The History of Carbon Monoxide Intoxication
Abstract
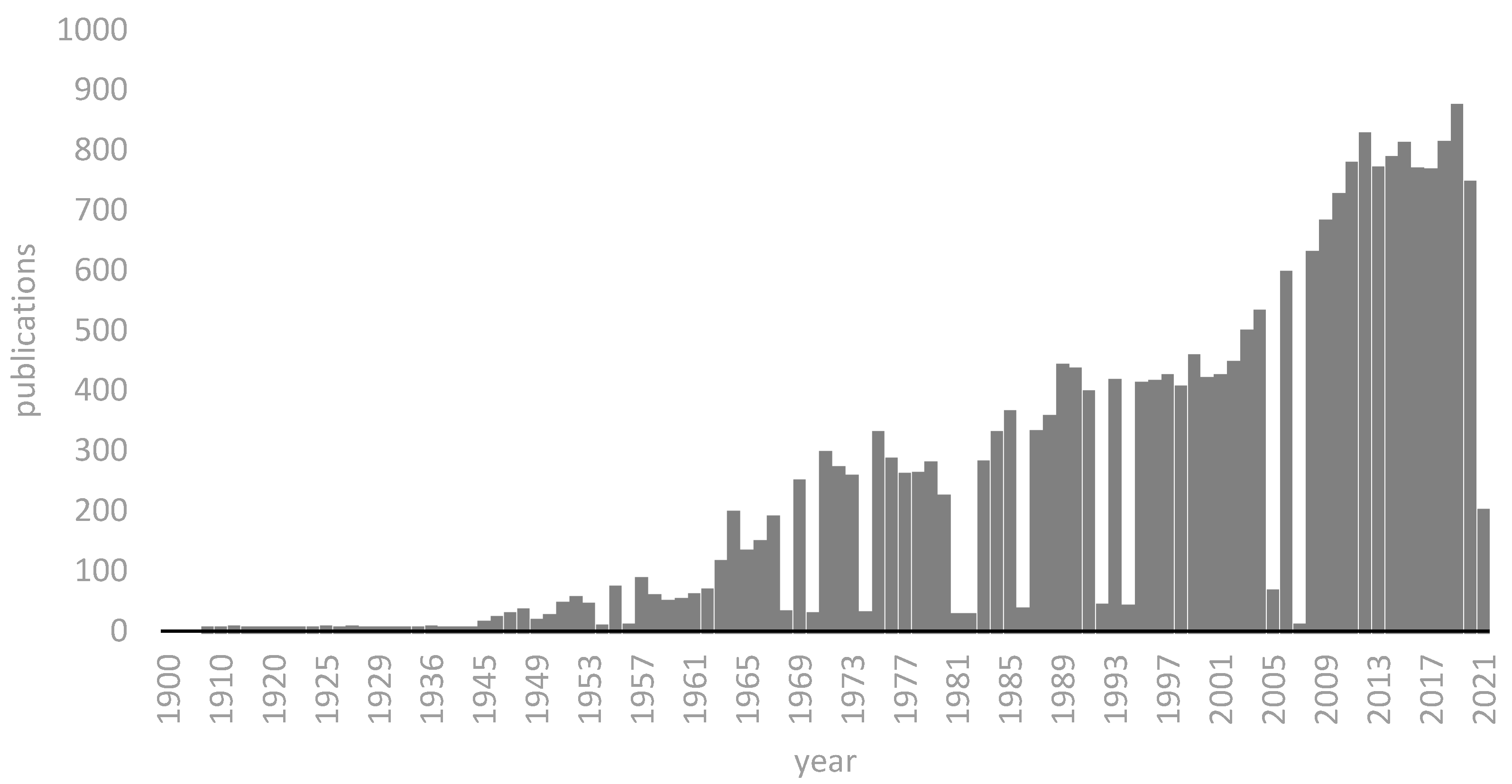
1. Introduction and Overview
2. Literature Research
3. 1900 to 1945
Twelve guinea pigs were taken, six males and six females, and placed in two cages in the gas chamber after making careful estimations of the white and red blood corpuscles and haemoglobin. Gas was then allowed to mix with the air drawn through the chamber until the mixture was of such a strength that 250/0 of the haemoglobin of the guinea-pigs was saturated with carbon monoxide.
