Contribution of [18F]FET PET in the Management of Gliomas, from Diagnosis to Follow-Up: A Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Synthesizing
3. Results
3.1. Literature Search
3.2. Diagnosis
3.3. Grading
3.4. IDH Status Determination
3.5. Prediction of Oligodendroglial Components
3.6. Guided Resection or Biopsy
3.7. Detection of Residual Tumor
3.8. Guided Radiotherapy
3.9. Detection of Malignant Transformation in Low-Grade Gliomas
3.10. Recurrence vs. Treatment-Related Changes
3.11. Prognosis and Treatment Response Evaluation
3.12. Radiomics
4. Discussion/Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Darlix, A.; Zouaoui, S.; Rigau, V.; Bessaoud, F.; Figarella-Branger, D.; Mathieu-Daudé, H.; Trétarre, B.; Bauchet, F.; Duffau, H.; Taillandier, L.; et al. Epidemiology for primary brain tumors: A nationwide population-based study. J. Neuro Oncol. 2017, 131, 525–546. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; Van Den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Brada, M.; Van Den Bent, M.J.; Tonn, J.-C.; Pentheroudakis, G. High-grade glioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2014, 25, iii93–iii101. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.Y.; Macdonald, D.R.; Reardon, D.A.; Cloughesy, T.F.; Sorensen, A.G.; Galanis, E.; DeGroot, J.; Wick, W.; Gilbert, M.R.; Lassman, A.B.; et al. Updated Response Assessment Criteria for High-Grade Gliomas: Response Assessment in Neuro-Oncology Working Group. J. Clin. Oncol. 2010, 28, 1963–1972. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; He, Q.; Zhang, B.; Li, N.; Zeng, X.; Li, W. Diagnostic accuracy of diffusion-weighted imaging in differentiating glioma recurrence from posttreatment-related changes: A meta-analysis. Expert. Rev. Anticancer Ther. 2022, 22, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Y.; Wang, Y.; Xiao, H.; Chen, X.; Lei, Y.; Feng, Z.; Ma, X.; Ma, L. Perfusion magnetic resonance imaging in the differentiation between glioma recurrence and pseudoprogression: A systematic review, meta-analysis and meta-regression. Quant. Imaging Med. Surg. 2022, 12, 4805–4822. [Google Scholar] [CrossRef]
- El-Abtah, M.E.; Talati, P.; Fu, M.; Chun, B.; Clark, P.; Peters, A.; Ranasinghe, A.; He, J.; Rapalino, O.; Batchelor, T.T.; et al. Magnetic resonance spectroscopy outperforms perfusion in distinguishing between pseudoprogression and disease progression in patients with glioblastoma. Neuro Oncol. Adv. 2022, 4, vdac128. [Google Scholar] [CrossRef]
- Law, I.; Albert, N.L.; Arbizu, J.; Boellaard, R.; Drzezga, A.; Galldiks, N.; la Fougère, C.; Langen, K.-J.; Lopci, E.; Lowe, V.; et al. Joint EANM/EANO/RANO practice guidelines/SNMMI procedure standards for imaging of gliomas using PET with radiolabelled amino acids and [18F]FDG: Version 1.0. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 540–557. [Google Scholar] [CrossRef]
- Galldiks, N.; Lohmann, P.; Fink, G.R.; Langen, K.-J. Amino Acid PET in Neurooncology. J. Nucl. Med. 2023, 64, 693–700. [Google Scholar] [CrossRef]
- He, Q.; Zhang, L.; Zhang, B.; Shi, X.; Yi, C.; Zhang, X. Diagnostic accuracy of 13N-ammonia PET, 11C-methionine PET and 18F-fluorodeoxyglucose PET: A comparative study in patients with suspected cerebral glioma. BMC Cancer 2019, 19, 332. [Google Scholar] [CrossRef]
- Deuschl, C.; Kirchner, J.; Poeppel, T.D.; Schaarschmidt, B.; Kebir, S.; El Hindy, N.; Hense, J.; Quick, H.H.; Glas, M.; Herrmann, K.; et al. 11C–MET PET/MRI for detection of recurrent glioma. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Kracht, L.W.; Burghaus, L.; Thomas, A.; Jacobs, A.H.; Heiss, W.; Herholz, K. Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, R.; Kimura, K.; Abe, K.; Sakai, S. 11C-methionine PET/CT findings in benign brain disease. Jpn. J. Radiol. 2017, 35, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Karunanithi, S.; Sharma, P.; Kumar, A.; Khangembam, B.C.; Bandopadhyaya, G.P.; Kumar, R.; Goenka, A.; Gupta, D.K.; Malhotra, A.; Bal, C. Comparative diagnostic accuracy of contrast-enhanced MRI and 18F-FDOPA PET-CT in recurrent glioma. Eur. Radiol. 2013, 23, 2628–2635. [Google Scholar] [CrossRef] [PubMed]
- Youland, R.S.; Pafundi, D.H.; Brinkmann, D.H.; Lowe, V.J.; Morris, J.M.; Kemp, B.J.; Hunt, C.H.; Giannini, C.; Parney, I.F.; Laack, N.N. Prospective trial evaluating the sensitivity and specificity of 3,4-dihydroxy-6-[18F]-fluoro-L-phenylalanine (18F-DOPA) PET and MRI in patients with recurrent gliomas. J. Neuro Oncol. 2018, 137, 583–591. [Google Scholar] [CrossRef]
- Sala, Q.; Metellus, P.; Taieb, D.; Kaphan, E.; Figarella-Branger, D.; Guedj, E. 18F-DOPA, a Clinically Available PET Tracer to Study Brain Inflammation? Clin. Nucl. Med. 2014, 39, e283–e285. [Google Scholar] [CrossRef]
- Hutterer, M.; Nowosielski, M.; Putzer, D.; Jansen, N.L.; Seiz, M.; Schocke, M.; McCoy, M.; Göbel, G.; la Fougère, C.; Virgolini, I.J.; et al. [18F]-fluoro-ethyl-l-tyrosine PET: A valuable diagnostic tool in neuro-oncology, but not all that glitters is glioma. Neuro Oncol. 2013, 15, 341–351. [Google Scholar] [CrossRef]
- Pöpperl, G.; Kreth, F.W.; Mehrkens, J.H.; Herms, J.; Seelos, K.; Koch, W.; Gildehaus, F.J.; Kretzschmar, H.A.; Tonn, J.C.; Tatsch, K. FET PET for the evaluation of untreated gliomas: Correlation of FET uptake and uptake kinetics with tumour grading. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 1933–1942. [Google Scholar] [CrossRef]
- Langen, K.-J.; Stoffels, G.; Filss, C.; Heinzel, A.; Stegmayr, C.; Lohmann, P.; Willuweit, A.; Neumaier, B.; Mottaghy, F.M.; Galldiks, N. Imaging of amino acid transport in brain tumours: Positron emission tomography with O-(2-[18F]fluoroethyl)- L -tyrosine (FET). Methods 2017, 130, 124–134. [Google Scholar] [CrossRef]
- Yu, P.; Wang, Y.; Su, F.; Chen, Y. Comparing [18F]FET PET and [18F]FDOPA PET for glioma recurrence diagnosis: A systematic review and meta-analysis. Front. Oncol. 2024, 13, 1346951. [Google Scholar] [CrossRef]
- Cui, M.; Zorrilla-Veloz, R.I.; Hu, J.; Guan, B.; Ma, X. Diagnostic Accuracy of PET for Differentiating True Glioma Progression From Post Treatment-Related Changes: A Systematic Review and Meta-Analysis. Front. Neurol. 2021, 12, 671867. [Google Scholar] [CrossRef]
- Ouyang, Z.-Q.; Zheng, G.-R.; Duan, X.-R.; Zhang, X.-R.; Ke, T.-F.; Bao, S.-S.; Yang, J.; He, B.; Liao, C.-D. Diagnostic accuracy of glioma pseudoprogression identification with positron emission tomography imaging: A systematic review and meta-analysis. Quant. Imaging Med. Surg. 2023, 13, 4943–4959. [Google Scholar] [CrossRef] [PubMed]
- Pauleit, D.; Stoffels, G.; Bachofner, A.; Floeth, F.W.; Sabel, M.; Herzog, H.; Tellmann, L.; Jansen, P.; Reifenberger, G.; Hamacher, K.; et al. Comparison of 18F-FET and 18F-FDG PET in brain tumors. Nucl. Med. Biol. 2009, 36, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Mauler, J.; Lohmann, P.; Maudsley, A.A.; Sheriff, S.; Hoevels, M.; Meissner, A.-K.; Hamisch, C.; Brunn, A.; Deckert, M.; Filss, C.P.; et al. Diagnostic Accuracy of MR Spectroscopic Imaging and 18F-FET PET for Identifying Glioma: A Biopsy-Controlled Hybrid PET/MRI Study. J. Nucl. Med. 2024, 65, 16–21. [Google Scholar] [CrossRef]
- Floeth, F.W.; Pauleit, D.; Wittsack, H.-J.; Langen, K.J.; Reifenberger, G.; Hamacher, K.; Messing-Jünger, M.; Zilles, K.; Weber, F.; Stummer, W.; et al. Multimodal metabolic imaging of cerebral gliomas: Positron emission tomography with [18F]fluoroethyl-l-tyrosine and magnetic resonance spectroscopy. J. Neurosurg. 2005, 102, 318–327. [Google Scholar] [CrossRef]
- Pauleit, D. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain 2005, 128, 678–687. [Google Scholar] [CrossRef]
- Jeong, S.Y.; Lim, S.M. Comparison of 3′-deoxy-3′-[18F]fluorothymidine PET and O-(2-[18F]fluoroethyl)-L-tyrosine PET in patients with newly diagnosed glioma. Nucl. Med. Biol. 2012, 39, 977–981. [Google Scholar] [CrossRef]
- Verger, A.; Filss, C.P.; Lohmann, P.; Stoffels, G.; Sabel, M.; Wittsack, H.J.; Kops, E.R.; Galldiks, N.; Fink, G.R.; Shah, N.J.; et al. Comparison of 18F-FET PET and perfusion-weighted MRI for glioma grading: A hybrid PET/MR study. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 2257–2265. [Google Scholar] [CrossRef]
- Lopez, W.O.C.; Cordeiro, J.G.; Albicker, U.; Doostkam, S.; Nikkhah, G.; Kirch, R.D.; Trippel, M.; Reithmeier, T. Correlation of 18F-fluoroethyl tyrosine positron-emission tomography uptake values and histomorphological findings by stereotactic serial biopsy in newly diagnosed brain tumors using a refined software tool. Onco Targets Ther. 2015, 8, 3803–3815. [Google Scholar] [CrossRef]
- Lohmann, P.; Herzog, H.; Rota Kops, E.; Stoffels, G.; Judov, N.; Filss, C.; Galldiks, N.; Tellmann, L.; Weiss, C.; Sabel, M.; et al. Dual-time-point O-(2-[18F]fluoroethyl)-L-tyrosine PET for grading of cerebral gliomas. Eur. Radiol. 2015, 25, 3017–3024. [Google Scholar] [CrossRef] [PubMed]
- Calcagni, M.L.; Galli, G.; Giordano, A.; Taralli, S.; Anile, C.; Niesen, A.; Baum, R.P. Dynamic O-(2-[18F]fluoroethyl)-L-tyrosine (F-18 FET) PET for Glioma Grading: Assessment of Individual Probability of Malignancy. Clin. Nucl. Med. 2011, 36, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.L.; Winkelmann, I.; Suchorska, B.; Wenter, V.; Schmid-Tannwald, C.; Mille, E.; Todica, A.; Brendel, M.; Tonn, J.-C.; Bartenstein, P.; et al. Early static 18F-FET-PET scans have a higher accuracy for glioma grading than the standard 20–40 min scans. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Hua, T.; Zhou, W.; Zhou, Z.; Guan, Y.; Li, M. Heterogeneous parameters based on 18F-FET PET imaging can non-invasively predict tumor grade and isocitrate dehydrogenase gene 1 mutation in untreated gliomas. Quant. Imaging Med. Surg. 2021, 11, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Kunz, M.; Thon, N.; Eigenbrod, S.; Hartmann, C.; Egensperger, R.; Herms, J.; Geisler, J.; la Fougere, C.; Lutz, J.; Linn, J.; et al. Hot spots in dynamic18FET-PET delineate malignant tumor parts within suspected WHO grade II gliomas. Neuro Oncol. 2011, 13, 307–316. [Google Scholar] [CrossRef]
- Röhrich, M.; Huang, K.; Schrimpf, D.; Albert, N.L.; Hielscher, T.; Von Deimling, A.; Schüller, U.; Dimitrakopoulou-Strauss, A.; Haberkorn, U. Integrated analysis of dynamic FET PET/CT parameters, histology, and methylation profiling of 44 gliomas. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1573–1584. [Google Scholar] [CrossRef]
- Jansen, N.L.; Graute, V.; Armbruster, L.; Suchorska, B.; Lutz, J.; Eigenbrod, S.; Cumming, P.; Bartenstein, P.; Tonn, J.-C.; Kreth, F.W.; et al. MRI-suspected low-grade glioma: Is there a need to perform dynamic FET PET? Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1021–1029. [Google Scholar] [CrossRef]
- Jansen, N.L.; Schwartz, C.; Graute, V.; Eigenbrod, S.; Lutz, J.; Egensperger, R.; Pöpperl, G.; Kretzschmar, H.A.; Cumming, P.; Bartenstein, P.; et al. Prediction of oligodendroglial histology and LOH 1p/19q using dynamic [18F]FET-PET imaging in intracranial WHO grade II and III gliomas. Neuro Oncol. 2012, 14, 1473–1480. [Google Scholar] [CrossRef]
- Pyka, T.; Gempt, J.; Hiob, D.; Ringel, F.; Schlegel, J.; Bette, S.; Wester, H.-J.; Meyer, B.; Förster, S. Textural analysis of pre-therapeutic [18F]-FET-PET and its correlation with tumor grade and patient survival in high-grade gliomas. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 133–141. [Google Scholar] [CrossRef]
- Zhou, W.; Huang, Q.; Wen, J.; Li, M.; Zhu, Y.; Liu, Y.; Dai, Y.; Guan, Y.; Zhou, Z.; Hua, T. Integrated CT Radiomics Features Could Enhance the Efficacy of 18F-FET PET for Non-Invasive Isocitrate Dehydrogenase Genotype Prediction in Adult Untreated Gliomas: A Retrospective Cohort Study. Front Oncol. 2021, 11, 772703. [Google Scholar] [CrossRef]
- Lohmann, P.; Lerche, C.; Bauer, E.K.; Steger, J.; Stoffels, G.; Blau, T.; Dunkl, V.; Kocher, M.; Viswanathan, S.; Filss, C.P.; et al. Predicting IDH genotype in gliomas using FET PET radiomics. Sci. Rep. 2018, 8, 13328. [Google Scholar] [CrossRef]
