
- Keramidas, E.; Brotherston, M. Extensive metastasis to the hand from undiagnosed adenocarcinoma of the lung. Scand. J. Plast. Reconstr. Surg. Hand Surg. 2005, 39, 113–115. [Google Scholar] [CrossRef]
- Mousavi, S.R.; Ghasemi, A.; Tajodini, A. Metastasis of esophageal cancer to finger. Arch. Iran. Med. 2005, 8, 319–320. [Google Scholar]
- Nakamura, H.; Shimizu, T.; Kodama, K.; Shimizu, H. Metastasis of lung cancer to the finger: A report of two cases. Int. J. Dermatol. 2005, 44, 47–49. [Google Scholar] [CrossRef] [PubMed]
- Tomas, X.; Conill, C.; Combalia, A.; Pomes, J.; Castel, T.; Nicolau, C. Malignant melanoma with metastasis into the capitate. Eur. J. Radiol. 2005, 56, 362–364. [Google Scholar] [CrossRef]
- Akjouj, S.; El Kettani, N.; Semlali, S.; Chaguar, B.; Chaouir, S.; Hanine, A.; Benameur, M. Acrométastase du pouce révélant un adénocarcinome pulmonaire: À propos d’un cas avec revue de la littérature. Chir. Main 2006, 25, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Bülbül, Y.; Özsu, S.; Özlü, T.; Öztuna, F. Acrometastasis in a case with bronchial carcinoma. Toraks. Dergisi. 2006, 7, 200–203. [Google Scholar]
- Carty, H.-M.; Simons, A.W.; Isgar, B. Breast carcinoma bone metastasis first presenting to single middle phalanx. Breast 2006, 15, 127–129. [Google Scholar] [CrossRef]
- Gallagher, B.; Yousef, G.; Bishop, L. Subungual Metastasis from a Rectal Primary: Case Report and Review of the Literature. Dermatologic Surg. 2006, 32, 592–595. [Google Scholar] [CrossRef]
- Gamblin, T.C.; Santos, R.S.; Baratz, M.; Landreneau, R.J. Metastatic colon cancer to the hand. Am. Surg. 2006, 72, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Heidarpour, M.; Rajabi, P.; Eftekhari, A.; Ghasemibasir, H.R. Lung carcinoma metastasis to the distal part of the index finger: A case report. Iran J. Pathol. 2006, 1, 173–176. [Google Scholar]
- Reparaz Padros, J.; Arenas Planelles, A.J.; Martinez Mariscal, J.; Garbayo Marturet, A.J. Acrometástasis. A propósito de dos Casos. Rev. Esp. Cir. Osteoartic. 2006, 41, 39–43. [Google Scholar]
- Afshar, A.; Ayatollahy, H.; Lotfinejad, S. A Rare Metastasis in the Hand: A Case of Cutaneous Metastasis of Choriocarcinoma to the Small Finger. J. Hand Surg. Am. 2007, 32, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Bigot, P.; Desbois, E.; Benoist, N.; Besnier, L.; Moui, Y. Acrométastase révélée par une douleur isolée de la main. À propos d’un cas. Chir. Main 2007, 26, 300–302. [Google Scholar] [CrossRef]
- Elhassan, B.; Fakhouri, A. Metastasis of squamous-cell carcinoma of the lung to the first web space of the hand. J. Bone Joint Surg. Br. 2007, 89-B, 1243–1246. [Google Scholar] [CrossRef]
- French, F.P.; Murray, P.M.; Perdikis, G. Extensive Cutaneous Metastatic Breast Carcinoma of the Hand and Upper Extremity: A Case Report. J. Hand Surg. Am. 2007, 32, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Kanbay, A.; Oguzulgen, K.I.; Ozturk, C.; Memis, L.; Demircan, S.; Kurkcuoglu, C.; Akyurek, N.; Kurul, C. Malignant Pleural Mesothelioma with Scalp, Cerebellar, and Finger Metastases: A Rare Case. South. Med. J. 2007, 100, 63–65. [Google Scholar] [CrossRef]
- Nikolic, I.; Patrnogic, A.; Stojiljkovic, B.; Bogdanovic, B.; Kukic, B. Thumb metastases in a patient with colorectal cancer. Arch. Oncol. 2007, 15, 97–98. [Google Scholar] [CrossRef]
- Preto, A.S.; Teixeira, R.; Cruz, R. Atypical presentation of lung cancer. Acta Reumatol. Port. 2007, 32, 282–286. [Google Scholar]
- Ramseier, L.E.; Dumont, C.E.; Ulrich Exner, G. Multiple subungual soft tissue metastases from a chondrosarcoma. Scand. J. Plast. Reconstr. Surg. Hand Surg. 2007, 41, 332–333. [Google Scholar] [CrossRef] [PubMed]
- Salesi, N.; Russillo, M.; Marandino, F.; Ruggeri, E.M.; Metro, G.; Cognetti, F.; Fabi, A. Bone of the hands as unusual metastastatic site of renal cell carcinoma. J. Exp. Clin. Cancer Res. 2007, 26, 595–597. [Google Scholar] [PubMed]
- Ahlmann, E.R.; Greene, N.W.; Menendez, L.R.; Stevanovic, M.V. Unusual locations for metastatic malignancy of the hand: A report of three cases. J. Surg. Orthop. Adv. 2008, 17, 267–270. [Google Scholar] [PubMed]
- Chen, T.-J.; Chiou, C.-C.; Chen, C.-H.; Kuo, T.; Hong, H.-S. Metastasis of Mediastinal Epithelioid Angiosarcoma to the Finger. Am. J. Clin. Dermatol. 2008, 9, 181–183. [Google Scholar] [CrossRef]
- Gaston, R.G.; Lourie, G.M.; Scott, C.C. Isolated metastatic lesion of the trapezium. Am. J. Orthop. 2008, 37, E144–E145. [Google Scholar]
- Miyamoto, W.; Yamamoto, S.; Uchio, Y. Metastasis of gastric cancer to the fifth metacarpal bone. Hand Surg. 2008, 13, 193–195. [Google Scholar] [CrossRef]
