Lychee Peel Extract Ameliorates Hyperuricemia by Regulating Uric Acid Production and Excretion in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Methods
2.2. Preparation of LPE
2.3. Determination of Total Polyphenol Content of LPE
2.4. Determination of Individual Components in LPE
2.5. The Effect of LPE on the Activity of XOD
2.6. Effect of LPE on Hyperuricemia Mice
2.6.1. Experimental Design and Administration
2.6.2. Treatment of Experimental Animals
2.6.3. Measurement of Biochemical Indicators in Mice
2.6.4. Extraction of Total Protein from Kidney Tissue
2.6.5. Western Blot
2.7. Statistical Analysis
3. Results and Discussion
3.1. Composition of LPE
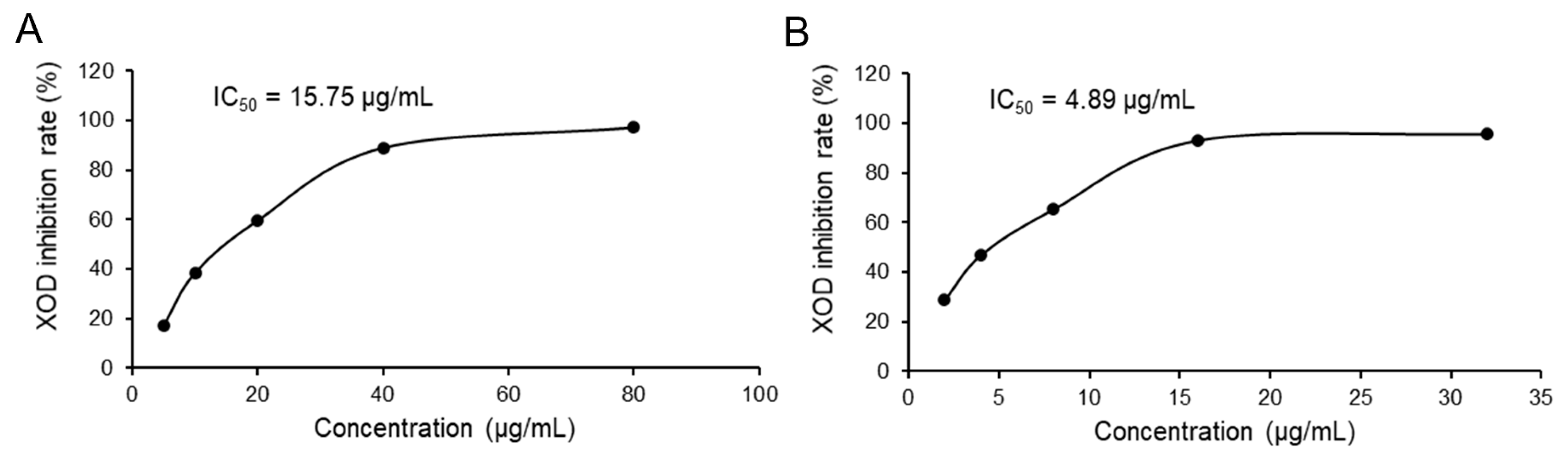
3.2. In Vitro Inhibitory Effect of LPE on XOD
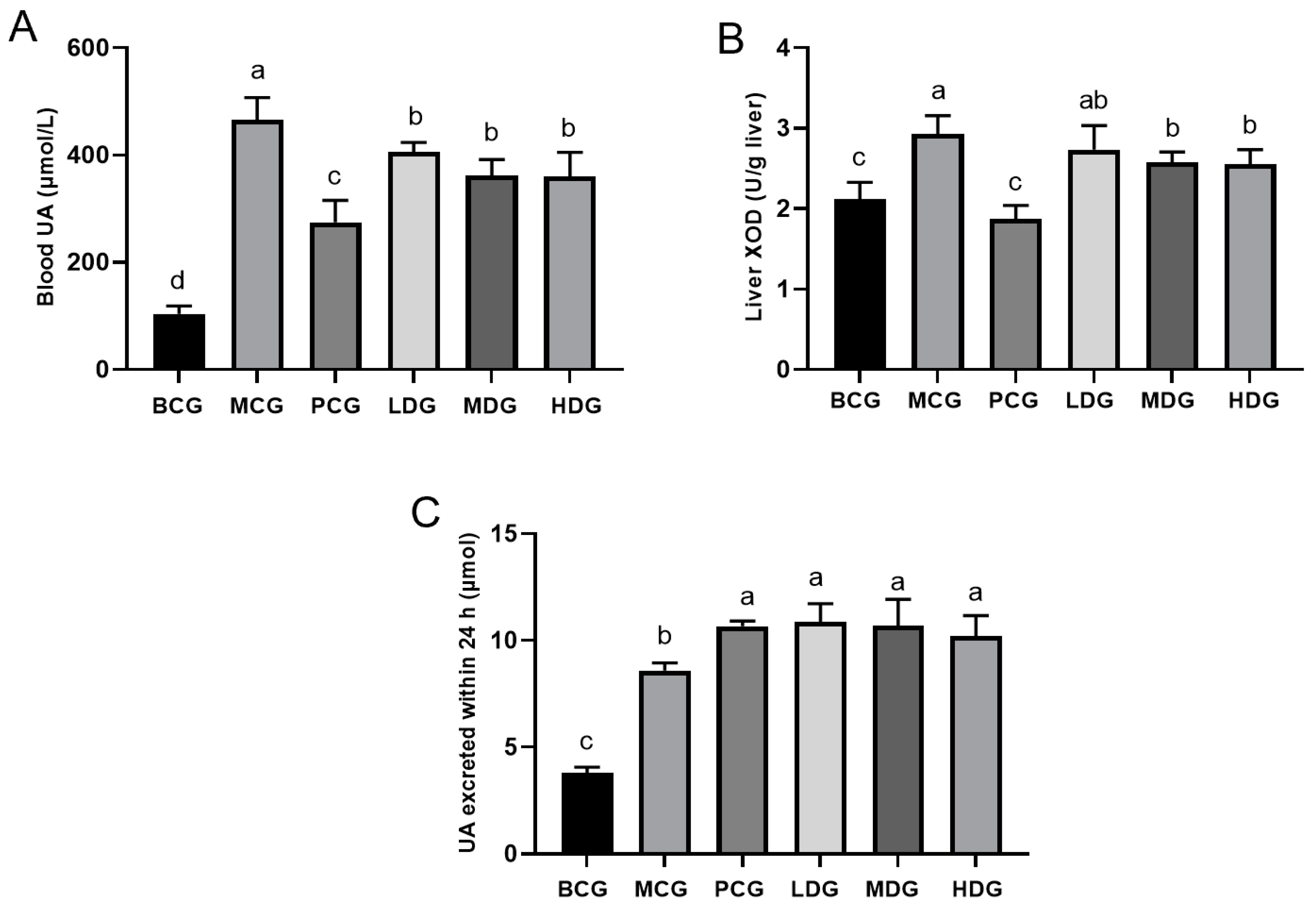
3.3. Effects of LPE on UA Content and XOD Activity in Hyperuricemia Mice
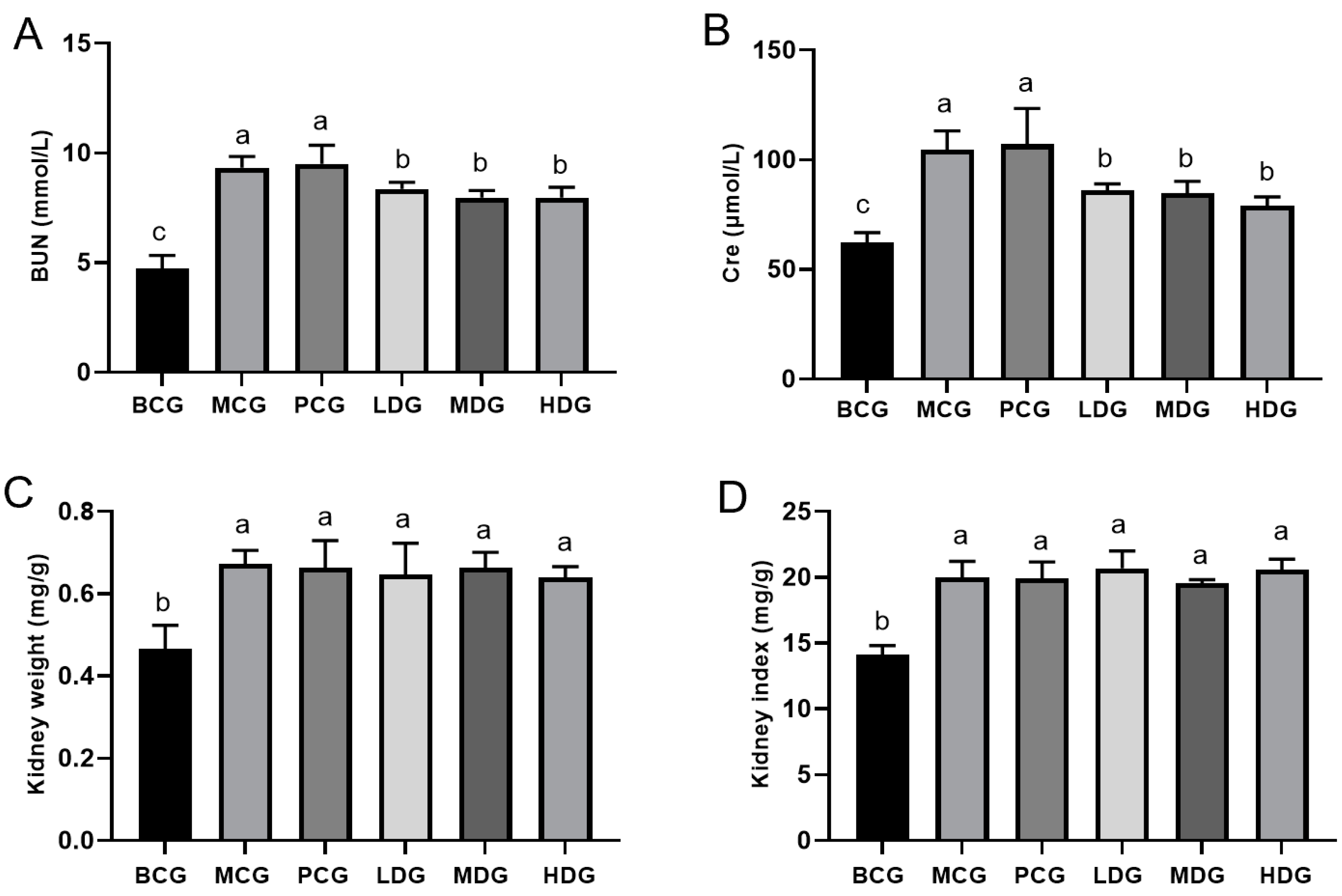
3.4. Effects of LPE on Kidney Function in Mice with Hyperuricemia
3.5. Effects of LPE on Liver Function in Hyperuricemia Mice
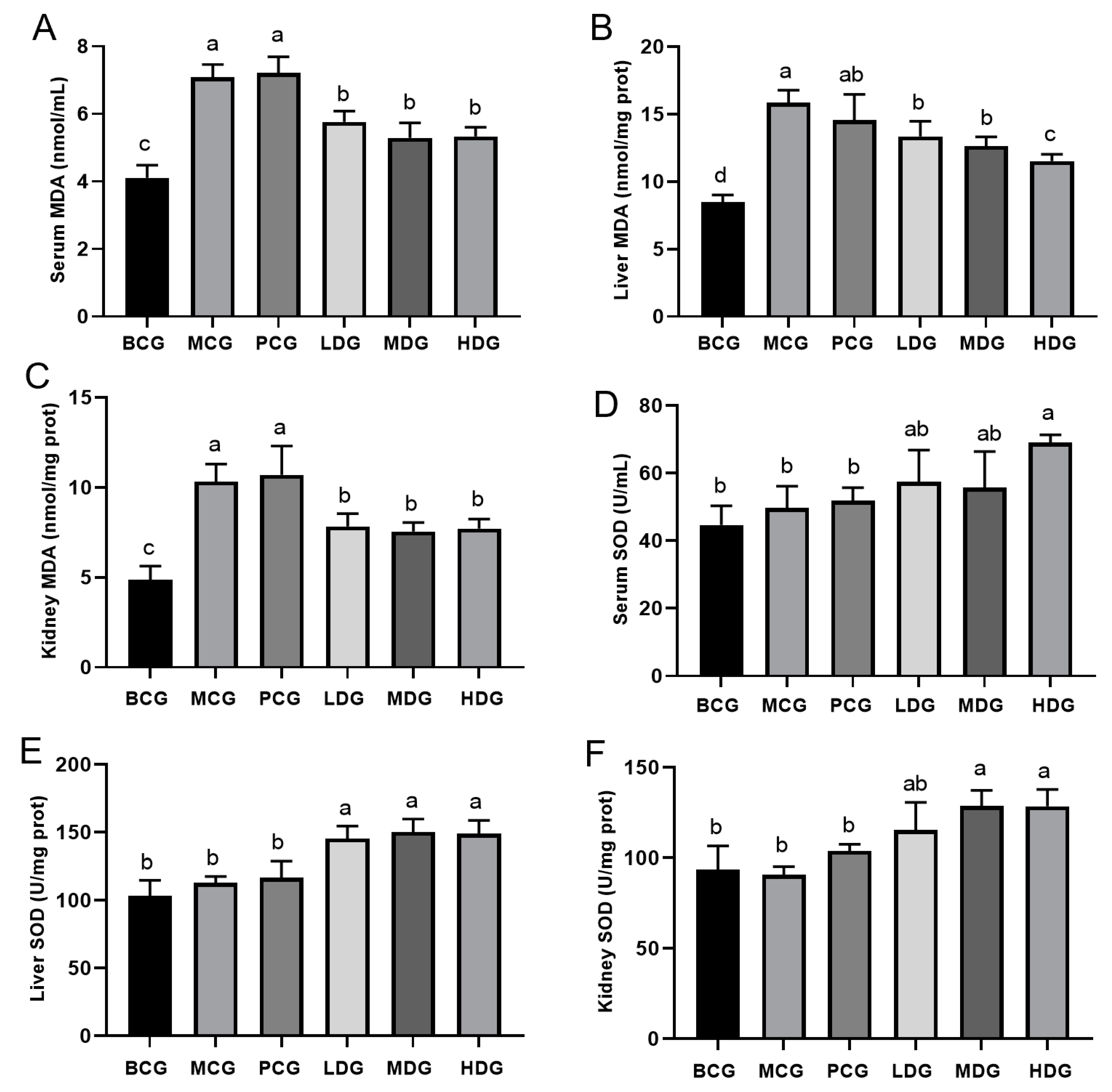
3.6. Effect of LPE on Antioxidant Capacity in Hyperuricemia Mice
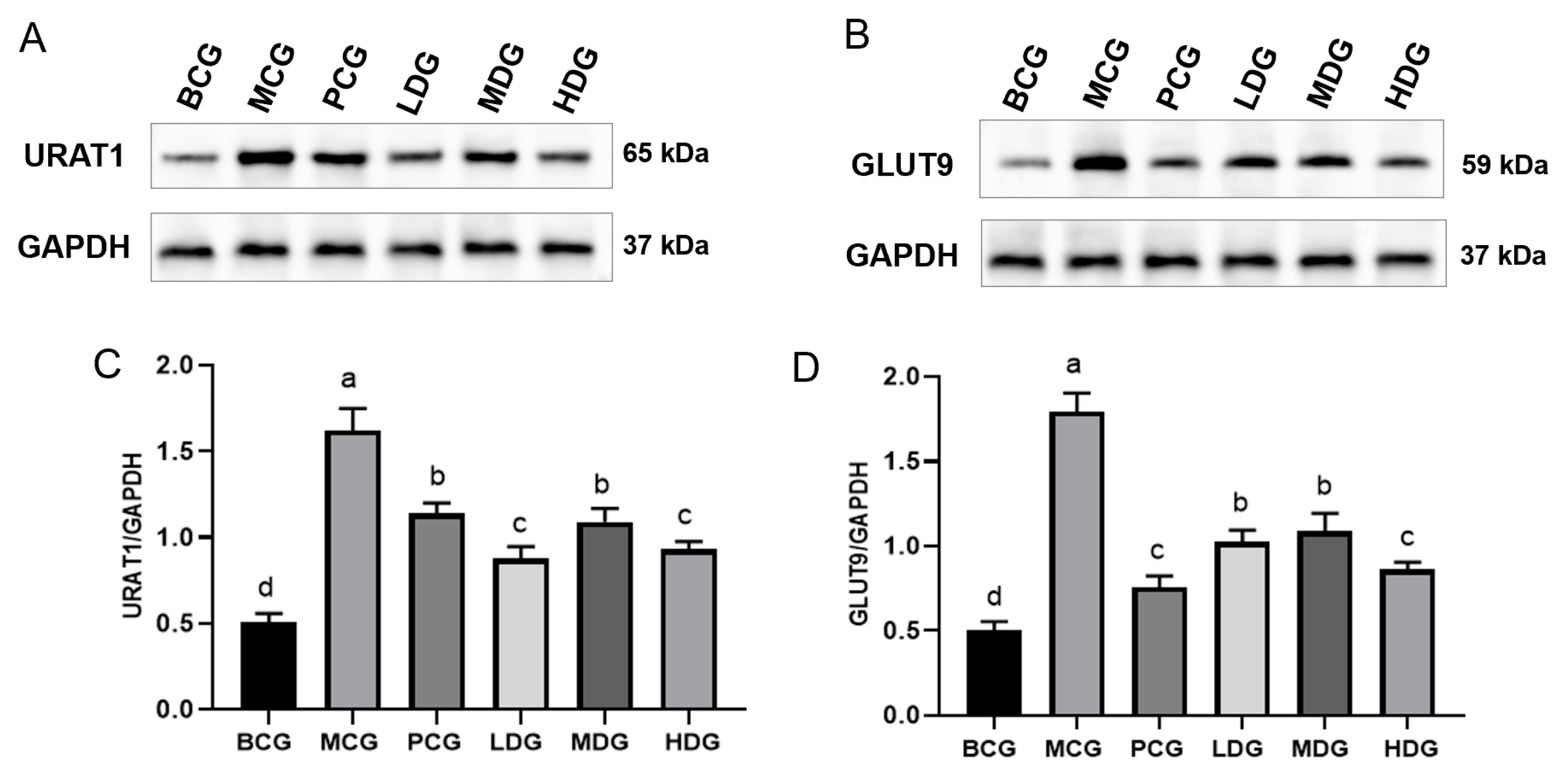
3.7. Impact of LPE on URAT1 and GLUT9 Protein Expression in Renal Tissues of Hyperuricemia Mice
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, H.; Hu, Z.; Wang, Y.; Chen, H.; Huang, X. Phenolic compounds and the antioxidant activities in litchi pericarp: Difference among cultivars. Sci. Hortic. 2011, 129, 784–789. [Google Scholar] [CrossRef]
