Mind-Body Interventions as Alternative and Complementary Therapies for Psoriasis: A Systematic Review of the English Literature
Abstract
1. Introduction
2. Methods
3. Results
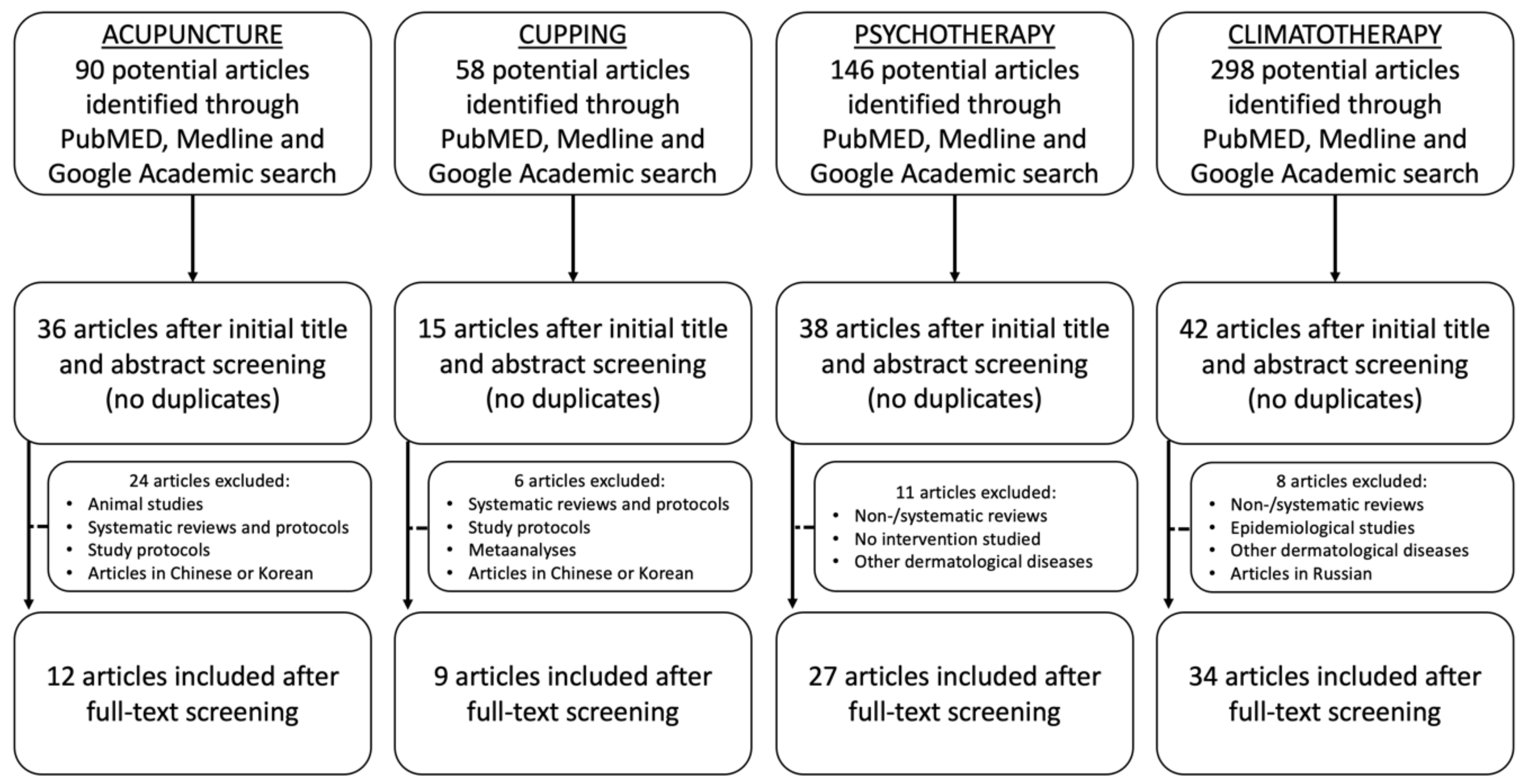
3.1. Data Acquisition
3.2. Patient Populations
| Author, Year | Procedure | Study Design | Study/Treatment Length | Patients Included | Gender | Mean Age (y) | Average Duration of Psoriasis | Comparison Therapy | Dropout No. (%) | Severity Measurement | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liao and Liao, 1992 [19] | Acupuncture | Retrospective report | 9.1 sessions (average) | 61 Therapy | 25M/36F | 51.84 | 16.26 y | N/A | N/A | Arbitrary, visual | On average, moderate improvement was achieved in psoriasis patients. However, no standard evaluation score was provided for accurate comparison. |
| Ye et al., 2004 [20] | Acupuncture plus Chinese herbs | Retrospective report | 8 y study (30–50 days courses) | 80 Therapy | 44M/36F | 9–65 | NS | N/A | N/A | Therapy effectiveness of 91.3% (41 cured, 18 markedly improved, 14 improved). | |
| Jerner et al., 1997 [28] | Electrostimulation via intramuscular acupuncture and ear acupuncture | Prospective RCT | 10 w | 54: 35 Therapy 19 Placebo | 31M/23F | 44 // 48 | 20 y // 20 y | sham (minimal) acupuncture | 2 (3.5%) | PASI | PASI lowered from 9.6 to 8.3 in the therapy group and from 9.2 to 6.9 in the placebo group, w/o statistically significant differences between the two. |
| Pan et al., 2018 [29] | Fire-needle therapy plus HXJDD and Vaseline cream | Prospective RCT | 4 w | 136: 68 Therapy 68 Control | 89M/47F | 44.92 // 45.19 | 13.72 y // 13.41 y | HXJDD and Vaseline cream alone | 15 (10%) | PASI, DLQI, HAMA, CM syndrome score | Therapy group did not attain significant improvement in PASI score compared to control group. However, significant differences were found between the two groups in alleviating CM syndrome and DLQI. |
| Lu et al., 2012 [30] | Auricular therapy with optimized Yinxieling Formula | Prospective single-blind RCT | 8 w | 84: 43 Therapy 41 Control | 61M/23F | 38.98 // 38.58 | 118.63 mo (9.9 y) // 136.79 mo (11.4 y) | optimized Yinxieling Formula alone | N/A | PASI, DLQI, VAS, SDS, SAS | PASI lowered in both, yet significantly more in the therapy group. DLQI decreased in both groups, though not statistically significant and w/o statistical significance in difference between the two. SAS, SDS, VAS showed no statistical difference |
| Jorge et al., 2016 [31] | Ear acupuncture | Prospective observational report | 14 w or symptom resolution | 7 | 2M/5F | 33 | 11.7 y | N/A | 3 (30%) | PASI | 5 of 7 patients presented complete disappearance of symptoms, two had significant recovery. |
| Jeon YC, 2016 [32] | Traditional Korean Medicine, especially Sa-Am acupuncture plus Chinese herbs | Case report | 14 mo therapy | 1 | F | 25 | 3 y | N/A | N/A | NS | The lesions on the patient’s back, abdomen right inner thigh, left foot, left side of the scalp had disappeared. |
| Lee et al., 2019 [33] | Traditional Korean medicine (acupuncture, herbal medicine, probiotics) and phototherapy | Case report | 5 mo (case 1), 8 mo (case 2) | 2 | 1M/1F | 37/33 | 4 y // 7 y | N/A | N/A | PASI, VAS | PASI decreased from 7 to 1.2 (Case 1), and 23.2 to 2.2 (Case 2). |
| Mahovic and Mrsic, 2016 [34] | Acupuncture | Case report | 1 mo | 1 | F | 49 | 20 y | N/A | N/A | Arbitrary, visual | Improvement of clinical aspect of psoriatic lesions. |
| Zhu et al., 2011 [35] | Needle acupuncture | Case report | 1 mo | 1 | F | 32 | 10 y | N/A | N/A | NS | Acupuncture induced Koebner phenomenon. |
| Wu et al., 2013 [36] | Acupuncture (for back pain) | Case report | Not reported | 1 | M | 73 | 20 y | N/A | N/A | NS | Acupuncture induced Koebner phenomenon. |
| Zhu et al., 2017 [37] | Acupuncture | Case report | 5 mo | 1 | F | 43 | 20 y | N/A | N/A | Arbitrary, visual | Improvement of clinical aspect of psoriatic lesions. |
| Author | Year | Procedure | Study Design | Study/Treatment Length | Patients Included | Gender | Age (y) | Average Duration of Disease | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| El-Domyati et al., 2013 [14] | 2013 | Wet cupping | Prospective single-arm cohort | 4–6 sessions (2–4 w between sessions) | 50 (8 with psoriasis) | M | Range 17–67 | NS | No improvement was demonstrated in the psoriasis group and Koebner phenomenon appeared on 3 of the patients’ sites of cupping and incisions (upper and lower back). |
| Sharquie et al., 2019 [15] | 2019 | Wet cupping | Retrospective report | 2 w | 24 (16 with psoriasis) | M | Range 25–40 (mean 32.5 for all cases) | NS | Koebner phenomenon appeared on the patients’ sites of cupping and incisions (back). |
| Pavlov and Dimitrova, 2019 [38] | 2019 | Bloodletting and cupping | Case report | NS | 1 | M | 26 | 3 mo | Koebner phenomenon appeared on the upper and lower back, at the site of cupping (paravertebral). |
| Malik et al., 2015 [39] | 2015 | Wet cupping | Case report | 3 w | 1 | M | 30 | 3 y | PASI was initially 2, and 90% of lesions had disappeared after 3 sessions. |
| Polat Ekinci and Pehlivan, 2000 [40] | 2020 | Wet cupping | Case report | 1 w | 1 | M | 39 | 8 mo | Koebner phenomenon appeared on the patient’s back at the site of the incisions and cupping (upper and lower back). |
| Suh et al., 2016 [41] | 2016 | Dry cupping | Case report | NS | 1 | M | 38 | 3 y | Koebner phenomenon appeared on the patients’ sites of cupping and incisions (back and buttocks) and aggravated psoriatic lesions on which cupping was performed. |
| Tang L et al., 2021 [42] | 2021 | Dry cupping | Case report | 2 w | 1 | M | 35 | 2 w | Koebner phenomenon appeared at the cupping area. |
| Vender and Vender, 2015 [43] | 2015 | Dry cupping | Case report | 2 w | 1 | M | 45 | 4 mo | Koebner phenomenon appeared on the patient’s lower back at the sites of cupping. |
| Yu et al, 2013 [44] | 2013 | Dry cupping | Case report | 5 d | 1 | M | 40 | 10 y | Koebner phenomenon appeared on the patient’s back, chest and abdomen, at the site of cupping. |
| Author | Procedure | Study Design | Study/Treatment Length | Patients Included | Gender | Age (y) | Mean Duration of Disease | Comparison Therapy | Severity Measurement | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Fortune et al., 2004 [16] | Psoriasis symptom management program | Patient-preference randomized controlled trial | 6 w | 93: 40 Therapy 53 Control | 31M/62F | 25.2 // 20.2 | 18.5 y // 22.5 y (Range 3–64 y) | Standard pharmacological care only | PASI | Significant difference on depression, anxiety, psoriasis life stress beliefs in severity of consequences of psoriasis, no/freq. of symptoms after therapy and follow-up. |
| Abel et al., 1990 [21] | Support group therapy | Case report | several w/1 mo | 2 | F | 62/56 | 1 d/25 y | N/A | Visual aspect of lesions | Improvement in clinical condition for both patients. |