- Ottomani, S.; Levadoux, M.; Cathelineau, O.; Nguyen, M.; Monchal, T. Double localisation métastatique d’un carcinome épidermoïde au niveau de la pulpe digitale. À propos d’un cas et revue de la littérature. Chir. Main 2008, 27, 243–245. [Google Scholar] [CrossRef]
- Plotkine, E.; Coscas, R.; Guigui, P. Digital metastatis of a nasopharyngeal carcinoma: A surgical trap. Chir. Main 2008, 27, 187–190. [Google Scholar] [CrossRef]
- Seth, R.; Athanassopoulos, A.; Mir, S. First Presentation of Lung Adenocarcinoma as a Subungual Metastasis. HAND 2008, 3, 69–71. [Google Scholar] [CrossRef]
- Tzaveas, A.; Paraskevas, G.; Pazis, I.; Dimitriadis, A.; Kitsoulis, P.; Vrettakos, A. Metastasis of bronchogenic carcinoma to the 5th metacarpal bone: A case report. Cases J. 2008, 1, 284. [Google Scholar] [CrossRef]
- Jakhar, S.; Dana, R.; Punia, D. Cancer lung: An unusual presentation. Indian J. Med. Paediatr. Oncol. 2009, 30, 149. [Google Scholar] [CrossRef][Green Version]
- Kodama, T.; Kikuchi, N.; Satoh, H.; Ohtsuka, M. Metacarpal Bone Metastasis from Lung Cancer. Oncol. Res. Treat. 2009, 32, 216–217. [Google Scholar] [CrossRef] [PubMed]
- Madjidi, A.; Cole, P.; Laucirica, R. Digital acrometastasis: A rare initial sign of occult pulmonary squamous cell carcinoma. J. Plast. Reconstr. Aesthetic Surg. 2009, 62, e365–e367. [Google Scholar] [CrossRef] [PubMed]
- Mitrovic, R.; Pakevic, N.; Jeremic, B.; Cesarevic, D.; Živanović, P. Renal cell carcinoma metastasis to hand: Rare localisations of solitary metastases. Eur. Urol. 2009, 8, 632. [Google Scholar] [CrossRef]
- Otsuji, M.; Matsunaga, S.; Koga, H.; Kawabata, N.; Imakiire, T.; Hiwaki, T.; Tashiro, Y.; Shirahama, H.; Komiya, S. An atypical extrahepatic metastasis of the distal phalanx from hepatocellular carcinoma. Int. J. Clin. Oncol. 2009, 14, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Rauh, M.A.; Duquin, T.R.; McGrath, B.E.; Mindell, E.R. Spread of Squamous Cell Carcinoma From the Thumb to the Small Finger via the Flexor Tendon Sheaths. J. Hand Surg. Am. 2009, 34, 1709–1713. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, R.; Singh, K.; Umbarker, B.; Karle, R.; Shrivastava, M. Phalange metastasis from carcinoma of alveolus. Indian J. Dent. Res. 2009, 20, 496. [Google Scholar] [CrossRef] [PubMed]
- Wavreille, G.; Baroncini, M.; Rtaimate, M. Une cause rare de pseudohippocratisme digital: L’acrométastase. À propos d’un cas. Chir. Main 2009, 28, 381–383. [Google Scholar] [CrossRef]
- Afshar, A.; Ilkhanizadeh, B. Leukaemia cutis of the distal phalanx of the right thumb. J. Hand Surg. 2010, 35, 153–154. [Google Scholar] [CrossRef]
- Anoop, T.M.; George, S.; Divya, K.P.; Jabbar, P.K. Metastatic phalangeal osteolysis as an initial presentation of carcinoma colon. Am. J. Surg. 2010, 200, e61–e63. [Google Scholar] [CrossRef] [PubMed]
- Anglada-Curado, F.J.; De Haro-Padilla, J.; Carrasco-Valiente, J.; Alvarez-Kindelan, J.; Ruiz-Garcia, J.; Requena-Tapia, M.J. Hand Metastasis From Renal Carcinoma. Urology 2010, 76, 846. [Google Scholar] [CrossRef] [PubMed]
- Biyi, A.; Oufroukhi, Y.; Doudouh, A. Acrométastases de la main et du pied compliquant un cancer mammaire. Chir. Main 2010, 29, 40–43. [Google Scholar] [CrossRef]
- Borobio León, G.; García Plaza, A.; García Cepeda, I.; González Alconada, R.; Hernández Cosido, L. Metástasis en mano de adenocarcinoma de recto. Un caso excepcional. Cirugía Española 2010, 88, 195–197. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, V.K.; Rao, N.D.C.; Chandra, S.T. Study of papillary carcinoma of thyroid with uncommon sites of metastasis. Indian J. Otolaryngol. Head Neck Surg. 2010, 62, 198–201. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Long, L.S.; Brickner, L.; Helfend, L.; Wong, T.; Kubota, D. Lung Cancer Presenting as Acrometastasis to the Finger: A Case Report. Case Rep. Med. 2010, 2010, 1–3. [Google Scholar] [CrossRef]
- Lozić, A.A.B.; Silconi, Z.B.; Misljenović, N. Metastases to rare locations as the initial manifestation of non-small cell lung cancer: Two case reports. Coll. Antropol. 2010, 34, 609–612. [Google Scholar]
- Lucilli, N.; Mattacheo, A.; Palacios, A. Acrometástasis por cáncer de pulmón. A propósito de un caso. Arch. Bronconeumol. 2010, 46, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Myrehaug, S.; Bezjak, A. Rapidly progressive bone destruction of the finger as first presentation of systemic metastases from lung cancer. Case Rep. 2010, 2010, bcr0520091912. [Google Scholar] [CrossRef]