- Joković, N.; Matejić, J.; Zvezdanović, J.; Stojanović-Radić, Z.; Stanković, N.; Mihajilov-Krstev, T.; Bernstein, N. Onion Peel as a Potential Source of Antioxidants and Antimicrobial Agents. Agronomy 2024, 14, 453. [Google Scholar] [CrossRef]
- Sarni-Manchado, P.; Roux, E.L.; Guernevé, C.L.; Lozano, Y.; Cheynier, V. Phenolic Composition of Litchi Fruit Pericarp. J. Agric. Food Chem. 2000, 48, 5995–6002. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liang, H.; Zhang, M.; Zhang, R.; Deng, Y.; Wei, Z.; Zhang, Y.; Tang, X. Phenolic Profiles and Antioxidant Activity of Litchi (Litchi chinensis Sonn.) Fruit Pericarp from Different Commercially Available Cultivars. Molecules 2012, 17, 14954–14967. [Google Scholar] [CrossRef]
- Kessy, H.N.E.; Hu, Z.; Zhao, L.; Zhou, M. Effect of Steam Blanching and Drying on Phenolic Compounds of Litchi Pericarp. Molecules 2016, 21, 729. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhang, L.; Liu, J.; Li, D. Litchi Pericarp Extract Treats Type 2 Diabetes Mellitus by Regulating Oxidative Stress, Inflammatory Response, and Energy Metabolism. Antioxidants 2024, 13, 495. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yuan, S.; Wang, J.; Lin, P.; Liu, G.; Lu, Y.; Zhang, J.; Wang, W.; Wei, Y. Anticancer activity of litchi fruit pericarp extract against human breast cancer in vitro and in vivo. Toxicol. Appl. Pharmacol. 2006, 215, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Rong, S.; Zhao, S.; Kai, X.; Zhang, L.; Zhao, Y.; Xiao, X.; Bao, W.; Liu, L. Procyanidins extracted from the litchi pericarp attenuate atherosclerosis and hyperlipidemia associated with consumption of a high fat diet in apolipoprotein-E knockout mice. Biomed. Pharmacother. 