| Price et al., 1990 [45] | Psychotherapy | RCT, pilot | 8 w | 23: 11 Therapy 12 Control | 12M/11F | 42.8/46 | 17 y/25 y | Control | VAS, HADS | Visible clinical improvement in within the active group. However, the VAS is a rather inaccurate measure of psoriasis, and this portion of the trial was handled in an open fashion. |
| Schmid-Ott, 2000 [46] | Psychotherapy | Case report | 2 y | 1 | F | 48 | 46 y | N/A | NS | Improvement at the end of and 2.5 y after therapy. |
| Shah and Bewly, 2014 [47] | Psychological intervention | Case report | 7 m | 1 | F | 46 | 40 y | N/A | PASI | Improvement of psoriasis (baseline PASI of 24.8, then 0.6 after therapy). |
| Shafii and Shafii, 1979 [48] | Psychodynamic psychotherapy | Case report | NS | 1 | M | NS | NS | N/A | NS | Improvement of lesions and recovery of symptoms after psychotherapy. |
| Tengattini et al., 2019 [49] | Psychological intervention | Prospective, non-randomized trial | 6 mo | 86: 33 Therapy 53 Control | NS | >18 | NS | Standard care | PASI, BSA, PSAB, GAD-7, DLQI, SF-12 | A statistically significant improvement in PASI and BSA was noticed in both groups at 6 months. |
| Zachariae et al., 1996 [50] | Psychotherapy (individual sessions) | Prospective RCT | 12 w | 44: 23 Therapy 21 Control | 18M/26F | 38.7 // 39.5 | NS | Control (no psychological or medical treatment) | PASI, TSS, LDBF | Slight but significant changes in TSS and LDBF in the therapy group, yet not in the control group. No differences between groups concerning PASI. The therapy group displayed significant reductions for all three psoriasis severity measures, w/o changes in the control group. |
| Kabat-Zinn et al., 1998 [51] | Mindfulness meditation-based stress reduction intervention plus UVB and PUVA | Prospective RCT | 13 w | 37: 8 Therapy 1 10 Therapy 2 8 Control 1 11 Control 2 | 17M/20F | 43 | 11.2 y | UVB and PUVA alone (no audiotapes) | Measure designed for this study | Statistically significant difference between tape and no-tape groups, attainment rate being approx. 3.8-times more likely in the former at halfway and clearing points. |
| Frankel and Misch, 1973 [52] | Hypnosis, psychotherapy | Case report | 4 y | 1 | M | 37 | 20 y | N/A | NS | Improvement of psoriatic lesions. |
| Kline et al, 1954 [53] | Hypnosis | Case report | 11 w | 1 | F | 45 | 20 y | N/A | Surface area and visual aspect | Improvement of psoriatic lesions. |
| Waxman, 1973 [54] | Hypnosis | Case report | NS | 1 | F | 38 | 20 y | N/A | NS | Improvement of skin condition (score not mentioned). |
| Tausk and Whitmore, 1999 [55] | Hypnosis with active suggestions of improvement | Prospective RCT, single blind | 3 mo | 11: 5 Therapy 6 Control | NS | NS | >6 mo | Neutral hypnosis with no mention of their disease process | PASI, VAS, Stanford Hypnotizability scale | In the therapy group, the 3 highly hypnotizable patients had 81%, 43%, and 8% reductions in PASI at 3 mo. The 2 highly hypnotizable subjects in control had 31% and 18% amendment in PASI, whereas 2 of the moderately hypnotizable patients showed 13% and 22%, and 2 had a worsening of 36% and 18%, correspondingly. |
| Gaston et al., 1991 [56] | Meditation (T1)/meditation and imagery (T2) | Prospective RCT | 12 w | 18: 5 Therapy 1 4 Therapy 2 5 Control 1 4 Control 2 | 5M/13F | 34.3 | 13.7 y | waiting list (C1)/no treatment (C2) | 4-item scale by Lowe et al | Significant difference between therapy and control groups regarding mean psoriasis ratings after period, w/o any additional impact from imagery. |
| Lazaroff and Shimshoni., 2000 [57] | Medical Resonance Therapy Music plus normal therapy | Prospective RCT | 14 d | 30: 20 Therapy 10 Control | 13M/17 F | Range 18–60 | NS | Normal therapy | Stimulus to scratch and the degree of sickness | Stimulus to scratch and degree of sickness were more reduced in therapy group (significance not specified). |
| Paradisi et al., 2010 [58] | Written emotional disclosure interventions and UVB | Prospective RCT | 4 mo | 40 | NS | UVB alone | PASI, SAPASI, Skindex-29, DHQ-12 | Significant differences in Skindex-29 values between emotional writing group and others. Additionally, patients allocated to emotional writing had a longer period of remission after phototherapy. | ||
| Vedhara et al., 2006 [59] | Written emotional disclosure intervention | Prospective RCT | 12 w | 59: 31 Therapy 28 Control | 32M/27F | 50 | 22 y | Control writing intervention | PASI, HADS, DLQI, POMS | Disease severity and quality of life improved in both groups at the follow-up, w/o significant difference between therapy and control patients. |
| Tabolli et al., 2012 [60] | Pennebaker’s writing emotional disclosure plus educational intervention | Prospective RCT | 2 y | 202: 97 Therapy 102 Control | 124M/78 | 47.94 | approx. 18 y | Educational intervention alone | PASI, SAPASI, Skindex-29, GHQ-12 | No significant differences in clinical or QoL between groups. Slight or no effect on the variables of interest by therapy. |
| Fordham et al., 2014 [61] | MBCT plus standard care | Prospective RCT | 8 w | 29: 13 Therapy 16 Control | 13M/16F | 41.17 | 21.21 y | Standard care | SAPASI, DLQI, HADS, PSS-10 | Those in the MBCT group had a significant improvement concerning symptoms and QoL (DLQI) when compared to control cases. |
| D’Alton et al., 2018 [62] | MBCT (T1), MBSCT (T2), and MBSCT-SH (T3) | Prospective RCT | 8 w | 94: 25 Therapy 1 25 Therapy 2 22 Therapy 3 22 Control | 42M/52F | 49.89 | 24.25 y | Standard care | PASI, DLQI, WHOQOL-BREF | No significant differences on psychological well-being, psoriasis symptom burden, or quality of life compared to TAU at post-treatment, 6- or 12-mo follow-up. |
| van Beugen et al., 2016 [63] | ICBT and standard care | Prospective RCT | 6 mo | 131: 65 Therapy 66 Control | 67M/64F | 52.69 // 53.45 | 18.03 y // 15.16 y | Standard care | PASI, SAPASI, ISDL, RAND-36 | Larger improvements in ICBT compared to CAU regarding physical functioning and impact on daily activities, though not in psychological functioning, at 6-mo follow-up. |
| Spillekom-Van Koulil, 2018 [64] | ICBT | Case report | 5 mo/6 mo | 2 | 1M/1F | 64/26 | 28 y/1 y | N/A | NS | Improved physical and psychological wellbeing, sustained at 6-mo follow-up. |
| Bundy et al., 2013 [65] | eTIPs—CBT | Prospective RCT | 6 w | 126: 61 Therapy 65 Control | 59M/67F | 45 | >16 y | usual care | PASI, HADS, DLQI | No significant difference in PASI scores after intervention. Results may be limited by the large quantity of unavailable data. |
| Seng and Nee, 1997 [66] | Support group therapy | Prospective single-arm cohort | 7 w | 10 | 6M/4F | 37.5 | 15 y | N/A | Knowledge, acceptance and coping with the disease | Patients felt that the program enhanced their knowledge of psoriasis and increased their confidence in coping with the disease. |
