In Vitro Assessment of Wound-Healing Efficacy of Stabilized Basic Fibroblast Growth Factor (FGF-2) Solutions
Abstract
1. Introduction
2. Results
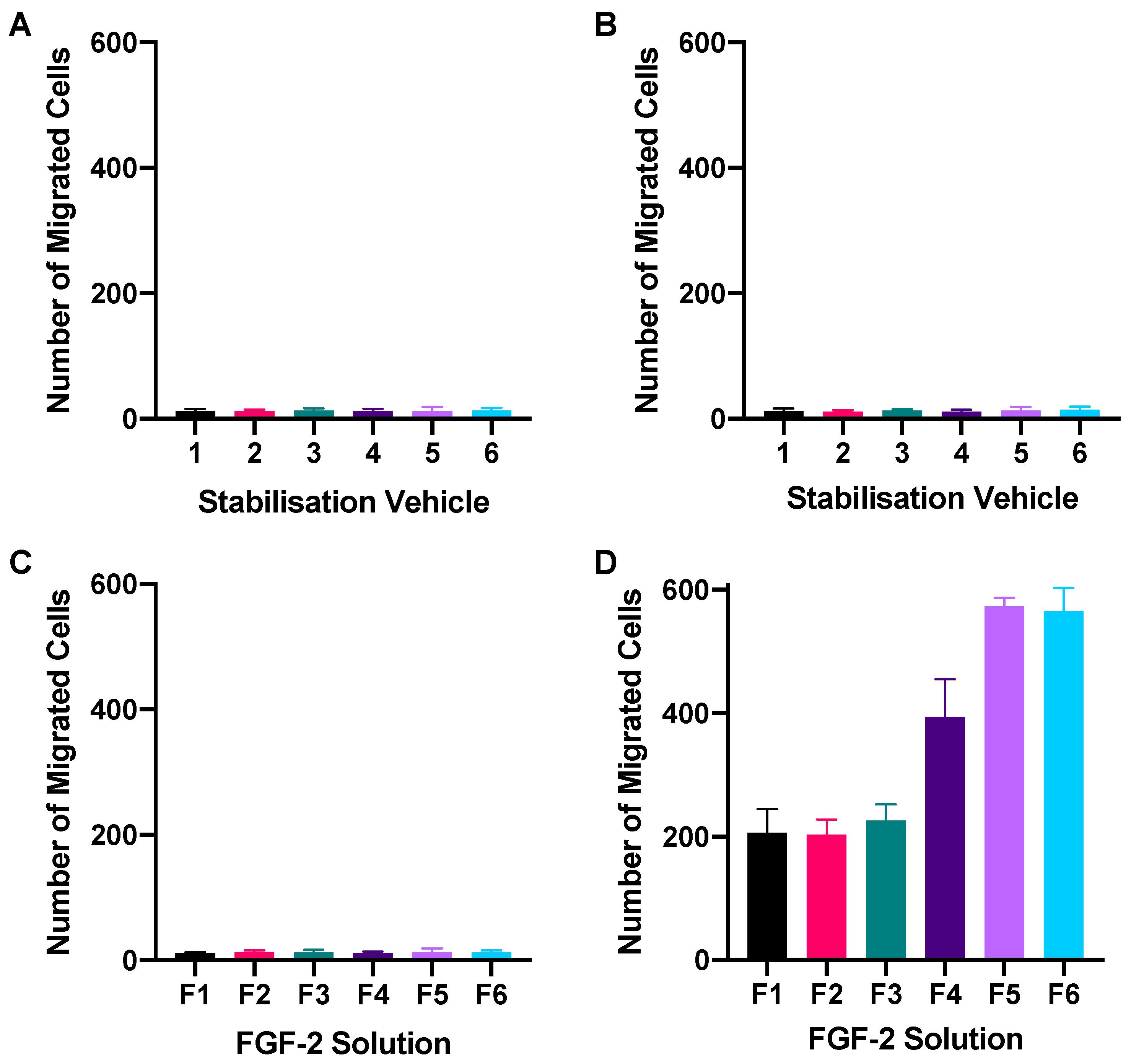
2.1. Dose-Response Assay
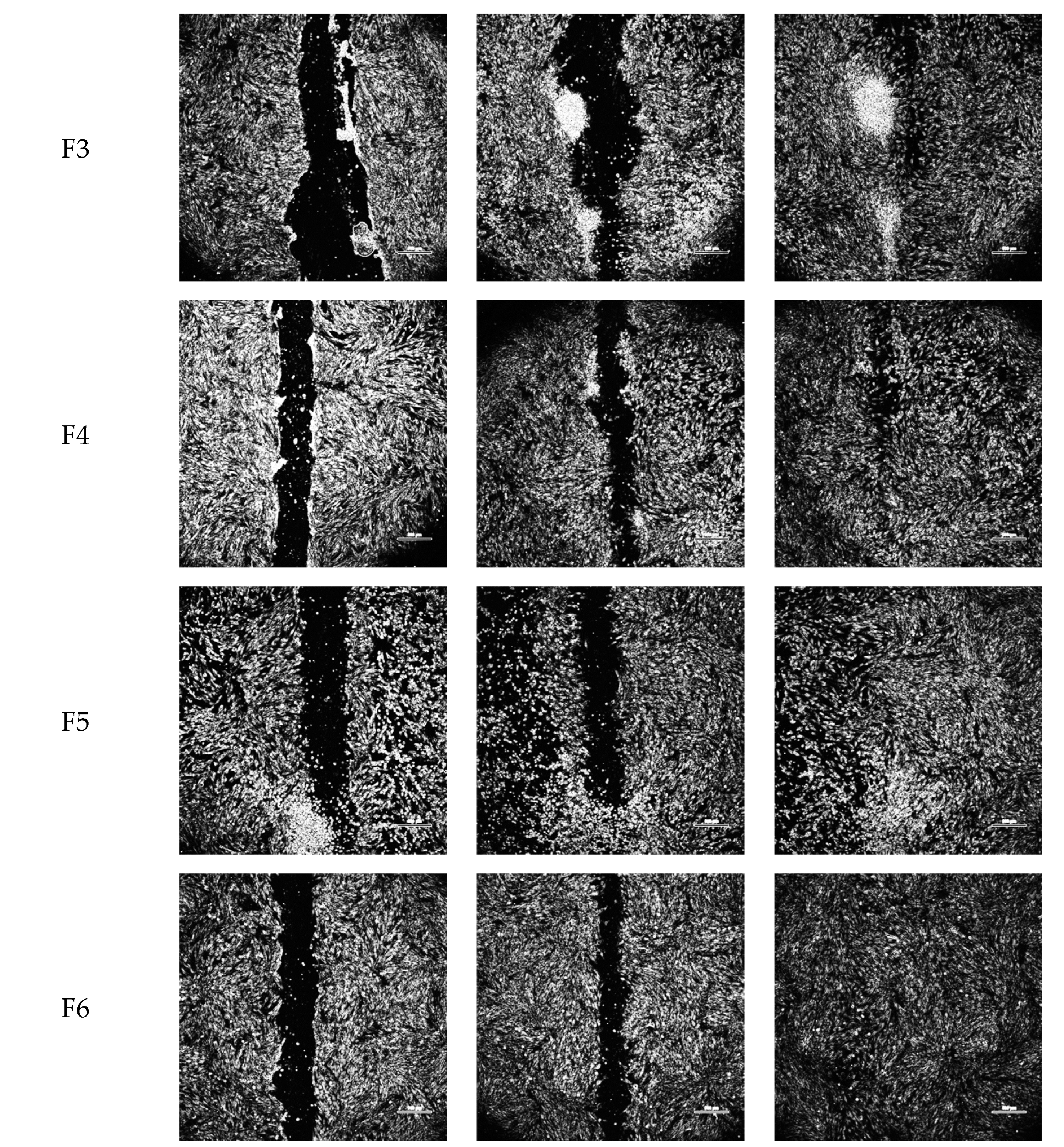
2.2. Wound-Healing Assay
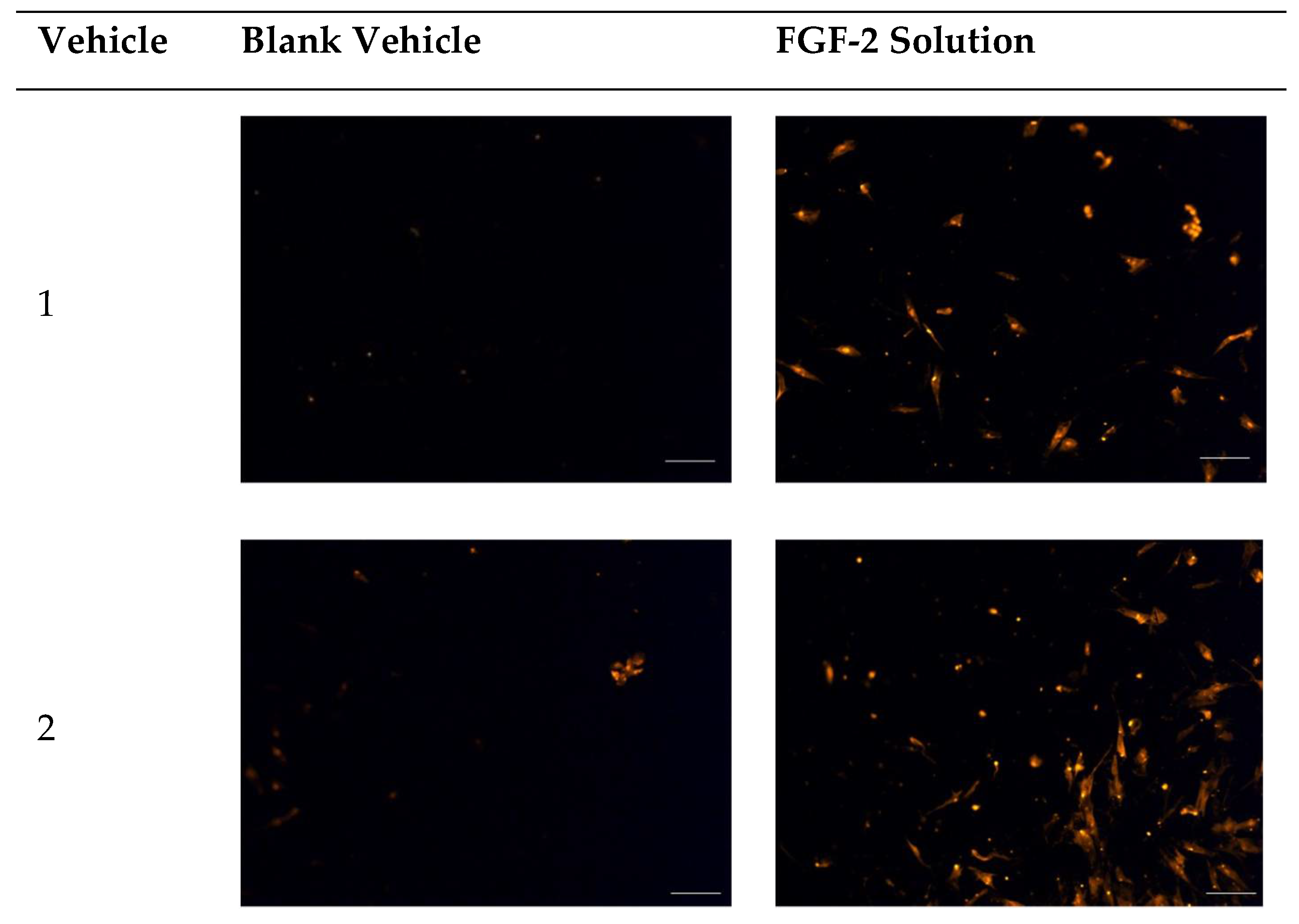
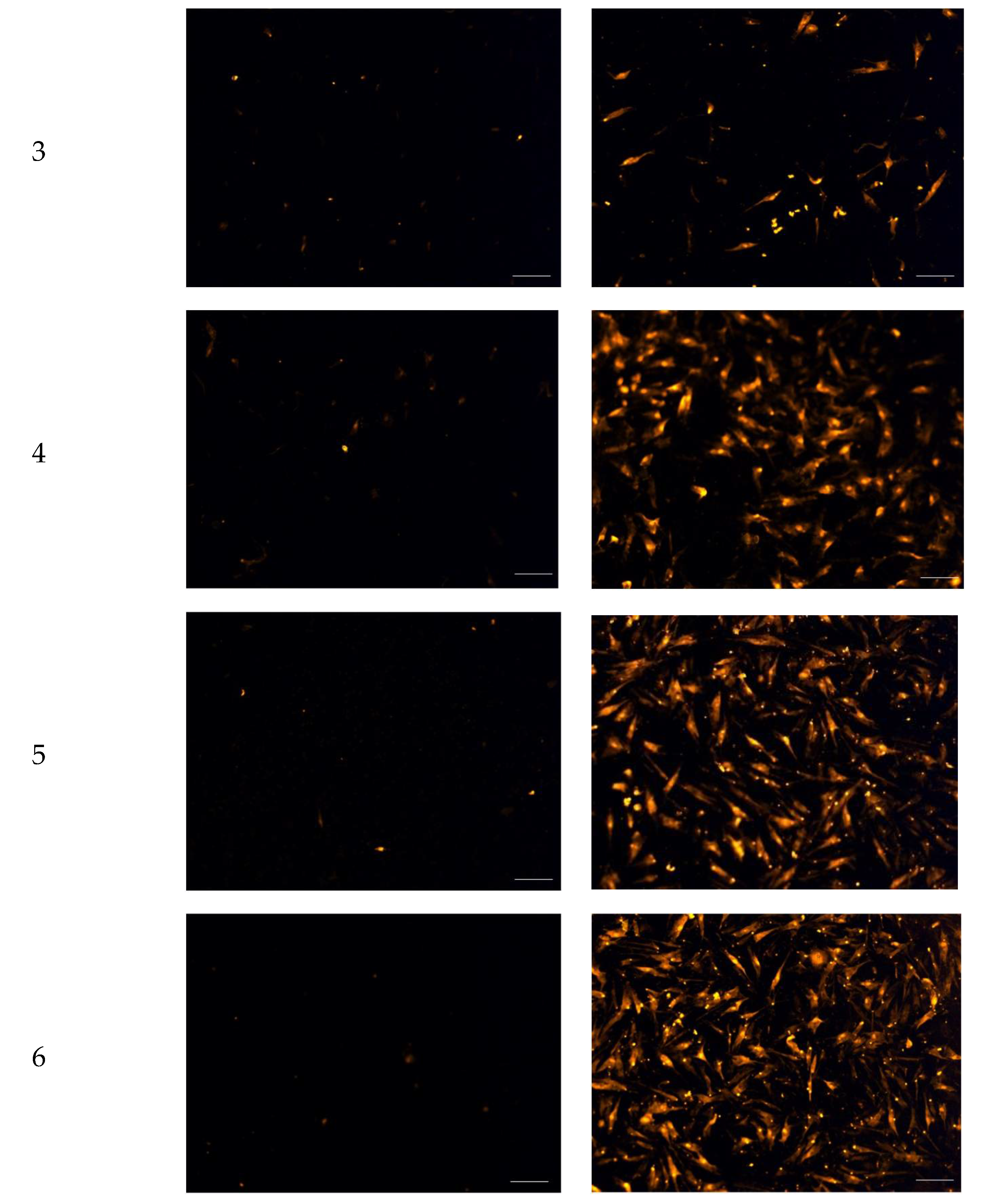
2.3. Chemotactic Migration Assay
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Sample Preparation and Quantification by ELISA
4.3. Cell Culture
4.4. Dose-Response Assay
4.5. In Vitro Wound-Healing Assay
4.6. Chemotactic Migration Assay
4.7. Data Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hakuba, N.; Hato, N.; Omotehara, Y.; Okada, M.; Gyo, K. Epithelial pearl formation following tympanic membrane regeneration therapy using an atelocollagen/silicone membrane and basic fibroblast growth factor: Our experience from a retrospective study of one hundred sixteen patients. Clin. Otolaryngol. 2013, 38, 394–397. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, H.B.; Jackler, R.K.; Varav, K. Tympanic membrane wound healing. An overview. Otolaryngol. Clin. N. Am. 1995, 28, 913–932. [Google Scholar] [CrossRef]
- O’Connor, T.E.; Perry, C.F.; Lannigan, F.J. Complications of otitis media in Indigenous and non-Indigenous children. Med. J. Aust. 2009, 191, S60–S64. [Google Scholar] [CrossRef]