- Verger, A.; Stoffels, G.; Bauer, E.K.; Lohmann, P.; Blau, T.; Fink, G.R.; Neumaier, B.; Shah, N.J.; Langen, K.-J.; Galldiks, N. Static and dynamic 18F–FET PET for the characterization of gliomas defined by IDH and 1p/19q status. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Blanc-Durand, P.; Van Der Gucht, A.; Verger, A.; Langen, K.-J.; Dunet, V.; Bloch, J.; Brouland, J.-P.; Nicod-Lalonde, M.; Schaefer, N.; Prior, J.O. Voxel-based 18F-FET PET segmentation and automatic clustering of tumor voxels: A significant association with IDH1 mutation status and survival in patients with gliomas. PLoS ONE 2018, 13, e0199379. [Google Scholar] [CrossRef]
- Bette, S.; Gempt, J.; Delbridge, C.; Kirschke, J.S.; Schlegel, J.; Foerster, S.; Huber, T.; Pyka, T.; Zimmer, C.; Meyer, B.; et al. Prognostic Value of O-(2-[18F]-Fluoroethyl)-L-Tyrosine-Positron Emission Tomography Imaging for Histopathologic Characteristics and Progression-Free Survival in Patients with Low-Grade Glioma. World Neurosurg. 2016, 89, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Ort, J.; Hamou, H.A.; Kernbach, J.M.; Hakvoort, K.; Blume, C.; Lohmann, P.; Galldiks, N.; Heiland, D.H.; Mottaghy, F.M.; Clusmann, H.; et al. 18F-FET-PET-guided gross total resection improves overall survival in patients with WHO grade III/IV glioma: Moving towards a multimodal imaging-guided resection. J. Neurooncol. 2021, 155, 71–80. [Google Scholar] [CrossRef]
- Floeth, F.W.; Sabel, M.; Ewelt, C.; Stummer, W.; Felsberg, J.; Reifenberger, G.; Steiger, H.J.; Stoffels, G.; Coenen, H.H.; Langen, K.-J. Comparison of 18F-FET PET and 5-ALA fluorescence in cerebral gliomas. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 731–741. [Google Scholar] [CrossRef]
- Ewelt, C.; Floeth, F.W.; Felsberg, J.; Steiger, H.J.; Sabel, M.; Langen, K.-J.; Stoffels, G.; Stummer, W. Finding the anaplastic focus in diffuse gliomas: The value of Gd-DTPA enhanced MRI, FET-PET, and intraoperative, ALA-derived tissue fluorescence. Clin. Neurol. Neurosurg. 2011, 113, 541–547. [Google Scholar] [CrossRef]
- Verburg, N.; Koopman, T.; Yaqub, M.M.; Hoekstra, O.S.; Lammertsma, A.A.; Barkhof, F.; Pouwels, P.J.W.; Reijneveld, J.C.; Heimans, J.J.; Rozemuller, A.J.M.; et al. Improved detection of diffuse glioma infiltration with imaging combinations: A diagnostic accuracy study. Neuro Oncol. 2020, 22, 412–422. [Google Scholar] [CrossRef]
- Buchmann, N.; Kläsner, B.; Gempt, J.; Bauer, J.S.; Pyka, T.; Delbridge, C.; Meyer, B.; Krause, B.J.; Ringel, F. 18F-Fluoroethyl-l-Thyrosine Positron Emission Tomography to Delineate Tumor Residuals After Glioblastoma Resection: A Comparison with Standard Postoperative Magnetic Resonance Imaging. World Neurosurg. 2016, 89, 420–426. [Google Scholar] [CrossRef]
- Kläsner, B.; Buchmann, N.; Gempt, J.; Ringel, F.; Lapa, C.; Krause, B.J. Early [18F]FET-PET in Gliomas after Surgical Resection: Comparison with MRI and Histopathology. PLoS ONE 2015, 10, e0141153. [Google Scholar] [CrossRef]
- Allard, B.; Dissaux, B.; Bourhis, D.; Dissaux, G.; Schick, U.; Salaün, P.-Y.; Abgral, R.; Querellou, S. Hotspot on 18F-FET PET/CT to Predict Aggressive Tumor Areas for Radiotherapy Dose Escalation Guiding in High-Grade Glioma. Cancers 2022, 15, 98. [Google Scholar] [CrossRef] [PubMed]
- Munck af Rosenschold, P.; Costa, J.; Engelholm, S.A.; Lundemann, M.J.; Law, I.; Ohlhues, L.; Engelholm, S. Impact of [18F]-fluoro-ethyl-tyrosine PET imaging on target definition for radiation therapy of high-grade glioma. Neuro Oncol. 2015, 17, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, D.F.; Unterrainer, M.; Schön, R.; Corradini, S.; Maihöfer, C.; Bartenstein, P.; Belka, C.; Albert, N.L.; Niyazi, M. Margin reduction in radiotherapy for glioblastoma through 18F-fluoroethyltyrosine PET?—A recurrence pattern analysis. Radiother. Oncol. 2020, 145, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Harat, M.; Małkowski, B.; Makarewicz, R. Pre-irradiation tumour volumes defined by MRI and dual time-point FET-PET for the prediction of glioblastoma multiforme recurrence: A prospective study. Radiother. Oncol. 2016, 120, 241–247. [Google Scholar] [CrossRef]
- Dissaux, G.; Dissaux, B.; Kabbaj, O.E.; Gujral, D.M.; Pradier, O.; Salaün, P.-Y.; Seizeur, R.; Bourhis, D.; Ben Salem, D.; Querellou, S.; et al. Radiotherapy target volume definition in newly diagnosed high grade glioma using 18F-FET PET imaging and multiparametric perfusion MRI: A prospective study (IMAGG). Radiother. Oncol. 2020, 150, 164–171. [Google Scholar] [CrossRef]
- Hayes, A.R.; Jayamanne, D.; Hsiao, E.; Schembri, G.P.; Bailey, D.L.; Roach, P.J.; Khasraw, M.; Newey, A.; Wheeler, H.R.; Back, M. Utilizing 18F-fluoroethyltyrosine (FET) positron emission tomography (PET) to define suspected nonenhancing tumor for radiation therapy planning of glioblastoma. Pract. Radiat. Oncol. 2018, 8, 230–238. [Google Scholar] [CrossRef]
- Galldiks, N.; Stoffels, G.; Ruge, M.I.; Rapp, M.; Sabel, M.; Reifenberger, G.; Erdem, Z.; Shah, N.J.; Fink, G.R.; Coenen, H.H.; et al. Role of O-(2-18F-Fluoroethyl)-l-Tyrosine PET as a Diagnostic Tool for Detection of Malignant Progression in Patients with Low-Grade Glioma. J. Nucl. Med. 2013, 54, 2046–2054. [Google Scholar] [CrossRef]
- Unterrainer, M.; Schweisthal, F.; Suchorska, B.; Wenter, V.; Schmid-Tannwald, C.; Fendler, W.P.; Schüller, U.; Bartenstein, P.; Tonn, J.-C.; Albert, N.L. Serial 18 F-FET PET Imaging of Primarily 18 F-FET–Negative Glioma: Does It Make Sense? J. Nucl. Med. 2016, 57, 1177–1182. [Google Scholar] [CrossRef]
- Bashir, A.; Brennum, J.; Broholm, H.; Law, I. The diagnostic accuracy of detecting malignant transformation of low-grade glioma using O-(2-[18F]fluoroethyl)-l-tyrosine positron emission tomography: A retrospective study. J. Neurosurg. 2018, 130, 451–464. [Google Scholar] [CrossRef]
- Jeong, S.Y.; Lee, T.H.; Rhee, C.H.; Cho, A.R.; Il Kim, B.; Cheon, G.J.; Choi, C.W.; Lim, S.M. 3′-Deoxy-3′-[18F]fluorothymidine and O-(2-[18F]fluoroethyl)-L-tyrosine PET in Patients with Suspicious Recurrence of Glioma after Multimodal Treatment: Initial Results of a Retrospective Comparative Study. Nucl. Med. Mol. Imaging 2010, 44, 45–54. [Google Scholar] [CrossRef][Green Version]
- Jansen, N.L.; Suchorska, B.; Schwarz, S.B.; Eigenbrod, S.; Lutz, J.; Graute, V.; Bartenstein, P.; Belka, C.; Kreth, F.W.; Fougère, C.L. [18F]Fluoroethyltyrosine–Positron Emission Tomography-Based Therapy Monitoring after Stereotactic Iodine-125 Brachytherapy in Patients with Recurrent High-Grade Glioma. Mol. Imaging 2013, 12, 7290.2012.00027. [Google Scholar] [CrossRef]
- Puranik, A.D.; Rangarajan, V.; Dev, I.D.; Jain, Y.; Purandare, N.C.; Sahu, A.; Choudhary, A.; Gupta, T.; Chatterjee, A.; Moiyadi, A.; et al. Brain FET PET tumor-to-white mater ratio to differentiate recurrence from post-treatment changes in high-grade gliomas. J. Neuroimaging 2021, 31, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Kertels, O.; Mihovilovic, M.I.; Linsenmann, T.; Kessler, A.F.; Tran-Gia, J.; Kircher, M.; Brumberg, J.; Monoranu, C.M.; Samnick, S.; Ernestus, R.-I.; et al. Clinical Utility of Different Approaches for Detection of Late Pseudoprogression in Glioblastoma with O-(2-[18F]Fluoroethyl)-l-Tyrosine PET. Clin. Nucl. Med. 2019, 44, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Verger, A.; Filss, C.P.; Lohmann, P.; Stoffels, G.; Sabel, M.; Wittsack, H.-J.; Kops, E.R.; Galldiks, N.; Fink, G.R.; Shah, N.J.; et al. Comparison of O-(2-18 F-Fluoroethyl)-L-Tyrosine Positron Emission Tomography and Perfusion-Weighted Magnetic Resonance Imaging in the Diagnosis of Patients with Progressive and Recurrent Glioma: A Hybrid Positron Emission Tomography/Magnetic Resonance Study. World Neurosurg. 2018, 113, e727–e737. [Google Scholar] [CrossRef]
- Pyka, T.; Hiob, D.; Preibisch, C.; Gempt, J.; Wiestler, B.; Schlegel, J.; Straube, C.; Zimmer, C. Diagnosis of glioma recurrence using multiparametric dynamic 18F-fluoroethyl-tyrosine PET-MRI. Eur. J. Radiol. 2018, 103, 32–37. [Google Scholar] [CrossRef]
- Werner, J.-M.; Weller, J.; Ceccon, G.; Schaub, C.; Tscherpel, C.; Lohmann, P.; Bauer, E.K.; Schäfer, N.; Stoffels, G.; Baues, C.; et al. Diagnosis of Pseudoprogression Following Lomustine-Temozolomide Chemoradiation in Newly Diagnosed Glioblastoma Patients Using FET-PET. Clin. Cancer Res. 2021, 27, 3704–3713. [Google Scholar] [CrossRef]
- Galldiks, N.; Dunkl, V.; Stoffels, G.; Hutterer, M.; Rapp, M.; Sabel, M.; Reifenberger, G.; Kebir, S.; Dorn, F.; Blau, T.; et al. Diagnosis of pseudoprogression in patients with glioblastoma using O-(2-[18F]fluoroethyl)-l-tyrosine PET. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 685–695. [Google Scholar] [CrossRef]
- Werner, J.-M.; Stoffels, G.; Lichtenstein, T.; Borggrefe, J.; Lohmann, P.; Ceccon, G.; Shah, N.J.; Fink, G.R.; Langen, K.-J.; Kabbasch, C.; et al. Differentiation of treatment-related changes from tumour progression: A direct comparison between dynamic FET PET and ADC values obtained from DWI MRI. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1889–1901. [Google Scholar] [CrossRef]
- Lohmann, P.; Elahmadawy, M.A.; Gutsche, R.; Werner, J.-M.; Bauer, E.K.; Ceccon, G.; Kocher, M.; Lerche, C.W.; Rapp, M.; Fink, G.R.; et al. FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation. Cancers 2020, 12, 3835. [Google Scholar] [CrossRef]
- Kebir, S.; Fimmers, R.; Galldiks, N.; Schäfer, N.; Mack, F.; Schaub, C.; Stuplich, M.; Niessen, M.; Tzaridis, T.; Simon, M.; et al. Late Pseudoprogression in Glioblastoma: Diagnostic Value of Dynamic O-(2-[18F]fluoroethyl)-L-Tyrosine PET. Clin. Cancer Res. 2016, 22, 2190–2196. [Google Scholar] [CrossRef]
- Rachinger, W.; Goetz, C.; Pöpperl, G.; Gildehaus, F.J.; Kreth, F.W.; Holtmannspötter, M.; Herms, J.; Koch, W.; Tatsch, K.; Tonn, J.-C. Positron Emission Tomography with O-(2-[18F]fluoroethyl)-l-tyrosine versus Magnetic Resonance Imaging in the Diagnosis of Recurrent Gliomas. Neurosurgery 2005, 57, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Lohmeier, J.; Bohner, G.; Siebert, E.; Brenner, W.; Hamm, B.; Makowski, M.R. Quantitative biparametric analysis of hybrid 18F-FET PET/MR-neuroimaging for differentiation between treatment response and recurrent glioma. Sci. Rep. 2019, 9, 14603. [Google Scholar] [CrossRef]
- Bashir, A.; Mathilde Jacobsen, S.; Mølby Henriksen, O.; Broholm, H.; Urup, T.; Grunnet, K.; Andrée Larsen, V.; Møller, S.; Skjøth-Rasmussen, J.; Skovgaard Poulsen, H.; et al. Recurrent glioblastoma versus late posttreatment changes: Diagnostic accuracy of O-(2-[18F]fluoroethyl)-L-tyrosine positron emission tomography (18F-FET PET). Neuro Oncol. 2019, 21, 1595–1606. [Google Scholar] [CrossRef] [PubMed]
- Steidl, E.; Langen, K.-J.; Hmeidan, S.A.; Polomac, N.; Filss, C.P.; Galldiks, N.; Lohmann, P.; Keil, F.; Filipski, K.; Mottaghy, F.M.; et al. Sequential implementation of DSC-MR perfusion and dynamic [18F]FET PET allows efficient differentiation of glioma progression from treatment-related changes. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1956–1965. [Google Scholar] [CrossRef] [PubMed]
- Pöpperl, G.; Götz, C.; Rachinger, W.; Schnell, O.; Gildehaus, F.J.; Tonn, J.C.; Tatsch, K. Serial O-(2-[18F]fluoroethyl)-L-tyrosine PET for monitoring the effects of intracavitary radioimmunotherapy in patients with malignant glioma. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 792–800. [Google Scholar] [CrossRef]
- Müller, M.; Winz, O.; Gutsche, R.; Leijenaar, R.T.H.; Kocher, M.; Lerche, C.; Filss, C.P.; Stoffels, G.; Steidl, E.; Hattingen, E.; et al. Static FET PET radiomics for the differentiation of treatment-related changes from glioma progression. J. Neurooncol. 2022, 159, 519–529. [Google Scholar] [CrossRef]
