Polylactic Acid—Lemongrass Essential Oil Nanocapsules with Antimicrobial Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Synthesis
2.2. Fluorescent Microscopy
2.3. Dynamic Light Scattering (DLS) and Z-Potential
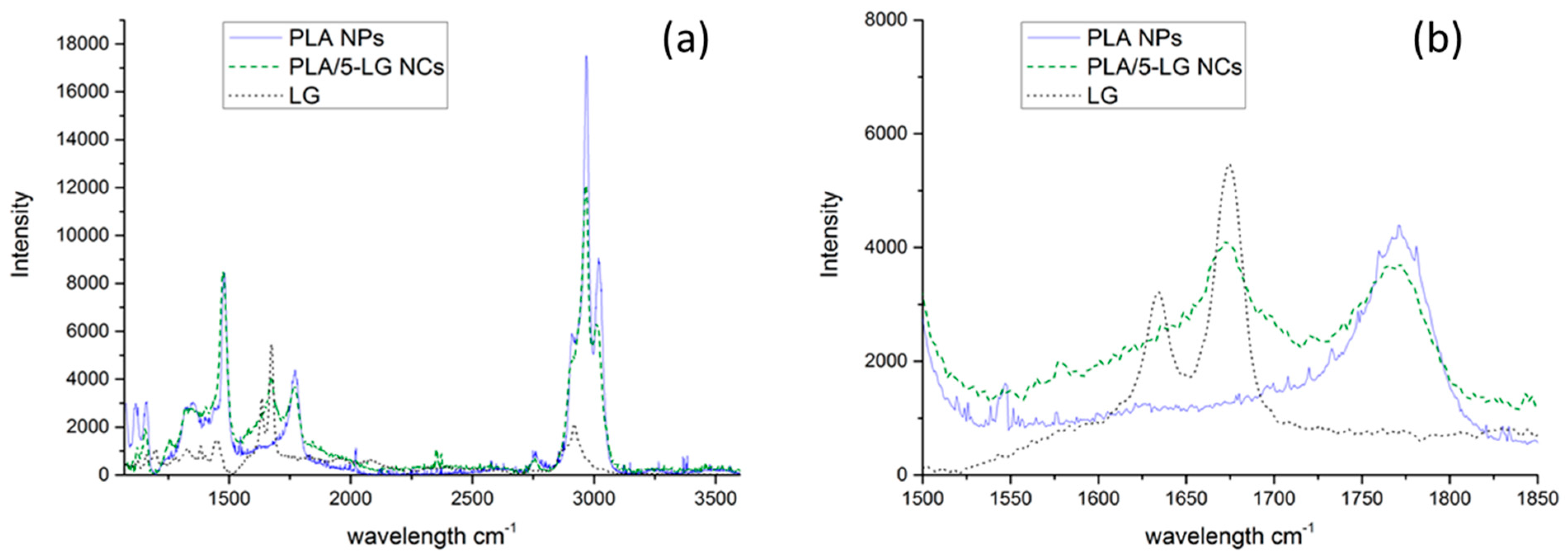
2.4. Raman Spectroscopy
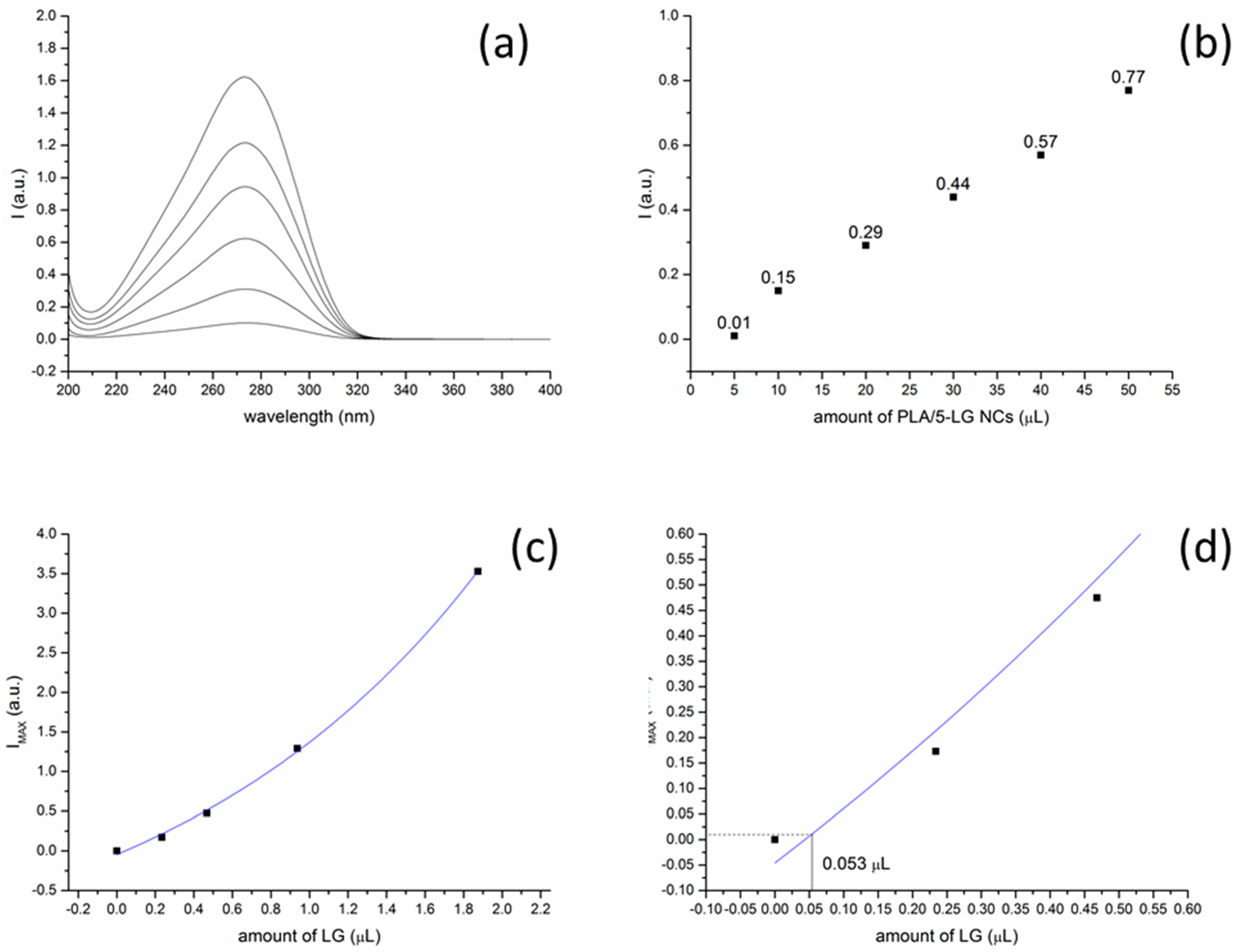
2.5. UV Absorption Spectroscopy
2.6. Antimicrobial Analysis
2.6.1. Microbial Strains and Growth Conditions
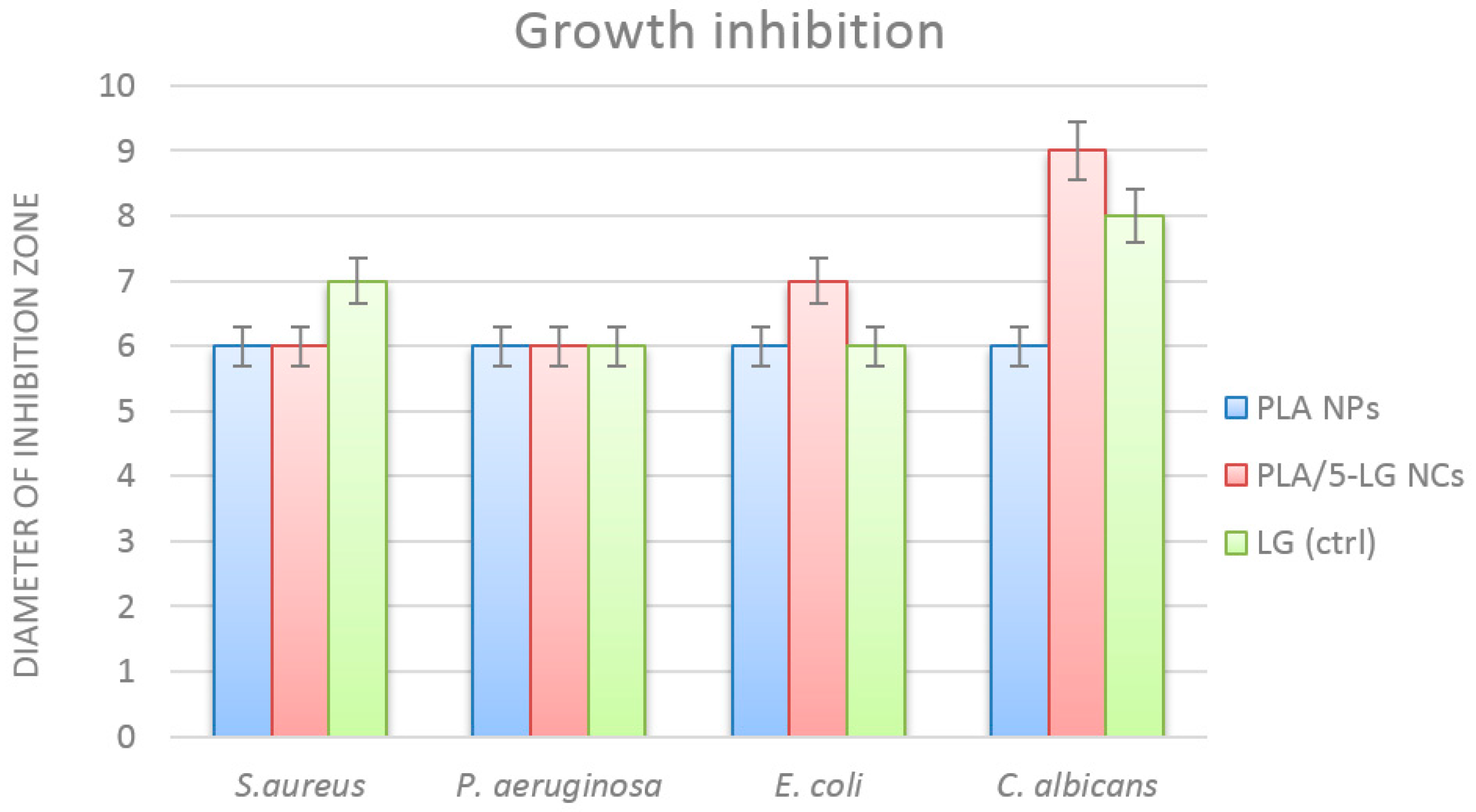
2.6.2. Qualitative Antimicrobial Assay
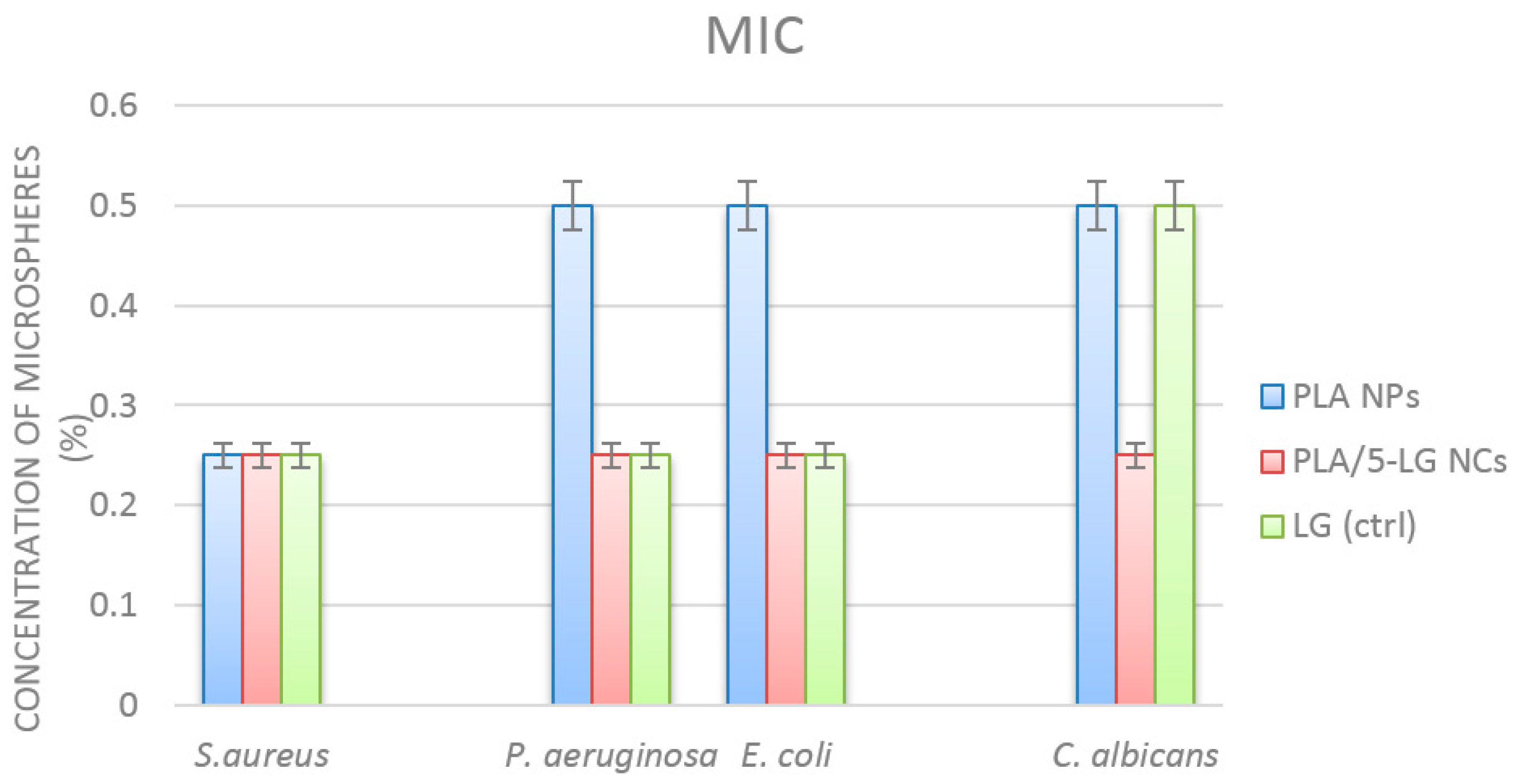
2.6.3. Minimum Inhibitory Concentration (MIC Assay)
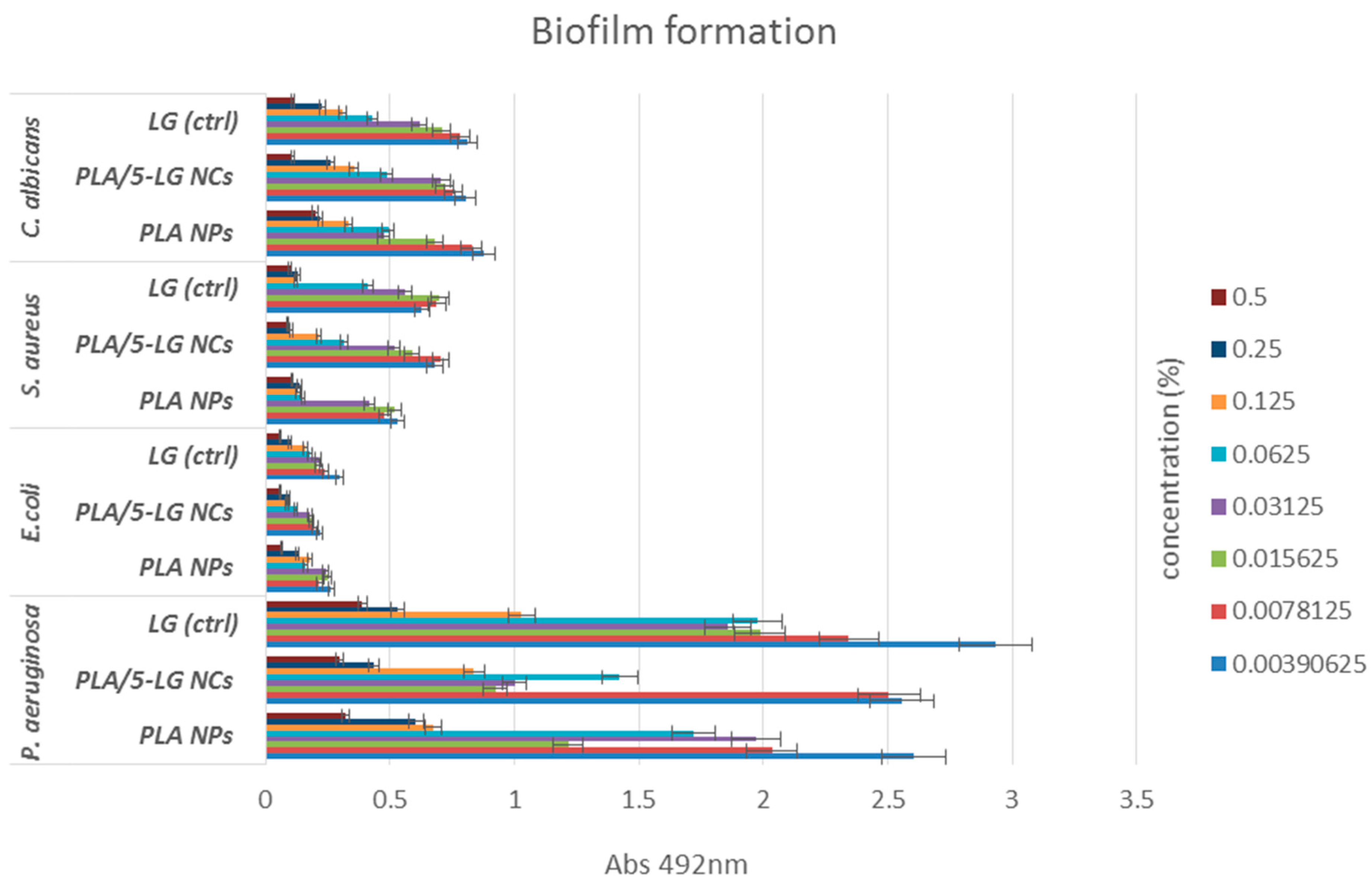
2.6.4. Biofilm Development
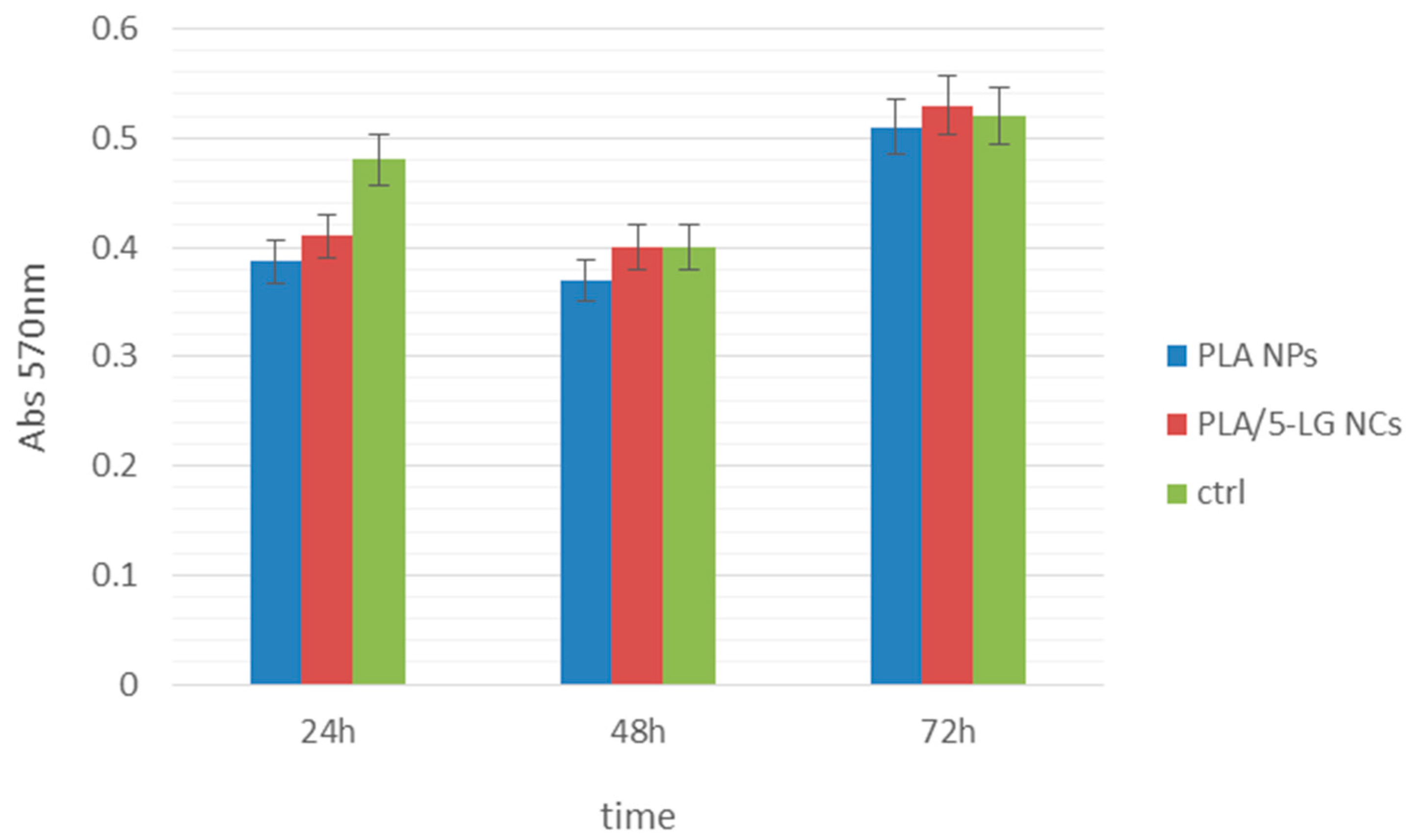
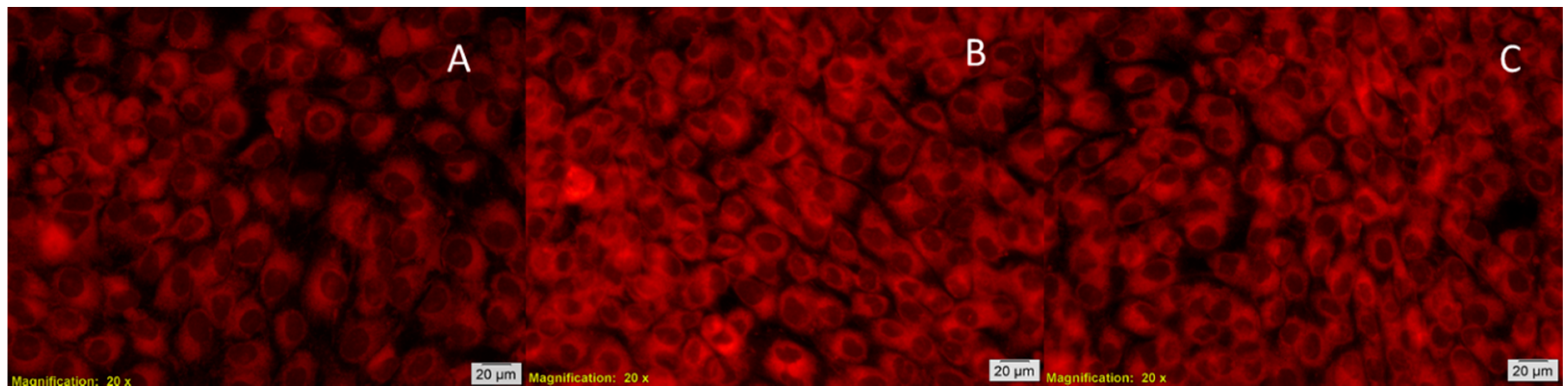
2.7. In Vitro Biocompatibility Tests
3. Results
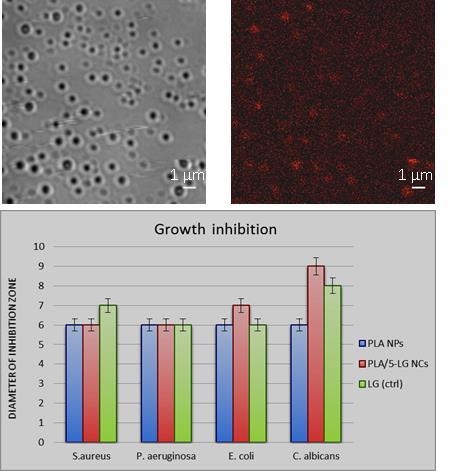
3.1. Fluorescence Microscopy
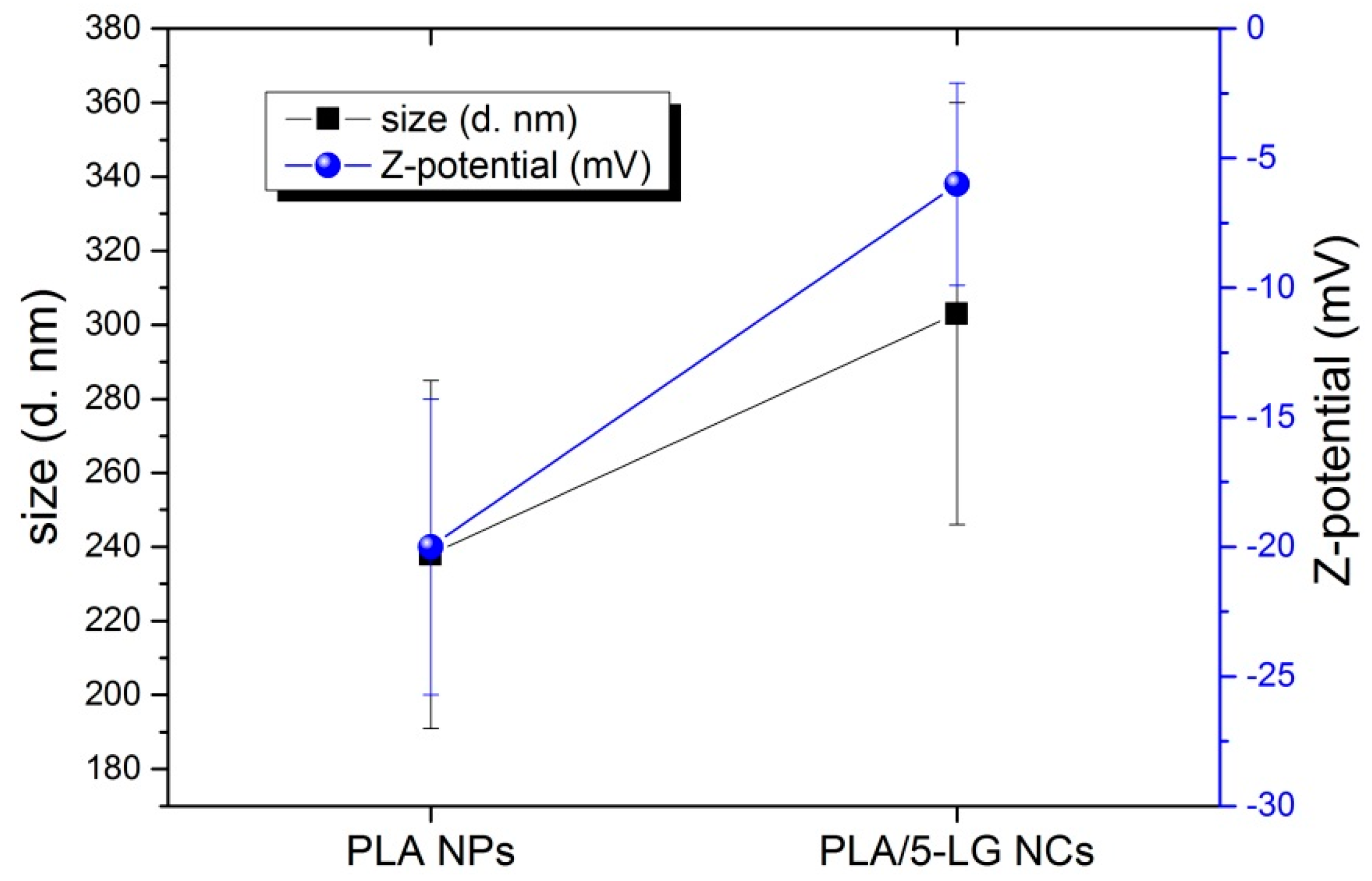
3.2. Dynamic Light Scattering (DLS) and Z-Potential
3.3. Raman Spectroscopy
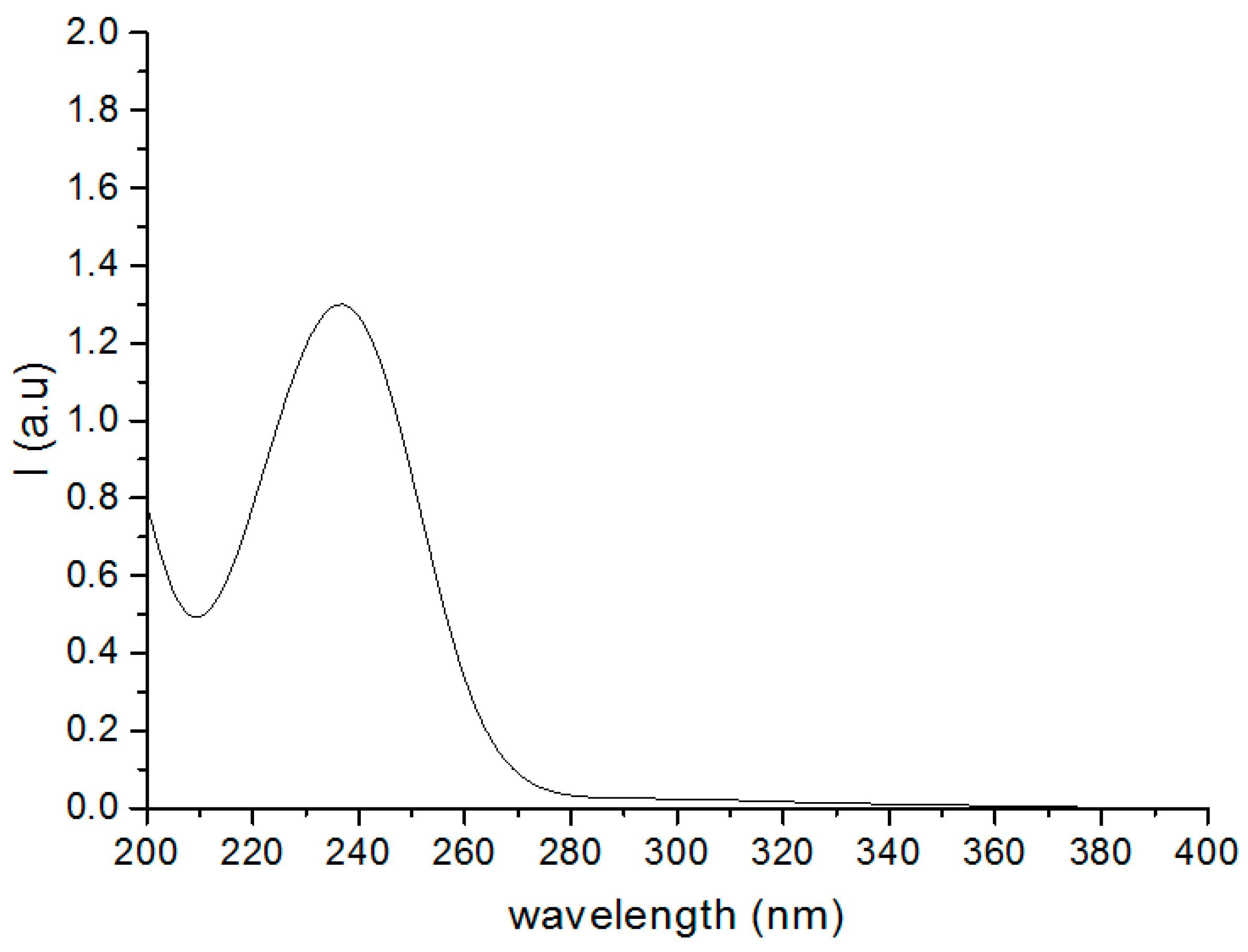
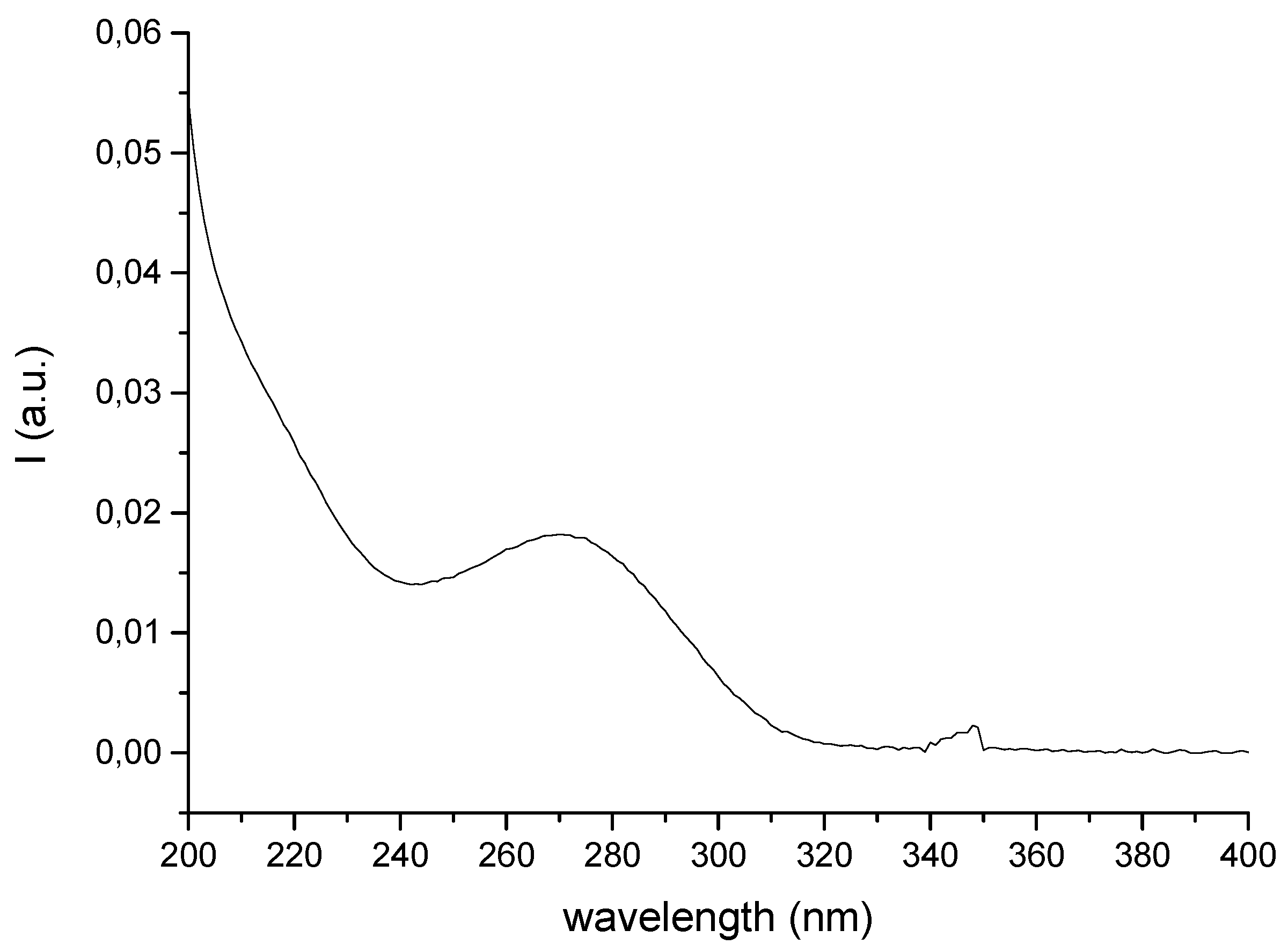
3.4. UV Absorption Spectroscopy
3.5. Antimicrobial Analysis
3.6. Biocompatibility Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
References
