Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health
Abstract
1. Introduction
2. Vegan Nutrition
2.1. Macronutrients
2.2. Micronutrients
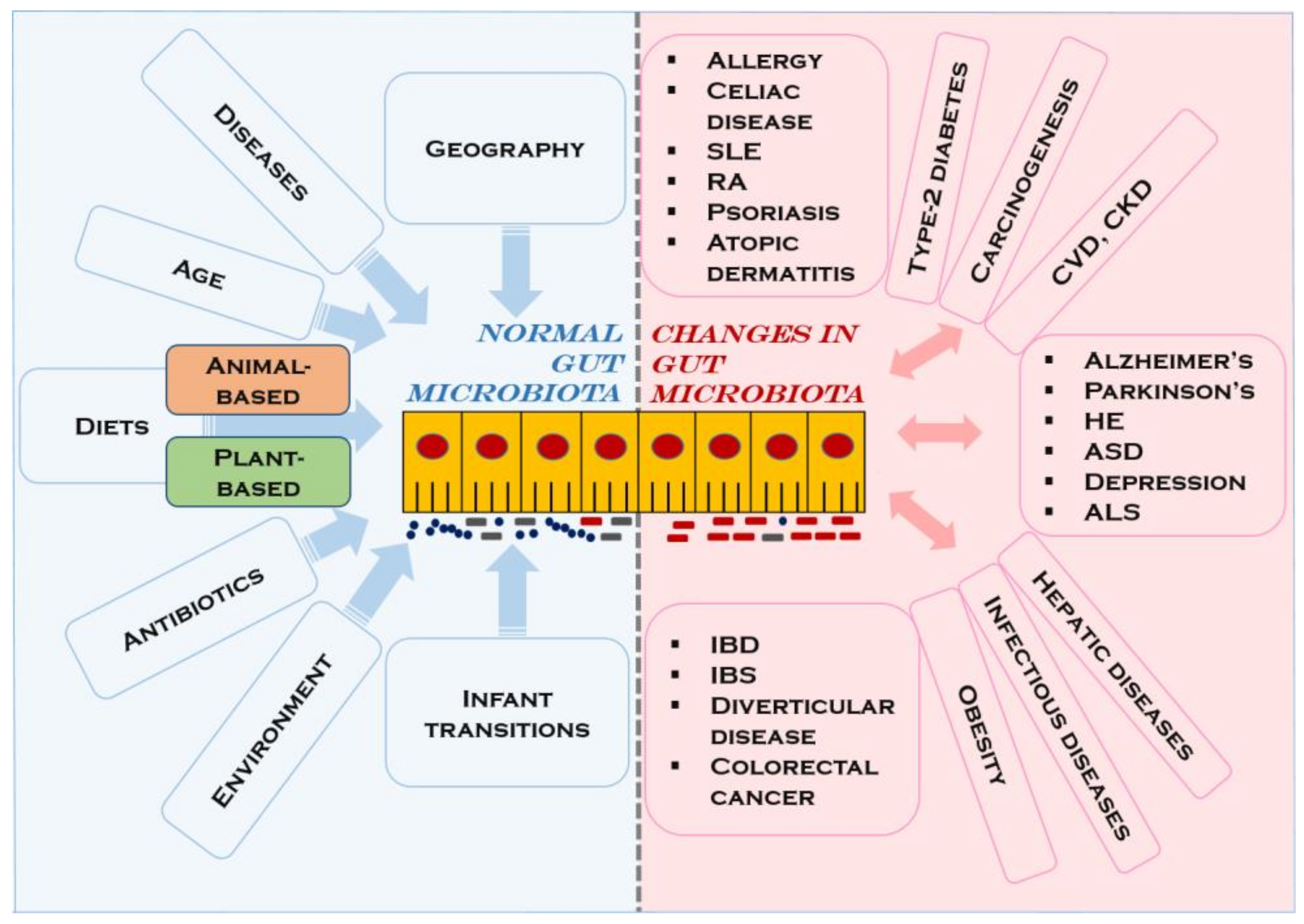
3. Influence of Vegan Diets on the Human Gut Microbiota
3.1. Gut Microbiota Composition and Functional Aspects
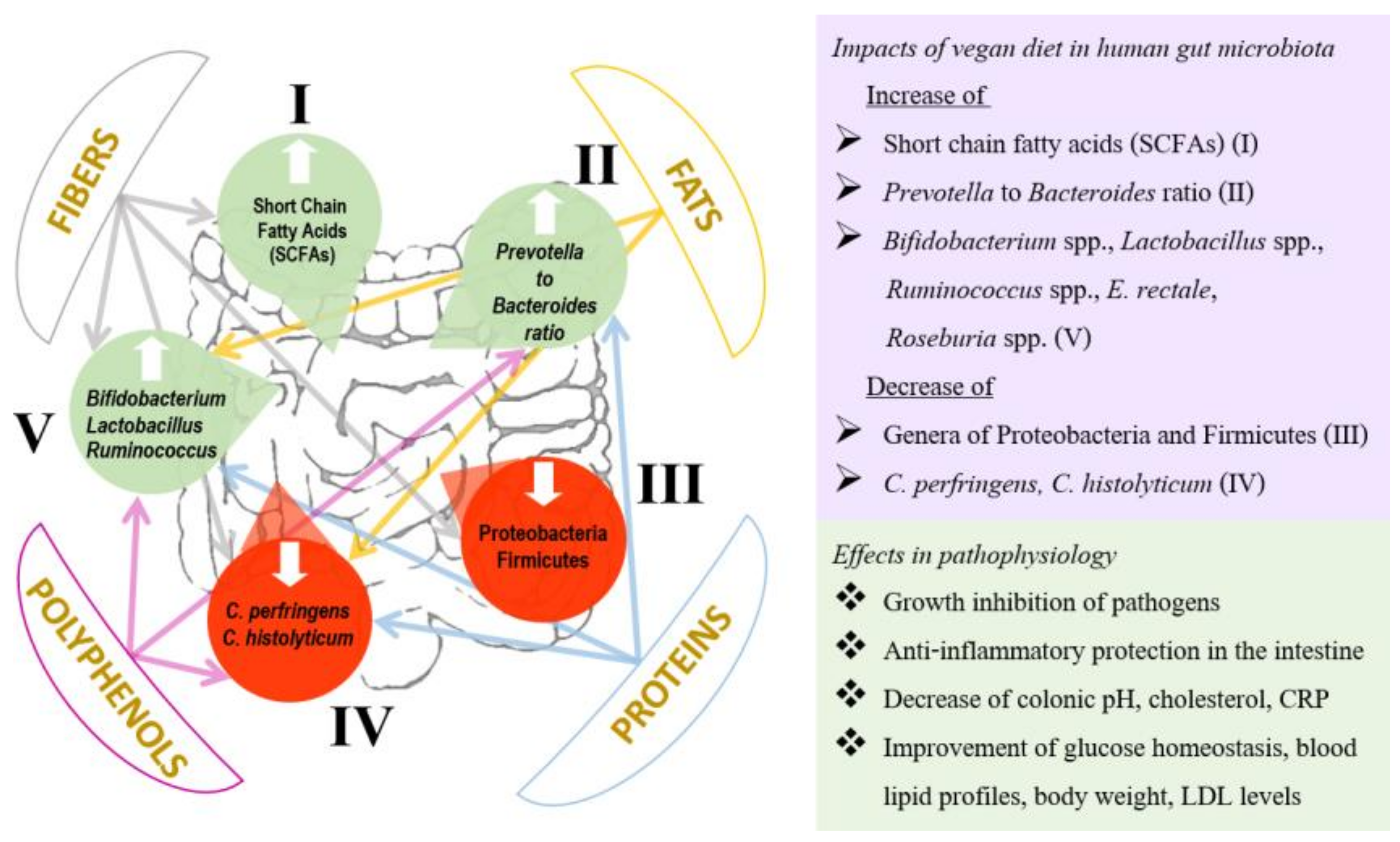
3.2. Impact of Vegan Food Components on the Human Gut Microbiota
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Craig, W.J. Health effects of vegan diets. Am. J. Clin. Nutr. 2009, 89, 1627–1633. [Google Scholar] [CrossRef] [PubMed]
- Medawar, E.; Huhn, S.; Villringer, A.; Witte, A.V. The effects of plant-based diets on the body and the brain: A systematic review. Transl. Psychiatr. 2019, 9, 226. [Google Scholar] [CrossRef]
- Cramer, H.; Kessler, C.; Sundberg, T.; Leach, M.; Schumann, D.; Adams, J.; Lauche, R. Characteristics of Americans Choosing Vegetarian and Vegan Diets for Health Reasons. J. Nutr. Educ. Behav. 2017, 49, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Lemale, J.; Mas, E.; Jung, C.; Bellaiche, M.; Tounian, P.; French-speaking Pediatric Hepatology, Gastroenterology and Nutrition Group (GFHGNP). Vegan diet in children and adolescents. Recommendations from the French-speaking Pediatric Hepatology, Gastroenterology and Nutrition Group (GFHGNP). Arch. Pediatr. 2019, 26, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Vergeer, L.; Vanderlee, L.; White, C.M.; Rynard, V.L.; Hammond, D. Vegetarianism and other eating practices among youth and young adults in major Canadian cities. Public Health Nutr. 2019, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Reginster, J.Y. The effects of calorie restriction, intermittent fasting and vegetarian diets on bone health. Aging Clin. Exp. Res. 2019, 31, 753. [Google Scholar] [CrossRef]
- Zick, S.M.; Snyder, D.; Abrams, D.I. Pros and Cons of dietary strategies popular among cancer patients. Oncology 2018, 32, 542–547. [Google Scholar]
- Le, L.T.; Sabaté, J. Beyond Meatless, the Health Effects of Vegan Diets: Findings from the Adventist Cohorts. Nutrients 2014, 6, 2131–2147. [Google Scholar] [CrossRef]
- Sebastiani, G.; Herranz Barbero, A.; Borrás-Novell, C.; Alsina Casanova, M.; Aldecoa-Bilbao, V.; Andreu-Fernández, V.; Pascual Tutusaus, M.; Ferrero Martínez, S.; Gómez Roig, M.D.; García-Algar, O. The Effects of Vegetarian and Vegan Diet during Pregnancy on the Health of Mothers and Offspring. Nutrients 2019, 11, 557. [Google Scholar] [CrossRef]
- Hawkins, I.; Mangels, A.R.; Goldman, R.; Wood, R.J. Dietetics Program Directors in the United States Support Teaching Vegetarian and Vegan Nutrition and Half Connect Vegetarian and Vegan Diets to Environmental Impact. Front. Nutr. 2019, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Patelakis, E.; Barbosa, C.L.; Haftenberger, M.; Brettschneider, A.K.; Lehmann, F.; Heide, K.; Frank, M.; Perlitz, H.; Richter, A.; Mensink, G.B.M. Prevalence of vegetarian diet among children and adolescents in Germany. Results from EsKiMo II. Ernahrungs Umschau 2019, 66, 85–91. [Google Scholar]
