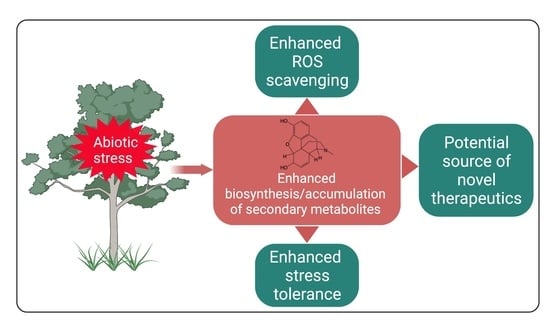
Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development
Abstract
1. Introduction
2. Plant Secondary Metabolites and Their Biological Roles
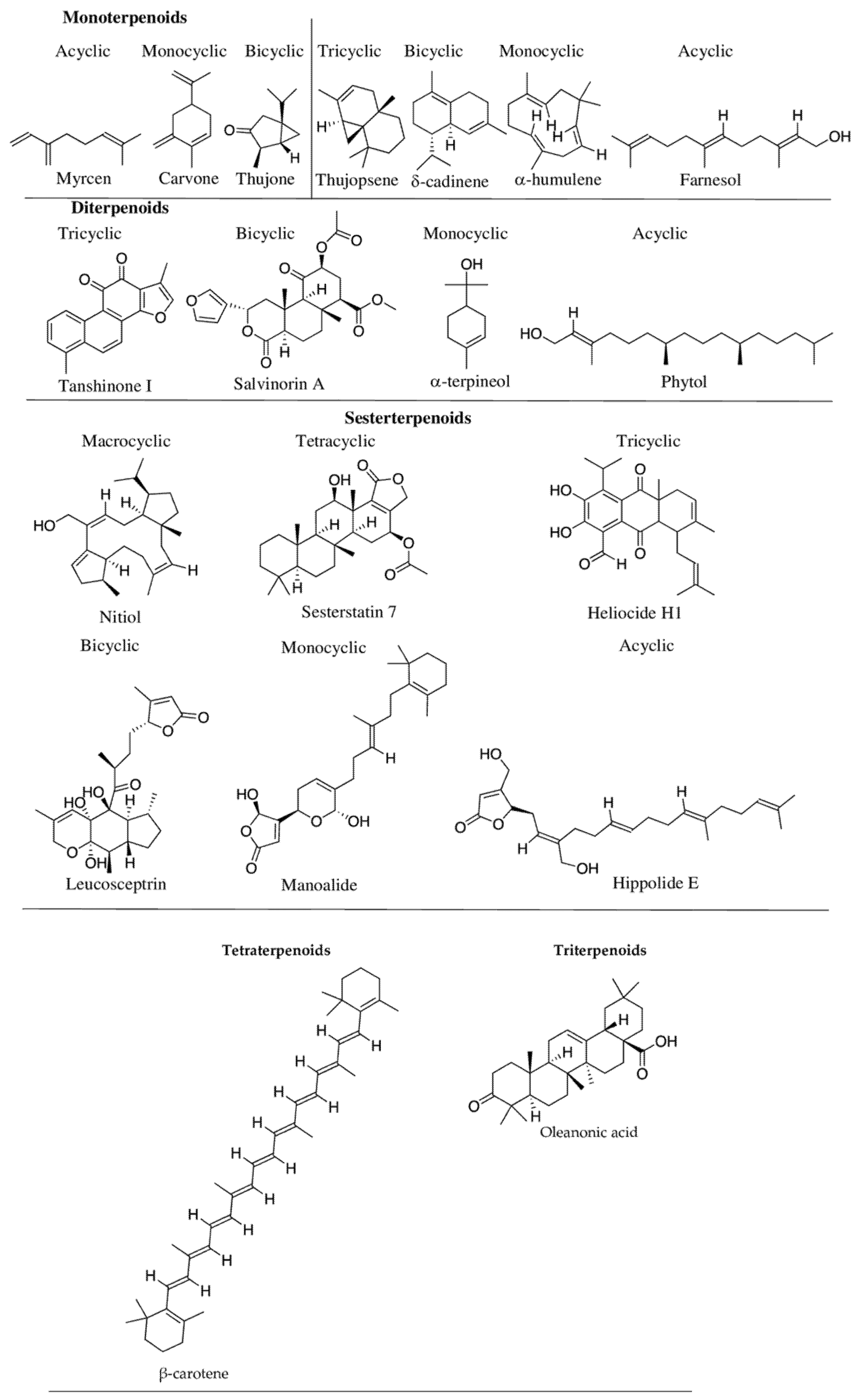
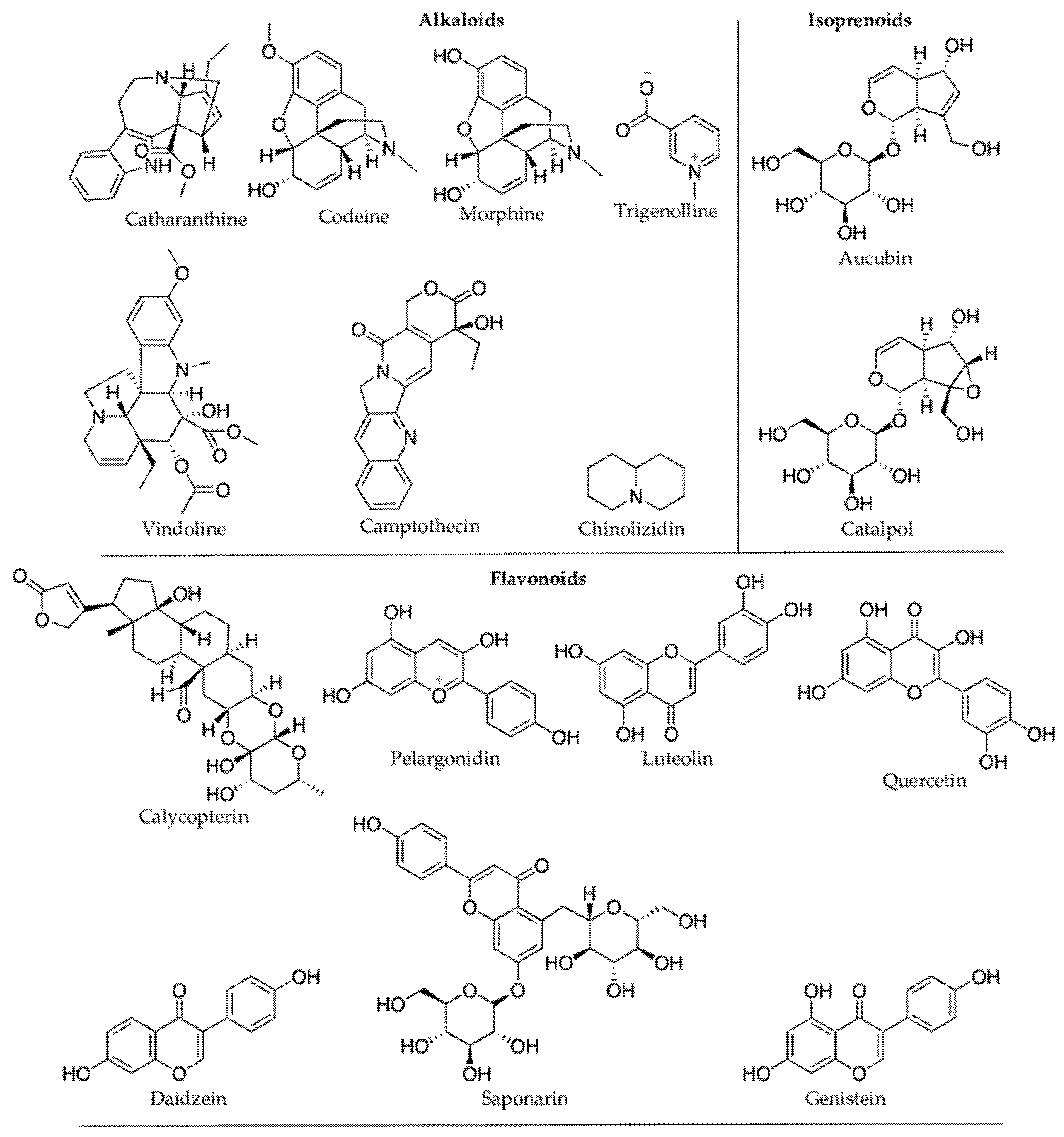
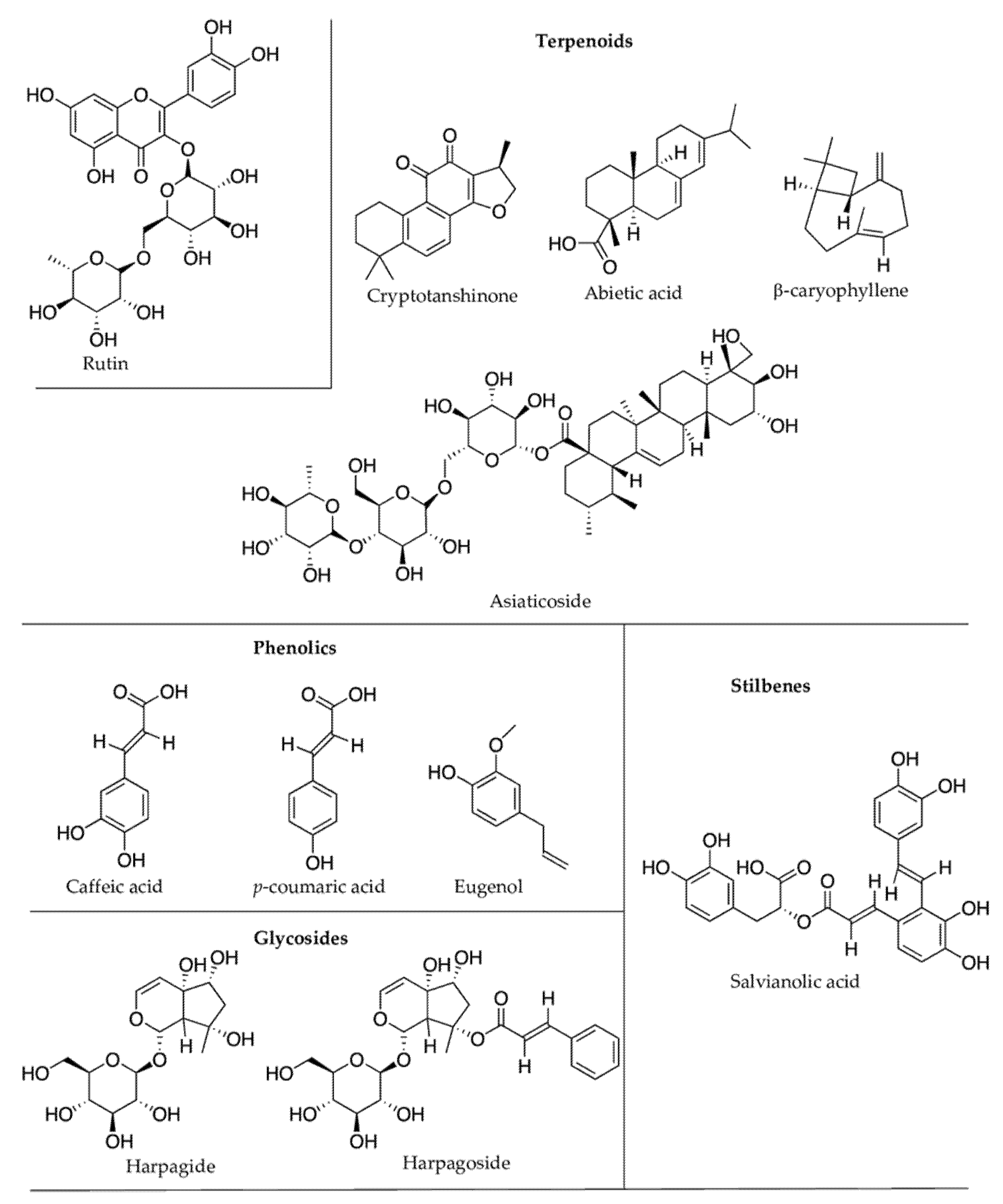
2.1. Terpenoids
- Monoterpenoids—two isoprene units (C-10 carbon atoms,)—e.g., linalool;
- Sesquiterpenoids—three isoprene units (C-15 carbon atoms)—e.g., β-caryophyllene;
- Diterpenoids—four isoprene units (C-20 carbon atoms)—e.g., abietic acid;
- Sesterterpenoids—five isoprene units (C-25 carbon atoms)—e.g., ophiobolin A;
- Triterpenoids—six isoprene units (C-30 carbon atoms)—e.g., ganoderic acid;
- Tetraterpenoids—eight isoprene units (C-40 carbon atoms)—e.g., α-carotene;
- Polyterpenoids—more than eight isoprene units (>C-40 carbon atoms)—e.g., trans-1,4-polyisoprene.