- Mehrkens, J.H.; Pöpperl, G.; Rachinger, W.; Herms, J.; Seelos, K.; Tatsch, K.; Tonn, J.C.; Kreth, F.W. The positive predictive value of O-(2-[18F]fluoroethyl)-l-tyrosine (FET) PET in the diagnosis of a glioma recurrence after multimodal treatment. J. Neurooncol. 2008, 88, 27–35. [Google Scholar] [CrossRef]
- Galldiks, N.; Stoffels, G.; Filss, C.; Rapp, M.; Blau, T.; Tscherpel, C.; Ceccon, G.; Dunkl, V.; Weinzierl, M.; Stoffel, M.; et al. The use of dynamic O-(2-18F-fluoroethyl)-l-tyrosine PET in the diagnosis of patients with progressive and recurrent glioma. Neuro Oncol. 2015, 17, 1293–1300. [Google Scholar] [CrossRef]
- Pöpperl, G.; Götz, C.; Rachinger, W.; Gildehaus, F.-J.; Tonn, J.-C.; Tatsch, K. Value of O-(2-[18F]fluoroethyl)-l-tyrosine PET for the diagnosis of recurrent glioma. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, 1464–1470. [Google Scholar] [CrossRef]
- Müther, M.; Koch, R.; Weckesser, M.; Sporns, P.; Schwindt, W.; Stummer, W. 5-Aminolevulinic Acid Fluorescence-Guided Resection of 18F-FET-PET Positive Tumor Beyond Gadolinium Enhancing Tumor Improves Survival in Glioblastoma. Neurosurgery 2019, 85, E1020–E1029. [Google Scholar] [CrossRef]
- Suchorska, B.; Unterrainer, M.; Biczok, A.; Sosnova, M.; Forbrig, R.; Bartenstein, P.; Tonn, J.-C.; Albert, N.L.; Kreth, F.-W. 18F-FET-PET as a biomarker for therapy response in non-contrast enhancing glioma following chemotherapy. J. Neurooncol. 2018, 139, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Langen, K.-J.; Holy, R.; Pinkawa, M.; Stoffels, G.; Nolte, K.W.; Kaiser, H.J.; Filss, C.P.; Fink, G.R.; Coenen, H.H.; et al. Assessment of Treatment Response in Patients with Glioblastoma Using O-(2-18F-Fluoroethyl)-l-Tyrosine PET in Comparison to MRI. J. Nucl. Med. 2012, 53, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Suchorska, B.; Jansen, N.L.; Linn, J.; Kretzschmar, H.; Janssen, H.; Eigenbrod, S.; Simon, M.; Pöpperl, G.; Kreth, F.W.; La Fougere, C.; et al. Biological tumor volume in 18FET-PET before radiochemotherapy correlates with survival in GBM. Neurology 2015, 84, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.L.; Suchorska, B.; Wenter, V.; Eigenbrod, S.; Schmid-Tannwald, C.; Zwergal, A.; Niyazi, M.; Drexler, M.; Bartenstein, P.; Schnell, O.; et al. Dynamic 18F-FET PET in Newly Diagnosed Astrocytic Low-Grade Glioma Identifies High-Risk Patients. J. Nucl. Med. 2014, 55, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Thon, N.; Kunz, M.; Lemke, L.; Jansen, N.L.; Eigenbrod, S.; Kreth, S.; Lutz, J.; Egensperger, R.; Giese, A.; Herms, J.; et al. Dynamic 18F-FET PET in suspected WHO grade II gliomas defines distinct biological subgroups with different clinical courses. Int. J. Cancer 2015, 136, 2132–2145. [Google Scholar] [CrossRef]
- Kunz, M.; Albert, N.L.; Unterrainer, M.; la Fougere, C.; Egensperger, R.; Schüller, U.; Lutz, J.; Kreth, S.; Tonn, J.-C.; Kreth, F.-W.; et al. Dynamic 18F-FET PET is a powerful imaging biomarker in gadolinium-negative gliomas. Neuro Oncol. 2019, 21, 274–284. [Google Scholar] [CrossRef]
- Ceccon, G.; Lohmann, P.; Werner, J.-M.; Tscherpel, C.; Dunkl, V.; Stoffels, G.; Rosen, J.; Rapp, M.; Sabel, M.; Herrlinger, U.; et al. Early Treatment Response Assessment Using 18F-FET PET Compared with Contrast-Enhanced MRI in Glioma Patients After Adjuvant Temozolomide Chemotherapy. J. Nucl. Med. 2021, 62, 918–925. [Google Scholar] [CrossRef]
- Galldiks, N.; Dunkl, V.; Ceccon, G.; Tscherpel, C.; Stoffels, G.; Law, I.; Henriksen, O.M.; Muhic, A.; Poulsen, H.S.; Steger, J.; et al. Early treatment response evaluation using FET PET compared to MRI in glioblastoma patients at first progression treated with bevacizumab plus lomustine. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2377–2386. [Google Scholar] [CrossRef]
- Carles, M.; Popp, I.; Starke, M.M.; Mix, M.; Urbach, H.; Schimek-Jasch, T.; Eckert, F.; Niyazi, M.; Baltas, D.; Grosu, A.L. FET-PET radiomics in recurrent glioblastoma: Prognostic value for outcome after re-irradiation? Radiat. Oncol. 2021, 16, 46. [Google Scholar] [CrossRef]
- Suchorska, B.; Giese, A.; Biczok, A.; Unterrainer, M.; Weller, M.; Drexler, M.; Bartenstein, P.; Schüller, U.; Tonn, J.-C.; Albert, N.L. Identification of time-to-peak on dynamic 18F-FET-PET as a prognostic marker specifically in IDH1/2 mutant diffuse astrocytoma. Neuro Oncol. 2018, 20, 279–288. [Google Scholar] [CrossRef]
- Wirsching, H.-G.; Roelcke, U.; Weller, J.; Hundsberger, T.; Hottinger, A.F.; von Moos, R.; Caparrotti, F.; Conen, K.; Remonda, L.; Roth, P.; et al. MRI and 18FET-PET predict survival benefit from bevacizumab plus radiotherapy in patients with IDH wild-type glioblastoma: Results from the randomized ARTE trial. Clin. Cancer Res. 2021, 27, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, R.; Polat, B.; Samnick, S.; Reiners, C.; Flentje, M.; Verburg, F.A. O-(2-[18F]fluoroethyl)-l-tyrosine uptake is an independent prognostic determinant in patients with glioma referred for radiation therapy. Ann. Nucl. Med. 2014, 28, 154–162. [Google Scholar] [CrossRef]
- Pyka, T.; Gempt, J.; Ringel, F.; Hüttinger, S.; van Marwick, S.; Nekolla, S.; Wester, H.-J.; Schwaiger, M.; Förster, S. Prediction of Glioma Recurrence Using Dynamic 18F-Fluoroethyltyrosine PET. AJNR Am. J. Neuroradiol. 2014, 35, 1924–1929. [Google Scholar] [CrossRef] [PubMed]
- Wollring, M.M.; Werner, J.-M.; Bauer, E.K.; Tscherpel, C.; Ceccon, G.S.; Lohmann, P.; Stoffels, G.; Kabbasch, C.; Goldbrunner, R.; Fink, G.R.; et al. Prediction of response to lomustine-based chemotherapy in glioma patients at recurrence using MRI and FET PET. Neuro Oncol. 2022, 25, 984–994. [Google Scholar] [CrossRef]
- Bauer, E.K.; Stoffels, G.; Blau, T.; Reifenberger, G.; Felsberg, J.; Werner, J.M.; Lohmann, P.; Rosen, J.; Ceccon, G.; Tscherpel, C.; et al. Prediction of survival in patients with IDH-wildtype astrocytic gliomas using dynamic O-(2-[18F]-fluoroethyl)-l-tyrosine PET. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Piroth, M.D.; Holy, R.; Pinkawa, M.; Stoffels, G.; Kaiser, H.J.; Galldiks, N.; Herzog, H.; Coenen, H.H.; Eble, M.J.; Langen, K.J. Prognostic impact of postoperative, pre-irradiation 18F-fluoroethyl-l-tyrosine uptake in glioblastoma patients treated with radiochemotherapy. Radiother. Oncol. 2011, 99, 218–224. [Google Scholar] [CrossRef]
- Jansen, N.L.; Suchorska, B.; Wenter, V.; Schmid-Tannwald, C.; Todica, A.; Eigenbrod, S.; Niyazi, M.; Tonn, J.-C.; Bartenstein, P.; Kreth, F.-W.; et al. Prognostic Significance of Dynamic 18F-FET PET in Newly Diagnosed Astrocytic High-Grade Glioma. J. Nucl. Med. 2015, 56, 9–15. [Google Scholar] [CrossRef]
- Moller, S.; Law, I.; Munck Af Rosenschold, P.; Costa, J.; Poulsen, H.S.; Engelholm, S.A.; Engelholm, S. Prognostic value of 18F-FET PET imaging in re-irradiation of high-grade glioma: Results of a phase I clinical trial. Radiother. Oncol. 2016, 121, 132–137. [Google Scholar] [CrossRef]
- Dissaux, G.; Basse, V.; Schick, U.; EL Kabbaj, O.; Auberger, B.; Magro, E.; Kassoul, A.; Abgral, R.; Salaun, P.-Y.; Bourhis, D.; et al. Prognostic value of 18F-FET PET/CT in newly diagnosed WHO 2016 high-grade glioma. Medicine 2020, 99, e19017. [Google Scholar] [CrossRef]
- Piroth, M.D.; Pinkawa, M.; Holy, R.; Klotz, J.; Nussen, S.; Stoffels, G.; Coenen, H.H.; Kaiser, H.J.; Langen, K.J.; Eble, M.J. Prognostic Value of Early [18F]Fluoroethyltyrosine Positron Emission Tomography After Radiochemotherapy in Glioblastoma Multiforme. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 176–184. [Google Scholar] [CrossRef]
- Schneider, F.; Wolpert, F.; Stolzmann, P.; Albatly, A.A.; Kenkel, D.; Weller, J.; Weller, M.; Kollias, S.S.; Rushing, E.J.; Veit-Haibach, P.; et al. Prognostic value of O-(2-[18F]-fluoroethyl)-L-tyrosine PET in relapsing oligodendroglioma. Acta Oncol. 2020, 59, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Kertels, O.; Kessler, A.F.; Mihovilovic, M.I.; Stolzenburg, A.; Linsenmann, T.; Samnick, S.; Brändlein, S.; Monoranu, C.M.; Ernestus, R.-I.; Buck, A.K.; et al. Prognostic Value of O-(2-[18F]Fluoroethyl)-L-Tyrosine PET/CT in Newly Diagnosed WHO 2016 Grade II and III Glioma. Mol. Imaging Biol. 2019, 21, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Floeth, F.W.; Pauleit, D.; Sabel, M.; Stoffels, G.; Reifenberger, G.; Riemenschneider, M.J.; Jansen, P.; Coenen, H.H.; Steiger, H.-J.; Langen, K.-J. Prognostic Value of O-(2-18F-Fluoroethyl)-L-Tyrosine PET and MRI in Low-Grade Glioma. J. Nucl. Med. 2007, 48, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Niyazi, M.; Jansen, N.; Ganswindt, U.; Schwarz, S.B.; Geisler, J.; Schnell, O.; Büsing, K.; Eigenbrod, S.; La Fougère, C.; Belka, C. Re-irradiation in recurrent malignant glioma: Prognostic value of [18F]FET–PET. J. Neurooncol. 2012, 110, 389–395. [Google Scholar] [CrossRef]
- Albert, N.L.; Galldiks, N.; Ellingson, B.M.; van den Bent, M.J.; Chang, S.M.; Cicone, F.; de Groot, J.; Koh, E.-S.; Law, I.; Rhun, E.L.; et al. PET-based response assessment criteria for diffuse gliomas (PET RANO 1.0): A report of the RANO group. Lancet Oncol. 2024, 25, e29–e41. [Google Scholar] [CrossRef]
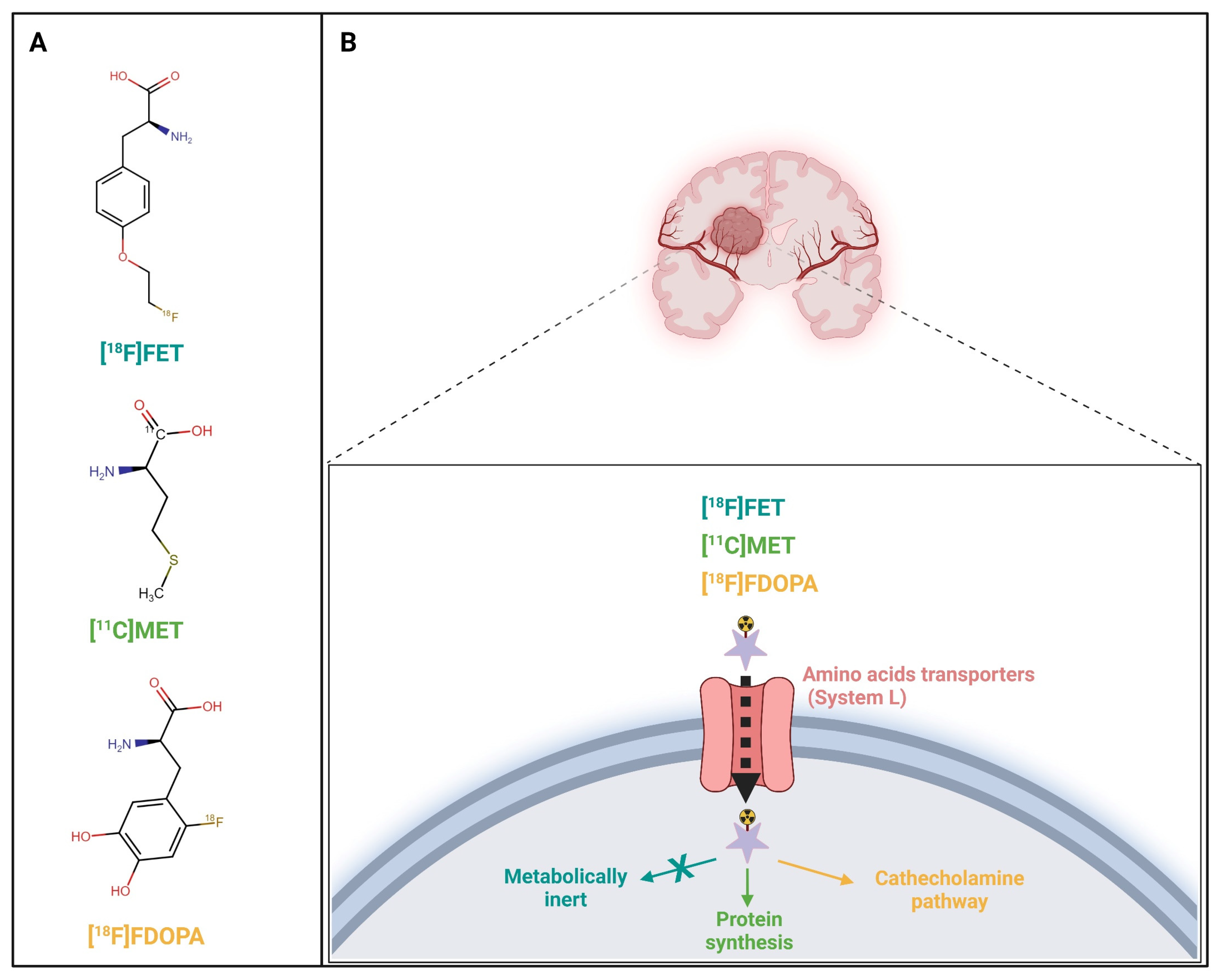

| Aspect | [11C]MET | [18F]F-DOPA | [18F]FET |
|---|---|---|---|
| Radiotracer Type | Amino acid analog | Amino acid precursor | Amino acid analog |
| Mechanism of Uptake | Uptake via L-type amino acid transporter (LAT) into tumor cells with high protein synthesis. | Uptake via amino acid transport (LAT) is overexpressed in tumor cells. Converted into dopamine in dopaminergic neurons. | Uptake via LAT, reflecting increased amino acid transport correlated to tumor proliferation. |
| Half-Life | 20 min | 110 min | 110 min |
| Production | Requires on-site cyclotron due to short half-life. | Can be produced off-site, longer shelf life. | Can be produced off-site, longer shelf life. |
| Sensitivity in Gliomas | High sensitivity, more effective in detecting high-grade gliomas. | High sensitivity in detecting glioma. | High sensitivity, more effective in detecting high-grade gliomas. |
| Specificity in Gliomas | Moderate specificity, possible uptake in inflammatory lesions. | High specificity, with potential uptake in inflammatory tissues. | High specificity, with less non-specific uptake in inflammatory tissues compared to [11C]MET. |
| Advantages | Rapid uptake, good lesion contrast. | Longer half-life allows broader clinical application. | Longer half-life allows broader clinical application. Dynamic acquisition allows additional information on tracer kinetics, particularly useful for tumor grading. |
| Disadvantages | Short half-life limits use to facilities with a cyclotron, potential uptake in inflammation. | May have false positives in inflamed tissues. High physiologic uptake in the basal ganglia. | Potential uptake in inflammatory lesions but less than [11C]MET. |
| Clinical Application | Primarily used in facilities with a cyclotron, used to detect tumor recurrence and in monitoring the response to therapy. | Mostly used for differentiating tumor recurrence from necrosis, especially in high-grade gliomas. | Widely used for differentiating high-grade glioma early and late progression from radiation effects. |
| Indication | Author, Year | Reference | Design | Number of Patients | Grade | Mean Age | Sex | Imaging Modality | Parameters | Optimal Cut-Off | Sensitivity | Specificity | AUC | Accuracy |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diagnosis | ||||||||||||||
| Pauleit et al., 2009 | [24] | Prospective | 52 | Not glioma:9 | 46 | 36 M 16 F | PET | Lmean/B # | - | |||||
| Grade 2:22 | Lmax/B # | - | ||||||||||||
| Grade 3:12 | Visual grading system # | - | ||||||||||||
| Grade 4:9 | ||||||||||||||
| Mauler et al., 2023 | [25] | Prospective | 30 | Not glioma:6 | 48 | 16 M 14 F | PET | 18F-FETn uptake | 1.4 x background | 76% | 80% | 0.89 | 78% | |
| Grade 2:7 | MRI | Cho/NAAn | 2.16 | 59% | 83% | 0.81 | 71% | |||||||
| Grade 3:7 | ||||||||||||||