- Hureaux, J.; Lagarce, F.; Gagnadoux, F.; Vecellio, L.; Clavreul, A.; Roger, E.; Kempf, M.; Racineux, J.-L.; Diot, P.; Benoit, J.-P.; et al. Lipid nanocapsules: Ready-to-use nanovectors for the aerosol delivery of paclitaxel. Eur. J. Pharm. Biopharm. 2009, 73, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Paranjpe, M.; Müller-Goymann, C. Nanoparticle-mediated pulmonary drug delivery: A review. Int. J. Mol. Sci. 2014, 15, 5852–5873. [Google Scholar] [CrossRef] [PubMed]
- Smola, M.; Vandamme, T.; Sokolowski, A. Nanocarriers as pulmonary drug delivery systems to treat and to diagnose respiratory and non respiratory diseases. Int. J. Nanomed. 2008, 3, 1–19. [Google Scholar]
- Willis, L.; Hayes, D.; Mansour, H.M. Therapeutic liposomal dry powder inhalation aerosols for targeted lung delivery. Lung 2012, 190, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Conhai, R.L.; Rodenkirch, L.A. Estimated functional diameter of alveolar septal microvessels at the zone i–ii border. Microcirculation 1997, 4, 51–59. [Google Scholar] [CrossRef]
- Uota, M.; Arakawa, H.; Kitamura, N.; Yoshimura, T.; Tanaka, J.; Kijima, T. Synthesis of high surface area hydroxyapatite nanoparticles by mixed surfactant-mediated approach. Langmuir 2005, 21, 4724–4728. [Google Scholar] [CrossRef] [PubMed]