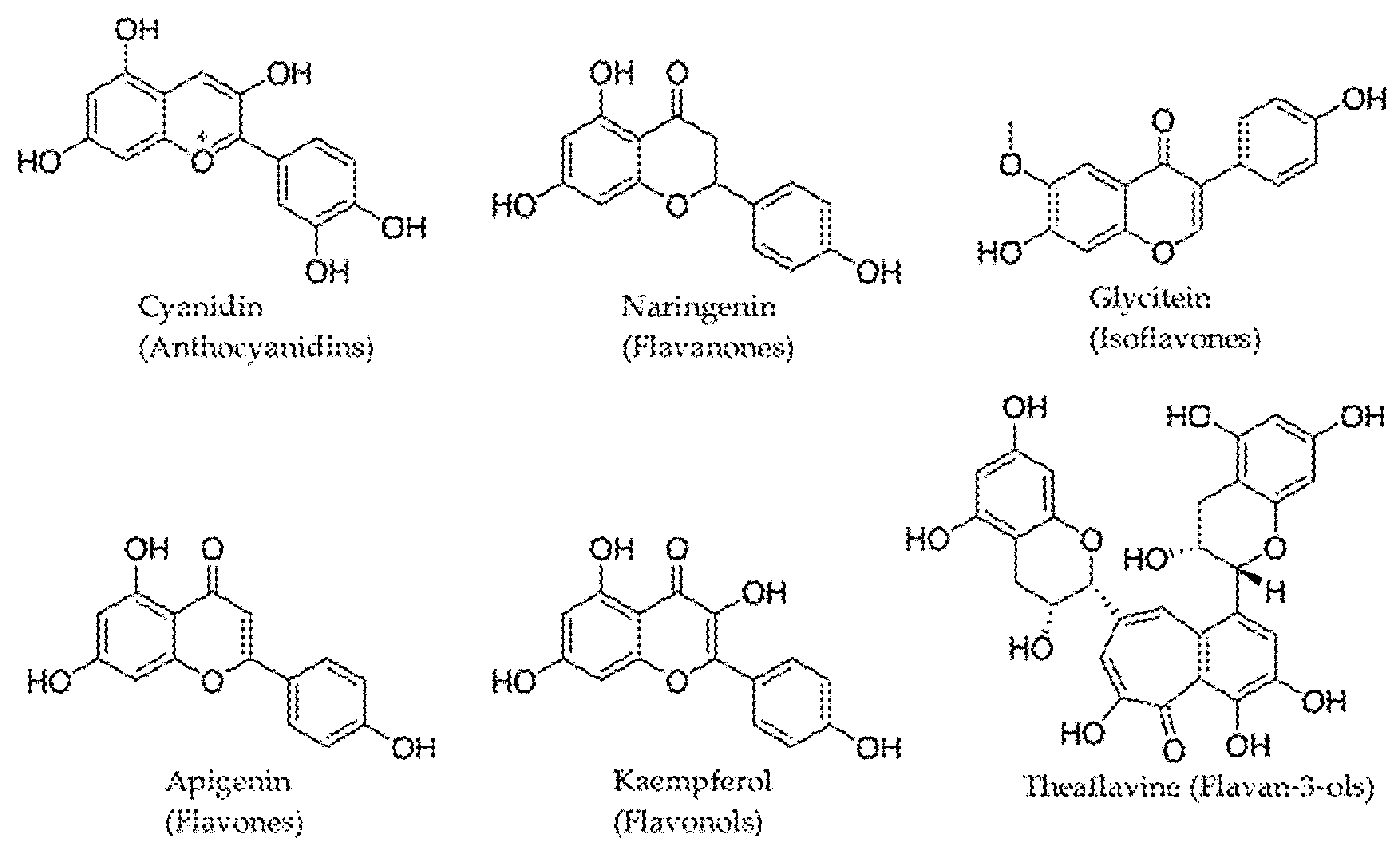
2.2. Phenolic Compounds
2.3. Nitrogen-Containing Compounds
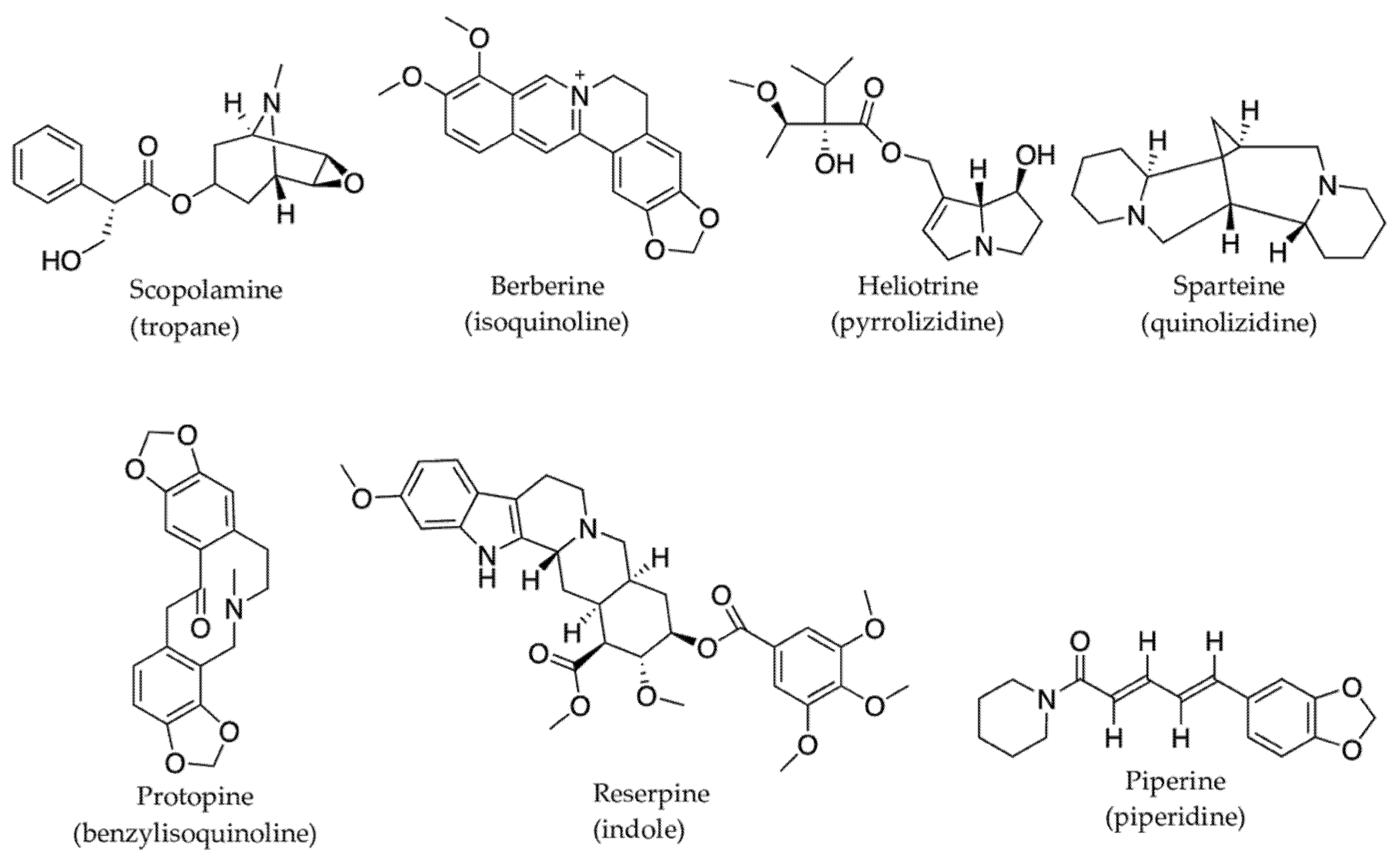
2.3.1. Alkaloids
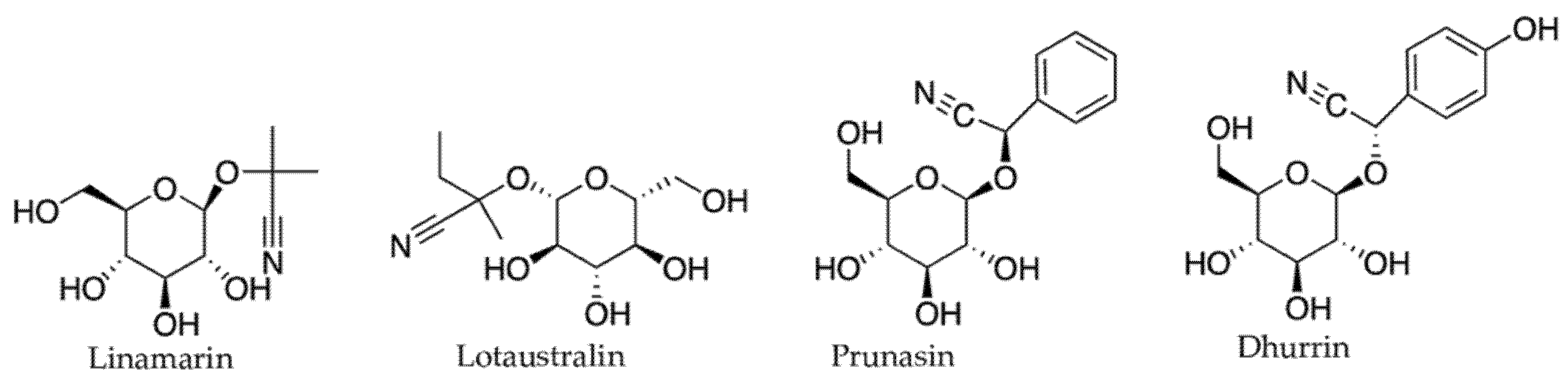
2.3.2. Cyanogenic Glycosides and Glucosinolates
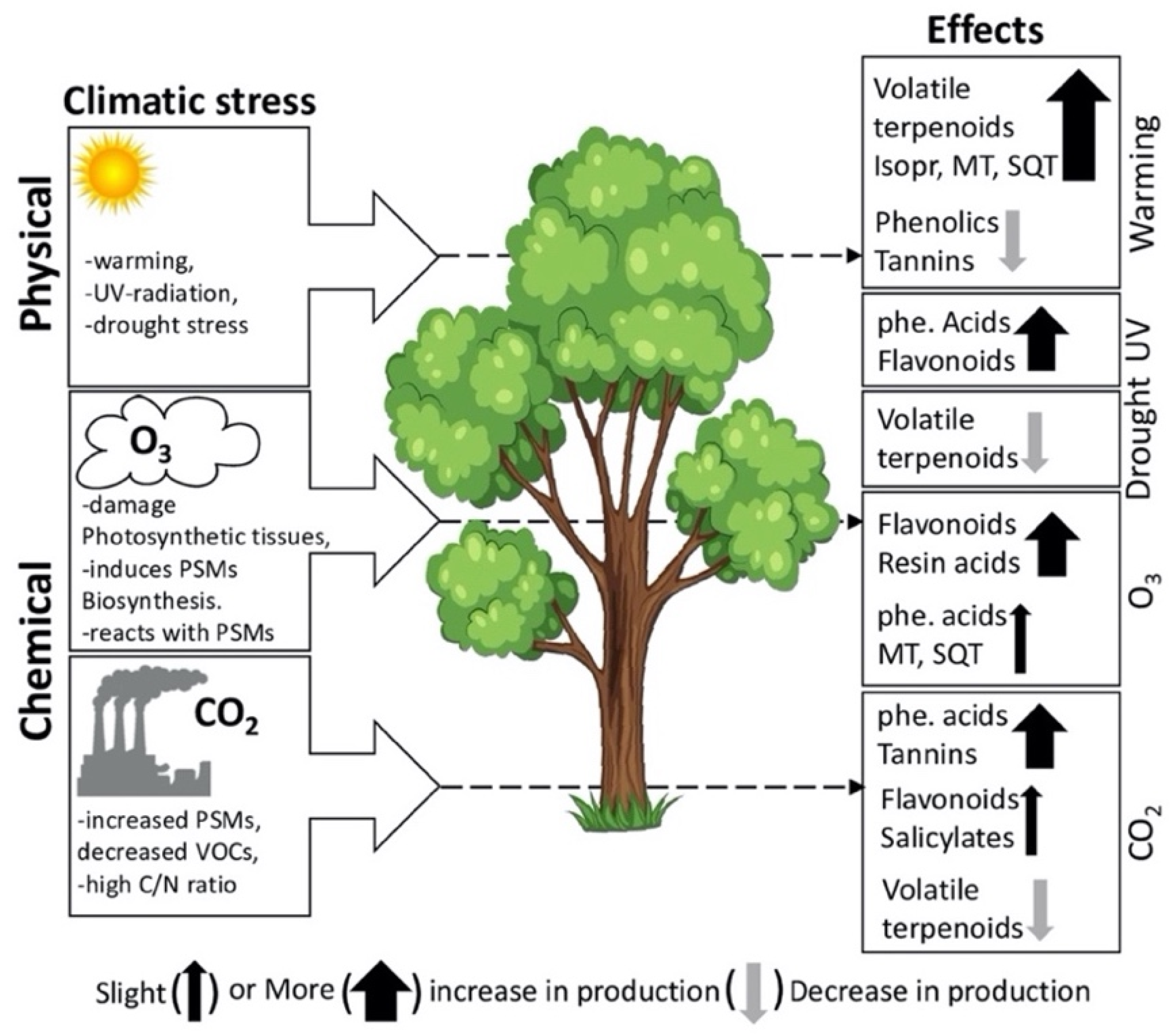
3. Factors Influencing PSMs Production in Plants
3.1. Effects of Heat Stress on PSMs
3.2. Effects of Cold Stress on PSMs
3.3. Effects of Drought Stress on PSMs
3.4. Effects of Ultraviolet (UV) Radiation on PSMs
3.5. Effects of Ozone on PSMs

4. Reported Pharmacological Properties of PSMs Present in Plants Affected by Ex Situ Abiotic Stresses
5. Biodiscovery Potential of Plants Growing under Ex-Situ Abiotic Stresses
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kessler, A.; Kalske, A. Plant secondary metabolite diversity and species interactions. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 115–138. [Google Scholar] [CrossRef]
- Sallas, L.; Luomala, E.M.; Utriainen, J.; Kainulainen, P.; Holopainen, J.K. Contrasting effects of elevated carbon dioxide concentration and temperature on rubisco activity, chlorophyll fluorescence, needle ultrastructure and secondary metabolites in conifer seedlings. Tree Physiol. 2003, 23, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Julkunen-Tiitto, R.; Nenadis, N.; Neugart, S.; Robson, M.; Agati, G.; Vepsäläinen, J.; Zipoli, G.; Nybakken, L.; Winkler, B.; Jansen, M.A.K. Assessing the response of plant flavonoids to UV radiation: An overview of appropriate techniques. Phytochem. Rev. 2014, 14, 273–297. [Google Scholar] [CrossRef]
- Kaling, M.; Kanawati, B.; Ghirardo, A.; Albert, A.; Winkler, J.B.; Heller, W.; Barta, C.; Loreto, F.; Schmitt-Kopplin, P.; Schnitzler, J.P. UV-B mediated metabolic rearrangements in poplar revealed by non-targeted metabolomics. Plant. Cell Environ. 2015, 38, 892–904. [Google Scholar] [CrossRef] [PubMed]
- Hošek, J.; Šmejkal, K. Flavonoids as anti-inflammatory agents. In Encyclopedia of Inflammatory Diseases; Miller, R., Ed.; Hayle Medical: New York, NY, USA, 2015; pp. 1–17. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Yeshi, K.; Yangdon, P.; Kashyap, S.; Wangchuk, P. Antioxidant activity and the polyphenolic and flavonoid contents of five high altitude medicinal plants used in Bhutanese sowa rigpa medicine. JBAPN 2017, 7, 18–26. [Google Scholar] [CrossRef]
- Pinto, D.M.; Blande, J.D.; Souza, S.R.; Nerg, A.M.; Holopainen, J.K. Plant volatile organic compounds (VOCs) in ozone (O3) polluted atmospheres: The ecological effects. J. Chem. Ecol. 2010, 36, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, A.C. Medicinal plants, conservation and livelihoods. Biodivers. Conserv. 2004, 13, 1477–1517. [Google Scholar] [CrossRef]
- Applequist, W.L.; Brinckmann, J.A.; Cunningham, A.B.; Hart, R.E.; Heinrich, M.; Katerere, D.R.; van Andel, T. Scientists’ warning on climate change and medicinal plants. Planta Med. 2020, 86, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.K.; Roy, D.K. Use of medicinal plant and its vulnerability due to climate change in northern part of Bangladesh. Am. J. Plant. Sci. 2016, 07, 1782–1793. [Google Scholar] [CrossRef]
- Gupta, A.; Singh, P.P.; Singh, P.; Singh, K.; Singh, A.V.; Singh, S.K.; Kumar, A. Medicinal Plants Under Climate Change: Impacts on Pharmaceutical Properties of Plants. In Climate Change and Agricultural Ecosystems; Elsevier: Amsterdam, The Netherlands, 2019; pp. 181–209. [Google Scholar]
- Trouvelot, S.; Heloir, M.C.; Poinssot, B.; Gauthier, A.; Paris, F.; Guillier, C.; Combier, M.; Trda, L.; Daire, X.; Adrian, M. Carbohydrates in plant immunity and plant protection: Roles and potential application as foliar sprays. Front. Plant. Sci. 2014, 5, 1–14. [Google Scholar] [CrossRef]
- Lee, S.; Choi, H.; Suh, S.; Doo, I.S.; Oh, K.Y.; Choi, E.J.; Taylor, A.S.; Low, P.S.; Lee, Y. Oligogalacturonic acid and chitosan reduce stomatal aperture by inducing the evolution of reactive oxygen species from guard cells of tomato and Commelina communis. Plant Physiol. 1999, 121, 147–152. [Google Scholar] [CrossRef]
- Albert, M. Peptides as triggers of plant defence. J. Exp. Bot. 2013, 64, 5269–5279. [Google Scholar] [CrossRef]
- Marmiroli, N.; Maestri, E. Plant peptides in defense and signaling. Peptides 2014, 56, 30–44. [Google Scholar] [CrossRef]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Kesselmeier, J.; Staudt, M. Biogenic volatile organic compounds (VOC): An overview on emission, physiology and ecology. J. Atmos. Chem. 1999, 33, 23–88. [Google Scholar] [CrossRef]
- Neilson, E.H.; Goodger, J.Q.D.; Woodrow, I.E.; Møller, B.L. Plant chemical defense: At what cost? Trends Plant Sci. 2013, 18, 250–258. [Google Scholar] [CrossRef]
- Willis, K.J. State of the World’s Plants 2017. Available online: https://www.kew.org/about-us/press-media/state-of-the-worlds-plants-2017 (accessed on 9 November 2021).
- van Wyk, B.E. The value of chemosystematics in clarifying relationships in the genistoid tribes of papilionoid legumes. Biochem. Syst. Ecol. 2003, 31, 875–884. [Google Scholar] [CrossRef]
- Acamovic, T.; Brooker, J.D. Biochemistry of plant secondary metabolites and their effects in animals. Proc. Nutr. Soc. 2005, 64, 403–412. [Google Scholar] [CrossRef]
- Ritmejeryte, E.; Boughton, B.A.; Bayly, M.J.; Miller, R.E. Unique and highly specific cyanogenic glycoside localization in stigmatic cells and pollen in the genus Lomatia (Proteaceae). Ann. Bot. 2020, 126, 387–400. [Google Scholar] [CrossRef]
- Sampaio, B.L.; Edrada-Ebel, R.; Da Costa, F.B. Effect of the environment on the secondary metabolic profile of Tithonia diversifolia: A model for environmental metabolomics of plants. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ryde, I.; Li, T.; Rieksta, J.; Dos Santos, B.M.; Neilson, E.H.J.; Gericke, O.; Jepsen, J.U.; Bork, L.R.H.; Holm, H.S.; Rinnan, R. Seasonal and elevational variability in the induction of specialized compounds from mountain birch (Betula pubescens var. pumila) by winter moth larvae (Operophtera brumata). Tree Physiol. 2021, 41, 1019–1033. [Google Scholar] [CrossRef] [PubMed]
- Grainger, T.N.; Gilbert, B. Multi-scale responses to warming in an experimental insect metacommunity. Glob. Chang. Biol. 2017, 23, 5151–5163. [Google Scholar] [CrossRef]
- Jogawat, A.; Yadav, B.; Chhaya; Lakra, N.; Singh, A.K.; Narayan, O.P. Crosstalk between phytohormones and secondary metabolites in the drought stress tolerance of crop plants: A review. Physiol. Plant. 2021, 172, 1106–1132. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kong, D.; Fu, Y.; Sussman, M.R.; Wu, H. The effect of developmental and environmental factors on secondary metabolites in medicinal plants. Plant. Physiol. Biochem. 2020, 148, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Yadav, B.; Jogawat, A.; Rahman, M.S.; Narayan, O.P. Secondary metabolites in the drought stress tolerance of crop plants: A review. Gene Reports 2021, 23, 1–14. [Google Scholar] [CrossRef]
- Jamwal, K.; Bhattacharya, S.; Puri, S. Plant growth regulator mediated consequences of secondary metabolites in medicinal plants. J. Appl. Res. Med. Aromat. Plants 2018, 9, 26–38. [Google Scholar] [CrossRef]
- Aharoni, A.; Jongsma, M.A.; Bouwmeester, H.J. Volatile science? Metabolic engineering of terpenoids in plants. Trends Plant Sci. 2005, 10, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Song, M.C.; Kim, E.J.; Kim, E.; Rathwell, K.; Nam, S.J.; Yoon, Y.J. Microbial biosynthesis of medicinally important plant secondary metabolites. Nat. Prod. Rep. 2014, 31, 1497–1509. [Google Scholar] [CrossRef]
- Breitmaier, E. Terpenes: Flavors, Fragrances, Pharmaca, Pheromones; Wiley-VCH: Weinheim, Germany, 2006; pp. 1–223. [Google Scholar]
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant. Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Kessler, A.; Baldwin, I.T. Defensive function of herbivore-induced plant volatile emissions in nature. Science 2001, 291, 2141–2144. [Google Scholar] [CrossRef]
- Singh, B.; Sharma, R.A. Plant terpenes: Defense responses, phylogenetic analysis, regulation and clinical applications. Three Biotech. 2015, 5, 129–151. [Google Scholar] [CrossRef]
- Gonzalez-Lamothe, R.; Mitchell, G.; Gattuso, M.; Diarra, M.S.; Malouin, F.; Bouarab, K. Plant antimicrobial agents and their effects on plant and human pathogens. Int. J. Mol. Sci. 2009, 10, 3400–3419. [Google Scholar] [CrossRef] [PubMed]
- Loreto, F.; Schnitzler, J.P. Abiotic stresses and induced BVOCs. Trends Plant. Sci 2010, 15, 154–166. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.K.; Ezzat, M.O.; Majid, A.S.A.; Majid, A.M.S.A. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-Caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef] [PubMed]
- Porres-Martinez, M.; Gonzalez-Burgos, E.; Carretero, M.E.; Gomez-Serranillos, M.P. In vitro neuroprotective potential of the monoterpenes alpha-pinene and 1,8-cineole against H2O2-induced oxidative stress in PC12 cells. Z. Nat. C J. Biosci. 2016, 71, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Blanch, J.S.; Peñuelas, J.; Sardans, J.; Llusià, J. Drought, warming and soil fertilization effects on leaf volatile terpene concentrations in Pinus halepensis and Quercus ilex. Acta. Physiol. Plant. 2009, 31, 207–218. [Google Scholar] [CrossRef]
- Bartwal, A.; Mall, R.; Lohani, P.; Guru, S.K.; Arora, S. Role of secondary metabolites and brassinosteroids in plant defense against environmental stresses. J. Plant. Growth Regul. 2013, 32, 216–232. [Google Scholar] [CrossRef]
- Kozukue, N.; Han, J.-S.; Lee, K.-R.; Friedman, M. Dehydrotomatine and alpha-tomatine content in tomato fruits and vegetative plant tissues. J. Agric. Food Chem. 2004, 52, 2079–2083. [Google Scholar] [CrossRef] [PubMed]