2018, 97, 1639–1644. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, E.R.; De Abreu, C.M.P.; Rocha, D.A.; Sousa, R.V.D.; Fráguas, R.M.; Braga, M.A.; César, P.H.S. Lychee (Litchi chinensis Sonn.) peel flour: Effects on hepatoprotection and dyslipidemia induced by a hypercholesterolemic diet. An. Acad. Bras. Cienc. 2018, 90, 267–281. [Google Scholar] [CrossRef]
- Park, C.H.; Noh, J.S.; Fujii, H.; Roh, S.; Song, Y.; Choi, J.S.; Chung, H.Y.; Yokozawa, T. Oligonol, a low-molecular-weight polyphenol derived from lychee fruit, attenuates glucolipotoxicity-mediated renal disorder in type 2 diabetic db/db mice. Drug Discov. Ther. 2015, 9, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Dehlin, M.; Jacobsson, L.; Roddy, E. Global epidemiology of gout: Prevalence, incidence, treatment patterns and risk factors. Nat. Rev. Rheumatol. 2020, 16, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H.; Adachi, H.; Hakoshima, M.; Katsuyama, H. Molecular Biological and Clinical Understanding of the Patho-Physiology and Treatments of Hyperuricemia and Its Association with Metabolic Syndrome, Cardiovascular Diseases and Chronic Kidney Disease. Int. J. Mol. Sci. 2021, 22, 9221. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Tanaka, A.; Node, K.; Kobayashi, Y. Uric acid and cardiovascular disease: A clinical review. J. Cardiol. 2021, 78, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Aihemaitijiang, S.; Zhang, Y.; Zhang, L.; Yang, J.; Ye, C.; Halimulati, M.; Zhang, W.; Zhang, Z. The Association between Purine Rich Food Intake and Hyperuricemia: A Cross-Sectional Study in Chinese Adult Residents. Nutrients 2020, 12, 3835. [Google Scholar] [CrossRef]