Treatment is summed up in warmth, oxygen, artificial respiration, rest. Circulatory stimulants, such as hot coffee, are valuable. Strychnine has been found useful for stimulating the respiratory centre, but chief reliance is to be placed upon artificial respiration by Schäfer’s method [20], combined with the administration of oxygen [19].
4. 1945 to 1980
In selecting a form of therapy there are two aspects to consider: firstly, the prevention of death and, secondly, the reduction of neuropsychiatric sequelae such as those described by H. Garland and J. Pearce. Carboxyhaemoglobin should be eliminated as quickly as possible because its presence alters the dissociation curve of the remaining oxyhaemoglobin, impeding oxygen release to the tissues [21].
5. From 1980 to Present
6. History of Hyperbaric Oxygen Therapy for CO Intoxication
The chief aim of the present investigation has been to determine experimentally the causes of the symptoms produced in man by carbonicoxide, and particularly the relation of the changes in the blood to the symptoms, to the percentage of carbonic oxide breathed, and to the period during which the inhalation is continued [33].
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, H.-H.; Choi, S. Therapeutic Aspects of Carbon Monoxide in Cardiovascular Disease. Int. J. Mol. Sci. 2018, 19, 2381. [Google Scholar] [CrossRef] [PubMed]
- Barret, L.; Danel, V.; Faure, J. Carbon Monoxide Poisoning, a Diagnosis Frequently Overlooked. J. Toxicol. Clin. Toxicol. 1985, 23, 309–313. [Google Scholar] [CrossRef]
- Silver, S.; Smith, C.; Worster, A.; BEEM (Best Evidence in Emergency Medicine) Team. Should hyperbaric oxygen be used for carbon monoxide poisoning? CJEM 2006, 8, 43–46. [Google Scholar] [CrossRef]
- Llano, A.L.; Raffin, T.A.; Ilano, A.L. Management of Carbon Monoxide Poisoning. Chest 1990, 97, 165–169. [Google Scholar] [CrossRef]
- Jaffe, F.A. Pathogenicity of Carbon Monoxide. Am. J. Forensic Med. Pathol. 1997, 18, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Goldbaum, L.R.; Ramirez, R.G.; Absalon, K.B. What is the mechanism of carbon monoxide toxicity? Aviat. Space Environ. Med. 1975, 46, 1289–1291. [Google Scholar]
- Hampson, N.B. Emergency department visits for carbon monoxide poisoning in the Pacific Northwest. J. Emerg. Med. 1998, 16, 695–698. [Google Scholar] [CrossRef]
- Thom, S.R. Hyperbaric-oxygen therapy for acute carbon monoxide poisoning. N. Engl. J. Med. 2002, 347, 1105–1106. [Google Scholar] [CrossRef]
- Grieb, G.; Simons, D.; Schmitz, L.; Piatkowski, A.; Grottke, O.; Pallua, N. Glasgow Coma Scale and laboratory markers are superi-or to COHb in predicting CO intoxication severity. Burns 2011, 37, 610–615. [Google Scholar] [CrossRef]
- Garland, H.; Pearce, J. Neurological complications of carbon monoxide poisoning. Q. J. Med. 1967, 36, 445–455. [Google Scholar]
- Piatkowski, A.; Ulrich, D.; Grieb, G.C.; Pallua, N. A new tool for the early diagnosis of carbon monoxide intoxication. Inhal. Toxicol. 2009, 21, 1144–1147. [Google Scholar] [CrossRef] [PubMed]
- Schimmel, J.; George, N.; Schwarz, J.; Yousif, S.; Suner, S.; Hack, J.B. Carboxyhemoglobin Levels Induced by Cigarette Smoking Outdoors in Smokers. J. Med. Toxicol. 2018, 14, 68–73. [Google Scholar] [CrossRef]
- Buehler, J.H.; Berns, A.S.; Webster, J.R.; Addington, W.W.; Cugell, D.W. Lactic acidosis from carboxyhemoglobinemia after smoke inhalation. Ann. Intern. Med. 1975, 82, 803–805. [Google Scholar] [CrossRef] [PubMed]
- Sokal, J.A. The effect of exposure duration on the blood level of glucose, pyruvate and lactate in acute carbon monoxide in-toxication in man. J. Appl. Toxicol. 1985, 5, 395–397. [Google Scholar] [CrossRef]
- Sokal, J.A.; Kralkowska, E. The relationship between exposure duration, carboxyhemoglobin, blood glucose, pyruvate and lactate and the severity of intoxication in 39 cases of acute carbon monoxide poisoning in man. Arch. Toxicol. 1985, 57, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Wallner, C.; Moormann, E.; Lulof, P.; Drysch, M.; Lehnhardt, M.; Behr, B. Burn Care in the Greek and Roman Antiquity. Medicina 2020, 56, 657. [Google Scholar] [CrossRef] [PubMed]
- Moiemen, N.S.; Lee, K.C.; Joory, K. History of burns: The past, present and the future. Burn. Trauma 2014, 2, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Nasmith, G.G.; Graham, D.A.L. The haematology of carbon-monoxide poisoning. J. Physiol. 1906, 35, 32–52. [Google Scholar] [CrossRef]
- Gas in Military Mines: The Symptoms of Carbon Monoxide Poisoning. Hospital 1916, 60, 169.