- Zhang, Q.; Lou, Z. Impact of basic fibroblast growth factor on healing of tympanic membrane perforations due to direct penetrating trauma: A prospective non-blinded/controlled study. Clin. Otolaryngol. 2012, 37, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Hardman, J.; Muzaffar, J.; Nankivell, P.; Coulson, C. Tympanoplasty for chronic tympanic membrane perforation in children: Systematic review and meta-analysis. Otol. Neurotol. 2015, 36, 796–804. [Google Scholar] [CrossRef]
- Lou, Z.; Tang, Y.; Wu, X. Analysis of the effectiveness of basic fibroblast growth factor treatment on traumatic perforation of the tympanic membrane at different time points. Am. J. Otolaryngol. 2012, 33, 244–249. [Google Scholar] [CrossRef]
- Knapik, M.; Saliba, I. Pediatric myringoplasty: A study of factors affecting outcome. Int. J. Pediatr. Otorhinolaryngol. 2011, 75, 818–823. [Google Scholar] [CrossRef]
- Kakigi, A.; Uchida, A.; Nishimura, M.; Takeda, T.; Takeda, S.; Nakatani, H. Expression of fibroblast growth factor receptors 1–4 in human chronic tympanic membrane perforation. ORL J. Otorhinolaryngol. Relat. Spec. 2009, 71, 67–70. [Google Scholar] [CrossRef]
