Impact of Contraception on Uterine Fibroids
Abstract
:1. Introduction
2. Materials and Methods
3. Results
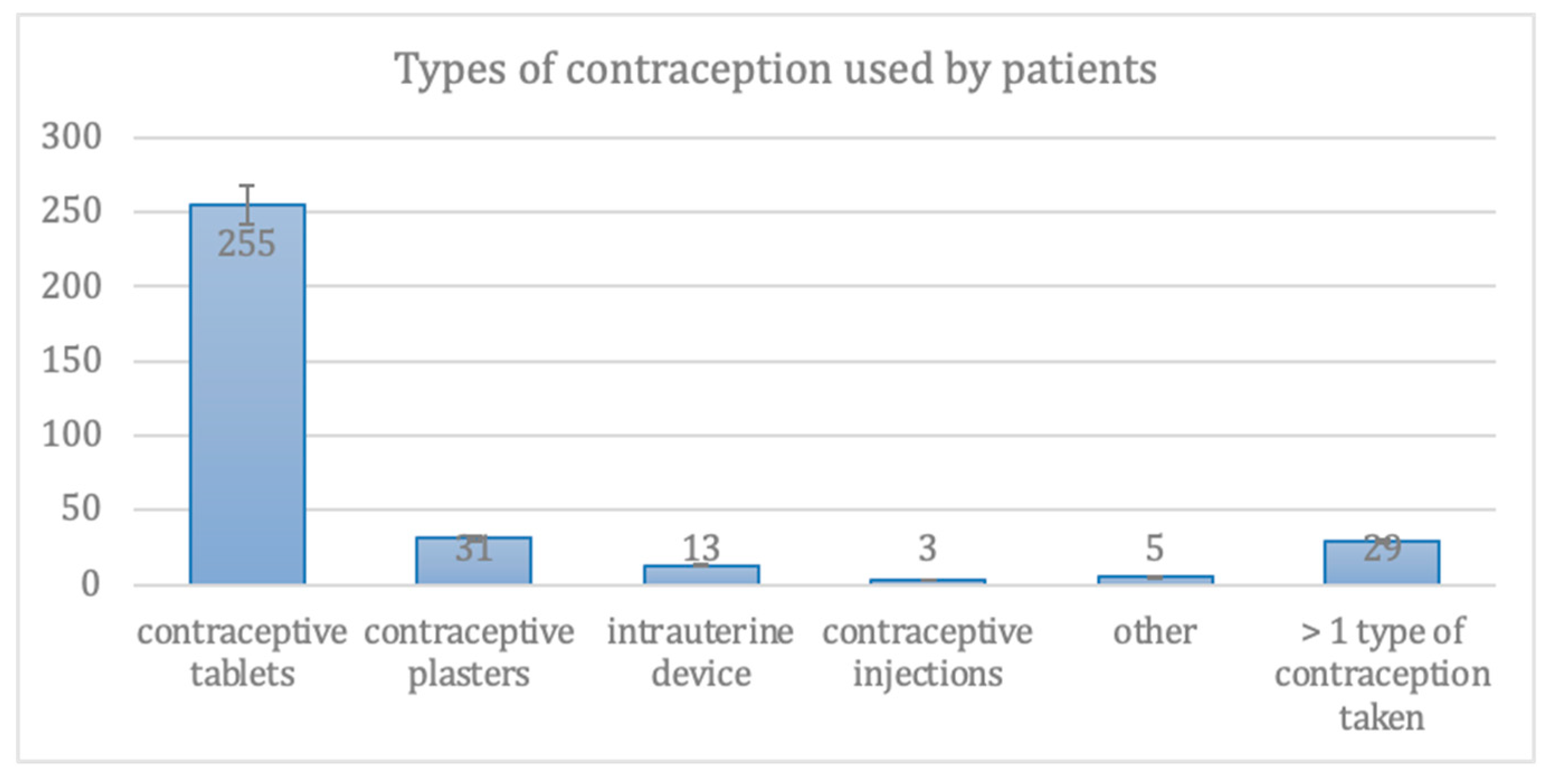
3.1. Contraception
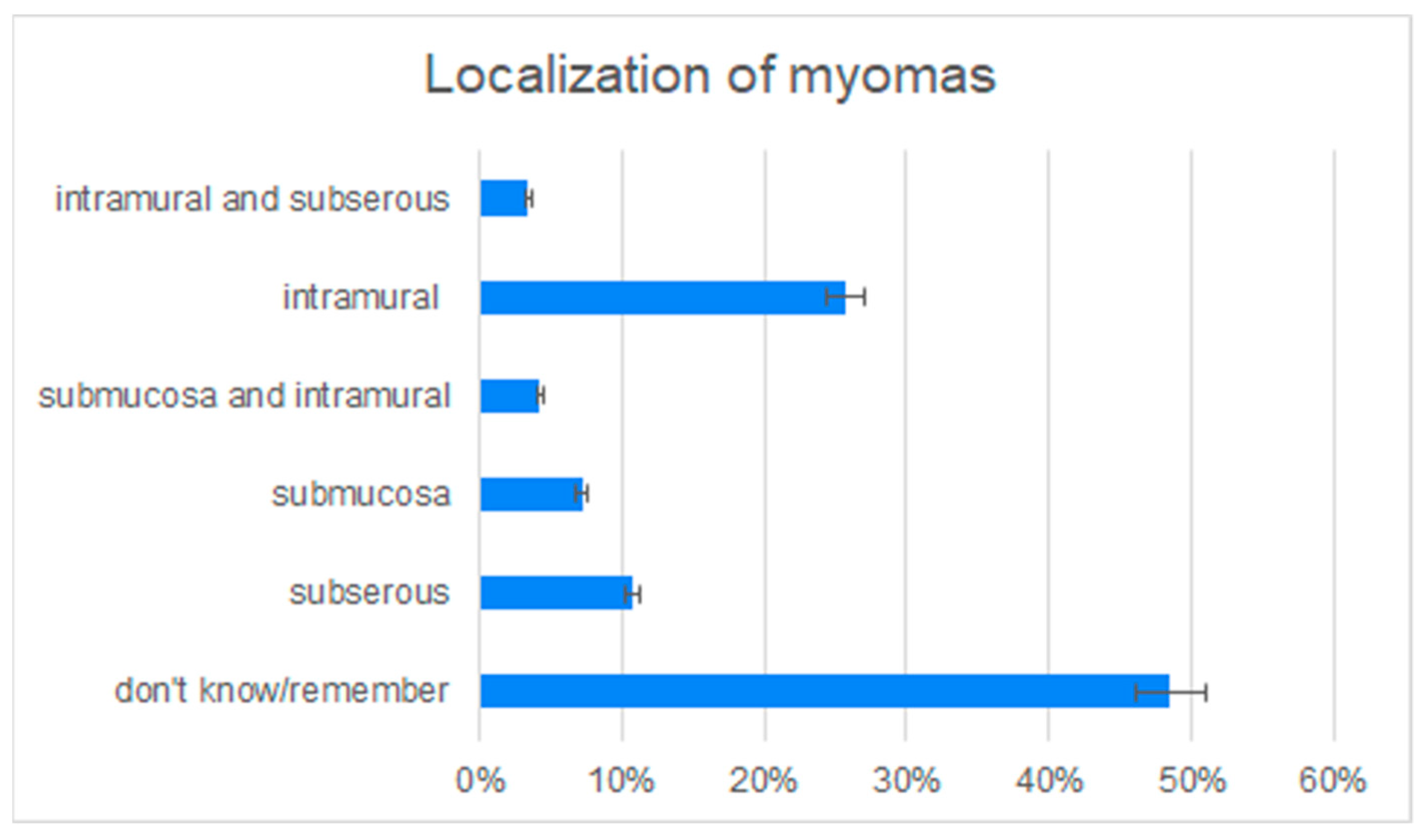
3.2. Medical-History
4. Discussion
5. Conclusions
Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De La Cruz, M.S.; Buchanan, E.M. Uterine Fibroids: Diagnosis and Treatment. Am. Fam. Physician 2017, 95, 100–107. [Google Scholar]
- Woźniak, S.; Pietrzak, B.; Paszkowski, T.; Radowicki, S.; Pawelczyk, S.; Wielgoś, M. Farmakoterapia mięśniaków macicy. Ginekol. Perinatol. Prakt. 2017, 2, 43–47. [Google Scholar]
- Giuliani, E.; As-Sanie, S.; Marsh, E.E. Epidemiology and management of uterine fibroids. Int. J. Gynecol. Obstet. 2020, 149, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.J.; Deshmukh, P.; Antell, K. Contraception Update: Oral Contraception. FP Essent. 2017, 462, 11–19. [Google Scholar]
- Chiaffarino, F.; Parazzini, F.; Vecchia, C.; Marsico, S.; Surace, M.; Ricci, E. Use of oral contraceptives and uterine fibroids: Results from a case-control study. BJOG Int. J. Obstet. Gynaecol. 1999, 106, 857–860. [Google Scholar] [CrossRef]
- Sangkomkamhang, U.S.; Lumbiganon, P.; Pattanittum, P. Progestogens or progestogen-releasing intrauterine systems for uterine fibroids (other than preoperative medical therapy). Cochrane Database Syst. Rev. 2020, 11, CD008994. [Google Scholar] [CrossRef]
- Bulun, S.E. Uterine Fibroids. N. Engl. J. Med. 2013, 369, 1344–1355. [Google Scholar] [CrossRef] [Green Version]
- Safrai, M.; Chill, H.H.; Salzman, A.R.; Shushan, A. Selective Progesterone Receptor Modulators for the Treatment of Uterine Leiomyomas. Obstet. Gynecol. 2017, 130, 315–318. [Google Scholar] [CrossRef]
- Donnez, J.; Dolmans, M.-M. Uterine fibroid management: From the present to the future. Hum. Reprod. Updat. 2016, 22, 665–686. [Google Scholar] [CrossRef]
- Reis, F.M.; Bloise, E.; Ortiga-Carvalho, T.M. Hormones and pathogenesis of uterine fibroids. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 34, 13–24. [Google Scholar] [CrossRef]
- Sayed, G.H.; Zakherah, M.S.; El-Nashar, S.A.; Shaaban, M.M. A randomized clinical trial of a levonorgestrel-releasing intrauterine system and a low-dose combined oral contraceptive for fibroid-related menorrhagia. Int. J. Gynecol. Obstet. 2010, 112, 126–130. [Google Scholar] [CrossRef]