- Vasic, L. Osteolysis of hand bones due to metastatic deposits from colon cancer: A case report. Med. Pregl. 2010, 63, 719–722. [Google Scholar] [CrossRef][Green Version]
- Wurapa, R.K.; Bickel, B.A.; Mayerson, J.; Mowbray, J.G. Metastatic esophageal adenocarcinoma of the carpus. Am. J. Orthop. 2010, 39, 283–285. [Google Scholar]
- Amar, M.F.; Almoubaker, S.; Lahrach, K.; Chbani, B.; Bennani, A.; Marzouki, A.; Boutayeb, F. Tumeur géante du pouce révélant un adénocarcinome pulmonaire (à propos d’un cas). Chir. Main 2011, 30, 133–135. [Google Scholar] [CrossRef]
- Çetın, B.; Büyükberber, S.; Yüksel, M.; Coşkun, U.; Yildiz, R.; Beneklı, M. Metastasis of rectal cancer to soft tissue of the hand: An unusual case. Turk. J. Gastroenterol. 2011, 22, 229–230. [Google Scholar] [CrossRef]
- Chao, D.; Harinarayanan, S.; Reynolds, D. The acromet. J. Thorac. Dis. 2011, 3, 209–210. [Google Scholar] [CrossRef] [PubMed]
- Elvey, M.H.; Aghasi, M.; Wasrbrout, Z.; Avisar, E. Metastasis of parotid basal cell adenocarcinoma to the hand-a case report. Hand 2011, 6, 321–323. [Google Scholar] [CrossRef] [PubMed]
- Huri, G. An atypical metastasis of follicular-type adenocarcinoma of the thyroid gland to thumb. Case Rep. Orthop. 2011, 2011, 735789. [Google Scholar] [CrossRef]
- Jenzer, A.; Badur, N.; Vögelin, E. Composite bone cement arthrodesis in acrometastasis of the proximal phalanx of the hand—A case report. J. Orthop. Surg. 2011, 19, 1–6. [Google Scholar]
- Kumar, R.; Kumar, P.; Ghosal, S.; Sharma, S.; Kumar, N.; Bera, A.; Angurana, S.; Srinivasan, R. Palliative and supportive care in acrometastasis to the hand: Case series. Indian J. Palliat. Care 2011, 17, 241. [Google Scholar] [CrossRef] [PubMed]
- Taleb, C.; Pelissier, P.; Choughri, H. Bladder urothelial carcinoma with acrometastasis: A case report and review of the literature. Chir. Main 2011, 30, 136–139. [Google Scholar] [CrossRef] [PubMed]
- Corrales Pinzón, R.; Alonso Sánchez, J.M.; de la Mano González, S.; El Karzazi Tarazona, K. A single metastasis in the carpal bones as the first clinical manifestation of a hepatocellular carcinoma. Radiologia 2014, 56, e42–e45. [Google Scholar] [CrossRef] [PubMed]
- Fadli, A.R.; Azmi, M.N.; David, O.; Zailani, M.H. Isolated metacarpal bone metastasis from advanced rectosigmoid carcinoma. Int. J. Malaysia. 2012, 11, 59–61. [Google Scholar]
- Ferraro, D.; Lucero, P. Metastatic lung adenocarcinoma presenting as thumb pain. Chest 2012, 142, 567A. [Google Scholar] [CrossRef]
- Gharwan, H.; Yarlagadda, L.; Duffy, A. Acrometastasis as the Initial Presentation of a KRAS -Positive Colon Cancer. Case Rep. Oncol. 2012, 5, 404–408. [Google Scholar] [CrossRef]
- Kamolz, L.-P.; Stiglbauer, W.; Längle, F. Palmar metastasis of an adenocarcinoma of the esophago-gastric-junction: First case report. Int. J. Surg. Case Rep. 2012, 3, 412–414. [Google Scholar] [CrossRef][Green Version]
- Kim, J.I.; Song, C.H.; Gong, H.S. Finger skin metastasis from hepatocellular carcinoma: A case report. Hand Surg. 2012, 17, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Ornetti, P.; Favier, L.; Varbedian, O.; Ansemant, T. Digital acrometastasis revealing endometrial cancer relapse. Arthritis Rheum. 2012, 64, 3167. [Google Scholar] [CrossRef] [PubMed]
- Rauf, M.S.; Motta, L.; Connolly, C. Digital acrometastases as first sign of hepatocellular carcinoma. Scott. Med. J. 2012, 57, 247. [Google Scholar] [CrossRef]
- Rinonapoli, G.; Caraffa, A.; Antenucci, R. Lung cancer presenting as a metastasis to the carpal bones: A case report. J. Med. Case Rep. 2012, 6, 384. [Google Scholar] [CrossRef]
- Roushdi, I.; Jeswani, T.; Clark, D. Lymphoma presenting as a metastasis to the hand. J. Hand Surg. Eur. Vol. 2012, 37, 286–287. [Google Scholar] [CrossRef]
- Stahl, S.; Santos Stahl, A.; Lotter, O.; Pfau, M.; Perner, S.; Schaller, H.-E. Palliative surgery for skeletal metastases from melanoma in the scaphoid--a critical case report appraisal. J. Plast. Reconstr. Aesthet. Surg. 2012, 65, 1111–1115. [Google Scholar] [CrossRef]
- Tan, K.T.; Simpson, C.; Chandrasekar, C.R. Hand metastasis: An unusual presentation of renal cell carcinoma. Hand 2012, 7, 204–206. [Google Scholar] [CrossRef]
- Khmamouche, M.R.; Aassab, R.; Debbagh, A.; Elbahraouy, A.; Mahfoud, T.; Tanz, R.; Ichou, M.; Errihani, H.; Khmamouche, M.R.; Aassab, R.; et al. A thumb acrometastasis revealing lung adenocarcinoma: A case report and review. J. Clin. Case Rep. 2013, 3, 100–101. [Google Scholar]
- Krishnamurthy, A.; Ramshankar, V. Metastatic iodophilic follicular carcinoma of thyroid to a hand bone. Thyroid. Res. Pr. 2013, 10, 36–37. [Google Scholar] [CrossRef]