- O’Daniel, T.G.; Petitjean, M.; Jones, S.C.; Zogg, J.; Martinez, S.A.; Nolph, M.B.; Schultz, G.S. Epidermal growth factor binding and action on tympanic membranes. Ann. Otol. Rhinol. Laryngol. 1990, 99, 80–84. [Google Scholar]
- Koba, R.; Kawabata, I. Immunohistochemical study of transforming growth factor-α expression in normal and perforated tympanic membrane. Ann. Otol. Rhinol. Laryngol. 1995, 104, 793–797. [Google Scholar] [CrossRef]
- Mondain, M.; Ryan, A. Effect of basic fibroblast growth factor on normal tympanic membrane. Am. J. Otolaryngol. 1994, 15, 344–350. [Google Scholar] [CrossRef]
- Dignass, A.U.; Tsunekawa, S.; Podolsky, D.K. Fibroblast growth factors modulate intestinal epithelial cell growth and migration. Gastroenterology 1994, 106, 1254–1262. [Google Scholar] [CrossRef]
- Holland, E.C.; Varmus, H.E. Basic fibroblast growth factor induces cell migration and proliferation after glia-specific gene transfer in mice. Proc. Natl. Acad. Sci. USA 1998, 95, 1218–1223. [Google Scholar] [CrossRef]
- Hutley, L.; Shurety, W.; Newell, F.; McGeary, R.; Pelton, N.; Grant, J.; Herington, A.; Cameron, D.; Whitehead, J.; Prins, J. Fibroblast growth factor 1: A key regulator of human adipogenesis. Diabetes 2004, 53, 3097–3106. [Google Scholar] [CrossRef]
- Farooq, M.; Khan, A.W.; Kim, M.S.; Choi, S. The Role of Fibroblast Growth Factor (FGF) Signaling in Tissue Repair and Regeneration. Cells 2021, 10, 3242. [Google Scholar] [CrossRef]