- Trojanowska, M.R.; Threlfall, D.R. Regulation of saponin biosynthesis in primary roots of oat. Acta. Bot. Gall. 1999, 146, 101–104. [Google Scholar] [CrossRef]
- Moses, T.; Papadopoulou, K.K.; Osbourn, A. Metabolic and functional diversity of saponins, biosynthetic intermediates and semi-synthetic derivatives. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 439–462. [Google Scholar] [CrossRef]
- Friedman, M. Potato glycoalkaloids and metabolites: Roles in the plants and in the diet. J. Agric. Food Chem. 2006, 54, 8655–8681. [Google Scholar] [CrossRef] [PubMed]
- Ndamba, J.; Lemmich, E.; Mølgaard, P. Investigation of the diurnal, ontogenetic and seasonal variation in the molluscicidal saponin content of Phytolacca dodecandra aqueous berry extracts. Phytochemistry 1994, 35, 95–99. [Google Scholar] [CrossRef]
- Li, J.T.; Chen, S.L.; Liu, S.C.; Yang, D.J. Effect of harvest time on saponins in Yam (Dioscorea pseudojaponica Yamamoto). J. Food Drug Anal. 2009, 17, 116–122. [Google Scholar]
- Hayat, M.; Abbas, M.; Munir, F. Potential of plant flavonoids in pharmaceutics and nutraceutics. J. Biomol. Biochem. 2017, 1, 12–17. [Google Scholar]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant. Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Hussein, R.A.; El-Anssary, A.A. Plants Secondary Metabolites: The Key Drivers of the Pharmacological Actions of Medicinal Plants. In Herbal Medicine; Intechopen: London, UK, 2019; pp. 11–30. [Google Scholar] [CrossRef]
- Winkel-Shirley, B. Biosynthesis of flavonoids and effects of stress. Curr. Opin. Plant. Biol. 2002, 5, 218–223. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Tattini, M. Multiple functional roles of flavonoids in photoprotection. New Phytol. 2010, 186, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Ohnishi, T. The significance of the study about the biological effects of solar ultraviolet radiation using the exposed facility on the international space station. Biol. Sci. Space 2004, 18, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B.; W’illiams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Samanta, A.; Das, G.; Das, S. Roles of flavonoids in plants. Int. J. Pharm. Sci. Tech. 2011, 6, 12–35. [Google Scholar]
- Schulz, E.; Tohge, T.; Zuther, E.; Fernie, A.R.; Hincha, D.K. Flavonoids are determinants of freezing tolerance and cold acclimation in Arabidopsis thaliana. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pastore, C.; Dal Santo, S.; Zenoni, S.; Movahed, N.; Allegro, G.; Valentini, G.; Filippetti, I.; Tornielli, G.B. Whole plant temperature manipulation affects flavonoid metabolism and the transcriptome of grapevine berries. Front. Plant. Sci. 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Bieza, K.; Lois, r. An Arabidopsis mutant tolerant to lethal ultraviolet-B levels shows constitutively elevated accumulation of flavonoids and other phenolics. Plant. Physiol. 2001, 126, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Kabtni, S.; Sdouga, D.; Bettaib Rebey, I.; Save, M.; Trifi-Farah, N.; Fauconnier, M.L.; Marghali, S. Influence of climate variation on phenolic composition and antioxidant capacity of Medicago minima populations. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tattini, M.; Landi, M.; Brunetti, C.; Giordano, C.; Remorini, D.; Gould, K.S.e.a. Epidermal coumaroyl anthocyanins protect sweet basil against excess light stress: Multiple consequences of light attenuation. Physiol. Plant 2014, 152, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Zhang, J.; Chen, C.; Zhang, D.; Li, P.; Ma, F. Anthocyanin contributes more to hydrogen peroxide scavenging than other phenolics in apple peel. Food Chem. 2014, 152, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant. Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Leopoldini, M.; Russo, N.; Chiodo, S.; Toscano, M. Iron chelation by the powerful antioxidant flavonoid quercetin. J. Agric. Food Chem. 2006, 54, 6343–6351. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1–21. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Kurek, J. Alkaloids—Their importance in nature and for human life. In Their Importance in Nature and for Human Life; Kurek, J., Ed.; IntechOpen: London, UK, 2019; pp. 1–7. [Google Scholar] [CrossRef]
- Levin, D.A. Alkaloids bearing plants: An ecogeographic perspective. Am. Nat. 1976, 110, 261–284. [Google Scholar] [CrossRef]
- Nowacki, E.; Waller, G.R. A study on the origin and protective function of alkaloids in plants. Flora 1973, 162, 108–117. [Google Scholar] [CrossRef]
- Kitamura, Y.; Yamashita, R.; Miura, H.; Watanabe, M. Phloem transport of tropane and pyridine alkaloids in Duboisia myoporoides. J. Plant. Physiol. 1993, 142, 635–637. [Google Scholar] [CrossRef]
- Wangchuk, P. Plant alkaloids: Classification, isolation and drug development. In Medicinal Plants: Chemistry, Pharmacology and Therapeutic Applications; Swamy, M.K., Patra, J.K., Rudramurthy, G.R., Eds.; Taylor & Francis Ltd.: Boca Raton, FL, USA, 2019; pp. 131–137. [Google Scholar]
- Saunders, J.A.; O’Neill, N.R.; Romeo, J.T. Alkaloids chemistry and feeding specificity of insect herbivores. In Alkaloids: Chemical and Biological Perspectives; Pelletier, S.W., Ed.; Springer: New York, NY, USA, 1992; pp. 151–196. [Google Scholar]
- Bastias, D.A.; Martinez-Ghersa, M.A.; Ballare, C.L.; Gundel, P.E. Epichloe fungal endophytes and plant defenses: Not just alkaloids. Trends Plant. Sci. 2017, 22, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Bastias, D.A.; Gianoli, E.; Gundel, P.E. Fungal endophytes can eliminate the plant growth–defence trade- off. New Phytol. 2021, 230, 2105–2113. [Google Scholar] [CrossRef] [PubMed]
- McCulley, R.L.; Bush, L.P.; Carlisle, A.E.; Ji, H.; Nelson, J.A. Warming reduces tall fescue abundance but stimulates toxic alkaloid concentrations in transition zone pastures of the U.S. Front. Chem. 2014, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Waller, G.R.; Nowacki, E.K. Alkaloid Biology and Metabolism in Plants; Springer: Boston, MA, USA, 1978; pp. 85–119. [Google Scholar]
- Vetter, J. Plant cyanogenic glycosides. Toxicon 2000, 38, 11–36. [Google Scholar] [CrossRef]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [PubMed]
- Gleadow, R.M.; Moller, B.L. Cyanogenic glycosides: Synthesis, physiology, and phenotypic plasticity. Annu. Rev. Plant Biol. 2014, 65, 155–185. [Google Scholar] [CrossRef] [PubMed]
- Nyirenda, K.K. Toxicity potential of cyanogenic glycosides in edible plants. In Medical Toxicology; Erkekoglu, P., Ogawa, T., Eds.; IntechOpen Limited: London, UK, 2020; pp. 1–19. [Google Scholar] [CrossRef]
- Cho, H.J.; Do, B.K.; Shim, S.M.; Kwon, H.; Lee, D.H.; Nah, A.H.; Choi, Y.J.; Lee, S.Y. Determination of cyanogenic compounds in edible plants by ion chromatography. Toxicol. Res. 2013, 29, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Gleadow, R.M.; Woodrow, I.E. Temporal and spatial variation in cyanogenic glycosides in Eucalyptus cladocalyx. Tree Physiol. 2000, 20, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.E.; Jensen, R.; Woodrow, I.E. Frequency of cyanogenesis in tropical rainforests of far north Queensland, Australia. Ann. Bot. 2006, 97, 1017–1044. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.C.; Sorensen, M.; Veiga, T.A.M.; Zibrandtsen, J.F.S.; Heskes, A.M.; Olsen, C.E.; Boughton, B.A.; Moller, B.L.; Neilson, E.H.J. Reconfigured Cyanogenic Glucoside Biosynthesis in Eucalyptus cladocalyx Involves a Cytochrome P450 CYP706C55. Plant Physiol. 2018, 178, 1081–1095. [Google Scholar] [CrossRef]
- Yulvianti, M.; Zidorn, C. Chemical Diversity of Plant Cyanogenic Glycosides: An Overview of Reported Natural Products. Molecules 2021, 26, 719. [Google Scholar] [CrossRef] [PubMed]
- Vickers, C.E.; Gershenzon, J.; Lerdau, M.T.; Loreto, F. A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat. Chem. Biol. 2009, 5, 283–291. [Google Scholar] [CrossRef]
- Wright, G.A.; Schiestl, F.P. The evolution of floral scent: The influence of olfactory learning by insect pollinators on the honest signalling of floral rewards. Funct. Ecol. 2009, 23, 841–851. [Google Scholar] [CrossRef]
- Munne-Bosch, S.; Falk, J. New insights into the function of tocopherols in plants. Planta 2004, 218, 323–326. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Alegre, L. The function of tocopherols and tocotrienols in plants. Crit. Rev. Plant Sci. 2010, 21, 31–57. [Google Scholar] [CrossRef]
- Yeshi, K.; Wangchuk, P. Essential oils and their bioactive molecules in healthcare. 1st ed. In Herbal Biomolecules in Healthcare Applications; Mandal, S.C., Nayak, A.K., Dhara, A.K., Eds.; Academic Press: London, UK, 2021; pp. 215–237. [Google Scholar]
- Peñuelas, J.; Llusià, J. The complexity of factors driving volatile organic compound emissions by plants. Biol. Plant 2001, 44, 481–487. [Google Scholar] [CrossRef]
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Janas, K.M.; Cvikrová, M.; Pałagiewicz, A.; Szafranska, K.; Posmyk, M.M. Constitutive elevated accumulation of phenylpropanoids in soybean roots at low temperature. Plant Sci. 2002, 163, 369–373. [Google Scholar] [CrossRef]
- Ono, M.; Takeshima, M.; Nishi, A.; Higuchi, T.; Nakano, S. Genistein suppresses v-Src-driven proliferative activity by arresting the cell-cycle at G2/M through increasing p21 level in Src-activated human gallbladder carcinoma cells. Nutr. Cancer 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.J.; Kim, G.H. Antiproliferative activity of daidzein and genistein may be related to ERalpha/c-erbB-2 expression in human breast cancer cells. Mol. Med. Rep. 2013, 7, 781–784. [Google Scholar] [CrossRef]
- Copolovici, L.; Kannaste, A.; Pazouki, L.; Niinemets, U. Emissions of green leaf volatiles and terpenoids from Solanum lycopersicum are quantitatively related to the severity of cold and heat shock treatments. J. Plant. Physiol. 2012, 169, 664–672. [Google Scholar] [CrossRef]
