Anticancer Potential of Flavonoids: An Overview with an Emphasis on Tangeretin
Abstract
1. Introduction
2. Polyphenols
3. Structure and Classification of Flavonoids
4. Antitumor Activity of Flavonoids
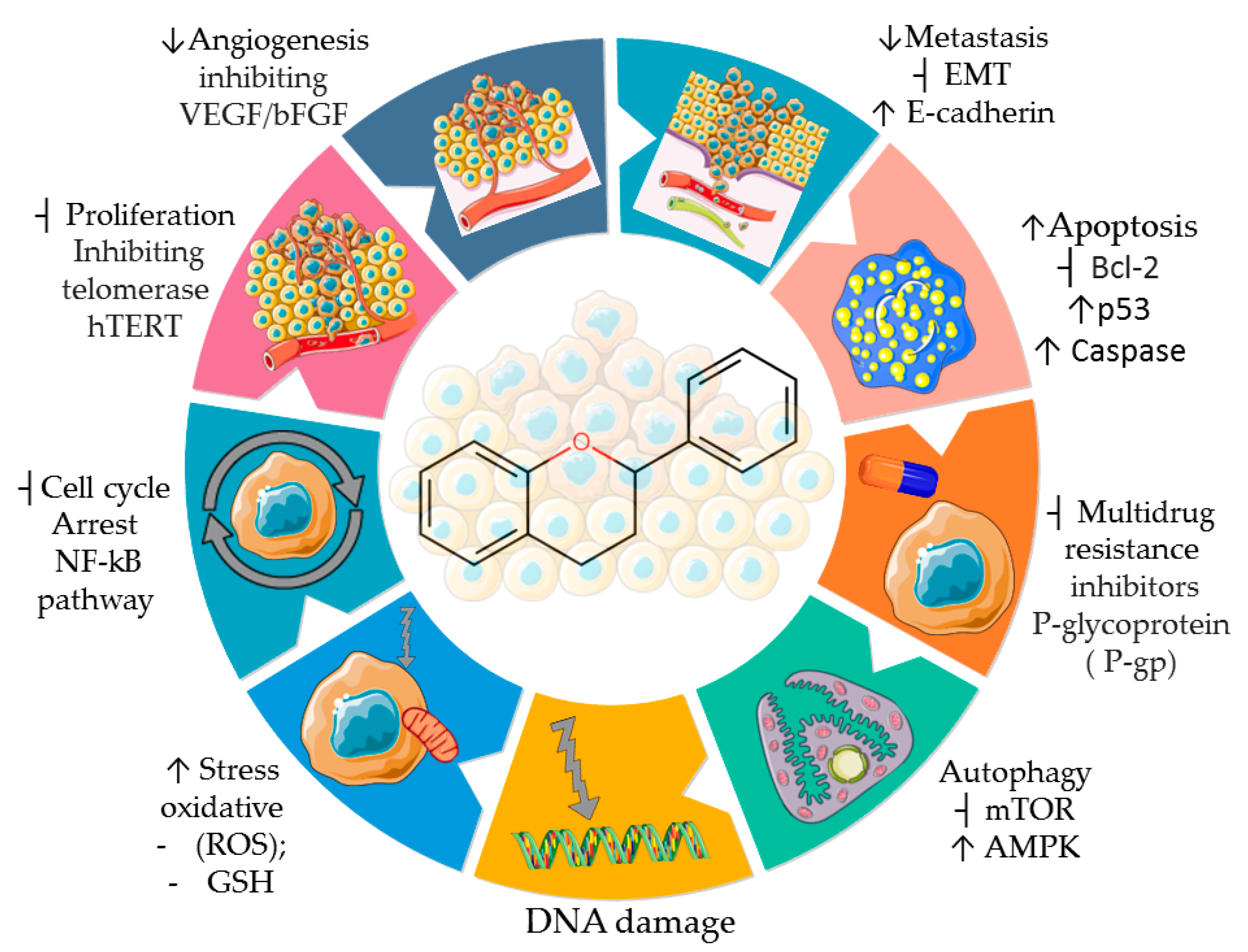
5. Antineoplastic Activity of Tangeretin
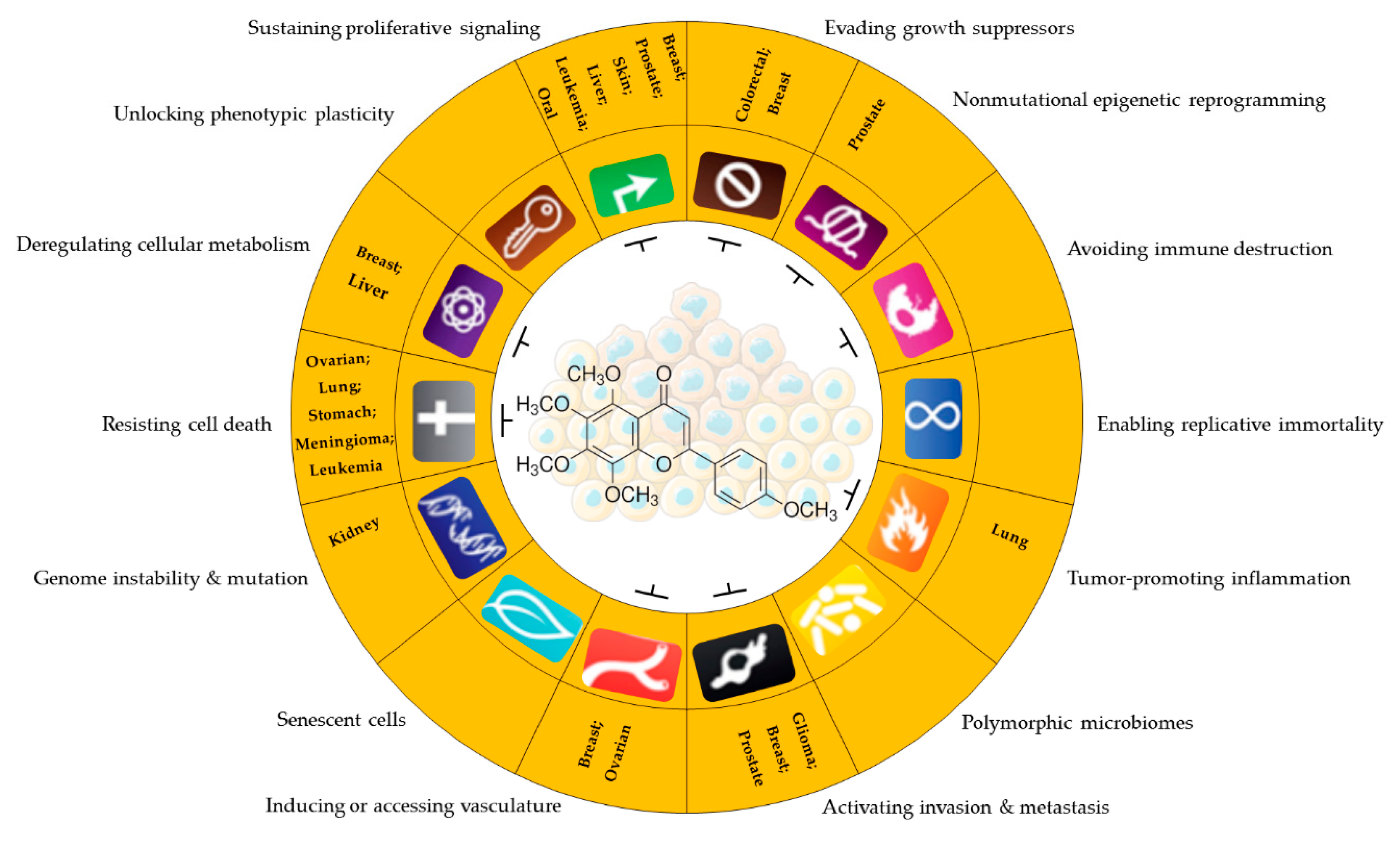
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Biological Hallmarks of Cancer. In Holland-Frei Cancer Medicine; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.A.; Sulaiman, A.A.; Balch, C.; Chauhan, H.; Alhadidi, Q.M.; Tiwari, A.K. Natural Polyphenols in Cancer Chemoresistance. Nutr. Cancer 2016, 68, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Jaitak, V. Natural Products as Multidrug Resistance Modulators in Cancer. Eur. J. Med. Chem. 2019, 176, 268–291. [Google Scholar] [CrossRef] [PubMed]
- Nirmaladevi, R. Epigenetic Alterations in Cancer. Front. Biosci. 2020, 25, 4847. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, R.; Gupta, S. Epigenetic Modifications in Cancer. Clin. Genet. 2012, 81, 303–311. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, H.; Gao, P. Metabolic Reprogramming and Epigenetic Modifications on the Path to Cancer. Protein Cell 2022, 13, 877–919. [Google Scholar] [CrossRef]
- Jones, P.A.; Baylin, S.B. The Epigenomics of Cancer. Cell 2007, 128, 683–692. [Google Scholar] [CrossRef]
- Arora, I.; Sharma, M.; Tollefsbol, T.O. Combinatorial Epigenetics Impact of Polyphenols and Phytochemicals in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2019, 20, 4567. [Google Scholar] [CrossRef]
- Rajendran, P.; Abdelsalam, S.A.; Renu, K.; Veeraraghavan, V.; Ben Ammar, R.; Ahmed, E.A. Polyphenols as Potent Epigenetics Agents for Cancer. Int. J. Mol. Sci. 2022, 23, 11712. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Mutha, R.E.; Tatiya, A.U.; Surana, S.J. Flavonoids as Natural Phenolic Compounds and Their Role in Therapeutics: An Overview. Future J. Pharm. Sci. 2021, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-Inflammatory Effects of Flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef] [PubMed]
- Sandu, M.; Bîrsă, L.M.; Bahrin, L.G. Flavonoids–Small Molecules, High Hopes. Acta Chem. Iasi 2017, 25, 6–23. [Google Scholar] [CrossRef][Green Version]
- Estrela, J.M.; Mena, S.; Obrador, E.; Benlloch, M.; Castellano, G.; Salvador, R.; Dellinger, R.W. Polyphenolic Phytochemicals in Cancer Prevention and Therapy: Bioavailability versus Bioefficacy. J. Med. Chem. 2017, 60, 9413–9436. [Google Scholar] [CrossRef] [PubMed]
- Afshari, K.; Haddadi, N.; Haj-Mirzaian, A.; Farzaei, M.H.; Rohani, M.M.; Akramian, F.; Naseri, R.; Sureda, A.; Ghanaatian, N.; Abdolghaffari, A.H. Natural Flavonoids for the Prevention of Colon Cancer: A Comprehensive Review of Preclinical and Clinical Studies. J. Cell. Physiol. 2019, 234, 21519–21546. [Google Scholar] [CrossRef]
- George, V.C.; Dellaire, G.; Rupasinghe, H.P.V. Plant Flavonoids in Cancer Chemoprevention: Role in Genome Stability. J. Nutr. Biochem. 2017, 45, 1–14. [Google Scholar] [CrossRef]
- Ross, J.A.; Kasum, C.M. DIETARY FLAVONOIDS: Bioavailability, Metabolic Effects, and Safety. Annu. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef]
- Kasprzak, M.M.; Erxleben, A.; Ochocki, J. Properties and Applications of Flavonoid Metal Complexes. RSC Adv. 2015, 5, 45853–45877. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016, 5, E47. [Google Scholar] [CrossRef] [PubMed]
- Rahnasto-Rilla, M.; Tyni, J.; Huovinen, M.; Jarho, E.; Kulikowicz, T.; Ravichandran, S.; Bohr, V.A.; Ferrucci, L.; Lahtela-Kakkonen, M.; Moaddel, R. Natural Polyphenols as Sirtuin 6 Modulators. Sci. Rep. 2018, 8, 4163. [Google Scholar] [CrossRef] [PubMed]
- Slámová, K.; Kapešová, J.; Valentová, K. “Sweet Flavonoids”: Glycosidase-Catalyzed Modifications. Int. J. Mol. Sci. 2018, 19, 2126. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.K.; Vishwavidyalaya, D.A.; Pradesh, M. Flavonoids Impact on Prevention and Treatment of Obesity and Related. Int. J. Pharm. Sci. Res. 2019, 10, 4420–4429. [Google Scholar] [CrossRef]
- Teles, Y.C.F.; Souza, M.S.R.; De Souza, M.d.F.V. Sulphated Flavonoids: Biosynthesis, Structures, and Biological Activities. Molecules 2018, 23, 480. [Google Scholar] [CrossRef] [PubMed]
- Veitch, N.C.; Grayer, R.J. Flavonoids and Their Glycosides, Including Anthocyanins. Nat. Prod. Rep. 2011, 28, 1626–1695. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Jakstas, V.; Savickas, A.; Bernatoniene, J. Flavonoids as Anticancer Agents. Nutrients 2020, 12, 457. [Google Scholar] [CrossRef]
- Wang, T.-y.; Li, Q.; Bi, K.-s. Bioactive Flavonoids in Medicinal Plants: Structure, Activity and Biological Fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Brodowska, K.M. European Journal of Biological Research Natural Flavonoids: Classification, Potential Role, and Application of Flavonoid Analogues. Eur. J. Biol. Res. 2017, 7, 108–123. [Google Scholar] [CrossRef]
- Khajuria, R.; Singh, S.; Bahl, A. General Introduction and Sources of Flavonoids. In Current Aspects of Flavonoids: Their Role in Cancer Treatment; Springer: Singapore, 2019; pp. 1–7. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, W.; Xie, M. Flavonoids: Recent Advances as Anticancer Drugs. Recent Pat. Anti-Cancer Drug Discov. 2010, 5, 152–164. [Google Scholar] [CrossRef]