- Lethaby, A.; Wise, M.R.; Weterings, M.A.; Rodriguez, M.B.; Brown, J. Combined hormonal contraceptives for heavy menstrual bleeding. Cochrane Database Syst. Rev. 2019, 2019, CD000154. [Google Scholar] [CrossRef]
- Asbell, B. The Pill: A Biography of the Drug That Changed the World; Random House: New York, NY, USA, 1995. [Google Scholar]
- Schindler, A.E. Non-Contraceptive Benefits of Oral Hormonal Contraceptives. Int. J. Endocrinol. Metab. 2012, 11, 41–47. [Google Scholar] [CrossRef] [Green Version]
- Jensen, J.T. The benefits of reduced menstrual bleeding associated with the use of contraceptive methods. Gynecol. Forum 2010, 15, 10–14. [Google Scholar]
- Fraser, I.S.; Porte, R.J.; Kouides, P.A.; Lukes, A.S. A Benefit-Risk Review of Systemic Haemostatic Agents: Part 1: In major surgery. Drug Saf. 2008, 31, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.A.; Cookson, C.L.; Gandolfo, R.A.; Schulze-Rath, R. Epidemiology of uterine fibroids: A systematic review. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 1501–1512. [Google Scholar] [CrossRef] [PubMed]
- Lethaby, A.; Vollenhoven, B. Fibroids (uterine myomatosis, leiomyomas). BMJ Clin. Evid. 2015, 2015, 0814. [Google Scholar]
- Wise, L.A.; Laughlin-Tommaso, S.K. Epidemiology of Uterine Fibroids: From Menarche to Menopause. Clin. Obstet. Gynecol. 2016, 59, 2–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McWilliams, M.M.; Chennathukuzhi, V.M. Recent Advances in Uterine Fibroid Etiology. Semin. Reprod. Med. 2017, 35, 181–189. [Google Scholar] [CrossRef] [Green Version]
- Van Voorhis, B.J.; Romitti, P.A.; Jones, M.P. Family history as a risk factor for development of uterine leiomyomas. Results of a pilot study. J. Reprod. Med. 2002, 47, 663–691. [Google Scholar]
- Kınay, T.; Başarır, Z.Ö.; Tuncer, S.F.; Akpınar, F.; Kayıkçıoğlu, F.; Koç, S. Prevalence of endometrial polyps coexisting with uterine fibroids and associated factors. J. Turk. Soc. Obstet. Gynecol. 2016, 13, 31–36. [Google Scholar] [CrossRef]
