The Overarching Influence of the Gut Microbiome on End-Organ Function: The Role of Live Probiotic Cultures
Abstract
:1. Introduction
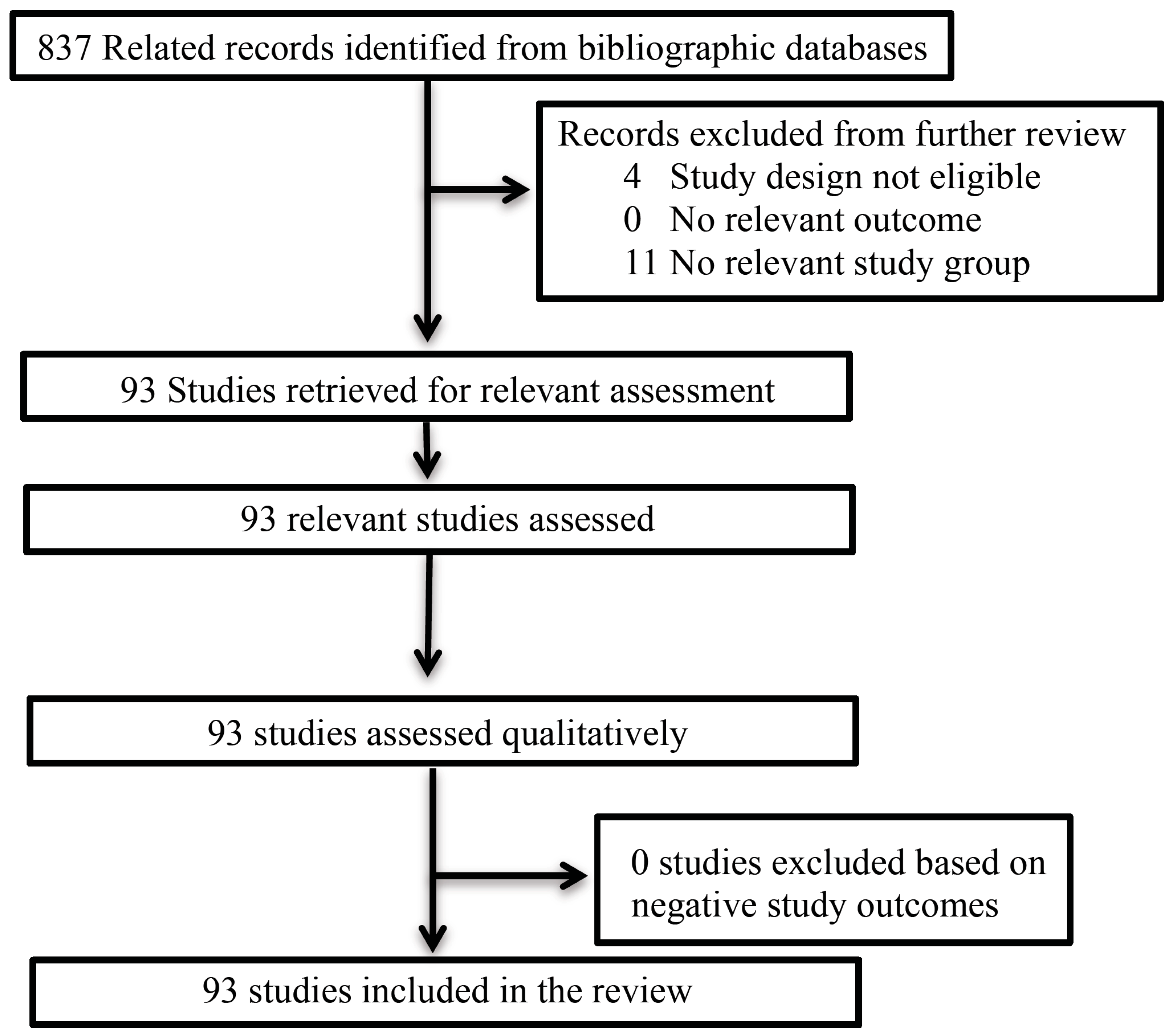
2. Methods
2.1. Search Terms
3. Clinical Studies
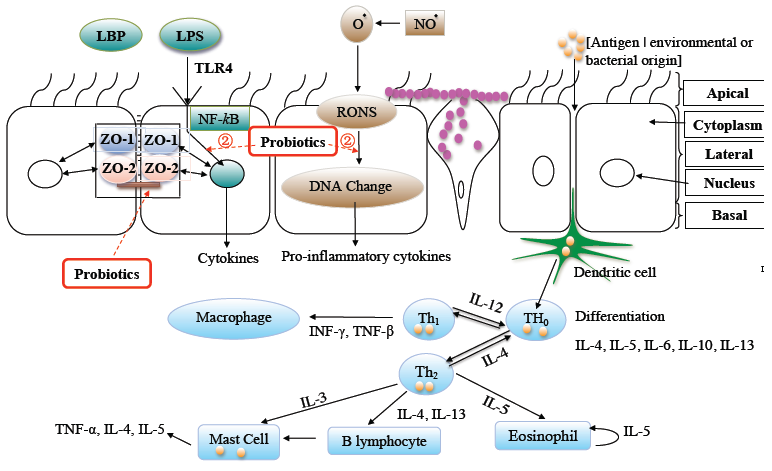
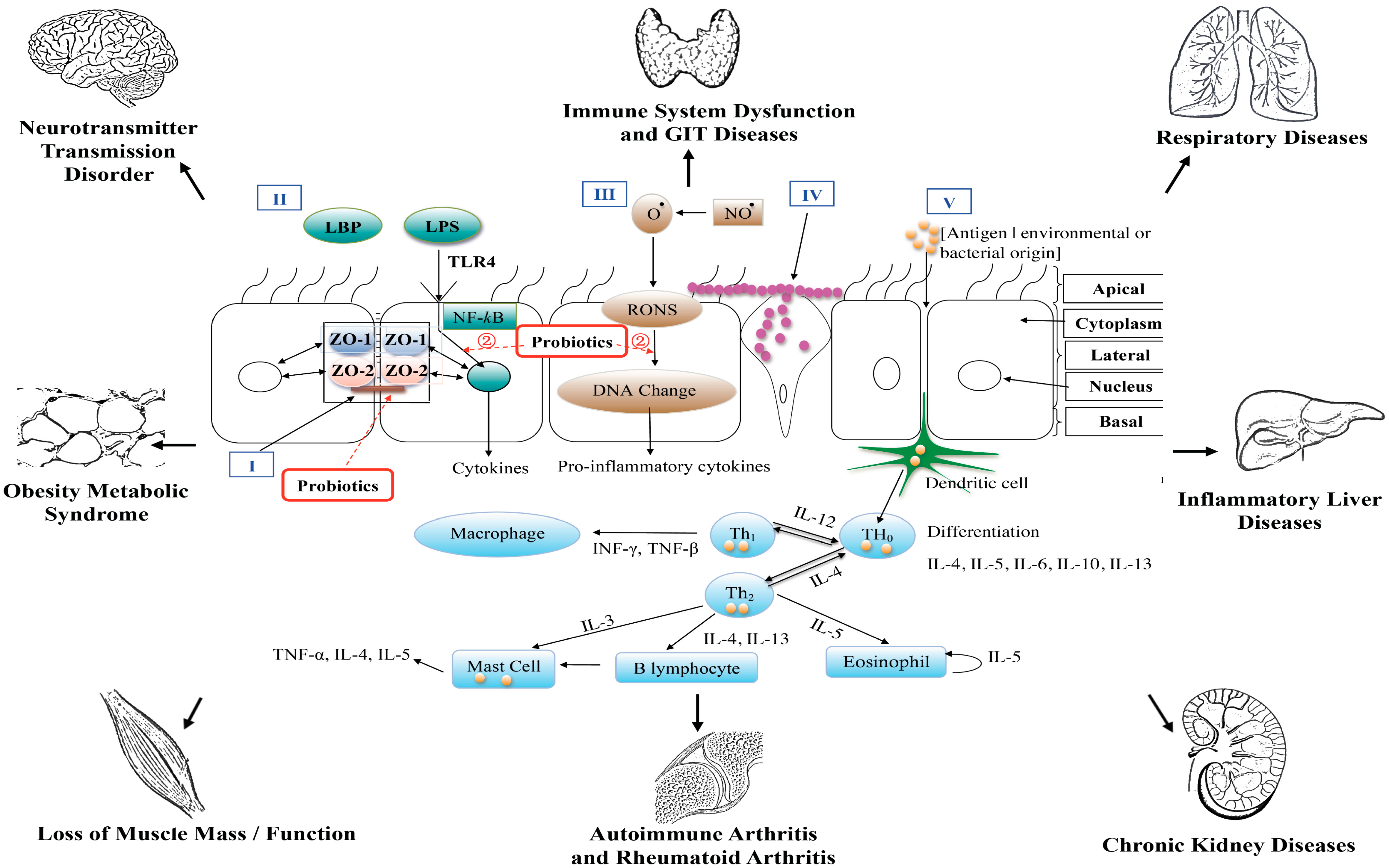
3.1. Probiotics and the GIT
3.2. Probiotics and the Liver
3.3. Probiotics and Obesity
| Participant Type | Study Type (N° Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Irritable Bowel Syndrome | |||||
| Irritable bowel syndrome –constipation predominant | DBPCT (34) | 1 × 108 CFU/g B. lactis DN-173 010 125g/b.i.d. | 4 weeks | ↑Abdominal girth and gastrointestinal transit ↓Symptoms scores of IBS | [10] |
| Irritable bowel syndrome | DBPCT (55) | 1 × 1010 CFU/cap L. rhamnosus GG/b.i.d. | 6 weeks | ↓Abdominal pain | [11] |
| Irritable bowel syndrome | RCT (77) | 1 × 1010 CFU B. infantis 35624/o.i.d. | 8 weeks | ↓Abdominal pain Normalization of Th1/Th2 balance | [12] |
| Irritable bowel syndrome | DBPCT (40) | 2 × 109 CFU/mL L. acidophilus-SDC 2012/o.i.d. | 4 weeks | ↓Abdominal pain or discomfort | [13] |
| Irritable bowel syndrome | DBPCT (52) | 2.5 × 1010 CFU/cap L. acidophilus CUL60 and CUL21 B. lactis CUL34 and B. bifidum CUL20/o.i.d. | 8 weeks | ↓Symptoms scores of IBS ↑Scores for quality of life, days without pain and satisfaction with bowel habit | [14] |
| Irritable bowel syndrome | DBPCT (52) | 5 × 107 CFU/mL L. paracasei ssp paracasei F19 5 × 107 CFU/mL L. acidophilus La5 5 × 107 CFU/mL B. lactis/Bb12 200 mL/b.i.d. | 8 weeks | No clear positive effect on IBS symptoms | [15] |
| Irritable bowel syndrome—diarrhea predominant | DBPCT (30) | 1 × 108 CFU/mL S. thermophiles/1 × 107 CFU/mL L. bulgaricus/ 1 × 107 CFU/mL L. acidophilus/1 × 107 CFU/mL B. Longum 200 mL/b.i.d. | 4 weeks | ↑IBS scores ↓Intestinal permeability | [16] |
| Irritable bowel syndrome | DBPCT(122) | 1 × 109 CFU/cap B. bifidum MIMBb75/o.i.d. | 4 weeks | ↑IBS scores | [17] |
| Irritable bowel syndrome –diarrhea predominant | DBPCT (297) | Inactivated L. acidophilus LB [dose administered not provided] | 6 weeks | ↓Number of stools | [18] |
| Functional Abdominal Pain/Associated Symptoms | |||||
| Functional gastroesphageal reflux | DBPCT(44) | 1 × 108 CFU/cap L. reuteri DSM 17938/o.i.d. | 4 weeks | ↓Median fasting antral area ↑Delta in gastric emptying rate ↓Median episodes per day of regurgitation | [19] |
| Functional abdominal pain | DBPCT (60) | 2 × 108 CFU/cap L. reuteri DSM 17938/b.i.d. | 4 weeks | ↓Abdominal pain | [20] |
| Functional gastrointestinal symptoms | DBPCT(17) | 1-5 × 1010 CFU L. rhamnosus GG/b.i.d | 2 weeks | No evidence of efficacy | [21] |
| Antibiotic Associated Diarrhea | |||||
| Antibiotic-associated diarrhea | DBPCT (2941) | 6 × 1010 CFU/cap L.acidophilus CUL60, and CUL21, B. bifidum CUL20, and B lactis CUL34/o.i.d | 8 weeks | No evidence of efficacy | [22] |
| Antibiotic-associated diarrhea | DBPCT (89) | 5 × 108 CFU/g L. acidophilus Cl1285 and L. casei 98 g/o.i.d. | During antibiotic treatment | Prevention of antibiotic-associated diarrhea in hospitalized patients | [23] |
| Antibiotic-associated diarrhea | DBPCT (255) | 50 or 100 × 109 CFU/cap L. acidophilus CL1285 and L. casei LBC80R/o.i.d. or b.i.d. during antibiotic treatment | 2 weeks | ↓Risk of antibiotic-associated diarrhea | [24] |
| Antibiotic-associated diarrhea | DBPCT (275) | 5 × 109 CFU/cap S. boulardii/b.i.d | during treatment + 1 week | No preventing effect on the development of antibiotic-associated diarrhea | [25] |
| Antibiotic-associated diarrhea | DBPCT (437) | 5 × 108 CFU/g L. acidophilus CL1285®/L. casei 98 g/o.i.d. during antibiotic treatment | 5 weeks | ↓Duration of diarrhea | [26] |
| Antibiotic-associated diarrhea | DBPCT (113) | 1 × 108 CFU/mL L. casei/L. bulgaricus/S. thermophilus 97mL/b.i.d. during antibiotic treatment | 2 weeks | ↓Risk of antibiotic-associated diarrhea | [27] |
| Antibiotic-associated diarrhea | DBPCT (229) | 4.5 × 1011 CFU/sachet B. breve, B. longum, B. infantis, L. acidophilus, L. plantarum, L. paracasei, L.delbrueckii subsp. bulgaricus, S. thermophilus/b.i.d | during antibiotic course+1 week | ↓Risk of antibiotic-associated diarrhea | [28] |
| Acute rotavirus diarrhea | DBPCT (64) | 4 × 1010 CFU/dose S. boulardii or 6.625 × 107 CFU/dose, L.acidophilus, 8.75 × 106 CFU/dose L. rhamnosus, B. longum and 1.375 × 107CFU/dose S. Boulardii/b.i.d | 5 days | ↓Median duration of diarrhea and fever in children who received the single species product ↓Vomiting in children who received the mixed species product | [29] |
| Helicobacter pylori eradication | |||||
| H. pylori therapy | DBPCT (107) | 1.25 × 109 CFU L. acidophilus, 1.25 × 109 CFU L.rhamnosus, 1.25 × 109 CFU B.bifidum and S. faecium (b.i.d) | 1 week | No evidence of increased efficacy | [30] |
| H. pylori therapy | Open label | 30 × 108 CFU B. infantis/b.i.d | 2 weeks | ↑ Cure rates | [31] |
| H. pylori therapy | DBPCT (88) | 1 × 106 CFU/g L. acidophilus LA-5/ 1 × 106 CFU/g B. lactis BB-12 125 g/b.i.d. during H. pylori eradication | 5 weeks | ↓Duration of antibiotics-associated diarrhea ↓Gastrointestinal complaints | [32] |
| H. pylori therapy | Open label (228) | 3 × 107 CFU L. acidophilus/o.i.d | 2 weeks | ↑ Cure rates | [33] |
| H. pylori therapy | Open label (90) | 1×108 CFU L.reuteri/o.i.d | 1 week | ↑ Cure rates ↓Frequency and the intensity of antibiotic-associated side-effects | [34] |
| H. pylori positive subjects | DBPCT (22) | 5 × 109 CFU/tablet dead L. reuteris DSMZ17648/4 tablets/b.i.d | 2 weeks | ↓ H. pylori | [35] |
| Functional gastrointestinal symptoms | |||||
| Chronic pouchitis | DBPCT (20) | (0.5–1) × 1010 CFU/capsule L. rhamnosus GG/2 caps, b.i.d | 12 weeks | ↑Ratio of total faecal lactobacilli to total faecal anaerobes ↑Frequency of lactobacilli-positive cultures in the pouch and afferent limb mucosal biopsy | [36] |
| Functional gastrointestinal symptoms | TBPCT (87) | 1.8 × 109 or 17.2 × 109 CFU/cap B. lactis HN019/o.i.d. | 2 weeks | ↓Whole gut transit time ↓Functional gastrointestinal symptoms | [37] |
| Healthy, postprandial intestinal gas-related symptom | DBPCT (61) | 2 × 109 CFU/cap B. coagulans/o.i.d. | 4 weeks | ↓Abdominal pain ↓Distension scoreNo significant differences in flatus, bloating and gas scores | [38] |
| Elderly patients receiving enteral feeding | DBPCT (123) | 2.5 × 1010 CFU/sachet B. longum BB536 or 5 × 1010CFU/sachet B. longum BB536/b.i.d | 16 weeks | ↑Bowel movements in patients with a low frequency of defecation ↓Bowel movements of patients with a high frequency of defecation | [39] |
| Elderly patients receiving enteral feeding | DBPCT (83) | 5 × 1010 CFU/sachet B. longum BB536/o.i.d | 16 weeks | No significant changes in the frequency of defecation | [39] |
| Women with mild digestive symptoms | DBPCT (197) | 1 × 108 CFU/g B. lactis DN-173 010 125g/b.i.d. | 4 weeks | ↑Gastrointestinal well-being ↓Digestive symptoms | [40] |
