The Skin and Natural Cannabinoids–Topical and Transdermal Applications
Abstract
1. Introduction
2. The Topical versus the Transdermal Route of Administration for Natural Cannabinoids
2.1. The Skin as a Potential Therapeutic Target
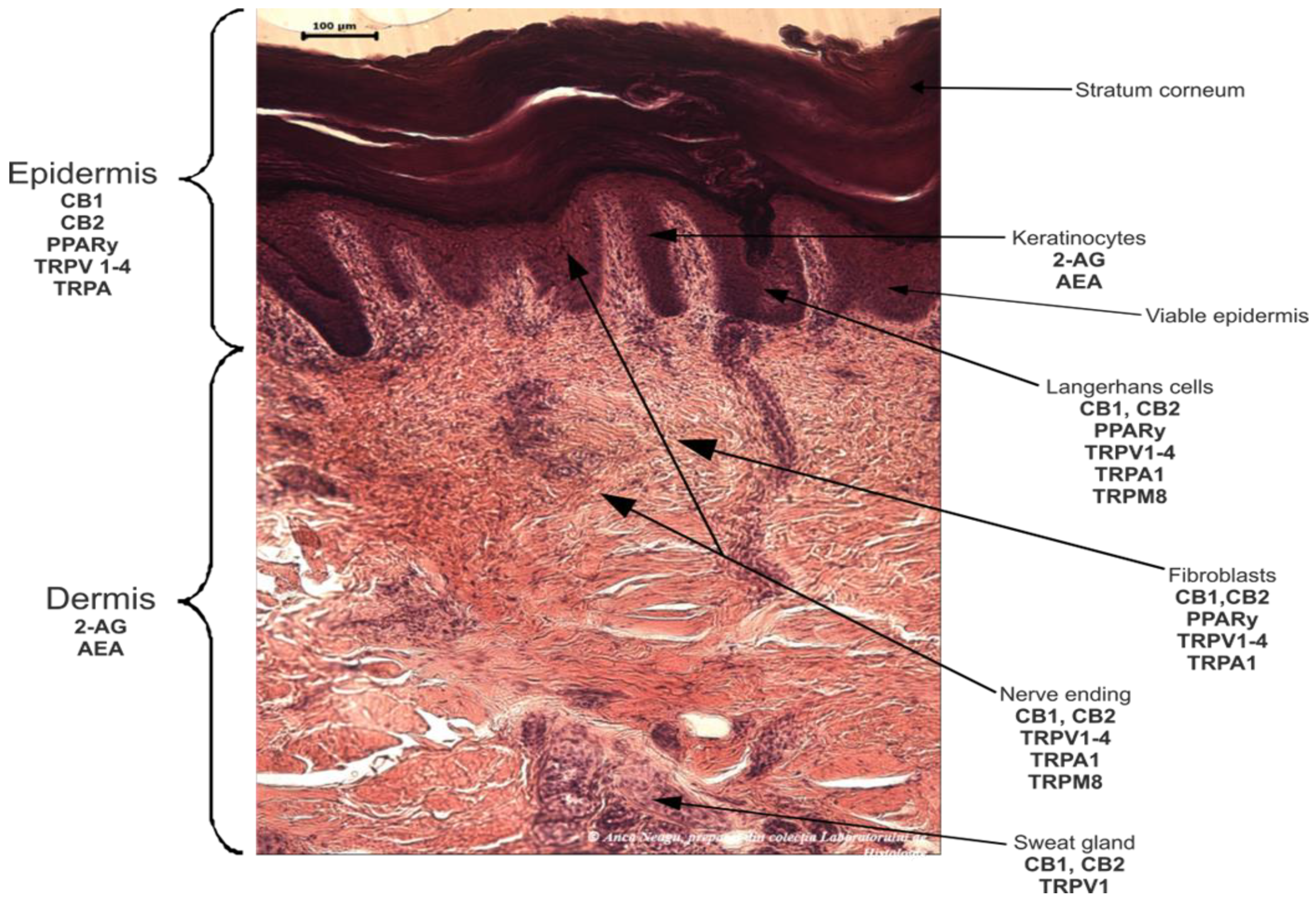
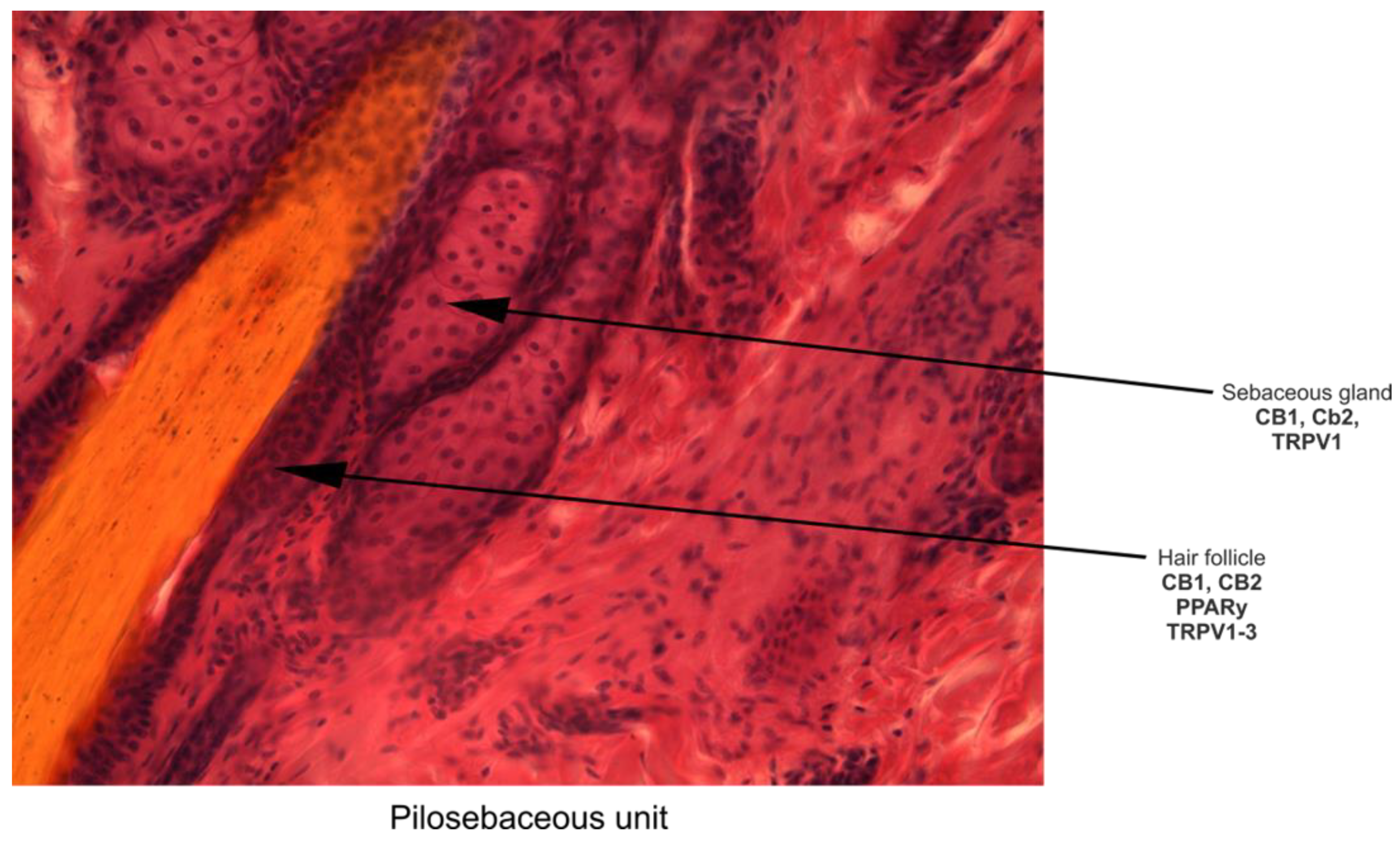
2.1.1. The Skin Endocannabinoid System
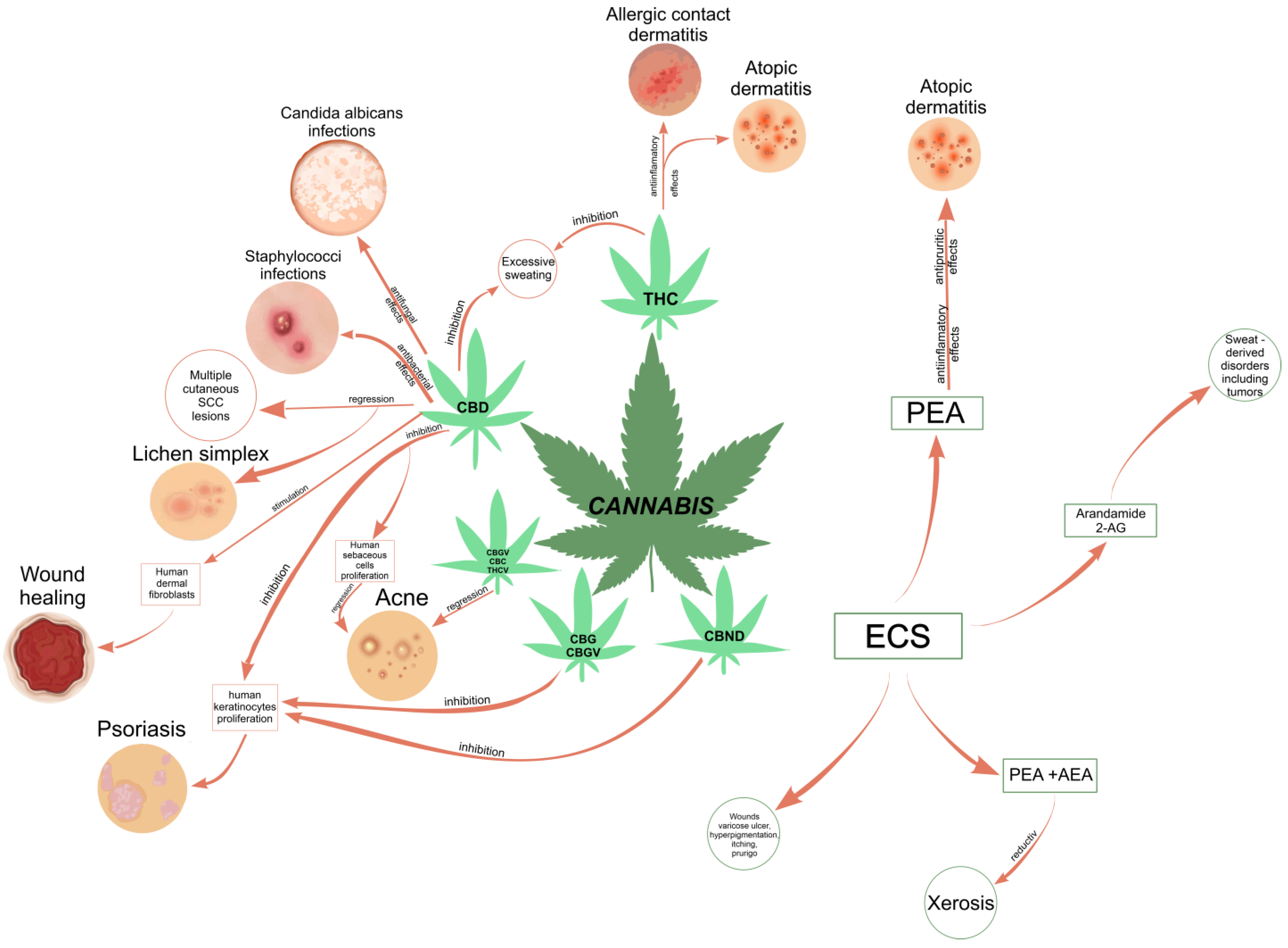
2.1.2. Skin Disorders Addressed by Cannabis-Based Medicines
| Affection (Estimative Costs around the Current Course of Therapy) | Current Drug Therapies | Drawbacks/ Most Significant Adverse Effects | Reference | |
|---|---|---|---|---|
| Psoriasis (between USD 23.9 and USD 35.4 billion annually in the US | topical agents | vitamin D analogues corticosteroids | poor therapy responses | [35,112] |
| phototherapy | NB-UVB PUVA | major risks of skin cancer | ||
| standard systemic | acitretin | dry skin, hair loss, hyperlipidemia, hepatotoxicity | ||
| ciclosporin | hypertension, irreversible renal toxicity | |||
| methotrexate | bone marrow suppression, liver fibrosis, teratogenicity, hepatitis | |||
| biologic agents | TNF IL-17 L-23 inhibitors | individualized therapy regimen | ||
| small molecule inhibitors | apremilast dimethyl fumarate | used only in studies | ||
| Atopic dermatitis (USD 3.8 billion annually in the US) | topical moisturizers | glycerin, alpha hydroxy acids, hyaluronic acid, sorbitol, urea, lanolin, mineral oil, olive oil, silicone, collagen, elastin, glyceryl stearate, shea acid, stearic acid | a large number of studies lacking any significant relevance regarding the efficacy | [113,114] |
| topical corticosteroids | clobetasol, fluocinonide, betamethasone, triamcinolone, fluticasone budesonide, hydrocortisone | atrophy, striae, rosacea, perioral dermatitis, acne, purpura, hypertrichosis, alteration of the skin’s pigment, sluggish wound healing, worsening of skin infections | [115] | |
| topical calcineurin inhibitors | tacrolimus pimecrolimus | skin burning, increased bone growth and decreased bone density, inhibition of the hypothalamic-pituitary-adrenal axis in children | [116,117,118] | |
| phototherapy | UVB | xerosis, erythema, actinic keratosis, skin damage tenderness, possible carcinogenic risk | [118,119] | |
| systemic immunotherapy | cyclosporine azathioprine | headache, serum lipid TSH elevation, teratogenicity, liver dysfunction, leukopenia, gastric ulcer, osteoporosis, glaucoma | [120] | |
| Allergic contact dermatitis (USD 5.3 billion in the US in 2015) | systemic corticosteroids | clobetasol, betamethasone, diflorasone, fluocinonide, halcinonide, mometasone furoate, fluocinolone acetonide, desoximetasone, triamcinolone acetonide, alclometasone dipropionate, hydrocortisone, dexamethasone, prednisolone, methylprednisolone | atrophy, infections, hypertrichosis, allergic reactions, problems with systemic absorption | [36,37,121] |