- Raffa, D.; Maggio, B.; Raimondi, M.V.; Plescia, F.; Daidone, G. Recent Discoveries of Anticancer Flavonoids. Eur. J. Med. Chem. 2017, 142, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Symonowicz, M.; Kolanek, M. Flavonoids and Their Properties to Form Chelate Complexes. Biotechnol. Food Sci. 2012, 76, 35–41. [Google Scholar]
- Ahn-Jarvis, J.H.; Parihar, A.; Doseff, A.I. Dietary Flavonoids for Immunoregulation and Cancer: Food Design for Targeting Disease. Antioxidants 2019, 8, 202. [Google Scholar] [CrossRef] [PubMed]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and Gut Health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Sudhakaran, M.; Sardesai, S.; Doseff, A.I. Flavonoids: New Frontier for Immuno-Regulation and Breast Cancer Control. Antioxidants 2019, 8, 103. [Google Scholar] [CrossRef] [PubMed]
- Al Aboody, M.S.; Mickymaray, S. Anti-Fungal Efficacy and Mechanisms of Flavonoids. Antibiotics 2020, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Li, X.; He, L.; Zheng, Y.; Lu, H.; Li, J.; Zhong, L.; Tong, R.; Jiang, Z.; Shi, J.; et al. Antidiabetic Potential of Flavonoids from Traditional Chinese Medicine: A Review. Am. J. Chin. Med. 2019, 47, 933–957. [Google Scholar] [CrossRef] [PubMed]
- Pietta, P.G. Flavonoids as Antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Ren, W.; Qiao, Z.; Wang, H.; Zhu, L.; Zhang, L. Flavonoids: Promising Anticancer Agents. Med. Res. Rev. 2003, 23, 519–534. [Google Scholar] [CrossRef]
- Weston, L.A.; Mathesius, U. Flavonoids: Their Structure, Biosynthesis and Role in the Rhizosphere, Including Allelopathy. J. Chem. Ecol. 2013, 39, 283–297. [Google Scholar] [CrossRef]
- Sharma, N.; Dobhal, M.; Joshi, Y.; Chahar, M. Flavonoids: A Versatile Source of Anticancer Drugs. Pharmacogn. Rev. 2011, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vizcaino, F.; Fraga, C.G. Research Trends in Flavonoids and Health. Arch. Biochem. Biophys. 2018, 646, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J.J.; Gaforio, J.J. Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants 2019, 8, 137. [Google Scholar] [CrossRef] [PubMed]
- Rudzińska, A.; Juchaniuk, P.; Oberda, J.; Wiśniewska, J.; Wojdan, W.; Szklener, K.; Mańdziuk, S. Phytochemicals in Cancer Treatment and Cancer Prevention—Review on Epidemiological Data and Clinical Trials. Nutrients 2023, 15, 1896. [Google Scholar] [CrossRef] [PubMed]
- Anantharaju, P.G.; Gowda, P.C.; Vimalambike, M.G.; Madhunapantula, S.V. An Overview on the Role of Dietary Phenolics for the Treatment of Cancers. Nutr. J. 2016, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- Mohana, S.; Ganesan, M.; Rajendra Prasad, N.; Ananthakrishnan, D.; Velmurugan, D. Flavonoids Modulate Multidrug Resistance through Wnt Signaling in P-Glycoprotein Overexpressing Cell Lines. BMC Cancer 2018, 18, 1168. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, A.; Poursoleiman, F.; Biregani, A.N.; Esmailzadeh, A. Flavonoids Target Different Molecules of Autophagic and Metastatic Pathways in Cancer Cells. Cancer Cell Int. 2023, 23, 114. [Google Scholar] [CrossRef]
- De Sousa Silva, G.V.; Lopes, A.L.V.F.G.; Viali, I.C.; Lima, L.Z.M.; Bizuti, M.R.; Haag, F.B.; Tavares de Resende e Silva, D. Therapeutic Properties of Flavonoids in Treatment of Cancer through Autophagic Modulation: A Systematic Review. Chin. J. Integr. Med. 2023, 29, 268–279. [Google Scholar] [CrossRef]
- Parekh, N.; Garg, A.; Choudhary, R.; Gupta, M.; Kaur, G.; Ramniwas, S.; Shahwan, M.; Tuli, H.S.; Sethi, G. The Role of Natural Flavonoids as Telomerase Inhibitors in Suppressing Cancer Growth. Pharmaceuticals 2023, 16, 605. [Google Scholar] [CrossRef]
- Rahman, N.; Khan, H.; Zia, A.; Khan, A.; Fakhri, S.; Aschner, M.; Gul, K.; Saso, L. Bcl-2 Modulation in P53 Signaling Pathway by Flavonoids: A Potential Strategy towards the Treatment of Cancer. Int. J. Mol. Sci. 2021, 22, 11315. [Google Scholar] [CrossRef]
- Kim, M.H. Flavonoids Inhibit VEGF/BFGF-Induced Angiogenesis in Vitro by Inhibiting the Matrix-Degrading Proteases. J. Cell. Biochem. 2003, 89, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.M.; Šupolíková, L.; Molčanová, L.; Šmejkal, K.; Bednar, D.; Slaninová, I. Screening of Natural Compounds as P-Glycoprotein Inhibitors against Multidrug Resistance. Biomedicines 2021, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Ding, K.; Qiao, Y.; Zhang, L.; Zheng, L.; Pan, T.; Zhang, L. Flavonoids Regulate Inflammation and Oxidative Stress in Cancer. Molecules 2020, 25, 5628. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Yang, J.; Xie, Y. Improvement Strategies for the Oral Bioavailability of Poorly Water-Soluble Flavonoids: An Overview. Int. J. Pharm. 2019, 570, 118642. [Google Scholar] [CrossRef] [PubMed]
- Raj, S.; Khurana, S.; Choudhari, R.; Kesari, K.K.; Kamal, M.A.; Garg, N.; Ruokolainen, J.; Das, B.C.; Kumar, D. Specific Targeting Cancer Cells with Nanoparticles and Drug Delivery in Cancer Therapy. Semin. Cancer Biol. 2021, 69, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced Targeted Therapies in Cancer: Drug Nanocarriers, the Future of Chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [PubMed]