| Women with minor digestive symptoms | DBPCT (324) | 1. 107 CFU/g B. lactis and 9.26 × 106 CFU/g S. thermophilus and L. bulgaricus/125 g/b.i.d | 4 weeks | No improvement in GI well-being | [41] |
| Very low weight infants/preterm infants | |||||
| Very low-birth weight infants | DBPCT (221) | 3.5 × 1018 CFU/mL L. sporogenes | from first feed until discharge | No significant difference in the incidence of death or necrotizing enterocolitis ↓Feeding intolerance | [42] |
| Preterm infants | BRCT (81) | 2 × 107 CFU/g of milk powder B. lactis (daily milk volume increasing during treatment) | 4 weeks | ↓Intestinal permeability ↑Head growth | [43] |
| Prophylatic use in newborn infants | DBRCT (589) | L reuteri DSM 17938 Dose of 1 × 108 CFU/day | 12 weeks | ↓the onset of functional gastrointestinal disorders | [44] |
| Participant Type | Study Type (N°. Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Alcoholic liver disease | Open-label (66) | 0.9 × 108CFU/cap B. bifidum/0.9 × 109CFU/cap L. plantarum/o.i.d. | 5 days | Restoration of the bowel flora Improvement in alcohol-induced liver injury | [47] |
| Alcoholic liver disease | DBPCT (49) | 2.5–25 × 109CFU/cap E. coli Nissle twice the amount after 5 days/o.i.d. | 6 weeks | Improvement of intestinal colonization in the E. coli, ↓Endotoxemia Improvement of liver functions | [48] |
| Alcoholic liver disease | Open-label (20) | 6.5 × 109CFU/cap L. casei Shirota/t.i.d. | 4 weeks | Restore neutrophil function, ex vivo endotoxin- stimulated levels of sTNFR1, sTNFR2 and IL10 normalized TLR4 expression | [49] |
| Alcoholic liver disease, Nonalcoholic Fatty Liver Disease | Open-label (78) | 4.5 × 1011CFU/cap S. thermophilus/B. breve/B. longum/B. infantis/L. acidophilus/L. plantarum/L. casei/L. bulgaricus/o.i.d. | 12 weeks | Improvement of plasma level of MDA and 4-HE, whereas cytokines (TNF-alpha, IL-6, and IL-10) improved only in ALD patients | [50] |
| Cirrhosis | RCT (39) | E. Nissle/o.i.d. [dose administered not provided] | 12 weeks | Improvement in intestinal colonisation Improvement in liver function assessed with the Child-Pugh classification. | [51] |
| Cirrhosis | RCT (81) | 109CFU/capsule B. bifidus/L. acidophilus/L.bulgaricus S. thermophilus/t.i.d. | 2 weeks | ↓Escherichia coli count ↓Intestinal flora imbalance Improvement in debilitation, food intake, appetite, abdominal distension, and ascitic fluid | [52] |
| Cirrhosis | DBPCT (36) | 2 × 1010 CFU/cap L. acidophilus/L. bulgaricus/B. lactis/S. thermophilus/o.i.d. | 24 weeks | ↓Ammonia levels starting after 1 month of treatment in patients with baseline ammonia levels > 50 mmol/LNo effect on liver enzyme | [53] |
| Cirrhosis | RCT (8) | 1.8 × 1012CFU/cap S. thermophiles/B. breve/B. longum/B. infantis/L. acidophilus/L. plantarum/L. casei/L. bulgaricus/b.i.d. | 8 weeks | ↑Serum TNF-α ↓Plasma aldosterone. | [54] |
| Cirrhosis | RCT (50) | 2.1 × 107CFU/cap Bifidobacterium/L. acidophilus/Enterococcus/t.i.d. or 9 × 108CFU/cap B. subtilis/1 × 108 CFU/cap E. faecium/t.i.d. | 2 weeks | ↑ Bifidobacterium count ↓Fecal pH, fecal and blood ammonia. ↓Endotoxin in cirrhotic patients with endotoxemia (probiotics containing Bacillus subtilis and Enterococcus faecium) | [55] |
| Hepathic encephalopathy | DBPCT (55) | 1010 CFU/cap P. pentoseceus 5–33:3/L. mesenteroides 32–77:1/L. paracasei subspecies paracasei 19/L. plantarum 2592/o.i.d. | 4 weeks | ↑Fecal content of non-urease-producing
Lactobacillus species at the expense of these other bacterial species ↓Blood ammonia levels and reversal of HE ↓Endotoxemia. | [56] |
| Hepathic encephalopathy | DBPCT (60) | B. longum/o.i.d. And FOS [dose administered not provided] | 12 weeks | Improving neuropsychological testing, serum ammonia levels | [57] |
3.4. Probiotics and the Brain
3.5. Probiotics and CKD
| Patients | Study Type (N° Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Healthy Infants | RCT (179) | 1 × 108 CFU/g L. paracasei ssp. paracasei F19/100 g b.i.d. | 28 weeks | ↓Palmitoleic acid ↑Putrescine | [60] |
| Adults with obese tendencies | DBPCT (87) | 5 × 108 CFU/g L. gasseri SBT2055 CFU/200 g daily | 12 weeks | ↓Abdominal visceral and subcutaneous fat areas ↓Body weight and other measures ↑High-molecular weight adiponectin in serum | [61] |
| Pregnant Women with obese tendencies | DBPCT (159) | 1 × 1010 CFU/cap L. rhamnosus GG/o.i.d. | 4 weeks | Moderation of the initial phase of excessive weight gain of the children, but not of the second phase of excessive weight gain | [62] |
| Obese Adults | DBPCT (75) | 1 × 108 CFU/mL L. acidophilus La5/ 1 × 108 CFU/mL B. BB12/ 1 × 108 CFU/mL L. casei DN001/o.i.d. | 8 weeks | ↓Expression of T-bet gene. | [63] |
| Overweight and obese children | TBPCT (70) | 2.0 × 108 CFU L. casei, L.s rhamnosus, S. thermophilus, B. breve, L. acidophilus, B. longum and L. bulgaricus with prebiotics (fructo oligosaccharides), Vitamin E, Vitamin A and Vitamin C/o.i.d | 8 weeks | ↓Serum triglycerides, total- and low density lipoprotein-cholesterol levels | [64] |
| Participant Type | Study Type (N° Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Anxiety-depressive symptoms | DBPCT (132) | 108 CFU/capsule L. casei/65mL/i.o.d. | 3 weeks | Improvement in mood scores | [71] |
| Chronic fatigue syndrome | DBPCT (39) | 8 × 107 CFU/sachet L. casei strain Shirota/t.i.d. | 8 weeks | ↑Fecal total Bifidobacteria and Lactobacillus ↓Anxiety symptoms | [72] |
| Healthy adults | DBPCT (25) | 3 × 109CFU/sachet L. helveticus R0052/3 × 109CFU/cap B. longum R0175/i.o.d. | 2 weeks | ↓Behaviors indicative of anxiety | [73] |
| Traumatic brain injury | SBCT (52) | 0.5 × 108CFU/sachet B. longum/ 0.5 × 107CFU/sachet L. bulgaricus 0.5 ×107CFU/cap S. thermophilues/t.i.d. | 3 weeks | Adjustment of the Th1/Th2 imbalance ↓Infection rate ↓Use of antibiotics ↑Level of IL-12 | [74] |
| Healthy women with no gastrointestinal or psychiatric symptoms | DBPCT (36) | 1.2×109 CFU/cup S. Thermophilues L. bulgaricus/b.i.d. | 4 weeks | ↓Task-related response of a distributed functional network containing affective, viscerosensory, and somatosensory cortices | [75] |
3.6. Probiotics and Joint Disease
3.7. Probiotics and Respiratory Diseases
3.8. Probiotics and Skin Conditions
| Patients | Study Type (N° Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Chronic kidney disease (stages 3 and 4) | DBPCT (13) | 1.5 × 109 CFU/cap L. acidophilus KB31/B. longum KB35/S. thermophilus KB27/2 capsules/t.i.d. | 24 weeks | Moderate changes in uric acid concentration No significant difference in serum creatinine concentration | [78] |
| Chronic kidney disease (stages 3 and 4) | DBPCT (246) | 1.5 × 109 CFU/cap L. acidophilus/B. longum/S. thermophilus/2 capsules/t.i.d. | 24 weeks | ↓Blood urea nitrogen. ↑Well-being with no serious adverse effects. | [79] |
| Chronic kidney disease | DBPCT (9) | 1 × 108 CFU L. casei strain Shirota/B. breve strain Yakult with 1.67 g galacto-oligosaccharides/t.i.d. | 4 weeks | ↑Quantity and normalization of the stools ↓Serum p-cresol level. | [80] |