- Yoneda, S.; Kinjo, T.; Nonomura, D.; Yamamoto, Y.; Nomura, H.; Tei, N.; Takada, S.; Matsumiya, K.; Fujioka, H. A case of bladder cancer with metastasis to the bone of the hand. Hinyokika Kiyo. 2013, 59, 129–131. [Google Scholar] [PubMed]
- Gilardi, R.; Della Rosa, N.; Pancaldi, G.; Landi, A. Acrometastasis showing an occult lung cancer. J. Plast. Surg. Hand Surg. 2013, 47, 550–552. [Google Scholar] [CrossRef] [PubMed]
- Rommer, E.; Leilabadi, S.N.; Lam, G.; Soltani, A.; Ellis, C.V.; Rizvi, M.; Wong, A.K. Metastasis of hepatocellular and renal cell carcinoma to the hand. Plast. Reconstr. Surgery. Glob. Open 2013, 1, e83. [Google Scholar] [CrossRef] [PubMed]
- van Veenendaal, L.M.; de Klerk, G.; van der Velde, D. A painful finger as first sign of a malignancy. Geriatr. Orthop. Surg. Rehabil. 2014, 5, 18–20. [Google Scholar] [CrossRef]
- Hernández-Cortés, P.; Caba-Molina, M.; Gómez-Sánchez, R.; Ríos-Peregrina, R. Renal Clear Cell Carcinoma Acrometastasis. An Unusual Terminal Condition. J. Hand Microsurg. 2015, 7, 149–151. [Google Scholar] [CrossRef]
- Sumodhee, S.; Huchot, E.; Peret, G.; Marchal, C.; Paganin, F.; Magnin, V. Radiotherapy for a phalanx bone metastasis of a lung adenocarcinoma. Case Rep. Oncol. 2014, 7, 727–731. [Google Scholar] [CrossRef]
- Liu, W.-C.; Ho, C.-J.; Lu, C.-K.; Wu, C.-C.; Fu, Y.-C.; Chien, S.-H. Acrometastasis to metacarpal bone disclosing an occult lung cancer. Case reports Plast. Surg. hand Surg. 2014, 1, 23–25. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lambe, G.; Le, P.; Clay, T.D. A finding with a diagnosis: I just can’t put my finger on it. BMJ Case Rep. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Gorospe Sarasúa, L.; Mezquita Pérez, L.; Pacios Blanco, R.E. Acrometastasis as the initial presentation of lung adenocarcinoma in a young woman. Arch. Bronconeumol. 2016, 52, 482–483. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.; Skandan, S.P. An uncommon presentation of non-small-cell lung cancer with acrometastases to the great toe and index finger. J. community Support. Oncol. 2016, 14, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, T.K.; Das, S.K.; Majumdar, S.K.D.; Senapati, S.N.; Parida, D.K. Digital Acrometastasis as Initial Presentation in Carcinoma of Lung A Case Report and Review of Literature. J. Clin. Diagn. Res. 2016, 10, XD01–XD02. [Google Scholar] [CrossRef]
- Asirvatham Gjorup, C.; Ottesen, S.S.; Gjerloff, C. Acrometastasis as the first sign of an occult non-small cell carcinoma of lung. J. Plast. Surg. Hand Surg. 2017, 51, 156–157. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Mahamud, E.; Combalia, A.; Carreño, A.; Arandes, J.M. Five cases of acrometastasis to the hand from a carcinoma and review of the literature. Hand Surg. Rehabil. 2017, 36, 12–16. [Google Scholar] [CrossRef]
- Xie, P.; Huang, J. Solitary Thumb Acrometastasis Identified on Bone Scintigraphy. Clin. Nucl. Med. 2017, 42, 549–550. [Google Scholar] [CrossRef]
- Lechmiannandan, S.; Aida, N.; Paul, A.G.; Thevarajah, S. “Thumb’s off” for acrometastasis of renal cell carcinoma: Is there a role for acrometastasectomy in the era of targeted therapy? Asian J. Urol. 2018, 5, 199–201. [Google Scholar] [CrossRef]
- El Idrissi, M.; Akasbi, N.; Elibrahimi, A.; Elmrini, A. Acrometastasis as the first manifestation of lung carcinoma. Eur. J. Rheumatol. 2018, 5, 212–213. [Google Scholar] [CrossRef]
- Tabrizi, A.; Afshar, A.; Shariyate, M.J.; Gharalari, F.H.; Aidenlou, A. Isolated Metastatic Carcinoma to the Hamate Bone: The First Manifestation of an Occult Malignancy. J. Hand Microsurg. 2019, 11, S01–S05. [Google Scholar] [CrossRef] [PubMed]
- Voskuil, R.T.; Smith, J.R.; Swafford, R.E.; Jemison, D.M. Colon adenocarcinoma with metastases to the scaphoid: A case report and review of the literature. J. Surg. Case Rep. 2019, 2019, rjz011. [Google Scholar] [CrossRef] [PubMed]
- Hirano, Y.; Sato, R.; Ohshima, N.; Matsui, H. Acrometastasis of Lung Cancer. Intern. Med. 2020, 59, 1919–1920. [Google Scholar] [CrossRef] [PubMed]
- Cruz, D.; Wild, T.; Glavynskyi, I.; Weissenberg, K.; Frenzel, S.; Florschütz, A.; Winter, J. Unusual Manifestation of Chronic Lymphocytic Leukemia in the Hand. J. Hand Surg. Am. 2021, 46, 74.e1–74.e8. [Google Scholar] [CrossRef]
- Gallardo-Alvarado, L.; Ramos, A.A.; Perez-Montiel, D.; Ramirez-Morales, R.; Diaz, E.; Leon, D.C. Hand metastasis in a patient with cervical cancer: A case report. Medicine 2020, 99, e20897. [Google Scholar] [CrossRef] [PubMed]