- Prudovsky, I. Cellular Mechanisms of FGF-Stimulated Tissue Repair. Cells 2021, 10, 1830. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.; Shu, E.; Lee, D.J.; Jung, D.H.; Quesnel, A.M.; Stankovic, K.M.; Abdul-Aziz, D.E.; Bay, C.P.; Quinkert, A.; Welling, D.B. Topical fibroblast growth factor-2 for treatment of chronic tympanic membrane perforations. Laryngoscope Investig. Otolaryngol. 2020, 5, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Wang, Y. Evaluation of the optimum time for direct application of fibroblast growth factor to human traumatic tympanic membrane perforations. Growth Factors 2015, 33, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Strohm, M. Trauma of the middle ear. Clinical findings, postmortem observations and results of experimental studies. Adv. Otorhinolaryngol. 1986, 35, 1–254. [Google Scholar] [PubMed]
- Chen, G.; Gulbranson, D.R.; Yu, P.; Hou, Z.; Thomson, J.A. Thermal stability of fibroblast growth factor protein is a determinant factor in regulating self-renewal, differentiation, and reprogramming in human pluripotent stem cells. Stem Cells 2012, 30, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Koledova, Z.; Sumbal, J.; Rabata, A.; de La Bourdonnaye, G.; Chaloupkova, R.; Hrdlickova, B.; Damborsky, J.; Stepankova, V. Fibroblast