| Patients | Study Type (N° Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Rheumatoid arthritis Sulfasalazine treated patients | PCT (12) | 0.9 × 108 CFU/sachet L. acidophilus L10/B. lactis B94/S. salivarius K12/b.i.d | 12 weeks | No influence on the Sulfasalazine metabolism. | [83] |
| Rheumatoid arthritis | DBPCT (45) | 2 × 109 CFU/caplet B. coagulans GBI-30/b.i.d. | 8 weeks | ↓Pain scores. Improvement of global assessment and self-assessed disability | [84] |
| Rheumatoid arthritis | DBPCT (29) | 2 × 109 CFU/cap L.s reuteri RC-14/L. rhamnosus GR-1/b.i.d. | 12 weeks | No differences observed | [85] |
| Spondyloarthritis | DBPCT (63) | 1 × 108 CFU/g S. salivarius K12/4 × 108 CFU/g B. lactis LAFTI B94 1 × 108 CFU/g L. acidophilus LAFTI L100.8 g/b.i.d. | 3 weeks | No significant difference was noted between groups in any of the core domains | [86] |
| Participant Type | Study Type (N°. Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| Asthma and allergic rhinitis | DBPCT (101) | 2 × 109 CFU/cap L.gasseri/o.i.d. | 8 weeks | ↓Clinical symptom scores ↓TNF-α, IFN-γ, IL-12, and IL-13 production by the PBMCs | [90] |
| Grass pollen-dependent allergic rhino-conjunctivitis | DBPCT (30) | 2.5–25 × 109 CFU/cap E.coli Nissle 1917/2 caps/o.i.d | 24 weeks | No clinical evidence of efficacy | [91] |
| Allergic asthma and/or rhinitis | DBCT (187) | 1 × 1010 CFU/mL L. casei/100 mL/o.i.d. | 52 weeks | No difference | [92] |
| Perennial allergic rhinitis | DBPCT (49) | 3 × 108 CFU/mL L. acidophilus strain L-92/100 mL/o.i.d. | 8 weeks | No difference in IgE level or Th1/Th2 | [93] |
| Seasonal allergic rhinitis | DBPCT (20) | 1 × 105 CFU/mL L. casei Shirota/65mL/o.i.d. | 20 weeks | ↓Antigen-induced IL-5, IL-6 and IFN- γ ↑IgG ↓IgE | [94] |
| High-risk allergy children | DBPCT (105) | 5 × 109 CFU/capsule L.GG/2 cap, o.i.d 6-4 weeks before delivery and 6 months after birth | 30 weeks | No evidence of efficacy | [95] |
| High risk allergic disease infants | DBPCT (1223) | 5 × 109 CFU/cap L. rhamnosus GG/5 × 109 CFU/capsule L. reuteri LC705 2 × 108 CFU/cap B. breve Bb99/2 × 108 CFU/cap P. freudenreichii subspecies shermanii JS/b.i.d [4 weeks before delivery + 24 weeks] | 30 weeks | Protection from allergic disease only to cesarean-delivered children | [96] |
| Respiratory illness | DBPCT (523) | 2.5 × 106 CFU/mL L.rhamnosus GG (130 mL, t.i.c) | 28 weeks | ↓Occurence of respiratory illness | [97] |
| Japanese cedar pollinosis | DBPCT (44) | 5 × 1010 CFU B. longum BB536/b.i.d | 13 weeks | ↑ Bacteroides fragilis group | [98] |
| Infants | DBPCT (81) | 1 × 109 CFU/cap L. rhamnosus GG and 1 × 1010 CFU/cap B. lactis Bb-12/o.i.d | 40 weeks | ↓Risk of recurrent respiratory infections ↓ Acute otitis media ↓ Antibiotic use | [99] |
| Grass pollen-dependent allergic rhinitis | DBPCT (20) | 2 × 109 CFU/g B. lactis NCC2818/2g/o.i.d | 8 weeks | ↓Th-2 cytokines, secreted by stimulated blood lymphocytes ↓Total nasal symptom scores ↓Activated CD63 expressing basophils | [100] |
| Allergic rhinitis | DBPCT (31) | 5 × 109 CFU/mL L. Helveticus NCC1643 and × 107 CFU/mL L. paracasei ST11 / 80 mL. o.i.d | 4 weeks | ↓Nasal congestion and nasal pruritus ↓IL-5, IL-8 and IL-10 secretion by peripheral blood mononuclear cells and serum allergen-specific IgG4 | [101] |
| Participant Type | Study Type (N°. Patients) | Treatment | Duration | Results | Ref. |
|---|---|---|---|---|---|
| UV induced skin damage | |||||
| Ultraviolet-induced skin damage | CT (139) | 5 × 108 CFU L. johnsoni/before UVR exposure | 3–6 weeks | Prevention the UV-induced decrease in Langerhans cell density ↑Factor XIIIa+ type I dermal dendrocytes ↓Dermal inflammatory cells ↑Minimal erythemal dose ↑ΔE* parameter | [103] |
| Pregnant women carrying high risk allergy babies | |||||
| High-risk allergy children | DBPCT (159) | 1 × 1010 CFU/cap L. rhamnosus strain GG/o.i.d. or b.i.d./3 weeks before delivery + 24 weeks | 4 weeks to mothers and 24 weeks to infants | ↓cumulative risk for developing eczema during the first 7 years of life | [104] |
| Pregnant women carrying high-risk allergy children | DBPCT (1223) mothers (925) infants | 5 × 109 CFU/cap L. rhamnosus GG 53103/5 × 109 CFU/cap/L. rhamnosus LC705 7061 CFU/cap/5 × 109 CFU/cap B. breve Bb99 13692 and 2 × 108 CFU/cap P. freudenreichii ssp. shermanii JS 7076/b.i.d.+ GOS daily | Mothers dosed with multi strain probiotics for 2 to 4 weeks before delivery then infants received probiotics +GOS for 24 weeks | Prevention of eczema at 2 years of age ↑Lactobacilli and Bifidobacteria in the gut. No effect on incidence of allergic diseases. ↑ CRP, IgA, IgE, IL-10 which were associated with ↓ risk of eczema. | [105,106] |
| Pre and post natal probiotic supplementation | DBPCT (61) | L. reuteri 1 × 108 CFU/day to mothers from week 36 of pregnancy and then to the infant for 24 months post delivery. | 52 weeks | ↓IgE-associated eczema and lowered allergen and mitogen responsiveness | [107] |
| Maternal probiotic supplementation during pregnancy | DBPCT (205) | L. rhamnosus LPR (CGMCC 1.3724) and B. longum BL999 (ATCC: BAA-999) or the combination ST11 and BL999 (ST11 BL999) consisting of L. paracasei ST11 (CNCM 1–2116) and B longum BL999. Dose 1 × 109 CFU/day provided in 1 sachet of 7 g/d (powder form) which was diluted in a glass of water. | 8 weeks | ↓risk of eczema in infants with allergic mothers positive for skin prick test. | [108] |
| Atopic dermatitis/eczema with/without cow’s milk/food allergies | |||||
| Atopic dermatitis | DBPCT (90) | 5 × 109 CFU/g L. acidophilus DDS-1/B. lactis UABLA-12/1g, b.i.d | 8 weeks | ↓SCORAD* score ↓CD4 and CD25 lymphocytes ↑CD8 Lymphocytes | [109] |
| High-risk atopic dermatitis children | PCT (15) | B. breve M-16V strain [dose administered not provided] | 4 weeks | ↑Proportion of Bifidobacteria in the fecal microflora ↓Proportion of aerobic bacteria ↓allergic symptoms | [110] |
| High-risk allergy children | DBPCT (132) | 0.5 × 106 CFU/cap LGG/2 capsules/o.i.d. | 28 weeks | Preventive effect on the incidence of eczema in high-risk children | [111] |
| High risk atopic eczema children | DBPCT (132) | 0.5 × 106 CFU/cap L. rhamnosus 53103 2 caps/o.i.d. | 28 weeks | Preventive effect on the incidence of eczema in high-risk children | [112] |
| Atopic dermatitis | DBPCT (58) | 1 × 106 CFU B. bifidum BGN4/1 × 106 CFU B. lactis AD011/1 × 106 CFU L. acidophilus AD031/o.i.d. | 32 weeks | ↓Cumulative incidence of eczema no difference in serum total IgE level or the sensitization against food allergens | [113] |
| High-risk atopic dermatitis children | DBPCT (102) | 1 × 109 CFU/sachet B. bifidum/1 × 109 CFU/sachet B. lactis/1 × 109 CFU/sachet L. lactis/o.i.d. 8 weeks before delivery + 58 weeks. | Prenatal administration to mothers and for 52 weeks to infants post birth | Preventive effect on the incidence of eczema in high-risk children | [114] |
| Atopic dermatitis | DBPCT (59) | 2 × 1010 CFU/g L. rhamnosus and B. Lactis [dose administered not provided] | 4 weeks | ↓SCORAD geometric mean score | [115] |
