Pathogenesis of Retraction Pocket of the Tympanic Membrane—A Narrative Review
Abstract
1. Introduction
2. The Eustachian Tube and Pathogenesis of the Retraction Pocket
3. The Relationship of Otitis Media and Pathogenesis of the Retraction Pocket
4. The Role of Inflammation in Pathogenesis of the Retraction Pocket
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Beer, B.A.; Schilder, A.G.M.; Zielhuis, G.A.; Graamans, K. Natural course of tympanic membrane pathology related to otitis media and ventilation tubes between ages 8 and 18 years. Otol. Neurotol. 2005, 26, 1016–1021. [Google Scholar] [CrossRef]
- Bluestone, C.; Klein, J. Pediatric Otolaryngology, 4th ed.; WB Saunders: Philadelphia, PA, USA, 2003. [Google Scholar]
- Bunne, M.; Falk, B.; Magnuson, B.; Hellstrom, M. Variability of Eustachian tubefunction: Comparison of ears with retraction dis-ease and normal ears. Laryngoscope 2000, 110, 1389–1395. [Google Scholar] [CrossRef]
- Sadé, J. Treatment of Cholesteatoma and Retraction Pockets. Eur. Arch. Oto-Rhino-Laryngol. 1993, 250, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Tos, M.; Poulsen, G. Attic Retractions Following Secretory Otitis. Acta Oto-Laryngol. 1980, 89, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Charachon, R. Classification of Retraction Pockets. Rev. Laryngol. Otol. Rhinol. 1988, 109, 205–207. [Google Scholar]
- Olszewska, E.; Rutkowska, J.; Özgirgin, N. Consensus-Based Recommendations on the Definition and Classification of Cho-lesteatoma. J. Int. Adv. Otol. 2015, 11, 81–87. [Google Scholar] [CrossRef]
- Makino, K.; Amatsu, M. Epithelial migration on the tympanic membrane and external canal. Eur. Arch. Oto-Rhino-Laryngol. 1986, 243, 39–42. [Google Scholar] [CrossRef]
- Lim, D.J. Tympanic Membrane: Electron Microscopic Observation Part I: Pars Tensa. Acta Oto-Laryngol. 1968, 66, 181–198. [Google Scholar] [CrossRef]