- Flynn, C.J.; Danjoux, C.; Wong, J.; Christakis, M.; Rubenstein, J.; Yee, A.; Yip, D.; Chow, E. Two Cases of Acrometastasis to the Hands and Review of the Literature. Curr. Oncol. 2008, 15, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, P.N.; Rangad, F.; Roul, R.K. Metastases to the Hand from Carcinoma of the Lower Alveolus. J. Hand Surg. Am. 1996, 21, 544–546. [Google Scholar] [CrossRef]
- Tolo, E.T.; Cooney, W.P.; Wenger, D.E. Renal cell carcinoma with metastases to the triquetrum: Case report. J. Hand Surg. Am. 2002, 27, 876–881. [Google Scholar] [CrossRef]
- Shannon, F.J.; Antonescu, C.R.; Athanasian, E.A. Metastatic thymic carcinoma in a digit: A case report. J. Hand Surg. Am. 2000, 25, 1169–1172. [Google Scholar] [CrossRef]
- Dar, A.M.; Kawoosa, N.u.N.; Sharma, M.L.; Bhat, M.A. Unusual metastasis to all the digits of both hands in a patient previously operated on for esophageal carcinoma. Gen. Thorac. Cardiovasc. Surg. 2011, 59, 225–227. [Google Scholar] [CrossRef]
- Bahk, W.-J.; Rhee, S.-K.; Kang, Y.-K.; Lee, A.-H.; Park, J.-M.; Chung, Y.-G. Gastric cancer acrometastases to all digits of one hand following closed intramedullary nailing. Skeletal Radiol. 2006, 35, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Sipahioglu, S.; Zehir, S.; Ozkanli, U. Nasopharyngeal carcinoma with hand metastasis. J. Hand Surg. (European Vol.) 2012, 37, 578–579. [Google Scholar] [CrossRef]
- De Abaffy, A.M.; Richter, R.H.; Grünert, J. Peripheral bone metastasis of a rare lung cancer. Arch. Orthop. Trauma Surg. 1998, 117, 477–478. [Google Scholar] [CrossRef] [PubMed]
- Mátrai, Z.; Péley, G.; Rényi Vámos, F.; Farkas, E.; Kovács, T.; Köves, I. The similarities between the mechanism of wound healing and tumor development--literature review on the occasion of a patient with colonic adenocarcinoma metastasis in a dog-bite wound. Orv. Hetil. 2005, 146, 99–109. [Google Scholar] [PubMed]
- Lewin, J.S.; Cleary, K.R.; Eicher, S.A. An Unusual Metastasis to the Thumb in a Laryngectomized Tracheoesophageal Speaker. Arch. Otolaryngol.—Head Neck Surg. 1997, 123, 1007–1009. [Google Scholar] [CrossRef] [PubMed]
- Ozcanli, H.; Ozdemir, H.; Ozenci, A.M.; Söyüncü, Y.; Aydin, A.T. Metastatic tumors of the hand in three cases. Acta Orthop. Traumatol. Turc. 2005, 39, 445–448. [Google Scholar] [PubMed]
- Hsieh, C.-Y.; Bai, L.-Y.; Lo, W.-C.; Huang, H.-H.; Chiu, C.-F. Esophageal Squamous Cell Carcinoma with a Solitary Phalangeal Metastasis. South. Med. J. 2008, 101, 1159–1160. [Google Scholar] [CrossRef] [PubMed]
- Stone, R.; Davies, J. Isolated digital metastasis in bronchogenic carcinoma. Palliat. Med. 1990, 4, 137–138. [Google Scholar] [CrossRef]
- Bhandari, T.; Brown, E. Acrometastasis and the Potential Benefits of Early Positron Emission Tomography Scanning. Ann. Plast. Surg. 2011, 67, 189–192. [Google Scholar] [CrossRef]
- Reichert, B.; Hoch, J.; Plötz, W.; Mailänder, P.; Moubayed, P. Metastatic Clear-Cell Sarcoma of the Capitate. J. Bone Jt. Surgery-American Vol. 2001, 83, 1713–1717. [Google Scholar] [CrossRef]
- Khosla, D.; Rai, B.; Patel, F.D.; Sapkota, S.; Srinvasan, R.; Sharma, S.C. Acrometastasis to hand in vaginal carcinoma: A rare entity. J. Cancer Res. Ther. 2012, 8, 430–432. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Yao, J. Trapezial Metastasis as the First Indication of Primary Non–small Cell Carcinoma of the Lung. J. Hand Surg. Am. 2012, 37, 1242–1244. [Google Scholar] [CrossRef] [PubMed]
- Ghert, M.A.; Harrelson, J.M.; Scully, S.P. Solitary renal cell carcinoma metastasis to the hand: The need for wide excision or amputation. J. Hand Surg. Am. 2001, 26, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Kosuda, S.; Gokan, T.; Tamura, K.; Dokiya, T.; Kubo, A.; Hashimoto, S. Radionuclide Imaging of Two Patients with Metastasis to a Distal Phalanx of the Hand. Clin. Nucl. Med. 1986, 11, 659–660. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, S.; Creditor, M. Metastasis to the hand. J. Natl. Med. Assoc. 1986, 78, 441–442. [Google Scholar] [PubMed]
- Buckley, N.; Brown, P. Metastatic tumors in the hand from adenocarcinoma of the colon. Dis. Colon Rectum 1987, 30, 141–143. [Google Scholar] [CrossRef]
- Hindley, C.J.; Metcalfe, J.W. A colonic metastatic tumor in the hand. J. Hand Surg. Am. 1987, 12, 803–805. [Google Scholar] [CrossRef]
- Bourne, M.H.; Amadio, P.C.; Wold, L.E.; Sim, F.H. Metastatic lesions of the hand: Report of a case. Orthopedics 1988, 11, 219–221. [Google Scholar] [CrossRef]