Growth Factor 2 Protein Stability Provides Decreased Dependence on Heparin for Induction of FGFR Signaling and Alters ERK Signaling Dynamics. Front. Cell Dev. Biol. 2019, 7, 331. [Google Scholar] [CrossRef]
- Kanemaru, S.I.; Umeda, H.; Kitani, Y.; Nakamura, T.; Hirano, S.; Ito, J. Regenerative treatment for tympanic membrane perforation. Otol. Neurotol. 2011, 32, 1218–1223. [Google Scholar] [CrossRef]
- Acharya, A.N.; Coates, H.; Tavora-Vièira, D.; Rajan, G.P. A pilot study investigating basic fibroblast growth factor for the repair of chronic tympanic membrane perforations in pediatric patients. Int. J. Pediatr. Otorhinolaryngol. 2015, 79, 332–335. [Google Scholar] [CrossRef]
- Hakuba, N.; Taniguchi, M.; Shimizu, Y.; Sugimoto, A.; Shinomori, Y.; Gyo, K. A new method for closing tympanic membrane perforations using basic fibroblast growth factor. Laryngoscope 2003, 113, 1352–1355. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, Y.; Yu, G. Effects of basic fibroblast growth factor dose on traumatic tympanic membrane perforation. Growth Factors 2014, 32, 150–154. [Google Scholar] [CrossRef]