- Sadé, J. Atelectatic Tympanic Membrane: Histologic Study. Ann. Otol. Rhinol. Laryngol. 1993, 102, 712–716. [Google Scholar] [CrossRef]
- Tos, M. Anatomy and histology of the middle ear. Clin. Rev. Allergy 1984, 2, 267–284. [Google Scholar]
- Jesic, S.D.; Rovcanin, L.H.; Jovicevic, O.D.; Jotic, A.D.; Slijepcevic, N.A.; Ljubic, V.R. Pars tensa retractions without cholesteatoma in children: Predictors for ossicular chain destruction, air conduction thresholds, and postoperative retractions. Otol. Neurotol. 2014, 35, 997–1002. [Google Scholar] [CrossRef]
- Swarts, J.D.; Alper, C.M.; Luntz, M. Panel 2: Eustachian tube, middle ear, and mastoid—Anatomy, physiology, pathophysi-ology, and pathogenesis. Otolaryngol. Head Neck Surg. 2013, 148 (Suppl. 4), E26–E36. [Google Scholar] [CrossRef]
- Marchioni, D.; Piccinini, A.; Alicandri-Ciufelli, M.; Presutti, L. Endoscopic Anatomy and Ventilation of the Epitympanum. Otolaryngol. Clin. N. Am. 2013, 46, 165–178. [Google Scholar] [CrossRef]
- Bezold, F. Perforation of Shrapnell’s membrane and occlusion of the tubes: An aetiological study. Arch Otolaryngol. 1890, 19, 232–254. [Google Scholar]
- Holmgren, L. Experimental tubal occlusion. Acta Otolaryngol. (Stock.) 1940, 28, 587–592. [Google Scholar] [CrossRef]
- Sade, J.; Carr, C.D.; Senturia, B.H. LXXI Middle Ear Effusions Produced Experimentally in Dogs. Ann. Otol. Rhinol. Laryngol. 1959, 68, 1017–1027. [Google Scholar] [CrossRef] [PubMed]
- Spilsbury, K.; Ha, J.F.; Semmens, J.B.; Lannigan, F. Cholesteatoma in cleft lip and palate: A population-based follow-up study of children after ventilation tubes. Laryngoscope 2013, 123, 2024–2029. [Google Scholar] [CrossRef]