- Craigen, M.; Chesney, R. Metastatic adenocarcinoma of the carpus: A case report. J. Hand Surg. J. Br. Soc. Surg. Hand 1988, 13, 306–307. [Google Scholar] [CrossRef]
- Dalicho, F.H.; Beckmann, D.; Hübner, L. Metastasis of cervical cancer to the end phalanx of the small finger. Zentralbl. Gynakol. 1988, 110, 111–114. [Google Scholar]
- Haas, O.; Bernard, A.; Cougard, P.; Viard, H. Phalangeal metastasis of cancer of the esophagus. Apropos of a case. Ann. Chir. 1988, 42, 37–38. [Google Scholar] [PubMed]
- Müller, S.; Dörner, A.; Dallek, M.; Supra, T. Handmetastasen—eine seltene Metastasierungsform des kolorektalen Karzinoms. DMW—Dtsch. Medizinische Wochenschrift 2008, 113, 728–730. [Google Scholar] [CrossRef]
- Sanjay, B.; Raj, G.A.; Vishwakarma, G. A small-cell osteosarcoma with multiple skeletal metastases. Arch. Orthop. Trauma. Surg. 1987, 107, 58–60. [Google Scholar] [CrossRef]
- Tenenbaum, F.; Reverberi, J.; Vacher, B.; Pulik, M. Metastasis in the carpal bones and cancer of the esophagus. J. Chir. 1988, 125, 443–444. [Google Scholar]
- Heymans, M.; Jardon-Jeghers, C.; Vanwijck, R. Hand metastases from urothelial tumor. J. Hand Surg. Am. 1990, 15, 509–511. [Google Scholar] [CrossRef]
- Lederer, A.; Flückiger, F.; Wildling, R.; Fruhwirth, J. A solitary metastasis in the trapezium bone. Radiologe 1990, 30, 79–80. [Google Scholar] [PubMed]
- Turan, I.; Sjöden, G.O.J.; Kalen, A. Ovarian carcinoma metastasis to the little finger. Acta Orthop. Scand. 1990, 61, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Desmanet, E.; Amrani, M.; Fievez, R.; Six, C. Les acrométastases. Ann. Chir. la Main du Memb. Supérieur 1991, 10, 154–157. [Google Scholar] [CrossRef]
- Henkert, K.; Berge, G. Malignant metastases on the hand. Zentralbl. Chir. 1991, 116, 337–341. [Google Scholar] [PubMed]
- Moutet, F.; Bellon-Champel, P.; Lebrun, C.; Sarrazin, R. A propos d’une métastase isolée au grand os. Ann. Chir. la Main du Memb. Supérieur 1991, 10, 148–150. [Google Scholar] [CrossRef]
- Rochet, N.; Pages, M.; Lassoued, S.; Poey, C.; Fournié, B.; Fournié, A. Phalangeal metastasis of the hand. Apropos of a case. Rev. Rhum. Mal. Osteoartic. 1991, 58, 73–74. [Google Scholar]
- Troncoso, A.; Ro, J.Y.; Grignon, D.J.; Han, W.S.; Wexler, H.; von Eschenbach, A.; Ayala, A.G. Renal cell carcinoma with acrometastasis: Report of two cases and review of the literature. Mod. Pathol. 1991, 4, 66–69. [Google Scholar]
- Bloom, R.A.; Sulkes, A.; Freilick, G.; Libson, E. Breast metastases to bones of the extremities: Simultaneous involvement of all four limbs. Clin. Oncol. 1992, 4, 58–59. [Google Scholar] [CrossRef]
- DiSpaltro, F.X.; Bickley, L.K.; Nissenblatt, M.J.; Devereux, D. Cutaneous acral metastasis in a patient with primary gastric adenocarcinoma. J. Am. Acad. Dermatol. 1992, 27, 117–118. [Google Scholar] [CrossRef]
- Hayes, M.M.; Jones, E.C.; Verma, A.K.; Lim, C.H.; Milne, G.; Tse, E. Transitional cell carcinoma of the renal pelvis metastatic to the metacarpal. A case report correlating cytologic and histologic findings. Acta Cytol. 1992, 36, 946–950. [Google Scholar]
- Jebson, P.; Buckwalter, J.; Blair, W.; Platz, C. Hand metastasis from renal carcinoma. Iowa Orthop. J. 1992, 12, 85–87. [Google Scholar]
- Kobus, R.J.; Leinberry, C.; Kirkpatrick, W.H. Metastatic renal carcinoma in the hand: Treatment with preoperative irradiation and ray resection. Orthop. Rev. 1992, 21, 983–984. [Google Scholar] [PubMed]
- Rousseau, A.; Madinier, J.F.; Favre, A.; Michenet, P. Tumeurs métastatiques des parties molles de la main A propos d’un cas. Revue de la littérature. Ann. Chir. la Main du Memb. Supérieur 1992, 11, 57–61. [Google Scholar] [CrossRef]
- Bibi, C.; Benmeir, P.; Maor, E.; Sagi, A. Hand Metastatis from Renal Cell Carcinoma with No Bone Involvement. Ann. Plast. Surg. 1993, 31, 377–378. [Google Scholar] [CrossRef]
- Marya, S.K.; Singh, S.; Jaswal, T.S.; Garg, P.; Gupta, A.K. Digital and clavicular bone metastasis from transitional cell carcinoma of urinary bladder: A case report. Indian J. Pathol. Microbiol. 1993, 36, 75–77. [Google Scholar] [PubMed]
- Roncaglio, C.; Arena, B. Metastasi alla mano da carcinoma broncopolmonare. Minerva Ortop. Traumatol. 1993, 44, 37–43. [Google Scholar]
- Knapp, D.; Abdul-Karim, F.W. Fine needle aspiration cytology of acrometastasis. A report of two cases. Acta Cytol. 1994, 38, 589–591. [Google Scholar] [PubMed]
- Walsh, T.M.; Mileski, R.A.; Ferlic, T.P. Metastatic transitional cell carcinoma to the hand. J. Hand Surg. Am. 1994, 19, 806–808. [Google Scholar] [CrossRef]