- Richter, M.; Boeing, H.; Grunewald-Funk, D.; Heseker, H.; Kroke, A.; Leschik-Bonnet, E.; Oberritter, H.; Strohm, D.; Watzl, B. for the German Nutrition Society (DGE). Vegan diet. Position of the German Nutrition Society (DGE). Ernahrungs Umschau 2016, 63, 92–102. [Google Scholar]
- Orlich, M.J.; Singh, P.N.; Sabaté, J.; Jaceldo-Siegl, K.; Knutsen, S.; Beeson, W.L.; Fraser, G.E. Vegetarian Dietary Patterns and Mortality in Adventist Health Study 2. JAMA Intern. Med. 2013, 173, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Shen, X.; Tang, W.; Zhao, Y.; Wu, F.; Zhu, Z.; Tang, Q.; Cai, W. Prevalence of vegetarians and vegetarian’s health dietary behavior survey in Shanghai. Wei Sheng Yan Jiu 2015, 44, 237–241. [Google Scholar]
- Ponzio, E.; Mazzarini, G.; Gasperi, G.; Bottoni, M.C.; Vallorani, S. The vegetarian habit in Italy: Prevalence and characteristics of consumers. Ecol. Food Nutr. 2015, 54, 370–379. [Google Scholar] [CrossRef]
- Gallego-Narbón, A.; Zapatera, B.; Vaquero, M.P. Physiological and Dietary Determinants of Iron Status in Spanish Vegetarians. Nutrients 2019, 11, 1734. [Google Scholar] [CrossRef]
- Larsson, C.L.; Klock, K.S.; Astrom, A.N.; Haugejorden, O.; Johansson, G. Food habits of young Swedish and Norwegian vegetarians and omnivores. Public Health Nutr. 2001, 4, 1005–1014. [Google Scholar] [CrossRef]
- Elorinne, A.L.; Alfthan, G.; Erlund, I.; Kivimäki, H.; Paju, A.; Salminen, I.; Turpeinen, U.; Voutilainen, S.; Laakso, J. Food and Nutrient Intake and Nutritional Status of Finnish Vegans and Non-Vegetarians. PLoS ONE 2016, 11, e0148235. [Google Scholar] [CrossRef]
- Kirse, A.; Karklina, D. Quality evaluation of new vegetarian bean spreads. Eur. Sci. J. 2013, 4, 453–457. [Google Scholar]
- Chandra-Hioe, M.V.; Lee, C.; Arcot, J. What is the cobalamin status among vegetarians and vegans in Australia? Int. J. Food Sci. Nutr. 2019, 70, 875–886. [Google Scholar] [CrossRef]
- Jaacks, L.M.; Kapoor, D.; Singh, K.; Narayan, K.M.; Ali, M.K.; Kadir, M.M.; Mohan, V.; Tandon, N.; Prabhakaran, D. Vegetarianism and cardiometabolic disease risk factors: Differences between South Asian and US adults. Nutrition 2016, 32, 975–984. [Google Scholar] [CrossRef]
- Worsley, A.; Skrzypiec, G. Teenage vegetarianism: Prevalence, social and cognitive contexts. Appetite 1998, 30, 151–170. [Google Scholar] [CrossRef] [PubMed]
- Allès, B.; Baudry, J.; Méjean, C.; Touvier, M.; Péneau, S.; Hercberg, S.; Kesse-Guyot, E. Comparison of Sociodemographic and Nutritional Characteristics between Self-Reported Vegetarians, Vegans, and Meat-Eaters from the NutriNet-Santé Study. Nutrients 2017, 9, 1023. [Google Scholar] [CrossRef]
- Woo, K.S.; Kwok, T.C.; Celermajer, D.S. Vegan Diet, Subnormal Vitamin B-12 Status and Cardiovascular Health. Nutrients 2014, 6, 3259–3273. [Google Scholar] [CrossRef] [PubMed]
- Glick-Bauer, M.; Yeh, M.-C. The Health Advantage of a Vegan Diet: Exploring the Gut Microbiota Connection. Nutrients 2014, 6, 4822–4838. [Google Scholar] [CrossRef] [PubMed]