| Piaserico et al, 2016 [67] | Biofeedback and CBT plus UVB | Prospective RCT | 8 w | 40: 20 Therapy 20 Control | 12M/28F | 49.7 | 17.7 y | UVB alone | PASI, GHQ-12, Skindex-29, STAI | Patients undergoing therapy had a significant reduction in PASI score, from 9 at baseline to 3.8 and 2.5 at 4 and 8 w, correspondingly. Likewise, 65% of cases in the therapy group achieved PASI75 whereas only 15% of standard UVB patients did so at 8 w. |
| Goodman, 1994 [68] | Thermal biofeedback | Case report | 13 w | 1 | F | 56 | 10 y | N/A | Skin temperature | All 11 presenting psoriasis lesions vanished, and any new lesions appearing during therapy disappeared w/o visible scarring. |
| Hughes et al., 1981 [69] | Biothermal feedback and supportive psychotherapy | Case report | 7 m | 1 | M | 31 | 2 y | N/A | Psoriasis Rating Scale (developed by the authors) | Marked dermatological improvement; inability to lower the temperature at the plaque site. |
| Author | Procedure | Study Design | Study/Treatment Length | Patients Included | Gender | Mean Age (y) | Average Duration of Psoriasis | Comparison Therapy | Severity Measurement | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Ben-Amitai and David., 2009 [17] | DSC | Open, uncontrolled prospective trial | 2 w | 17 | 8M/9F | Range 10–18 | NS | N/A | PASI | >75% improvement in six cases, and 50–75% amelioration (moderate) in five patients. At 6 mo. Follow-up, 12 patients were relapse-free, the other 5 presented mild relapse. |
| David et al., 2005 [18] | DSC | Multicenter controlled cross-sectional study | 224 d (mean), 140 d (median) | 1198: 460 Psoriasis 738 Control | 261 M/199 F (P) 296 M/442 F (C) | 48 (P) 47 (C) | 20 y | Benign skin conditions including contact dermatitis and seborrhea (Control) | NS | Elastosis, solar lentigines, poikiloderma, and facial wrinkles—significantly more common in psoriatic patients than controls, in relation with Dead Sea exposure time. No correlation with increased risk of malignant melanoma or NMSC in psoriatic patients. |
| Eysteinsdóttir et al., 2014 [19] | Balneotherapy with geothermal sea water and NB-UVB | Prospective RCT | 6 w | 68: 22 GSW 22 IT-GSW 24 UVB | 39M/29F | 41/42.2/ 37.9 | 20 y/ 16.4 y/ 12.3 y | Bathing in GSW+NB-UVB vs. intensive treatment with GSW and NB-UVB vs. NB-UVB therapy alone | PASI, DLQI, Lattice score, BMI | % of patients attaining PASI 75 and 90 was significantly greater for both GSW and IT-GSW regimens than for NB-UVB monotherapy. Faster clinical and histological improvement, longer remission period and lower NB-UVB doses than for GSW-regimens than standard NB-UVB monotherapy. |
| Baros et al., 2014 [23] | Balneotherapy | Prospective RCT | 6 w | 60: 19 T1 15 T2 26 C | 37M/27F | NS | NS | Standard therapy vs. balneotherapy vs. standard therapy plus balneotherapy | PASI, CRP, antistreptolysin O titer, iron, uric acid | Statistically significant difference in remission length for patients treated with combination therapy and patients treated with standard therapeutic modalities, with best results for combination therapy. |
| Gambichler et al., 2001 [24] | Highly concentrated salt water balneotherapy | Prospective RCT, single-blind, left-right | 8 w | 10 | 4M/6F | 36 y | 2.5 y | Tap water balneotherapy on opposite elbow | Severity score pertaining to desquamation, erythema, and infiltration of the psoriatic plaques. | Highly significant decrease in the baseline score, w/o significant difference between pre-treatment with salt water or tap water. |
| Cohen et al., 2005 [25] | DSC | Prospective single-arm cohort | 2 w (6–33 d) | 70 | 40M/30F | Range 19–78 y | Range 4–30 y | N/A | PASI, BPSS (Beer Sheva Psoriasis Severity Score) | 75.9% reduction in PASI and 57.5% reduction in the mean BPSS. |
| * Langeland et al., 2013 [26] | Gran canaria climatotherapy and patient education program | Prospective single-arm cohort | 4 mo (3 w program) | 254 | 152M/102F | 47 | 24 y | N/A | PASI, MHC-SF | Positive mental health and health-related emotional distress recovered markedly after treatment. The longer the duration of psoriasis, and the presence of comorbidities, the greater the aforementioned improvement. |
| * Wahl et al., 2015 [27] | Gran Canaria climate therapy | Prospective single-arm cohort | 3 w | 254 | 152M/102F | 47 | 24 y | N/A | PASI, SAPASI, Health Education Impact Questionnaire (heiQ) | SAPASI score improved significantly, as well as self-management; at 3 mo. follow-up, only emotional distress and disease severity stayed significantly ameliorated. |
| Hodak et al., 2003 [70] | DSC | Prospective single-arm cohort | 4 w | 27 | 18M/9F | Range 24–73 y | 4–30 y | N/A | PASI, quantitative histologic measures | Average 81.5% decrease in PASI score, with complete therapeutic response in 13 subjects, marked in 5, moderate in 6, and slight in 2. |
| Czarnowicki et al., 2011 [71] | DSC | Retrospective comparison | 4 w | 40 | 25M/15F | NS | NS | Climatotherapy alone vs. climatotherapy plus MTX | PASI, BMI, BSA | DSC did not show better results in patients treated simultaneously with methotrexate |
| Emmanuel et al., 2020 [72] | DSC | Prospective single-arm cohort | 4 w | 18 | 12M/6F | 52.2 | 34.2 | N/A | PASI, IGA, NAPSI, NAPPA, DLQI, EQ-5D-3L, BMI | DSC led to PASI reduction of 88%, a mean decrease of 2.3 (76.7%) on the 5-IGA, and a QoL improvement as measured by DLQI and EuroQol 5D index. |
| Emmanuel et al., 2019 [73] | DSC | Retrospective | NS | 6 | 5M/1F | NS | NS | N/A | PASI, BSA, histological specimens | 60.2% of new plaque areas reemerged within the site of former plaques, proved by histopathology. |
| Even-Paz et al. [74] | DSC | Prospective, non-randomized | 4 w | 45 | 24M/21F | NS | NS | Sun exposure time of 3.0/4.5/6.0 h daily | PASI | 3 h of daily sun exposure at the Dead Sea, in two equal sessions from 09:00 and 14:00, respectively, were sufficient in treating psoriasis in July and August. |
| Frentz et al., 1999 [75] | DSC | Retrospective nation-wide cohort | 6.1 y (0–22) treatment length NS | 1738 | 872M/866F | 43 | NS | N/A | Presence of NMSC or other cancers at follow-up | Overall risk of cancer in patients undergoing DSC surpassed that expected in the general population, due to NMSC with an unusual distribution among body sites, age groups and sexes in these patients: young individuals and at multiple sites, numerous BCC being frequent in young women. |
| Harari et al., 2011 [76] | DSC | Retrospective | 4 w treatment (study 2003–2007) | 605 | 441M/164F | 48.08 | 25.36 y | DSC for <40 y age at onset of psoriasis vs. >40 y at onset | PASI, BSA | 74% of the patients <40 y at psoriasis onset had PASI 95, as opposed to 62% for >40 y. Therapeutic effect was inversely associated with the age of the patient at disease onset. |