- Liakos, I.L.; D’autilia, F.; Garzoni, A.; Bonferoni, C.; Scarpellini, A.; Brunetti, V.; Carzino, R.; Bianchini, P.; Pompa, P.P.; Athanassiou, A. All natural cellulose acetate—Lemongrass essential oil antimicrobial nanocapsules. Int. J. Pharm. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Pornpattananangkul, D.; Hu, C.M.J.; Huang, C.M. Development of nanoparticles for antimicrobial drug delivery. Curr. Med. Chem. 2010, 17, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Morais, J.M.; Papadimitrakopoulos, F.; Burgess, D.J. Biomaterials/tissue interactions: Possible solutions to overcome foreign body response. AAPS J. 2010, 12, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Boreham, A.; Pikkemaat, J.; Volz, P.; Brodwolf, R.; Kuehne, C.; Licha, K.; Haag, R.; Dernedde, J.; Alexiev, U. Detecting and quantifying biomolecular interactions of a dendritic polyglycerol sulfate nanoparticle using fluorescence lifetime measurements. Molecules 2016, 21, 22. [Google Scholar] [CrossRef] [PubMed]
- Hirsjärvi, S. Preparation and Characterization of Poly(Lactic Acid) Nanoparticles for Pharmaceutical Use; University of Helsinki, Faculty of Pharmacy, Division of Pharmaceutical Technology, University of Helsinki, Department of Chemistry, Laboratory of Analytical Chemistry: Helsinki, Finland, 2008. [Google Scholar]