- Liu, Y.; Jarman, J.B.; Low, Y.S.; Augustijn, H.E.; Huang, S.; Chen, H.; DeFeo, M.E.; Sekiba, K.; Hou, B.; Meng, X.; et al. A widely dis-tributed gene cluster compensates for uricase loss in hominids. Cell 2023, 186, 3400–3413. [Google Scholar] [CrossRef]
- Zhu, J.; Shi, Y.; Wang, H.; Li, M. Two new phenylpropanoid glycosides from Lagotis brachystachya Maxim and their Xanthione Oxidase inhibitions. Nat. Prod. Res. 2021, 35, 2131–2136. [Google Scholar] [CrossRef] [PubMed]
- Adomako, E.; Moe, O.W. Uric Acid and Urate in Urolithiasis: The Innocent Bystander, Instigator, and Perpetrator. Semin. Nephrol. 2020, 40, 564–573. [Google Scholar] [CrossRef]
- Dalbeth, N.; Gosling, A.L.; Gaffo, A.; Abhishek, A. Gout. Lancet 2021, 397, 1843–1855. [Google Scholar] [CrossRef]
- Shen, Q.; Wang, J.; Roy, D.; Sun, L.; Jiang, Z.; Zhang, L.; Huang, X. Organic anion transporter 1 and 3 contribute to traditional Chinese medicine-induced nephrotoxicity. Chin. J. Nat. Med. 2020, 18, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Rundles, R.W.; Metz, E.N.; Silberman, H.R. Allopurinol in the treatment of gout. Ann. Intern. Med. 1966, 64, 229–258. [Google Scholar] [PubMed]
- Stamp, L.K.; Chapman, P.T. Allopurinol hypersensitivity: Pathogenesis and prevention. Best Pract. Res. Clin. Rheumatol. 2020, 34, 101501. [Google Scholar] [CrossRef] [PubMed]
- Quintana, M.J.; Shum, A.Z.; Folse, M.S.; Ramesh, P.C.; Ahmadzadeh, S.; Varrassi, G.; Shekoohi, S.; Kaye, A.D. Gout Treatment and Clinical Considerations: The Role of Pegloticase, Colchicine, and Febuxostat. Cureus 2023, 15, e46649. [Google Scholar] [CrossRef]