- Tos, M.; Stangerup, S.-E.; Holm-Jensen, S.; Sørensen, C.H. Spontaneous Course of Secretory Otitis and Changes of the Eardrum. Arch. Otolaryngol. Head Neck Surg. 1984, 110, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Bluestone, C. Eustachian Tube: Structure, Function, Role in Otitis Media, 1st ed.; BCDecker: Hamilton, ON, Canada; London, UK, 2005. [Google Scholar]
- Kuijpers, W.; Van Der Beek, J.M.; Jap, P.H. Experimental model for the study of otitis media with effusion. Acta Oto-Rhino-Laryngol. Belg. 1983, 37, 135–137. [Google Scholar]
- Gerber, M.J.; Mason, J.C.; Lambert, P.R. Hearing Results after Primary Cartilage Tympanoplasty. Laryngoscope 2000, 110, 1994–1999. [Google Scholar] [CrossRef] [PubMed]
- Tos, M. Incidence, Etiology and Pathogenesis of Cholesteatoma in Children. Adv. Oto-Rhino-Laryngol. 1988, 40, 110–117. [Google Scholar] [CrossRef]
- Bhide, A. Etiology of the Retraction Pocket in the Posterosuperior Quadrant of the Eardrum. Arch. Otolaryngol. 1977, 103, 707–711. [Google Scholar] [CrossRef]
- Meyerhoff, W.L.; Wright, C.G.; Gerken, G.M. Effects of middle ear ventilation on cholesteatoma development in experimental animals. Acta Oto-Laryngol. 1990, 110, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Sudhoff, H.; Tos, M. Pathogenesis of attic cholesteatoma: Clinical and immunohistochemical support for combination of retrac-tion theory and proliferation theory. Am. J. Otol. 2000, 21, 786–792. [Google Scholar] [PubMed]
- Işlek, A.; Songu, M.; Balcı, M.K.; Aladağ, I. The Role of Serum Adhesion Molecules s-ICAM-1 and s-VCAM-1 in Chronic Otitis Media Pathogenesis: A Prospective Controlled Study. Indian J. Otolaryngol. Head Neck Surg. 2020, 1–6. [Google Scholar] [CrossRef]
- Hüttenbrink, K.B. A New Theory Interprets the Development of a Retraction Pocket as a Natural Self-Healing Process. Eur. Arch. Oto-Rhino-Laryngol. 2019, 276, 367–373. [Google Scholar] [CrossRef]