| Grade 4:10 | ||||||||||||||
| Floeth et al., 2005 | [26] | Prospective | 50 | Not glioma:16 | 44 | 21 M 29 F | PET | FET lesion/brain ratio | 1.6 | 88% | 88% | - | ||
| Grade 1:2 | MRI | Gd enhancement | - | 44% | 69% | 68% | ||||||||
| Grade 2:13 | NAA/Cho ratio | 0.7 | 100% | 81% | - | |||||||||
| Grade 3:14 | ||||||||||||||
| Grade 4:5 | ||||||||||||||
| Pauleit et al., 2005 | [27] | Prospective | 28 | Not glioma:5 | 42 | 9 M 19 F | PET | FET ratio | 1.6 | 92% | 81% | - | ||
| Grade 1:2 | MRI | T1 ratio | 1.0 | 85% | 12% | - | ||||||||
| Grade 2:7 | Gd-T1 ratio | 1.0 | 38% | 96% | - | |||||||||
| Grade 3:12 | FLAIR ratio | 1.0 | 96% | 4% | - | |||||||||
| Grade 4:2 | T1/Gd-T1/FLAIR ratio | - | 96% | 53% | 68% | |||||||||
| PET/CT + MRI | FET/T1/Gd-T1/FLAIR ratio | - | 93% | 94% | 94% | |||||||||
| Grading (LGG vs. HGG) | ||||||||||||||
| Jeong and Lim, 2012 | [28] | Prospective | 20 | Grade 2:3 | 52 | 13 M 7 F | PET | SUVmax | - | |||||
| Grade 3:2 | TNR | - | ||||||||||||
| Grade 4:15 | ||||||||||||||
| Verger et al., 2017 | [29] | Retrospective | 72 | Grade 1:1 | 49 | 42 M 30 F | PET | TBRmax | 2.62 | 82% | 68% | 0.83 | 78% | |
| Grade 2:21 | TBRmean | 1.69 | 82% | 68% | 0.80 | 78% | ||||||||
| Grade 3:25 | TTP | 30 min | 54% | 91% | 0.78 | 65% | ||||||||
| Grade 4:25 | Slope | −0.03 SUV/h | 64% | 91% | 0.78 | 72% | ||||||||
| PWI rCBF | TBRmax | 1.51 | 64% | 64% | 0.74 | 64% | ||||||||
| TBRmean | 0.69 | 62% | 59% | 0.66 | 61% | |||||||||
| PWI rCBV | TBRmax | 1.80 | 88% | 72% | 0.81 | 83% | ||||||||
| TBRmean | 1.14 | 72% | 77% | 0.80 | 74% | |||||||||
| PWI MTT | TBRmax § | 1.16 | 64% | 50% | 0.58 | 60% | ||||||||
| TBRmean § | 0.98 | 54% | 36% | 0.43 | 49% | |||||||||
| Lopez et al., 2015 | [30] | Prospective | 23 | No-grade:2 | 56 | 18 M 5 F | PET | UR | 3.0 | |||||
| Grade 1:1 | ||||||||||||||
| Grade 2:7 | ||||||||||||||
| Grade 3:2 | ||||||||||||||
| Grade 4:11 | ||||||||||||||
| Lohmann et al., 2015 | [31] | Prospective | 36 | Grade 2:12 | 49 | 19 M 17 F | PET | TBRmean § | 2 | 83% | 58% | 0.65 | 75% | |
| Grade 3:8 | ∆TBRmean 20–40 min/70–90 min | −8% | 83% | 75% | 0.85 | 81% | ||||||||
| Grade 4:16 | TTP | 35 min | 58% | 92% | 0.76 | 69% | ||||||||
| Kinetic pattern | II/III | 88% | 75% | - | 83% | |||||||||
| Calcagni et al., 2011 | [32] | Prospective | 32 | Grade 1:3 | 41 | 21 M 11 F | PET | TAC # | I/II vs. III | 73% | 100% | 87% | ||
| Grade 2:14 | Early SUV | 2.32 | 73% | 71% | 72% | |||||||||
| Grade 3:11 | Middle SUV § | - | - | - | - | |||||||||
| Grade 4:4 | Late SUV § | - | - | - | - | |||||||||
| e-m ratio | 0.93 | 93% | 94% | 94% | ||||||||||
| e-l ratio | 0.95 | 87% | 88% | 87% | ||||||||||
| Tpeak | 25 min | 87% | 100% | 94% | ||||||||||
| SoD | 0.5 | 93% | 82% | 87% | ||||||||||
| Logistic regression using Early SUV + SoD § | 50% | 93% | 100% | 97% | ||||||||||
| Albert et al., 2016 | [33] | Retrospective | 314 | Grade 1:3 | 49 | 181 M 133 F | PET | TBRmax (20–40 min) | 2.7 | 67% | 78% | 70% | ||
| Grade 2:128 | TBRmax (0–10 min) | 2.8 | 76% | 79% | 76% | |||||||||
| Grade 3:95 | TBRmax (5–15 min) | 2.7 | 78% | 76% | 77% | |||||||||
| Grade 4:88 | TBRmax (5–20 min) | 2.6 | 80% | 74% | 76% | |||||||||
| TBRmax (10–30 min) | 2.5 | 75% | 75% | 74% | ||||||||||
| Kinetic pattern # | Decreasing | 90% | 66% | 80% | ||||||||||
| Pöpperl et al., 2007 | [19] | Prospective | 54 | Grade 2:15 | 49 | 30 M 24 F | PET | SUVmax/BG | 2.58 | 71% | 85% | 0.798 | ||
| Grade 3:21 | SUV90 10–60 min | 0.20 | 94% | 100% | 0.969 | |||||||||
| Grade 4:18 | SUV90 15–60 min | −0.41 | 94% | 100% | 0.965 | |||||||||
| Grade 2/3 vs. grade 4 | Hua et al., 2021 | [34] | Retrospective | 58 | Grade 2:33 | 42 | 37 M 21 F | PET | TBRmax | 2.67 | 92% | 61% | 0.824 | 67% |
| Grade 3:13 | TBRpeak | 2.35 | 92% | 61% | 0.832 | 67% | ||||||||
| Grade 4:12 | TBRmean | 2.31 | 58% | 93% | 0.791 | 86% | ||||||||
| COV | 27.21 | 58% | 91% | 0.808 | 84% | |||||||||
| HI | 1.77 | 67% | 87% | 0.826 | 83% | |||||||||
| MTV | 20.13 | 75% | 80% | 0.801 | 79% | |||||||||
| TLU | 50.93 | 75% | 83% | 0.841 | 81% | |||||||||
| SUVsd | 0.45 | 67% | 87% | 0.816 | 83% | |||||||||
| TBRmax + SUVsd + TBRmean | - | 75% | 85% | 0.850 | 83% | |||||||||
| HI + SUVsd + MTV | - | 75% | 83% | 0.848 | 81% | |||||||||
| HI + SUVsd + TLU | - | 75% | 84% | 0.848 | 81% | |||||||||
| Kunz et al., 2011 | [35] | Prospective | 55 | Grade 2:31 | 44 | 33 M 22 F | PET | TAC | Increasing vs. decreasing | 96% | 94% | |||
| Grade 3:22 | MRI | Tumor volume § | - | - | - | |||||||||
| Grade 4:2 | ||||||||||||||
| Grade 2/3 vs. grade 4 | Röhrich et al., 2018 | [36] | Retrospective | 44 | Grade 2:10 | 53 | - | PET | TAC # | LGG-like vs. mixed vs. HGG-like | - | - | - | |
| Grade 3:13 | SUVmax/BG | - | - | - | - | |||||||||
| Grade 4:21 | TTP § | - | - | - | - | |||||||||
| Relative K1 | - | 85% | 60% | 0.766 | ||||||||||
| Relative K2 § | - | - | - | - | ||||||||||
| Relative K3 § | - | - | - | - | ||||||||||
| Relative FD | - | 67% | 78% | 0.716 | ||||||||||
| SUVmax/BG + TTP | - | - | - | 0.745 | ||||||||||
| SUVmax/BG + TTP + relative K1 + relative FD | - | - | - | 0.799 | ||||||||||
| Jansen et al., 2012 | [37] | Retrospective | 127 | No tumor:7 | 46 | 72 M 55 F | PET | TAC # | Increasing vs. decreasing | 95% | 72% | |||
| Grade 1:4 | FET uptake # | Reduced vs. normal vs. increased | - | - | ||||||||||
| Grade 2:69 | FET uptake pattern § | Inhomogeneous vs. diffuse vs. focal | - | - | ||||||||||
| Grade 3:42 | SUVmax/BG § | - | - | - | ||||||||||
| Grade 4:5 | SUVmean/BG § | - | - | - | ||||||||||
| BTV § | - | - | - | |||||||||||
| grade 2 vs. 3 | Jansen et al., 2012 | [38] | Prospective | 144 | Grade 2:79 | 45 | 84 M 60 F | PET | TAC # | Decreasing | 88% | 63% | ||
| Grade 3:65 | SUVmax/BG § | - | - | - | ||||||||||
| BTV § | - | - | - | |||||||||||
| SUVtotal/BG § | - | - | - | |||||||||||
| SUVmean/BG § | - | - | - | |||||||||||
| grade 3 vs. 4 | Pyka et al., 2016 | [39] | Retrospective | 113 | Grade 3:26 | 59 | 43 M 70 F | PET | TBRmax § | 2.74 | 0.614 | |||
| Grade 4:87 | TBRmean | 1.68 | 0.644 | |||||||||||
| MTV | 19.7 | 0.710 | ||||||||||||
| TLU | 46.2 | 0.704 | ||||||||||||
| Textural parameters: | ||||||||||||||
| Coarseness | 0.607 | 0.757 | ||||||||||||
| Contrast | 0.203 | 0.775 | ||||||||||||
| Busyness | 1.12 | 0.737 | ||||||||||||
| Complexity | 0.069 | 0.633 | ||||||||||||
| Combined | 2.05 | 0.830 | ||||||||||||
| IDH status determination | ||||||||||||||
| Hua et al., 2021 | [34] | Retrospective | 58 | Grade 2:33 | 42 | 37 M 21 F | PET | TBRmax | 2.21 | 48% | 87% | 0.658 | 72% | |
| Grade 3:13 | TBRpeak § | 2.15 | 57% | 73% | 0.638 | 67% | ||||||||
| Grade 4:12 | TBRmean § | 1.84 | 62% | 68% | 0.633 | 66% | ||||||||
| COV | 8.85 | 52% | 76% | 0.650 | 67% | |||||||||
| HI | 1.26 | 48% | 87% | 0.676 | 72% | |||||||||
| MTV | 19.48 | 90% | 46% | 0.660 | 62% | |||||||||
| TLU | 28.95 | 81% | 57% | 0.698 | 66% | |||||||||
| SUVsd | 0.11 | 47% | 57% | 0.710 | 66% | |||||||||
| TBRmax + SUVsd + TBRmean | - | 76% | 84% | 0.821 | 81% | |||||||||
| HI + SUVsd + MTV | - | 86% | 81% | 0.804 | 83% | |||||||||
| HI + SUVsd + TLU | - | 76% | 84% | 0.799 | 81% | |||||||||
| Zhou et al., 2021 | [40] | Retrospective | 58 | Grade 2:31 | - | 26 M 22 F | PET | SUVSD | 0.23 | - | - | - | - | |
| Grade 3:14 | TLU § | - | - | - | - | - | ||||||||
| Grade 4:13 | MTV § | - | - | - | - | - | ||||||||
| TBRmax § | - | - | - | - | - | |||||||||
| TBRmean § | - | - | - | - | - | |||||||||
| TBRpeak § | - | - | - | - | - | |||||||||
| Midline involvement | Yes vs. no | - | - | - | - | |||||||||
| Simple predictive model | - | 85% | 71% | 0.786 | 76% | |||||||||
| Radiomics models: | ||||||||||||||
| PET-Rad model | - | 80% | 74% | 0.812 | 76% | |||||||||
| CT | CT-Rad model | - | 85% | 76% | 0.883 | 79% | ||||||||
| PET/CT | PET/CT-Rad model | - | 85% | 87% | 0.912 | 86% | ||||||||
| Lohmann et al., 2018 | [41] | Retrospective | 84 | Grade 2:7 | 54 | 50 M 34 F | PET | TBRmean | 1.68 | 12% | 100% | 0.66 | 73% | |
| Grade 3:26 | TBRmax § | 2.07 | 8% | 100% | 0.59 | 71% | ||||||||
| Grade 4:51 | TTP | 45 min | 27% | 93% | 0.75 | 73% | ||||||||
| Slope | 0.30 SUV/h | 58% | 90% | 0.79 | 80% | |||||||||
| Slope + Radiomic feature SZHGE | - | 54% | 93% | - | 81% | |||||||||
| Radiomic features: | ||||||||||||||
| SkewnessH § | - | 31% | 90% | 0.53 | 71% | |||||||||
| LRHGE § | - | 8% | 100% | 0.52 | 71% | |||||||||
| Verger et al., 2018 | [42] | Retrospective | 90 | Grade 2:16 | 51 | 55 M 35 F | PET | TBRmean | 1.85 | 44% | 92% | 0.73 | 69% | |
| Grade 3:27 | TBRmax | 2.15 | 56% | 77% | 0.68 | 67% | ||||||||
| Grade 4:47 | TTP | 25 min | 86% | 60% | 0.75 | 72% | ||||||||
| Slope | −0.26 SUV/h | 81% | 60% | 0.75 | 70% | |||||||||
| TBRmean + TBRmax | 1.85 and 2.15 | 44% | 91% | - | 69% | |||||||||
| TTP + Slope | 25 min and −0.26 SUV/h | 77% | 70% | - | 73% | |||||||||
| TBRmean + TTP | 1.85 and 25 min | 40% | 96% | - | 69% | |||||||||
| TBRmax + TTP | 2.15 and 25 min | 51% | 94% | - | 73% | |||||||||
| TBRmean + Slope | 1.85 and −0.26 SUV/h | 40% | 94% | - | 68% | |||||||||
| TBRmax + Slope | 2.15 and −0.26 SUV/h | 47% | 91% | - | 70% | |||||||||
| Blanc-Durand et al., 2018 | [43] | Retrospective | 37 | Grade 1:3 | 45 | 23 M 14 F | PET | TBRmax | - | - | ||||
| Grade 2:15 | TBRmean | - | - | |||||||||||
| Grade 3:14 | TTP | - | - | |||||||||||
| Grade 4:5 | Slope | - | - | |||||||||||
| TAC | Centroid #1 vs. centroid #3 | - | - | |||||||||||
| Bette et al., 2016 | [44] | Retrospective | 65 | Grade 1:11 | 38 | 36 M 29 F | PET | TBR # | 1.3 | 89% | 36% | |||
| Grade 2:54 | TBR # | 1.6 | 71% | 53% | ||||||||||
| TBR # | 2.0 | 57% | 68% | |||||||||||
| TBRmax § | - | - | - | |||||||||||
| Prediction of oligodendroglial components | ||||||||||||||
| Jansen et al., 2012 | [38] | Prospective | 144 | Grade 2:79 | 45 | 84 M 60 F | PET | SUVmax/BG | 2.6 | 70% | 72% | |||
| Grade 3:65 | BTV | 4.0 | 71% | 69% | ||||||||||
| SUVmean/BG | 2.1 | 61% | 59% | |||||||||||
| SUVtotal/BG | 6.9 | 75% | 66% | |||||||||||
| Bette et al., 2016 | [44] | Retrospective | 65 | Grade 1:11 | 38 | 36 M 29 F | PET | TBR # | 1.3 | 100% | 23% | |||
| Grade 2:54 | TBR # | 1.6 | 93% | 48% | ||||||||||
| TBR # | 2.0 | 86% | 65% | |||||||||||
| TBRmax | - | - | - | |||||||||||
| Guided resection/biopsy | ||||||||||||||
| Ort et al., 2021 | [45] | Retrospective | 30 | Grade 3:5 | 59 | 19 M 11 F | PET | BTV | 1 cm3 | |||||
| Grade 4:25 | ||||||||||||||
| Floeth et al., 2011 | [46] | Prospective | 30 patients/38 biopsies | Grade 2:17 | 43 | 20 M 10 F | PET | TBR | 1.6 | |||||
| Grade 3:19 | MRI | Gd-DTPA enhancement | - | |||||||||||
| Grade 4:2 | 5-ALA-fluorescence | Fluorescent areas | - | |||||||||||
| Ewelt et al., 2011 | [47] | Prospective | 30 | Grade 2:13 | 42 | 20 M 10 F | LGG subgroup: | |||||||
| Grade 3:15 | PET | Tumor/brain tissue ratio | 1.6 | 54% | 12% | |||||||||
| Grade 4:2 | MRI | Gd enhancement | - | 8% | 36% | |||||||||
| 5-ALA-fluorescence | Fluorescent areas | - | 8% | 29% | ||||||||||
| PET/MRI | - | - | 8% | 35% | ||||||||||
| MRI/5-ALA | - | - | 8% | 41% | ||||||||||
| PET/5-ALA | - | - | 8% | 29% | ||||||||||
| PET/MRI/5-ALA | - | - | 8% | 41% | ||||||||||
| HGG subgroup: | ||||||||||||||
| PET | Tumor/brain tissue ratio | 1.6 | 88% | 46% | ||||||||||
| MRI | Gd enhancement | - | 65% | 92% | ||||||||||
| 5-ALA-fluorescence | Fluorescent areas | - | 71% | 92% | ||||||||||
| PET/MRI | - | - | 65% | 92% | ||||||||||
| MRI/5-ALA | - | - | 59% | 92% | ||||||||||
| PET/5-ALA | - | - | 71% | 92% | ||||||||||
| PET/MRI/5-ALA | - | - | 59% | 92% | ||||||||||
| Verburg et al., 2020 | [48] | Prospective | 20 | Grade 2:8 | - | 12 M 8 F | PET | TBR | - | - | - | 0.76 | ||
| Grade 4:12 | T1G-MRI | - | - | - | - | 0.56 | ||||||||
| PET/MRI | ADC + TBR | - | - | - | 0.89 | |||||||||
| Detection of residual tumor | ||||||||||||||