- Dutta, A.; Sen, J.; Deswal, R. Downregulation of terpenoid indole alkaloid biosynthetic pathway by low temperature and cloning of a AP2 type C-repeat binding factor (CBF) from Catharanthus roseus (L). G. Don. Plant Cell Rep. 2007, 26, 1869–1878. [Google Scholar] [CrossRef]
- Goboza, M.; Aboua, Y.G.; Chegou, N.; Oguntibeju, O.O. Vindoline effectively ameliorated diabetes-induced hepatotoxicity by docking oxidative stress, inflammation and hypertriglyceridemia in type 2 diabetes-induced male Wistar rats. Biomed. Pharmacother 2019, 112, 1–11. [Google Scholar] [CrossRef]
- Kubo, A.; Lunde, C.S.; Kubo, I. Indole and (E)-2-hexenal, phytochemical potentiators of polymyxins against Pseudomonas aeruginosa and Escherichia coli. Antimicrob. Agents Chemother. 1996, 40, 1438–1441. [Google Scholar] [CrossRef]
- Wang, X.-S.; Yang, W.; Tao, S.-J.; Li, K.; Li, M.; Dong, J.-H.; Wang, M.-W. The effect of delta-elemene on hela cell lines by apoptosis induction. Yakugaku Zasshi 2006, 126, 979–990. [Google Scholar] [CrossRef]
- Legault, J.; Pichette, A. Potentiating effect of beta-caryophyllene on anticancer activity of alpha-humulene, isocaryophyllene and paclitaxel. J. Pharm. Pharmacol. 2007, 59, 1643–1647. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Khadeer Ahamed, M.B.; Abdul Majid, A.M.S. In vivo anti-inflammatory activity of β-caryophyllene, evaluated by molecular imaging. Molecules Med. Chem. 2016, 162, 108–117. [Google Scholar] [CrossRef]
- Christie, P.J.; Alfenito, M.R.; Walbot, V. Impact of low-temperature stress on general phenylpropanoid and anthocyanin pathways: Enhancement of transcript abundance and anthocyanin pigmentation in maize seedlings. Planta 1994, 194, 541–549. [Google Scholar] [CrossRef]
- Ku, S.K.; Yoon, E.K.; Lee, W.; Kwon, S.; Lee, T.; Bae, J.S. Antithrombotic and antiplatelet activities of pelargonidin in vivo and in vitro. Arch. Pharm. Res. 2016, 39, 398–408. [Google Scholar] [CrossRef]
- Jeon, J.; Kim, J.K.; Wu, Q.; Park, S.U. Effects of cold stress on transcripts and metabolites in tartary buckwheat (Fagopyrum tataricum). Environ. Exp. Bot. 2018, 155, 488–496. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Nurulhuda, M.H.; Ismail, A.; Abas, F.; Hamid, M.; Roowi, S. Antioxidative and cardioprotective properties of anthocyanins from defatted dabai extracts. Evid. Based Complement. Alternat. Med. 2013, 2013, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mir, B.A.; Mir, S.A.; Khazir, J.; Tonfack, L.B.; Cowan, D.A.; Vyas, D.; Koul, S. Cold stress affects antioxidative response and accumulation of medicinally important withanolides in Withania somnifera (L.) Dunal. Ind. Crops. Prod. 2015, 74, 1008–1016. [Google Scholar] [CrossRef]
- Akhoon, B.A.; Pandey, S.; Tiwari, S.; Pandey, R. Withanolide A offers neuroprotection, ameliorates stress resistance and prolongs the life expectancy of Caenorhabditis elegans. Exp. Gerontol. 2016, 78, 47–56. [Google Scholar] [CrossRef]
- Hahm, E.R.; Moura, M.B.; Kelley, E.E.; Van Houten, B.; Shiva, S.; Singh, S.V. Withaferin A-induced apoptosis in human breast cancer cells is mediated by reactive oxygen species. PLoS ONE 2011, 6, 1–12. [Google Scholar] [CrossRef]
- Zhao, M.; Zhang, N.; Gao, T.; Jin, J.; Jing, T.; Wang, J.; Wu, Y.; Wan, X.; Schwab, W.; Song, C. Sesquiterpene glucosylation mediated by glucosyltransferase UGT91Q2 is involved in the modulation of cold stress tolerance in tea plants. New Phytol. 2020, 226, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Sarker, U.; Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant. Biol. 2018, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Akojie, F.O.; Fung, L.W. Antisickling activity of hydroxybenzoic acids in Cajanus cajan. Planta Med. 1992, 58, 317–320. [Google Scholar] [CrossRef]
- Hernandez, I.; Alegre, L.; Munne-Bosch, S. Enhanced oxidation of flavan-3-ols and proanthocyanidin accumulation in water-stressed tea plants. Phytochemistry 2006, 67, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Anitha, S.; Krishnan, S.; Senthilkumar, K.; Sasirekha, V. Theoretical investigation on the structure and antioxidant activity of (+) catechin and (−) epicatechin—A comparative study. Mol. Phys. 2020, 118, 1–12. [Google Scholar] [CrossRef]
- Liu, Z. Drought-induced in vivo synthesis of camptothecin in Camptotheca acuminata seedlings. Physiol. Plant 2000, 110, 483–488. [Google Scholar] [CrossRef]
- Adams, D.J.; Dewhirst, M.W.; Flowers, J.L.; Gamcsik, M.P.; Colvin, O.M.; Manikumar, G.; Wani, M.C.; Wall, M.E. Camptothecin analogues with enhanced antitumor activity at acidic pH. Cancer Chemother. Pharmacol. 2000, 46, 263–271. [Google Scholar] [CrossRef]
- Liu, Y.; Meng, Q.; Duan, X.; Zhang, Z.; Li, D. Effects of PEG-induced drought stress on regulation of indole alkaloid biosynthesis in Catharanthus roseus. J. Plant Interact. 2017, 12, 87–91. [Google Scholar] [CrossRef]
- Zhou, X.; Xu, Z.; Li, A.; Zhang, Z.; Xu, S. Double-sides sticking mechanism of vinblastine interacting with alpha, beta-tubulin to get activity against cancer cells. J. Biomol. Struct. Dyn. 2019, 37, 4080–4091. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, I.; Alegre, L.; Munne-Bosch, S. Drought-induced changes in flavonoids and other low molecular weight antioxidants in Cistus clusii grown under Mediterranean field conditions. Tree Physiol. 2004, 24, 1303–1311. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.D.; Pagidas, K. Epigallocatechin-3-gallate, a natural polyphenol, inhibits cell proliferation and induces apoptosis in human ovarian cancer Cclls. Anticancer Res. 2010, 30, 2519–2524. [Google Scholar]
- Falcinelli, S.D.; Shi, M.C.; Friedlander, A.M.; Chua, J. Green tea and epigallocatechin-3-gallate are bactericidal against Bacillus anthracis. FEMS Microbiol. Lett. 2017, 364, 1–8. [Google Scholar] [CrossRef][Green Version]
- Kirakosyan, A.; Kaufman, P.; Warber, S.; Zick, S.; Aaronson, K.; Bolling, S.; Chul Chang, S. Applied environmental stresses to enhance the levels of polyphenolics in leaves of hawthorn plants. Physiol. Plant 2004, 121, 182–186. [Google Scholar] [CrossRef]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm. 2011, 403, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.Z.; Yeung, S.Y.; Chang, Q.; Huang, Y.; Chen, Z.Y. Comparison of antioxidant activity and bioavailability of tea epicatechins with their epimers. Br. J. Nutr. 2004, 91, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Njiti, V.N.; Lightfoot, D.A.; Wood, A.J. Trigonelline concentration in field-grown soybean in response to irrigation. Biol. Plant 2003, 46, 405–410. [Google Scholar] [CrossRef]
- Yoshinari, O.; Sato, H.; Igarashi, K. Anti-diabetic effects of pumpkin and its components, trigonelline and nicotinic acid, on Goto-Kakizaki rats. Biosci. Biotechnol. Biochem. 2009, 73, 1033–1041. [Google Scholar] [CrossRef] [PubMed]
- Nacif de Abreu, I.; Mazzafera, P. Effect of water and temperature stress on the content of active constituents of Hypericum brasiliense Choisy. Plant Physiol. Biochem. 2005, 43, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Bridi, H.; Dischkaln Stolz, E.; Maikon Corrêa de Barros, F.; Elingson da Silva Costa, B.; Guerini, L.; Maris Kuze Rates, S.; Lino von Poser, G. Antinociceptive activity of phloroglucinol derivatives isolated from southern Brazilian Hypericum species. Chem. Biodivers 2018, 15, 66–87. [Google Scholar] [CrossRef]
- Lin, J.P.; Yang, J.S.; Lin, J.J.; Lai, K.C.; Lu, H.F.; Ma, C.Y.; Sai-Chuen Wu, R.; Wu, K.C.; Chueh, F.S.; Gibson Wood, W.; et al. Rutin inhibits human leukemia tumor growth in a murine xenograft model in vivo. Environ. Toxicol. 2012, 27, 480–484. [Google Scholar] [CrossRef]
- Zuco, V.; Supino, R.; Righetti, S.C.; Cleris, L.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar] [CrossRef]
- Christiansen, J.L.; Jornsgard, B.; Buskov, S.; Oslen, C.E. Effect of drought stress on content and composition of seed alkaloids in narrow-leafed lupin, Lupinus angustifolius L. Eur. J. Agron. 1997, 7, 307–314. [Google Scholar] [CrossRef]
- Szabó, B.; Tyihák, E.; Szabó, G.; Botz, L. Mycotoxin and drought stress induced change of alkaloid content of Papaver somniferum plantlets. Acta. Bot. Hung. 2003, 45, 409–417. [Google Scholar] [CrossRef]
- Stein, C.; Comisel, K.; Haimerl, E.; Yassouridis, A.; Lehrberger, K.; Herz, A.; Peter, K. Analgesic effect of intraarticular morphine after arthroscopic knee surgery. N. Engl. J. Med. 1991, 325, 1123–1126. [Google Scholar] [CrossRef]
- Clark, E.; Plint, A.C.; Correll, R.; Gaboury, I.; Passi, B. A randomized, controlled trial of acetaminophen, ibuprofen, and codeine for acute pain relief in children with musculoskeletal trauma. Pediatrics 2007, 119, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Turtola, S.; Manninen, A.M.; Rikala, R.; Kainulainen, P. Drought stress alters the concentration of wood terpenoids in Scots pine and Norway spruce seedlings. J. Chem. Ecol. 2003, 29, 1981–1995. [Google Scholar] [CrossRef]
- Ulusu, N.N.; Ercil, D.; Sakar, M.K.; Tezcan, E.F. Abietic acid inhibits lipoxygenase activity. Phytother Res. 2002, 16, 88–90. [Google Scholar] [CrossRef]
- Fernandez, M.A.; Tornos, M.P.; GarcIa, M.D.; de las Heras, B.; Villar, A.M.; Saenz, M.T. Anti-inflammatory activity of abietic acid, a diterpene isolated from Pimenta racemosa var. grissea. J. Pharm. Pharmacol. 2001, 53, 867–872. [Google Scholar] [CrossRef]
- Liu, H.; Wang, X.; Wang, D.; Zou, Z.; Liang, Z. Effect of drought stress on growth and ccumulation of active constituents in Salvia miltiorrhiza Bunge. Ind. Crops. Prod. 2011, 33, 84–88. [Google Scholar] [CrossRef]
- Chen, W.; Lu, Y.; Chen, G.; Huang, S. Molecular evidence of cryptotanshinone for treatment and prevention of human cancer. Anticancer Agents Med. Chem. 2013, 13, 979–987. [Google Scholar] [CrossRef]