- Tomova, A.; Bukovsly, I.; Rembert, E.; Yonas, W.; Alwarith, J.; Barnard, N.D.; Kahleova, H. The effects of vegetarian and vegan diets on gut microbiota. Front. Nutr. 2019, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.W.; Yi, C.H.; Liu, T.T.; Hung, J.S.; Lin, C.L.; Lin, S.Z.; Chen, C.L. Impact of vegan diets on gut microbiota: An update on the clinical implications. Tzu Chi Med. J. 2018, 30, 200–203. [Google Scholar]
- Appleby, P.N.; Allen, N.E.; Key, T.J. Diet, vegetarianism, and cataract risk. Am. J. Clin. Nutr. 2011, 93, 1128–1135. [Google Scholar] [CrossRef]
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef]
- Wilson, A.K.; Ball, M.J. Nutrient intake and iron status of Australian male vegetarians. Eur. J. Clin. Nutr. 1999, 53, 189–194. [Google Scholar] [CrossRef]
- Mangels, A.R. Bone nutrients for vegetarians. Am. J. Clin. Nutr. 2014, 100, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Appleby, P.N.; Key, T.J. The long-term health of vegetarians and vegans. Proc. Nutr. Soc. 2016, 75, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Heiss, S.; Coffino, J.A.; Hormes, J.M. Eating and health behaviors in vegans compared to omnivores: Dispelling common myths. Appetite 2017, 118, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Costa, I.; Gill, P.R.; Morda, R.; Ali, L. “More than a diet”: A qualitative investigation of young vegan women’s relationship to food. Appetite 2019, 143, 104418. [Google Scholar] [CrossRef]
- Rogerson, D. Vegan diets: Practical advice for athletes and exercisers. J. Int. Soc. Sports Nutr. 2017, 14, 36. [Google Scholar] [CrossRef]
- Fujioka, N.; Fritz, V.; Upadhyaya, P.; Kassie, F.; Hecht, S. Research on cruciferous vegetables, indole-3-carbinol, and cancer prevention: A tribute to Lee, W. Wattenberg. Mol. Nutr. Food Res. 2016, 60, 1228–1238. [Google Scholar] [CrossRef]
- Lee, A.; Beaubernard, L.; Lamothe, V.; Bennetau-Pelissero, C. New Evaluation of Isoflavone Exposure in the French Population. Nutrients 2019, 11, 2308. [Google Scholar] [CrossRef]
- Rajaram, S.; Jones, J.; Lee, G.J. Plant-based dietary patterns, plant foods, and age-related cognitive decline. Adv. Nutr. 2019, 10, S422–S436. [Google Scholar] [CrossRef]
- Verneau, F.; La Barbera, F.; Furno, M. The Role of Health Information in Consumers’ Willingness to Pay for Canned Crushed Tomatoes Enriched with Lycopene. Nutrients 2019, 11, 2173. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and Prebiotics: Mechanisms and Health Benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Miqdady, M.; Al Mistarihi, J.; Azaz, A.; Rawat, D. Prebiotics in the infant microbiome: The past, present and future. Pediatr. Gastroenterol. Hepatol. Nutr. 2020, 23, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.L.; Erickson, J.M.; Lloyd, B.B.; Slavin, J.L. Health effects and sources of prebiotic dietary fiber. Curr. Dev. Nutr. 2018, 2, nzy005. [Google Scholar] [CrossRef] [PubMed]
- Desmond, M.A.; Sobiecki, J.; Fewtrell, M.; Wells, J.C.K. Plant-based diets for children as a means of improving adult cardiometabolic health. Nutr. Rev. 2018, 76, 260–273. [Google Scholar] [CrossRef]
