Effects of High Intensity Plank Exercise on Physical Fitness and Immunocyte Function in a Middle-Aged Man: A Case Report
Abstract
1. Introduction
2. Materials and Methods
2.1. Participant
2.2. Experimental Design
2.3. Measurement Methods
2.3.1. Blood Sampling and Immunocyte Measures
2.3.2. Physical Fitness Measures
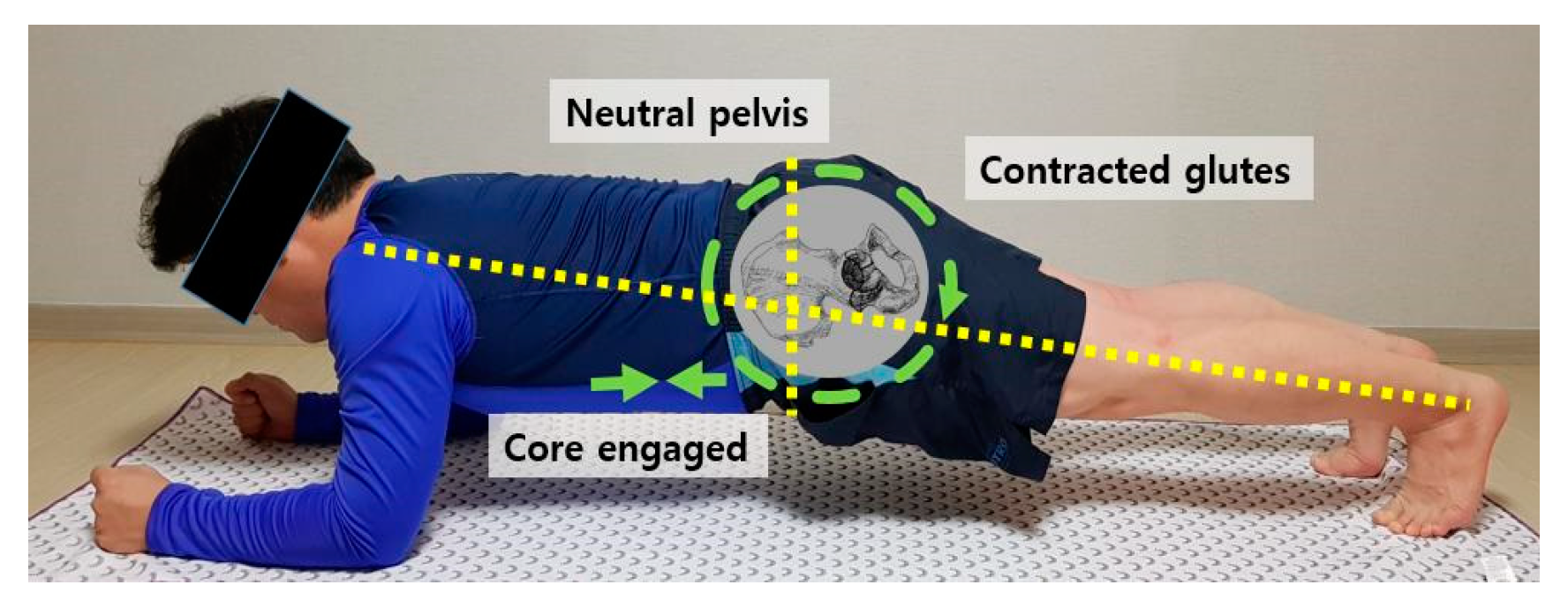
2.4. Exercise Program
2.5. Data Analyses
3. Results
3.1. Effect of Plank Exercise on Complete Blood Count
3.2. Effect of Plank Exercise on Lymphocytes and Granulocytes
3.3. Effect of Plank Exercise on Body Composition
3.4. Effect of Plank Exercise on Physical Fitness Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Korea Disease Control and Prevention Agency. 2017 Korea National Health and Nutrition Examination Survey; Korea Disease Control and Prevention Agency: Cheongju-si, Korea, 2017.