- Kaufmann, E.; Staemmler, M. Lehrbuch der Speziellen Pathologischen Anatomie. Band 1; 1. Hälfte; De Gruyter: Berlin, Germany, 1956. [Google Scholar]
- Olszewska, E.; Jakimowicz-Rudy, J.; Knas, M.; Chilimoniuk, M.; Pietruski, J.K.; Sieskiewicz, A. Cholesteatoma-associated patho-genicity: Potential role of lysosomal exoglycosidases. Otol. Neurotol. 2012, 33, 596–603. [Google Scholar] [CrossRef]
- Urík, M.; Hurník, P.; Žiak, D.; Machac, J.; Šlapák, I.; Motyka, O.; Vaculová, J.; Dvořáčková, J. Histological analysis of retraction pocket pars tensa of tympanic membrane in children. Int. J. Pediatr. Otorhinolaryngol. 2016, 86, 213–217. [Google Scholar] [CrossRef]
- Yoon, T.H.; A Schachern, P.; Paparella, M.M.; Aeppli, D.M. Pathology and pathogenesis of tympanic membrane retraction. Am. J. Otolaryngol. 1990, 11, 10–17. [Google Scholar] [CrossRef]
- Olszewska, E.; Sudhoff, H. Comparative cytokeratin distribution patterns in cholesteatoma epithelium. Histol. Histopathol. 2007, 22, 37–42. [Google Scholar]
- Urík, M.; Hurník, P.; Žiak, D.; Machač, J.; Šlapák, I.; Motyka, O.; Jabandžiev, P. Immunohistochemical analysis of retraction pocket pars tensa of tympanic membrane in children. Int. J. Pediatr. Otorhinolaryngol. 2019, 122, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Alper, C.M.; Olszewska, E. Assessment and management of retraction pockets. Pol. J. Otolaryngol. 2017, 71, 1–21. [Google Scholar]
- Shunyu, N.B.; Gupta, S.D.; Thakar, A.; Sharma, S.C. Histological and Immunohistochemical Study of Pars Tensa Retraction Pocket. Otolaryngol. Head Neck Surg. 2011, 145, 628–634. [Google Scholar] [CrossRef]
- Tos, M. Upon the relationship between secretory otitis in childhood and chronic otitis and its sequelae in adults. J. Laryngol. Otol. 1981, 95, 1011–1022. [Google Scholar] [CrossRef] [PubMed]