| calcineurin antagonists | cyclosporine | risk of malignity | [36,38] | |
| ultraviolet therapy | PUVA UVB | lentigines (freckling), photoaging, precancerous lesions, actinic keratoses, skin cancer, hyperpigmentation, redness, dryness, pruritus, herpes simplex virus reactivation, polymorphic light eruption | [36,39] | |
| Asteatotic eczema (USD 5.3 billion annually) | topical steroid | hydrocortisone, glucocorticoid, nonfluorinated steroids (hydrocortisone valerate, hydrocortisone probutate, mometasone furoate), fluorinated steroids (dexamethasone, triamcinolone, fluocortolone, flumethasone, betamethasone) | skin atrophy, striae atrophicans, xeroderma, delayed wound repair, capillary telangiectasia, rosacea-like dermatitis, steroid purpura, steroid acne milium, pigmentation disorder, acne uncontrollable, hyper-pilosis | [36,40,41] |
| phototherapy | UVA (UVA-1) UVB PUVA | suppressing immune system | [40] | |
| Acne (acne is thought to cost the economy USD 3 billion annually) | topical retinoids | adapalene, isotretinoin, motretinide, retinoyl-β-glucuronide, tazarotene, tretinoin | used in various combinations, all of these topical treatments disrupt the skin’s natural barrier, requiring frequent treatment changes | [42,43,44] |
| topical antibiotics | clindamycin, erythromycin | disruption of the skin’s natural barrier | ||
| diverse topical agents | azelaic acid, benzoyl peroxide, chemical peels, corticosteroids, dapsone, hydrogen peroxide, niacinamide, salicylic acid, sodium sulfacetamide, sulfur triclosan | specific adverse events | ||
| systemic retinoids | isotretinoin | hematological/lymphatic disorders, immune system disorders, metabolic and nutritional disorders | ||
| systemic antibiotics | azithromycin, clindamycin, co-trimoxazole, doxycycline, erythromycin, levofloxacin, minocycline, roxithromycin | specific adverse events | ||
| other systemic agents | hormones, clofazimine, corticosteroids, ibuprofen, zinc sulfate | specific adverse events | ||
| Seborrhea (in 2021, the value of the global seborrheic dermatitis market was estimated to be USD 2.2 billion) | antifungals | itraconazole, terbinafine, fluconazole, ketoconazole, pramiconazole | itching, burning sensation, dryness | [108,109,122] |
| corticosteroids | prednisone | skin atrophy, telangiectasias, folliculitis, hypertrichosis, hypopigmentation | [109] | |
| calcineurin inhibitors | pimecrolimus | skin malignancy lymphoma | [123] | |
| phototherapy | UVB PUVA red and blue LED light | burning itching sensation genital tumor | [123] | |