- Lou, Z.; Yang, J.; Tang, Y.; Xiao, J. Risk factors affecting human traumatic tympanic membrane perforation regeneration therapy using fibroblast growth factor-2. Growth Factors 2015, 33, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.C.; Lou, Z.H. The short- and long-term adverse effects of FGF-2 on tympanic membrane perforations. Acta Otorhinolaryngol. Ital. 2018, 38, 264–272. [Google Scholar]
- Benington, L.R.; Rajan, G.; Locher, C.; Lim, L.Y. Stabilisation of Recombinant Human Basic Fibroblast Growth Factor (FGF-2) against Stressors Encountered in Medicinal Product Processing and Evaluation. Pharmaceutics 2021, 13, 1762. [Google Scholar] [CrossRef] [PubMed]
- Shah, D. Effect of Various Additives on the Stability of Basic Fibroblast Growth Factor and Development of an Intradermal Injectable Formulation; University of Missouri: Kansas City, MO, USA, 1998. [Google Scholar]
- Tomoko, K.; Kenji, K.; Koichi, K. Quantitative colorimetric assay for basic fibroblast growth factor using bovine endothelial cells and heparin. J. Pharmacol. Toxicol. Methods 1992, 28, 9–14. [Google Scholar] [CrossRef]
- Bryant, J.C. Methylcellulose effect on cell proliferation and glucose utilization in chemically defined medium in large stationary cultures. Biotechnol. Bioeng. 1969, 11, 155–179. [Google Scholar] [CrossRef] [PubMed]