- Ambrose, C.G.; Clanton, T.O. Bioabsorbable implants: Review of clinical experience in orthopedic surgery. Ann. Biomed. Eng. 2004, 32, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomaterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- Middleton, J.C.; Tipton, A.J. Synthetic biodegradable polymers as orthopedic devices. Biomaterials 2000, 21, 2335–2346. [Google Scholar] [CrossRef]
- Jeevitha, D.; Malathy, B.R.; Pradeep, P.S.; Srividya, S. Antibacterial activity of anthraquinone encapsulated chitosan/poly(lactic acid) nanoparticles. Int. J. Pharm. Bio. Sci. 2014, 5, 20–28. [Google Scholar]
- Stoica, P.; Râpă, M.; Chifiriuc, M.-C.; Lungu, M.; Tatia, R.; Niță, M.I.; Grumezescu, A.M.; Bertesteanu, S.; Bezirtzoglou, E.; Lazăr, V. Antifungal bionanocomposites based on poly(lactic acid) and silver nanoparticles for potential medical devices. Romanian Biotechnol. Lett. 2015, 20, 10696–10707. [Google Scholar]
- Mosqueira, V.C.F.; Loiseau, P.M.; Bories, C.; Legrand, P.; Devissaguet, J.-P.; Barratt, G. Efficacy and pharmacokinetics of intravenous nanocapsule formulations of halofantrine in plasmodium berghei-infected mice. Antimicrob. Agents Chemother. 2004, 48, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.; Moreira, R.G.; Castell-Perez, E. Poly (dl-lactide-co-glycolide) (PLGA) nanoparticles with entrapped trans-cinnamaldehyde and eugenol for antimicrobial delivery applications. J. Food Sci. 2011, 76, N16–N24. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.S.; Kumar, P.S.; Vijayalakshmi, S. Anticancer studies of drug encapsulated polyethylene terephthalate-co-polylactic acid nanocapsules. J. Pharm. Bioallied Sci. 2011, 3, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Jia, D.; Barwal, I.; Thakur, S.; Yadav, S.C. Methodology to nanoencapsulate hepatoprotective components from picrorhiza kurroa as food supplement. Food Biosci. 2015, 9, 28–35. [Google Scholar] [CrossRef]
- Parry, E.J. The Chemistry of Essential Oils and Artificial Perfumes, 4th ed.; Scott, Greenwood and Son: London, UK, 1922. [Google Scholar]
- Shah, G.; Shri, R.; Panchal, V.; Sharma, N.; Singh, B.; Mann, A.S. Scientific basis for the therapeutic use of cymbopogon citratus, stapf (lemon grass). J. Adv. Pharm. Technol. Res. 2011, 2, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Liakos, I.; Rizzello, L.; Hajiali, H.; Brunetti, V.; Carzino, R.; Pompa, P.P.; Athanassiou, A.; Mele, E. Fibrous wound dressings encapsulating essential oils as natural antimicrobial agents. J. Mater. Chem. B 2015, 3, 1583–1589. [Google Scholar] [CrossRef]
- Maizura, M.; Fazilah, A.; Norziah, M.H.; Karim, A.A. Antibacterial activity and mechanical properties of partially hydrolyzed sago starch–alginate edible film containing lemongrass oil. J. Food Sci. 2007, 27, C324–C330. [Google Scholar] [CrossRef] [PubMed]