2.2. The Skin as a Potential Route for Systemic Administration
2.2.1. Why Transdermal Administration of Cannabinoids Could Be the Solution?
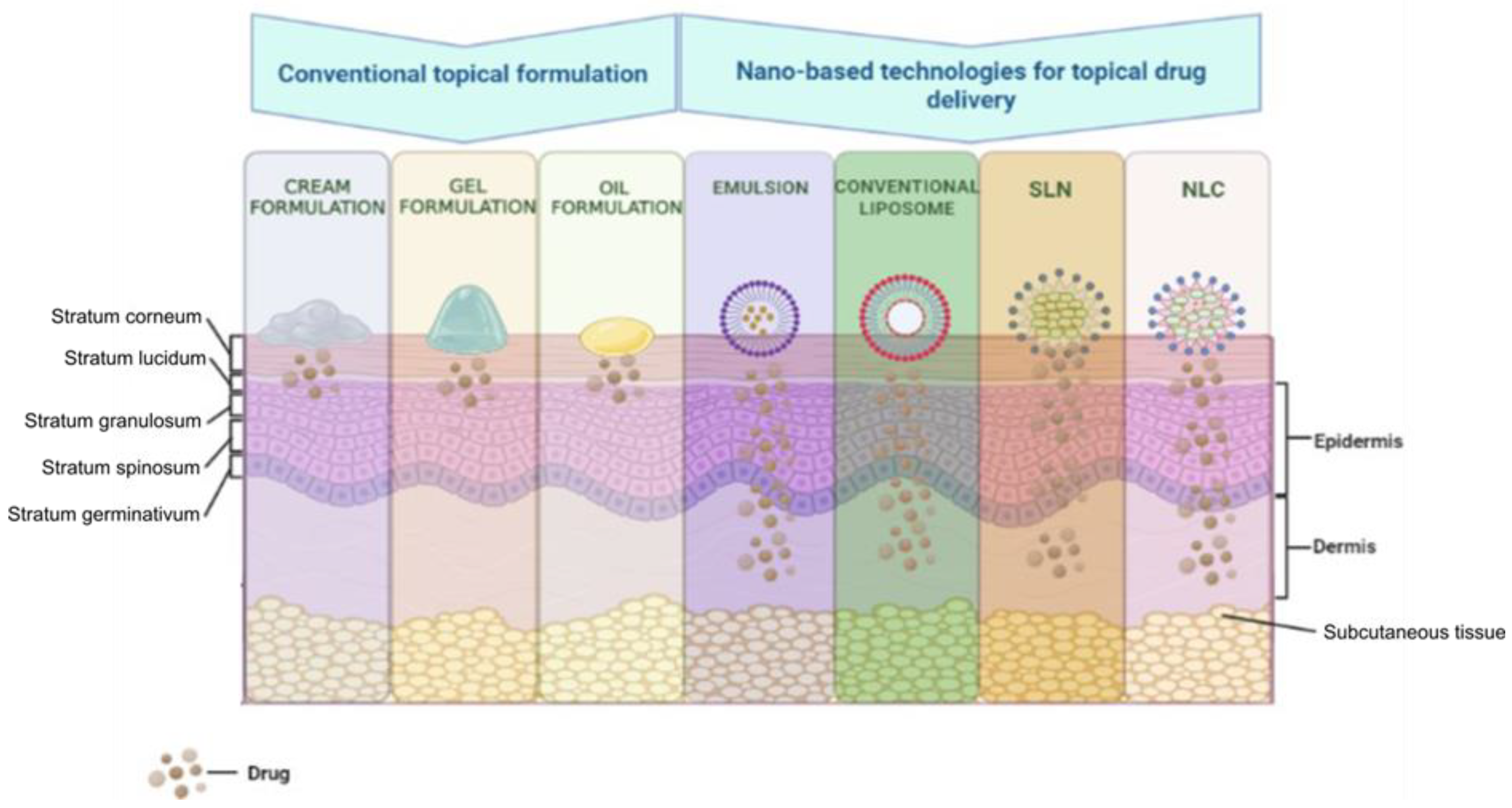
2.2.2. The Use of Nanotechnologies for Cannabinoid Transdermal Administration
3. Future Perspectives on the Transdermal Delivery of Cannabinoids
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Abel, E.L.; Emboden, W. Marihuana. The First Twelve Thousand Years. Ernest L. Abel. Q. Rev. Biol. 1981, 56, 514. [Google Scholar] [CrossRef]
- Filipiuc, L.E.; Ababei, D.C.; Alexa-Stratulat, T.; Pricope, C.V.; Bild, V.; Stefanescu, R.; Stanciu, G.D.; Tamba, B.-I. Major Phytocannabinoids and Their Related Compounds: Should We Only Search for Drugs That Act on Cannabinoid Receptors? Pharmaceutics 2021, 13, 1823. [Google Scholar] [CrossRef]
- Johal, H.; Vannabouathong, C.; Chang, Y.; Zhu, M.; Bhandari, M. Medical cannabis for orthopaedic patients with chronic musculoskeletal pain: Does evidence support its use? Ther. Adv. Musculoskelet. Dis. 2020, 12, 1759720X2093796. [Google Scholar] [CrossRef]
- Butrica, J.L. The Medical Use of Cannabis Among the Greeks and Romans. J. Cannabis Ther. 2002, 2, 51–70. [Google Scholar] [CrossRef]
- Turgeman, I.; Bar-Sela, G. Cannabis for cancer–illusion or the tip of an iceberg: A review of the evidence for the use of Cannabis and synthetic cannabinoids in oncology. Expert Opin. Investig. Drugs 2019, 28, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Zuardi, A.W. History of cannabis as a medicine: A review. Rev. Bras. Psiquiatr. 2006, 28, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Gomes, A.; Boas, I.; Marto, J.; Ribeiro, H. Cannabis-Based Products for the Treatment of Skin Inflammatory Diseases: A Timely Review. Pharmaceuticals 2022, 15, 210. [Google Scholar] [CrossRef]
- McPartland, J.M. Cannabis Systematics at the Levels of Family, Genus, and Species. Cannabis Cannabinoid Res. 2018, 3, 203–212. [Google Scholar] [CrossRef]
- Gloss, D. An Overview of Products and Bias in Research. Neurotherapeutics 2015, 12, 731–734. [Google Scholar] [CrossRef]
- Pollio, A. The Name of Cannabis: A Short Guide for Nonbotanists. Cannabis Cannabinoid Res. 2016, 1, 234–238. [Google Scholar] [CrossRef]
- Punja, Z.K.; Holmes, J.E. Hermaphroditism in Marijuana (Cannabis sativa L.) Inflorescences—Impact on Floral Morphology, Seed Formation, Progeny Sex Ratios, and Genetic Variation. Front. Plant Sci. 2020, 11, 718. [Google Scholar] [CrossRef]
- Farag, S.; Kayser, O. The Cannabis Plant: Botanical Aspects. In Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis, and Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 3–12. ISBN 9780128008270. [Google Scholar]
- Chayasirisobhon, S. Mechanisms of Action and Pharmacokinetics of Cannabis. Perm. J. 2021, 25, 1–3. [Google Scholar] [CrossRef]
- Milay, L.; Berman, P.; Shapira, A.; Guberman, O.; Meiri, D. Metabolic Profiling of Cannabis Secondary Metabolites for Evaluation of Optimal Postharvest Storage Conditions. Front. Plant Sci. 2020, 11, 583605. [Google Scholar] [CrossRef] [PubMed]
- Tamba, B.I.; Stanciu, G.D.; Urîtu, C.M.; Rezus, E.; Stefanescu, R.; Mihai, C.T.; Luca, A.; Rusu-zota, G.; Leon-constantin, M.M.; Cojocaru, E.; et al. Challenges and opportunities in preclinical research of synthetic cannabinoids for pain therapy. Medicina 2020, 56, 24. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. History of Cannabis and Its Preparations in Saga, Science, and Sobriquet. Chem. Biodivers. 2007, 4, 1614–1648. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.; Rosado, T.; Soares, S.; Simão, A.Y.; Caramelo, D.; Luís, Â.; Fernández, N.; Barroso, M.; Gallardo, E.; Duarte, A.P. Cannabis and Its Secondary Metabolites: Their Use as Therapeutic Drugs, Toxicological Aspects, and Analytical Determination. Medicines 2019, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Cannabinoid pharmacology: The first 66 years. Br. J. Pharmacol. 2006, 147, 163–171. [Google Scholar] [CrossRef]
- Curran, H.V.; Freeman, T.P.; Mokrysz, C.; Lewis, D.A.; Morgan, C.J.A.; Parsons, L.H. Keep off the grass? Cannabis, cognition and addiction. Nat. Rev. Neurosci. 2016, 17, 293–306. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. In Progress in the Chemistry of Organic Natural Products; Kinghorn, A., Falk, H., Gibbons, S., Kobayashi, J., Eds.; Springer: Cham, Switzerland, 2017; Volume 103, pp. 1–36. [Google Scholar]
- Palrasu, M.; Wright, L.; Patel, M.; Leech, L.; Branch, S.; Harrelson, S.; Khan, S. Perspectives on Challenges in Cannabis Drug Delivery Systems: Where Are We? Med. Cannabis Cannabinoids 2022, 5, 102–119. [Google Scholar] [CrossRef]
- Mahmoudinoodezh, H.; Telukutla, S.R.; Bhangu, S.K.; Bachari, A.; Cavalieri, F.; Mantri, N. The Transdermal Delivery of Therapeutic Cannabinoids. Pharmaceutics 2022, 14, 438. [Google Scholar] [CrossRef]
- Mechoulam, R.; Parker, L.A.; Gallily, R. Cannabidiol: An Overview of Some Pharmacological Aspects. J. Clin. Pharmacol. 2002, 42, 11S–19S. [Google Scholar] [CrossRef] [PubMed]
- Paudel, K.S.; Hammell, D.C.; Agu, R.U.; Valiveti, S.; Stinchcomb, A.L. Cannabidiol bioavailability after nasal and transdermal application: Effect of permeation enhancers. Drug Dev. Ind. Pharm. 2010, 36, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Bruni, N.; Della Pepa, C.; Oliaro-Bosso, S.; Pessione, E.; Gastaldi, D.; Dosio, F. Cannabinoid Delivery Systems for Pain and Inflammation Treatment. Molecules 2018, 23, 2478. [Google Scholar] [CrossRef] [PubMed]
- Vacek, J.; Vostalova, J.; Papouskova, B.; Skarupova, D.; Kos, M.; Kabelac, M.; Storch, J. Antioxidant function of phytocannabinoids: Molecular basis of their stability and cytoprotective properties under UV-irradiation. Free Radic. Biol. Med. 2021, 164, 258–270. [Google Scholar] [CrossRef]