- Lai, S.; Liao, K.; Kuo, Y.; Hwang, B.; Liu, C. Comparison of benzbromarone and allopurinol on the risk of chronic kidney disease in people with asymptomatic hyperuricemia. Eur. J. Intern. Med. 2023, 113, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Abdellatif, A.; Zhao, L.; Chamberlain, J.; Katya Cherny, K.; Xin, Y.; Marder, B.A.; Scandling, J.D.; Saag, K. Pegloticase efficacy and safety in kidney transplant recipients; results of the phase IV, open-label PROTECT clinical trial. Clin. Transplant. 2023, 37, e14993. [Google Scholar] [CrossRef]
- Qaed, E.; Almaamari, A.; Almoiliqy, M.; Alyafeai, E.; Sultan, M.; Aldahmash, W.; Mahyoub, M.A.; Tang, Z. Phosphocreatine attenuates doxorubicin-induced nephrotoxicity through inhibition of apoptosis, and restore mitochondrial function via activation of Nrf2 and PGC-1α pathways. Chem. Biol. Interact. 2024, 400, 111147. [Google Scholar] [CrossRef]
- Lu, J.; Dalbeth, N.; Yin, H.; Li, C.; Merriman, T.R.; Wei, W. Mouse models for human hyperuricaemia: A critical review. Nat. Rev. Rheumatol. 2019, 15, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Ji, H.; Song, W.; Zhang, D.; Su, W.; Liu, S. Anserine beneficial effects in hyperuricemic rats by inhibiting XOD, regulating uric acid transporter and repairing hepatorenal injury. Food Funct. 2022, 13, 9434–9442. [Google Scholar] [CrossRef]
- Hou, P.; Mi, C.; He, Y.; Zhang, J.; Wang, S.; Yu, F.; Anderson, S.; Zhang, Y.; Wu, X. Pallidifloside D from Smilax riparia enhanced allopurinol effects in hyperuricemia mice. Fitoterapia 2015, 105, 43–48. [Google Scholar] [CrossRef]