- Liskova, A.; Samec, M.; Koklesova, L.; Brockmueller, A.; Zhai, K.; Abdellatif, B.; Siddiqui, M.; Biringer, K.; Kudela, E.; Pec, M.; et al. Flavonoids as an Effective Sensitizer for Anti-Cancer Therapy: Insights into Multi-Faceted Mechanisms and Applicability towards Individualized Patient Profiles. EPMA J. 2021, 12, 155–176. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.; Costa, D.; Sousa, Â. Flavonoids-Based Delivery Systems towards Cancer Therapies. Bioengineering 2022, 9, 197. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Martorell, M.; Valdes, S.E.; Belwal, T.; Tejada, S.; Sureda, A.; Kamal, M.A. Flavonoids Nanoparticles in Cancer: Treatment, Prevention and Clinical Prospects. Semin. Cancer Biol. 2019, 69, 200–211. [Google Scholar] [CrossRef]
- Wei, Q.Y.; He, K.M.; Chen, J.L.; Xu, Y.M.; Lau, A.T.Y. Phytofabrication of Nanoparticles as Novel Drugs for Anticancer Applications. Molecules 2019, 24, 4246. [Google Scholar] [CrossRef]
- Siddiqui, I.A.; Bharali, D.J.; Nihal, M.; Adhami, V.M.; Khan, N.; Chamcheu, J.C.; Khan, M.I.; Shabana, S.; Mousa, S.A.; Mukhtar, H. Excellent Anti-Proliferative and pro-Apoptotic Effects of (−)-Epigallocatechin-3-Gallate Encapsulated in Chitosan Nanoparticles on Human Melanoma Cell Growth Both in Vitro and in Vivo. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1619–1626. [Google Scholar] [CrossRef] [PubMed]
- Nihal, M.; Ahmad, N.; Mukhtar, H.; Wood, G.S. Anti-Proliferative and Proapoptotic Effects of (−)-Epigallocatechin-3-Gallate on Human Melanoma: Possible Implications for the Chemoprevention of Melanoma. Int. J. Cancer 2005, 114, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.A.; Adhami, V.M.; Bharali, D.J.; Hafeez, B.B.; Asim, M.; Khwaja, S.I.; Ahmad, N.; Cui, H.; Mousa, S.A.; Mukhtar, H. Introducing Nanochemoprevention as a Novel Approach for Cancer Control: Proof of Principle with Green Tea Polyphenol Epigallocatechin-3-Gallate. Cancer Res. 2009, 69, 1712–1716. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Hsieh, C.; Tsai, S.; Wang, C.-Y.; Wang, C. Anticancer Effects of Epigallocatechin-3-Gallate Nanoemulsion on Lung Cancer Cells through the Activation of AMP-Activated Protein Kinase Signaling Pathway. Sci. Rep. 2020, 10, 5163. [Google Scholar] [CrossRef] [PubMed]
- Trefts, E.; Shaw, R.J. AMPK: Restoring Metabolic Homeostasis over Space and Time. Mol. Cell 2021, 81, 3677–3690. [Google Scholar] [CrossRef] [PubMed]
- Thirupathi, A.; Chang, Y.Z. Role of AMPK and Its Molecular Intermediates in Subjugating Cancer Survival Mechanism. Life Sci. 2019, 227, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Gorrini, C.; Harris, I.S.; Mak, T.W. Modulation of Oxidative Stress as an Anticancer Strategy. Nat. Rev. Drug Discov. 2013, 12, 931–947. [Google Scholar] [CrossRef]
- Huang, M.Z.; Li, J.Y. Physiological Regulation of Reactive Oxygen Species in Organisms Based on Their Physicochemical Properties. Acta Physiol. 2020, 228, e13351. [Google Scholar] [CrossRef]
- Marengo, B.; Nitti, M.; Furfaro, A.L.; Colla, R.; De Ciucis, C.; Marinari, U.M.; Pronzato, M.A.; Traverso, N.; Domenicotti, C. Redox Homeostasis and Cellular Antioxidant Systems: Crucial Players in Cancer Growth and Therapy. Oxid. Med. Cell. Longev. 2016, 2016, 6235641. [Google Scholar] [CrossRef]
- Lv, H.; Zhen, C.; Liu, J.; Yang, P.; Hu, L. Unraveling the Potential Role of Glutathione in Multiple Forms of Cell Death in Cancer Therapy. Oxid. Med. Cell. Longev. 2019, 2019, 3150145. [Google Scholar] [CrossRef]
- Zaidieh, T.; Smith, J.R.; Ball, K.E.; An, Q. ROS as a Novel Indicator to Predict Anticancer Drug Efficacy. BMC Cancer 2019, 19, 1224. [Google Scholar] [CrossRef] [PubMed]
- Perillo, B.; Di Donato, M.; Pezone, A.; Di Zazzo, E.; Castoria, G.; Migliaccio, A. ROS in Cancer Therapy: The Bright Side of the Moon. Exp. Mol. Med. 2020, 52, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Liou, G.-Y.; Storz, P. Reactive Oxygen Species in Cancer. Free Radic. Res. 2010, 44, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and Prooxidant Behavior of Flavonoids: Structure-Activity Relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Havsteen, B.H. The Biochemistry and Medical Significance of the Flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef] [PubMed]
- Tavsan, Z.; Kayali, H.A. Flavonoids Showed Anticancer Effects on the Ovarian Cancer Cells: Involvement of Reactive Oxygen Species, Apoptosis, Cell Cycle and Invasion. Biomed. Pharmacother. 2019, 116, 109004. [Google Scholar] [CrossRef] [PubMed]
- Biswas, P.; Dey, D.; Biswas, P.K.; Rahaman, T.I.; Saha, S.; Parvez, A.; Khan, D.A.; Lily, N.J.; Saha, K.; Sohel, M.; et al. A Comprehensive Analysis and Anti-Cancer Activities of Quercetin in ROS-Mediated Cancer and Cancer Stem Cells. Int. J. Mol. Sci. 2022, 23, 11746. [Google Scholar] [CrossRef] [PubMed]
- Ai, L.; Kim, W.J.; Demircan, B.; Dyer, L.M.; Bray, K.J.; Skehan, R.R.; Massoll, N.A.; Brown, K.D. The Transglutaminase 2 Gene (TGM2), a Potential Molecular Marker for Chemotherapeutic Drug Sensitivity, Is Epigenetically Silenced in Breast Cancer. Carcinogenesis 2008, 29, 510–518. [Google Scholar] [CrossRef]
- Eckert, R.L. Transglutaminase 2 Takes Center Stage as a Cancer Cell Survival Factor and Therapy Target. Mol. Carcinog. 2019, 58, 837–853. [Google Scholar] [CrossRef]
- Mehta, K.; Han, A. Tissue Transglutaminase (TG2)-Induced Inflammation in Initiation, Progression, and Pathogenesis of Pancreatic Cancer. Cancers 2011, 3, 897–912. [Google Scholar] [CrossRef]