| Harari et al., 2007 [77] | DSC | Prospective, single-arm cohort | 4 w | 64 | 42M/22F | 41.4 | 16.8 y | N/A | PASI, QoL VAS | All subjects attained PASI 50, and 75.9% of cases reached PASI 75 after 1 mo of DSC, with a median remission length of 23.1 w and median therapeutic effect duration of 33.6 w. Younger patient age at therapy was correlated with a longer remission time. |
| Harari et al., 2016 [78] | DSC | Retrospective report | 3–4 w | 719 | 505M/214F | 51.24 | 28.01 y | N/A | PASI, BMI, body surface involvement | Previous DSC sessions were a positive predictor for an improved PASI, with a positive association between psoriasis duration (and younger age at onset) and PASI 90. More patients with photo skin type II (Fitzpatrick) achieved PASI 90 than other types. |
| Harari and Shani., 1997 [79] | DSC | Prospective, non-randomized trial | 4 w | 740 | 428M/312F | Range 10–72 y | NS | N/A | BSA, rheumatologic index | Percentage of clearance was best (>72%) for patients staying in the sun at least 7 h daily; lack of psychologically support led to disease clearance in 68.9% of patients, compared to 75.8% of those supported. Previous DSC sessions, moderate to severe skin surface involvement, and coexistence of arthritis increased chances of psoriasis clearance. |
| Kushelevsky et al., 1998 [80] | DSC | Retrospective comparison | 4 w | 80 | NS | NS | NS | Exposure to UVB exposure in other climate therapy regions (Sweden, Switzerland, Germany, Bulgaria, New Zealand) | MED | Mean UVB exposure dose in DSC stands among the lowest reported for psoriasis clearance. Despite comparable on a monthly basis, cumulative annual phototherapy exposure is noticeably higher than the UVB doses provided on a 4-w DSC session. |
| Kushelevsky et al., 1996 [81] | DSC | Prospective, non-randomized | 4 w | 688 (study 1) 502 (study 2) 1142 (study 3) | 320M/368F // 238M/264F // 583M/559F | <10–>60 // >65 | NS | N/A | BSA | Duration of psoriasis of 40–49 y resulted in a higher clearance rate of 78.6%. The clearance rate in patients with disease onset at the ages of 10–19 y (77.8%) was superior to those in which the disease appeared 60 y (56.2%) // A mean decrease in systolic and diastolic blood pressures during DSC was noticed, regardless of sex and age, in all groups. |
| Nissen et al., 1998 [82] | DSC and bathing | Prospective, non-randomized | 4 w | 21: 10 Psoriasis 11 Healthy | NS | NS | NS | Psoriasis vs. healthy skin exposed to UV | RIA for enkephalin | Total clearance of psoriasis at sample sites; average reduction of 21% in enk levels. |
| Schewach-Millet et al., 1989 [83] | DSC | Retrospective study | 3 y (between 2 and 8 y)—3–4 w yearly | 19 | NS | NS | NS | N/A | Histological specimens | Intensification of epidermal pigmentation when compared to pretreatment biopsy specimens in certain cases, w/o epidermal dystrophy or melanocytic atypia. |
| Trøstrup et al., 2019 [84] | DSC | Prospective single-arm cohort | 4 w | 49 | 28M/21F | 51.86 | NS | N/A | PASI, DLQI | 11/49 patients reached PASI; 10/49 presented increased PASI; age, sex, previous DSC sessions, and duration of observation period did not affect endpoints. DSC led to a significantly increased DLQI score in 60% of cases, even several mo follow-up, whereas 20% of patients presented a marked decrease in PASI. |
| Bogdanov et al., 2012 [85] | Climatotherapy and phototherapy | Prospective, non-randomized | 2 w | 93: 45 psoriasis placate 12 psoriasis palmoplantaris 36 control | 55M/38F | 45 | NS | Narrowband phototherapy alone | DLQI | Combined climatotherapy has a significantly better beneficial effect on the QoL of the patients with psoriasis placate than phototherapy alone. |
| Golusin et al., 2015 [86] | Rusanda Spa balneotherapy plus calcipotriol | Prospective RCT | 3 w | 60: 30 Therapy 30 Control | 28M/32F | 55.46 (T) 41.73 (C) | NS | Balneotherapy plus calcipotriol vs. calcipotriol alone | PASI | Therapy group showed a decrease in PASI score by 59.45%, whereas in the control group it was 39.34%. Topical calcipotriol associated with Rusanda Spa balneotherapy is more efficient than topical calcipotriol alone. |
| Peroni et al., 2008 [87] | Comano spa balneotherapy | Prospective, non-randomized | 1–2 w | 280: 124 Therapy 156 Control | 176M/104F | Range 18–85 y | NS | BPT (therapy) vs. balneotherapy alone | PASI, SAPASI, Skindex-29, BSA | 1 w balneotherapy or BPT was enough to obtain statistically significant PASI score reduction. Both 2 w balneotherapy and BPT groups brought greater psoriasis amelioration with reduction in both PASI score and BSA. |
| Péter et al., 2017 [88] | Balneotherapy | Prospective single-arm cohort | 3 w | 80 | 35M/45F | 63.7 | NS | N/A | PASI, CRP level | After therapy, both PASI and CRP levels presented significant improvement. |
| Tabolli et al., 2009 [89] | Balneotherapy and BPT | Prospective, non-randomized | 2 w | 111: 66 Therapy 45 Control | 67M/24F | >18 y | 25 < 10 y; 81 > 10 y | BPT vs. Comano balneotherapy alone | SAPASI, Skindex-29, GHQ-12, SF-36 | A decrease >50% after therapy in SAPASI 50 score was attained by 42% and 37% of patients in the BPT and BT groups, correspondingly. BPT group showed a statistically significant reduction in the number of GHQ-12 positive cases. |
| Nilsen et al., 2009 [90] | Gran Canaria climate therapy | Prospective single-arm cohort | 2 w | 20 | 14M/6F | 47.2 | NS | N/A | PASI, UV exposure, Spectral UVB (280–315 nm), UVA (315–400 nm) and CIE-weighted UV | Reduction in overall PASI score in all patients; no significant correlation between the reduction in psoriasis area severity index scores and UV doses; |
| Martin et al., 2015 [91] | Balneotherapy with selenium-rich spa water (La Roche-Posay) | Prospective single-arm cohort | 3 w | 29 | 32M/22F | 59 | NS | Composition of skin microbial communities associated with unaffected and affected skin. | PASI, PGA, Shannon Diversity Index | PASI scores decreased post-balneotherapy. Poor bacterial biodiversity was observed, with the bacterial communities being similar on both unaffected and affected adjacent skin. Family analysis identified, for the first time, Xanthomonadaceae belonging to Proteobacteria phylum and recognized as keratolytic, was linked with clinical improvement after 3 w balneotherapy. |
| Pinton et al., 1995 [92] | Balneotherapy with selenium-rich spa water (La Roche-Posay) | Prospective single-arm cohort | 3 w | 92 | 46M/46F | 47.3 | NS | N/A | PASI, circulating soluble interleukin 2 receptor (sCD25) level | Average PASI score decreased by 47 ± 4%. In total, 44 patients improved by >50%, 80 subjects responded with a mean decrease in the PASI score of 52-6 and 10 were stable with 10% improvement or less. Men responded markedly better than women. An association between PASI score decrease and the rise in the plasma selenium levels was noticed. |