- World Health Organization. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Nieman, D.C.; Wentz, L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2018, 8, 201–217. [Google Scholar] [CrossRef]
- Stiegler, P.; Cunliffe, A. The Role of Diet and Exercise for the Maintenance of Fat-Free Mass and Resting Metabolic Rate During Weight Loss. Sports Med. 2006, 36, 239–262. [Google Scholar] [CrossRef] [PubMed]
- Jee, Y.-S. Exercise is an antigen for vaccination: First series of scientific evidence. J. Exerc. Rehabil. 2019, 15, 339–340. [Google Scholar] [CrossRef] [PubMed]
- Jee, Y.-S. How much exercise do we need to improve our immune system? Second series of scientific evidence. J. Exerc. Rehabil. 2020, 16, 113–114. [Google Scholar] [CrossRef] [PubMed]
- Jee, Y.-S. Influences of acute and/or chronic exercise on human immunity: Third series of scientific evidence. J. Exerc. Rehabil. 2020, 16, 205–206. [Google Scholar] [CrossRef] [PubMed]
- Jee, Y.-S. Physical exercise for strengthening innate immunity during COVID-19 pandemic: 4th series of scientific evidence. J. Exerc. Rehabil. 2020, 16, 383–384. [Google Scholar] [CrossRef]
- Jee, Y.-S. Acquired immunity and moderate physical exercise: 5th series of scientific evidence. J. Exerc. Rehabil. 2021, 17, 2–3. [Google Scholar] [CrossRef]
- Jee, Y.-S. Cancer and exercise immunity: 6th series of scientific evidence. J. Exerc. Rehabil. 2021, 17, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Nosaka, K.; Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 2005, 11, 64–85. [Google Scholar] [PubMed]
- Nyenhuis, S.M.; Greiwe, J.; Zeiger, J.S.; Nanda, A.; Cooke, A. Exercise and Fitness in the Age of Social Distancing During the COVID-19 Pandemic. J. Allergy Clin. Immunol. Pr. 2020, 8, 2152–2155. [Google Scholar] [CrossRef]
- Schultz, D.; Jones, S.S.; Pinder, W.M.; Wiprovnick, A.E.; Groth, E.C.; Shanty, L.M.; Duggan, A. Effective Home Visiting Training: Key Principles and Findings to Guide Training Developers and Evaluators. Matern. Child. Heal. J. 2018, 22, 1563–1567. [Google Scholar] [CrossRef] [PubMed]
- Park, D.-J.; Park, S.-Y. Which trunk exercise most effectively activates abdominal muscles? A comparative study of plank and isometric bilateral leg raise exercises. J. Back Musculoskelet. Rehabil. 2019, 32, 797–802. [Google Scholar] [CrossRef]
- Akuthota, V.; Ferreiro, A.; Moore, T.; Fredericson, M. Core Stability Exercise Principles. Curr. Sports Med. Rep. 2008, 7, 39–44. [Google Scholar] [CrossRef]
- Behm, D.G.; Drinkwater, E.J.; Willardson, J.M.; Cowley, P.M. Canadian Society for Exercise Physiology position stand: The use of instability to train the core in athletic and nonathletic conditioning. Appl. Physiol. Nutr. Metab. 2010, 35, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Kline, J.B.; Krauss, J.R.; Maher, S.F.; Qu, X. Core strength training using a combination of home exercises and a dynamic sling system for the management of low back pain in pre-professional ballet dancers: A case series. J. Dance Med. Sci. 2013, 17, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Granacher, U.; Gollhofer, A.; Hortobágyi, T.; Kressig, R.W.; Muehlbauer, T. The Importance of Trunk Muscle Strength for Balance, Functional Performance, and Fall Prevention in Seniors: A Systematic Review. Sports Med. 2013, 43, 627–641. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.-Y.; Kim, J.-H.; Hong, J.; Choi, Y.-T.; Kim, M.-H.; Cho, J.-H.; Ko, I.-G.; Jee, Y.-S. A 12-week rehabilitation program improves body composition, pain sensation, and internal/external torques of baseball pitchers with shoulder impingement symptom. J. Exerc. Rehabil. 2014, 10, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Muscle strength: Clinical and prognostic value of hand-grip dynamometry. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Jemni, M.; Thomas, E.; Patti, A.; Paoli, A.; Roque, J.R.; Palma, A.; Mammina, C.; Tabacchi, G. A systematic review to determine reliability and usefulness of the field-based test batteries for the assessment of physical fitness in adolescents—The ASSO Project. Int. J. Occup. Med. Environ. Health 2015, 28, 445–478. [Google Scholar] [CrossRef]
- Park, S.; Park, J.; Yoo, J.; Jee, Y.-S. Effect of playing soccer on stress, sociality, and physical fitness in alienated youth: A retrospective study. J. Exerc. Rehabil. 2020, 16, 154–161. [Google Scholar] [CrossRef]
- Jee, Y.-S. The efficacy and safety of whole-body electromyostimulation in applying to human body: Based from graded exercise test. J. Exerc. Rehabil. 2018, 14, 49–57. [Google Scholar] [CrossRef]
- Calatayud, J.; Casaña, J.; Martín, F.; Jakobsen, M.D.; Colado, J.C.; Gargallo, P.; Juesas, A.; Muñoz, V.; Andersen, L.L. Trunk muscle activity during different variations of the supine plank exercise. Musculoskelet. Sci. Pract. 2017, 28, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Kim, D.-E.; Cynn, H.-S. Comparison of Trunk Muscle Activity Between Traditional Plank Exercise and Plank Exercise with Isometric Contraction of Ankle Muscles in Subjects with Chronic Low Back Pain. J. Strength Cond. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Giacco, S.R.; Tocco, F.; Melis, F.; Crisafulli, A.; Gessa, M.; Santoboni, U.; Caria, M.; Tavéra, C.; Giacco, S.G.; Concu, A. Responsiveness of human natural killer cells during acute, incremental exercise up to exhaustion. Sport Sci. Health 2004, 1, 36–40. [Google Scholar] [CrossRef]
- Pedersen, B.K. Special feature for the Olympics: Effects of exercise on the immune system: Exercise and cytokines. Immunol. Cell Biol. 2000, 78, 532–535. [Google Scholar] [CrossRef] [PubMed]
- Inkabi, S.E.; Pushpamithran, G.; Richter, P.; Attakora, K. Exercise immunology: Involved components and varieties in different types of physical exercise. Scientect. J. Life Sci. 2017, 1, 31–35. Available online: http://www.scientect.com/journals/index.php/SJLS (accessed on 10 July 2021).
- Gleeson, M. Immune function in sport and exercise. J. Appl. Physiol. 2007, 103, 693–699. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Hoffman-Goetz, L. Exercise and the Immune System: Regulation, Integration, and Adaptation. Physiol. Rev. 2000, 80, 1055–1081. [Google Scholar] [CrossRef] [PubMed]