- Wang, F.; Wang, L.; Qu, C.; Chen, L.; Geng, Y.; Cheng, C.; Yu, S.; Wang, D.; Yang, L.; Meng, Z.; et al. Kaempferol Induces ROS-Dependent Apoptosis in Pancreatic Cancer Cells via TGM2-Mediated Akt/MTOR Signaling. BMC Cancer 2021, 21, 396. [Google Scholar] [CrossRef]
- Reyes-Farias, M.; Carrasco-Pozo, C. The Anti-Cancer Effect of Quercetin: Molecular Implications in Cancer Metabolism. Int. J. Mol. Sci. 2019, 20, 3177. [Google Scholar] [CrossRef] [PubMed]
- Ortega, A.L.; Mena, S.; Estrela, J.M. Glutathione in Cancer Cell Death. Cancers 2011, 3, 1285–1310. [Google Scholar] [CrossRef] [PubMed]
- Pakfetrat, A.; Dalirsani, Z.; Hashemy, S.I.; Ghazi, A.; Mostaan, L.V.; Anvari, K.; Pour, A.M. Evaluation of Serum Levels of Oxidized and Reduced Glutathione and Total Antioxidant Capacity in Patients with Head and Neck Squamous Cell Carcinoma. J. Cancer Res. Ther. 2018, 14, 428–431. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Simon, M.C. Glutathione Metabolism in Cancer Progression and Treatment Resistance. J. Cell Biol. 2018, 217, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.; Jung, E.; Noh, J.; Hyun, H.; Seon, S.; Hong, S.; Kim, D.; Lee, D. Glutathione-Depleting Pro-Oxidant as a Selective Anticancer Therapeutic Agent. ACS Omega 2019, 4, 10070–10077. [Google Scholar] [CrossRef]
- Traverso, N.; Ricciarelli, R.; Nitti, M.; Marengo, B.; Furfaro, A.L.; Pronzato, M.A.; Marinari, U.M.; Domenicotti, C. Role of Glutathione in Cancer Progression and Chemoresistance. Oxid. Med. Cell. Longev. 2013, 2013, 972913. [Google Scholar] [CrossRef]
- Desideri, E.; Ciccarone, F.; Ciriolo, M.R. Targeting Glutathione Metabolism: Partner in Crime in Anticancer Therapy. Nutrients 2019, 11, 1926. [Google Scholar] [CrossRef]
- Liang, F.; Fang, Y.; Cao, W.; Zhang, Z.; Pan, S.; Xu, X. Attenuation of tert-Butyl Hydroperoxide (t-BHP)-Induced Oxidative Damage in HepG2 Cells by Tangeretin: Relevance of the Nrf2-ARE and MAPK Signaling Pathways. J. Agric. Food Chem. 2018, 66, 6317–6325. [Google Scholar] [CrossRef]
- Omar, H.A.; Mohamed, W.R.; Arab, H.H.; Arafa, E.S.A. Tangeretin Alleviates Cisplatin-Induced Acute Hepatic Injury in Rats: Targeting MAPKs and Apoptosis. PLoS ONE 2016, 11, e0151649. [Google Scholar] [CrossRef]
- Portela, A.; Esteller, M. Epigenetic Modifications and Human Disease. Nat. Biotechnol. 2010, 28, 1057–1068. [Google Scholar] [CrossRef]
- Hernando-Herraez, I.; Garcia-Perez, R.; Sharp, A.J.; Marques-Bonet, T. DNA Methylation: Insights into Human Evolution. PLoS Genet. 2015, 11, e1005661. [Google Scholar] [CrossRef] [PubMed]
- Parry, L.; Clarke, A.R. The Roles of the Methyl-CpG Binding Proteins in Cancer. Genes Cancer 2011, 2, 618–630. [Google Scholar] [CrossRef]
- Bergman, Y.; Cedar, H. DNA Methylation Dynamics in Health and Disease. Nat. Struct. Mol. Biol. 2013, 20, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Ning, B.; Li, W.; Zhao, W.; Wang, R. Targeting Epigenetic Regulations in Cancer. Acta Biochim. Biophys. Sin. 2015, 48, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Stefansson, O.A.; Moran, S.; Gomez, A.; Sayols, S.; Arribas-Jorba, C.; Sandoval, J.; Hilmarsdottir, H.; Olafsdottir, E.; Tryggvadottir, L.; Jonasson, J.G.; et al. A DNA Methylation-Based Definition of Biologically Distinct Breast Cancer Subtypes. Mol. Oncol. 2015, 9, 555–568. [Google Scholar] [CrossRef] [PubMed]
- Zakhari, S. Alcohol Metabolism and Epigenetics Changes. Alcohol Res. 2013, 35, 6–16. [Google Scholar] [PubMed]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and Other Polyphenols Act as Epigenetic Modifiers in Breast Cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Cadet, J.; McCoy, M.; Jayanthi, S. Epigenetics and Addiction. Clin. Pharmacol. Ther. 2016, 99, 502–511. [Google Scholar] [CrossRef]
- Fatima, N.; Baqri, S.S.R.; Bhattacharya, A.; Koney, N.K.K.; Husain, K.; Abbas, A.; Ansari, R.A. Role of Flavonoids as Epigenetic Modulators in Cancer Prevention and Therapy. Front. Genet. 2021, 12, 758733. [Google Scholar] [CrossRef]
- Goldar, S.; Khaniani, M.S.; Derakhshan, S.M.; Baradaran, B. Molecular Mechanisms of Apoptosis and Roles in Cancer Development and Treatment. Asian Pac. J. Cancer Prev. 2015, 16, 2129–2144. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.; Watari, H.; Abualmaaty, A.; Ohba, Y.; Sakuragi, N. Apoptosis and Molecular Targeting Therapy in Cancer. Biomed Res. Int. 2014, 2014, 150845. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, B.A.; El-Deiry, W.S. Targeting Apoptosis in Cancer Therapy. Nat. Rev. Clin. Oncol. 2020, 17, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Tewary, P.; Gunatilaka, A.A.L.; Sayers, T.J. Using Natural Products to Promote Caspase-8-Dependent Cancer Cell Death. Cancer Immunol. Immunother. 2017, 66, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Kong, A.N.T.; Yu, R.; Chen, C.; Mandlekar, S.; Primiano, T. Signal Transduction Events Elicited by Natural Products: Role of MAPK and Caspase Pathways in Homeostatic Response and Induction of Apoptosis. Arch. Pharm. Res. 2000, 23, 1–16. [Google Scholar] [CrossRef]
- McIlwain, D.R.; Berger, T.; Mak, T.W. Caspase Functions in Cell Death and Disease. Cold Spring Harb. Perspect. Biol. 2015, 7, a026716. [Google Scholar] [CrossRef] [PubMed]
- Ghavami, S.; Hashemi, M.; Ande, S.R.; Yeganeh, B.; Xiao, W.; Eshraghi, M.; Bus, C.J.; Kadkhoda, K.; Wiechec, E.; Halayko, A.J.; et al. Apoptosis and Cancer: Mutations within Caspase Genes. J. Med. Genet. 2009, 46, 497–510. [Google Scholar] [CrossRef]