- Wong, J.Y.Y.; Gold, E.B.; Johnson, W.O.; Lee, J.S. Circulating Sex Hormones and Risk of Uterine Fibroids: Study of Women’s Health Across the Nation (SWAN). J. Clin. Endocrinol. Metab. 2016, 101, 123–130. [Google Scholar] [CrossRef] [Green Version]
- Gallicchio, L.; Schilling, C.; Romani, W.A.; Miller, S.; Zacur, H.; Flaws, J.A. Endogenous hormones, participant characteristics, and symptoms among midlife women. Maturitas 2008, 59, 114–127. [Google Scholar] [CrossRef] [Green Version]
- Uimari, O.; Järvelä, I.; Ryynänen, M. Do symptomatic endometriosis and uterine fibroids appear together? J. Hum. Reprod. Sci. 2011, 4, 34–38. [Google Scholar] [CrossRef]
- Ross, R.K.; Pike, M.C.; Vessey, M.P.; Bull, D.; Yeates, D.; Casagrande, J.T. Risk factors for uterine fibroids: Reduced risk associated with oral contraceptives. BMJ 1986, 293, 359–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramcharan, S.; Pellegrin, F.A.; Ray, R.M.; Hsu, J.P. The Walnut Creek Contraceptive Drug Study. A prospective study of the side effects of oral contraceptives. Volume III, an interim report: A comparison of disease occurrence leading to hospitalization or death in users and nonusers of oral contraceptives. J. Reprod. Med. 1980, 25, 345–372. [Google Scholar] [PubMed]
- Eskenazi, B.; Warner, M.; Samuels, S.; Young, J.; Gerthoux, P.M.; Needham, L.; Patterson, D.; Olive, D.; Gavoni, N.; Vercellini, P.; et al. Serum Dioxin Concentrations and Risk of Uterine Leiomyoma in the Seveso Women’s Health Study. Am. J. Epidemiol. 2007, 166, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A.; Palmer, J.R.; Harlow, B.L.; Spiegelman, D.; Stewart, E.A.; Adams-Campbell, L.L.; Rosenberg, L. Reproductive Factors, Hormonal Contraception, and Risk of Uterine Leiomyomata in African-American Women: A Prospective Study. Am. J. Epidemiol. 2004, 159, 113–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parazzini, F.; Negri, E.; La Vecchia, C.; Chatenoud, L.; Ricci, E.; Guarnerio, P. Reproductive Factors and Risk of Uterine Fibroids. Epidemiology 1996, 7, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Ying, J.; Nie, D.; Chen, S. The relationship between the use of oral contraceptives and the risk of uterine leiomyoma in women of child bearing age of Han Chines. Pharm. Bioprocess. 2018, 6, 48–52. [Google Scholar]