- Adomako-Bonsu, A.G.; Chan, S.L.F.; Pratten, M.; Fry, J.R. Antioxidant activity of rosmarinic acid and its principal metabolites in chemical and cellular systems: Importance of physico-chemical characteristics. Toxicol. In Vitro 2017, 40, 248–255. [Google Scholar] [CrossRef]
- Sun, Y.; Zhu, H.; Wang, J.; Liu, Z.; Bi, J. Isolation and purification of salvianolic acid A and salvianolic acid B from Salvia miltiorrhiza by high-speed counter-current chromatography and comparison of their antioxidant activity. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2009, 877, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.H.; Du, F.; Liu, H.Y.; Liang, Z.S. Drought stress increases iridoid glycosides biosynthesis in the roots of Scrophularia ningpoensis seedlings. J. Med. Plant. Res. 2010, 4, 2691–2699. [Google Scholar]
- Liu, L.; Cao, X.; Li, T.; Li, X. Effects of catalpol on the activity of human liver cytochrome P450 enzymes. Xenobiotica 2019, 49, 1289–1295. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Han, M.; Kim Kwon, Y. Effect of aucubin on neural precursor cell survival during neuronal differentiation. Int. J. Neurosci. 2018, 128, 899–905. [Google Scholar] [CrossRef]
- Marquardt, D.; Williams, J.A.; Kucerka, N.; Atkinson, J.; Wassall, S.R.; Katsaras, J.; Harroun, T.A. Tocopherol activity correlates with its location in a membrane: A new perspective on the antioxidant vitamin E. J. Am. Chem Soc. 2013, 135, 7523–7533. [Google Scholar] [CrossRef]
- Kawata, A.; Murakami, Y.; Suzuki, S.; Fujisawa, S. Anti-inflammatory activity of beta-carotene, lycopene and tri-n-butylborane, a scavenger of reactive oxygen species. In Vivo 2018, 32, 255–264. [Google Scholar] [CrossRef]
- Rufino, A.T.; Ribeiro, M.; Judas, F.; Salgueiro, L.; Lopes, M.C.; Cavaleiro, C.; Mendes, A.F. Anti-inflammatory and chondroprotective activity of (+)-alpha-pinene: Structural and enantiomeric selectivity. J. Nat. Prod. 2014, 77, 264–269. [Google Scholar] [CrossRef]
- Du, Y.; Luan, J.; Jiang, R.P.; Liu, J.; Ma, Y. Myrcene exerts anti-asthmatic activity in neonatal rats via modulating the matrix remodeling. Int. J. Immunopathol. Pharmacol. 2020, 34, 1–10. [Google Scholar] [CrossRef]
- Roberto, D.; Micucci, P.; Sebastian, T.; Graciela, F.; Anesini, C. Antioxidant activity of limonene on normal murine lymphocytes: Relation to H2O2 modulation and cell proliferation. Basic. Clin. Pharmacol. Toxicol. 2010, 106, 38–44. [Google Scholar] [CrossRef]
- Fernandes, E.S.; Passos, G.F.; Medeiros, R.; da Cunha, F.M.; Ferreira, J.; Campos, M.M.; Pianowski, L.F.; Calixto, J.B. Anti-inflammatory effects of compounds alpha-humulene and (-)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur. J. Pharmacol. 2007, 569, 228–236. [Google Scholar] [CrossRef]
- He, X.; Huang, W.; Chen, W.; Dong, T.; Liu, C.; Chen, Z.; Xu, S.; Ruan, Y. Changes of main secondary metabolites in leaves of Ginkgo biloba in response to ozone fumigation. J. Environ. Sci. 2009, 21, 199–203. [Google Scholar] [CrossRef]
- Feng, Z.; Sun, Q.; Chen, W.; Bai, Y.; Hu, D.; Xie, X. The neuroprotective mechanisms of ginkgolides and bilobalide in cerebral ischemic injury: A literature review. Mol. Med. 2019, 25, 1–8. [Google Scholar] [CrossRef]
- Ormrod, D.P.; Landry, L.G.; Coclin, P.L. Short-term UV-B radiation and ozone exposure effects on aromatic secondary metabolite accumulation and shoot growth of flavonoid-deficient Arabidopsis mutants. Physiol. Plant 1995, 93, 602–610. [Google Scholar] [CrossRef]
- Olsson, L.C.; Veit, M.; Weissenbock, G.; Bornman, J.F. Flavonoid response to UV-B radiation in Brassica napus. Phytochemistry 1998, 49, 1021–1028. [Google Scholar] [CrossRef]
- Gitz, D.C.; Liu, L.; McClure, J.W. Phenolic metabolism, growth and UV-B tolerance in phenylalanine ammonia lyase inhibited red cabbage seedlings. Phytochemistry 1998, 49, 377–386. [Google Scholar] [CrossRef]
- Ramani, S.; Jayabaskaran, C. Enhanced catharanthine and vindoline production in suspension cultures of Catharanthus roseus by ultraviolet-B light. J. Mol. Signal. 2008, 3, 1–6. [Google Scholar] [CrossRef]
- Ramani, S.; Chelliah, J. UV-B-induced signaling events leading to enhanced-production of catharanthine in Catharanthus roseus cell suspension cultures. BMC Plant. Biol. 2007, 7, 1–17. [Google Scholar] [CrossRef]
- Siddiqui, M.J.; Ismail, Z.; Aisha, A.F.A.; Abdul Majid, A.M.S. Cytotoxic activity of Catharanthus roseus (Apocynaceae) crude extracts and pure compounds against human colorectal carcinoma cell line. Int. J. Pharmacol. 2010, 6, 43–47. [Google Scholar] [CrossRef]
- Wang, J.; Dudareva, N.; Bhakta, S.; Raguso, R.A.; Pichersky, E. Floral scent production in Clarkia breweri (Onagraceae). II. Localization and developmental modulation of the enzyme SAM:(Iso)eugenol O-methyltransferase and phenylpropanoid emission. Plant. Physiol. 1997, 114, 213–221. [Google Scholar] [CrossRef]
- Campaniello, D.; Corbo, M.R.; Sinigaglia, M. Antifungal activity of eugenol against Penicillium, Aspergillus, and Fusarium species. J. Food Prot. 2010, 73, 1124–1128. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Hung, S.L.; Pai, S.F.; Lee, Y.H.; Yang, S.F. Eugenol suppressed the expression of lipopolysaccharide-induced proinflammatory mediators in human macrophages. J. Endod. 2007, 33, 698–702. [Google Scholar] [CrossRef]
- Regvar, M.; Bukovnik, U.; Likar, M.; Kreft, I. UV-B radiation affects flavonoids and fungal colonisation in Fagopyrum esculentum and F. tataricum. Open Life Sci. 2012, 7, 275–283. [Google Scholar] [CrossRef]
- Zheng, Y.Z.; Deng, G.; Liang, Q.; Chen, D.F.; Guo, R.; Lai, R.C. Antioxidant activity of quercetin and its glucosides from Propolis: A theoretical study. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Sun, H.; Yin, M.; Hao, D.; Shen, Y. Anti-Cancer Activity of Catechin against A549 Lung Carcinoma Cells by Induction of Cyclin Kinase Inhibitor p21 and Suppression of Cyclin E1 and P–AKT. Appl. Sci. 2020, 10, 2065. [Google Scholar] [CrossRef]
- Simos, Y.V.; Verginadis, I.I.; Toliopoulos, I.K.; Velalopoulou, A.P.; Karagounis, I.V.; Karkabounas, S.C.; Evangelou, A.M. Effects of catechin and epicatechin on superoxide dismutase and glutathione peroxidase activity, in vivo. Redox. Rep. 2012, 17, 181–186. [Google Scholar] [CrossRef]
- Cuadra, P.; Harborne, J.B.; Waterman, P.G. Increases in surface flavonols and photosynthetic pigments in Gnaphalium luteo-album in response to UV-B radiation. Phytochemistry 1997, 45, 1377–1383. [Google Scholar] [CrossRef]
- Faham, N.; Javidnia, K.; Bahmani, M.; Amirghofran, Z. Calycopterin, an immunoinhibitory compound from the extract of Dracocephalum kotschyi. Phytother. Res. 2008, 22, 1154–1158. [Google Scholar] [CrossRef]
- Cuadra, P.; Harborne, J.B. Changes in epicuticular flavonoids and photosynthetic pigments as a plant response to UV-B radiation. Z. Naturforsch. 1996, 51c, 671–680. [Google Scholar] [CrossRef]
- Reuber, S.; Bornman, J.F.; Weissenbock, G. A flavonoid mutant of barley exhibits increased sensitivity to UV-B radiation in primary leaves. Plant Cell Environ. 1996, 19, 593–599. [Google Scholar] [CrossRef]
- Ra, J.-E.; Woo, S.-Y.; Jin, H.; Lee, M.J.; Kim, H.Y.; Ham, H.; Chung, I.-M.; Seo, W.D. Evaluation of antihypertensive polyphenols of barley (Hordeum vulgare L.) seedlings via their effects on angiotensin-converting enzyme (ACE) inhibition. Appl. Biol. Chem. 2020, 63, 1–9. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, Y.; Zhang, Z.; Chen, M.; Zhang, D.; Tian, C.; Liu, M.; Jiang, G. The antibacterial activity and mechanism of action of luteolin against Trueperella pyogenes. Infect. Drug Resist. 2020, 13, 1697–1711. [Google Scholar] [CrossRef] [PubMed]
- Markham, K.R.; Ryan, K.G.; Bloor, S.J.; Mitchell, K.A. An increase in the luteolin-apigenin ratio in Marchantia polymorpha on UV-B enhancement. Phytochemistry 1998, 48, 791–794. [Google Scholar] [CrossRef]
- Skaltsa, H.; Verykokidou, E.; Harvala, C.; Karabourniotis, G.; Manetas, Y. UV-B protective potential and flavonoid content of leaf hairs in Quercus ilex. Phytochemistry 1994, 37, 987–990. [Google Scholar] [CrossRef]
- Lee, K.M.; Lee, K.W.; Jung, S.K.; Lee, E.J.; Heo, Y.S.; Bode, A.M.; Lubet, R.A.; Lee, H.J.; Dong, Z. Kaempferol inhibits UVB-induced COX-2 expression by suppressing Src kinase activity. Biochem. Pharmacol. 2010, 80, 2042–2049. [Google Scholar] [CrossRef]
- Kadioglu, O.; Nass, J.; Saeed, M.E.M.; Schuler, B.; Efferth, T. Kaempferol is an anti-inflammatory compound with activity towards NF-κB pathway proteins. Anticancer Res. 2015, 35, 2645–2650. [Google Scholar]
- Zu, Y.G.; Tang, Z.H.; Yu, J.H.; Liu, S.G.; Wang, W.; Guo, X.R. Different responses of camptothecin and 10-hydroxycamptothecin to heat shock in Camptotheca acuminata seedlings. Acta. Bot. Sin. 2003, 45, 809–814. [Google Scholar]
- Ping, Y.-H.; Lee, H.-C.; Lee, J.-Y.; Wu, P.-H.; Ho, L.-K.; Chi, C.-W.; Lu, M.-F.; Wang, J.-J. Anticancer effects of low-dose 10-hydroxycamptothecin in human colon cancer. Oncol. Rep. 2006, 15, 1273–1279. [Google Scholar] [CrossRef]
- Rosenfeld, H.J.; Aaby, K.; Lea, P. Influence of temperature and plant density on sensory quality and volatile terpenoids of carrot (Daucus carota L.) root. J. Sci. Food Agric. 2002, 82, 1384–1390. [Google Scholar] [CrossRef]
- Helmig, D.; Ortega, J.; Duhl, T.; Tanner, D.; Guenther, A.; Harley, P.; Wiedinmyer, C.; Milford, J.; Sakulyanontvittaya, T. Sesquiterpene emissions from pine trees-identifications, emission rates and flux estimates for the contiguous United States. Environ. Sci. Technol. 2007, 41, 1545–1553. [Google Scholar] [CrossRef]
- Commisso, M.; Toffali, K.; Strazzer, P.; Stocchero, M.; Ceoldo, S.; Baldan, B.; Levi, M.; Guzzo, F. Impact of phenylpropanoid compounds on heat stress tolerance in carrot cell cultures. Front. Plant Sci. 2016, 7, 1–16. [Google Scholar] [CrossRef]