- De Maeseneer, M.; Machiels, F.; Naegels, S.; Verhaeghe, W. Hand and foot acrometastases in a patient with bronchial carcinoma. J. Belge Radiol. 1995, 78, 274–275. [Google Scholar]
- Castello, J.R.; Garro, L.; Romero, F.; Campo, M.; Najera, A. Metastatic Tumours of the Hand. J. Hand Surg. Am. 1996, 21, 547–550. [Google Scholar] [CrossRef]
- Hetzel, D.J.; Olt, G.J.; Sorosky, J.I.; Mortel, R.; Singapuri, K.; Podczaski, E.S. Gestational Trophoblastic Disease Presenting asa Large Metastasis to the Finger. Gynecol. Oncol. 1996, 63, 123–126. [Google Scholar] [CrossRef]
- Königsberger, H.; Goth, D. Bone metastasis of bronchial carcinoma to the wrist. Case report. Handchir. Mikrochir. Plast. Chir. 1996, 28, 46–49. [Google Scholar]
- Saglike, Y.; Demirtas, M.; Demirors, H. Metastatic tumours of the hand. Int. Orthop. 1996, 20, 123–124. [Google Scholar] [CrossRef]
- Vine, J.E.; Cohen, P.R. Renal cell carcinoma metastatic to the thumb: A case report and review of subungual metastases from all primary sites. Clin. Exp. Dermatol. 1996, 21, 377–380. [Google Scholar] [CrossRef]
- Bauer, P.; Saalfeld, U.; Schmidt, G.; Partecke, B.D. A rare case of metastasis of urinary bladder carcinoma in the pisiform bone. A case report. Handchir. Mikrochir. Plast. Chir. 1997, 29, 154–157. [Google Scholar]
- Hatakeyama, S.; Tachibana, A.; Suzuki, K.; Okano, H. A case of metastasis to the metacarpal bone of the right thumb from squamous cell carcinoma of the lung. Jpn. J. Lung Cancer 1997, 37, 531–535. [Google Scholar] [CrossRef][Green Version]
- Méndez López, J.M.; García Mas, R.; Salvà Coll, G. Metastasis of an adenocarcinoma of the colon to the 1st metacarpal bone. Ann. Chir. Main Memb. Super. 1997, 16, 134–137. [Google Scholar]
- Rümenapf, G.; Rupprecht, H.; Groitl, H.; Grunert, J.; Hohenberger, W. Fingermetastase eines Kolonkarzinoms. Coloproctology 1997, 19, 169–172. [Google Scholar] [CrossRef]
- Baron, J.M.; Borrego, P.; Solis, J.A. Metastasis en la mano, a proposito de un caso. Oncologia 1998, 21, 184–186. [Google Scholar]
- Delsmann, B.; Lienemann, A.; Nerlich, A.; Hoffmann, E.; Caselmann, W.; Refior, H. Primärmanifestation eines hepatozellulären Carcinoms als osteolytische Handmetastase—Ein Fallbericht. Z. Orthop. Ihre Grenzgeb. 2008, 136, 571–573. [Google Scholar] [CrossRef]
- Baran; Guillot; Tosti Metastasis from carcinoma of the bronchus to the distal aspect of two digits. Br. J. Dermatol. 1998, 138, 708. [CrossRef] [PubMed]
- Celik, G.; Saryal, S.; Enacar, N. Mixed squamous and small cell lung carcinoma with separate histologic subtypes in eye and phalanx metastases. J. Exp. Clin. Cancer Res. 1998, 17, 129–130. [Google Scholar]
- Chirodian, N.; Dickson, M.G.; Kerr, P.S. Fingertip metastasis presenting with a history of trauma. Hosp. Med. 1998, 59, 819. [Google Scholar] [PubMed]
- Galmarini, C.; Kertesz, A.; Oliva, R.; Porta, J.; Galmarini, F.C. Metastasis of bronchogenic carcinoma to the thumb. Med. Oncol. 1998, 15, 282–285. [Google Scholar] [CrossRef] [PubMed]
- Massraf, A.B.; Wand, J.S. Haemorrhagic secondary prostatic metastasis of the terminal phalanx of the thumb. Injury 1998, 29, 243–245. [Google Scholar] [CrossRef]
- Adegboyega, P.A.; Adesokan, A.; Viegas, S.F. Acrometastasis in Renal Cell Carcinoma. South. Med. J. 1999, 92, 1009–1012. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.E.; Choi, J.H.; Sung, K.J.; Moon, K.C.; Koh, J.K. Metastatic squamous cell carcinoma of the nail bed: A presenting sign of lung cancer. Br. J. Dermatol. 1999, 141, 939–940. [Google Scholar] [CrossRef]
- Giberti, C.; Mantero, R.; Schienone, M.; Costa, M.; Lavagna, M. Unusual location for secondary bone metastasis from advanced renal cell carcinoma. Acta Urol. Ital. 1999, 13, 177–178. [Google Scholar]
- Lee, K.-S.; Lee, S.-H.; Kang, K.-H.; Oh, K.-J. Metastatic hepatocellular carcinoma of the distal phalanx of the thumb: A case report. Hand Surg. 1999, 4, 95–100. [Google Scholar] [CrossRef]
- Okada, H.; Weller, M.; Huang, R.; Finocchiaro, G.; Gilbert, M.R.; Wick, W.; Ellingson, B.M.; Hashimoto, N.; Pollack, I.F.; Brandes, A.A.; et al. Immunotherapy response assessment in neuro-oncology: A report of the RANO working group. Lancet Oncol. 2015, 16, e534–e542. [Google Scholar] [CrossRef]
- Vanhooteghem, O.; Dumont, M.; André, J.; Leempoel, M.; de la Brassinne, M. Bilateral subungual metastasis from squamous cell carcinoma of the lung: A diagnostic trap! Rev. Med. Liege 1999, 54, 653–654. [Google Scholar] [PubMed]
- Esther, R.J.; Bos, G.D. Management of metastatic disease of other bones. Orthop. Clin. North Am. 2000, 31, 647–659. [Google Scholar] [CrossRef]