| Wang et al., 2020 [93] | Balneotherapy plus Chinese herbal medicine | Prospective RCT | NS | 190: 97 Therapy 93 Control | 103M/87F | NS | NS | Consolidated CHM balneotherapy and NB-UVB (CTG) vs. unconsolidated (stopped after PASI drop to 1.8–2.0) (UTG) | PASI | No significant difference in PASI score between the two groups at the initiation and the termination of therapy. However, the mean remission length in CTG was 10.99 mo, significantly longer than UTG (7.94 mo). |
| Tsoureli-Nikita et al., 2002 [94] | Balneotherapy with Leopoldine spa water | Prospective single-arm cohort | 4 w | 10 | 7M/3F | Range 28–53 y | NS | Leopoldine water vs. double-distilled water | PASI, immunohistological study | Average PASI improvement for Leopoldine spa water treated arms was 85.9%, whereas double-distilled water treated arms had 50.5% PASI improvement. Significant differences between cutaneous samples taken before and after 4 w of Leopoldine spa water therapy. |
| Melandri et al., 2019 [95] | Liman peloid baths and heliotherapy at Cervia spa, Emilia, Italy | Prospective RCT | 2.5 w | 91: 56 Therapy 35 Control | 57M/24F | 52.3/57.9 | NS | Liman peloid application followed by bath therapy vs. clay peloid mixed with tap water | PASI | Compared with the control group, there was a significant improvement in PASI score and fewer psoriasis recurrences in the therapy group, aside from a marked reduction in the topical use of cortisone and nonsteroid drugs. |
4. Discussion
4.1. Acupuncture
4.2. Cupping Therapy
4.3. Psychotherapy, Stress Management and Meditation
4.4. Balneotherapy and Climatotherapy
5. Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Harden, J.L.; Krueger, J.G.; Bowcock, A.M. The immunogenetics of Psoriasis: A comprehensive review. J. Autoimmun. 2015, 64, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Colombo, D.; Cassano, N.; Bellia, G.; Vena, G.A. Gender medicine and psoriasis. World J. Dermatol. 2014, 3, 36–44. [Google Scholar] [CrossRef]
- Langley, R.G.; Ellis, C.N. Evaluating psoriasis with Psoriasis Area and Severity Index, Psoriasis Global Assess-ment, and Lattice System Physician’s Global Assessment. J. Am. Acad. Dermatol. 2004, 51, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.A.; Jones, H.; Mallbris, L.; Duffin, K.C.; Krueger, G.G.; Clegg, D.O.; Szumski, A. The Physician Global Assessment and Body Surface Area composite tool is a simple alternative to the Psoriasis Area and Severity Index for assessment of psoriasis: Post hoc analysis from PRISTINE and PRESTA. Psoriasis Targets Ther. 2018, 8, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Cardiff University Department of Dermatology Website. Available online: www.cardiff.ac.uk/dermatology/quality-of-life/dermatology-quality-of-life-index-dlq (accessed on 20 February 2021).
- Rønholt, K.; Iversen, L. Old and New Biological Therapies for Psoriasis. Int. J. Mol. Sci. 2017, 18, 2297. [Google Scholar] [CrossRef]
- Iannone, L.F.; Bennardo, L.; Palleria, C.; Roberti, R.; De Sarro, C.; Naturale, M.D.; Dastoli, S.; Donato, L.; Manti, A.; Valenti, G.; et al. Safety profile of biologic drugs for psoriasis in clinical practice: An Italian prospective pharmacovigilance study. PLoS ONE 2020, 15, e0241575. [Google Scholar] [CrossRef]
- Sbidian, E.; Chaimani, A.; Garcia-Doval, I.; Do, G.; Hua, C.; Mazaud, C.; Droitcourt, C.; Hughes, C.; Ingram, J.R.; Naldi, L.; et al. Systemic pharmacological treatments for chronic plaque psoriasis: A network meta-analysis. Cochrane Database Syst. Rev. 2017, 12, CD011535. [Google Scholar] [CrossRef]
- Dattola, A.; Silvestri, M.; Tamburi, F.; Amoruso, G.F.; Bennardo, L.; Nisticò, S.P. Emerging role of anti-IL23 in the treatment of psoriasis: When humanized is very promising. Dermatol. Ther. 2020, 33, e14504. [Google Scholar] [CrossRef]
- Dattola, A.; Silvestri, M.; Bennardo, L.; Passante, M.; Rizzuto, F.; Dastoli, S.; Patruno, C.; Bianchi, L.; Nisticò, S.P. A novel vehicle for the treatment of psoriasis. Dermatol. Ther. 2020, 33, e13185. [Google Scholar] [CrossRef]
- Talbott, W.; Duffy, N. Complementary and Alternative Medicine for Psoriasis: What the Dermatologist Needs to Know. Am. J. Clin. Dermatol. 2015, 16, 147–165. [Google Scholar] [CrossRef]
- Gamret, A.C.; Price, A.; Fertig, R.M.; Lev-Tov, H.; Nichols, A.J. Complementary and alternative medicine therapies for psoriasis: A systematic review. JAMA Dermatol. 2018, 154, 1330–1337. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Robertson, A.D.; Wu, J.; Schupp, C.; Lebwohl, M.G. Undertreatment, Treatment Trends, and Treatment Dissatisfaction Among Patients with Psoriasis and Psoriatic Arthritis in the United States: Findings from the National Psoriasis Foundation Surveys, 2003–2011. JAMA Dermatol. 2013, 149, 1180–1185. [Google Scholar] [CrossRef]
- El-Domyati, M.; Saleh, F.; Barakat, M.; Mohamed, N. Evaluation of cupping therapy in some dermatoses. Egypt Dermatol. Online J. 2013, 9, 2. [Google Scholar]
- Al-Jaralla, F.A.; Sharquie, K.E. Cupping (Hijama) in Skin diseases with positive Koebner’s Phenomenon: What is New? J. Clin. Exp. Investig. 2019, 10, em00726. [Google Scholar] [CrossRef]
- Fortune, D.G.; Richards, H.L.; Griffiths, C.E.M.; Main, C.J. Targeting cognitive-behaviour therapy to patients’ implicit model of psoriasis: Results from a patient preference controlled trial. Br. J. Clin. Psychol. 2004, 43, 65–82. [Google Scholar] [CrossRef]
- Ben-Amitai, D.; David, M. Climatotherapy at the Dead Sea for Pediatric-Onset Psoriasis Vulgaris. Pediatr. Dermatol. 2009, 26, 103–104. [Google Scholar] [CrossRef]
- David, M.; Tsukrov, B.; Adler, B.; Hershko, K.; Pavlotski, F.; Rozenman, D.; Hodak, E.; Paltiel, O. Actinic damage among patients with psoriasis treated by climatotherapy at the Dead Sea. J. Am. Acad. Dermatol. 2005, 52, 445–450. [Google Scholar] [CrossRef]
- Liao, S.J.; Liao, T.A. Acupuncture Treatment for Psoriasis: A Retrospective Case Report. Acupunct. Electro Ther. Res. 1992, 17, 195–208. [Google Scholar] [CrossRef]
- Ye, L. Treatment of psoriasis by acupuncture plus Chinese herbs: A report of 80 cases. J. Acupunct. Tuina Sci. 2004, 2, 16–17. [Google Scholar] [CrossRef]
- Abel, E.A.; Moore, U.S.; Glathe, J.P. Psoriasis Patient Support Group and Self-Care Efficacy as an Adjunct to Day Care Center Treatment. Int. J. Dermatol. 1990, 29, 640–643. [Google Scholar] [CrossRef]