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian diets. J. Acad. Nutr. Diet. 2016, 116, 1970–1980. [Google Scholar] [CrossRef] [PubMed]
- Satija, A.; Bhupathiraju, S.; Spiegelman, D.; Chiuve, S.; Manson, J.; Willett, W.; Rexrode, K.; Rimm, E.; Hu, F. Healthful and unhealthful plant-based diets and the risk of coronary heart disease in US adults. J. Am. Coll. Cardiol. 2017, 70, 411–422. [Google Scholar] [CrossRef]
- Russo, G.L. Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochem. Pharmacol. 2009, 15, 937–946. [Google Scholar] [CrossRef]
- Thumann, T.A.; Pferschy-Wenzig, E.-M.; Moissl-Eichinger, C. The role of gut microbiota for the activity of medicinal plants traditionally used in the European Union for gastrointestinal disorders. J. Ethnopharmacol. 2019, 245, 112153. [Google Scholar] [CrossRef]
- Schmidt, J.A.; Rinaldi, S.; Ferrari, P.; Carayol, M.; Achaintre, D.; Scalbert, A.; Cross, A.J.; Gunter, M.J.; Fensom, G.K.; Appleby, P.N.; et al. Metabolic profiles of male meat eaters, fish eaters, vegetarians, and vegans from the EPIC-Oxford cohort. Am. J. Clin. Nutr. 2015, 102, 1518–1526. [Google Scholar] [CrossRef]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Papa, M.; Pellegrini, N.; Sbarbati, R.; Scarino, M.L.; Siani, V.; et al. Position paper on vegetarians diets from the working group of the Italian Society of Human Nutrition. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1037–1052. [Google Scholar] [CrossRef]
- Afshin, A.; Micha, R.; Khatibzadeh, S.; Mozaffarian, D. Consumption of nuts and legumes and risk of ischemic heart disease, stroke and diabetes: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 278–288. [Google Scholar] [CrossRef]
- Olza, J.; Aranceta-Bartrina, J.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Gil, Á. Reported Dietary Intake and Food Sources of Zinc, Selenium, and Vitamins A, E and C in the Spanish Population: Findings from the ANIBES Study. Nutrients 2017, 9, 697. [Google Scholar] [CrossRef]
- Irimie, A.I.; Braicu, C.; Pasca, S.; Magdo, L.; Gulei, D.; Cojocneanu, R.; Ciocan, C.; Olariu, A.; Coza, O.; Berindan-Neagoe, I. Role of Key Micronutrients from Nutrigenetic and Nutrigenomic Perspectives in Cancer Prevention. Medicina 2019, 55, 283. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.C.; La Ferrera, G.M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef] [PubMed]
- Tong, T.Y.N.; Key, T.J.; Gaitskell, K.; Green, T.J.; Guo, W.; Sanders, T.A.; Bradbury, K.E. Hematological parameters and prevalence of anemia in white and British Indian vegetarians and nonvegetarians in the UK Biobank. Am. J. Clin. Nutr. 2019, 110, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Wang, B.; Cui, X.; Tang, Q.; Cai, W.; Shen, X. Young adult vegetarians in Shanghai have comparable bone health to omnivores despite lower serum 25(OH) vitamin D in vegans: A cross-sectional study. Asia Pasific J. Clin. Nutr. 2019, 28, 383–388. [Google Scholar]