| Buchmann et al., 2016 | [49] | Retrospective | 62 | Grade 4:62 | 61 | 37 M 25 F | PET | TBR | 1.6 | |||||
| MRI | Contrast-enhanced tissue areas | - | ||||||||||||
| Kläsner et al., 2015 | [50] | Prospective | 25 | Grade 2:4 | 62 | 16 M 9 F | PET | Visual uptake | >Background | |||||
| Grade 3:3 | MRI | Contrast-enhancement volume | 0.175 cm2 | |||||||||||
| Grade 4:18 | ||||||||||||||
| Guided radiotherapy | ||||||||||||||
| Allard et al., 2022 | [51] | Prospective | 23 | Grade 3:3 | 59 | 14 M 9 F | PET | TBRmax # | 1.6 | |||||
| Grade 4:20 | SUVmax # | 30% | ||||||||||||
| SUVmax # | 40% | |||||||||||||
| SUVmax # | 50% | |||||||||||||
| SUVmax # | 60% | |||||||||||||
| SUVmax # | 70% | |||||||||||||
| SUVmax # | 80% | |||||||||||||
| SUVmax # | 90% | |||||||||||||
| CE-MRI | Visual analysis # | - | ||||||||||||
| Munck af Rosenschold et al., 2015 | [52] | Prospective | 54 | Grade 3:19 | 55 | - | PET | TBR # | 1.6 | |||||
| Grade 4:35 | CE-MRI | Visual analysis # | - | |||||||||||
| Fleischmann et al., 2020 | [53] | Retrospective | 36 | Grade 4:36 | 66 | 20 M 16 F | PET | TBRmax # | 1.6 | |||||
| MRI | Visual analysis # | |||||||||||||
| Harat et al., 2016 | [54] | Prospective | 34 | Grade 4:34 | - | - | PET | FET uptake # | 1.6 x SUVmean | |||||
| MRI | Visual analysis # | - | ||||||||||||
| Dissaux et al., 2020 | [55] | Prospective | 30 | Grade 3:5 | 63 | 20 M 10 F | PET | TBR# | 1.6 | |||||
| Grade 4:25 | MRI | Visual analysis # | - | |||||||||||
| Hayes et al., 2018 | [56] | Retrospective | 26 | Grade 3:5 | 61 | 17 M 9 F | PET | TBR # | 1.6 | |||||
| Grade 4:21 | CE-MRI | Visual analysis # | - | |||||||||||
| FLAIR-MRI | Visual analysis # | - | ||||||||||||
| Detection of malignant transformation in LGG | ||||||||||||||
| Galldiks et al., 2013 | [57] | Prospective | 27 | Grade 2:27 | 44 | 18 M 9 F | PET | TBRmax | ∆33% | 72% | 89% | 0.87 | 78% | |
| TBRmean | ∆13% | 72% | 78% | 0.80 | 74% | |||||||||
| TTP | ∆-6 min | 72% | 89% | 0.78 | 78% | |||||||||
| Kinetic pattern change | I to II/III | 72% | 89% | - | 78% | |||||||||
| TBRmax + TTP + Kinetic pattern change | ∆ + 33% or ∆-6 min or I to II/III | 83% | 78% | - | 81% | |||||||||
| MRI | Contrast enhancement change | - | 44% | 100% | - | 63% | ||||||||
| Unterrainer et al., 2016 | [58] | Retrospective | 31 | Grade 2:26 | 38 | 18 M 13 F | PET | TBRmax | 2.46 | 82% | 89% | 0.92 | 85% | |
| Grade 3:5 | TTPmin | 17.5 min | 73% | 67% | - | 70% | ||||||||
| Bashir et al., 2018 | [59] | Retrospective | 42 patients/47 PET | Inconclusive:2 | 41 | 18 M 24 F | PET | TBRmax § | - | 57% | 41% | 0.476 | ||
| Grade 1:1 | TAC § | - | 71% | 41% | 0.549 | |||||||||
| Grade 1/2:1 | TTP § | 25 min | 57% | 47% | 0.511 | |||||||||
| Grade 2:43 | TBRmax + TAC + TTP § | 1.6 + II/III + 25 min | 65% | 58% | 0.634 | |||||||||
| TBRmax + TAC§ | 1.6 + II/III | 65% | 58% | 0.639 | ||||||||||
| TBRmax + TTP § | 1.6 + 25 min | 96% | 25% | 0.591 | ||||||||||
| MRI | Contrast enhancement § (CE) | new area | 43% | 77% | 0.597 | |||||||||
| PET/MRI | TBRmax + TAC + TTP + CE § | - | 70% | 50% | 0.643 | |||||||||
| TBRmax + TAC + CE § | - | 52% | 75% | 0.656 | ||||||||||
| TBRmax + TTP + CE § | - | 57% | 58% | 0.620 | ||||||||||
| Recurrence vs. treatment-related changes | ||||||||||||||
| Jeong et al., 2010 | [60] | Retrospective | 32 | Grade 2:10 | 47 | 12 M 20 F | PET | SUVmax | 1.66 | 87% | 100% | 0.978 | ||
| Grade 3:8 | LNR | 2.18 | 86% | 88% | 0.940 | |||||||||
| Grade 4:14 | LGG subgroup: | |||||||||||||
| SUVmax | 1.48 | 88% | 89% | 0.951 | ||||||||||
| LNR | 1.64 | 100% | 75% | 0.893 | ||||||||||
| HGG subgroup: | ||||||||||||||
| SUVmax | 1.66 | 93% | 100% | 0.993 | ||||||||||
| LNR | 2.46 | 86% | 100% | 0.964 | ||||||||||
| Jansen et al., 2013 | [61] | Prospective | 33 | Grade 3:20 | - | - | PET | BTV after 6 months | - | |||||
| Grade 4:13 | SUVmax/BG after 6 months | - | ||||||||||||
| Puranik et al., 2021 | [62] | Retrospective | 72 | Grade 3:13 | - | 47 M 25 F | PET | T/Wm | 2.65 | 80% | 88% | |||
| Grade 4:59 | ||||||||||||||
| Kertels et al., 2019 | [63] | Retrospective | 36 | Grade 4:36 | 54 | 22 M 14 F | PET | TBRmax | 3.69 | 79% | 88% | 0.86 | ||
| TBRmax | 3.58 | 64% | 100% | 0.84 | ||||||||||
| TBRmax | 3.44 | 86% | 88% | 0.86 | ||||||||||
| TBRmean | 2.31 | 61% | 100% | 0.83 | ||||||||||
| TBRmean | 2.19 | 71% | 88% | 0.80 | ||||||||||
| TBR16 mm | 2.44 | 82% | 75% | 0.82 | ||||||||||
| TBR10 mm | 2.86 | 86% | 75% | 0.81 | ||||||||||
| TBR90% | 3.23 | 71% | 100% | 0.85 | ||||||||||
| TBR80% | 3.08 | 82% | 88% | 0.88 | ||||||||||
| TBR70% | 2.72 | 86% | 88% | 0.87 | ||||||||||
| Verger et al., 2018 | [64] | Retrospective | 31 patients/32 tumors | Grade 2:2 | 52 | 16 M 15 F | PET | TBRmax | 2.61 | 80% | 86% | 0.78 | 81% | |
| Grade 3:3 | TBRmean § | - | - | - | 0.74 | - | ||||||||
| Grade 4:27 | TTP § | - | - | - | 0.71 | - | ||||||||
| Slope § | - | - | - | 0.70 | - | |||||||||
| PWI rCBF | TBRmax § | - | - | - | 0.65 | - | ||||||||
| TBRmean § | - | - | - | 0.55 | - | |||||||||
| PWI rCBV | TBRmax § | - | - | - | 0.58 | - | ||||||||
| TBRmean § | - | - | - | 0.64 | - | |||||||||
| PWI MTT | TBRmax § | - | - | - | 0.59 | - | ||||||||
| TBRmean § | - | - | - | 0.59 | - | |||||||||
| Pyka et al., 2018 | [65] | Retrospective | 47 patients/63 lesions | Grade 2:5 | 54 | 22 M 25 F | PET | TBR30–40 min | 2.07 | 80% | 85% | 0.863 | ||
| Grade 3:20 | TBR10–20 min | 1.71 | 76% | 85% | 0.848 | |||||||||
| Grade 4:38 | TTP | 20 min | 64% | 79% | 0.728 | |||||||||
| PWI MRI | rCBVuncor | 4.32 | 62% | 77% | 0.726 | |||||||||
| rCBVcor | 3.35 | 66% | 77% | 0.708 | ||||||||||
| DWI MRI | ADC | 1610 × 10−6 mm2/s | 50% | 77% | 0.688 | |||||||||
| nADC | 1.22 | 62% | 77% | 0.697 | ||||||||||
| FA § | 98.9 | 65% | 62% | 0.593 | ||||||||||
| PET/MRI | TBR30–40 min + TTP + rCBVcor + nADC | - | 78% | 92% | 0.891 | |||||||||
| Werner et al., 2021 | [66] | Retrospective | 23 | Grade 4:23 | 58 | 13 M 10 F | PET | TBRmax | 2.85 | 64% | 92% | 0.75 | 78% | |
| TBRmean | 1.95 | 82% | 92% | 0.77 | 87% | |||||||||
| Slope § | 0.02 SUV/h | 73% | 75% | 0.72 | 74% | |||||||||
| TTP | 35 min | 64% | 83% | 0.82 | 74% | |||||||||
| TBRmax + TTP | 2.85 and 35 min | 36% | 100% | - | 70% | |||||||||
| TBRmean + TTP | 1.95 and 35 min | 55% | 100% | - | 78% | |||||||||
| MRI | RANO criteria § | - | 30% | 79% | - | 58% | ||||||||
| Galldiks et al., 2015 | [67] | Retrospective | 22 | Grade 4:22 | 56 | 14 M 8 F | PET | TBRmax | 2.3 | 100% | 91% | 0.94 | 96% | |
| TBRmean | 2.0 | 82% | 82% | 0.91 | 82% | |||||||||
| Kinetic pattern | II/III | - | - | - | - | |||||||||
| TBRmax+ Kinetic pattern | 2.3 and II/III | 80% | 91% | - | 86% | |||||||||
| TBRmean+ Kinetic pattern | 2.0 and II/III | 60% | 91% | - | 76% | |||||||||
| Werner et al., 2019 | [68] | Retrospective | 48 | Grade 3:8 | 50 | 29 M 19 F | PET | TBRmax | 1.95 | 100% | 79% | 0.89 | 83% | |
| Grade 4:40 | TBRmean | 1.95 | 100% | 79% | 0.89 | 83% | ||||||||
| TTP | 32.5 min | 80% | 69% | 0.79 | 72% | |||||||||
| Slope | 0.32 SUV/h | 70% | 75% | 0.82 | 74% | |||||||||
| TBRmax/mean + TTP | 1.95 and 32.5 min | 89% | 91% | - | 90% | |||||||||
| TBRmax/mean + Slope | 1.95 and 0.32 SUV/h | 78% | 97% | - | 93% | |||||||||
| DWI-MRI | Visual assessment § | - | 70% | 66% | - | 67% | ||||||||
| ADC § | 1.09×10−3 mm2/s | 60% | 71% | 0.73 | 69% | |||||||||
| PET/MRI | TBRmax/mean + ADC | - | 67% | 94% | - | 89% | ||||||||
| Lohmann et al., 2020 | [69] | Retrospective | 34 | Grade 3:1 | 57 | 21 M 13 F | PET | TBRmax | 2.25 | 81% | 67% | 0.79 | 74% | |
| Grade 4:33 | TBRmean | 1.95 | 75% | 61% | 0.73 | 68% | ||||||||
| TTP § | 25 min | 75% | 44% | 0.61 | 59% | |||||||||
| Slope § | 0.3 SUV/h | 56% | 61% | 0.55 | 59% | |||||||||
| TBRmean + TBRmax | - | 75% | 72% | - | 74% | |||||||||
| TBRmean + TTP | - | 69% | 78% | - | 74% | |||||||||
| TBRmean + Slope § | - | 50% | 78% | - | 65% | |||||||||
| TBRmax + TTP | - | 69% | 83% | - | 76% | |||||||||
| TBRmax + Slope | - | 50% | 89% | - | 71% | |||||||||
| TTP + Slope § | - | 56% | 61% | - | 59% | |||||||||
| TBRmax + TBRmean + TTP | - | 69% | 89% | - | 79% | |||||||||
| Radiomics features | - | 100% | 40% | 0.74 | 70% | |||||||||
| Kebir et al., 2016 | [70] | Retrospective | 26 | Grade 4:26 | 58 | 21 M 5 F | PET | TBRmax | 1.9 | 84% | 86% | 0.88 | 85% | |
| TBRmean | 1.9 | 74% | 86% | 0.86 | 77% | |||||||||
| TAC | II/III | 84% | 100% | - | 89% | |||||||||
| TTP | - | - | - | 0.86 | - | |||||||||
| Rachinger et al., 2005 | [71] | Retrospective | 45 | Grade 1:1 | 45 | 23 M 22 F | PET | SUVmax | 2.2 | 100% | 93% | |||
| Grade 2:10 | MRI | Volume/Gd-enhancing area | ∆25%/new area | 94% | 50% | |||||||||
| Grade 3:12 | ||||||||||||||
| Grade 4:22 | ||||||||||||||
| Lohmeier et al., 2019 | [72] | Retrospective | 42 | Grade 1–2:2 | 47 | 32 M 10 F | PET | SUVmax § | - | - | - | - | ||
| Grade 3–4:40 | SUV80mean § | - | - | - | - | |||||||||
| SUV-BG § | - | - | - | - | ||||||||||
| TBR80mean | - | - | - | - | ||||||||||
| TBRmax | 2.0 | 81% | 60% | 0.81 | ||||||||||
| DWI-MRI | ADCmean | 1254 × 10−6 mm2/s | 62% | 100% | 0.82 | |||||||||
| ADC-BG § | - | - | - | - | ||||||||||
| rADCmean | - | - | - | - | ||||||||||
| PET/MRI | TBRmax + ADCmean | - | 97% | 60% | 0.90 | |||||||||
| Bashir et al., 2019 | [73] | Retrospective | 146 | Grade 4:146 | 60 | 96 M 50 F | PET | TBRmax | 2.0 | 99% | 94% | 0.970 | 99% | |
| TBRmean | 1.8 | 96% | 94% | 0.977 | 96% | |||||||||
| BTV | 0.55 cm3 | 98% | 94% | 0.955 | 98% | |||||||||
| Steidl et al., 2020 | [74] | Retrospective | 104 | Grade 2:9 | 52 | 68 M 36 F | PET | TBRmax | 1.95 | 70% | 60% | 0.72 | 68% | |
| Grade 3:24 | TBRmean | - | - | - | 0.72 | - | ||||||||
| Grade 4:71 | TTP § | - | - | - | 0.60 | - | ||||||||
| Slope | 0.69 SUV/h | 84% | 62% | 0.69 | 80% | |||||||||
| TBRmax + Slope # | 1.95 and/or 0.69 SUV/h | 96% | 43% | - | 86% | |||||||||
| MRI | rCBVmax | 2.85 | 54% | 100% | 0.75 | 63% | ||||||||
| PET/MRI | rCBVmax + TBRmax + Slope # | - | 98% | 43% | - | 87% | ||||||||
| Pöpperl et al., 2006 | [75] | Prospective | 24 | Grade 3:5 | 49 | 15 M 9 F | PET | Tumax/BG # | 2.0 | 100% | 78% | |||
| Grade 4:19 | Tumax/BG # | 2.1 | 97% | 91% | ||||||||||
| Tumax/BG # | 2.2 | 82% | 95% | |||||||||||
| Tumax/BG # | 2.3 | 74% | 98% | |||||||||||
| Tumax/BG # | 2.4 | 74% | 100% | |||||||||||
| Tumax/BG # | 2.5 | 62% | 100% | |||||||||||
| Visual analysis # | Nodular vs. non-nodular | 94% | 94% | |||||||||||
| Müller et al., 2022 | [76] | Retrospective | 151 | Grade 2:28 | 52 | 97 M 54 F | PET | TBRmax | - | - | - | - | ||
| Grade 3:40 | TBRmean | - | - | - | - | |||||||||
| Grade 4:83 | TBRmax + TBRmean # | - | 66% | 80% | 0.78 | |||||||||
| Radiomics features # | - | 73% | 80% | 0.85 | ||||||||||
| TBRmax + TBRmean + radiomics features # | - | 81% | 70% | 0.85 | ||||||||||
| Mehrkens et al., 2008 | [77] | Prospective | 31 | Grade 2:17 | 46 | 17 M 14 F | PET | SUVmax/BG § | 2.0 | |||||
| Grade 3:6 | ||||||||||||||
| Grade 4:8 | ||||||||||||||
| Galldiks et al., 2015 | [78] | Retrospective | 124 | Grade 2:55 | 52 | 81 M 43 F | PET | TBRmax | 2.3 | 68% | 100% | 0.85 | 71% | |
| Grade 3:19 | TBRmean | 2.0 | 74% | 91% | 0.91 | 75% | ||||||||
| Grade 4:50 | TTP | 45 min | 82% | 73% | 0.81 | 81% | ||||||||
| Curve pattern | II/III | 78% | 73% | - | 77% | |||||||||
| TBRmax + Curve pattern | 2.3 and/or II/III | 93% | 73% | - | 91% | |||||||||
| TBRmean + Curve pattern | 2.0 and/or II/III | 93% | 73% | - | 91% | |||||||||
| TBRmax + TTP | 2.3 and/or 45 min | 92% | 73% | - | 90% | |||||||||
| TBRmean + TTP | 2.0 and/or 45 min | 93% | 100% | - | 93% | |||||||||
| MRI | RANO criteria § | - | 92% | 9% | - | 85% | ||||||||
| Pöpperl et al., 2004 | [79] | Prospective | 53 | Grade 1:1 | - | 28 M 25 F | PET | SUVmax | 2.2 | |||||
| Grade 2:9 | SUVmax/BG | 2.0 | ||||||||||||
| Grade 3:16 | SUV80/BG | - | ||||||||||||
| Grade 4:27 | SUV70/BG | - | ||||||||||||
| Prognosis/Treatment response evaluation | ||||||||||||||