- Eysteinsdóttir, J.H.; Ólafsson, J.H.; Agnarsson, B.A.; Lúðvíksson, B.R.; Sigurgeirsson, B. Psoriasis treatment: Faster and long-standing results after bathing in geothermal seawater. A randomized trial of three UVB phototherapy regimens. Photodermatol. Photoimmunol. Photomed. 2013, 30, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Baros, D.N.; Gajanin, V.S.; Gajanin, R.B.; Zrnić, B. Comparative analysis of success of psoriasis treatment with standard therapeutic modalities and balneotherapy. Med. Pregl. 2014, 67, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Gambichler, T.; Rapp, S.; Senger, E.; Altmeyer, P.; Hoffmann, K. Balneophototherapy of psoriasis: Highly concentrated salt water versus tap water—A randomized, one-blind, right/left comparative study. Photodermatol. Photoimmunol. Photomed. 2001, 17, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.D.; Van-Dijk, D.; Naggan, L.; Vardy, D.A. Effectiveness of climatotherapy at the Dead Sea for psoriasis vulgaris: A community-oriented study introducing the ‘Beer Sheva Psoriasis Severity Score’. J. Dermatol. Treat. 2005, 16, 308–313. [Google Scholar] [CrossRef]
- Langeland, E.; Robinson, H.S.; Moum, T.; Larsen, M.H.; Wahl, A.K. Mental health among people with psoriasis undergoing patient education in climate therapy. Scand. J. Psychol. 2013, 54, 508–514. [Google Scholar] [CrossRef]
- Wahl, A.; Langeland, E.; Larsen, M.; Robinson, H.; Osborne, R.; Krogstad, A. Positive Changes in Self-management and Disease Severity Following Climate Therapy in People with Psoriasis. Acta Derm. Venereol. 2015, 95, 317–321. [Google Scholar] [CrossRef]
- Jerner, B.; Skogh, M.; Vahlquist, A. A controlled trial of acupuncture in psoriasis: No convincing effect. Acta Derm. Venereol. 1997, 77, 154–156. [Google Scholar]
- Pan, H.-D.; Qi, X.-L.; Wang, L.; He, J.-Y.; Liu, J.-L.; Zhang, Y.-B.; Tian, Y.; Duan, X.-W.; Yang, H.-Y.; Bai, Y.-P. Whether Fire-needle Therapy Benefits Plaque Psoriasis: A Multicenter, Randomized, and Controlled Trial. Chin. J. Integr. Med. 2019, 25, 259–263. [Google Scholar] [CrossRef]
- Lu, C.-J.; Xiang, Y.; Xie, X.-L.; Xuan, M.-L.; He, Z.-H. A randomized controlled single-blind clinical trial on 84 outpatients with psoriasis vulgaris by auricular therapy combined with optimized Yinxieling Formula. Chin. J. Integr. Med. 2012, 18, 186–191. [Google Scholar] [CrossRef]
- Jorge, M.; Silverio-Lopes, S.; Carneiro-Suliano, L. Case Report: Ear Acupuncture Therapy for Psoriasis. J. Complement. Altern. Med Res. 2016, 1, 1–8. [Google Scholar] [CrossRef]
- Jeon, Y.-C. Treatment for an Adult Patient with Psoriasis with Traditional Korean Medicine, Especially Sa-Am Acupuncture and Herbal Medicine. J. Acupunct. Meridian Stud. 2016, 9, 88–92. [Google Scholar] [CrossRef]
- Lee, S.-R.; Kim, S.; Park, C.-E.; Lee, J.-H.; Lee, D.-H. Effect of Korean medicine as add-on therapy to phototherapy for psoriasis: Two case report. Medicine 2019, 98, e14526. [Google Scholar] [CrossRef]
- Mahović, D.; Mrsić, F. Acupuncture as a Complementary Method of Traditional Psoriasis Treatment: Myth or Reality? Acta Dermatovenerol. Croat. 2016, 24, 221–222. [Google Scholar]
- Zhu, J.; Arsovska, B.; Kozovsk, K. Acupuncture Treatment for Psoriasis Present on the Soles. J. Surg. 2017, 1, 5. [Google Scholar] [CrossRef][Green Version]
- Wu, J.J.; Caperton, C. Psoriasis Flare from Koebner’s Phenomenon after Acupuncture. N. Engl. J. Med. 2013. [Google Scholar] [CrossRef]
- Zhu, L.-L.; Hong, Y.; Zhang, L.; Huo, W.; Zhang, L.; Chen, H.-D.; Gao, X.-H. Needle Acupuncture–Induced Koebner Phenomenon in a Psoriatic Patient. J. Altern. Complement. Med. 2011, 17, 1097–1098. [Google Scholar] [CrossRef]
- Pavlov, S.; Dimitrova, M. Koebner phenomenon caused by bloodletting and cupping therapy in a patient with psoriasis. Scr. Sci. Med. 2019, 51, 32–36. [Google Scholar] [CrossRef]
- Malik, I.A.; Akhter, S.; Kamal, M.A. Treatment of psoriasis by using Hijamah: A case report. Saudi J. Biol. Sci. 2015, 22, 117–121. [Google Scholar] [CrossRef]
- Ekinci, A.P.; Pehlivan, G. Cupping therapy as alternative medicine turns into a trigger of disease via the Koebner phenomenon: A case report of Hijama-induced psoriasis and review of the literature. Dermatol. Ther. 2020, 33, e14264. [Google Scholar] [CrossRef]
- Suh, H.Y.; Kim, K.H.; Jeon, J.W.; Ahn, J.Y.; Park, M.Y.; Youn, J.I. Koebner Phenomenon Related to Cupping Therapy in a Patient with Psoriasis. Korean J. Dermatol. 2016, 54, 499–500. [Google Scholar]
- Tang, L.; Liao, Y.; Xu, J.; Li, C. Koebner phenomenon induced by cupping therapy in the unstable stage of psoriasis: A case report. Dermatol. Ther. 2021, e14852. [Google Scholar] [CrossRef]
- Vender, R.; Vender, R. Paradoxical, Cupping-Induced Localized Psoriasis: A Koebner Phenomenon. J. Cutan. Med. Surg. 2015, 19, 320–322. [Google Scholar] [CrossRef]
- Yu, R.-X.; Hui, Y.; Li, C.-R. Köebner phenomenon induced by cupping therapy in a psoriasis patient. Dermatol. Online J. 2013, 19, 18575. [Google Scholar]
- Price, M.L.; Mottahedin, I.; Mayo, P.R. Can psychotherapy help patients with psoriasis? Clin. Exp. Dermatol. 1991, 16, 114–117. [Google Scholar] [CrossRef]
- Schmid-Ott, G. Depth Psychology-Founded Psychotherapy of Psoriatic Patients—Treatment of a Patient with Chronic Recidivating Psoriasis vulgaris. Dermatol. Psychosom. 2000, 1, 112–116. [Google Scholar] [CrossRef]
- Shah, R.; Bewley, A. Psoriasis: ‘the badge of shame’. A case report of a psychological intervention to reduce and potentially clear chronic skin disease. Clin. Exp. Dermatol. 2014, 39, 600–603. [Google Scholar] [CrossRef]
- Shafii, M.; Shafii, S.L. Exploratory Psychotherapy in the Treatment of Psoriasis. Twelve hundred years ago. Arch. Gen. Psychiatry 1979, 36, 1242–1245. [Google Scholar] [CrossRef]
- Tengattini, V.; Foschi, L.; Rucci, P.; Iommi, M.; Fantini, M.P.; Patrizi, A.; Bardazzi, F. Effectiveness of psychological intervention in psoriatic patients relative to standard care. Eur. J. Dermatol. 2019, 29, 231–233. [Google Scholar] [CrossRef]
- Zachariae, R.; Øster, H.; Bjerring, P.; Kragballe, K. Effects of psychologic intervention on psoriasis: A preliminary report. J. Am. Acad. Dermatol. 1996, 34, 1008–1015. [Google Scholar] [CrossRef]
- Kabat-Zinn, J.; Wheeler, E.; Light, T.; Skillings, A.; Scharf, M.J.; Cropley, T.G.; Hosmer, D.; Bernhard, J. Influence of a mindfulness meditation-based stress reduction intervention on rates of skin clearing in patients with moderate to severe psoriasis undergoing phototherapy (UVB) and photochemotherapy (PUVA). Psychosom. Med. 1998, 60, 625–632. [Google Scholar] [CrossRef]
- Frankel, F.H.; Misch, R.C. Hypnosis in a case of long-standing psoriasis in a person with character problems. Int. J. Clin. Exp. Hypn. 1973, 21, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Kline, M.V. Psoriasis and hypnotherapy: A case report. J. Clin. Exp. Hypn. 1954, 2, 318–322. [Google Scholar] [CrossRef]
- Waxman, D. Behaviour therapy of psoriasis—A hypnoanalytic and counter-conditioning technique. Postgrad. Med. J. 1973, 49, 591–595. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tausk, F.; Whitmore, S.E. A pilot study of hypnosis in the treatment of patients with psoriasis. Psychother. Psychosom. 1999, 68, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Gaston, L.; Crombez, J.C.; Lassonde, M.; Bernier-Buzzanga, J.; Hodgins, S. Psychological stress and psoriasis: Experimental and prospective correlational studies. Acta Derm. Venereol. Suppl. 1991, 156, 37–43. [Google Scholar]