- Tretiakow, D.; Kuczkowsji, J.; Tukaj, C. Ultrastructural alterations of the tympanic membrane in retraction pockets from patients with chronic otitis media. Pol. J. Pathol. 2020, 71, 347–352. [Google Scholar] [CrossRef]
 Malleus,
Malleus,  Erosion of the incudostapedial joint.
Erosion of the incudostapedial joint.
 Malleus,
Malleus, 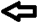 Erosion of the incudostapedial joint.
Erosion of the incudostapedial joint.
 External epithelial layer,
External epithelial layer,  Basal membrane,
Basal membrane,  Fragmented elastic fibers in the middle layer.
Fragmented elastic fibers in the middle layer.
 External epithelial layer,
External epithelial layer,  Basal membrane,
Basal membrane,  Fragmented elastic fibers in the middle layer.
Fragmented elastic fibers in the middle layer.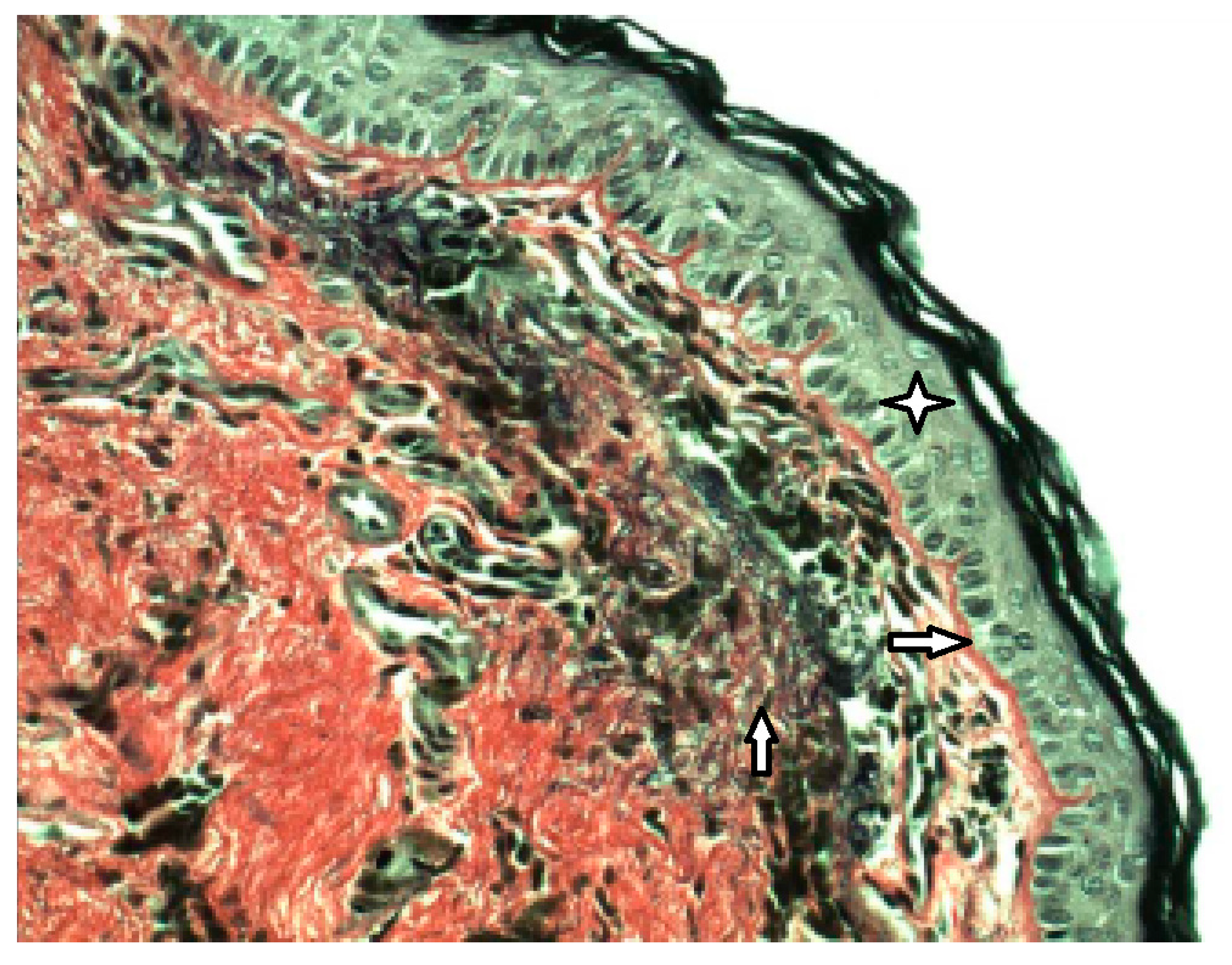
 External epithelial layer,
External epithelial layer,  Basal membrane,
Basal membrane,  Rete peg (area of ingrowing squamous epithelium),
Rete peg (area of ingrowing squamous epithelium),  White blood cells of all categories (CD45 LCA+).
White blood cells of all categories (CD45 LCA+).
 External epithelial layer,
External epithelial layer,  Basal membrane,
Basal membrane,  Rete peg (area of ingrowing squamous epithelium),
Rete peg (area of ingrowing squamous epithelium),  White blood cells of all categories (CD45 LCA+).
White blood cells of all categories (CD45 LCA+).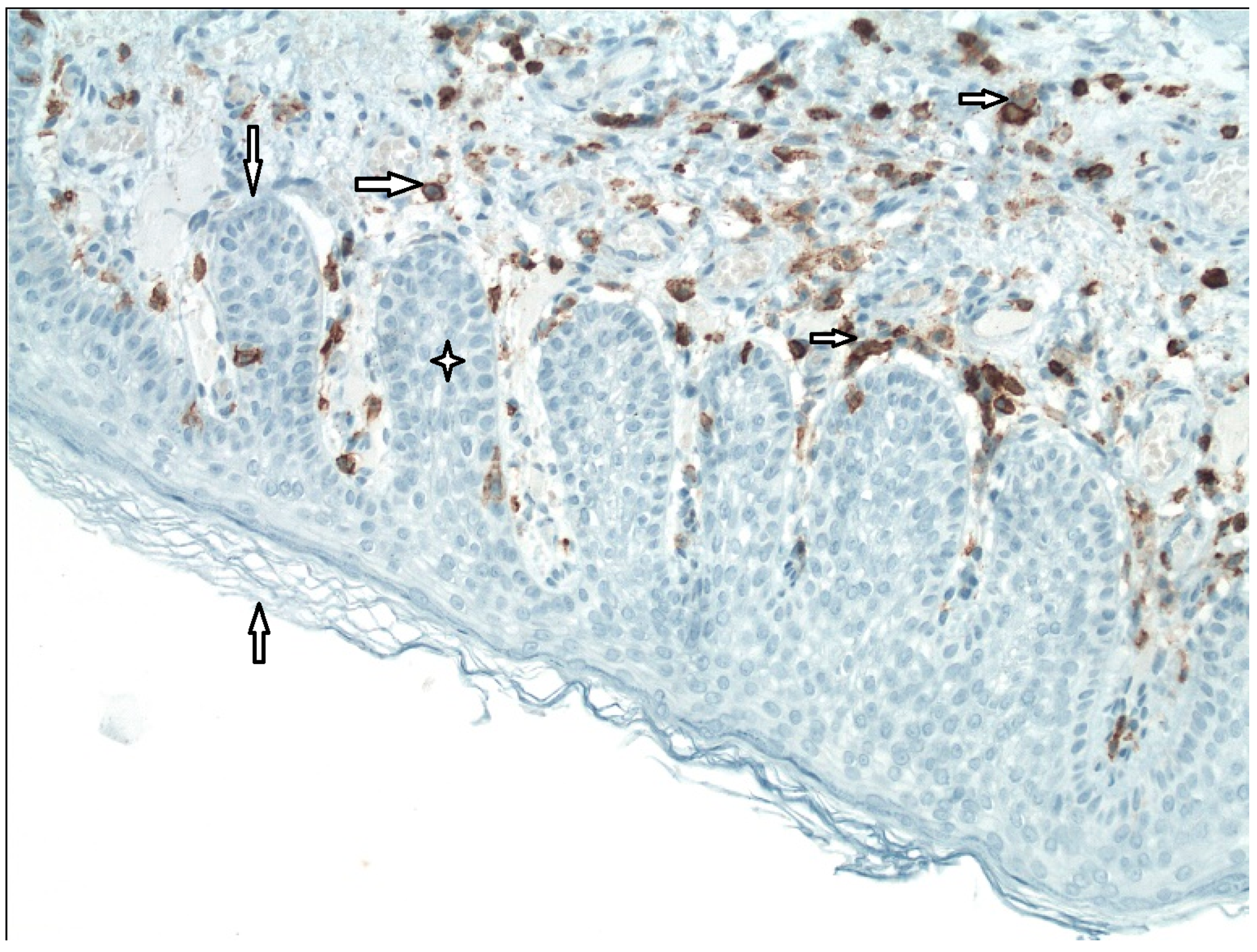
 External epithelial layer,
External epithelial layer,  Middle layer of the tympanic membrane,
Middle layer of the tympanic membrane,  MMP9+.
MMP9+.
 External epithelial layer,
External epithelial layer,  Middle layer of the tympanic membrane,
Middle layer of the tympanic membrane,  MMP9+.