- Menal-Puey, S.; Martínez-Biarge, M.; Marques-Lopes, I. Developing a Food Exchange System for Meal Planning in Vegan Children and Adolescents. Nutrients 2019, 11, 43. [Google Scholar] [CrossRef]
- Yeliosof, O.; Silverman, L.A. Veganism as a cause of iodine deficient hypothyroidism. J. Pediatr. Endocrinol. Metab. 2018, 31, 91–94. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Tanprasertsuk, J.; Kris-Etherton, P.; Weaver, C.M.; Johnson, E.J. Perspective: The role of beverages as a source of nutrients and phytonutrients. Adv. Nutr. 2019, nmz115. [Google Scholar] [CrossRef]
- Weaver, C.M.; Proulx, W.R.; Heaney, R. Choices for achieving adequate dietary calcium with a vegetarian diet. Am. J. Clin. Nutr. 1999, 70, 543–548. [Google Scholar] [CrossRef]
- Haider, L.M.; Schwingshackl, L.; Hoffmann, G.; Ekmekcioglu, C. The effect of vegetarian diets on iron status in adults: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1359–1374. [Google Scholar] [CrossRef] [PubMed]
- Abdelhaleim, A.; Amer, A.Y.; Abdo Soliman, J.S. Association of zinc deficiency with iron deficiency anemia and its symptoms: Results from a case-control study. Cureus 2019, 11, e3811. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Rajilic-Stojanovic, M.; de Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047. [Google Scholar] [CrossRef]
- Graf, D.; Di Cagno, R.; Fak, F.; Flint, H.J.; Nyman, M.; Saarela, M.; Watzl, B. Contribution of diet to the composition of the human gut microbiota. Microb. Ecol. Health Dis. 2015, 26, 26164. [Google Scholar] [CrossRef]
- Klement, R.J.; Pazienza, V. Impact of Different Types of Diet on Gut Microbiota Profiles and Cancer Prevention and Treatment. Medicina 2019, 55, 84. [Google Scholar] [CrossRef]
- Milanovic, V.; Osimani, A.; Cardinali, F.; Litta-Mulondo, A.; Vignaroli, C.; Citterio, B.; Mangiaterra, G.; Aquilanti, L.; Garofalo, C.; Biavasco, F.; et al. Erythromycin-resistant lactic acid bacteria in the healthy gut of vegans, ovo-lacto vegetarians and omnivores. PLoS ONE 2019, 14, e0220549. [Google Scholar] [CrossRef]
- Simpson, H.L.; Campbell, B.J. Review article: Dietary fibre-microbiota interactions. Alimentary Pharmacol. Ther. 2015, 42, 158–179. [Google Scholar] [CrossRef]
- Karl, J.P.; Margolis, L.M.; Madslien, E.H.; Murphy, N.E.; Castellani, J.W.; Gundersen, Y.; Hoke, A.V.; Levangie, M.W.; Kumar, R.; Chakraborty, N.; et al. Changes in intestinal microbiota composition and metabolism coincide with increased intestinal permeability in young adults under prolonged physiological stress. Am. J. Physiol. Gastrointest. Liv. Physiol. 2017, 312, G559–G571. [Google Scholar] [CrossRef]
- Li, Z.; Quan, G.; Jiang, X.; Yang, Y.; Ding, X.; Zhang, D.; Wang, X.; Hardwidge, P.R.; Ren, W.; Zhu, G. Effects of metabolites derived from gut microbiota and hosts on pathogens. Front. Cell. Infect. Microbiol. 2018, 8, 314. [Google Scholar] [CrossRef]