- Lazaroff, I.; Shimshoni, R. Effects of Medical Resonance Therapy Music on patients with psoriasis and neuro-dermatitis—A pilot study. Integr. Physiol. Behav. Sci. 2000, 35, 189–198. [Google Scholar] [CrossRef]
- Paradisi, A.; Abeni, D.; Finore, E.; Di Pietro, C.; Sampogna, F.; Mazzanti, C.; Pilla, M.A.; Tabolli, S. Effect of written emotional disclosure interventions in persons with psoriasis undergoing narrow band ultraviolet B phototherapy. Eur. J. Dermatol. 2010, 20, 599–605. [Google Scholar] [CrossRef]
- Vedhara, K.; Morris, R.M.; Booth, R.; Horgan, M.; Lawrence, M.; Birchall, N. Changes in mood predict disease activity and quality of life in patients with psoriasis following emotional disclosure. J. Psychosom. Res. 2007, 62, 611–619. [Google Scholar] [CrossRef]
- Tabolli, S.; Naldi, L.; Pagliarello, C.; Sampogna, F.; Di Pietro, C.; Spagnoli, A.; Abeni, D.; The Italian Writing Exercise Study Group. Evaluation of the impact of writing exercises interventions on quality of life in patients with psoriasis undergoing systemic treatments. Br. J. Dermatol. 2012, 167, 1254–1264. [Google Scholar] [CrossRef]
- Fordham, B.; Griffiths, C.; Bundy, C. A pilot study examining mindfulness-based cognitive therapy in psoriasis. Psychol. Heal. Med. 2014, 20, 121–127. [Google Scholar] [CrossRef]
- D’Alton, P.; Kinsella, L.; Walsh, O.; Sweeney, C.; Timoney, I.; Lynch, M.; O’Connor, M.; Kirby, B. Mindfulness-Based Interventions for Psoriasis: A Randomized Controlled Trial. Mindfulness 2019, 10, 288–300. [Google Scholar] [CrossRef]
- Van Beugen, S.; Ferwerda, M.; Koulil, S.S.-V.; Smit, J.V.; Zeeuwen-Franssen, M.E.; Kroft, E.B.; De Jong, E.M.; Otero, M.E.; Donders, A.R.T.; Van De Kerkhof, P.C.; et al. Tailored Therapist-Guided Internet-Based Cognitive Behavioral Treatment for Psoriasis: A Randomized Controlled Trial. Psychother. Psychosom. 2016, 85, 297–307. [Google Scholar] [CrossRef]
- Koulil, S.; Ferwerda, M.; Beugen, S.; Middendorp, H.; Kerkhof, P.; Riel, P.A.; Evers, A. Tailored Therapist-guided Internet-based Cognitive-behavioural Treatment for Psoriasis and Rheumatoid Arthritis: Two Case Reports. Acta Derm. Venereol. 2018, 98, 225–233. [Google Scholar] [CrossRef]
- Bundy, C.; Pinder, B.; Bucci, S.; Reeves, D.; Griffiths, C.; Tarrier, N. A novel, web-based, psychological intervention for people with psoriasis: The electronic Targeted Intervention for Psoriasis (eTIPs) study. Br. J. Dermatol. 2013, 169, 329–336. [Google Scholar] [CrossRef]
- Seng, T.K.; Nee, T.S. Group therapy: A useful and supportive treatment for psoriasis patients. Int. J. Dermatol. 1997, 36, 110–112. [Google Scholar] [CrossRef]
- Piaserico, S.; Marinello, E.; Dessi, A.; Linder, M.; Coccarielli, D.; Peserico, A. Efficacy of Biofeedback and Cognitive-behavioural Therapy in Psoriatic PatientsA Single-blind, Randomized and Controlled Study with Added Narrow-band Ultraviolet B Therapy. Acta Derm. Venereol. 2014, 964, 91–95. [Google Scholar] [CrossRef]
- Goodman, M. An hypothesis explaining the successful treatment of psoriasis with thermal biofeedback: A case report. Biofeedback Self Regul. 1994, 19, 347–352. [Google Scholar] [CrossRef]
- Hughes, J.E.; Barraclough, B.M.; Hamblin, L.G.; White, J.E. Psychiatric Symptoms in Dermatology Patients. Br. J. Psychiatry 1983, 143, 51–54. [Google Scholar] [CrossRef]
- Hodak, E.; Gottlieb, A.B.; Segal, T.; Politi, Y.; Maron, L.; Sulkes, J.; David, M. Climatotherapy at the Dead Sea is a remittive therapy for psoriasis: Combined effects on epidermal and immunologic activation. J. Am. Acad. Dermatol. 2003, 49, 451–457. [Google Scholar] [CrossRef]
- Czarnowicki, T.; Ingber, A.; Ruzicka, T.; Novack, L.; Harari, M. Does Simultaneous Methotrexate Treatment Improve Dead Sea Climatotherapy Outcomes in Plaque Psoriasis? Psoriasis Forum 2011, 17, 220–224. [Google Scholar] [CrossRef]
- Emmanuel, T.; Lybæk, D.; Johansen, C.; Iversen, L. Effect of Dead Sea Climatotherapy on Psoriasis; A Prospective Cohort Study. Front. Med. 2020, 7, 83. [Google Scholar] [CrossRef]
- Emmanuel, T.; Lybaek, D.; Johansen, C.; Iversen, L. Non-random Plaque-site Recurrence of Psoriasis in Patients Treated with Dead Sea Climatotherapy. Acta Derm. Venereol. 2019, 99, 909–910. [Google Scholar] [CrossRef]
- Even-Paz, Z.; Efron, D.; Kipnis, V.; Abels, D. How much Dead Sea sun for psoriasis? J. Dermatol. Treat. 1996, 7, 17–19. [Google Scholar] [CrossRef]
- Frentz, G.; Olsen, J.; Avrach, W. Malignant tumours and psoriasis: Climatotherapy at the Dead Sea. Br. J. Dermatol. 1999, 141, 1088–1091. [Google Scholar] [CrossRef]
- Harari, M.; Czarnowicki, T.; Fluss, R.; Ruzicka, T.; Ingber, A. Patients with early-onset psoriasis achieve better results following Dead Sea climatotherapy. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 554–559. [Google Scholar] [CrossRef]
- Harari, M.; Novack, L.; Barth, J.; David, M.; Friger, M.; Moses, S.W. The percentage of patients achieving PASI 75 after 1 month and remission time after climatotherapy at the Dead Sea. Int. J. Dermatol. 2007, 46, 1087–1091. [Google Scholar] [CrossRef]
- Harari, M.; Sela, Y.; Ingber, A.; Vardy, D. Dead sea climatotherapy for psoriasis vulgaris: Analysis of short-term results. Glob. Dermatol. 2016, 3, 295–301. [Google Scholar] [CrossRef][Green Version]
- Harari, M.; Shani, J. Demographic evaluation of successful antipsoriatic climatotherapy at the Dead Sea (Israel) DMZ Clinic. Int. J. Dermatol. 1997, 36, 304–308. [Google Scholar] [CrossRef]
- Kushelevsky, A.P.; Harari, M.; Kudish, A.I.; Hristakieva, E.; Ingber, A.; Shani, J. Safety of solar phototherapy at the Dead Sea. J. Am. Acad. Dermatol. 1998, 38, 447–452. [Google Scholar] [CrossRef]
- Kushelevsky, A. Climatotherapy of Psoriasis and Hypertension in Elderly Patients at the Dead-Sea. Pharmacol. Res. 1996, 34, 87–91. [Google Scholar] [CrossRef]
- Nissen, J.B.; Avrach, W.W.; Hansen, E.S.; Stengaard-Pedersen, K.; Kragballe, K. Increased levels of enkephalin following natural sunlight (combined with salt water bathing at the Dead Sea) and ultraviolet A irradiation. Br. J. Dermatol. 1998, 139, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Schewach-Millet, M.; Feinstein, A.; Trau, H.; Abel, E.A.; Cox, A.J. Histologic studies in psoriatic patients treated at the Dead Sea: Comparison with photochemotherapy. J. Am. Acad. Dermatol. 1989, 20, 502–503. [Google Scholar] [CrossRef]
- Trøstrup, H.; Riis, P.T.; Heidenheim, M.; Bryld, L.E.; Jemec, G.B. Long-term evaluation of climatotherapy for psoriasis. Dermatol. Ther. 2020, 33, e13432. [Google Scholar] [CrossRef]
- Bogdanov, I.; Kircheva, K.; Miteva, L.; Tsankov, N. Quality of life in patients undergoing combined climatotherapy and phototherapy. Rev. Med. Românå 2012, 59, 251–254. [Google Scholar]