| Atopic eczema/dermatitis syndrome and food allergy | DBPCT (230) | 5 × 109 CFU/cap L. rhamnosus GG 53103 or 5 × 109 CFU/cap L. rhamnosus GG/5 × 109 CFU L. rhamnosus LC705/2 × 108 CFU/cap/B. breve Bbi99 and 2 × 109 CFU/capsule P. freudenreichii ssp. Shermanii JS/b.i.d. | 4 weeks | ↑Fecal IgA ↓Fecal alpha1-antitrypsin | [116] |
| Atopic dermatitis | DBPCT (66) | 1 × 109/sachet L. fermentum VRI-033 PCC (b.i.d) | 8 weeks | ↓SCORAD total scores | [117] |
| High-risk allergy children | DBPCT (425) | 6 × 109 CFU/day L. rhamnosus HN001 or 9 × 109 CFU/day B.animalis subsp lactis HN019/from 35 weeks gestation to 2 years after birth | 109 weeks | Protective effect of HN001 against eczema, when given for the first 2 years of life only, extended to at least 4 years of age. Protective effect against rhino-conjunctivitis | [118] |
| High-risk allergy children | DBPCT (474) | 6 × 109 CFU/cap L. rhamnosus/3 weeks before delivery + 2 years | 119 weeks | ↓Cumulative prevalence of eczema No effect on atopy | [119] |
4. Discussion
5. Future Prospective
Acknowledgments
Conflicts of interest
Author Contributions
References
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar]
- Koren, O.; Knights, D.; Gonzalez, A.; Waldron, L.; Segata, N.; Knight, R.; Huttenhower, C.; Ley, R.E. A guide to enterotypes across the human body: Meta-analysis of microbial community structures in human microbiome datasets. PLoS Comput. Biol. 2013, 9, e1002863. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Roager, H.M.; Licht, T.R.; Poulsen, S.K.; Larsen, T.M.; Bahl, M.I. Microbial enterotypes, inferred by the prevotella-to-bacteroides ratio, remained stable during a 6-month randomized controlled diet intervention with the new nordic diet. Appl. Environ. Microbiol. 2014, 80, 1142–1149. [Google Scholar] [CrossRef]
- Morelli, L.; Capurso, L. FAO/WHO Guidelines on Probiotics: 10 Years on. J. Clin. Gastroenterol. 2012, 46, S1–S2. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, H.M.; Koning, C.J.; Mulder, L.; Rombouts, F.M.; Beynen, A.C. Monostrain, multistrain and multispecies probiotics—A comparison of functionality and efficacy. Int. J. Food Microbiol. 2004, 96, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Briskey, D.; Hayes, E.; Shing, C.; Peake, J. A review of the pharmacobiotic regulation of gastrointestinal inflammation by probiotics, commensal bacteria and prebiotics. Inflammopharmacology 2012, 20, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Rijkers, G.T.; Bengmark, S.; Enck, P.; Haller, D.; Herz, U.; Kalliomaki, M.; Kudo, S.; Lenoir-Wijnkoop, I.; Mercenier, A.; Myllyluoma, E.; et al. Guidance for Substantiating the Evidence for Beneficial Effects of Probiotics: Current Status and Recommendations for Future Research. J. Nutr. 2010, 140, 671S–676S. [Google Scholar]
- Agrawal, A.; Houghton, L.A.; Morris, J.; Reilly, B.; Guyonnet, D.; Goupil Feuillerat, N.; Schlumberger, A.; Jakob, S.; Whorwell, P.J. Clinical trial: The effects of a fermented milk product containing Bifidobacterium lactis DN-173 010 on abdominal distension and gastrointestinal transit in irritable bowel syndrome with constipation. Aliment. Pharmacol. Ther. 2009, 29, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Bausserman, M.; Michail, S. The use of Lactobacillus GG in irritable bowel syndrome in children: A double-blind randomized control trial. J. Pediatr. 2005, 147, 197–201. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, L.; McCarthy, J.; Kelly, P.; Hurley, G.; Luo, F.; Chen, K.; O’Sullivan, G.C.; Kiely, B.; Collins, J.K.; Shanahan, F.; Quigley, E.M. Lactobacillus and bifidobacterium in irritable bowel syndrome: Symptom responses and relationship to cytokine profiles. Gastroenterology 2005, 128, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Sinn, D.H.; Song, J.H.; Kim, H.J.; Lee, J.H.; Son, H.J.; Chang, D.K.; Kim, Y.H.; Kim, J.J.; Rhee, J.C.; Rhee, P.L. Therapeutic effect of Lactobacillus acidophilus-SDC 2012, 2013 in patients with irritable bowel syndrome. Dig. Dis. Sci. 2008, 53, 2714–2718. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.A.; Stimpson, J.; Wang, D.; Plummer, S.; Garaiova, I.; Barker, M.E.; Corfe, B.M. Clinical trial: A multistrain probiotic preparation significantly reduces symptoms of irritable bowel syndrome in a double-blind placebo-controlled study. Aliment. Pharmacol. Ther. 2009, 29, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Sondergaard, B.; Olsson, J.; Ohlson, K.; Svensson, U.; Bytzer, P.; Ekesbo, R. Effects of probiotic fermented milk on symptoms and intestinal flora in patients with irritable bowel syndrome: A randomized, placebo-controlled trial. Scand. J. Gastroenterol. 2011, 46, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Li, Y.Q.; Zuo, X.L.; Zhen, Y.B.; Yang, J.; Liu, C.H. Clinical trial: Effect of active lactic acid bacteria on mucosal barrier function in patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharmacol. Ther. 2008, 28, 994–1002. [Google Scholar] [CrossRef] [PubMed]
- Guglielmetti, S.; Mora, D.; Gschwender, M.; Popp, K. Randomised clinical trial: Bifidobacterium bifidum MIMBb75 significantly alleviates irritable bowel syndrome and improves quality of life—A double-blind, placebo-controlled study. Aliment. Pharmacol. Ther. 2011, 33, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Tarrerias, A.L.; Costil, V.; Vicari, F.; Letard, J.C.; Adenis-Lamarre, P.; Aisene, A.; Batistelli, D.; Bonnaud, G.; Carpentier, S.; Dalbies, P.; et al. The effect of inactivated Lactobacillus LB fermented culture medium on symptom severity: Observational investigation in 297 patients with diarrhea-predominant irritable bowel syndrome. Dig. Dis. 2011, 29, 588–591. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Riezzo, G.; Raimondi, F.; Bisceglia, M.; Filannino, A.; Cavallo, L.; Francavilla, R. Lactobacillus reuteri accelerates gastric emptying and improves regurgitation in infants. Eur. J. Clin. Invest. 2011, 41, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Romano, C.; Ferrau, V.; Cavataio, F.; Iacono, G.; Spina, M.; Lionetti, E.; Comisi, F.; Famiani, A.; Comito, D. Lactobacillus reuteri in children with functional abdominal pain (FAP). J. Paediatr. Child Health 2010. [Google Scholar] [CrossRef]
- Salminen, M.K.; Tynkkynen, S.; Rautelin, H.; Poussa, T.; Saxelin, M.; Ristola, M.; Valtonen, V.; Järvinen, A. The efficacy and safety of probiotic Lactobacillus rhamnosus GG on prolonged, noninfectious diarrhea in HIV Patients on antiretroviral therapy: A randomized, placebo-controlled, crossover study. HIV Clin. Trials 2004, 5, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.J.; Wareham, K.; Wang, D.; Bradley, C.; Hutchings, H.; Harris, W.; Dhar, A.; Brown, H.; Foden, A.; Gravenor, M.B. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2013, 382, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Beausoleil, M.; Fortier, N.; Guenette, S.; L’Ecuyer, A.; Savoie, M.; Franco, M.; Lachaine, J.; Weiss, K. Effect of a fermented milk combining Lactobacillus acidophilus Cl1285 and Lactobacillus casei in the prevention of antibiotic-associated diarrhea: A randomized, double-blind, placebo-controlled trial. Can. J. Gastroenterol. 2007, 21, 732–736. [Google Scholar] [PubMed]