- Timmons, B.W.; Cieslak, T. Human natural killer cell subsets and acute exercise: A brief review. Exerc. Immunol. Rev. 2008, 14, 8–23. [Google Scholar] [PubMed]
- Lee, H.Y.; Baek, J.O.; Lee, J.R.; Park, S.H.; Jeon, I.S.; Roh, J.Y. Atypical Hydroa Vacciniforme-Like Epstein-Barr Virus Associated T/NK-Cell Lymphoproliferative Disorder. Am. J. Dermatopathol. 2012, 34, e119–e124. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, M. T lymphocytes. Int. J. Biochem. Cell Biol. 2003, 35, 1004–1008. [Google Scholar] [CrossRef]
- McLeod, I.X.; He, Y. Roles of autophagy in lymphocytes: Reflections and directions. Cell. Mol. Immunol. 2010, 7, 104–107. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luckheeram, R.V.; Zhou, R.; Verma, A.D.; Xia, B. CD4+T Cells: Differentiation and Functions. Clin. Dev. Immunol. 2012, 2012, 1–12. [Google Scholar] [CrossRef]
- Pedersen, B.K. Effects of exercise on lymphocytes and cytokines. Br. J. Sports Med. 2000, 34, 246–251. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Nieman, D.C. Exercise immunology: Integration and regulation. Immunol. Today 1998, 19, 204–206. [Google Scholar] [CrossRef]
- Petersen, A.M.W.; Pedersen, B.K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 2005, 98, 1154–1162. [Google Scholar] [CrossRef]
- Northoff, H.; Weinstock, C.; Berg, A. The Cytokine Response to Strenuous Exercise. Int. J. Sports Med. 1994, 15, S167–S171. [Google Scholar] [CrossRef]
- Shephard, R.J.; Shek, P.N.; DiNubile, N.A. Exercise, Immunity, and Susceptibility to Infection: A j-shaped relationship? Physician Sportsmed. 1999, 27, 47–71. [Google Scholar] [CrossRef] [PubMed]
- Natale, V.M.; Brenner, I.K.; Moldoveanu, A.I.; Vasiliou, P.; Shek, P.; Shephard, R.J. Effects of three different types of exercise on blood leukocyte count during and following exercise. Sao Paulo Med. J. 2003, 121, 9–14. [Google Scholar] [CrossRef] [PubMed]
| Days | Plank Exercise | Resting Time | Total (Minutes) | |||
|---|---|---|---|---|---|---|
| Seconds | Minutes | Seconds | Minutes | |||
| Week 1 | Day 1 | 1079.00 | 17.98 | 840.00 | 14 | 31.98 |
| Day 2 | 1049.00 | 17.48 | 840.00 | 14 | 31.48 | |
| Day 3 | 1026.00 | 17.10 | 840.00 | 14 | 31.10 | |
| Day 4 | 1028.00 | 17.13 | 840.00 | 14 | 31.13 | |
| Day 5 | 985.00 | 16.42 | 840.00 | 14 | 30.42 | |
| Mean | 17.22 | 14.00 | 31.22 | |||
| Week 2 | Day 1 | 1052.00 | 17.53 | 840.00 | 14 | 31.53 |
| Day 2 | 1034.00 | 17.23 | 780.00 | 13 | 30.23 | |
| Day 3 | 1023.00 | 17.05 | 780.00 | 13 | 30.05 | |
| Day 4 | 983.00 | 16.38 | 780.00 | 13 | 29.38 | |
| Day 5 | 951.00 | 15.85 | 840.00 | 14 | 29.85 | |
| Mean | 16.81 | 13.40 | 30.21 | |||
| Week 3 | Day 1 | 1108.00 | 18.47 | 780.00 | 13 | 31.47 |
| Day 2 | 1051.00 | 17.52 | 780.00 | 13 | 30.52 | |
| Day 3 | 1066.00 | 17.77 | 780.00 | 13 | 30.77 | |
| Day 4 | 1079.00 | 17.98 | 780.00 | 13 | 30.98 | |
| Day 5 | 999.00 | 16.65 | 780.00 | 13 | 29.65 | |