- Hagglund, B.; Sandberg, G. Effect of L-alanine and some other amino acids on thymocyte proliferation in vivo. Immunobiology 1993, 188, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Francis, G.L. Albumin and mammalian cell culture: Implications for biotechnology applications. Cytotechnology 2010, 62, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kanda, S.; Naba, A.; Miyata, Y. Inhibition of endothelial cell chemotaxis toward FGF-2 by gefitinib associates with downregulation of Fes activity. Int. J. Oncol. 2009, 35, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Levy, Y.; Hershfield, M.S.; Fernandez-Mejia, C.; Polmar, S.H.; Scudiery, D.; Berger, M.; Sorensen, R.U. Adenosine deaminase deficiency with late onset of recurrent infections: Response to treatment with polyethylene glycol-modified adenosine deaminase. J. Pediatr. 1988, 113, 312–317. [Google Scholar] [CrossRef]
- Ringsdorf, H. Structure and properties of pharmacologically active polymers. J. Polym. Sci. Polym. Symp. 1975, 51, 135–153. [Google Scholar] [CrossRef]
- Chen, H.C. Boyden chamber assay. Methods Mol. Biol. 2005, 294, 15–22. [Google Scholar]
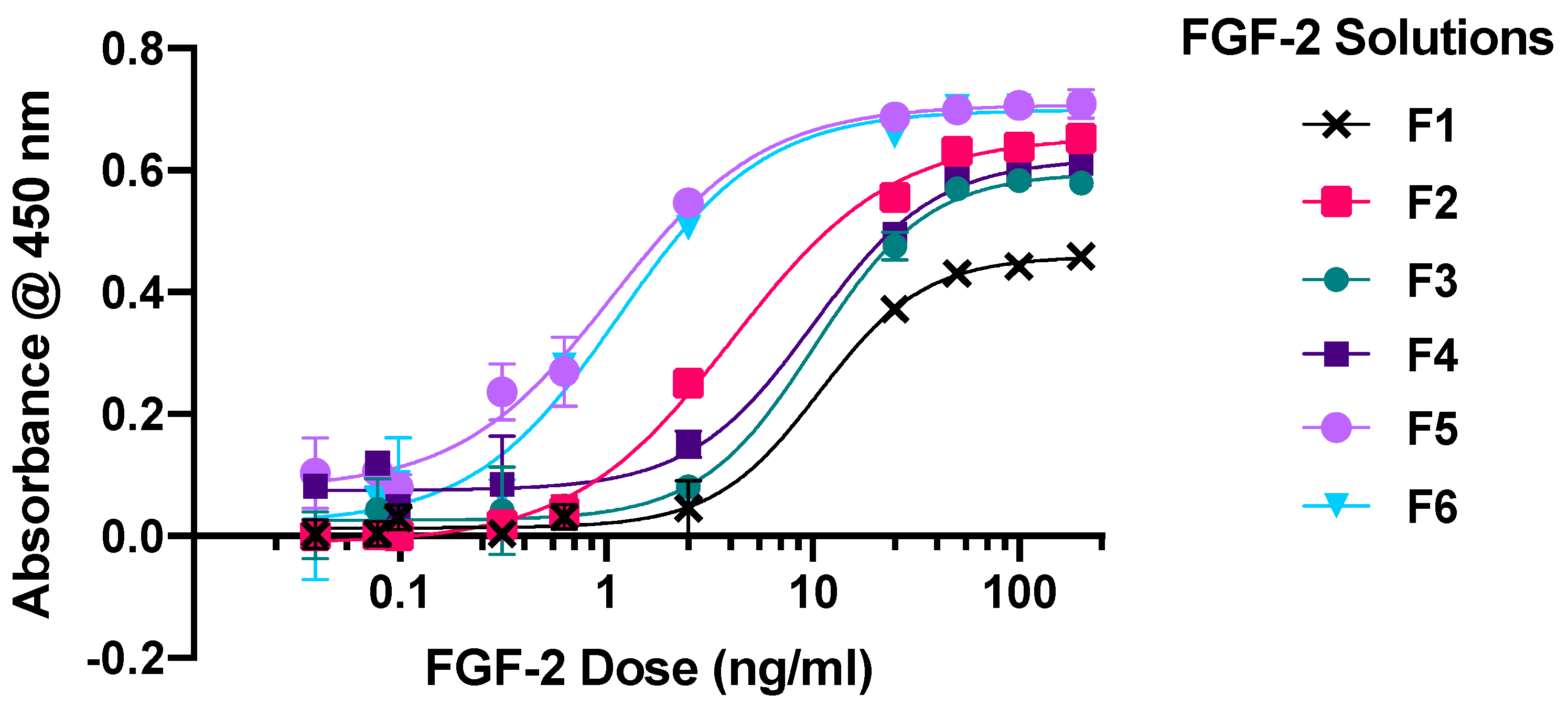
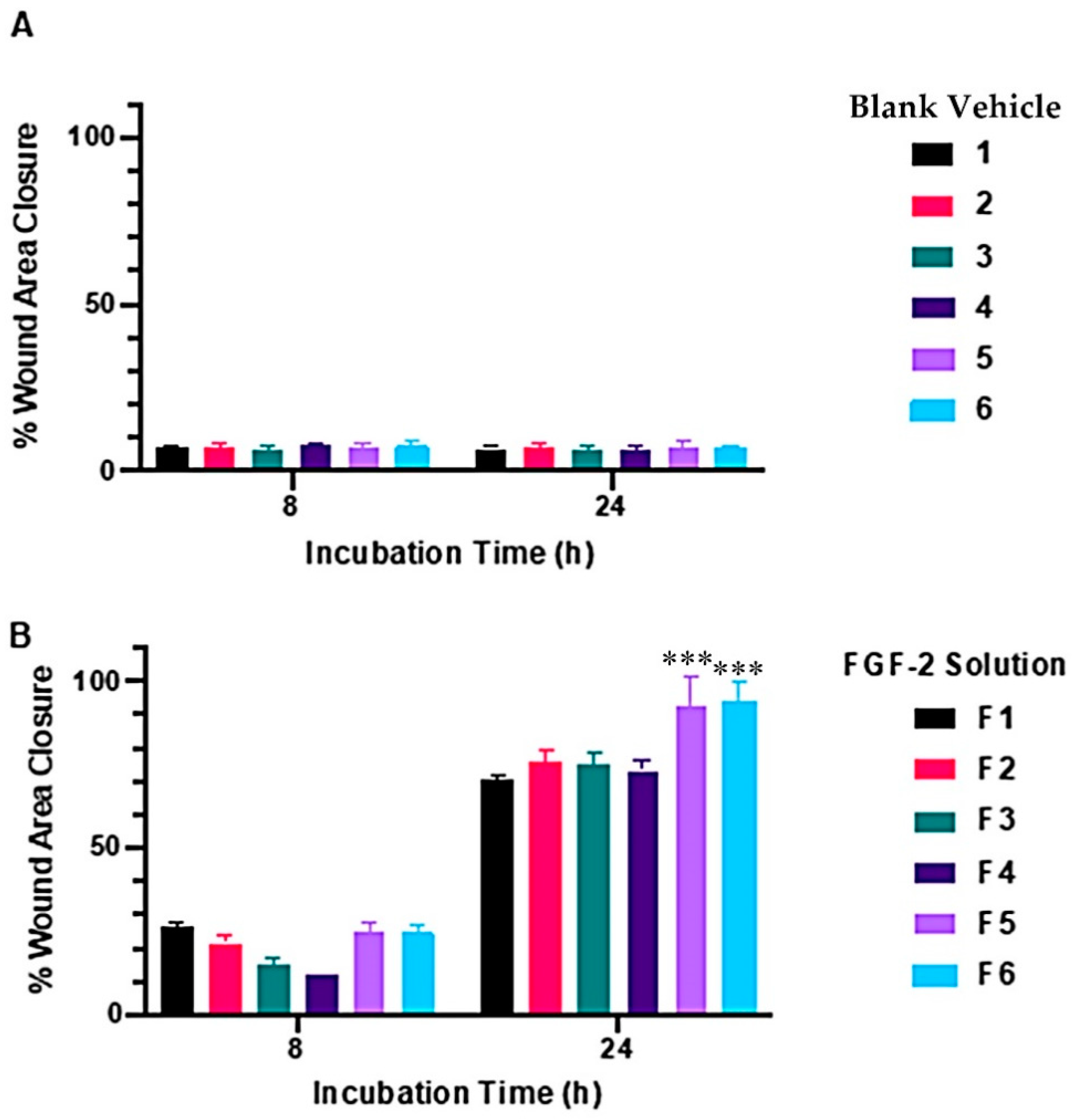
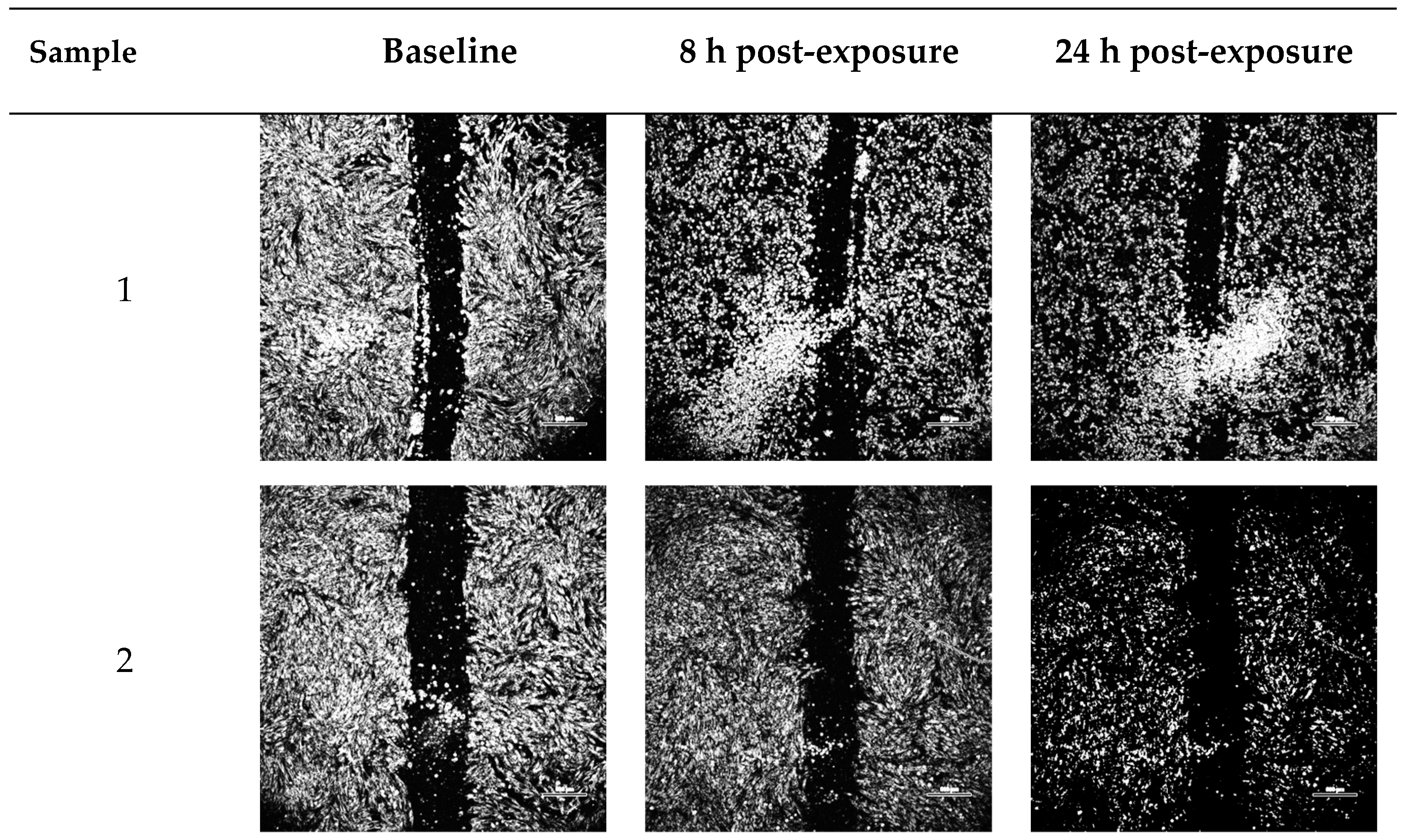
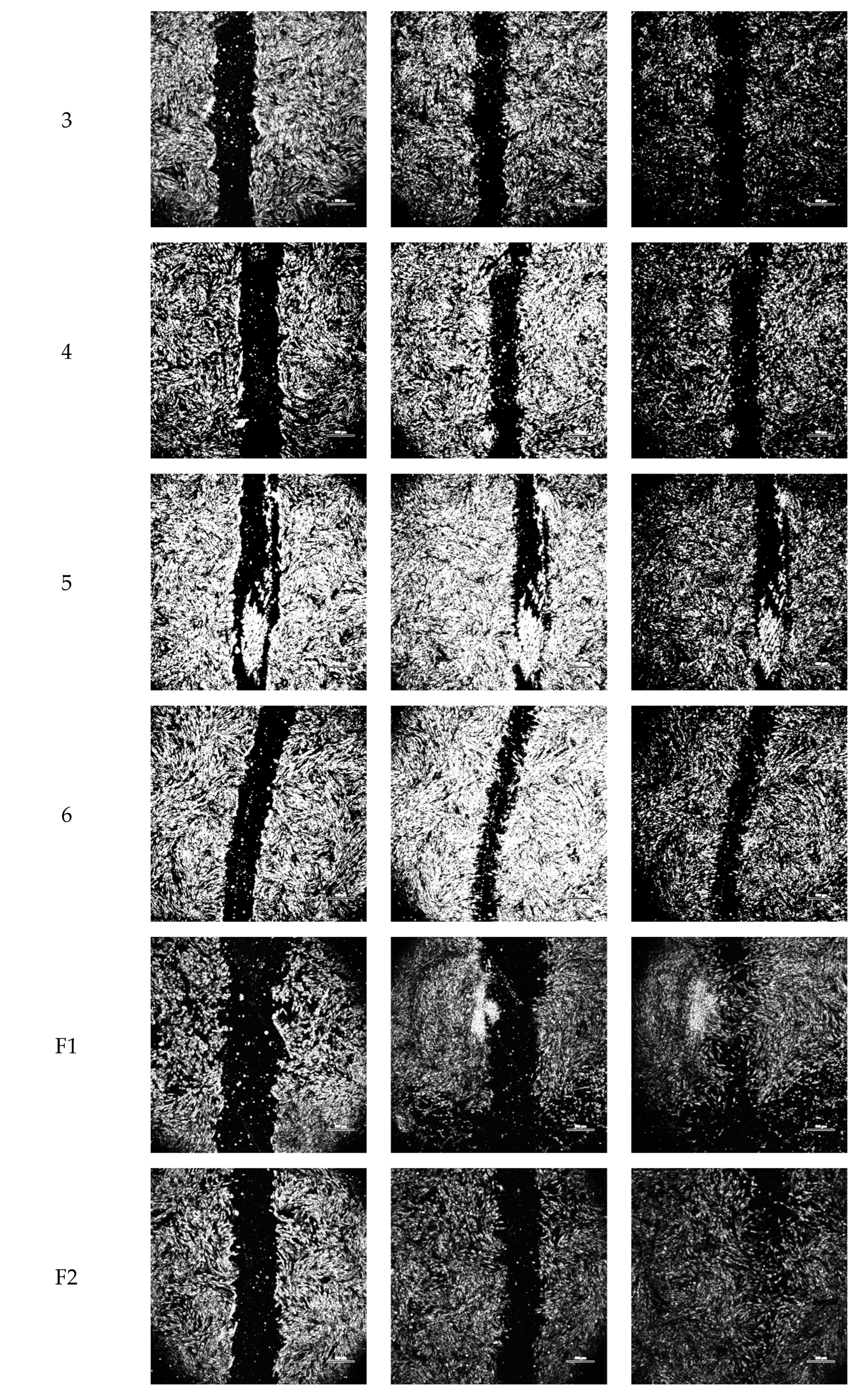
| Vehicle Constituents | Notation for Blank Vehicle | Notation for Solutions Containing 1600 ng/mL Bioactive FGF-2 in Vehicle |
|---|---|---|
| Water (control) | 1 | F1 |