- Gao, X.W.; Mubasher, M.; Fang, C.Y.; Reifer, C.; Miller, L.E. Dose-response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile-associated diarrhea prophylaxis in adult patients. Am. J. Gastroenterol. 2010, 105, 1636–1641. [Google Scholar] [CrossRef] [PubMed]
- Pozzoni, P.; Riva, A.; Bellatorre, A.G.; Amigoni, M.; Redaelli, E.; Ronchetti, A.; Stefani, M.; Tironi, R.; Molteni, E.E.; Conte, D.; et al. Saccharomyces boulardii for the prevention of antibiotic-associated diarrhea in adult hospitalized patients: A single-center, randomized, double-blind, placebo-controlled trial. Am. J. Gastroenterol. 2012, 107, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Sampalis, J.; Psaradellis, E.; Rampakakis, E. Efficacy of BIO K+ CL1285 in the reduction of antibiotic-associated diarrhea—A placebo controlled double-blind randomized, multi-center study. Arch. Med. Sci. 2010, 6, 56–64. [Google Scholar] [PubMed]
- Hickson, M.; D’Souza, A.L.; Muthu, N.; Rogers, T.R.; Want, S.; Rajkumar, C.; Bulpitt, C.J. Use of probiotic Lactobacillus preparation to prevent diarrhoea associated with antibiotics: Randomised double blind placebo controlled trial. BMJ 2007, 335, 80. [Google Scholar] [CrossRef] [PubMed]
- Selinger, C.P.; Bell, A.; Cairns, A.; Lockett, M.; Sebastian, S.; Haslam, N. Probiotic VSL#3 prevents antibiotic-associated diarrhoea in a double-blind, randomized, placebo-controlled clinical trial. J. Hosp Infect. 2013, 84, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Grandy, G.; Medina, M.; Soria, R.; Teran, C.G.; Araya, M. Probiotics in the treatment of acute rotavirus diarrhoea. A randomized, double-blind, controlled trial using two different probiotic preparations in Bolivian children. BMC Infect. Dis. 2010, 10, 253. [Google Scholar]
- Navarro-Rodriguez, T.; Silva, F.M.; Barbuti, R.C.; Mattar, R.; Moraes-Filho, J.P.; de Oliveira, M.N.; Bogsan, C.S.; Chinzon, D.; Eisig, J.N. Association of a probiotic to a Helicobacter pylori eradication regimen does not increase efficacy or decreases the adverse effects of the treatment: A prospective, randomized, double-blind, placebo-controlled study. BMC Gastroenterol. 2013, 13, 56. [Google Scholar] [CrossRef] [PubMed]
- Dajani, A.I.; Abu Hammour, A.M.; Yang, D.H.; Chung, P.C.; Nounou, M.A.; Yuan, K.Y.; Zakaria, M.A.; Schi, H.S. Do probiotics improve eradication response to Helicobacter pylori on standard triple or sequential therapy? Saudi J. Gastroenterol. 2013, 19, 113–120. [Google Scholar]
- De Vrese, M.; Kristen, H.; Rautenberg, P.; Laue, C.; Schrezenmeir, J. Probiotic lactobacilli and bifidobacteria in a fermented milk product with added fruit preparation reduce antibiotic associated diarrhea and Helicobacter pylori activity. J. Dairy Res. 2011, 78, 396–403. [Google Scholar]
- Du, Y.Q.; Su, T.; Fan, J.G.; Lu, Y.X.; Zheng, P.; Li, X.H.; Guo, C.Y.; Xu, P.; Gong, Y.F.; Li, Z.S. Adjuvant probiotics improve the eradication effect of triple therapy for Helicobacter pylori infection. World J. Gastroenterol. 2012, 18, 6302–6307. [Google Scholar] [CrossRef] [PubMed]
- Efrati, C.; Nicolini, G.; Cannaviello, C.; O’Sed, N.P.; Valabrega, S. Helicobacter pylori eradication: Sequential therapy and Lactobacillus reuteri supplementation. World J. Gastroenterol. 2012, 18, 6250–6254. [Google Scholar] [CrossRef] [PubMed]
- Mehling, H.; Busjahn, A. Non-viable Lactobacillus reuteri DSMZ 17648 (Pylopass) as a new approach to Helicobacter pylori control in humans. Nutrients 2013, 5, 3062–3073. [Google Scholar] [CrossRef] [PubMed]
- Kuisma, J.; Mentula, S.; Jarvinen, H.; Kahri, A.; Saxelin, M.; Farkkila, M. Effect of Lactobacillus rhamnosus GG on ileal pouch inflammation and microbial flora. Aliment. Pharmacol. Ther. 2003, 17, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Waller, P.A.; Gopal, P.K.; Leyer, G.J.; Ouwehand, A.C.; Reifer, C.; Stewart, M.E.; Miller, L.E. Dose-response effect of Bifidobacterium lactis HN019 on whole gut transit time and functional gastrointestinal symptoms in adults. Scand J. Gastroenterol. 2011, 46, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Kalman, D.S.; Schwartz, H.I.; Alvarez, P.; Feldman, S.; Pezzullo, J.C.; Krieger, D.R. A prospective, randomized, double-blind, placebo-controlled parallel-group dual site trial to evaluate the effects of a Bacillus coagulans-based product on functional intestinal gas symptoms. BMC Gastroenterol. 2009, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Kondo, J.; Xiao, J.Z.; Shirahata, A.; Baba, M.; Abe, A.; Ogawa, K.; Shimoda, T. Modulatory effects of Bifidobacterium longum BB536 on defecation in elderly patients receiving enteral feeding. World J. Gastroenterol. 2013, 19, 2162–2170. [Google Scholar] [CrossRef] [PubMed]
- Guyonnet, D.; Schlumberger, A.; Mhamdi, L.; Jakob, S.; Chassany, O. Fermented milk containing Bifidobacterium lactis DN-173 010 improves gastrointestinal well-being and digestive symptoms in women reporting minor digestive symptoms: A randomised, double-blind, parallel, controlled study. Br. J. Nutr. 2009, 102, 1654–1662. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Guyonnet, D.; Lafaye de Micheaux, P.; Gelu, S. A randomized, double-blind, controlled study and pooled analysis of two identical trials of fermented milk containing probiotic Bifidobacterium lactis CNCM I-2494 in healthy women reporting minor digestive symptoms. Neurogastroenterol. Motil. 2013, 25, 331–e252. [Google Scholar] [CrossRef] [PubMed]
- Sari, F.N.; Dizdar, E.A.; Oguz, S.; Erdeve, O.; Uras, N.; Dilmen, U. Oral probiotics: Lactobacillus sporogenes for prevention of necrotizing enterocolitis in very low-birth weight infants: A randomized, controlled trial. Eur. J. Clin. Nutr. 2011, 65, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Stratiki, Z.; Costalos, C.; Sevastiadou, S.; Kastanidou, O.; Skouroliakou, M.; Giakoumatou, A.; Petrohilou, V. The effect of a bifidobacter supplemented bovine milk on intestinal permeability of preterm infants. Early Hum. Dev. 2007, 83, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; di Mauro, A.; Riezzo, G.; Civardi, E.; Intini, C.; Corvaglia, L.; Ballardini, E.; Bisceglia, M.; Cinquetti, M.; Brazzoduro, E.; et al. Prophylactic use of a probiotic in the prevention of colic, regurgitation, and functional constipation: A randomized clinical trial. JAMA Pediatr. 2014, 168, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; de Simone, T.; Federico, A.; Terracciano, F.; Tuccillo, C.; di Chicco, M.; Carteni, M. Gut-liver axis: A new point of attack to treat chronic liver damage? Am. J. Gastroenterol. 2002, 97, 2144–2146. [Google Scholar]
- Polson, J.; Lee, W.M. AASLD position paper: The management of acute liver failure. Hepatology 2005, 41, 1179–1197. [Google Scholar] [CrossRef] [PubMed]