- Cani, P.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Lyte, J.M.; Gabler, N.K.; Hollis, J.H. Postprandial serum endotoxin in healthy humans is modulated by dietary fat in a randomized, controlled, cross-over study. Lipids Health Disease 2016, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Candido, T.L.N.; Bressan, J.; Alfenas, R. Dysbiosis and metabolic endotoxemia induced by high-fat diet. Nutr. Hosp. 2018, 35, 1432–1440. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Q.; Zhang, A.H.; Miao, J.H.; Sun, H.; Yan, G.L.; Wu, F.F.; Wang, X.J. Gut microbiota as important modulator of metabolism in health and disease. RSC Adv. 2018, 8, 42380–42389. [Google Scholar] [CrossRef]
- Wang, H.; Wei, C.X.; Min, L.; Zhu, L.Y. Good or bad: Gut bacteria in human health and diseases. Biotechnol. Biotechnol. Equip. 2018, 32, 1075–1080. [Google Scholar] [CrossRef]
- Ilhan, N. Gut microbiota and metabolism. Int. J. Med. Biochem. 2018, 1, 115–128. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Kho, Z.; Lai, S.K. The human gut microbiome—A potential controller of wellness and disease. Front. Microbiol. 2018, 9, 1835. [Google Scholar] [CrossRef]
- De Angelis, M.; Garruti, G.; Minervini, F.; Bonfrate, L.; Portincasa, P.; Gobbetti, M. The food-gut human axis: The effects of diet on gut microbiota and metabolome. Curr. Med. Chem. 2019, 26, 3567–3583. [Google Scholar] [CrossRef]
- Heijtz, R.D.; Wang, S.; Anuar, F.; Qian, Y.; Bjorkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef]
- Derovs, A.; Laivacuma, S.; Krumina, A. Targeting Microbiota: What Do We Know about It at Present? Medicina 2019, 55, 459. [Google Scholar] [CrossRef] [PubMed]
- Attaye, I.; Pinto-Sietsma, S.J.; Herrema, H.; Nieuwdorp, M. A crucial role for diet in the relationship between gut microbiota and cardiometabolic disease. Ann. Rev. Med. 2020, 71, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Kitai, T.; Hazen, S.L. Gut microbiota in cardiovascular health and disease. Circ. Res. 2017, 120, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- McBurney, M.I.; Davis, C.; Fraser, C.M.; Schneeman, B.O.; Huttenhower, C.; Verbeke, K.; Walter, J.; Latulippe, M.E. Establishing what constitutes a healthy human gut microbiome: State of the science, regulatory considerations, and future directions. J. Nutr. 2019, 149, 1882–1895. [Google Scholar] [CrossRef] [PubMed]
- Li, B.-Y.; Xu, X.-Y.; Gan, R.-Y.; Sun, Q.-C.; Meng, J.-M.; Shang, A.; Mao, Q.-Q.; Li, H.-B. Targeting Gut Microbiota for the Prevention and Management of Diabetes Mellitus by Dietary Natural Products. Foods 2019, 8, 440. [Google Scholar] [CrossRef]
- Ramezani, A.; Raj, D.S. The gut microbiome, kidney disease, and targeted interventions. J. Am. Soc. Nephrol. 2014, 25, 657–670. [Google Scholar] [CrossRef]
- Singh, R.K.; Chang, H.-W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Frahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 73. [Google Scholar] [CrossRef]