| Mean | 17.68 | 13.00 | 30.68 | |||
| Week 4 | Day 1 | 1071.00 | 17.85 | 720.00 | 12 | 29.85 |
| Day 2 | 1085.00 | 18.08 | 780.00 | 13 | 31.08 | |
| Day 3 | 1029.00 | 17.15 | 720.00 | 12 | 29.15 | |
| Day 4 | 1106.00 | 18.43 | 780.00 | 13 | 31.43 | |
| Day 5 | 1130.00 | 18.83 | 780.00 | 13 | 31.83 | |
| Mean | 18.07 | 12.6 | 30.67 | |||
| Baseline | Week 4 | Δ% | |
|---|---|---|---|
| White blood cell (×103/μL) | 6.1 | 6.7 | 9.84 |
| Red blood cell (×106/μL) | 4.8 | 5.1 | 6.25 |
| Hemoglobin (g/dL) | 15.4 | 16.2 | 5.19 |
| Hematocrit (%) | 43.4 | 45.9 | 5.76 |
| Platelets (×103/μL) | 266 | 274 | 3.01 |
| Mean corpuscular volume (fL) | 89.9 | 89.5 | −0.44 |
| Mean corpuscular hemoglobin (pg) | 31.9 | 31.6 | −0.94 |
| Mean corpuscular hemoglobin concentration (g/dL) | 35.5 | 35.3 | −0.56 |
| Erythrocyte sedimentation rate (mm/hr) | 4 | 4 | 0 |
| Red cell distribution width (%) | 11.6 | 11.4 | −1.72 |
| Platelet distribution width (%) | 13.4 | 16.6 | 23.88 |
| Baseline | Week 4 | Δ% | |
|---|---|---|---|
| Neutrophil (%) | 53.9 | 55.7 | 3.34 |
| Lymphocyte (%) | 34.8 | 34.5 | −0.86 |
| Monocyte (%) | 7.7 | 6.4 | −16.88 |
| Eosinophil (%) | 3.3 | 3 | −9.09 |
| Basophil (%) | 0.3 | 0.4 | 33.33 |
| CD3 (%) | 67.5 | 68 | 0.74 |
| CD4 (%) | 38.4 | 33.7 | −12.24 |
| CD8 (%) | 29.1 | 34.3 | 17.87 |
| CD56 (%) | 7.26 | 7.33 | 0.96 |
| CD4/CD8 (%) | 1.32 | 0.98 | −25.76 |
| Cytotoxicity (%) | 25.54 | 7.71 | −69.81 |
| Baseline | Week 4 | Δ% | |
|---|---|---|---|
| Weight (kg) | 80.4 | 81.5 | 1.37 |
| Skeletal muscle mass (kg) | 32.3 | 33.3 | 3.1 |
| Body fat mass (kg) | 23.5 | 22.8 | −2.98 |
| Body mass index (kg/m2) | 28.6 | 28.7 | 0.35 |
| Body fat percentage (%) | 29.2 | 28.6 | −2.05 |
| Basal metabolism rate (kcal) | 1599 | 1638 | 2.44 |
| Baseline | Week 4 | Δ% | |
|---|---|---|---|
| Muscle strength (kg) | 41.75 | 42.65 | 2.16 |
| Muscle endurance (reps) | 28 | 35 | 25 |
| Flexibility (cm) | −6 | 3 | 150 |
| VO2max (ml/kg/min) | 35.6 | 36.3 | 9.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.-K.; Lee, K.-S.; Heo, S.-J.; Jee, Y.-S. Effects of High Intensity Plank Exercise on Physical Fitness and Immunocyte Function in a Middle-Aged Man: A Case Report. Medicina 2021, 57, 845. https://doi.org/10.3390/medicina57080845
Park S-K, Lee K-S, Heo S-J, Jee Y-S. Effects of High Intensity Plank Exercise on Physical Fitness and Immunocyte Function in a Middle-Aged Man: A Case Report. Medicina. 2021; 57(8):845. https://doi.org/10.3390/medicina57080845
Chicago/Turabian StylePark, Sang-Kyun, Ki-Soo Lee, Seung-Jae Heo, and Yong-Seok Jee. 2021. "Effects of High Intensity Plank Exercise on Physical Fitness and Immunocyte Function in a Middle-Aged Man: A Case Report" Medicina 57, no. 8: 845. https://doi.org/10.3390/medicina57080845
APA StylePark, S.-K., Lee, K.-S., Heo, S.-J., & Jee, Y.-S. (2021). Effects of High Intensity Plank Exercise on Physical Fitness and Immunocyte Function in a Middle-Aged Man: A Case Report. Medicina, 57(8), 845. https://doi.org/10.3390/medicina57080845