- Golusin, Z.; Jovanovic, M.; Magda, N.; Stojanovic, S.; Matic, M.; Petrovic, A. Effects of Rusanda Spa balneotherapy combined with calcipotriol on plaque psoriasis. Vojn. Pregl. 2015, 72, 1010–1017. [Google Scholar] [CrossRef]
- Peroni, A.; Gisondi, P.; Zanoni, M.; Girolomoni, G. Balneotherapy for chronic plaque psoriasis at Comano spa in Trentino, Italy. Dermatol. Ther. 2008, 21 (Suppl. 1), S31–S38. [Google Scholar] [CrossRef] [PubMed]
- Péter, I.; Jagicza, A.; Ajtay, Z.; Boncz, I.; Kiss, I.; Szendi, K.; Kustán, P.; Németh, B. Balneotherapy in Psoriasis Rehabilitation. In Vivo 2017, 31, 1163–1168. [Google Scholar] [CrossRef]
- Tabolli, S.; Calza, A.; Di Pietro, C.; Sampogna, F.; Abeni, D. Quality of Life of Psoriasis Patients before and after Balneo- or Balneophototherapy. Yonsei Med. J. 2009, 50, 215–221. [Google Scholar] [CrossRef]
- Nilsen, L.T.N.; Søyland, E.; Krogstad, A.L. Estimated ultraviolet doses to psoriasis patients during climate therapy. Photodermatol. Photoimmunol. Photomed. 2009, 25, 202–208. [Google Scholar] [CrossRef]
- Martin, R.; Henley, J.B.; Sarrazin, P.; Seité, S. Skin Microbiome in Patients with Psoriasis Before and After Balneo-therapy at the Thermal Care Center of La Roche-Posay. J. Drugs Dermatol. 2015, 14, 1400–1405. [Google Scholar]
- Pinton, J.; Friden, H.; Kettaneh-Wold, N.; Wold, S.; Dreno, B.; Richard, A.; Bieber, T. Clinical and biological effects of balneotherapy with selenium-rich spa water in patients with psoriasis vulgaris. Br. J. Dermatol. 1995, 133, 344–347. [Google Scholar] [CrossRef]
- Wang, G.; Bai, F.; Tian, Z.; Fan, L.; Lei, M. Balneotherapy with Chinese herbal medicine prolongs the remission period in patients with psoriasis vulgaris. Dermatol. Ther. 2020, 33, e13351. [Google Scholar] [CrossRef]
- Tsoureli-Nikita, E.; Menchini, G.; Ghersetich, I.; Hercogova, J. Alternative treatment of psoriasis with balneotherapy using Leopoldine spa water. J. Eur. Acad. Dermatol. Venereol. 2002, 16, 260–262. [Google Scholar] [CrossRef]
- Melandri, D.; Albano, V.M.; Venturi, M.; Flamigni, A.; Vairetti, M. Efficacy of combined liman peloid baths and heliotherapy in the treatment of psoriasis at Cervia spa, Emilia, Italy. Int. J. Biometeorol. 2020, 64, 1145–1152. [Google Scholar] [CrossRef]
- Wang, Y.; Fu, Y.; Zhang, L.; Fu, J.; Li, B.; Zhao, L.; Di, T.; Meng, Y.; Li, N.; Guo, J.; et al. Acupuncture Needling, Electroacupuncture, and Fire Needling Improve Imiquimod-Induced Psoriasis-Like Skin Lesions through Reducing Local Inflammatory Responses. Evid. Based Complement. Altern. Med. 2019, 2019, 4706865. [Google Scholar] [CrossRef]
- Chen, Z.; Zhou, D.; Wang, Y.; Lan, H.; Duan, X.; Li, B.; Zhao, J.; Li, W.; Liu, Z.; Di, T.; et al. Fire needle acupuncture or moxibustion for chronic plaque psoriasis: Study protocol for a randomized controlled trial. Trials 2019, 20, 1–11. [Google Scholar] [CrossRef]
- Koo, J.; Desai, R. Traditional Chinese medicine in dermatology. Dermatol. Ther. 2003, 16, 98–105. [Google Scholar] [CrossRef]
- Yeh, M.-L.; Ko, S.-H.; Wang, M.-H.; Chi, C.-C.; Chung, Y.-C. Acupuncture-Related Techniques for Psoriasis: A Systematic Review with Pairwise and Network Meta-Analyses of Randomized Controlled Trials. J. Altern. Complement. Med. 2017, 23, 930–940. [Google Scholar] [CrossRef]
- Xiao, S.; Li, B.; Feng, S.; Liu, C.; Zhang, G. Acupuncture combined with herbal medicine versus herbal medicine alone for plaque psoriasis: A systematic review protocol. Ann. Transl. Med. 2019, 7, 115. [Google Scholar] [CrossRef]
- Sanchez, D.P.; Sonthalia, S. Koebner Phenomenon. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Deng, J.; Lu, C.; Xiang, Y.; Deng, H.; Wen, Z.; Yao, D.; Xuan, M.; Yan, Y. Auricular acupressure as a complementary therapy for psoriasis vulgaris: Study protocol for a multicenter randomized controlled trial. Trials 2019, 20, 358. [Google Scholar] [CrossRef]
- Xing, M.; Yan, X.; Yang, S.; Li, L.; Gong, L.; Liu, H.; Xu, R.; Chen, J.; Ying, L.; Zhao, Y.; et al. Effects of moving cupping therapy for plaque psoriasis: Study protocol for a randomized multicenter clinical trial. Trials 2020, 21, 229. [Google Scholar] [CrossRef]
- Lowe, D.T. Cupping therapy: An analysis of the effects of suction on skin and the possible influence on human health. Complement. Ther. Clin. Pract. 2017, 29, 162–168. [Google Scholar] [CrossRef]
- Lee, H.Y.; Lee, H.J.; Kim, G.C.; Choi, J.H.; Hong, J.W. Plasma cupping induces VEGF expression in skin cells through nitric oxide-mediated activation of hypoxia inducible factor 1. Sci. Rep. 2019, 9, 3821. [Google Scholar] [CrossRef]
- Mehta, P.; Dhapte, V. Cupping therapy: A prudent remedy for a plethora of medical ailments. J. Tradit. Complement. Med. 2015, 5, 127–134. [Google Scholar] [CrossRef]
- Xing, M.; Ding, X.; Zhang, J.; Kuai, L.; Ru, Y.; Sun, X.; Ma, T.; Miao, X.; Liu, L.; Wang, Y.; et al. Moving cupping therapy for plaque psoriasis: A PRISMA-compliant study of 16 randomized controlled trials. Medicine 2020, 99, e22539. [Google Scholar] [CrossRef]
- Basavaraj, K.H.; Navya, M.A.; Rashmi, R. Stress and quality of life in psoriasis: An update. Int. J. Dermatol. 2011, 50, 783–792. [Google Scholar] [CrossRef]
- Shenefelt, P.D. Hypnosis in Dermatology. Arch. Dermatol. 2000, 136, 393–399. [Google Scholar] [CrossRef]
- Capella, G.L.; Finzi, A.F. Complementary therapy for psoriasis. Dermatol. Ther. 2003, 16, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.; Shapiro, J.; Michael, D.; Hodak, E.; Van-Dijk, D.; Naggan, L.; Vardy, D. Outcome of “Short-term” Dead Sea Climatotherapy for Psoriasis. Acta Derm. Venereol. 2008, 88, 90–91. [Google Scholar] [CrossRef][Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timis, T.L.; Florian, I.A.; Mitrea, D.R.; Orasan, R. Mind-Body Interventions as Alternative and Complementary Therapies for Psoriasis: A Systematic Review of the English Literature. Medicina 2021, 57, 410. https://doi.org/10.3390/medicina57050410
Timis TL, Florian IA, Mitrea DR, Orasan R. Mind-Body Interventions as Alternative and Complementary Therapies for Psoriasis: A Systematic Review of the English Literature. Medicina. 2021; 57(5):410. https://doi.org/10.3390/medicina57050410
Chicago/Turabian StyleTimis, Teodora Larisa, Ioan Alexandru Florian, Daniela Rodica Mitrea, and Remus Orasan. 2021. "Mind-Body Interventions as Alternative and Complementary Therapies for Psoriasis: A Systematic Review of the English Literature" Medicina 57, no. 5: 410. https://doi.org/10.3390/medicina57050410
APA StyleTimis, T. L., Florian, I. A., Mitrea, D. R., & Orasan, R. (2021). Mind-Body Interventions as Alternative and Complementary Therapies for Psoriasis: A Systematic Review of the English Literature. Medicina, 57(5), 410. https://doi.org/10.3390/medicina57050410