- Xu, D.C.; Arthurton, L.; Baena-Lopez, L.A. Learning on the Fly: The Interplay between Caspases and Cancer. Biomed Res. Int. 2018, 2018, 5473180. [Google Scholar] [CrossRef]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef]
- Warren, C.F.A.; Wong-Brown, M.W.; Bowden, N.A. BCL-2 Family Isoforms in Apoptosis and Cancer. Cell Death Dis. 2019, 10, 177. [Google Scholar] [CrossRef]
- Lomonosova, E.; Chinnadurai, G. BH3-Only Proteins in Apoptosis and beyond: An Overview. Oncogene 2008, 27, S2–S19. [Google Scholar] [CrossRef] [PubMed]
- Elango, R.; Athinarayanan, J.; Subbarayan, V.P.; Lei, D.K.Y.; Alshatwi, A.A. Hesperetin Induces an Apoptosis-Triggered Extrinsic Pathway and a P53-Independent Pathway in Human Lung Cancer H522 Cells. J. Asian Nat. Prod. Res. 2018, 20, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Dahlawi, H. Effect of Malvidin on Induction of Apoptosis and Inhibition of Cell Proliferation on Myeloid and Lymphoid Leukemia. Sch. J. Appl. Med. Sci. 2022, 10, 150–156. [Google Scholar] [CrossRef]
- Wang, Y.X.; Cai, H.; Jiang, G.; Zhou, T.B.; Wu, H. Silibinin Inhibits Proliferation, Induces Apoptosis and Causes Cell Cycle Arrest in Human Gastric Cancer MGC803 Cells via STAT3 Pathway Inhibition. Asian Pac. J. Cancer Prev. 2014, 15, 6791–6798. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, D.; Hao, Y.; Liu, Q.; Wu, Y.; Liu, X.; Luo, J.; Zhou, T.; Sun, B.; Luo, X.; et al. Cyanidin Curtails Renal Cell Carcinoma Tumorigenesis. Cell. Physiol. Biochem. 2018, 46, 2517–2531. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Yun, S.M.; Song, M.Y.; Jung, K.; Kim, E.H. Cyanidin Chloride Induces Apoptosis by Inhibiting NF-κB Signaling through Activation of Nrf2 in Colorectal Cancer Cells. Antioxidants 2020, 9, 285. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.; Zhu, Y.; Han, B.; Peng, J.; Deng, X.; Chen, W.; Du, J.; Ou, Y.; Peng, X.; Yu, X. Delphinidin Induces Cell Cycle Arrest and Apoptosis in HER-2 Positive Breast Cancer Cell Lines by Regulating the NF-κB and MAPK Signaling Pathways. Oncol. Lett. 2021, 22, 832. [Google Scholar] [CrossRef] [PubMed]
- Kim, U.; Kim, C.-Y.; Lee, J.M.; Oh, H.; Ryu, B.; Kim, J.; Park, J.-H. Phloretin Inhibits the Human Prostate Cancer Cells Through the Generation of Reactive Oxygen Species. Pathol. Oncol. Res. 2020, 26, 977–984. [Google Scholar] [CrossRef]
- Zhou, J.; Xia, L.; Zhang, Y. Naringin Inhibits Thyroid Cancer Cell Proliferation and Induces Cell Apoptosis through Repressing PI3K/AKT Pathway. Pathol. Res. Pract. 2019, 215, 152707. [Google Scholar] [CrossRef]
- He, M.; Qiu, C.; Wang, J.; Li, B.; Wang, G.; Ji, X. Naringin Targets Zeb1 to Suppress Osteosarcoma Cell Proliferation and Metastasis. Aging 2018, 10, 4141–4151. [Google Scholar] [CrossRef]
- Raha, S.; Kim, S.M.; Lee, H.J.; Yumnam, S.; Saralamma, V.V.G.; Ha, S.E.; Lee, W.S.; Kim, G.S. Naringin Induces Lysosomal Permeabilization and Autophagy Cell Death in AGS Gastric Cancer Cells. Am. J. Chin. Med. 2020, 48, 679–702. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, R.; Ni, H. Basic Research Eriodictyol Exerts Potent Anticancer Activity against A549 Human Lung Cancer Cell Line by Inducing Mitochondrial-Mediated Apoptosis, G2/M Cell Cycle Arrest and Inhibition of m-TOR/PI3K/Akt Signalling Pathway. Arch. Med. Sci. 2020, 16, 446–452. [Google Scholar] [CrossRef]
- Li, W.; Du, Q.; Li, X.; Zheng, X.; Lv, F.; Xi, X.; Huang, G. Eriodictyol Inhibits Proliferation, Metastasis and Induces Apoptosis of Glioma Cells via PI3K/Akt/NF-κB Signaling Pathway. Front. Pharmacol. 2020, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Ma, X.; Zhao, X.; Zhang, S. Baicalein Induces Apoptosis and Autophagy of Breast Cancer Cells via Inhibiting PI3K/AKT Pathway In Vivo and Vitro. Drug Des. Dev. Ther. 2018, 12, 3961–3972. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Chen, W.J.; Lin-Shiau, S.Y.; Ho, C.T.; Lin, J.K. Tangeretin Induces Cell-Cyle G1 Arrest through Inhibiting Cyclin-Dependent Kinases 2 and 4 Activities as Well as Elevating Cdk Inhibitors P21 and P27 in Human Colorectal Carcinoma Cells. Carcinogenesis 2002, 23, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Abe, K.; Gotoh, M. Citrus Flavone Tangeretin Inhibits Leukaemic HL-60 Cell Growth Partially through Induction of Apoptosis with Less Cytotoxicity on Normal Lymphocytes. Br. J. Cancer 1995, 72, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Cao, A.; Shi, J.; Yin, P.; Wang, L.; Ji, G.; Xie, J.; Wu, D. Tangeretin, a Citrus Polymethoxyflavonoid, Induces Apoptosis of Human Gastric Cancer AGS Cells through Extrinsic and Intrinsic Signaling Pathways. Oncol. Rep. 2014, 31, 1788–1794. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.A.; Piao, M.J.; Hyun, Y.J.; Zhen, A.X.; Cho, S.J.; Ahn, M.J.; Yi, J.M.; Hyun, J.W. Luteolin Promotes Apoptotic Cell Death via Upregulation of Nrf2 Expression by DNA Demethylase and the Interaction of Nrf2 with P53 in Human Colon Cancer Cells. Exp. Mol. Med. 2019, 51, 1–14. [Google Scholar] [CrossRef]
- Mahbub, A.A.; Le Maitre, C.L.; Cross, N.A.; Mahy, N.J. The Effect of Apigenin and Chemotherapy Combination Treatments on Apoptosis-Related Genes and Proteins in Acute Leukaemia Cell Lines. Sci. Rep. 2022, 12, 8858. [Google Scholar] [CrossRef]