MMP9+.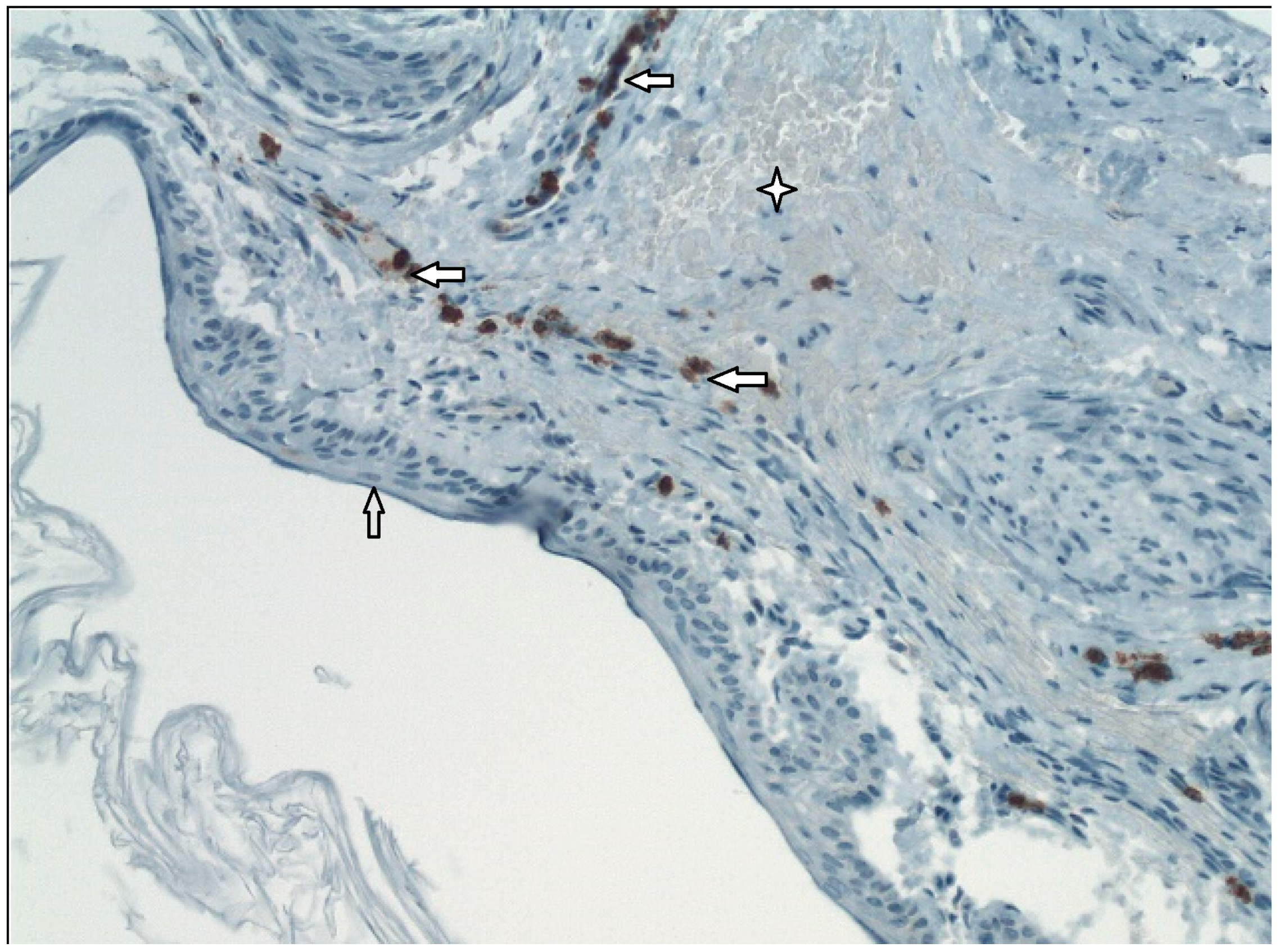
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urík, M.; Tedla, M.; Hurník, P. Pathogenesis of Retraction Pocket of the Tympanic Membrane—A Narrative Review. Medicina 2021, 57, 425. https://doi.org/10.3390/medicina57050425
Urík M, Tedla M, Hurník P. Pathogenesis of Retraction Pocket of the Tympanic Membrane—A Narrative Review. Medicina. 2021; 57(5):425. https://doi.org/10.3390/medicina57050425
Chicago/Turabian StyleUrík, Milan, Miroslav Tedla, and Pavel Hurník. 2021. "Pathogenesis of Retraction Pocket of the Tympanic Membrane—A Narrative Review" Medicina 57, no. 5: 425. https://doi.org/10.3390/medicina57050425
APA StyleUrík, M., Tedla, M., & Hurník, P. (2021). Pathogenesis of Retraction Pocket of the Tympanic Membrane—A Narrative Review. Medicina, 57(5), 425. https://doi.org/10.3390/medicina57050425