- Schäfer, E.A. Description of a Simple and Efficient Method of Performing Artificial Respiration in the Human Subject, espe-cially in Cases of Drowning; to which is appended Instructions for the Treatment of the Apparently Drowned. Med. Chir. Trans. 1904, 87, 609–623. [Google Scholar]
- Carbon monoxide poisoning. Br. Med. J. 1970, 3, 180. [CrossRef][Green Version]
- Smith, G.; Ledingham, I.; Sharp, G.; Norman, J.; Bates, E. Treatment of coal-gas poisoning with oxygen at 2 atmospheres pressure. Lancet 1962, 279, 816–819. [Google Scholar] [CrossRef]
- Lawson, D.D.; Mcallister, R.A.; Smith, G. Treatment of acute experimental carbon-monoxide poisoning with oxygen under pressure. Lancet 1961, 1, 800–802. [Google Scholar] [CrossRef]
- Davis, N. Carbon monoxide poisoning. Br. Med. J. 1979, 2, 1584. [Google Scholar] [CrossRef]
- Smith, J.S.; Brandon, S. Morbidity from Acute Carbon Monoxide Poisoning at Three-year Follow-up. BMJ 1973, 1, 318–321. [Google Scholar] [CrossRef][Green Version]
- Ernst, A.; Zibrak, J.D. Carbon monoxide poisoning. N. Engl. J. Med. 1998, 339, 1603–1608. [Google Scholar] [CrossRef]
- Weaver, L.K.; Howe, S.; Hopkins, R.; Chan, K.J. Carboxyhemoglobin half-life in carbon monoxide-poisoned patients treated with 100% oxygen at atmospheric pressure. Chest 2000, 117, 801–808. [Google Scholar] [CrossRef]
- Weaver, L.K. Clinical practice. Carbon monoxide poisoning. N. Engl. J. Med. 2009, 360, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- Acute Respiratory Distress Syndrome Network; Brower, R.G.; Matthay, M.A.; Morris, A.; Schoenfeld, D.; Thompson, B.T.; Wheeler, A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N. Engl. J. Med. 2000, 342, 1301–1308. [Google Scholar] [PubMed]
- Park, G.Y.; Park, J.W.; Jeong, D.H.; Jeong, S.H. Prolonged airway and systemic inflammatory reactions after smoke inhalation. Chest 2003, 123, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Danesh-Sani, S.A.; Shariati-Sarabi, Z.; Feiz, M.R. Comprehensive Review of Hyperbaric Oxygen Therapy. J. Craniofacial Surg. 2012, 23, e483–e491. [Google Scholar] [CrossRef] [PubMed]
- Yoshinoya, Y.; Böcker, A.H.; Ruhl, T.; Siekmann, U.; Pallua, N.; Beier, J.P.; Kim, B.-S. The Effect of Hyperbaric Oxygen Therapy on Human Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2020, 146, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Haldane, J. The Action of Carbonic Oxide on Man. J. Physiol. 1895, 18, 430–462. [Google Scholar] [CrossRef] [PubMed]
- Churchill-Davidson, I. Therapeutic uses of hyperbaric oxygen. Ann. R. Coll. Surg. Engl. 1966, 39, 164–168. [Google Scholar]
- Buckley, N.A.; Juurlink, D.N.; Isbister, G.; Bennett, M.H.; Lavonas, E.J. Hyperbaric oxygen for carbon monoxide poisoning. Cochrane Database Syst. Rev. 2011, CD002041. [Google Scholar] [CrossRef] [PubMed]
- Raphael, J.C.; Elkharrat, D.; Jars-Guincestre, M.C.; Chastang, C.; Chasles, V.; Vercken, J.B.; Gajdos, P. Trial of normobaric and hyper-baric oxygen for acute carbon monoxide intoxication. Lancet 1989, 2, 414–419. [Google Scholar] [CrossRef]