- Jîtcă, G.; Ősz, B.E.; Vari, C.E.; Rusz, C.-M.; Tero-Vescan, A.; Pușcaș, A. Cannabidiol: Bridge between Antioxidant Effect, Cellular Protection, and Cognitive and Physical Performance. Antioxidants 2023, 12, 485. [Google Scholar] [CrossRef]
- Palmieri, B.; Laurino, C.; Vadalà, M. A therapeutic effect of cbd-enriched ointment in inflammatory skin diseases and cutaneous scars. Clin. Ther. 2019, 170, e93–e99. [Google Scholar] [CrossRef]
- Tortolani, D.; Di Meo, C.; Standoli, S.; Ciaramellano, F.; Kadhim, S.; Hsu, E.; Rapino, C.; Maccarrone, M. Rare Phytocannabinoids Exert Anti-Inflammatory Effects on Human Keratinocytes via the Endocannabinoid System and MAPK Signaling Pathway. Int. J. Mol. Sci. 2023, 24, 2721. [Google Scholar] [CrossRef]
- Oláh, A.; Markovics, A.; Szabó-Papp, J.; Szabó, P.; Stott, C.; Zouboulis, C.; Bíró, T. Differential effectiveness of selected non-psychotropic phytocannabinoids on human sebocyte functions implicates their introduction in dry/seborrheic skin and acne treatment. Exp. Dermatol. 2016, 25, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Czifra, G.; Szöllősi, A.G.; Tóth, B.I.; Demaude, J.; Bouez, C.; Breton, L.; Bíró, T. Endocannabinoids Regulate Growth and Survival of Human Eccrine Sweat Gland–Derived Epithelial Cells. J. Investig. Dermatol. 2012, 132, 1967–1976. [Google Scholar] [CrossRef]
- Marta, G.; Robinson, G.; Groves, A.; Haselhorst, L.; Nandakumar, S.; Stahl, C.; Kovalchuk, O.; Kovalchuk, I. Phytocannabinoids Stimulate Rejuvenation and Prevent Cellular Senescence in Human Dermal Fibroblasts. Cells 2022, 11, 3939. [Google Scholar] [CrossRef]
- Mazuz, M.; Tiroler, A.; Moyal, L.; Hodak, E.; Nadarajan, S.; Vinayaka, A.C.; Gorovitz-Haris, B.; Lubin, I.; Drori, A.; Drori, G.; et al. Synergistic cytotoxic activity of cannabinoids from cannabis sativa against cutaneous T-cell lymphoma (CTCL) in-vitro and ex-vivo. Oncotarget 2020, 11, 1141–1156. [Google Scholar] [CrossRef]
- Richard, M.A.; Paul, C.; Nijsten, T.; Gisondi, P.; Salavastru, C.; Taieb, C.; Trakatelli, M.; Puig, L.; Stratigos, A.; EADV Burden of Skin Diseases Project Team. Prevalence of most common skin diseases in Europe: A population-based study. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Raharja, A.; Mahil, S.K.; Barker, J.N. Psoriasis: A brief overview. Clin. Med. 2021, 21, 170–173. [Google Scholar] [CrossRef]
- Cohen, D.E.; Heidary, N. Treatment of irritant and allergic contact dermatitis. Dermatol. Ther. 2004, 17, 334–340. [Google Scholar] [CrossRef]
- Smith Begolka, W.; Chovatiya, R.; Thibau, I.J.; Silverberg, J.I. Financial Burden of Atopic Dermatitis Out-of-Pocket Health Care Expenses in the United States. Dermatitis 2021, 32, S62–S70. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.B.; Franco, A.I.; Beck, L.A.; Prezzano, J.C. Treatment resistant atopic dermatitis: Challenges and solutions. Clin. Cosmet. Investig. Dermatol. 2019, 12, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Vangipuram, R.; Feldman, S. Ultraviolet phototherapy for cutaneous diseases: A concise review. Oral Dis. 2016, 22, 253–259. [Google Scholar] [CrossRef]
- Plötz, S.G.; Wiesender, M.; Todorova, A.; Ring, J. What is new in atopic dermatitis/eczema? Expert Opin. Emerg. Drugs 2014, 19, 441–458. [Google Scholar] [CrossRef]
- Hino, H. Topical use of steroids in the aged. J. Jpn. Med. Assoc. 1999, 122, 142–147. [Google Scholar]
- Fox, L.; Csongradi, C.; Aucamp, M.; du Plessis, J.; Gerber, M. Treatment Modalities for Acne. Molecules 2016, 21, 1063. [Google Scholar] [CrossRef]
- Tucker, D.; Masood, S. Seborrheic Dermatitis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Brownstone, N. Meeting the Challenges of Acne in Adults. Dermatol. Times 2022, 43, 36–38. [Google Scholar]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Stasiewicz, A.; Jarocka-Karpowicz, I.; Markowska, A.; Wroński, A.; Gęgotek, A.; Skrzydlewska, E. Antioxidant and Anti-inflammatory Effect of Cannabidiol Contributes to the Decreased Lipid Peroxidation of Keratinocytes of Rat Skin Exposed to UV Radiation. Oxid. Med. Cell. Longev. 2021, 2021, 6647222. [Google Scholar] [CrossRef]
- Klahn, P. Cannabinoids-Promising Antimicrobial Drugs or Intoxicants with Benefits? Antibiotics 2020, 9, 297. [Google Scholar] [CrossRef]
- Luz-Veiga, M.; Amorim, M.; Pinto-Ribeiro, I.; Oliveira, A.L.S.; Silva, S.; Pimentel, L.L.; Rodríguez-Alcalá, L.M.; Madureira, R.; Pintado, M.; Azevedo-Silva, J.; et al. Cannabidiol and Cannabigerol Exert Antimicrobial Activity without Compromising Skin Microbiota. Int. J. Mol. Sci. 2023, 24, 2389. [Google Scholar] [CrossRef]
- Feldman, M.; Sionov, R.; Mechoulam, R.; Steinberg, D. Anti-Biofilm Activity of Cannabidiol against Candida albicans. Microorganisms 2021, 9, 441. [Google Scholar] [CrossRef] [PubMed]
- Kühbacher, A.; Burger-Kentischer, A.; Rupp, S. Interaction of Candida Species with the Skin. Microorganisms 2017, 5, 32. [Google Scholar] [CrossRef]
- Wroński, A.; Jarocka-Karpowicz, I.; Stasiewicz, A.; Skrzydlewska, E. Phytocannabinoids in the Pharmacotherapy of Psoriasis. Molecules 2023, 28, 1192. [Google Scholar] [CrossRef]
- Morin, C.; Raef, H.; Elmariah, S. Neuropathic itch successfully treated with oral cannabinoids: A case series. JAAD Case Rep. 2021, 17, 38–42. [Google Scholar] [CrossRef]
- Martinelli, G.; Magnavacca, A.; Fumagalli, M.; Dell’Agli, M.; Piazza, S.; Sangiovanni, E. Cannabis sativa and Skin Health: Dissecting the Role of Phytocannabinoids. Planta Med. 2021, 88, 492–506. [Google Scholar] [CrossRef]
- Schräder, N.H.B.; Gorell, E.S.; Stewart, R.E.; Duipmans, J.C.; Harris, N.; Perez, V.A.; Tang, J.Y.; Wolff, A.P.; Bolling, M.C. Cannabinoid use and effects in patients with epidermolysis bullosa: An international cross-sectional survey study. Orphanet J. Rare Dis. 2021, 16, 377. [Google Scholar] [CrossRef]
- Bachari, A.; Piva, T.; Salami, S.A.; Jamshidi, N.; Mantri, N. Roles of Cannabinoids in Melanoma: Evidence from In Vivo Studies. Int. J. Mol. Sci. 2020, 21, 6040. [Google Scholar] [CrossRef] [PubMed]