- Aydin, E.; Turkez, H.; Tasdemir, S. Anticancer and antioxidant properties of terpinolene in rat brain cells. Arh. Hig. Rada. Toksikol. 2013, 64, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Song, X.; Li, L.; Sun, J.; Jaiswal, Y.; Huang, J.; Liu, C.; Yang, W.; Williams, L.; Zhang, H.; et al. Protective effects of p-coumaric acid against oxidant and hyperlipidemia-an in vitro and in vivo evaluation. Biomed. Pharmacother. 2019, 111, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Spagnol, C.M.; Assis, R.P.; Brunetti, I.L.; Isaac, V.L.B.; Salgado, H.R.N.; Correa, M.A. In vitro methods to determine the antioxidant activity of caffeic acid. Spectrochim Acta A Mol. Biomol. Spectrosc. 2019, 219, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Hanson, D.T.; Sharkey, T.D. Effect of growth conditions on isoprene emission and other thermotolerance-enhancing compounds. Plant Cell Environ. 2001, 24, 929–936. [Google Scholar] [CrossRef]
- Zhang, J.H.; Sun, H.L.; Chen, S.Y.; Zeng, L.; Wang, T.T. Anti-fungal activity, mechanism studies on alpha-phellandrene and nonanal against Penicillium cyclopium. Bot. Stud. 2017, 58, 1–9. [Google Scholar] [CrossRef]
- Hadri, A.; Gomez del Rio, M.A.; Sanz, J.; Coloma, A.G.; Idaomar, M.; Ozonas, B.R.; Gonzalez, J.B.; Reus, M.I.S. Cytotoxic activity of α-humulene and trans-caryophyllene from Salvia officinalis in animal and human tumor cells. An. R. Acad. Nac. Farm. 2010, 76, 343–356. [Google Scholar]
- Randriamampionona, D.; Diallo, B.; Rakotoniriana, F.; Rabemanantsoa, C.; Cheuk, K.; Corbisier, A.M.; Jaziri, M. Comparative analysis of active constituents in Centella asiatica samples from Madagascar: Application for ex situ conservation and clonal propagation. Fitoterapia 2007, 78, 482–489. [Google Scholar] [CrossRef]
- Sondari, D.; Harmami, S.B.; Ghozali, M.; Randy, A.; Amanda, A.; Irawan, Y. Determination of the active asiaticoside content in Centella asiatica as anti-cellulite agent. Indones J. Cancer Chemoprev. 2011, 2, 222–227. [Google Scholar] [CrossRef]
- Sewelam, N.; Kazan, K.; Schenk, P.M. Global plant stress signaling: Reactive oxygen species at the cross-road. Front. Plant. Sci. 2016, 7, 1–21. [Google Scholar] [CrossRef]
- Nogués, I.; Medori, M.; Calfapietra, C. Limitations of monoterpene emissions and their antioxidant role in Cistus sp. under mild and severe treatments of drought and warming. Environ. Exp. Bot. 2015, 119, 76–86. [Google Scholar] [CrossRef]
- Fall, R.; Karl, T.; Hansel, A.; Jordan, A.; Lindinger, W. Volatile organic compounds emitted after leaf wounding: On-line analysis by proton-transfer-reaction mass spectrometry. J. Geophys. Res. 1999, 104, 15963–15974. [Google Scholar] [CrossRef]
- D’Auria, J.C.; Pichersky, E.; Schaub, A.; Hansel, A.; Gershenzon, J. Characterization of a BAHD acyltransferase responsible for producing the green leaf volatile (Z)-3-hexen-1-yl acetate in Arabidopsis thaliana. Plant J. 2007, 49, 194–207. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Kunishima, M.; Mizutani, M.; Sugimoto, Y. Reactive short-chain leaf volatiles act as powerful inducers of abiotic stress-related gene expression. Sci. Rep. 2015, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, A.G. Lipid melting and cuticular permeability: New insights into an old problem. J. Insect Physiol. 2002, 48, 391–400. [Google Scholar] [CrossRef]
- Boncan, D.A.T.; Tsang, S.S.K.; Li, C.; Lee, I.H.T.; Lam, H.M.; Chan, T.F.; Hui, J.H.L. Terpenes and terpenoids in plants: Interactions with environment and insects. Int. J. Mol. Sci. 2020, 21, 7382. [Google Scholar] [CrossRef] [PubMed]
- Korankye, E.A.; Lada, R.R.; Asiedu, S.; Caldwell, C. Plant senescence: The role of volatile terpene compounds (VTCs). Am. J. Plant. Sci. 2017, 8, 3120–3139. [Google Scholar] [CrossRef]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B.; Rowshan, V.; Niazi, A.; Moghadam, A. Effect of salt stress on terpenoid biosynthesis in Salvia mirzayanii: From gene to metabolite. J. Hortic. Sci. Biotechnol. 2018, 94, 389–399. [Google Scholar] [CrossRef]
- Spicher, L.; Glauser, G.; Kessler, F. Lipid antioxidant and galactolipid remodeling under temperature stress in tomato plants. Front Plant Sci. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Sakuragi, Y.; Maeda, H.; Dellapenna, D.; Bryant, D.A. alpha-tocopherol plays a role in photosynthesis and macronutrient homeostasis of the cyanobacterium Synechocystis sp. PCC 6803 that is independent of its antioxidant function. Plant Physiol. 2006, 141, 508–521. [Google Scholar] [CrossRef]
- Havaux, M. Plastoquinone in and beyond photosynthesis. Trends Plant Sci. 2020, 25, 1252–1265. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Chen, X.Y.; Yeh, S. Isoprene increases thermotolerance of fosmidomycin-fed leaves. Plant Physiol. 2001, 125, 2001–2006. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Wiberley, A.E.; Donohue, A.R. Isoprene emission from plants: Why and how. Ann. Bot. 2008, 101, 5–18. [Google Scholar] [CrossRef]
- Loreto, F.; Velikova, V. Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol. 2001, 127, 1781–1787. [Google Scholar] [CrossRef]
- Rinnan, R.; Rinnan, Å.; Faubert, P.; Tiiva, P.; Holopainen, J.K.; Michelsen, A. Few long-term effects of simulated climate change on volatile organic compound emissions and leaf chemistry of three subarctic dwarf shrubs. Environ. Exp. Bot. 2011, 72, 377–386. [Google Scholar] [CrossRef]
- Harvaux, M.; Kloppstech, K. The protective functions of carotenoid and flavonoid pigments against excess visible radiation at chilling temperature investigated in Arabidopsis npq and tt mutants. Planta 2001, 213, 953–966. [Google Scholar] [CrossRef] [PubMed]
- Camejo, D.; Jimenez, A.; Alarcon, J.J.; Torres, W.; Gomez, J.M.; Sevilla, F. Changes in photosynthetic parameters and antioxidant activities following heat-shock treatment in tomato plants. Funct. Plant Biol. 2006, 33, 177–187. [Google Scholar] [CrossRef]
- Al-Huqail, A.; El-Dakak, R.M.; Sanad, M.N.; Badr, R.H.; Ibrahim, M.M.; Soliman, D.; Khan, F. Effects of climate temperature and water stress on plant growth and accumulation of antioxidant compounds in Sweet Basil (Ocimum basilicum L.) leafy vegetable. Scientifica 2020, 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Thakur, P.; Nayyar, H. Facing the Cold Stress by Plants in the Changing Environment: Sensing, Signaling, and Defending Mechanisms. In Plant. Acclimation to Environmental Stress; Tuteja, N., Singh, G.S., Eds.; Springer: New York, NY, USA, 2013; pp. 29–69. [Google Scholar]
- Chinnusamy, V.; Zhu, J.; Zhu, J.K. Cold stress regulation of gene expression in plants. Trends Plant Sci. 2007, 12, 444–451. [Google Scholar] [CrossRef]
- Krol, A.; Amarowicz, R.; Weidner, S. The effects of cold stress on the phenolic compounds and antioxidant capacity of grapevine (Vitis vinifera L.) leaves. J. Plant Physiol. 2015, 189, 97–104. [Google Scholar] [CrossRef]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef]
- Ahmed, S.; Griffin, T.S.; Kraner, D.; Schaffner, M.K.; Sharma, D.; Hazel, M.; Leitch, A.R.; Orians, C.M.; Han, W.; Stepp, J.R.; et al. Environmental Factors Variably Impact Tea Secondary Metabolites in the Context of Climate Change. Front. Plant. Sci. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Dat, J.; Vandenabeele, S.; Vranova, E.; Van Montagu, M.; Inze, D.; Van Breusegem, F. Dual action of the active oxygen species during plant stress responses. Cell Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Overmyer, K.; Brosche, M.; Kangasjarvi, J. Reactive oxygen species and hormonal control of cell death. Trends Plant Sci. 2003, 8, 335–342. [Google Scholar] [CrossRef]
- Nakabayashi, R.; Mori, T.; Saito, K. Alternation of flavonoid accumulation under drought stress in Arabidopsis thaliana. Plant Signal. Behav. 2014, 9, 1–3. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant. Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Massacci, A.; Nabiev, S.M.; Pietrosanti, L.; Nematov, S.K.; Chernikova, T.N.; Thor, K.; Leipner, J. Response of the photosynthetic apparatus of cotton (Gossypium hirsutum) to the onset of drought stress under field conditions studied by gas-exchange analysis and chlorophyll fluorescence imaging. Plant Physiol. Biochem. 2008, 46, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, R.; Yonekura-Sakakibara, K.; Urano, K.; Suzuki, M.; Yamada, Y.; Nishizawa, T.; Matsuda, F.; Kojima, M.; Sakakibara, H.; Shinozaki, K.; et al. Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J. 2014, 77, 367–379. [Google Scholar] [CrossRef]
- Ma, D.; Sun, D.; Wang, C.; Li, Y.; Guo, T. Expression of flavonoid biosynthesis genes and accumulation of flavonoid in wheat leaves in response to drought stress. Plant Physiol. Biochem. 2014, 80, 60–66. [Google Scholar] [CrossRef]
- Von Wettberg, E.J.; Stanton, M.L.; Whittall, J.B. How anthocyanin mutants respond to stress: The need to distinguish between stress tolerance and maximal vigour. Evol. Ecol. Res. 2010, 12, 457–476. [Google Scholar]
- Li, X.; Lv, X.; Wang, X.; Wang, L.; Zhang, M.; Ren, M. Effects of abiotic stress on anthocyanin accumulation and grain weight in purple wheat. Crop. Pasture. Sci. 2018, 69, 1208–1214. [Google Scholar] [CrossRef]
- Sun, J.; Qiu, C.; Ding, Y.; Wang, Y.; Sun, L.; Fan, K.; Gai, Z.; Dong, G.; Wang, J.; Li, X.; et al. Fulvic acid ameliorates drought stress-induced damage in tea plants by regulating the ascorbate metabolism and flavonoids biosynthesis. BMC Genom. 2020, 21, 1–13. [Google Scholar] [CrossRef]