- Piecak, K.; Milart, P.; Woźniakowska, E.; Paszkowski, T. Ulipristal acetate as a treatment option for uterine fibroids. Menopause Rev. 2017, 16, 133–136. [Google Scholar] [CrossRef] [Green Version]
- Marci, R.; Maiorana, A.; Carinelli, S.; Biglia, N.; Monte, G.L.; D’Alonzo, M. Ulipristal acetate: A novel pharmacological approach for the treatment of uterine fibroids. Drug Des. Dev. Ther. 2014, 8, 285–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Q.; Ohara, N.; Chen, W.; Liu, J.; Sasaki, H.; Morikawa, A.; Sitruk-Ware, R.; Johansson, E.D.; Maruo, T. Progesterone receptor modulator CDB-2914 down-regulates vascular endothelial growth factor, adrenomedullin and their receptors and modulates progesterone receptor content in cultured human uterine leiomyoma cells. Hum. Reprod. 2006, 21, 2408–2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horak, P.; Mára, M.; Dundr, P.; Kubinova, K.; Kužel, D.; Hudecek, R.; Chmel, R. Effect of a Selective Progesterone Receptor Modulator on Induction of Apoptosis in Uterine Fibroids In Vivo. Int. J. Endocrinol. 2012, 2012, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Del Forno, S.; Degli Esposti, E.; Salucci, P.; Leonardi, D.; Iodice, R.; Arena, A.; Raimondo, D.; Paradisi, R.; Seracchioli, R. Liver function, tolerability and satisfaction during treatment with ulipristal acetate in women with fibroids: A single center experience. Gynecol. Endocrinol. 2019, 36, 445–447. [Google Scholar] [CrossRef]
| Characteristics | Patients with Myomas (Study Group) n = 140 Mean ± SD | Patients without Myomas (Control Group) n = 206 Mean ± SD | p Value |
|---|---|---|---|
| Mean age (years) | 36.61 ± 8.60 | 24.77 ± 6.17 | p < 0.0001 |
| Weight (kg) | 67.61 ± 12.91 | 62.35 ± 12.48 | p = 0.000004 |
| Mean age of menarche (years) | 12.99 SD = 2.08 | 12.74 SD = 1.59 | p = 0.2298 |
| Number of pregnancies (median) | 0 | 1 | p < 0.000001 |
| Mean age of first pregnancy (years) | 25.26 SD = 5.37 | 24.14 SD = 4.24 | p = 0.0897 |
| Median number of life births | 1 | 1 | p = 0.725 |
| % of patients with natural labor (73 patients–100%) | 63.01% | 26.99% | |
| % of patients with cesarean labor (47 patients–100%) | 63.83% | 26.17% |
| Patients with Myomas (Study Group) n = 94 | Patients without Myomas (Control Group) n = 180 | p Value | |