- Laborada, J.; Cohen, P. Cutaneous Squamous Cell Carcinoma and Lichen Simplex Chronicus Successfully Treated with Topical Cannabinoid Oil: A Case Report and Summary of Cannabinoids in Dermatology. Cureus 2022, 14, e23850. [Google Scholar] [CrossRef] [PubMed]
- Sheriff, T.; Lin, M.J.; Dubin, D.; Khorasani, H. The potential role of cannabinoids in dermatology. J. Dermatolog. Treat. 2020, 31, 839–845. [Google Scholar] [CrossRef]
- Peyravian, N.; Deo, S.; Daunert, S.; Jimenez, J. The Anti-Inflammatory Effects of Cannabidiol (CBD) on Acne. J. Inflamm. Res. 2022, 15, 2795–2801. [Google Scholar] [CrossRef] [PubMed]
- Baswan, S.M.; Klosner, A.E.; Glynn, K.; Rajgopal, A.; Malik, K.; Yim, S.; Stern, N. Therapeutic Potential of Cannabidiol (CBD) for Skin Health and Disorders. Clin. Cosmet. Investig. Dermatol. 2020, 13, 927–942. [Google Scholar] [CrossRef]
- Chelliah, M.P.; Zinn, Z.; Khuu, P.; Teng, J.M.C. Self-initiated use of topical cannabidiol oil for epidermolysis bullosa. Pediatr. Dermatol. 2018, 35, e224–e227. [Google Scholar] [CrossRef]
- Maida, V.; Shi, R.B.; Fazzari, F.G.T.; Zomparelli, L. Topical cannabis-based medicines—A novel adjuvant treatment for venous leg ulcers: An open-label trial. Exp. Dermatol. 2021, 30, 1258–1267. [Google Scholar] [CrossRef]
- Makhakhe, L. Topical cannabidiol (CBD) in skin pathology—A comprehensive review and prospects for new therapeutic opportunities. S. Afr. Fam. Pract. 2022, 64, 4. [Google Scholar] [CrossRef]
- Irving, A.; Abdulrazzaq, G.; Chan, S.L.F.; Penman, J.; Harvey, J.; Alexander, S.P.H. Cannabinoid Receptor-Related Orphan G Protein-Coupled Receptors. Adv. Pharmacol. 2017, 80, 223–247. [Google Scholar]
- Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Della Pina, S.; Tambaro, S.; Memo, M.; Mastinu, A. Cannabidiol: Recent advances and new insights for neuropsychiatric disorders treatment. Life Sci. 2019, 224, 120–127. [Google Scholar] [CrossRef]
- Taylor, A.H.; Ang, C.; Bell, S.C.; Konje, J.C. The role of the endocannabinoid system in gametogenesis, implantation and early pregnancy. Hum. Reprod. Update 2007, 13, 501–513. [Google Scholar] [CrossRef]
- Gillies, R.; Lee, K.; Vanin, S.; Laviolette, S.R.; Holloway, A.C.; Arany, E.; Hardy, D.B. Maternal exposure to Δ9-tetrahydrocannabinol impairs female offspring glucose homeostasis and endocrine pancreatic development in the rat. Reprod. Toxicol. 2020, 94, 84–91. [Google Scholar] [CrossRef] [PubMed]
- González-Mariscal, I.; Krzysik-Walker, S.M.; Doyle, M.E.; Liu, Q.R.; Cimbro, R.; Santa-Cruz Calvo, S.; Ghosh, S.; Cieala, A.; Moaddel, R.; Carlson, O.D.; et al. Human CB1 Receptor Isoforms, present in Hepatocytes and β-cells, are Involved in Regulating Metabolism. Sci. Rep. 2016, 6, 33302. [Google Scholar] [CrossRef]
- Joshi, N.; Onaivi, E.S. Endocannabinoid System Components: Overview and Tissue Distribution. Adv. Exp. Med. Biol. 2019, 1162, 1–12. [Google Scholar]
- Deiana, S. Potential Medical Uses of Cannabigerol: A Brief Overview. In Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis, and Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 958–967. ISBN 9780128008270. [Google Scholar]
- Bodine, M.; Kemp, A.K. Medical Cannabis Use in Oncology; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Donvito, G.; Nass, S.R.; Wilkerson, J.L.; Curry, Z.A.; Schurman, L.D.; Kinsey, S.G.; Lichtman, A.H. The Endogenous Cannabinoid System: A Budding Source of Targets for Treating Inflammatory and Neuropathic Pain. Neuropsychopharmacology 2018, 43, 52–79. [Google Scholar] [CrossRef]
- del Río, C.; Millán, E.; García, V.; Appendino, G.; DeMesa, J.; Muñoz, E. The endocannabinoid system of the skin. A potential approach for the treatment of skin disorders. Biochem. Pharmacol. 2018, 157, 122–133. [Google Scholar] [CrossRef]
- Alkilani, A.; McCrudden, M.T.; Donnelly, R. Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Barrier Properties of the Stratum Corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef]
- Roberts, M.S. Targeted drug delivery to the skin and deeper tissues: Role of physiology, solute structure and disease. Clin. Exp. Pharmacol. Physiol. 1997, 24, 874–879. [Google Scholar] [CrossRef] [PubMed]
- Khabir, Z.; Partalis, C.; Panchal, J.V.; Deva, A.; Khatri, A.; Garcia-Bennett, A. Enhanced Skin Penetration of Cannabidiol Using Organosilane Particles as Transdermal Delivery Vehicles. Pharmaceutics 2023, 15, 798. [Google Scholar] [CrossRef]
- Tijani, A.O.; Thakur, D.; Mishra, D.; Frempong, D.; Chukwunyere, U.I.; Puri, A. Delivering therapeutic cannabinoids via skin: Current state and future perspectives. J. Control. Release 2021, 334, 427–451. [Google Scholar] [CrossRef]
- Landis, E.; Davis, S.; Feldman, S.; Taylor, S. Complementary and Alternative Medicine Use in Dermatology in the United States. J. Altern. Complement. Med. 2014, 20, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Nickles, M.; Lio, P. Cannabinoids in Dermatology: Hope or Hype? Cannabis Cannabinoid Res. 2020, 5, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Shao, K.; Stewart, C.; Grant-Kels, J.M. Cannabis and the skin. Clin. Dermatol. 2021, 39, 784–795. [Google Scholar] [CrossRef]
- House, W. Global Report on Atopic Dermatitis 2022; The Global Atopic Dermatitis Atlas (GADA): Madagascar, Tunisia, 2022. [Google Scholar]
- Zuberbier, T.; Lötvall, J.; Simoens, S.; Subramanian, S.V.; Church, M.K. Economic burden of inadequate management of allergic diseases in the European Union: A GA 2 LEN review. Allergy 2014, 69, 1275–1279. [Google Scholar] [CrossRef]
- Dickel, H. Management of contact dermatitis. Allergo J. Int. 2023, 32, 57–76. [Google Scholar] [CrossRef]
- Michalek, I.M.; Loring, B.; John, S.M. World Health Organization. Global Report on Psoriasis; World Health Organization: Geneva, Switzerland, 2019; ISBN 9789241565189. [Google Scholar]