| Müther et al., 2019 | [80] | Prospective | 31 | Grade 4:31 | 67 | 13 M 18 F | PET | Volume | 4.3 cm3 | |||||
| Jansen et al., 2013 | [61] | Prospective | 33 | Grade 3:20 | - | - | PET | Uptake kinetics | Increasing | |||||
| Grade 4:13 | ||||||||||||||
| Suchorska et al., 2018 | [81] | Retrospective | 61 | Grade 2:44 | 46 | 31 M 30 F | PET | Initial BTV § | - | |||||
| Grade 3:17 | Initial TBRmax § | - | ||||||||||||
| Initial TAC § | Increasing vs. decreasing | |||||||||||||
| BTV after 6 months | - | |||||||||||||
| TBRmax after 6 months § | - | |||||||||||||
| TAC after 6 months § | Increasing vs. decreasing | |||||||||||||
| BTV response | ∆ ± 25% | |||||||||||||
| TBRmax response | ∆ ± 10% | |||||||||||||
| TAC response § | Stable increasing vs. Decreasing to increasing vs. Increasing to decreasing vs. Stable decreasing | |||||||||||||
| FET-PET response | Yes vs. no | |||||||||||||
| MRI | Initial T2 volume | - | ||||||||||||
| T2 volume after 6 months | - | |||||||||||||
| T2 volume response § | RD vs. SD vs. PD | |||||||||||||
| Galldiks et al., 2012 | [82] | Prospective | 25 | Grade 4:25 | 54 | 15 M 10 F | PET | TBRmax change | ∆-10% (PFS)/∆-20% (OS) | 83% (OS) | 67% (OS) | 0.75 (OS) | ||
| TBRmean change | ∆-5% | 67% | 75% | 0.72 | ||||||||||
| Tvol 1.6 change | ∆0% (PFS) | - | - | - | ||||||||||
| MRI | Gd-volume § | ∆0%/∆-25% | - | - | - | |||||||||
| Suchorska et al., 2015 | [83] | Prospective | 79 | Grade 4:79 | - | - | PET | BTVpreRCx | 9.5 cm3 | 64% | 70% | |||
| LBRmax-preRCx | 2.9 (OS) | 68% | 73% | |||||||||||
| Initial TAC | Increasing vs. decreasing (OS) | - | - | |||||||||||
| MRI | Gd+ volume | - | - | - | ||||||||||
| Jansen et al., 2014 | [84] | Retrospective | 59 | Grade 2:59 | 43 | 32 M 27 F | PET | TAC | Increasing vs. decreasing | |||||
| Uptake § | Positive vs. negative | |||||||||||||
| SUVmax/BG § | - | |||||||||||||
| SUVmean/BG § | - | |||||||||||||
| SUVtotal/BG § | - | |||||||||||||
| BTV § | - | |||||||||||||
| MRI | Contrast enhancement § | Yes vs. no | ||||||||||||
| Largest diameter | 6 cm (PFS) | |||||||||||||
| Tumor crossing midline § | Yes vs. no | |||||||||||||
| Thon et al., 2015 | [85] | Prospective | 98 | Grade 2:54 | - | 56 M 42 F | PET | TAC | Homogeneous decreasing vs. focal decreasing vs. homogeneous increasing | |||||
| Grade 3:40 | SUVmax § | 2.3 | ||||||||||||
| Grade 4:4 | MRI | Tumor volume § | 35 mL | |||||||||||
| Kunz et al., 2018 | [86] | Prospective | 98 | Grade 2:59 | - | - | PET | TAC | Homogeneous increasing vs. mixed vs. homogeneous decreasing | |||||
| Grade 3:35 | TTPmin | >25 min vs. 12.5 < t ≤ 25 min vs. ≤12.5 min | ||||||||||||
| Grade 4:4 | SUVmax § | 2.3 | ||||||||||||
| MRI | Tumor volume § | 35 mL | ||||||||||||
| Ceccon et al., 2021 | [87] | Prospective | 41 | Grade 2:1 | 52 | 22 M 19 F | PET | TBRmax baseline | 2.0 (PFS)/1.9 § (OS) | |||||
| Grade 3:2 | TBRmean baseline § | 1.9 (PFS)/1.8 (OS) | ||||||||||||
| Grade 4:38 | MTV baseline | 28.2 mL (PFS)/13.8 mL (OS) | ||||||||||||
| TBRmax change | 0% | |||||||||||||
| TBRmean change § | 0% | |||||||||||||
| MTV change | 0% | |||||||||||||
| MRI | RANO criteria § | SD/PR/CR vs. PD | ||||||||||||
| Galldiks et al., 2018 | [88] | Prospective | 21 | Grade 4:21 | 55 | 13 M 8 F | PET | TBRmax relative reduction § | 27% | 92% | 63% | 0.78 | ||
| TBRmean relative reduction § | 16% | 92% | 63% | 0.81 | ||||||||||
| MTV relative reduction § | 27% | 77% | 63% | 0.82 | ||||||||||
| Absolute MTV at follow-up | 5 mL | 85% | 88% | 0.92 | ||||||||||
| MRI | RANO criteria § | PR or SD | 63% | 69% | - | |||||||||
| Carles et al., 2021 | [89] | Prospective | 32 | Grade 4:32 | 52 | 17 M 15 F | PET | Radiomic features: | ||||||
| SUVmin & | - | |||||||||||||
| SUVmean & | - | |||||||||||||
| GLV & | - | |||||||||||||
| GLV2 & | - | |||||||||||||
| WF_GLV & | - | |||||||||||||
| Qacor & | - | |||||||||||||
| QHGZE & | - | |||||||||||||
| QSZHGE & | - | |||||||||||||
| QGLN2 & | - | |||||||||||||
| QHGRE & | - | |||||||||||||
| QSRHGE & | - | |||||||||||||
| QLRHGE & | - | |||||||||||||
| SZLGE | - | |||||||||||||
| Busyness & | - | |||||||||||||
| WF_TS & | - | |||||||||||||
| QvarianceCM & | - | |||||||||||||
| Eccentricity & | - | |||||||||||||
| SUVmean + WF_GLV + QLRHGE + SUVmin | - | |||||||||||||
| SZLGE + Busyness + QVarianceCM + Eccentricity | - | |||||||||||||
| Suchorska et al., 2018 | [90] | Retrospective | 300 | Grade 2:121 | 48 | 166 M 134 F | PET | TBRmax § | 1.6 | |||||
| Grade 3:106 | TBRmax § | 2.6 | ||||||||||||
| Grade 4:73 | TTPmin | 17.5 min (OS) | ||||||||||||
| MRI | Contrast enhancement § | Yes vs. no | ||||||||||||
| T2 volume § | 49 mL | |||||||||||||
| Wirsching et al., 2021 | [91] | Retrospective | 31 | Grade 4:31 | - | - | PET | TBR in non-contrast enhancing tumor portions at follow-up | High vs. low | |||||
| MRI | Contrast enhancement at baseline | - | ||||||||||||
| ADC at baseline | - | |||||||||||||
| Contrast enhancement at follow-up | - | |||||||||||||
| Sweeney et al., 2013 | [92] | Retrospective | 28 | Grade 2:5 | - | 21 M 7 F | PET | SUVmax | 2.6 | |||||
| Grade 3:12 | TBRmax § | - | ||||||||||||
| Grade 4:11 | TBRmean§ | - | ||||||||||||
| Tumor volume § | ||||||||||||||
| VolSUVmax ≥ 2.2 | - | |||||||||||||
| Vol ≥ 40%SUVmax | - | |||||||||||||
| MRI | VolMRI | - | ||||||||||||
| PET/MRI | VolMRI + VolSUVmax ≥ 2.2 | - | ||||||||||||
| VolMRI + Vol≥ 40%SUVmax | - | |||||||||||||
| Non-overlap, VolMRI + VolSUVmax ≥ 2.2 | - | |||||||||||||
| Non-overlap, VolMRI + Vol ≥ 40%SUVmax | - | |||||||||||||
| Pyka et al., 2014 | [93] | Retrospective | 34 | Grade 1:2 | 41 | 22 M 12 F | PET | TBRmax | 2.5 | 0.696 | ||||
| Grade 2:19 | TBRmean | 2.3 | 0.696 | |||||||||||
| Grade 3:3 | TTP | 20 min | 0.848 | |||||||||||
| Grade 4:10 | Peak TBR | 2.2 | 0.704 | |||||||||||
| Slope-to-peak | 7 × 10−5/s | 0.711 | ||||||||||||
| Wollring et al., 2022 | [94] | Retrospective | 36 | Grade 3:8 | 54 | 20 M 16 F | PET | New distant FET hotspot | Yes vs. no | |||||
| Grade 4:28 | TBRmax change | 0% | ||||||||||||
| TBRmean change § | 0% | |||||||||||||
| MTV change | 0% | |||||||||||||
| TTP change § | 0% | |||||||||||||
| MRI | RANO criteria | SD/PR/CR vs. PD | ||||||||||||
| Bauer et al., 2020 | [95] | Retrospective | 60 | Grade 3:15 | 55 | 35 M 25 F | PET | TBRmax § | 2.55 | 70% | 57% | 0.63 | ||
| Grade 4:45 | TBRmean § | 2.05 | 60% | 70% | 0.69 | |||||||||
| MTV § | 11.15 mL | 72% | 54% | 0.56 | ||||||||||
| TTP | 25 min | 90% | 87% | 0.90 | ||||||||||
| Slope § | −0.103 SUV/h | 70% | 90% | 0.77 | ||||||||||
| Piroth et al., 2011 | [96] | Prospective | 44 | Grade 4:44 | 57 | 16 M 28 F | PET | VolTBR ≥ 1.6 | 25 mL | |||||
| VolTBR ≥ 2.0 | 10 mL | |||||||||||||
| TBRmax | 2.4 | |||||||||||||
| TBRmean | 2.0 | |||||||||||||
| MRI | Gd-volume § | 10 mL | ||||||||||||
| Jansen et al., 2015 | [97] | Retrospective | 121 | Grade 3:51 | 54 | 73 M 48 F | PET | TTPmin | 12.5 min | |||||
| Grade 4:70 | SUVmax/BG § | - | ||||||||||||
| SUVmean/BG § | - | |||||||||||||
| BTV § | - | |||||||||||||
| MRI | contrast enhancement § | Yes vs. no | ||||||||||||
| Moller et al., 2016 | [98] | Prospective | 31 | Grade 3:6 | 54 | - | PET | BTV baseline | - | |||||
| Grade 4:25 | Tmax/B baseline # | - | ||||||||||||
| ∆BTV scan 2 § | - | |||||||||||||
| ∆BTV scan 3 § | - | |||||||||||||
| ∆Tmax/B scan 2 # | - | |||||||||||||
| ∆Tmax/B scan 3 # | - | |||||||||||||
| MRI | Volume (+necrosis) § | - | ||||||||||||
| Volume (−necrosis) | - | |||||||||||||
| Dissaux et al., 2020 | [99] | Prospective | 29 | Grade 3:3 | 60 | 17 M 12 F | PET | TBRmax | Median (5.03) | |||||
| Grade 4:26 | TBRmean § | Median | ||||||||||||
| SUVmax § | Median | |||||||||||||
| SUVmean § | Median | |||||||||||||
| SUVpeak § | Median | |||||||||||||
| TLG § | Median | |||||||||||||
| Volume § | Median | |||||||||||||
| Piroth et al., 2011 | [100] | Prospective | 22 | Grade 4:22 | 56 | 13 M 9 F | PET | Volume | 20 mL | |||||
| TBRmax § | 3.0 | |||||||||||||
| TBRmean § | 2.0 | |||||||||||||
| TBRmean | 2.4 | |||||||||||||
| Early TBRmax response | ∆-10% | |||||||||||||
| Early TBRmean response | ∆-10% | |||||||||||||
| MRI | Diameter of contrast-enhanced area | 4 cm | ||||||||||||
| Schneider et al., 2020 | [101] | Retrospective | 42 | Grade 2:19 | 46 | 26 M 16 F | PET | SUVmax | 3.4 | |||||
| Grade 3:23 | TBRmax | 3.03 | ||||||||||||
| BTV | 10 cm3 | |||||||||||||
| Kertels et al., 2019 | [102] | Retrospective | 35 | Grade 2:14 | 48 | 20 M 15 F | PET | FET positivity | Yes vs. no | |||||
| Grade 3:21 | ||||||||||||||
| Floeth et al., 2007 | [103] | Prospective | 33 | Grade 2:33 | - | 20 M 13 F | PET | Mean FET uptake | 1.1 | |||||
| Maximum FET uptake § | 2.0 | |||||||||||||
| MRI | Hemisphere§ | Right vs. left | ||||||||||||
| Brain lobe location § | - | |||||||||||||
| Extension § | Deep vs. superficial | |||||||||||||
| Size § | 3 cm | |||||||||||||
| Mass shift § | Yes vs. no | |||||||||||||
| Appearance | Circumscribed vs. diffuse | |||||||||||||
| PET/MRI | Mean FET uptake + MRI appearance | - | ||||||||||||
| Niyazi et al., 2012 | [104] | Retrospective | 56 | Grade 3:13 | 50 | 34 M 22 F | PET | Kinetics pre re-RT | G1–2 vs. G3 vs. G4–5 | |||||
| Grade 4:43 | Kinetics post re-RT § | G1–2 vs. G3 vs. G4–5 | ||||||||||||
| SUVmax/BG pre re-RT § | 3.3 | |||||||||||||
| SUVmax/BG post re-RT § | 2.6 | |||||||||||||
| SUVmean/BG pre re-RT § | 2.2 | |||||||||||||
| SUVmean/BG post re-RT § | 2.3 | |||||||||||||
| BTV pre re-RT § | 13.7 cc | |||||||||||||
| BTV post re-RT § | 7.3 cc | |||||||||||||
| Pyka et al., 2016 | [39] | Retrospective | 113 | Grade 3:26 | 59 | 43 M 70 F | PET | TBRmax § | 2.5 | |||||
| Grade 4:87 | TBRmean § | 1.56 (PFS)/1.57 (OS) | ||||||||||||
| MTV | 19.4 (PFS) §/18.9 (OS) | |||||||||||||
| TLU | 35.0 (PFS) §/17.1 (OS) | |||||||||||||
| Textural parameters: | ||||||||||||||
| Coarseness | 5.96 × 10−3 (PFS)/6.88 × 10−3 (OS) | |||||||||||||
| Contrast | 0.427 | |||||||||||||
| Busyness | 1.366 (PFS)/0.984 (OS) | |||||||||||||
| Complexity | 0.085 (PFS)/0.094 (OS) | |||||||||||||
| Blanc-Durand et al., 2018 | [43] | Retrospective | 37 | Grade 1:3 | 45 | 23 M 14 F | PET | TBRmax § | - | |||||
| Grade 2:15 | TBRmean § | - | ||||||||||||
| Grade 3:14 | TTP | - | ||||||||||||
| Grade 4:5 | Slope | - | ||||||||||||
| TAC | - |
| Parameter | Definition |
|---|---|
| TBRmean | Mean uptake in the tumor area with a TBR ≥ 1.6 divided by mean uptake in the normal brain |
| TBRmax | Maximal uptake in the tumor area divided by mean uptake in the normal brain |
| TBR10/16mm | Mean uptake in a ROI/VOI with a diameter of 10/16 mm centered on the tumor area with the highest uptake divided by mean uptake in the normal brain |
| TBR25mm2 | Mean uptake in a standardized ROI/VOI with a size of 25 mm2 placed manually at the biopsy sites centered to the titanium pellets on postoperative images divided by mean uptake in the normal brain |
| TBR3SD | Mean uptake in an isocontour region around the lesion maximum using a cutoff of three standard deviations above average activity in the reference region divided by mean uptake in the normal brain |
| TBR70/80% | Mean in a 70/80% isocontour region divided by mean uptake in the normal brain |
| TBR | Uptake in the tumor area (unspecified) divided by mean uptake in the normal brain |
| SUVmax/mean/BG | SUVmax/mean of the tumor area divided by maximal uptake in the normal brain |
| Indication | Number of Studies | Grade | Parameters | Threshold | Sensitivity | Specificity | AUC | Accuracy | Significance |
|---|---|---|---|---|---|---|---|---|---|
| Diagnosis | |||||||||
| 1 | LGG and HGG | Visual grading system | - | - | - | - | - | NA | |
| 1 | LGG and HGG | TBRmax | - | - | - | NA | |||
| 1 | LGG and HGG | TBR25mm2 | 1.6 | 92% | 81% | - | * | ||
| 1 | LGG and HGG | TBR3SD | - | - | - | NA | |||
| 1 | LGG and HGG | TBR | 1.6 | 88% | 88% | - | * | ||