- Li, N.; Sun, C.; Zhou, B.; Xing, H.; Ma, D.; Chen, G.; Weng, D. Low Concentration of Quercetin Antagonizes the Cytotoxic Effects of Anti-Neoplastic Drugs in Ovarian Cancer. PLoS ONE 2014, 9, e100314. [Google Scholar] [CrossRef]
- Nafees, S.; Mehdi, S.H.; Zafaryab, M.; Zeya, B.; Sarwar, T.; Rizvi, M.A. Synergistic Interaction of Rutin and Silibinin on Human Colon Cancer Cell Line. Arch. Med. Res. 2018, 49, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Fan, J.; Cheng, L.; Hu, P.; Liu, R. The Anticancer Activity of Genistein Is Increased in Estrogen Receptor Beta 1-Positive Breast Cancer Cells. OncoTargets. Ther. 2018, 11, 8153–8163. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Wang, Y.; Li, Y.; Li, Y.; Feng, C.; Li, Z. Daidzein-Rich Iso Fl Avones Aglycone Inhibits Lung Cancer Growth through Inhibition of NF-κB Signaling Pathway. Immunol. Lett. 2020, 222, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Gao, F.; Li, W.; Zhou, L.; Liu, W.; Li, M. Formononetin Inhibits Tumor Growth by Suppression of EGFR-Akt-Mcl-1 Axis in Non-Small Cell Lung Cancer. J. Exp. Clin. Cancer Res. 2020, 39, 62. [Google Scholar] [CrossRef] [PubMed]
- Won, D.; Kim, L.; Jang, B.; Yang, I.; Kwon, H.; Jin, B.; Oh, S.H.; Kang, J. In Vitro and in Vivo Anti-Cancer Activity of Silymarin on Oral Cancer. Tumor Biol. 2018, 40, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhu, X.; Wang, Q.; Li, X.; Wang, E.; Zhao, Q.; Wang, Q.; Cao, H. The Anti-Tumor Effect of Taxifolin on Lung Cancer via Suppressing Stemness and Epithelial-Mesenchymal Transition in Vitro and Oncogenesis in Nude Mice. Ann. Transl. Med. 2020, 8, 590. [Google Scholar] [CrossRef] [PubMed]
- Razak, S.; Afsar, T.; Ullah, A.; Almajwal, A.; Alkholief, M.; Alshamsan, A.; Jahan, S. Taxifolin, a Natural Flavonoid Interacts with Cell Cycle Regulators Causes Cell Cycle Arrest and Causes Tumor Regression by Activating Wnt/β-Catenin Signaling Pathway. BMC Cancer 2018, 18, 1043. [Google Scholar] [CrossRef]
- Silva, C.; Correia-Branco, A.; Andrade, N.; Ferreira, A.C.; Soares, M.L.; Sonveaux, P.; Stephenne, J.; Martel, F. Selective Pro-Apoptotic and Antimigratory Effects of Polyphenol Complex Catechin:Lysine 1:2 in Breast, Pancreatic and Colorectal Cancer Cell Lines. Eur. J. Pharmacol. 2019, 859, 172533. [Google Scholar] [CrossRef]
- Pereyra-Vergara, F.; Olivares-Corichi, I.M.; Perez-Ruiz, A.G.; Luna-Arias, J.P.; García-Sánchez, J.R. Apoptosis Induced by (−)-Epicatechin in Human Breast Cancer Cells Is Mediated by Reactive Oxygen Species. Molecules 2020, 25, 1020. [Google Scholar] [CrossRef]
- Wu, D.; Liu, Z.; Li, J.; Zhang, Q.; Zhong, P.; Teng, T.; Chen, M.; Xie, Z.; Ji, A.; Li, Y. Epigallocatechin-3-Gallate Inhibits the Growth and Increases the Apoptosis of Human Thyroid Carcinoma Cells through Suppression of EGFR/RAS/RAF/MEK/ERK Signaling Pathway. Cancer Cell Int. 2019, 19, 43. [Google Scholar] [CrossRef]
- Lakshmi, A.; Subramanian, S.P. Tangeretin Ameliorates Oxidative Stress in the Renal Tissues of Rats with Experimental Breast Cancer Induced by 7,12-Dimethylbenz[a]anthracene. Toxicol. Lett. 2014, 229, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Braidy, N.; Behzad, S.; Habtemariam, S.; Ahmed, T.; Daglia, M.; Nabavi, S.M.; Sobarzo-Sanchez, E.; Nabavi, S.F. Neuroprotective Effects of Citrus Fruit-Derived Flavonoids, Nobiletin and Tangeretin in Alzheimer’s and Parkinson’s Disease. CNS Neurol. Disord. Drug Targets 2017, 16, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.Y.; Wu, M.Y.; Lo, Y.C. Tangeretin Sensitizes SGS1-Deficient Cells by Inducing DNA Damage. J. Agric. Food Chem. 2013, 61, 6376–6382. [Google Scholar] [CrossRef] [PubMed]
- Arafa, E.S.A.; Shurrab, N.T.; Buabeid, M.A. Therapeutic Implications of a Polymethoxylated Flavone, Tangeretin, in the Management of Cancer via Modulation of Different Molecular Pathways. Adv. Pharmacol. Pharm. Sci. 2021, 2021, 4709818. [Google Scholar] [CrossRef] [PubMed]
- Koolaji, N.; Shammugasamy, B.; Schindeler, A.; Dong, Q.; Dehghani, F.; Valtchev, P. Citrus Peel Flavonoids as Potential Cancer Prevention Agents. Curr. Dev. Nutr. 2020, 4, nzaa025. [Google Scholar] [CrossRef] [PubMed]
- Ortuno, A.; Benavente-Garcia, O.; Castillo, J.; Alcaraz, M.; Vicente, V.; Del Rio, J. Beneficial Action of Citrus Flavonoids on Multiple Cancer-Related Biological Pathways. Curr. Cancer Drug Targets 2007, 7, 795–809. [Google Scholar] [CrossRef] [PubMed]
- Kandaswami, C.; Perkins, E.; Soloniuk, D.S.; Drzewiecki, G.; Middleton, E. Antitproliferative Effects of Citrus Flavonoids on a Human Squamous Cell Carcinoma in Vitro. Cancer Lett. 1991, 56, 147–152. [Google Scholar] [CrossRef]
- Lin, J.J.; Huang, C.C.; Su, Y.L.; Luo, H.L.; Lee, N.L.; Sung, M.T.; Wu, Y.J. Proteomics Analysis of Tangeretin-Induced Apoptosis through Mitochondrial Dysfunction in Bladder Cancer Cells. Int. J. Mol. Sci. 2019, 20, 1017. [Google Scholar] [CrossRef]
- Rooprai, H.K.; Christidou, M.; Murray, S.A.; Davies, D.; Selway, R.; Gullan, R.W.; Pilkington, G.J. Inhibition of Invasion by Polyphenols from Citrus Fruit and Berries in Human Malignant Glioma Cells In Vitro. Anticancer Res. 2021, 41, 619–633. [Google Scholar] [CrossRef]