- Yang, C.; Kuo, W.; Wang, J.; Hsiang, Y.; Lin, Y.; Wang, Y.; Tsai, F.; Lee, C.; Chou, J.; Chang, H.; et al. Improvement in the Blood Urea Nitrogen and Serum Creatinine Using New Cultivation of Cordyceps militaris. Evid.-Based Complement. Altern. Med. 2022, 2022, 4321298. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Gao, H.; Zhang, Y.; Wang, S.; Lu, M.; Dai, X.; Liu, Y.; Shi, H.; Xu, T.; Yin, J.; et al. Apigenin Ameliorates Hyperuricemia and Renal Injury Through Regulation of Uric Acid Metabolism and JAK2/STAT3 Signaling Pathway. Pharmaceuticals 2022, 15, 1442. [Google Scholar] [CrossRef] [PubMed]
- van Beek, J.H.D.A.; de Moor, M.H.M.; de Geus, E.J.C.; Lubke, G.H.; Vink, J.M.; Willemsen, G.; Boomsma, D.I. The genetic architecture of liver enzyme levels: GGT, ALT and AST. Behav. Genet. 2013, 43, 329–339. [Google Scholar] [CrossRef]
- Wang, S.; Fang, Y.; Yu, X.; Guo, L.; Zhang, X.; Xia, D. The flavonoid-rich fraction from rhizomes of Smilax glabra Roxb. ameliorates renal oxidative stress and inflammation in uric acid nephropathy rats through promoting uric acid excretion. Biomed. Pharmacother. 2019, 111, 162–168. [Google Scholar] [CrossRef]
- Duan, X.; Jiang, Y.; Su, X.; Zhang, Z.; Shi, J. Antioxidant properties of anthocyanins extracted from litchi (Litchi chinenesis Sonn.) fruit pericarp tissues in relation to their role in the pericarp browning. Food Chem. 2007, 101, 1365–1371. [Google Scholar] [CrossRef]