| 1 | LGG and HGG | 18F-FETn uptake | 1.4 x background | 76% | 80% | 0.89 | 78% | * | |
| Grading (LGG vs. HGG) | |||||||||
| 1 | LGG and HGG | FET uptake | Reduced vs. normal vs. increased | - | - | NA | |||
| 1 | LGG and HGG | FET uptake pattern | Inhomogeneous vs. diffuse vs. focal | - | - | X | |||
| 1 | LGG and HGG | Early SUV | 2.32 | 73% | 71% | 72% | * | ||
| 1 | LGG and HGG | Middle SUV | - | - | - | - | - | X | |
| 1 | LGG and HGG | Late SUV | - | - | - | - | - | X | |
| 1 | LGG and HGG | e-m Ratio | 0.93 | 93% | 94% | 94% | * | ||
| 1 | LGG and HGG | e-l Ratio | 0.95 | 87% | 88% | 87% | * | ||
| 1 | LGG and HGG | SoD | 0.5 | 93% | 82% | 87% | * | ||
| 1 | LGG and HGG | SUVmax | - | - | - | * | |||
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | SUVsd | 0.45 | 67% | 87% | 0.816 | 83% | * |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | SUVmax/BG | - | - | - | * | ||
| 2 | LGG and HGG | SUVmean/BG | - | - | - | X | |||
| Grade 2 vs. 3 | LGG and HGG | - | - | - | X | ||||
| Grade 2 vs. 3 | 1 | LGG and HGG | SUVtotal/BG | - | - | - | X | ||
| 1 | LGG and HGG | SUV90 10–60 min | 0.2 | 94% | 100% | 0.969 | * | ||
| 1 | LGG and HGG | SUV90 15–60 min | −0.41 | 94% | 100% | 0.965 | * | ||
| 1 | LGG and HGG | TBRmax(0–10min) | 2.8 | 76% | 79% | 76% | * | ||
| 1 | LGG and HGG | TBRmax(5–15min) | 2.7 | 78% | 76% | 77% | * | ||
| 1 | LGG and HGG | TBRmax(5–20min) | 2.6 | 80% | 74% | 76% | * | ||
| 1 | LGG and HGG | TBRmax(10–30min) | 2.5 | 75% | 75% | 74% | * | ||
| 7 | LGG and HGG | TBRmax | 2.58 | 71% | 85% | 0.798 | * | ||
| LGG and HGG | 2.62 | 82% | 68% | 0.83 | 78% | * | |||
| Grade 2/3 vs. Grade 4 | LGG and HGG | 2.67 | 92% | 61% | 0.824 | 67% | * | ||
| LGG and HGG | 2.7 | 67% | 78% | 70% | * | ||||
| LGG and HGG | - | - | - | * | |||||
| LGG and HGG | - | - | - | X | |||||
| Grade 2 vs. 3 | LGG and HGG | - | - | - | X | ||||
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | TBRpeak | 2.35 | 92% | 61% | 0.832 | 67% | * |
| 2 | LGG and HGG | TBRmean | 2 | 83% | 58% | 0.65 | 75% | X | |
| Grade 2/3 vs. Grade 4 | LGG and HGG | 2.31 | 58% | 93% | 0.791 | 86% | * | ||
| 1 | LGG and HGG | ∆TBRmean 20–40 min/70–90 min | −8% | 83% | 75% | 0.85 | 81% | * | |
| 1 | LGG and HGG | TBR16mm | 1.69 | 82% | 68% | 0.8 | 78% | * | |
| Grade 3 vs. 4 | 3 | HGG | TBR | 1.68 | - | - | 0.644 | * | |
| Grade 3 vs. 4 | HGG | 2.74 | - | - | 0.614 | X | |||
| LGG and HGG | 3 | - | - | * | |||||
| 4 | LGG and HGG | TTP | 25 min | 87% | 100% | 94% | * | ||
| LGG and HGG | 30 min | 54% | 91% | 0.78 | 65% | * | |||
| LGG and HGG | 35 min | 58% | 92% | 0.76 | 69% | * | |||
| Grade 2/3 vs. Grade 4 | LGG and HGG | - | - | - | - | X | |||
| 1 | LGG and HGG | Slope | −0.03 SUV/h | 64% | 91% | 0.78 | 72% | * | |
| 7 | LGG and HGG | TAC | II/III | 88% | 75% | 83% | * | ||
| LGG and HGG | I/II vs. III | 73% | 100% | 87% | NA | ||||
| LGG and HGG | Decreasing | 90% | 66% | 80% | NA | ||||
| Grade 2 vs. 3 | LGG and HGG | 88% | 63% | NA | |||||
| LGG and HGG | Increasing vs. Decreasing | 95% | 72% | NA | |||||
| LGG and HGG | 96% | 94% | * | ||||||
| Grade 2/3 vs. Grade 4 | LGG and HGG | LGG-like vs. mixed vs. HGG-like | - | - | - | NA | |||
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | COV | 27.21 | 58% | 91% | 0.808 | 84% | * |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | HI | 1.77 | 67% | 87% | 0.826 | 83% | * |
| Grade 3 vs. 4 | 4 | HGG | BTV | 19.7 | - | - | 0.71 | * | |
| Grade 2/3 vs. Grade 4 | LGG and HGG | 20.13 | 75% | 80% | 0.801 | 79% | * | ||
| LGG and HGG | - | - | - | X | |||||
| Grade 2 vs. 3 | LGG and HGG | - | - | - | X | ||||
| Grade 3 vs. 4 | 2 | HGG | TLU | 46.2 | - | - | 0.704 | * | |
| Grade 2/3 vs. Grade 4 | LGG and HGG | 50.93 | 75% | 83% | 0.841 | 81% | * | ||
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | Relative K1 | - | 85% | 60% | 0.766 | * | |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | Relative K2 | - | - | - | - | X | |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | Relative K3 | - | - | - | - | X | |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | Relative FD | - | 67% | 78% | 0.716 | * | |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | TBRmax + SUVsd + TBRmean | - | 75% | 85% | 0.850 | 83% | * |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | HI + SUVsd + MTV | - | 75% | 83% | 0.848 | 81% | * |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | HI + SUVsd + TLU | - | 75% | 84% | 0.848 | 81% | * |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | SUVmax/BG + TTP | - | - | - | 0.745 | * | |
| Grade 2/3 vs. Grade 4 | 1 | LGG and HGG | SUVmax/BG + TTP + relative K1 + relative FD | - | - | - | 0.799 | * | |
| 1 | LGG and HGG | Logistic regression using early SUV + SoD | 50% | 93% | 100% | 97% | X | ||
| Radiomic features: | * | ||||||||
| Grade 3 vs. 4 | 1 | HGG | Coarseness | 0.607 | - | - | 0.757 | * | |
| Grade 3 vs. 4 | 1 | HGG | Contrast | 0.203 | - | - | 0.775 | * | |
| Grade 3 vs. 4 | 1 | HGG | Busyness | 1.12 | - | - | 0.737 | * | |
| Grade 3 vs. 4 | 1 | HGG | Complexity | 0.069 | - | - | 0.633 | * | |
| Grade 3 vs. 4 | 1 | HGG | Combined | 2.05 | - | - | 0.830 | * | |
| IDH status determination | |||||||||
| 2 | LGG and HGG | SUVsd | 0.11 | 47% | 57% | 0.710 | 66% | * | |
| LGG and HGG | 0.23 | - | - | - | - | * | |||
| 5 | LGG and HGG | TBRmax | 2.07 | 8% | 100% | 0.59 | 71% | X | |
| LGG and HGG | 2.21 | 48% | 87% | 0.658 | 72% | * | |||
| LGG | - | - | - | - | - | X | |||
| LGG and HGG | - | - | - | - | - | X | |||
| LGG and HGG | - | - | - | - | - | * | |||
| 2 | LGG and HGG | TBRpeak | 2.15 | 57% | 73% | 0.638 | 67% | X | |
| LGG and HGG | - | - | - | - | - | X | |||
| 5 | LGG and HGG | TBRmean | 1.68 | 12% | 100% | 0.66 | 73% | * | |
| LGG and HGG | 1.84 | 62% | 68% | 0.633 | 66% | X | |||
| LGG and HGG | 1.85 | 44% | 92% | 0.73 | 69% | * | |||
| LGG and HGG | - | - | - | - | - | X | |||
| LGG and HGG | - | - | - | - | - | * | |||
| 1 | LGG and HGG | TBR16mm | 2.15 | 56% | 77% | 0.68 | 67% | * | |
| 3 | LGG | TBR | 1.3 | 89% | 36% | - | - | NA | |
| LGG | 1.6 | 71% | 53% | - | - | NA | |||
| LGG | 2.0 | 57% | 68% | - | - | NA | |||
| 3 | LGG and HGG | TTP | 25 min | 86% | 60% | 0.75 | 72% | * | |
| LGG and HGG | 45 min | 27% | 93% | 0.75 | 73% | * | |||
| LGG and HGG | - | - | - | - | - | * | |||
| 3 | LGG and HGG | Slope | −0.26 SUV/h | 81% | 60% | 0.75 | 70% | * | |
| LGG and HGG | 0.30 SUV/h | 58% | 90% | 0.79 | 80% | * | |||
| LGG and HGG | - | - | - | - | - | * | |||
| 1 | LGG and HGG | TAC | centroid #1 vs. centroid #3 | - | - | - | - | * | |
| 1 | LGG and HGG | COV | 8.85 | 52% | 76% | 0.65 | 67% | * | |
| 1 | LGG and HGG | HI | 1.26 | 48% | 87% | 0.676 | 72% | * | |
| 2 | LGG and HGG | BTV | 19.48 | 90% | 46% | 0.66 | 62% | * | |
| LGG and HGG | - | - | - | - | - | X | |||
| 2 | LGG and HGG | TLU | 28.95 | 81% | 57% | 0.698 | 66% | * | |
| LGG and HGG | - | - | - | - | - | X | |||
| 1 | LGG and HGG | TBRmean + TBR16mm | 1.85 and 2.15 | 44% | 91% | - | 69% | * | |
| 1 | LGG and HGG | TTP + Slope | 25 min and −0.26 SUV/h | 77% | 70% | - | 73% | * | |
| 1 | LGG and HGG | TBRmean + TTP | 1.85 and 25 min | 40% | 96% | - | 69% | * | |
| 1 | LGG and HGG | TBR16mm + TTP | 2.15 and 25 min | 51% | 94% | - | 73% | * | |
| 1 | LGG and HGG | TBRmean + Slope | 1.85 and −0.26 SUV/h | 40% | 94% | - | 68% | * | |
| 1 | LGG and HGG | TBR16mm + Slope | 2.15 and −0.26 SUV/h | 47% | 91% | - | 70% | * | |
| 1 | LGG and HGG | TBRmax + SUVsd + TBRmean | - | 76% | 84% | 0.821 | 81% | * | |
| 1 | LGG and HGG | HI + SUVsd + MTV | - | 86% | 81% | 0.804 | 83% | * | |
| 1 | LGG and HGG | HI + SUVsd + TLU | - | 76% | 84% | 0.799 | 81% | * | |
| 1 | LGG and HGG | Midline involvement | Yes vs. no | - | - | - | - | * | |
| 1 | LGG and HGG | Simple predictive model | - | 85% | 71% | 0.786 | 76% | * | |
| 1 | LGG and HGG | PET-Radiomics model | - | 80% | 74% | 0.812 | 76% | * | |
| 1 | LGG and HGG | Slope + Radiomic feature SZHGE | - | 54% | 93% | - | 81% | * | |
| Radiomic features: | * | ||||||||
| 1 | LGG and HGG | SkewnessH | - | 31% | 90% | 0.53 | 71% | * | |
| 1 | LGG and HGG | LRHGE | - | 8% | 100% | 0.52 | 71% | * | |
| Prediction of oligodendroglial components | |||||||||
| 1 | LGG and HGG | SUVmean/BG | 2.1 | 61% | 59% | * | |||
| 1 | LGG and HGG | SUVtotal/BG | 6.9 | 75% | 66% | * | |||
| 2 | LGG and HGG | TBRmax | 2.6 | 70% | 72% | * | |||
| LGG | - | - | - | * | |||||
| 3 | LGG | TBR | 1.3 | 100% | 23% | NA | |||
| LGG | 1.6 | 93% | 48% | NA | |||||
| LGG | 2 | 86% | 65% | NA | |||||
| 1 | LGG and HGG | BTV | 4 mL | 71% | 69% | * | |||
| Guided resection/biopsy | |||||||||
| 1 | HGG | BTV | 1 cm3 | * | |||||
| 1 | LGG and HGG | TBR25mm2 | 1.6 | - | - | * | |||
| 3 | LGG | TBR | 1.6 | 54% | 12% | * | |||
| HGG | 88% | 46% | * | ||||||
| LGG and HGG | - | - | - | 0.76 | * | ||||
| Detection of residual tumor | |||||||||
| 1 | HGG | TBR | 1.6 | - | - | * | |||
| 1 | LGG and HGG | Visual uptake | >Background | - | - | * | |||
| Guided radiotherapy | |||||||||
| 7 | HGG | SUVmax | 30% | - | - | NA | |||
| HGG | 40% | - | - | NA | |||||
| HGG | 50% | - | - | NA | |||||
| HGG | 60% | - | - | NA | |||||
| HGG | 70% | - | - | NA | |||||
| HGG | 80% | - | - | NA | |||||
| HGG | 90% | - | - | NA | |||||
| 1 | HGG | TBRmax | 1.6 | - | - | NA | |||
| 5 | HGG | TBR | 1.6 | - | - | NA | |||
| HGG | - | - | NA | ||||||
| HGG | - | - | NA | ||||||
| HGG | - | - | NA | ||||||
| HGG | - | - | NA | ||||||
| Detection of malignant transformation in LGG | |||||||||
| 3 | LGG | TBRmax | ∆ + 33% | 72% | 89% | 0.87 | 78% | * | |
| LGG and HGG | 2.46 | 82% | 89% | 0.92 | 85% | * | |||
| LGG | - | 57% | 41% | 0.476 | X | ||||
| 1 | LGG | TBRmean | ∆ + 13% | 72% | 78% | 0.8 | 74% | * | |
| 2 | LGG | TTP | ∆-6 min | 72% | 89% | 0.78 | 78% | * | |
| LGG | 25 min | 57% | 47% | 0.511 | X | ||||
| 1 | LGG and HGG | TTPmin | 17.5 min | 73% | 67% | - | 70% | * | |
| 1 | LGG | TAC | - | 71% | 41% | 0.549 | X | ||
| 1 | LGG | TAC change | I to II/III | 72% | 89% | - | 78% | * | |
| 1 | LGG | TBRmax + TTP + TAC change | ∆ + 33% or ∆-6 min or I to II/III | 83% | 78% | - | 81% | * | |
| 1 | LGG | TBRmax + TAC + TTP | 1.6 + II/III + 25 min | 65% | 58% | 0.634 | X | ||
| 1 | LGG | TBRmax + TAC | 1.6 + II/III | 65% | 58% | 0.639 | X | ||
| 1 | LGG | TBRmax + TTP | 1.6 + 25 min | 96% | 25% | 0.591 | X | ||
| Recurrence vs. treatment-related changes | |||||||||
| 1 | HGG | Visual analysis | Nodular vs. non-nodular | 94% | 94% | NA | |||
| 6 | LGG | SUVmax | 1.48 | 88% | 89% | 0.951 | * | ||
| LGG and HGG | 1.66 | 87% | 100% | 0.978 | * | ||||
| HGG | 93% | 100% | 0.993 | * | |||||
| LGG and HGG | 2.2 | 100% | 93% | * | |||||
| LGG and HGG | - | - | * | ||||||
| LGG and HGG | - | - | - | X | |||||
| 1 | LGG and HGG | SUV80mean | - | - | - | X | |||
| 1 | LGG and HGG | SUV-BG | - | - | - | X | |||
| 20 | LGG | TBRmax | 1.64 | 100% | 75% | 0.893 | * | ||
| LGG and HGG | 2 | 81% | 60% | 0.81 | * | ||||
| LGG and HGG | - | - | X | ||||||
| LGG and HGG | - | - | * | ||||||
| HGG | 99% | 94% | 0.970 | 99% | * | ||||
| HGG | 100% | 78% | NA | ||||||
| HGG | 2.1 | 97% | 91% | NA | |||||
| LGG and HGG | 2.18 | 86% | 88% | 0.940 | * | ||||
| HGG | 2.2 | 82% | 95% | NA | |||||
| HGG | 2.3 | 74% | 98% | NA | |||||
| HGG | 2.4 | 74% | 100% | NA | |||||
| HGG | 2.46 | 86% | 100% | 0.964 | * | ||||
| HGG | 2.5 | 62% | 100% | NA | |||||
| LGG and HGG | 2.61 | 80% | 86% | 0.78 | 81% | * | |||
| HGG | 2.65 | 80% | 88% | * | |||||
| HGG | 2.85 | 64% | 92% | 0.75 | 78% | * | |||
| HGG | 3.44 | 86% | 88% | 0.86 | * | ||||
| HGG | 3.58 | 64% | 100% | 0.84 | * | ||||
| HGG | 3.69 | 79% | 88% | 0.86 | * | ||||
| LGG and HGG | - | - | - | - | * | ||||
| 1 | HGG | TBRmax after 6 months | - | - | - | * | |||
| 11 | HGG | TBRmean | 1.8 | 96% | 94% | 0.977 | 96% | * | |
| HGG | 1.9 | 74% | 86% | 0.86 | 77% | * | |||
| HGG | 1.95 | 82% | 92% | 0.77 | 87% | * | |||