- Nair, P.A.; Badri, T. Psoriasis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Benson, H.; Grice, J.; Mohammed, Y.; Namjoshi, S.; Roberts, M. Topical and Transdermal Drug Delivery: From Simple Potions to Smart Technologies. Curr. Drug Deliv. 2019, 16, 444–460. [Google Scholar] [CrossRef]
- Korkina, L. Metabolic and redox barriers in the skin exposed to drugs and xenobiotics. Expert Opin. Drug Metab. Toxicol. 2016, 12, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Hewitt, N.J.; Merk, H.F.; Reisinger, K. Dermal Xenobiotic Metabolism: A Comparison between Native Human Skin, Four in vitro Skin Test Systems and a Liver System. Ski. Pharmacol. Physiol. 2014, 27, 263–275. [Google Scholar] [CrossRef]
- Gorzelanny, C.; Meß, C.; Schneider, S.; Huck, V.; Brandner, J. Skin Barriers in Dermal Drug Delivery: Which Barriers Have to Be Overcome and How Can We Measure Them? Pharmaceutics 2020, 12, 684. [Google Scholar] [CrossRef]
- Eyerich, S.; Eyerich, K.; Traidl-Hoffmann, C.; Biedermann, T. Cutaneous Barriers and Skin Immunity: Differentiating A Connected Network. Trends Immunol. 2018, 39, 315–327. [Google Scholar] [CrossRef]
- Agarwal, R.; Prakash, O.; Vyas, S. The pilosebaceous unit: A pivotal route for topical drug delivery. Methods Find. Exp. Clin. Pharmacol. 2000, 22, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Kirk, R.D.; Akanji, T.; Li, H.; Shen, J.; Allababidi, S.; Seeram, N.P.; Bertin, M.J.; Ma, H. Evaluations of Skin Permeability of Cannabidiol and Its Topical Formulations by Skin Membrane-Based Parallel Artificial Membrane Permeability Assay and Franz Cell Diffusion Assay. Med. Cannabis Cannabinoids 2022, 5, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Oláh, A.; Toth, B.; Borbíró, I.; Sugawara, K.; Szöllõsi, A.; Czifra, G.; Pal, B.; Ambrus, L.; Hundt, J.; Camera, E.; et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J. Clin. Investig. 2014, 124, 3713–3724. [Google Scholar] [CrossRef] [PubMed]
- Cobo-Golpe, M.; de-Castro-Ríos, A.; Cruz, A.; López-Rivadulla, M.; Lendoiro, E. Determination and Distribution of Cannabinoids in Nail and Hair Samples. J. Anal. Toxicol. 2021, 45, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Pitliuk, R.; Coelho, C.; Fucci, T. Unexpected improvement of hyperhidrosis with cannabidiol. Einstein 2022, 20, eRC5795. [Google Scholar] [CrossRef]
- Kaemmerer, T.; Clanner-Engelshofen, B.M.; Lesmeister, T.; French, L.E.; Reinholz, M. Cannabinoids in hyperhidrosis. J. Dermatolog. Treat. 2023, 34, 2127308. [Google Scholar] [CrossRef] [PubMed]
- Mounessa, J.S.; Siegel, J.A.; Dunnick, C.A.; Dellavalle, R.P. The role of cannabinoids in dermatology. J. Am. Acad. Dermatol. 2017, 77, 188–190. [Google Scholar] [CrossRef]
- Tóth, K.; Ádám, D.; Bíró, T.; Oláh, A. Cannabinoid Signaling in the Skin: Therapeutic Potential of the “C(ut)annabinoid” System. Molecules 2019, 24, 918. [Google Scholar] [CrossRef]
- Tüting, T.; Gaffal, E. Regulatory Role of Cannabinoids for Skin Barrier Functions and Cutaneous Inflammation. In Handbook of Cannabis and Related Pathologies; Elsevier: Amsterdam, The Netherlands, 2017; pp. 543–549. [Google Scholar]
- Kendall, A.C.; Pilkington, S.M.; Massey, K.A.; Sassano, G.; Rhodes, L.E.; Nicolaou, A. Distribution of Bioactive Lipid Mediators in Human Skin. J. Investig. Dermatol. 2015, 135, 1510–1520. [Google Scholar] [CrossRef]
- Gęgotek, A.; Biernacki, M.; Ambrożewicz, E.; Surażyński, A.; Wroński, A.; Skrzydlewska, E. The cross-talk between electrophiles, antioxidant defence and the endocannabinoid system in fibroblasts and keratinocytes after UVA and UVB irradiation. J. Dermatol. Sci. 2016, 81, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M.; Di Rienzo, M.; Battista, N.; Gasperi, V.; Guerrieri, P.; Rossi, A.; Finazzi-Agrò, A. The Endocannabinoid System in Human Keratinocytes. J. Biol. Chem. 2003, 278, 33896–33903. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, A.; Pasquariello, N.; Barcaroli, D.; Maccarrone, M. Anandamide Regulates Keratinocyte Differentiation by Inducing DNA Methylation in a CB1 Receptor-dependent Manner. J. Biol. Chem. 2008, 283, 6005–6012. [Google Scholar] [CrossRef]
- Ständer, S.; Schmelz, M.; Metze, D.; Luger, T.; Rukwied, R. Distribution of cannabinoid receptor 1 (CB1) and 2 (CB2) on sensory nerve fibers and adnexal structures in human skin. J. Dermatol. Sci. 2005, 38, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Pagano, C.; Navarra, G.; Coppola, L.; Avilia, G.; Bifulco, M.; Laezza, C. Cannabinoids: Therapeutic Use in Clinical Practice. Int. J. Mol. Sci. 2022, 23, 3344. [Google Scholar] [CrossRef] [PubMed]
- Eagleston, L.R.M.; Kalani, N.K.; Patel, R.R.; Flaten, H.K.; Dunnick, C.A.; Dellavalle, R.P. Cannabinoids in dermatology: A scoping review. Dermatol. Online J. 2018, 24, 1–17. [Google Scholar] [CrossRef]
- Wilkinson, J.D.; Williamson, E.M. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J. Dermatol. Sci. 2007, 45, 87–92. [Google Scholar] [CrossRef]
- Ramot, Y.; Sugawara, K.; Zákány, N.; Tóth, B.I.; Bíró, T.; Paus, R. A novel control of human keratin expression: Cannabinoid receptor 1-mediated signaling down-regulates the expression of keratins K6 and K16 in human keratinocytes in vitro and in situ. PeerJ 2013, 1, e40. [Google Scholar] [CrossRef]
- Borda, L.J.; Wikramanayake, T.C. Seborrheic Dermatitis and Dandruff: A Comprehensive Review. J. Clin. Investig. Dermatol. 2015, 3. [Google Scholar] [CrossRef]
- Gupta, A.K.; Richardson, M.; Paquet, M. Systematic Review of Oral Treatments for Seborrheic Dermatitis. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 16–26. [Google Scholar] [CrossRef]
- Bolognia, J.L.; Schaffer, J.V.; Cerroni, L. Dermatology: 2-Volume Set, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 2, pp. 425–478. [Google Scholar]