- Kirpich, I.A.; Solovieva, N.V.; Leikhter, S.N.; Shidakova, N.A.; Lebedeva, O.V.; Sidorov, P.I.; Bazhukova, T.A.; Soloviev, A.G.; Barve, S.S.; McClain, C.J.; et al. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: A pilot study. Alcohol 2008, 42, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Stadlbauer, V.; Mookerjee, R.P.; Hodges, S.; Wright, G.A.; Davies, N.A.; Jalan, R. Effect of probiotic treatment on deranged neutrophil function and cytokine responses in patients with compensated alcoholic cirrhosis. J. Hepatol. 2008, 48, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Federico, A.; Tuccillo, C.; Terracciano, F.; D’Auria, M.V.; de Simone, C.; del Vecchio Blanco, C. Beneficial effects of a probiotic VSL#3 on parameters of liver dysfunction in chronic liver diseases. J. Clin. Gastroenterol. 2005, 39, 540–543. [Google Scholar] [CrossRef] [PubMed]
- Lata, J.; Jurankova, J.; Pribramska, V.; Fric, P.; Senkyrik, M.; Dite, P.; Kroupa, R. Effect of administration of Escherichia coli Nissle (Mutaflor) on intestinal colonisation, endo-toxemia, liver function and minimal hepatic encephalopathy in patients with liver cirrhosis. Vnitr. Lek. 2006, 52, 215–219. [Google Scholar] [PubMed]
- Lata, J.; Novotny, I.; Pribramska, V.; Jurankova, J.; Fric, P.; Kroupa, R.; Stiburek, O. The effect of probiotics on gut flora, level of endotoxin and Child-Pugh score in cirrhotic patients: Results of a double-blind randomized study. Eur. J. Gastroenterol. Hepatol. 2007, 19, 1111–1113. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.E.; Zhang, Y.; Zhang, J.; Dong, P.L.; Chen, M.; Duan, Z.P. Probiotic yogurt effects on intestinal flora of patients with chronic liver disease. Nurs. Res. 2010, 59, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Pereg, D.; Kotliroff, A.; Gadoth, N.; Hadary, R.; Lishner, M.; Kitay-Cohen, Y. Probiotics for patients with compensated liver cirrhosis: A double-blind placebo-controlled study. Nutrition 2011, 27, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.Y.; Wang, H.J.; Lu, Z.; Xu, S.Z. Intestinal microflora in patients with liver cirrhosis. Chin. J. Dig. Dis. 2004, 5, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Tandon, P.; Moncrief, K.; Madsen, K.; Arrieta, M.C.; Owen, R.J.; Bain, V.G.; Wong, W.W.; Ma, M.M. Effects of probiotic therapy on portal pressure in patients with cirrhosis: A pilot study. Liver Int. 2009, 29, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Duan, Z.P.; Ha, D.K.; Bengmark, S.; Kurtovic, J.; Riordan, S.M. Synbiotic modulation of gut flora: Effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology 2004, 39, 1441–1449. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Greco, F.; Barone, G.; Gargante, M.P.; Malaguarnera, M.; Toscano, M.A. Bifidobacterium longum with fructo-oligosaccharide (FOS) treatment in minimal hepatic encephalopathy: A randomized, double-blind, placebo-controlled study. Dig. Dis. Sci. 2007, 52, 3259–3265. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.; Gibson, G.R. Cholesterol assimilation by lactic acid bacteria and bifidobacteria isolated from the human gut. Appl. Environ. Microbiol. 2002, 68, 4689–4693. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Lund, P.K. Role of intestinal inflammation as an early event in obesity and insulin resistance. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Chorell, E.; Karlsson Videhult, F.; Hernell, O.; Antti, H.; West, C.E. Impact of probiotic feeding during weaning on the serum lipid profile and plasma metabolome in infants. Br. J. Nutr. 2013, 110, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Kadooka, Y.; Sato, M.; Imaizumi, K.; Ogawa, A.; Ikuyama, K.; Akai, Y.; Okano, M.; Kagoshima, M.; Tsuchida, T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur. J. Clin. Nutr. 2010, 64, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Kalliomaki, M.; Laitinen, K.; Isolauri, E. The impact of perinatal probiotic intervention on the development of overweight and obesity: Follow-up study from birth to 10 years. Int. J. Obes. (Lond) 2010, 34, 1531–1537. [Google Scholar] [CrossRef]
- Zarrati, M.; Shidfar, F.; Nourijelyani, K.; Mofid, V.; Hossein zadeh-Attar, M.J.; Bidad, K.; Najafi, F.; Gheflati, Z.; Chamari, M.; Salehi, E. Lactobacillus acidophilus La5, Bifidobacterium BB12, and Lactobacillus casei DN001 modulate gene expression of subset specific transcription factors and cytokines in peripheral blood mononuclear cells of obese and overweight people. Biofactors 2013, 39, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Safavi, M.; Farajian, S.; Kelishadi, R.; Mirlohi, M.; Hashemipour, M. The effects of synbiotic supplementation on some cardio-metabolic risk factors in overweight and obese children: A randomized triple-masked controlled trial. Int. J. Food Sci. Nutr. 2013, 64, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Tien, M.T.; Girardin, S.E.; Regnault, B.; le Bourhis, L.; Dillies, M.A.; Coppée, J.Y.; Bourdet-Sicard, R.; Sansonetti, P.J.; Pédron, T. Anti-inflammatory effect of Lactobacillus casei on Shigella-infected human intestinal epithelial cells. J. Immunol. 2006, 176, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.K.; Omaye, S.T. Metabolic diseases and pro- and prebiotics: Mechanistic insights. Nutr. Metab. (Lond.) 2012, 9, 60. [Google Scholar] [CrossRef]
- Amaral, F.A.; Sachs, D.; Costa, V.V.; Fagundes, C.T.; Cisalpino, D.; Cunha, T.M.; Ferreira, S.H.; Cunha, F.Q.; Silva, T.A.; Nicoli, J.R. Commensal microbiota is fundamental for the development of inflammatory pain. Proc. Natl. Acad. Sci. USA 2008, 105, 2193–2197. [Google Scholar] [CrossRef] [PubMed]
- Diaz Heijtz, R.; Wang, S.; Anuar, F.; Qian, Y.; Björkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255–264, e119. [Google Scholar]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Williams, C.; Brown, A. Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur. J. Clin. Nutr. 2007, 61, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.V.; Bested, A.C.; Beaulne, T.M.; Katzman, M.A.; Iorio, C.; Berardi, J.M.; Logan, A.C. A randomized, double-blind, placebo-controlled pilot study of a probiotic in emotional symptoms of chronic fatigue syndrome. Gut Pathog. 2009, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, M.; Lalonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M.; et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Zhu, J.C.; Du, J.; Zhang, L.M.; Yin, H.H. Effects of probiotics on serum levels of Th1/Th2 cytokine and clinical outcomes in severe traumatic brain-injured patients: A prospective randomized pilot study. Crit. Care 2011, 15, R290. [Google Scholar] [CrossRef] [PubMed]
- Tillisch, K.; Labus, J.; Kilpatrick, L.; Jiang, Z.; Stains, J.; Ebrat, B.; Guyonnet, D.; Legrain-Raspaud, S.; Trotin, B.; Naliboff, B.; et al. Consumption of fermented milk product with probiotic modulates brain activity. Gastroenterology 2013, 144, 1394–1401. [Google Scholar]