- Muller-Xing, R.; Xing, Q.; Goodrich, J. Footprints of the sun: Memory of UV and light stress in plants. Front Plant Sci. 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Del Valle, J.C.; Buide, M.L.; Whittall, J.B.; Valladares, F.; Narbona, E. UV radiation increases phenolic compound protection but decreases reproduction in Silene littorea. PLoS ONE 2020, 15, 1–18. [Google Scholar] [CrossRef]
- Stapleton, A.E.; Walbot, V. Flavonoids can protect maize DNA from the induction of ultraviolet radiation damage. Plant Physiol. 1994, 105, 881–889. [Google Scholar] [CrossRef]
- Jordan, B.R. Molecular response of plant cells to UV-B stress. Funct. Plant Biol. 2002, 29, 909–916. [Google Scholar] [CrossRef]
- Agati, G.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Pollastri, S.; Tattini, M. Functional roles of flavonoids in photoprotection: New evidence, lessons from the past. Plant Physiol. Biochem. 2013, 72, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Tattini, M.; Gravano, E.; Pinelli, P.; Mullinacci, N.; Romani, A. Flavonoids accumulate in leaves and glandular trichomes of phillyrea latifolia exposed to excess solar radiation. New Phytol. 2000, 148, 69–77. [Google Scholar] [CrossRef]
- Tattini, M.; Galardi, C.; Pinelli, P.; Massai, R.; Remorini, D.; Agati, G. Differential accumulation of flavonoids and hydroxycinnamates in leaves of Ligustrum vulgare under excess light and drought stress. New Phytol. 2004, 163, 547–561. [Google Scholar] [CrossRef]
- Nascimento, L.; Leal-Costa, M.V.; Menezes, E.A.; Lopes, V.R.; Muzitano, M.F.; Costa, S.S.; Tavares, E.S. Ultraviolet-B radiation effects on phenolic profile and flavonoid content of Kalanchoe pinnata. J. Photochem. Photobiol. B 2015, 148, 73–81. [Google Scholar] [CrossRef]
- Herrlich, P.; Blattner, C.; Knebel, A.; Bender, K.; Rahmsdorf, H.F. Nuclear and non-nuclear targets of genotoxic agents in the induction of gene expression. Shared principles in yeast, rodents, man and plants. Biol. Chem. 1997, 378, 1217–1229. [Google Scholar]
- Lindroth, R.L. Impacts of elevated atmospheric CO2 and O3 on forests: Phytochemistry, trophic interactions, and ecosystem dynamics. J. Chem. Ecol. 2010, 36, 2–21. [Google Scholar] [CrossRef]
- Iriti, M.; Faoro, F. Chemical diversity and defence metabolism: How plants cope with pathogens and ozone pollution. Int. J. Mol. Sci. 2009, 10, 3371–3399. [Google Scholar] [CrossRef]
- Hoshika, Y.; Omasa, K.; Paoletti, E. Both ozone exposure and soil water stress are able to induce stomatal sluggishness. Environ. Exp. Bot. 2013, 88, 19–23. [Google Scholar] [CrossRef]
- Mao, B.; Yin, H.; Wang, Y.; Zhao, T.H.; Tian, R.R.; Wang, W.; Ye, J.S. Combined effects of O3 and UV radiation on secondary metabolites and endogenous hormones of soybean leaves. PLoS ONE 2017, 12, 1–16. [Google Scholar] [CrossRef]
- Kangasj¨arvi, J.; Talvinen, J.; Utriaine, M.; Karjalainen, R. Plant defence systems induced by ozone. Plant. Cell Environ. 1994, 17, 783–794. [Google Scholar] [CrossRef]
- Sharma, Y.K.; Davis, K.R. Ozone-induced expression of stress-related genes in Arabidopsis thaliana. Plant Physiol. 1994, 105, 1089–1096. [Google Scholar] [CrossRef]
- Eckey-Kaltenbach, H.E.; Ernst, D.; Heller, W.; Sandermann, H. Biochemical plant response to ozone IV. Cross-induction of defensive pathways in parsley (Petroselinum crispum L.) plants. Plant Physiol. 1994, 104, 67–74. [Google Scholar] [CrossRef]
- Martin-Tanguy, J. Metabolism and function of polyamines in plants: Recent development (new approaches). Plant Growth Regul. 2001, 34, 135–148. [Google Scholar] [CrossRef]
- Marchica, A.; Cotrozzi, L.; Detti, R.; Lorenzini, G.; Pellegrini, E.; Petersen, M.; Nali, C. The biosynthesis of phenolic compounds is an integrated defence mechanism to prevent ozone injury in Salvia officinalis. Antioxidants 2020, 9, 1274. [Google Scholar] [CrossRef]
- Peñuelas, J.; Staudt, M. BVOCs and global change. Trends Plant Sci. 2010, 15, 133–144. [Google Scholar] [CrossRef]
- Valkama, E.; Koricheva, J.; Oksanen, E. Effects of elevated O3, alone and in combination with elevated CO2, on tree leaf chemistry and insect herbivore performance: A meta-analysis. Glob. Chang. Biol. 2007, 13, 184–201. [Google Scholar] [CrossRef]
- Rao, M.V.; Davis, K.R. The physiology of ozone-induced cell death. Planta 2001, 213, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Heiden, A.C.; Hoffmann, T.; Kahl, J.; Kley, D.; Klockow, D.; Langebartels, C.; Mehlhorn, H.; Sandermann, H.; Schraudner, M.; Schuh, G.; et al. Emission of volatile organic compounds from ozone-exposed plants. Ecolog. Appl. 1999, 9, 1160–1167. [Google Scholar] [CrossRef]
- Langebartels, C.; Kerner, K.; Leonardi, S.; Schraudner, M.; Trost, M.; Heller, W.; Sandermann, H. Biochemical plant resonses to ozone I: Differential induction of polyamine and ethylene biosynthesis in tobacco. Plant Physiol. 1991, 9, 882–889. [Google Scholar] [CrossRef]
- Piao, M.J.; Kim, K.C.; Chae, S.; Keum, Y.S.; Kim, H.S.; Hyun, J.W. Protective effect of fisetin (3,7,3’,4’-tetrahydroxyflavone) against gamma-irradiation-induced oxidative stress and cell damage. Biomol. Ther. 2013, 21, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Maher, P. A comparison of the neurotrophic activities of the flavonoid fisetin and some of its derivatives. Free Radic. Res. 2006, 40, 1105–1111. [Google Scholar] [CrossRef]
- Pinto, D.M.; Blande, J.D.; Nykanen, R.; WenXia, D.; Ner, A.M.; Holopainen, J.K. Ozone degrades common herbivore-induced plant volatiles: Does this affect herbivore prey location by predators and parasitoids? J. Chem. Ecol. 2007, 33, 683–694. [Google Scholar] [CrossRef]
- Liu, J.-H.; Kitashiba, H.; Wang, J.; Ban, Y.; Moriguchi, T. Polyamines and their ability to provide environmental stress tolerance to plants. Plant Biotechn. 2007, 24, 117–126. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta Gen. Subj. BBA-GEN SUBJECTS 2013, 1830, 3670–3695. [Google Scholar] [CrossRef]
- Williams, C.A.; Grayer, R.J. Anthocyanins and other flavonoids. Nat. Prod. Rep. 2004, 12, 539–573. [Google Scholar] [CrossRef]
- Melidou, M.; Riganakos, K.; Galaris, D. Protection against nuclear DNA damage offered by flavonoids in cells exposed to hydrogen peroxide: The role of iron chelation. Free Radic. Biol. Med. 2005, 39, 1591–1600. [Google Scholar] [CrossRef] [PubMed]
- Selway, J.T. Antiviral activity of flavones and flavans. Prog Clin. Biol. Res. 1986, 213, 521–536. [Google Scholar]
- Maher, P. The flavonoid fisetin promotes nerve cell survival from trophic factor withdrawal by enhancement of proteasome activity. Arch. Biochem. Biophy. 2008, 476, 139–144. [Google Scholar] [CrossRef]
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev. 2010, 29, 405–434. [Google Scholar] [CrossRef]
- Sung, B.; Pandey, M.K.; Aggarwal, B.B. Fisetin, an inhibitor of cyclin-dependent kinase 6, down-regulates nuclear factor-κB-regulated cell proliferation, antiapoptotic and metastatic gene products through the suppression of TAK-1 and receptor-interacting protein-regulated IκBα kinase activation. Mol. Pharmacol. 2007, 71, 1703–1714. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Al-Attas, O.S.; Alamery, S.; Ahmed, M.; Odeibat, H.A.M.; Alrokayan, S. The plant flavonoid, fisetin alleviates cigarette smoke-induced oxidative stress, and inflammation in Wistar rat lungs. J. Food Biochem. 2019, 43, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.S.; Choi, E.Y.; Jin, J.Y.; Park, H.R.; Choi, J.I.; Kim, S.J. Kaempferol inhibits P. intermedia lipopolysaccharide-induced production of nitric oxide through translational regulation in murine macrophages: Critical role of heme oxygenase-1-mediated ROS reduction. J. Periodontol. 2013, 84, 545–555. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Calderon-Montano, J.M.; Burgos-Moron, E.; Perez-Guerrero, C.; Lopez-Lazaro, M. A review on the dietary flavonoid kaempferol. Mini. Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Kalantari, H.; Das, D.K. Physiological effects of resveratrol. Biofactors 2010, 36, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Gao, W.; Huang, L. Tanshinones, critical pharmacological components in Salvia miltiorhiza. Front. Pharmacol. 2019, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Oliviera, A.B.; Dolabela, M.F.; Braga, F.C.; Jacome, R.L.R.P.; Varotti, F.P.; Povoa, M.M. Plant-derived antimalarial agents: New leads and efficient phytomedicines. Part I. Alkaloids. An. Acad. Bras. Cienc. 2009, 81, 715–740. [Google Scholar] [CrossRef] [PubMed]
- Binder, B.Y.K.; Peebles, C.A.M.; Shanks, J.V.; San, K.-Y. The effects of UV-B stress on the production of terpenoid indole alkaloids in Catharanthus roseus hairy roots. Biotechnol. Prog. 2009, 25, 861–865. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.; Creswell, K.M.; Chin, D. The mechanism of action of vinblastine. Binding of [acetyl-3H]vinblastine to embryonic chick brain tubulin and tubulin from sea urchin sperm tail outer doublet microtubules. Biochemistry 1975, 14, 5586–5592. [Google Scholar] [CrossRef] [PubMed]
- Zi, J.; Mafu, S.; Peters, R.J. To gibberellins and beyond! surveying the evolution of (di) terpenoid metabolism. Annu. Rev. Plant Biol. 2014, 65, 259–286. [Google Scholar] [CrossRef]
- Salminen, A.; Lehtonen, M.; Suuronen, T.; Kaarniranta, K.; Huuskonen, J. Terpenoids: Natural inhibitors of NF-kB signaling with anti-inflammatory and anticancer potential Cell Mol. Life Sci. 2008, 65, 2979–2999. [Google Scholar]
- Geu-Flores, F.; Sherden, N.H.; Courdavault, V.; Burlat, V.; Glenn, W.S.; Wu, C.; Nims, E.; Cui, Y.; O’Connor, S.E. An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis. Nature 2012, 492, 138–142. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Butler, M.S.; Robertson, A.A.; Cooper, M.A. Natural product and natural product derived drugs in clinical trials. Nat. Prod. Rep. 2014, 31, 1612–1661. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; International Natural Product Sciences, T.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Zemp, M.; Hoelzle, M.; Haeberli, W. Six decades of glacier mass balance observations—A review of the worldwide monitoring network. Ann. Glaciol. 2009, 50, 101–111. [Google Scholar] [CrossRef]