- Yarandi, S.S.; Peterson, D.A.; Treisman, G.J.; Moran, T.H.; Pasricha, P.J. Modulatory effects of gut microbiota on the central nervous system: How gut could play a role in neuropsychiatric health and diseases. J. Neurogastroenterol. Motil. 2016, 22, 201–212. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, S.; Varma, Y.; Fischbach, M.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Just, S.; Mondot, S.; Ecker, J.; Wegner, K.; Rath, E.; Gau, L.; Streidl, T.; Hery-Arnaud, G.; Schmidt, S.; Lesker, T.B.; et al. The gut microbiota drives the impact of bile acids and fat source in diet on mouse metabolism. Microbiome 2018, 6, 134. [Google Scholar] [CrossRef]
- Ramirez-Perez, O.; Cruz-Ramon, V.; Chinchilla-Lopez, P.; Mendez-Sanchez, N. The role of gut microbiota in bile acid metabolism. Ann. Hepatol. 2017, 16, S21–S26. [Google Scholar] [CrossRef] [PubMed]
- Losasso, C.; Di Cesare, A.; Mastrorilli, E.; Patuzzi, I.; Cibin, V.; Eckert, E.M.; Fontaneto, D.; Vanzo, A.; Ricci, A.; Corno, G. Assessing antimicrobial resistance gene load in vegan, vegetarian and omnivore human gut microbiota. Int. J. Antimicrob. Agents 2018, 52, 702–705. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, M.F.; Blaedel, T.; Bendtsen, L.Q.; Lorenzen, J.K.; Holm, J.B.; Kiilerich, P.; Roager, H.M.; Kristiansen, K.; Larsen, L.H.; Astrup, A. Prevotella-to Bacteroides ratio predicts body weight and fat loss success on 24-week diets varying in macronutrient composition and dietary fiber: Results from a post-hoc analysis. Int. J. Obes. 2019, 43, 149–157. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef]
- Wong, J.M.W. Gut microbiota and cardiometabolic outcomes: Influence of dietary patterns and their associated components. Am. J. Clin. Nutr. 2014, 100, 369S–377S. [Google Scholar] [CrossRef]
- Marietta, E.V.; Murray, J.A.; Luckey, D.H.; Jeraldo, P.R.; Lamba, A.; Patel, R.; Luthra, H.S.; Mangalam, A.; Taneja, V. Human gut-derived Prevotella histicola suppresses inflammatory arthritis in humanized mice. Arthritis Rheumatol. 2016, 68, 2878–2888. [Google Scholar] [CrossRef]
- Mangalam, A.; Murray, J. Microbial monotherapy with Prevotella histicola for patients with multiple sclerosis. Expert Rev. Neurother. 2019, 19, 45–53. [Google Scholar] [CrossRef]
- Wexler, H.M. Bacteroides: The good, the bad, and the nitty-gritty. Clin. Microb. Rev. 2007, 20, 593–621. [Google Scholar] [CrossRef]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef]
- Zhou, K. Strategies to promote abundance of Akkermansia muciniphila, an emerging probiotics in the gut, evidence from dietary intervention studies. J. Funct. Foods 2017, 33, 194–201. [Google Scholar] [CrossRef]
- Plovier, H.; Everard, A.; Druart, C.; Depommier, C.; Van Hul, M.; Geurts, L.; Chillux, J.; Ottman, N.; Duparc, T.; Lichtenstein, N.; et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 2017, 23, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Suriano, F.; Bindels, L.B.; Verspreet, J.; Courtin, C.M.; Verbeke, K.; Cani, P.D.; Neyrinck, A.M.; Delzenne, N.M. Fat binding capacity and modulation of the gut microbiota both determine the effect of wheat bran fractions on adiposity. Sci. Rep. 2017, 7, 5621. [Google Scholar] [CrossRef] [PubMed]