- Vitetta, L.; Gobe, G. Uremia and chronic kidney disease: The role of the gut microflora and therapies with pro- and prebiotics. Mol. Nutr. Food Res. 2013, 57, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Alford, H. The Pharmacobiotic Potential of the Gastrointestinal Tract Micro-Biometabolome–Probiotic Connect: A Brief Commentary. Drug Dev. Res. 2013. [Google Scholar] [CrossRef]
- Ranganathan, N.; Friedman, E.A.; Tam, P.; Rao, V.; Ranganathan, P.; Dheer, R. Probiotic dietary supplementation in patients with stage 3 and 4 chronic kidney disease: A 6-month pilot scale trial in Canada. Curr. Med. Res. Opin. 2009, 25, 1919–1930. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, N.; Ranganathan, P.; Friedman, E.A.; Joseph, A.; Delano, B.; Goldfarb, D.S.; Tam, P.; Rao, A.V.; Anteyi, E.; Musso, C.G. Pilot study of probiotic dietary supplementation for promoting healthy kidney function in patients with chronic kidney disease. Adv. Ther. 2010, 27, 634–647. [Google Scholar] [PubMed]
- Nakabayashi, I.; Nakamura, M.; Kawakami, K.; Ohta, T.; Kato, I.; Uchida, K.; Yoshida, M. Effects of synbiotic treatment on serum level of p-cresol in haemodialysis patients: A preliminary study. Nephrol. Dial. Transplant. 2011, 26, 1094–1098. [Google Scholar] [CrossRef] [PubMed]
- Chong, V.H.; Wang, C.L. Higher prevalence of gastrointestinal symptoms among patients with rheumatic disorders. Singap. Med. J. 2008, 49, 419–424. [Google Scholar]
- Hatakka, K.; Martio, J.; Korpela, M.; Herranen, M.; Poussa, T.; Laasanen, T.; Saxelin, M.; Vapaatalo, H.; Moilanen, E.; Korpela, R. Effects of probiotic therapy on the activity and activation of mild rheumatoid arthritis—A pilot study. Scand J. Rheumatol. 2003, 32, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Waller, R.D.; Stebbings, S.; Highton, J.; Orlovich, D.A.; Schmierer, D.; Fawcett, J.P. The effects of an orally administered probiotic on sulfasalazine metabolism in individuals with rheumatoid arthritis: A preliminary study. Int. J. Rheum. Dis. 2010, 13, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Mandel, D.R.; Eichas, K.; Holmes, J. Bacillus coagulans: A viable adjunct therapy for relieving symptoms of rheumatoid arthritis according to a randomized, controlled trial. BMC Complement. Altern. Med. 2010, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Pineda Mde, L.; Thompson, S.F.; Summers, K.; de Leon, F.; Pope, J.; Reid, G. A randomized, double-blinded, placebo-controlled pilot study of probiotics in active rheumatoid arthritis. Med. Sci. Monit. 2011, 17, CR347–CR354. [Google Scholar]
- Jenks, K.; Stebbings, S.; Burton, J.; Schultz, M.; Herbison, P.; Highton, J. Probiotic therapy for the treatment of spondyloarthritis: A randomized controlled trial. J. Rheumatol. 2010, 37, 2118–2125. [Google Scholar] [CrossRef] [PubMed]
- So, J.S.; Song, M.K.; Kwon, H.K.; Lee, C.G.; Chae, C.S.; Sahoo, A.; Jash, A.; Lee, S.H.; Park, Z.Y.; Im, S.H. Lactobacillus casei enhances type II collagen/glucosamine-mediated suppression of inflammatory responses in experimental osteoarthritis. Life Sci. 2011, 88, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Sironi, M.; Clerici, M. The hygiene hypothesis: An evolutionary perspective. Microbes Infect. 2010, 12, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.S.; Lin, Y.L.; Jan, R.L.; Chen, H.H.; Wang, J.Y. Randomized placebo-controlled trial of lactobacillus on asthmatic children with allergic rhinitis. Pediatr. Pulmonol. 2010, 45, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Dolle, S.; Berg, J.; Rasche, C.; Worm, M. Tolerability and clinical outcome of coseasonal treatment with Escherichia coli strain Nissle 1917 in grass pollen-allergic subjects. Int. Arch. Allergy Immunol. 2014, 163, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Giovannini, M.; Agostoni, C.; Riva, E.; Salvini, F.; Ruscitto, A.; Zuccotti, G.V.; Radaelli, G.; Felicita Study Group. A randomized prospective double blind controlled trial on effects of long-term consumption of fermented milk containing Lactobacillus casei in pre-school children with allergic asthma and/or rhinitis. Pediatr. Res. 2007, 62, 215–220. [Google Scholar]
- Ishida, Y.; Nakamura, F.; Kanzato, H.; Sawada, D.; Hirata, H.; Nishimura, A.; Kajimoto, O.; Fujiwara, S. Clinical effects of Lactobacillus acidophilus strain L-92 on perennial allergic rhinitis: A double-blind, placebo-controlled study. J. Dairy Sci. 2005, 88, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Ivory, K.; Chambers, S.J.; Pin, C.; Prieto, E.; Arqués, J.L.; Nicoletti, C. Oral delivery of Lactobacillus casei Shirota modifies allergen-induced immune responses in allergic rhinitis. Clin. Exp. Allergy 2008, 38, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
- Kopp, M.V.; Hennemuth, I.; Heinzmann, A.; Urbanek, R. Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: No clinical effects of Lactobacillus GG supplementation. Pediatrics 2008, 121, e850–e856. [Google Scholar] [CrossRef] [PubMed]
- Kuitunen, M.; Kukkonen, K.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Haahtela, T.; Savilahti, E. Probiotics prevent IgE-associated allergy until age 5 years in cesarean-delivered children but not in the total cohort. J Allergy Clin. Immunol. 2009, 123, 335–341. [Google Scholar]
- Kumpu, M.; Kekkonen, R.A.; Kautiainen, H.; Jarvenpaa, S.; Kristo, A.; Huovinen, P.; Pitkäranta, A.; Korpela, R.; Hatakka, K. Milk containing probiotic Lactobacillus rhamnosus GG and respiratory illness in children: A randomized, double-blind, placebo-controlled trial. Eur. J. Clin. Nutr. 2012, 66, 1020–1023. [Google Scholar]
- Odamaki, T.; Xiao, J.Z.; Iwabuchi, N.; Sakamoto, M.; Takahashi, N.; Kondo, S.; Miyaji, K.; Iwatsuki, K.; Togashi, H.; Enomoto, T.; et al. Influence of Bifidobacterium longum BB536 intake on faecal microbiota in individuals with Japanese cedar pollinosis during the pollen season. J. Med. Microbiol. 2007, 56, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S.; Salminen, S.; Isolauri, E. Specific probiotics in reducing the risk of acute infections in infancy—A randomised, double-blind, placebo-controlled study. Br. J. Nutr. 2009, 101, 1722–1726. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Hacini-Rachinel, F.; Gosoniu, M.L.; Bourdeau, T.; Holvoet, S.; Doucet-Ladeveze, R.; Beaumont, M.; Mercenier, A.; Nutten, S. Immune-modulatory effect of probiotic Bifidobacterium lactis NCC2818 in individuals suffering from seasonal allergic rhinitis to grass pollen: An exploratory, randomized, placebo-controlled clinical trial. Eur. J. Clin. Nutr. 2013, 67, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Wassenberg, J.; Nutten, S.; Audran, R.; Barbier, N.; Aubert, V.; Moulin, J.; Mercenier, A.; Spertini, F. Effect of Lactobacillus paracasei ST11 on a nasal provocation test with grass pollen in allergic rhinitis. Clin. Exp. Allergy 2011, 41, 565–573. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, T.T.; di Sabatino, A. The exposure of infants to Lactobacillus rhamnosus GG in Finland. J. Pediatr. Gastroenterol. Nutr. 2006, 42, 476–478. [Google Scholar] [CrossRef] [PubMed]