- Scheinkestel, C.D.; Myles, P.S.; Cooper, D.J.; Millar, I.L.; Tuxen, D.V.; Bailey, M.; Jones, K. Hyperbaric or normobaric oxygen for acute carbon monoxide poisoning: A randomised controlled clinical trial. Med. J. Aust. 1999, 170, 203–210. [Google Scholar] [CrossRef]
- Annane, D.; Chadda, K.; Gajdos, P.; Jars-Guincestre, M.-C.; Chevret, S.; Raphael, J.-C. Hyperbaric oxygen therapy for acute domestic carbon monoxide poisoning: Two randomized controlled trials. Intensiv. Care Med. 2010, 37, 486–492. [Google Scholar] [CrossRef]
- Sinkovic, A.; Smolle-Juettner, F.M.; Krunic, B.; Marinšekz, M. Severe Carbon Monoxide Poisoning Treated by Hyperbaric Oxygen Therapy—A Case Report. Inhal. Toxicol. 2006, 18, 211–214. [Google Scholar] [CrossRef]
- Stoller, K.P. Hyperbaric oxygen and carbon monoxide poisoning: A critical review. Neurol. Res. 2007, 29, 146–155. [Google Scholar] [CrossRef]
- Juurlink, D.N.; Buckley, N.; Stanbrook, M.B.; Isbister, G.; Bennett, M.H.; McGuigan, M. Hyperbaric oxygen for carbon monoxide poisoning. Cochrane Database Syst. Rev. 2005, 24, 75–92. [Google Scholar] [CrossRef]
- Weaver, L.K.; Hopkins, R.O.; Chan, K.J.; Churchill, S.; Elliott, C.G.; Clemmer, T.P.; Orme, J.F.; Thomas, F.O.; Morris, A.H. Hyperbaric oxygen for acute carbon monoxide poisoning. N. Engl. J. Med. 2002, 347, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- U.S. CDC (The Centers for Disease Control and Prevention): Clinical Guidance for Carbon Monoxide Poisoning|Natural Disasters and Severe Weather. 2020. Available online: https://www.cdc.gov/disasters/co_guidance.html (accessed on 11 March 2021).
- Mutluoglu, M.; Metin, S.; Arziman, I.; Uzun, G.; Yildiz, S. The use of hyperbaric oxygen therapy for carbon monox-ide poisoning in Europe. Undersea Hyperb Med. 2016, 43, 49–56. [Google Scholar] [PubMed]
- U. K. Department of Health and the NHS England: Carbon Monoxide Poisoning. GOV.UK. Available online: https://www.gov.uk/government/publications/carbon-monoxide-poisoning (accessed on 11 March 2021).
- DIVI (German Interdisciplinary Association for Intensive Care and Emergency Medicine): AWMF. Available online: https://www.awmf.org/leitlinien/detail/anmeldung/1/ll/040-012.html (accessed on 11 March 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Megas, I.-F.; Beier, J.P.; Grieb, G. The History of Carbon Monoxide Intoxication. Medicina 2021, 57, 400. https://doi.org/10.3390/medicina57050400
Megas I-F, Beier JP, Grieb G. The History of Carbon Monoxide Intoxication. Medicina. 2021; 57(5):400. https://doi.org/10.3390/medicina57050400
Chicago/Turabian StyleMegas, Ioannis-Fivos, Justus P. Beier, and Gerrit Grieb. 2021. "The History of Carbon Monoxide Intoxication" Medicina 57, no. 5: 400. https://doi.org/10.3390/medicina57050400
APA StyleMegas, I.-F., Beier, J. P., & Grieb, G. (2021). The History of Carbon Monoxide Intoxication. Medicina, 57(5), 400. https://doi.org/10.3390/medicina57050400