- Weismann, D.; Hartvigsen, K.; Lauer, N.; Bennett, K.L.; Scholl, H.P.N.; Issa, P.C.; Cano, M.; Brandstätter, H.; Tsimikas, S.; Skerka, C.; et al. Complement factor H binds malondialdehyde epitopes and protects from oxidative stress. Nature 2011, 478, 76–81. [Google Scholar] [CrossRef]
- Borgstahl, G.E.O.; Oberley-Deegan, R.E. Superoxide Dismutases (SODs) and SOD Mimetics. Antioxidants 2018, 7, 156. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Jiang, Y.; Chen, Y.; Wu, T.; Lan, Q.; Li, Y.; Li, L.; Yang, Y.; Lin, C.; Cao, Y.; et al. CDER167, a dual inhibitor of URAT1 and GLUT9, is a novel and potent uricosuric candidate for the treatment of hyperuricemia. Acta. Pharmacol. Sin. 2022, 43, 121–132. [Google Scholar] [CrossRef]
- Arakawa, H.; Amezawa, N.; Kawakatsu, Y.; Tamai, I. Renal Reabsorptive Transport of Uric Acid Precursor Xanthine by URAT1 and GLUT9. Biol. Pharm. Bull. 2020, 43, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, A.; Kimura, H.; Chairoungdua, A.; Shigeta, Y.; Jutabha, P.; Cha, S.H.; Hosoyamada, M.; Takeda, M.; Sekine, T.; Igarashi, T.; et al. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 2002, 417, 447–452. [Google Scholar] [CrossRef]
- Yanai, H.; Adachi, H.; Hakoshima, M.; Iida, S.; Katsuyama, H. A Possible Therapeutic Application of the Selective Inhibitor of Urate Transporter 1, Dotinurad, for Metabolic Syndrome, Chronic Kidney Disease, and Cardiovascular Disease. Cells 2024, 13, 450. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.; Cai, S.; Sun, Y.; Chu, C. Chinese Sumac (Rhus chinensis Mill.) Fruits Prevent Hyperuricemia and Uric Acid Nephropathy in Mice Fed a High-Purine Yeast Diet. Nutrients 2024, 16, 184. [Google Scholar] [CrossRef]