| Methylcellulose (MC; 0.05% w/v) in water | 2 | F2 |
| Alanine (20 mM) in water | 3 | F3 |
| Human serum albumin (HSA; 1 mg/mL) in water | 4 | F4 |
| MC (0.05% w/v) and alanine (20 mM) in water | 5 | F5 |
| MC (0.05% w/v) and HSA (1 mg/mL) in water | 6 | F6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benington, L.; Mo, J.; Li, M.; Rajan, G.; Locher, C.; Lim, L.Y. In Vitro Assessment of Wound-Healing Efficacy of Stabilized Basic Fibroblast Growth Factor (FGF-2) Solutions. Pharmaceuticals 2024, 17, 247. https://doi.org/10.3390/ph17020247
Benington L, Mo J, Li M, Rajan G, Locher C, Lim LY. In Vitro Assessment of Wound-Healing Efficacy of Stabilized Basic Fibroblast Growth Factor (FGF-2) Solutions. Pharmaceuticals. 2024; 17(2):247. https://doi.org/10.3390/ph17020247
Chicago/Turabian StyleBenington, Leah, Jingxin Mo, Mingxin Li, Gunesh Rajan, Cornelia Locher, and Lee Yong Lim. 2024. "In Vitro Assessment of Wound-Healing Efficacy of Stabilized Basic Fibroblast Growth Factor (FGF-2) Solutions" Pharmaceuticals 17, no. 2: 247. https://doi.org/10.3390/ph17020247
APA StyleBenington, L., Mo, J., Li, M., Rajan, G., Locher, C., & Lim, L. Y. (2024). In Vitro Assessment of Wound-Healing Efficacy of Stabilized Basic Fibroblast Growth Factor (FGF-2) Solutions. Pharmaceuticals, 17(2), 247. https://doi.org/10.3390/ph17020247