- Hayden, R.J.; Sullivan, L.G.; Jebson, P.J. The hand in metastatic disease and acral manifestations of paraneoplastic syndromes. Hand Clin. 2004, 20, 335–343. [Google Scholar] [CrossRef]
- Umana, G.E.; Passanisi, M.; Tranchina, M.G.; Fricia, M.; Nicoletti, G.F.; Cicero, S.; Scalia, G. Letter to the Editor Regarding “Rare Thymoma Metastases to the Spine: Case Reports and Review of the Literature. World Neurosurg. 2020, 138, 567–569. [Google Scholar] [CrossRef]
- Afshar, A.; Farhadnia, P.; Khalkhali, H. Metastases to the Hand and Wrist: An Analysis of 221 Cases. J. Hand Surg. Am. 2014, 39, 923–932. [Google Scholar] [CrossRef]
- Ponzo, G.; Umana, G.E.; Giuffrida, M.; Furnari, M.; Nicoletti, G.F.; Scalia, G. Intramedullary craniovertebral junction metastasis leading to the diagnosis of underlying renal cell carcinoma. Surg. Neurol. Int. 2020, 11, 152. [Google Scholar] [CrossRef] [PubMed]
- Fiori, R.; Coco, I.; Nezzo, M.; Kabunda, G.; Umana, G.E.; Fraioli, M.F.; Simonetti, G. Spinal Hydatidosis Relapse: A Case Report. Case Rep. Orthop. 2014, 2014, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Passanisi, M.; Scalia, G.; Palmisciano, P.; Franceschini, D.; Crea, A.; Capone, C.; Tranchina, M.G.; Nicoletti, G.F.; Cicero, S.; Umana, G.E. Difficulty differentiating between a posterior extradural lumbar tumor versus sequestered disc even with gadolinum-enhanced MRI. Surg. Neurol. Int. 2021, 12, 267. [Google Scholar] [CrossRef] [PubMed]
- Libson, E.; Bloom, R.A.; Husband, J.E.; Stoker, D.J. Metastatic tumours of bones of the hand and foot. Skeletal Radiol. 1987, 16, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Kerin, R. The hand in metastatic disease. J. Hand Surg. Am. 1987, 12, 77–83. [Google Scholar] [CrossRef]
- Sonoda, L.I.; Halim, M.Y.; Balan, K.K. Solitary Phalangeal Metastasis of Renal Cell Carcinoma on Bone Scintigram. Clin. Nucl. Med. 2011, 36, 237–239. [Google Scholar] [CrossRef]
- Healey, J.H.; Turnbull, A.D.; Miedema, B.; Lane, J.M. Acrometastases. A study of twenty-nine patients with osseous involvement of the hands and feet. J. Bone Joint Surg. Am. 1986, 68, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Tewari, M.K.; Khosla, V.K.; Sharma, B.S.; Vashistha, R.K.; Khandelwal, N.K.; Kak, V.K. Brain metastasis from urachal carcinoma: Case report. Surg. Neurol. 1994, 42, 340–342. [Google Scholar] [CrossRef]
- Rodrigues, L.; Cornelis, F.H.; Reina, N.; Chevret, S. Prevention of Pathological Fracture of the Proximal Femur: A Systematic Review of Surgical and Percutaneous Image-Guided Techniques Used in Interventional Oncology. Medicina. 2019, 55, 755. [Google Scholar] [CrossRef]
- Toliušis, V.; Kalesinskas, R.; Kiudelis, M.; Maleckas, A. Surgical treatment of humeral metastatic tumors. Medicina 2009, 45, 607. [Google Scholar] [CrossRef]

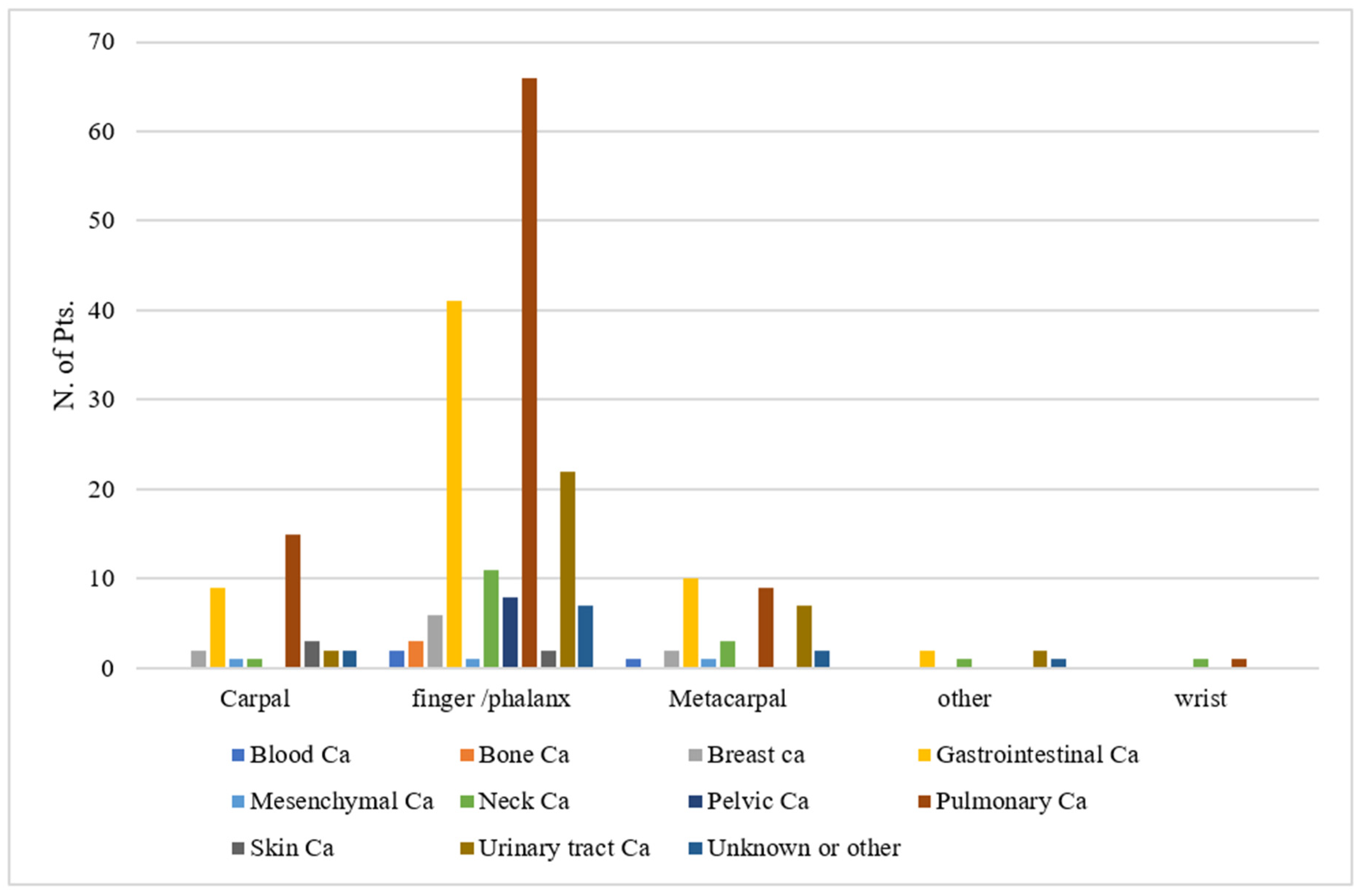

| Primary Tumor Type | No. of Patients | Average Interval from Primary Tumor Diagnosis to Acrometastasis (Months) |
|---|---|---|
| Blood Ca | 3 | 48 |
| Bone Ca | 3 | 44 |
| Breast Ca | 10 | 64 |
| Gastrointestinal Ca | 62 | 26.1 |
| Mesenchymal Ca | 3 | 2 |
| Neck Ca | 17 | 32.2 |
| Pelvic Ca | 8 | 6 |
| Pulmonary Ca | 91 | 13.4 |
| Skin Ca | 5 | 58 |
| Urinary tract Ca | 33 | 40.5 |
| Other or unknown | 12 | 27.3 |
| Total | 247 | 29.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umana, G.E.; Scalia, G.; Palmisciano, P.; Passanisi, M.; Da Ros, V.; Pompili, G.; Barone, F.; Amico, P.; Tomasi, S.O.; Graziano, F.; et al. Acrometastases to the Hand: A Systematic Review. Medicina 2021, 57, 950. https://doi.org/10.3390/medicina57090950
Umana GE, Scalia G, Palmisciano P, Passanisi M, Da Ros V, Pompili G, Barone F, Amico P, Tomasi SO, Graziano F, et al. Acrometastases to the Hand: A Systematic Review. Medicina. 2021; 57(9):950. https://doi.org/10.3390/medicina57090950
Chicago/Turabian StyleUmana, Giuseppe Emmanuele, Gianluca Scalia, Paolo Palmisciano, Maurizio Passanisi, Valerio Da Ros, Gianluca Pompili, Fabio Barone, Paolo Amico, Santino Ottavio Tomasi, Francesca Graziano, and et al. 2021. "Acrometastases to the Hand: A Systematic Review" Medicina 57, no. 9: 950. https://doi.org/10.3390/medicina57090950
APA StyleUmana, G. E., Scalia, G., Palmisciano, P., Passanisi, M., Da Ros, V., Pompili, G., Barone, F., Amico, P., Tomasi, S. O., Graziano, F., Patti, I. V., Mele, S., Maugeri, R., Raffa, G., Giammalva, G. R., Iacopino, G. D., Germanò, A., Nicoletti, G. F., Ippolito, M., ... Cuttone, G. (2021). Acrometastases to the Hand: A Systematic Review. Medicina, 57(9), 950. https://doi.org/10.3390/medicina57090950