| HGG | 100% | 79% | 0.89 | 83% | * | ||||
| HGG | 75% | 61% | 0.73 | 68% | * | ||||
| LGG and HGG | 2.0 | 74% | 91% | 0.91 | 75% | * | |||
| HGG | 82% | 82% | 0.91 | 82% | * | ||||
| HGG | 2.19 | 71% | 88% | 0.80 | * | ||||
| HGG | 2.31 | 61% | 100% | 0.83 | * | ||||
| LGG and HGG | - | - | - | 0.72 | * | ||||
| LGG and HGG | - | - | - | - | * | ||||
| 1 | LGG and HGG | TBR30–40min | 2.07 | 80% | 85% | 0.863 | * | ||
| 1 | LGG and HGG | TBR10–20min | 1.71 | 76% | 85% | 0.848 | * | ||
| 1 | HGG | TBR10mm | 2.86 | 86% | 75% | 0.81 | * | ||
| 8 | HGG | TBR16mm | 1.9 | 84% | 86% | 0.88 | 85% | * | |
| LGG and HGG | 1.95 | 70% | 60% | 0.72 | 68% | * | |||
| HGG | 100% | 79% | 0.89 | 83% | * | ||||
| HGG | 2.25 | 81% | 67% | 0.79 | 74% | * | |||
| LGG and HGG | 2.3 | 68% | 100% | 0.85 | 71% | * | |||
| HGG | 100% | 91% | 0.94 | 96% | * | ||||
| HGG | 2.44 | 82% | 75% | 0.82 | * | ||||
| LGG and HGG | - | - | - | 0.74 | X | ||||
| 2 | HGG | TBR70% | 2.72 | 86% | 88% | 0.87 | * | ||
| LGG and HGG | - | - | - | * | |||||
| 2 | HGG | TBR80% | 3.08 | 82% | 88% | 0.88 | * | ||
| LGG and HGG | - | - | - | * | |||||
| 1 | HGG | TBR90% | 3.23 | 71% | 100% | 0.85 | * | ||
| 1 | LGG and HGG | TBR80mean | - | - | - | * | |||
| 8 | LGG and HGG | TTP | 20 min | 64% | 79% | 0.728 | * | ||
| HGG | 25 min | 75% | 44% | 0.61 | 59% | X | |||
| HGG | 32.5 min | 80% | 69% | 0.79 | 72% | * | |||
| HGG | 35 min | 64% | 83% | 0.82 | 74% | * | |||
| LGG and HGG | 45 min | 82% | 73% | 0.81 | 81% | * | |||
| LGG and HGG | - | - | - | 0.60 | X | ||||
| LGG and HGG | - | - | - | 0.71 | * | ||||
| HGG | - | - | - | 0.86 | - | * | |||
| 5 | HGG | Slope | 0.02 SUV/h | 73% | 75% | 0.72 | 74% | X | |
| HGG | 0.3 SUV/h | 56% | 61% | 0.55 | 59% | X | |||
| HGG | 0.32 SUV/h | 70% | 75% | 0.82 | 74% | * | |||
| LGG and HGG | 0.69 SUV/h | 84% | 62% | 0.69 | 80% | * | |||
| LGG and HGG | - | - | - | 0.70 | * | ||||
| 3 | LGG and HGG | TAC | II/III | 78% | 73% | - | 77% | * | |
| HGG | 84% | 100% | - | 89% | * | ||||
| HGG | - | - | - | - | * | ||||
| 1 | HGG | BTV | 0.55 cm3 | 98% | 94% | 0.955 | 98% | * | |
| 1 | HGG | BTV after 6 months | - | * | |||||
| 1 | LGG and HGG | TBRmean + TBRmax | - | 66% | 80% | 0.78 | NA | ||
| 1 | HGG | TBRmean + TBR16mm | - | 75% | 72% | - | 74% | * | |
| 1 | HGG | TBRmax + TTP | 2.85 and 35 min | 36% | 100% | 70% | * | ||
| 3 | LGG and HGG | TBRmean + TTP | 2.0 and/or 45 min | 93% | 100% | 93% | * | ||
| HGG | 1.95 and 35 min | 55% | 100% | 78% | * | ||||
| HGG | - | 69% | 78% | - | 74% | * | |||
| 2 | LGG and HGG | TBR16mm + TTP | 2.3 and/or 45 min | 92% | 73% | 90% | * | ||
| HGG | - | 69% | 83% | 76% | * | ||||
| 1 | HGG | TBR16mm/mean + TTP | 1.95 and 32.5 min | 89% | 91% | 90% | * | ||
| 1 | HGG | TBRmax+ TAC | 2.3 and II/III | 80% | 91% | 86% | * | ||
| 2 | LGG and HGG | TBRmean + TAC | 2.0 and/or II/III | 93% | 73% | 91% | * | ||
| HGG | 2.0 and II/III | 60% | 91% | 76% | * | ||||
| 1 | LGG and HGG | TBR16mm + TAC | 2.3 and/or II/III | 93% | 73% | 91% | * | ||
| 1 | HGG | TBRmean + Slope | - | 50% | 78% | 65% | X | ||
| 2 | LGG and HGG | TBR16mm + Slope | 1.95 and/or 0.69 SUV/h | 96% | 43% | 86% | NA | ||
| HGG | - | 50% | 89% | 71% | * | ||||
| 1 | HGG | TBR16mm/mean + Slope | 1.95 and 0.32 SUV/h | 78% | 97% | 93% | * | ||
| 1 | HGG | TTP + Slope | - | 56% | 61% | 59% | X | ||
| 1 | HGG | TBR16mm + TBRmean + TTP | - | 69% | 89% | 79% | * | ||
| 2 | LGG and HGG | Radiomics features | - | 73% | 80% | 0.85 | NA | ||
| HGG | - | 100% | 40% | 0.74 | 70% | * | |||
| 1 | LGG and HGG | TBRmax + TBRmean + radiomics features | - | 81% | 70% | 0.85 | NA | ||
| Prognosis/Treatment response evaluation | |||||||||
| 1 | LGG | Uptake | Positive vs. negative | - | - | X | |||
| 1 | LGG and HGG | FET positivity | Yes vs. no | - | - | * | |||
| 1 | HGG | New distant FET hotspot | Yes vs. no | * | |||||
| 1 | LGG and HGG | FET-PET response | Yes vs. no | - | - | * | |||
| 3 | LGG and HGG | SUVmax/BG | - | - | - | X | |||
| LGG and HGG | - | - | - | X | |||||
| LGG and HGG | - | - | - | X | |||||
| 1 | LGG and HGG | Initial SUVmax/BG | - | - | - | X | |||
| 2 | LGG | SUVmean/BG | - | - | - | X | |||
| HGG | - | - | - | X | |||||
| 1 | HGG | SUVmean/BG pre re-RT | 2.2 | - | - | X | |||
| 1 | HGG | SUVmean/BG post re-RT | 2.3 | - | - | X | |||
| 1 | LGG | SUVtotal/BG | - | - | - | X | |||
| 5 | LGG and HGG | SUVmax | 2.3 | - | - | X | |||
| LGG and HGG | - | - | X | ||||||
| LGG and HGG | 2.6 | - | - | * | |||||
| LGG and HGG | 3.4 | - | - | * | |||||
| HGG | Median | - | - | X | |||||
| 1 | HGG | SUVmean | Median | - | - | X | |||
| 1 | HGG | SUVpeak | Median | - | - | X | |||
| 12 | LGG and HGG | TBRmax | 1.6 | - | - | X | |||
| LGG | 2 | - | - | X | |||||
| HGG | 2.4 | - | - | * | |||||
| LGG and HGG | 2.5 | - | - | 0.696 | * | ||||
| LGG and HGG | 2.6 | - | - | X | |||||
| HGG | 3 | - | - | X | |||||
| LGG and HGG | 3.03 | - | - | * | |||||
| HGG | Median (5.03) | * | |||||||
| LGG | - | - | - | X | |||||
| LGG and HGG | - | - | - | X | |||||
| LGG and HGG | - | - | - | X | |||||
| HGG | - | - | - | X | |||||
| 1 | HGG | TBRmax-preRCx | 2.9 (OS) | 68% | 73% | * | |||
| 2 | LGG and HGG | TBRmax baseline | 2.0 (PFS)/1.9 (OS) | - | - | * (PFS) | |||
| HGG | - | - | - | NA | |||||
| 1 | LGG and HGG | TBRmax after 6 months | - | - | - | X | |||
| 1 | HGG | Early TBRmax response | ∆-10% | - | - | * | |||
| 1 | LGG and HGG | TBRmax response | ∆ ± 10% | - | - | * | |||
| 3 | LGG and HGG | TBRmax change | 0% | - | - | * | |||
| HGG | - | - | * | ||||||
| HGG | ∆-10% (PFS)/∆-20% (OS) | 83% (OS) | 67% (OS) | 0.75 (OS) | * | ||||
| 1 | HGG | TBRmax pre re-RT | 3.3 | - | - | X | |||
| 1 | HGG | TBRmax post re-RT | 2.6 | - | - | X | |||
| 1 | HGG | TBR16mm relative reduction | 27% | 92% | 63% | 0.78 | NA | ||
| 1 | HGG | ∆TBRmax scan 2 | - | - | - | NA | |||
| 1 | HGG | ∆TBRmax scan 3 | - | - | - | NA | |||
| 2 | HGG | TBRmean | 2 | - | - | * | |||
| HGG | 2.05 | 60% | 70% | 0.69 | X | ||||
| 1 | HGG | TBRmean relative reduction | 16% | 92% | 63% | 0.81 | NA | ||
| 1 | LGG and HGG | TBR16mm baseline | 1.9 (PFS)/1.8 (OS) | - | - | X | |||
| 3 | LGG and HGG | TBR16mm change | 0% | - | - | X | |||
| HGG | - | - | X | ||||||
| HGG | ∆-5% | 67% | 75% | 0.72 | * | ||||
| 1 | HGG | TBR in non-contrast enhancing tumor portions at follow-up | High vs. low | - | - | * | |||
| 1 | LGG | TBR3SD | 1.1 | - | - | * | |||
| 1 | LGG and HGG | TBR10mm | 2.3 | - | - | 0.696 | * | ||
| 1 | HGG | TBR16mm | 2.55 | 70% | 57% | 0.63 | X | ||
| 5 | HGG | TBR | 1.56 (PFS)/1.57 (OS) | - | - | X | |||
| HGG | 2 | - | - | X | |||||
| HGG | 2.4 | - | - | * | |||||
| HGG | 2.5 | - | - | X | |||||
| HGG | Median | - | - | X | |||||
| 1 | HGG | Early TBR response | ∆-10% | * | |||||
| 1 | HGG | TLG | Median | X | |||||
| 1 | HGG | TLU | 35.0 (PFS)/17.1 (OS) | - | - | * (OS) | |||
| 1 | LGG and HGG | TTP | 20 min | - | - | 0.848 | * | ||
| 1 | HGG | 25 min | 90% | 87% | 0.90 | * | |||
| 1 | LGG and HGG | - | - | - | * | ||||
| 1 | HGG | TTP change | 0% | - | - | X | |||
| 1 | HGG | TTPmin | 12.5 min | - | - | * | |||
| 1 | LGG and HGG | >25 min vs. 12.5 < t ≤ 25 min vs. ≤12.5 min | - | - | * | ||||
| 1 | LGG and HGG | 17.5 min | - | - | * | ||||
| 1 | HGG | Slope | −0.103 SUV/h | 70% | 90% | 0.77 | X | ||
| 1 | LGG and HGG | - | - | - | * | ||||
| 1 | LGG and HGG | Slope-to-peak | 7 × 10−5/s | - | - | 0.711 | * | ||
| 5 | LGG | TAC | Increasing vs. decreasing | - | - | * | |||
| LGG and HGG | Homogeneous increasing vs. mixed vs. homogeneous decreasing | - | - | * | |||||
| LGG and HGG | Homogeneous decreasing vs. focal decreasing vs. homogeneous increasing | - | - | * | |||||
| HGG | Increasing | - | - | * | |||||
| LGG and HGG | - | - | - | * | |||||
| 1 | HGG | TAC pre re-RT | G1–2 vs. G3 vs. G4–5 | * | |||||
| 1 | HGG | TAC post re-RT | G1–2 vs. G3 vs. G4–5 | X | |||||
| 1 | LGG and HGG | Initial TAC | Increasing vs. decreasing | - | - | X | |||
| 1 | HGG | Increasing vs. decreasing (OS) | - | - | * | ||||
| 1 | LGG and HGG | TAC after 6 months | Increasing vs. decreasing | - | - | X | |||
| 1 | LGG and HGG | TAC response | Stable increasing vs. decreasing to increasing vs. Increasing to decreasing vs. Stable decreasing | - | - | X | |||
| 1 | LGG and HGG | Peak TBR | 2.2 | - | - | 0.704 | * | ||
| 8 | HGG | BTV | 4.3 cm3 | - | - | * | |||
| LGG and HGG | 10 cm3 | * | |||||||
| HGG | 11.15 mL | 72% | 54% | 0.56 | X | ||||
| HGG | 19.4 (PFS)/18.9 (OS) | - | - | * (OS) | |||||
| HGG | 20 mL | - | - | * | |||||
| HGG | Median | X | |||||||
| LGG | - | - | - | X | |||||
| HGG | - | - | - | X | |||||
| 1 | HGG | BTVpreRCx | 9.5 cm3 | 64% | 70% | * | |||
| 1 | LGG and HGG | Initial BTV | - | - | - | X | |||
| 1 | LGG and HGG | BTV baseline | 28.2 mL (PFS)/13.8 mL (OS) | - | - | * | |||
| 1 | HGG | - | - | - | * | ||||
| 1 | LGG and HGG | BTV after 6 months | - | - | - | * | |||
| 1 | HGG | Absolute BTV at follow-up | 5 mL | 85% | 88% | 0.92 | * | ||
| 1 | LGG and HGG | BTV response | ∆ ± 25% | - | - | * | |||
| 3 | LGG and HGG | BTV change | 0% | - | - | * | |||
| HGG | 0% | - | - | * | |||||
| HGG | 0% (PFS) | - | - | - | * | ||||
| 1 | HGG | BTV relative reduction | 27% | 77% | 63% | 0.82 | NA | ||
| 1 | HGG | ∆BTV scan 2 | - | - | - | X | |||
| 1 | HGG | ∆BTV scan 3 | - | - | - | X | |||
| 1 | LGG and HGG | BTVSUVmax≥2.2 | - | - | - | X | |||
| 1 | LGG and HGG | BTV≥40%SUVmax | - | - | - | X | |||
| 1 | HGG | BTVTBR≥ 1.6 | 25 mL | - | - | * | |||
| 1 | HGG | BTVTBR≥ 2.0 | 10 mL | - | - | * | |||
| 1 | HGG | BTV pre re-RT | 13.7 cc | - | - | X | |||
| 1 | HGG | BTV post re-RT | 7.3 cc | - | - | X | |||
| Radiomic features: | * | ||||||||
| 1 | HGG | SUVmin | - | - | - | *, & | |||
| 1 | HGG | SUVmean | - | - | - | *, & | |||
| 1 | HGG | GLV | - | - | - | *, & | |||
| 1 | HGG | GLV2 | - | - | - | *, & | |||
| 1 | HGG | WF_GLV | - | - | - | *, & | |||
| 1 | HGG | Qacor | - | - | - | *, & | |||
| 1 | HGG | QHGZE | - | - | - | *, & | |||
| 1 | HGG | QSZHGE | - | - | - | *, & | |||
| 1 | HGG | QGLN2 | - | - | - | *, & | |||
| 1 | HGG | QHGRE | - | - | - | *, & | |||
| 1 | HGG | QSRHGE | - | - | - | *, & | |||
| 1 | HGG | QLRHGE | - | - | - | *, & | |||
| 1 | HGG | SZLGE | - | - | - | * | |||
| 1 | HGG | Busyness | 1.366 (PFS)/0.984 (OS) | - | - | * | |||
| 1 | HGG | - | - | - | *, & | ||||
| 1 | HGG | WF_TS | - | - | - | *, & | |||
| 1 | HGG | QvarianceCM | - | - | - | *, & | |||
| 1 | HGG | Eccentricity | - | - | - | *, & | |||
| 1 | HGG | Coarseness | 5.96 × 10−3 (PFS)/6.88 × 10−3 (OS) | - | - | * | |||
| 1 | HGG | Contrast | 0.427 | - | - | * | |||
| 1 | HGG | Complexity | 0.085 (PFS)/0.094 (OS) | - | - | * | |||
| 1 | HGG | SUVmean + WF_GLV + QLRHGE + SUVmin | - | - | - | * | |||
| 1 | HGG | SZLGE + Busyness + QVarianceCM + Eccentricity | - | - | - | * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robert, J.A.; Leclerc, A.; Ducloie, M.; Emery, E.; Agostini, D.; Vigne, J. Contribution of [18F]FET PET in the Management of Gliomas, from Diagnosis to Follow-Up: A Review. Pharmaceuticals 2024, 17, 1228. https://doi.org/10.3390/ph17091228
Robert JA, Leclerc A, Ducloie M, Emery E, Agostini D, Vigne J. Contribution of [18F]FET PET in the Management of Gliomas, from Diagnosis to Follow-Up: A Review. Pharmaceuticals. 2024; 17(9):1228. https://doi.org/10.3390/ph17091228
Chicago/Turabian StyleRobert, Jade Apolline, Arthur Leclerc, Mathilde Ducloie, Evelyne Emery, Denis Agostini, and Jonathan Vigne. 2024. "Contribution of [18F]FET PET in the Management of Gliomas, from Diagnosis to Follow-Up: A Review" Pharmaceuticals 17, no. 9: 1228. https://doi.org/10.3390/ph17091228
APA StyleRobert, J. A., Leclerc, A., Ducloie, M., Emery, E., Agostini, D., & Vigne, J. (2024). Contribution of [18F]FET PET in the Management of Gliomas, from Diagnosis to Follow-Up: A Review. Pharmaceuticals, 17(9), 1228. https://doi.org/10.3390/ph17091228