- Suriano, F.; Neyrick, A.M.; Verspreet, J.; Olivares, M.; Leclercq, S.; Van de Wiele, T.; Courtin, C.M.; Cani, P.D.; Bindels, L.B.; Delzenne, N.M. Particle size determines the anti-inflammatory effect of wheat bran in a model of fructose over-consumption: Implication of the gut microbiota. J. Funct. Foods 2018, 41, 155–162. [Google Scholar] [CrossRef]
- Daliri, E.B.M.; Wei, S.; Oh, D.H.; Lee, B.H. The human microbiome and metabolomics: Current concepts and applications. Crit. Rev. Food Sci. Nutr. 2017, 57, 3565–3576. [Google Scholar] [CrossRef]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases Bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef]
- Yamakoshi, J.; Tokutake, S.; Kikuchi, M.; Kubota, Y.; Konishi, H.; Mitsuoka, T. Effect of proanthocyanidin-rich extract from grape seeds on human fecal flora and fecal odor. Microb. Ecol. Health Dis. 2001, 13, 25–31. [Google Scholar]
- Queipo-Ortuno, M.I.; Boto-Ordonez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Diaz, F.C.; Andres-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and biochemical biomarkers. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef]
- Gluba-Brzózka, A.; Franczyk, B.; Rysz, J. Vegetarian Diet in Chronic Kidney Disease—A Friend or Foe. Nutrients 2017, 9, 374. [Google Scholar] [CrossRef]
- Koeth, R.; Wang, Z.; Levison, B.; Buffa, J.; Org, E.; Sheehy, B.; Britt, E.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- Molan, A.-L.; Liu, Z.; Plimmer, G. Evaluation of the effect of blackcurrant products on gut microbiota and on markers of risk for colon cancer in humans. Phytother. Res. 2014, 28, 416–422. [Google Scholar] [CrossRef] [PubMed]
| Bacteroidetes | Firmicutes | Actinobacteria |
|---|---|---|
| Bacteroides | Clostridium | Bifidobacterium |
| Prevotella | Faecalibacterium | |
| Enterococcus | Verrucomicrobia | |
| Proteobacteria | Streptococcus | Akkermansia |
| Escherichia | Roseburia | |
| Shigella | Lactobacillus | Fusobacteria |
| Bacillus | Fusobacterium | |
| Eubacterium | ||
| Ruminococcus |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakkas, H.; Bozidis, P.; Touzios, C.; Kolios, D.; Athanasiou, G.; Athanasopoulou, E.; Gerou, I.; Gartzonika, C. Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health. Medicina 2020, 56, 88. https://doi.org/10.3390/medicina56020088
Sakkas H, Bozidis P, Touzios C, Kolios D, Athanasiou G, Athanasopoulou E, Gerou I, Gartzonika C. Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health. Medicina. 2020; 56(2):88. https://doi.org/10.3390/medicina56020088
Chicago/Turabian StyleSakkas, Hercules, Petros Bozidis, Christos Touzios, Damianos Kolios, Georgia Athanasiou, Eirini Athanasopoulou, Ioanna Gerou, and Constantina Gartzonika. 2020. "Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health" Medicina 56, no. 2: 88. https://doi.org/10.3390/medicina56020088
APA StyleSakkas, H., Bozidis, P., Touzios, C., Kolios, D., Athanasiou, G., Athanasopoulou, E., Gerou, I., & Gartzonika, C. (2020). Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health. Medicina, 56(2), 88. https://doi.org/10.3390/medicina56020088