- Surichan, S.; Arroo, R.R.; Tsatsakis, A.M.; Androutsopoulos, V.P. Tangeretin Inhibits the Proliferation of Human Breast Cancer Cells via CYP1A1/CYP1B1 Enzyme Induction and CYP1A1/CYP1B1–Mediated Metabolism to the Product 4′ Hydroxy Tangeretin. Toxicol. Vitr. 2018, 50, 274–284. [Google Scholar] [CrossRef]
- Weng, C.J.; Yen, G.C. Flavonoids, a Ubiquitous Dietary Phenolic Subclass, Exert Extensive In Vitro Anti-Invasive and in Vivo Anti-Metastatic Activities. Cancer Metastasis Rev. 2012, 31, 323–351. [Google Scholar] [CrossRef] [PubMed]
- Yumnam, S.; Raha, S.; Kim, S.; Venkatarame Gowda Saralamma, V.; Lee, H.; Ha, S.; Heo, J.; Lee, S.; Kim, E.; Lee, W.; et al. Identification of a Novel Biomarker in Tangeretin-induced Cell Death in AGS Human Gastric Cancer Cells. Oncol. Rep. 2018, 40, 3249–3260. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, Y.; Pal, I.; Banik, P.; Chakraborty, S.; Borkar, S.A.; Dey, G.; Mukherjee, A.; Mandal, M. Insights into Molecular Therapy of Glioma: Current Challenges and next Generation Blueprint. Acta Pharmacol. Sin. 2017, 38, 591–613. [Google Scholar] [CrossRef] [PubMed]
- Erices, J.I.; Torres, Á.; Niechi, I.; Bernales, I.; Quezada, C. Current Natural Therapies in the Treatment against Glioblastoma. Phyther. Res. 2018, 32, 2191–2201. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.L.; Wang, D.; Yu, X.D.; Zhou, Y.L. Tangeretin Induces Cell Cycle Arrest and Apoptosis through Upregulation of PTEN Expression in Glioma Cells. Biomed. Pharmacother. 2016, 81, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Arivazhagan, L.; Sorimuthu Pillai, S. Tangeretin, a citrus pentamethoxyflavone, exerts cytostatic effect via p53/p21 up-regulation and suppresses metastasis in 7,12-dimethylbenz(α)anthracene-induced rat mammary carcinoma. J. Nutr. Biochem. 2014, 24, 1140–1153. [Google Scholar] [CrossRef] [PubMed]
- Sangavi, P.; Langeswaran, K. Anti-Tumorigenic Efficacy of Tangeretin in Liver Cancer—An in-Silico Approach. Curr. Comput.-Aided. Drug Des. 2020, 17, 337–343. [Google Scholar] [CrossRef]
- Yoon, J.H.; Lim, T.G.; Lee, K.M.; Jeon, A.J.; Kim, S.Y.; Lee, K.W. Tangeretin Reduces Ultraviolet B (UVB)-Induced Cyclooxygenase-2 Expression in Mouse Epidermal Cells by Blocking Mitogen-Activated Protein Kinase (MAPK) Activation and Reactive Oxygen Species (ROS) Generation. J. Agric. Food Chem. 2011, 59, 222–228. [Google Scholar] [CrossRef]
- Chen, Q.; Gu, Y.; Tan, C.; Sundararajan, B.; Li, Z.; Wang, D.; Zhou, Z. Comparative Effects of Five Polymethoxyflavones Purified from Citrus Tangerina on Inflammation and Cancer. Front. Nutr. 2022, 9, 963662. [Google Scholar] [CrossRef]
- Zhu, G.; Lin, C.; Cheng, Z.; Wang, Q.; Hoffman, R.M.; Singh, S.R.; Huang, Y.; Zheng, W.; Yang, S.; Ye, J. TRAF6-Mediated Inflammatory Cytokines Secretion in LPS-Induced Colorectal Cancer Cells Is Regulated by MiR-140. Cancer Genom. Proteom. 2020, 17, 23–33. [Google Scholar] [CrossRef]
- Jain, S.; Dash, P.; Minz, A.P.; Satpathi, S.; Samal, A.G.; Behera, P.K.; Satpathi, P.S.; Senapati, S. Lipopolysaccharide (LPS) Enhances Prostate Cancer Metastasis Potentially through NF-κB Activation and Recurrent Dexamethasone Administration Fails to Suppress It In Vivo. Prostate 2019, 79, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yin, J.; Shen, W.; Gao, R.; Liu, Y.; Chen, Y.; Li, X.; Liu, C.; Xiang, R.; Luo, N. TLR4 Promotes Breast Cancer Metastasis via Akt/GSK3β/β-Catenin Pathway upon LPS Stimulation. Anat. Rec. 2017, 300, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xu, D.; Wang, Q.; Zheng, D.; Jiang, X.; Xu, L. LPS Induced miR-181a Promotes Pancreatic Cancer Cell Migration via Targeting PTEN and MAP2K4. Dig. Dis. Sci. 2014, 59, 1452–1460. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.K.; Chang, S.N.; Vadlamudi, Y.; Park, J.G.; Kang, S.C. Synergistic Therapy with Tangeretin and 5-Fluorouracil Accelerates the ROS/JNK Mediated Apoptotic Pathway in Human Colorectal Cancer Cell. Food Chem. Toxicol. 2020, 143, 111529. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.J.; Chao, Y.H.; Tung, Y.C.; Wu, T.Y.; Su, Z.Y. A Tangeretin Derivative Inhibits the Growth of Human Prostate Cancer LNCaP Cells by Epigenetically Restoring P21 Gene Expression and Inhibiting Cancer Stem-like Cell Proliferation. AAPS J. 2019, 21, 86. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.G.; Peacock, J.J.; Bourland, T.C.; Taylor, S.E.; Wright, J.M.; Patil, B.S.; Miller, E.G. Inhibition of Oral Carcinogenesis by Citrus Flavonoids. Nutr. Cancer 2007, 60, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.-H.; Weng, M.-S.; Lin, J.-K. Tangeretin Suppresses IL-1β-Induced Cyclooxygenase (COX)-2 Expression through Inhibition of P38 MAPK, JNK, and AKT Activation in Human Lung Carcinoma Cells. Biochem. Pharmacol. 2007, 73, 215–227. [Google Scholar] [CrossRef]
- Zhu, W.B.; Xiao, N.; Liu, X.J. Dietary Flavonoid Tangeretin Induces Reprogramming of Epithelial to Mesenchymal Transition in Prostate Cancer Cells by Targeting the PI3K/Akt/MTOR Signaling Pathway. Oncol. Lett. 2018, 15, 433–440. [Google Scholar] [CrossRef]
- He, Z.; Li, B.; Rankin, G.O.; Rojanasakul, Y.; Chen, Y.C. Selecting Bioactive Phenolic Compounds as Potential Agents to Inhibit Proliferation and VEGF Expression in Human Ovarian Cancer Cells. Oncol. Lett. 2015, 9, 1444–1450. [Google Scholar] [CrossRef]
- Jaboin, J.; Iii, W.A.V.; Banik, N.L.; Giglio, P. A Novel Component from Citrus, Ginger, and Mushroom Family Exhibits Antitumor Activity on Human Meningioma Cells through Suppressing the Wnt/β-Catenin Signaling Pathway. Tumor Biol. 2022, 36, 7027–7034. [Google Scholar] [CrossRef]