|---|---|---|---|
| Mean age [years] | 23.71 SD = 7.00 | 19.63 SD = 4.04 | p = 0.000000004 |
| Mean duration of contraceptive use [months] | 73.29 SD = 72.6 | 39.04 SD = 46.90 | p = 0.00007 |
| Age [years] | Myoma Presence | No Contraception Intake | Contraception Intake | Statistical Significance |
|---|---|---|---|---|
| 20–30 | Yes | 9 | 15 | p = 0.1159 |
| no | 21 | 77 | ||
| 30–40 | yes | 23 | 29 | p = 0.00299 |
| no | 3 | 25 | ||
| 40–50 | yes | 14 | 26 | p = 0.5422 |
| no | 0 | 3 |
| Relationship between Uterine Fibroids | |||
|---|---|---|---|
| uterine fibroids present “+” lack of uterine fibroids “–” | statistical test used in analysis | p Value | |
| Contraception | – | Chi2 Test | p = 0.00001 |
| Morning-after pill taken | + | Chi2 Test | p = 0.67559 |
| Positive family history | + | Chi2 Test | p = 0.00004 |
| Gynecological surgeries | + | Chi2 Test | p < 0.00001 |
| Gyn. coexisting diseases | + | Chi2 Test | p = 0.00275 |
| Hypertension occurrence | + | Fisher’s exact two-sided test | p = 0.05593 |
| Pregnancy number | – | U Mann–Whitney Test | p < 0.00001 |
| Natural labor | – | Chi2 Test | p = 0.9278 |
| Assisted labor | + | Chi2 Test | p = 0.1024 |
| Age of first pregnancy | + | U Mann–Whitney Test | p = 0.0897 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwas, K.; Nowakowska, A.; Fornalczyk, A.; Krzycka, M.; Nowak, A.; Wilczyński, J.; Szubert, M. Impact of Contraception on Uterine Fibroids. Medicina 2021, 57, 717. https://doi.org/10.3390/medicina57070717
Kwas K, Nowakowska A, Fornalczyk A, Krzycka M, Nowak A, Wilczyński J, Szubert M. Impact of Contraception on Uterine Fibroids. Medicina. 2021; 57(7):717. https://doi.org/10.3390/medicina57070717
Chicago/Turabian StyleKwas, Katarzyna, Aleksandra Nowakowska, Angelika Fornalczyk, Magda Krzycka, Anna Nowak, Jacek Wilczyński, and Maria Szubert. 2021. "Impact of Contraception on Uterine Fibroids" Medicina 57, no. 7: 717. https://doi.org/10.3390/medicina57070717
APA StyleKwas, K., Nowakowska, A., Fornalczyk, A., Krzycka, M., Nowak, A., Wilczyński, J., & Szubert, M. (2021). Impact of Contraception on Uterine Fibroids. Medicina, 57(7), 717. https://doi.org/10.3390/medicina57070717