- Bouilly-Gauthier, D.; Jeannes, C.; Maubert, Y.; Duteil, L.; Queille-Roussel, C.; Piccardi, N.; Montastier, C.; Manissier, P.; Piérard, G.; Ortonne, J.P. Clinical evidence of benefits of a dietary supplement containing probiotic and carotenoids on ultraviolet-induced skin damage. Br. J. Dermatol. 2010, 163, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Kalliomaki, M.; Salminen, S.; Poussa, T.; Isolauri, E. Probiotics during the first 7 years of life: A cumulative risk reduction of eczema in a randomized, placebo-controlled trial. J. Allergy Clin. Immunol. 2007, 119, 1019–1021. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, K.; Savilahti, E.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Kuitunen, M. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: A randomized, double-blind, placebo-controlled trial. J. Allergy Clin. Immunol. 2007, 119, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Marschan, E.; Kuitunen, M.; Kukkonen, K.; Poussa, T.; Sarnesto, A.; Haahtela, T.; Korpela, R.; Savilahti, E.; Vaarala, O. Probiotics in infancy induce protective immune profiles that are characteristic for chronic low-grade inflammation. Clin. Exp. Allergy 2008, 38, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, A.; Abrahamsson, T.R.; Bjorksten, B.; Jenmalm, M.C. Pre- and post-natal Lactobacillus reuteri supplementation decreases allergen responsiveness in infancy. Clin. Exp. Allergy 2013, 43, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S.; Kainonen, E.; Salminen, S.; Isolauri, E. Maternal probiotic supplementation during pregnancy and breast-feeding reduces the risk of eczema in the infant. J. Allergy Clin. Immunol. 2012, 130, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Gerasimov, S.V.; Vasjuta, V.V.; Myhovych, O.O.; Bondarchuk, L.I. Probiotic supplement reduces atopic dermatitis in preschool children: A randomized, double-blind, placebo-controlled, clinical trial. Am. J. Clin. Dermatol. 2010, 11, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Hattori, K.; Yamamoto, A.; Sasai, M.; Taniuchi, S.; Kojima, T.; Kobayashi, Y.; Iwamoto, H.; Namba, K.; Yaeshima, T. Effects of administration of bifidobacteria on fecal microflora and clinical symptoms in infants with atopic dermatitis. Arerugi 2003, 52, 20–30. [Google Scholar] [PubMed]
- Kalliomaki, M.; Salminen, S.; Arvilommi, H.; Kero, P.; Koskinen, P.; Isolauri, E. Probiotics in primary prevention of atopic disease: A randomised placebo-controlled trial. Lancet 2001, 357, 1076–1079. [Google Scholar] [CrossRef] [PubMed]
- Kalliomaki, M.; Salminen, S.; Poussa, T.; Arvilommi, H.; Isolauri, E. Probiotics and prevention of atopic disease: 4-Year follow-up of a randomised placebo-controlled trial. Lancet 2003, 361, 1869–1871. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kwon, J.H.; Ahn, S.H.; Lee, S.I.; Han, Y.S.; Choi, Y.O.; Lee, S.Y.; Ahn, K.M.; Ji, G.E. Effect of probiotic mix (Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus) in the primary prevention of eczema: A double-blind, randomized, placebo-controlled trial. Pediatr. Allergy Immunol. 2010, 21, e386–e393. [Google Scholar] [CrossRef] [PubMed]
- Niers, L.; Martin, R.; Rijkers, G.; Sengers, F.; Timmerman, H.; van Uden, N.; Smidt, H.; Kimpen, J.; Hoekstra, M. The effects of selected probiotic strains on the development of eczema (the PandA study). Allergy 2009, 64, 1349–1358. [Google Scholar]
- Sistek, D.; Kelly, R.; Wickens, K.; Stanley, T.; Fitzharris, P.; Crane, J. Is the effect of probiotics on atopic dermatitis confined to food sensitized children? Clin. Exp. Allergy 2006, 36, 629–633. [Google Scholar] [CrossRef]
- Viljanen, M.; Kuitunen, M.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Savilahti, E. Probiotic effects on faecal inflammatory markers and on faecal IgA in food allergic atopic eczema/dermatitis syndrome infants. Pediatr. Allergy Immunol. 2005, 16, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Weston, S.; Halbert, A.; Richmond, P.; Prescott, S.L. Effects of probiotics on atopic dermatitis: A randomised controlled trial. Arch. Dis. Child. 2005, 90, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Wickens, K.; Black, P.; Stanley, T.V.; Mitchell, E.; Barthow, C.; Fitzharris, P.; Purdie, G.; Crane, J. A protective effect of Lactobacillus rhamnosus HN001 against eczema in the first 2 years of life persists to age 4 years. Clin. Exp. Allergy 2012, 42, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Wickens, K.; Black, P.N.; Stanley, T.V.; Mitchell, E.; Fitzharris, P.; Tannock, G.W.; Purdie, G.; Crane, J.; Probiotic Study Group. A differential effect of 2 probiotics in the prevention of eczema and atopy: A double-blind, randomized, placebo-controlled trial. J. Allergy Clin. Immunol. 2008, 122, 788–794. [Google Scholar]
- Shornikova, A.V.; Casas, I.A.; Isolauri, E.; Mykkanen, H.; Vesikari, T. Lactobacillus reuteri as a therapeutic agent in acute diarrhea in young children. J. Pediatr. Gastroenterol. Nutr. 1997, 24, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Saxelin, M. Probiotic formulations and applications, the current probiotics market, and changes in the marketplace: A European perspective. Clin. Infect Dis. 2008, 46, S76–S79. [Google Scholar] [CrossRef] [PubMed]
- Hatakka, K.; Savilahti, E.; Ponka, A.; Meurman, J.H.; Poussa, T.; Näse, L.; Saxelin, M.; Korpela, R. Effect of long term consumption of probiotic milk on infections in children attending day care centres: Double blind, randomised trial. BMJ 2001, 322, 1327. [Google Scholar] [CrossRef] [PubMed]
- Prilassnig, M.; Wenisch, C.; Daxboeck, F.; Feierl, G. Are probiotics detectable in human feces after oral uptake by healthy volunteers? Wien. Klin. Wochenschr. 2007, 119, 456–462. [Google Scholar] [CrossRef]
- Saxelin, M.; Pessi, T.; Salminen, S. Fecal recovery following oral administration of Lactobacillus Strain GG (ATCC 53103) in gelatine capsules to healthy volunteers. Int. J. Food Microbiol. 1995, 25, 199–203. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vitetta, L.; Manuel, R.; Zhou, J.Y.; Linnane, A.W.; Hall, S.; Coulson, S. The Overarching Influence of the Gut Microbiome on End-Organ Function: The Role of Live Probiotic Cultures. Pharmaceuticals 2014, 7, 954-989. https://doi.org/10.3390/ph7090954
Vitetta L, Manuel R, Zhou JY, Linnane AW, Hall S, Coulson S. The Overarching Influence of the Gut Microbiome on End-Organ Function: The Role of Live Probiotic Cultures. Pharmaceuticals. 2014; 7(9):954-989. https://doi.org/10.3390/ph7090954
Chicago/Turabian StyleVitetta, Luis, Rachel Manuel, Joyce Yusi Zhou, Anthony W. Linnane, Sean Hall, and Samantha Coulson. 2014. "The Overarching Influence of the Gut Microbiome on End-Organ Function: The Role of Live Probiotic Cultures" Pharmaceuticals 7, no. 9: 954-989. https://doi.org/10.3390/ph7090954
APA StyleVitetta, L., Manuel, R., Zhou, J. Y., Linnane, A. W., Hall, S., & Coulson, S. (2014). The Overarching Influence of the Gut Microbiome on End-Organ Function: The Role of Live Probiotic Cultures. Pharmaceuticals, 7(9), 954-989. https://doi.org/10.3390/ph7090954