- Liakos, I.; Rizzello, L.; Scurr, D.J.; Pompa, P.P.; Bayer, I.S.; Athanassiou, A. All-natural composite wound dressing films of essential oils encapsulated in sodium alginate with antimicrobial properties. Int. J. Pharm. 2014, 463, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Mohamed Hanaa, A.R.; Sallam, Y.I.; El-Leithy, A.S.; Aly, S.E. Lemongrass (cymbopogon citratus) essential oil as affected by drying methods. Ann. Agric. Sci. 2012, 57, 113–116. [Google Scholar] [CrossRef]
- Osmundsen, C.M.; Egeblad, K.; Taarning, E. Chapter 4—Trends and challenges in catalytic biomass conversion a2—Suib, steven l. In New and Future Developments in Catalysis; Elsevier: Amsterdam, The Netherlands, 2013; pp. 73–89. [Google Scholar]
- Gref, R.; Lück, M.; Quellec, P.; Marchand, M.; Dellacherie, E.; Harnisch, S.; Blunk, T.; Müller, R.H. ‘Stealth’ corona-core nanoparticles surface modified by polyethylene glycol (PEG): Influences of the corona (peg chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf. B Biointerfaces 2000, 18, 301–313. [Google Scholar] [CrossRef]
- Musumeci, T.; Ventura, C.A.; Giannone, I.; Ruozi, B.; Montenegro, L.; Pignatello, R.; Puglisi, G. Pla/plga nanoparticles for sustained release of docetaxel. Int. J. Pharm. 