- Namazi, M. Cannabinoids, loratadine and allopurinol as novel additions to the antipsoriatic ammunition. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Norooznezhad, A.H.; Norooznezhad, F. Cannabinoids: Possible agents for treatment of psoriasis via suppression of angiogenesis and inflammation. Med. Hypotheses 2017, 99, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Maya-Rico, A.M.; Londoño-García, Á.; Palacios-Barahona, A.U.; Jimenez-Tamayo, S.B.; Muriel-Lopera, E. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An. Bras. Dermatol. 2021, 96, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Escobar, L.G.; Mora-Ochoa, H.; Vargas Villanueva, A.; Spineli, L.; Sanclemente, G.; Couban, R.; García, E.; Chapman, E.; Yepes-Nuñez, J.J. Effectiveness and adverse events of topical and allergen immunotherapy for atopic dermatitis: A systematic review and network meta-analysis protocol. Syst. Rev. 2020, 9, 222. [Google Scholar] [CrossRef] [PubMed]
- Giam, Y.C.; Hebert, A.A.; Dizon, M.V.; Van Bever, H.; Tiongco-Recto, M.; Kim, K.-H.; Soebono, H.; Munasir, Z.; Diana, I.A.; Luk, D.C.K. A review on the role of moisturizers for atopic dermatitis. Asia Pac. Allergy 2016, 6, 120–128. [Google Scholar] [CrossRef]
- Coondoo, A.; Phiske, M.; Verma, S.; Lahiri, K. Side-effects of topical steroids: A long overdue revisit. Indian Dermatol. Online J. 2014, 5, 416. [Google Scholar] [CrossRef]
- Varga, E.; Kemény, L. The use of calcineurin inhibitors in atopic dermatitis. Bőrgyógyászati és Venerológiai Szle 2017, 93, 235–239. [Google Scholar] [CrossRef]
- Carr, W.W. Topical Calcineurin Inhibitors for Atopic Dermatitis: Review and Treatment Recommendations. Pediatr. Drugs 2013, 15, 303–310. [Google Scholar] [CrossRef]
- Frazier, W.; Bhardwaj, N. Atopic Dermatitis: Diagnosis and Treatment. Am. Fam. Physician 2020, 101, 590–598. [Google Scholar] [PubMed]
- Patrizi, A.; Raone, B.; Ravaioli, G.M. Management of atopic dermatitis: Safety and efficacy of phototherapy. Clin. Cosmet. Investig. Dermatol. 2015, 8, 511–520. [Google Scholar] [CrossRef]
- Megna, M.; Napolitano, M.; Patruno, C.; Villani, A.; Balato, A.; Monfrecola, G.; Ayala, F.; Balato, N. Systemic Treatment of Adult Atopic Dermatitis: A Review. Dermatol. Ther. 2017, 7, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Saraswat, A. Contact allergy to topical corticosteroids and sunscreens. Indian J. Dermatol. Venereol. Leprol. 2012, 78, 552. [Google Scholar] [CrossRef] [PubMed]
- FnF Research Global Report on Seborrheic Dermatitis Market Size & Share Worth USD 5 Billion, to Record a 11.1% CAGR by 2030. Available online: https://www.prnewswire.co.uk/news-releases/global-report-on-seborrheic-dermatitis-market-size--share-worth-usd-5-billion-to-record-a-11-1-cagr-by-2030--seborrheic-dermatitis-industry-trends-segmentation-analysis--forecast-by-fnf-301736378.html (accessed on 8 April 2023).
- Curkova, A.K.; Simaljakova, M. Seborrheic Dermatitis. In European Handbook of Dermatological Treatments; Springer: Berlin/Heidelberg, Germany, 2015; pp. 867–877. [Google Scholar]
- Gaffal, E.; Glodde, N.; Jakobs, M.; Bald, T.; Tüting, T. Cannabinoid 1 receptors in keratinocytes attenuate fluorescein isothiocyanate-induced mouse atopic-like dermatitis. Exp. Dermatol. 2014, 23, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Eberlein, B.; Eicke, C.; Reinhardt, H.-W.; Ring, J. Adjuvant treatment of atopic eczema: Assessment of an emollient containing N-palmitoylethanolamine (ATOPA study). J. Eur. Acad. Dermatol. Venereol. 2007, 22, 73–82. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, B.; Park, B.M.; Jeon, J.E.; Lee, S.H.; Mann, S.; Ahn, S.K.; Hong, S.-P.; Jeong, S.K. Topical cannabinoid receptor 1 agonist attenuates the cutaneous inflammatory responses in oxazolone-induced atopic dermatitis model. Int. J. Dermatol. 2015, 54, e401–e408. [Google Scholar] [CrossRef]
- Petrosino, S.; Verde, R.; Vaia, M.; Allarà, M.; Iuvone, T.; Di Marzo, V. Anti-inflammatory Properties of Cannabidiol, a Nonpsychotropic Cannabinoid, in Experimental Allergic Contact Dermatitis. J. Pharmacol. Exp. Ther. 2018, 365, 652–663. [Google Scholar] [CrossRef]
- Gupta, A.K.; Talukder, M. Cannabinoids for skin diseases and hair regrowth. J. Cosmet. Dermatol. 2021, 20, 2703–2711. [Google Scholar] [CrossRef]
- Dobrosi, N.; Tóth, B.I.; Nagy, G.; Dózsa, A.; Géczy, T.; Nagy, L.; Zouboulis, C.C.; Paus, R.; Kovács, L.; Bíró, T. Endocannabinoids enhance lipid synthesis and apoptosis of human sebocytes via cannabinoid receptor-2-mediated signaling. FASEB J. 2008, 22, 3685–3695. [Google Scholar] [CrossRef]
- Ali, A.; Akhtar, N. The safety and efficacy of 3% Cannabis seeds extract cream for reduction of human cheek skin sebum and erythema content. Pak. J. Pharm. Sci. 2015, 28, 1389–1395. [Google Scholar]
- Gaffal, E.; Cron, M.; Glodde, N.; Tüting, T. Anti-inflammatory activity of topical THC in DNFB-mediated mouse allergic contact dermatitis independent of CB1 and CB2 receptors. Allergy 2013, 68, 994–1000. [Google Scholar] [CrossRef]
- Soliman, E.; Van Dross, R. Anandamide-induced endoplasmic reticulum stress and apoptosis are mediated by oxidative stress in non-melanoma skin cancer: Receptor-independent endocannabinoid signaling. Mol. Carcinog. 2016, 55, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Soliman, E.; Henderson, K.L.; Danell, A.S.; Van Dross, R. Arachidonoyl-ethanolamide activates endoplasmic reticulum stress-apoptosis in tumorigenic keratinocytes: Role of cyclooxygenase-2 and novel J-series prostamides. Mol. Carcinog. 2016, 55, 117–130. [Google Scholar] [CrossRef]
- Ständer, S.; Reinhardt, H.W.; Luger, T.A. Topical cannabinoid agonists. An effective new possibility for treating chronic pruritus. Der Hautarzt 2006, 57, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Szepietowski, J.C.; Szepietowski, T.; Reich, A. Efficacy and tolerance of the cream containing structured physiological lipids with endocannabinoids in the treatment of uremic pruritus: A preliminary study. Acta Dermatovenerol. Croat. 2005, 13, 97–103. [Google Scholar] [PubMed]
- Broughton, G.; Janis, J.E.; Attinger, C.E. The Basic Science of Wound Healing. Plast. Reconstr. Surg. 2006, 117, 12S–34S. [Google Scholar] [CrossRef]