- Li, Y.R.; Li, S.; Ho, C.T.; Chang, Y.H.; Tan, K.T.; Chung, T.W.; Wang, B.Y.; Chen, Y.K.; Lin, C.C. Tangeretin Derivative, 5-Acetyloxy-6,7,8,4′-Tetramethoxyflavone Induces G2/M Arrest, Apoptosis and Autophagy in Human Non-Small Cell Lung Cancer Cells In Vitro and In Vivo. Cancer Biol. Ther. 2016, 17, 48–64. [Google Scholar] [CrossRef]
- Periyasamy, K.; Baskaran, K.; Ilakkia, A.; Vanitha, K.; Selvaraj, S.; Sakthisekaran, D. Antitumor Efficacy of Tangeretin by Targeting the Oxidative Stress Mediated on 7,12-Dimethylbenz(a) Anthracene-Induced Proliferative Breast Cancer in Sprague-Dawley Rats. Cancer Chemother. Pharmacol. 2015, 75, 263–272. [Google Scholar] [CrossRef]



| Subclasses | Compounds | Antitumor Activity | Cancer/Cell |
|---|---|---|---|
 Anthocyanin |  Cyanidin | Anti-proliferative Anti-metastatic Apoptosis ↓ (NF-κB) Anti-metastatic Apoptosis ↓ (NF-κB) | Kidney/ 786-O; ACHN; [117] Colorectal/ HCT116; HT29; SW620; [118] |
 Delphinidin | Apoptosis ┤ (ERK; NF-κB) | Breast/ MDA; MB-453; BT-474; [119] | |
 Malvidin | Anti-proliferative Apoptosis | Leukemia/ SUP-B15; KG-1; [115] | |
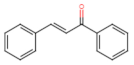 Chalcones | 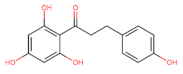 Phloretin | Anti-proliferative ┤ Migration ↑ ROS | Prostate/ PC3; DU145; [120] |
 Flavanones | 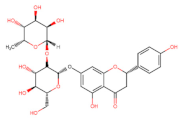 Naringin | Apoptosis ↓ (PI3K/AKT) Anti-proliferative Anti-metastatic ┤ (Zeb1) Autophagy ↓ (PI3K/AKT/mTOR) | Thyroid/ TPC-1; SW1736; [121] Osteosarcoma/ MG63, U2OS; [122] Gastric/ AGS; [123] |
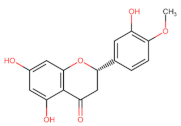 Hesperidin | Apoptosis ↑ (FADD/caspase-8) | Lung/ H522; [114] | |
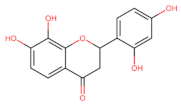 Eriodictyol | Anti-proliferative Apoptosis ┤ mTOR/PI3K/Akt Anti-proliferative Apoptosis -Anti-metastatic ┤ PI3K/Akt/NF-κB | Lung/ A549; [124] Glioma/ U87MG; CHG-5; [125] | |
 Flavones |  Baicalein | Induction apoptosis Autophagy ┤ (PI3K/AKT) | Breast/ MCF-7; MDA-MB-231; [126] |
 Tangeretin | Anti-proliferative ┤ (Cdk2/Cdk4) Anti-proliferative Apoptosis ↓ (MMP) ↑ Caspases-3, -8, -9 | Colorectal/ COLO 205; [127] Leukemia/ HL-60 [128] Gastric/ AGS; [129] | |
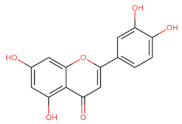 Luteolin | ↑ p53 Apoptosis ┤ DNA metiltransferas | Colo/ HT-29 [130] | |
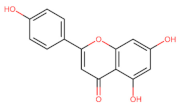 Apegenin | Apoptosis ↑ BAX, CYT c ,SMAC/DIABLO, HTRA2/OMI, CASP-3 and -9 | Leukemia/ THP-1; Jukart; [131] | |
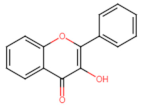 Flavonols |  Quercetin | Antioxidant ↓ (ROS) | Ovarian/ C13* cisplatin-resistant (C13*) [132] |
 Rutin | Induction apoptosis ↑ (caspases-3, -8, -9) | Colo/ HT-29 [133] | |
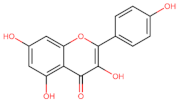 Kaempferol | Apoptosis ┤ Akt/mTOR | Pancreas/ PANC-1; Mia PaCa-2; [83] | |
 Isoflavones | 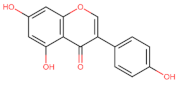 Genistein | Anti-proliferative Cell cycle arrest G2/M | Breast/ MCF-7; ERβ1; MDA-MB-231/ERβ1; [134] |
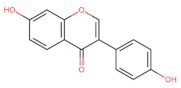 Daidzein | Anti-proliferative ┤ NF-κB | Lung/ A594 e 95D; [135] | |
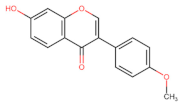 Formononetin | Anti-proliferative ┤ EGFR-Akt | Lung/ HCC827; H3255; H1975; A549; H1299; [136] | |
 Flavanonols | 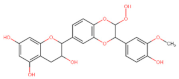 Silibinin | Anti-proliferative Induction apoptosis Cell cycle arrest G2/M ┤ (STAT3) | Gastric/ MGC803; [116] |
 Sylimarin | Anti-proliferative Apoptosis ↑ (Caspases-5, -8) | Oral/ HSC-4; YD15; Ca9.22; [137] | |
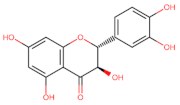 Taxifolin | Anti-proliferative ┤ (EMT) ↑ E-cadherin Cytotoxicity Cell cycle arrest G2/M | Lung/ A549; H1975; [138] Colorectal/ HCT116; HT29; [139] | |
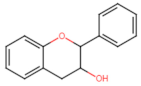 Flavanol | 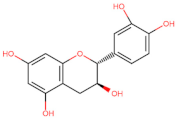 Catechin | Anti-metastatic ┤ (Wnt) | Breast/ MCF-7; HTB-26; Pancreas/ PANC-1; AsPC-1 Colorectal/ HT-29; Caco-2; [140] |
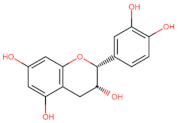 Epicatechin | Apoptosis ↑ (DR4/DR5) | Breast/ MDA-MB-231; MCF-7; [141] | |
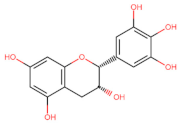 Epigallocatechin (EGCG) | Anti-proliferative Anti-metastatic ↑ AMPK Anti-proliferative Apoptosis Cell cycle arrest S ┤ EGFR/RAS/RAF/MEK/ERK | H1299, A549; [66] Thyroid/ TT; TPC-1; ARO; [142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Luna, F.C.F.; Ferreira, W.A.S.; Casseb, S.M.M.; de Oliveira, E.H.C. Anticancer Potential of Flavonoids: An Overview with an Emphasis on Tangeretin. Pharmaceuticals 2023, 16, 1229. https://doi.org/10.3390/ph16091229
de Luna FCF, Ferreira WAS, Casseb SMM, de Oliveira EHC. Anticancer Potential of Flavonoids: An Overview with an Emphasis on Tangeretin. Pharmaceuticals. 2023; 16(9):1229. https://doi.org/10.3390/ph16091229
Chicago/Turabian Stylede Luna, Francisco Canindé Ferreira, Wallax Augusto Silva Ferreira, Samir Mansour Moraes Casseb, and Edivaldo Herculano Correa de Oliveira. 2023. "Anticancer Potential of Flavonoids: An Overview with an Emphasis on Tangeretin" Pharmaceuticals 16, no. 9: 1229. https://doi.org/10.3390/ph16091229
APA Stylede Luna, F. C. F., Ferreira, W. A. S., Casseb, S. M. M., & de Oliveira, E. H. C. (2023). Anticancer Potential of Flavonoids: An Overview with an Emphasis on Tangeretin. Pharmaceuticals, 16(9), 1229. https://doi.org/10.3390/ph16091229