2006, 325, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Schulz, H. Rapid Analysis of Medicinal and Aromatic Plants by Non-Destructive Vibrational Spectroscopy Methods. In Proceedings WOCMAP III, Quality, Efficacy, Safety, Processing & Trade in MAPs; ISHS Acta Horticulturae: White River, South Africa, 2005. [Google Scholar]
- Grumezescu, V.; Socol, G.; Grumezescu, A.M.; Holban, A.M.; Ficai, A.; Truşcǎ, R.; Bleotu, C.; Balaure, P.C.; Cristescu, R.; Chifiriuc, M.C. Functionalized antibiofilm thin coatings based on pla–pva microspheres loaded with usnic acid natural compounds fabricated by maple. Appl. Surf. Sci. 2014, 302, 262–267. [Google Scholar] [CrossRef]
- Salmieri, S.; Islam, F.; Khan, R.A.; Hossain, F.M.; Ibrahim, H.M.M.; Miao, C.; Hamad, W.Y.; Lacroix, M. Antimicrobial nanocomposite films made of poly(lactic acid)–cellulose nanocrystals (PLA–CNC) in food applications—Part B: Effect of oregano essential oil release on the inactivation of listeria monocytogenes in mixed vegetables. Cellulose 2014, 21, 4271–4285. [Google Scholar] [CrossRef]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential oils loaded in nanosystems: A developing strategy for a successful therapeutic approach. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver nanoparticles as potential antibacterial agents. Molecules 2015, 20, 8856–8874. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Bachiega, T.F.; Sforcin, J.M. Lemongrass and citral effect on cytokines production by murine macrophages. J. Ethnopharmacol. 2011, 137, 909–913. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liakos, I.L.; Grumezescu, A.M.; Holban, A.M.; Florin, I.; D’Autilia, F.; Carzino, R.; Bianchini, P.; Athanassiou, A. Polylactic Acid—Lemongrass Essential Oil Nanocapsules with Antimicrobial Properties. Pharmaceuticals 2016, 9, 42. https://doi.org/10.3390/ph9030042
Liakos IL, Grumezescu AM, Holban AM, Florin I, D’Autilia F, Carzino R, Bianchini P, Athanassiou A. Polylactic Acid—Lemongrass Essential Oil Nanocapsules with Antimicrobial Properties. Pharmaceuticals. 2016; 9(3):42. https://doi.org/10.3390/ph9030042
Chicago/Turabian StyleLiakos, Ioannis L., Alexandru Mihai Grumezescu, Alina Maria Holban, Iordache Florin, Francesca D’Autilia, Riccardo Carzino, Paolo Bianchini, and Athanassia Athanassiou. 2016. "Polylactic Acid—Lemongrass Essential Oil Nanocapsules with Antimicrobial Properties" Pharmaceuticals 9, no. 3: 42. https://doi.org/10.3390/ph9030042
APA StyleLiakos, I. L., Grumezescu, A. M., Holban, A. M., Florin, I., D’Autilia, F., Carzino, R., Bianchini, P., & Athanassiou, A. (2016). Polylactic Acid—Lemongrass Essential Oil Nanocapsules with Antimicrobial Properties. Pharmaceuticals, 9(3), 42. https://doi.org/10.3390/ph9030042