- Cazzolla Gatti, R.; Callaghan, T.; Velichevskaya, A.; Dudko, A.; Fabbio, L.; Battipaglia, G.; Liang, J. Accelerating upward treeline shift in the Altai Mountains under last-century climate change. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Williams, S.E.; Bolitho, E.E.; Fox, S. Climate change in Australian tropical rainforests: An impending environmental catastrophe. Proc. Biol. Sci. 2003, 270, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- Steinbauer, M.J.; Grytnes, J.A.; Jurasinski, G.; Kulonen, A.; Lenoir, J.; Pauli, H.; Rixen, C.; Winkler, M.; Bardy-Durchhalter, M.; Barni, E.; et al. Accelerated increase in plant species richness on mountain summits is linked to warming. Nature 2018, 556, 231–234. [Google Scholar] [CrossRef]
- Toscano, S.; Trivellini, A.; Cocetta, G.; Bulgari, R.; Francini, A.; Romano, D.; Ferrante, A. Effect of preharvest abiotic stresses on the accumulation of bioactive compounds in horticultural produce. Front. Plant. Sci. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef]
| Stress Condition(s) | Plant Species (Family) | PSMs Produced | Effects on PSMs Concentration | Compound Class | Bioactive Compounds | Reported Pharmacological Properties |
|---|---|---|---|---|---|---|
| Cold stress | Catharanthus roseus (Apocynaceae) [98] | vindoline | Decrease | Alkaloids | vindoline | Antidiabetic [99] |
| Cold stress | Glycine max (Fabaceae) [94] | genistein, daidzein | Increase | Phenolics | genistein, daidzein | Antiproliferative [95,96] |
| Cold stress | Solanum lycopersicon (Solanaceae) [87,97] | (Z)-3-hexenol and (E)-2-hexenal (dominant); 1-hexanol and 1,4-hexadienal (smaller quantities) | Increase | Fatty Acyls | (E)-2-hexenal | Antibacterial [100] |
| Cold stress | β-phellandrene, (E)-β-ocimene | Increase | Terpenoids | NA | NA | |
| Cold stress | δ-elemene, α-humulene and β-caryophyllene (dominant); in severe cold: β-elemene is produced. | Increase | Terpenoids | δ-elemene, α-humulene and β-caryophyllene | Antiproliferative [101]; anticancer [102]; anti-inflammatory [103] | |
| Cold stress | Zea mays (Poaceae) [104] | pelargonidin | Increase | Phenolics | pelargonidin | Antithrombotic [105] |
| Cold stress | Fagopyrum tartaricum (Polygonaceae) [106] | anthocyanins (e.g.,3-O-galactosides) and anthocyanidins (e.g., malvidin) | Increase | Phenolics | anthocyanins | Antioxidant [107] |
| Cold stress | Withania somnifera (Solanaceae) [108] | withanolide A, withaferin A | Increase | Terpenoids | withanolide A; withferin A | Neuroprotective [109]; anticancer [110] |
| Cold stress | Camellia sinensis (Theaceae) [111] | nerolidol glucoside | Increase | Terpenoids | NA | NA |
| Drought | Amaranthus tricolor (Amaranthaceae) [112] | hydroxybenzoic acids (gallic acid, vanillic acid, syringic acid, p-hydroxybenzoic acid, salicylic acid, ellagic acid), hydroxycinnamic acids (caffeic acid, chlorogenic acid, p-coumaric acid, ferulic acid, m-coumaric acid, sinapic acid, trans-cinnamic acid), flavonoids (iso-quercetin, hyperoside, rutin). | Increase | Phenolics (Flavonoids) | p-hydroxybenzoic acid | Antisickling activity [113] |
| Drought | Camellia sinensis (Theaceae) [114] | Epicatechins | Increase | Phenolics (Flavonoids) | epicatechins | Antioxidant [115] |
| Drought | Camptotheca acuminata (Nyssaceae) [116] | camptothecin | Increase | Alkaloids | camptothecin | Antitumour [117] |
| Drought (PEG-induced) | Catharanthus roseus (Apocyanaceae) [118] | vinblastine | Increase | Alkaloids | vinblastine | Anticancer [119] |
| Drought | Cistus clusii (Cistaceae) [120] | epigallocatechin gallate, epicatechin, epicatechin gallate, and ascorbic acid. | Increase | Phenolics (Flavonols) | epigallocatechin gallate | Anticancer [121]; antibacterial [122] |
| Drought | Crataegus laevigata, C. monogyna (Rosaceae) [123] | chlorogenic acid, catechin, (−)-epicatechin | Increase | Phenolics | chlorogenic acid, (−)-epicatechin | Antioxidant [124,125] |
| Drought | Glycine max (Fabaceae) [126] | trigonelline | Increase | Alkaloids | trigonelline | Antidiabetic [127] |
| Drought | Hypericum brasiliense (Hypericaceae) [128] | isouliginosin B, rutin, 1,5-dihydroxyxanthone | Increase | Phenolics | isouliginosin B, rutin, | Antinociceptive [129]; Anticancer [130] |
| betulinic acid | Terpenoids | betulinic acid | Anticancer [131] | |||
| Drought | Lupinus angustifolius (Fabaceae) [132] | chinolizidin | Increase | Alkaloids | NA | NA |
| Drought | Papaver somniferum (Papaveraceae) [133] | morphine, codeine | Increase | Alkaloids | morphine, codeine | Analgesic [134,135] |
| Drought | Pinus sylvestris (Pinaceae) [136] | abietic acid | Increase | Terpenoids | abietic acid | Antiallergic [137]; anti-inflammatory [138] |
| Drought | Salvia miltiorrhiza (Lamiaceae) [139] | tanshinones, cryptotanshinone | Increase | Terpenoids | cryptotanshinone | Anticancer [140]. |
| Drought | S. miltiorrhiza [139] | rosmarinic acid | Decrease | Phenolics | rosmarinic acid | Antioxidant [141] |
| salvianolic acid | Increase | salvianolic acids | Antioxidant [142] | |||
| Drought | Scrophularia ningpoensis (Scrophulariaceae) [143] | catalpol, harpagide, aucubin, harpagoside | Increase | Glycosides | catalpol, aucubin | Hepatoprotective [144]; neuroprotective [145] |
| Ozone (O3) stress | S. lycopersicon [87,97] | α-carotene, β-carotene, violoxanthin | Increase | Terpenoids | β-carotene | Antioxidants [146]; anti-inflammatory [147] |
| isoprene, α-pinene, β-pinene, myrcene, limonene, sabinene, (E)-β-ocimene, (Z)-β-ocimene, α-humulene, (E)-β-farnesene, (E,E)-α-farnesene, (E)-β-caryophyllene, δ-cadinene | Increase | Terpenoids | α-pinene; myrcene; limonene; α-humulene. | Anti-inflammatory [148]; anti-asthmatic [149]; antioxidant [150]; anti-inflammatory [151] | ||
| O3 | Gingko biloba (Ginkgoaceae) [152] | ginkgolide A | Increase | Terpenoids | ginkgolide A | Neuroprotective [153] |
| Ultraviolet radiation-B (UV-B) | Arabidopsis thaliana (Brassicaceae) [154] | kaempferol 3-gentiobioside-7-rhamnoside; kaempferol 3,7-dirhamnoside. | Increase | Phenolics (Flavonoids) | NA | NA |
| UV-B | Brassica napus (Brassicaceae) [155] | quercetin 3-sophoroide-7-glucoside; quercetin 3-sinapyl sophoroside-7-glucoside | Increase | Phenolics (Flavonoids) | NA | NA |
| UV-B | Brassica oleracea (Brassicaceae) [156] | cyanidine glycosides; sinapyl alcohol | Increase | Phenolics (Flavoboids) | NA | NA |
| UV-B | C. roseus (Apocynaceae) [157,158] | catharanthine, vindoline | Increase | Alkaloids | catharanthine | Anticancer [159] |
| Clarkia breweri (Onagraceae) [160] | eugenol, isoeugenol, methyleugenol, and isomethyleugenol | Increase | Phenolics | eugenol | Antifungal [161]; anti-inflammatory [162] | |
| UV-B | Fagopyrum esculentum (Polygonaceae) [163] | rutin, quercetin, catechin | Increase | Phenolics | quercetin; catechin | Antioxidant [164]; anticancer and antioxidant [165,166] |
| UV-B | Gnaphalium luteoalbum (Asteraceae) [167] | calycopterin; 3’-methoxycalycopterin | Increase | Phenolics (Flavonoids) | calycopterin | Anticancer [168] |
| UV-B | G. viravira [169] | 7-O-methyl araneol | Increase | Phenolics (Flavonoids) | NA | NA |
| UV-B | Hordeum vulgare (Poaceae) [170] | saponarin; luteolin | Increase | Phenolics (Flavonoids) | saponarin; luteolin | Antihypertensive [171]; antibacterial [172] |
| UV-B | Marchantia polymorpha (Marchantiaceae) [173] | luteolin 7-glucuronide; luteolin 3,4’-di-p-coumaryl-quercetin 3-glucoside. | Increase | Phenolics (Flavonoids) | NA | NA |
| UV-B | Quercus ilex (Fagaceae) [174] | acylated kaempferol glycosides | Increase | Phenolics (Flavonoids) | kaempferol | Anticancer [175]; anti-inflammatory [176] |
| Heat stress | C. acuminata [177] | 10-hydroxycamptothecin | Increase | Alkaloids | 10-hydroxycamptothecin | Anticancer [178] |
| Heat stress | Daucus carota (Apiaceae) [179,180,181] | α-terpinolene | Decrease | Terpenoids | α-terpinolene | Antioxidant and anticancer [182] |
| α-caryophyllene, β-farnesene | Increase | NA | NA | |||
| anthocyanins, coumaric and caffeic acid; | Increase | Phenolics | p-coumaric acid and caffeic acid | Antioxidant [183,184] | ||
| Heat stress | Q. rubra (Fagaceae) [185] | isoprene (2-methyl-1,3-butadiene) | Increase | Terpenoids | NA | NA |
| Heat stress | S. lycopersicon [87,97] | β-phellandrene (dominant), 2-carene, α-phellandrene, limonene; increased emission of (E)-β-ocimene after treatment above 46 °C; β-caryophyllene. | Increase | Terpenoids | α-phellandrene; β-caryophyllene | Antifungal [186]; anticancer and anti-inflammatory [102,103] |
| α-humulene | Decrease | α-humulene | Anticancer [187] | |||
| Heat stress (increased humidity) | Centella asiatica (Apiaceae) [188] | asiaticoside | Increase | Phenolics | asiaticoside | Anti-cellulite agent [189] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeshi, K.; Crayn, D.; Ritmejerytė, E.; Wangchuk, P. Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules 2022, 27, 313. https://doi.org/10.3390/molecules27010313
Yeshi K, Crayn D, Ritmejerytė E, Wangchuk P. Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules. 2022; 27(1):313. https://doi.org/10.3390/molecules27010313
Chicago/Turabian StyleYeshi, Karma, Darren Crayn, Edita Ritmejerytė, and Phurpa Wangchuk. 2022. "Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development" Molecules 27, no. 1: 313. https://doi.org/10.3390/molecules27010313
APA StyleYeshi, K., Crayn, D., Ritmejerytė, E., & Wangchuk, P. (2022). Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules, 27(1), 313. https://doi.org/10.3390/molecules27010313