- Hou, X.; Wang, Y.; Yang, Y.; Xiao, Z. Discovery of Novel Biphenyl Carboxylic Acid Derivatives as Potent URAT1 Inhibitors. Molecules 2023, 28, 7415. [Google Scholar] [CrossRef] [PubMed]

| Item | LPE |
|---|---|
| Total polyphenols (‰) | 517.04 ± 18.12 |
| (-)-Epicatechin (‰) | 102.96 ± 2.49 |
| (-)-Epigallocatechin (‰) | 31.12 ± 0.25 |
| Procyanidin A2 (‰) | 16.92 ± 0.21 |
| Procyanidin B1 (‰) | 13.57 ± 0.58 |
| Procyanidin B2 (‰) | 5.75 ± 0.08 |
| (+)-Catechin (‰) | 2.34 ± 0.26 |
| Vanillic acid (‰) | 0.28 ± 0.01 |
| Rutin (‰) | 0.21 ± 0.02 |
| Protocatechuic acid (‰) | 0.16 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Z.; Zhang, L.; Liu, J.; Yang, Z. Lychee Peel Extract Ameliorates Hyperuricemia by Regulating Uric Acid Production and Excretion in Mice. Curr. Issues Mol. Biol. 2025, 47, 76. https://doi.org/10.3390/cimb47020076
Guo Z, Zhang L, Liu J, Yang Z. Lychee Peel Extract Ameliorates Hyperuricemia by Regulating Uric Acid Production and Excretion in Mice. Current Issues in Molecular Biology. 2025; 47(2):76. https://doi.org/10.3390/cimb47020076
Chicago/Turabian StyleGuo, Zhenwang, Li Zhang, Jinlei Liu, and Ziming Yang. 2025. "Lychee Peel Extract Ameliorates Hyperuricemia by Regulating Uric Acid Production and Excretion in Mice" Current Issues in Molecular Biology 47, no. 2: 76. https://doi.org/10.3390/cimb47020076
APA StyleGuo, Z., Zhang, L., Liu, J., & Yang, Z. (2025). Lychee Peel Extract Ameliorates Hyperuricemia by Regulating Uric Acid Production and Excretion in Mice. Current Issues in Molecular Biology, 47(2), 76. https://doi.org/10.3390/cimb47020076