- Ellis, S.; Lin, E.J.; Tartar, D. Immunology of Wound Healing. Curr. Dermatol. Rep. 2018, 7, 350–358. [Google Scholar] [CrossRef]
- Zhao, Z.; Guan, D.; Liu, W.; Wang, T.; Fan, Y.; Cheng, Z.; Zheng, J.; Hu, G. Expression of cannabinoid receptor I during mice skin incised wound healing course. Fa Yi Xue Za Zhi 2010, 26, 241–245. [Google Scholar]
- Zheng, J.-L.; Yu, T.-S.; Li, X.-N.; Fan, Y.-Y.; Ma, W.-X.; Du, Y.; Zhao, R.; Guan, D.-W. Cannabinoid receptor type 2 is time-dependently expressed during skin wound healing in mice. Int. J. Leg. Med. 2012, 126, 807–814. [Google Scholar] [CrossRef]
- Wang, L.-L.; Zhao, R.; Li, J.-Y.; Li, S.-S.; Liu, M.; Wang, M.; Zhang, M.-Z.; Dong, W.-W.; Jiang, S.-K.; Zhang, M.; et al. Pharmacological activation of cannabinoid 2 receptor attenuates inflammation, fibrogenesis, and promotes re-epithelialization during skin wound healing. Eur. J. Pharmacol. 2016, 786, 128–136. [Google Scholar] [CrossRef]
- Bort, A.; Alvarado-Vazquez, P.A.; Moracho-Vilrriales, C.; Virga, K.G.; Gumina, G.; Romero-Sandoval, A.; Asbill, S. Effects of JWH015 in cytokine secretion in primary human keratinocytes and fibroblasts and its suitability for topical/transdermal delivery. Mol. Pain 2017, 13. [Google Scholar] [CrossRef]
- Ramot, Y.; Oláh, A.; Paus, R. Cover Image: Neuroendocrine treatment of inherited keratin disorders by cannabinoids? Br. J. Dermatol. 2018, 178, 1469. [Google Scholar] [CrossRef] [PubMed]
- Sangiovanni, E.; Fumagalli, M.; Pacchetti, B.; Piazza, S.; Magnavacca, A.; Khalilpour, S.; Melzi, G.; Martinelli, G.; Dell’Agli, M. Cannabis sativa L. extract and cannabidiol inhibit in vitro mediators of skin inflammation and wound injury. Phyther. Res. 2019, 33, 2083–2093. [Google Scholar] [CrossRef] [PubMed]
- Bolzinger, M.-A.; Briançon, S.; Pelletier, J.; Chevalier, Y. Penetration of drugs through skin, a complex rate-controlling membrane. Curr. Opin. Colloid Interface Sci. 2012, 17, 156–165. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Kweon, D.K.; Park, H.J. Investigations on skin permeation of hyaluronic acid based nanoemulsion as transdermal carrier. Carbohydr. Polym. 2011, 86, 837–843. [Google Scholar] [CrossRef]
- Larese Filon, F.; Mauro, M.; Adami, G.; Bovenzi, M.; Crosera, M. Nanoparticles skin absorption: New aspects for a safety profile evaluation. Regul. Toxicol. Pharmacol. 2015, 72, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Larrañeta, E.; Lutton, R.E.M.; Woolfson, A.D.; Donnelly, R.F. Microneedle arrays as transdermal and intradermal drug delivery systems: Materials science, manufacture and commercial development. Mater. Sci. Eng. R Rep. 2016, 104, 1–32. [Google Scholar] [CrossRef]
- Moser, K. Passive skin penetration enhancement and its quantification in vitro. Eur. J. Pharm. Biopharm. 2001, 52, 103–112. [Google Scholar] [CrossRef]
- Godin, B.; Touitou, E. Transdermal skin delivery: Predictions for humans from in vivo, ex vivo and animal models. Adv. Drug Deliv. Rev. 2007, 59, 1152–1161. [Google Scholar] [CrossRef]
- Goyal, R.; Macri, L.K.; Kaplan, H.M.; Kohn, J. Nanoparticles and nanofibers for topical drug delivery. J. Control. Release 2016, 240, 77–92. [Google Scholar] [CrossRef]
- Lucas, C.J.; Galettis, P.; Schneider, J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br. J. Clin. Pharmacol. 2018, 84, 2477–2482. [Google Scholar] [CrossRef]
- Stella, B.; Baratta, F.; Della Pepa, C.; Arpicco, S.; Gastaldi, D.; Dosio, F. Cannabinoid Formulations and Delivery Systems: Current and Future Options to Treat Pain. Drugs 2021, 81, 1513–1557. [Google Scholar] [CrossRef] [PubMed]
- Madej, K.; Chmiołek, A.; Szlachta, K.; Piekoszewski, W. HPLC-DAD Analysis of Hemp Oil Supplements for Determination of Four Cannabinoids: Cannabidiol, Cannabidiolic Acid, Cannabinol and Delta 9-Tetrahydrocannabinol. Separations 2021, 8, 227. [Google Scholar] [CrossRef]
- Toprak, Ş. Theoretical calculation of some chemical properties of the cannabidiol (CBD) molecule. Int. J. Sci. Lett. 2021, 3, 129–142. [Google Scholar] [CrossRef]
- Thomas, B.F.; Compton, D.R.; Martin, B.R. Characterization of the lipophilicity of natural and synthetic analogs of delta 9-tetrahydrocannabinol and its relationship to pharmacological potency. J. Pharmacol. Exp. Ther. 1990, 255, 624–630. [Google Scholar]
- Furqan, T.; Batool, S.; Habib, R.; Shah, M.; Kalasz, H.; Darvas, F.; Kuca, K.; Nepovimova, E.; Batool, S.; Nurulain, S.M. Cannabis Constituents and Acetylcholinesterase Interaction: Molecular Docking, In Vitro Studies and Association with CNR1 rs806368 and ACHE rs17228602. Biomolecules 2020, 10, 758. [Google Scholar] [CrossRef]
- ChemSpider. Cannabigerol. Available online: http://www.chemspider.com/Chemical-Structure.4474921.html (accessed on 12 January 2023).
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic Properties of Cannabidiol at 5-HT1a Receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef]
- Costa, B.; Giagnoni, G.; Franke, C.; Trovato, A.E.; Colleoni, M. Vanilloid TRPV1 receptor mediates the antihyperalgesic effect of the nonpsychoactive cannabinoid, cannabidiol, in a rat model of acute inflammation. Br. J. Pharmacol. 2004, 143, 247–250. [Google Scholar] [CrossRef]
- Blaskovich, M.A.T.; Kavanagh, A.M.; Elliott, A.G.; Zhang, B.; Ramu, S.; Amado, M.; Lowe, G.J.; Hinton, A.O.; Pham, D.M.T.; Zuegg, J.; et al. The antimicrobial potential of cannabidiol. Commun. Biol. 2021, 4, 7. [Google Scholar] [CrossRef]
- Morakul, B.; Junyaprasert, V.B.; Sakchaisri, K.; Teeranachaideekul, V. Cannabidiol-Loaded Nanostructured Lipid Carriers (NLCs) for Dermal Delivery: Enhancement of Photostability, Cell Viability, and Anti-Inflammatory Activity. Pharmaceutics 2023, 15, 537. [Google Scholar] [CrossRef]
- Grifoni, L.; Vanti, G.; Donato, R.; Sacco, C.; Bilia, A.R. Promising Nanocarriers to Enhance Solubility and Bioavailability of Cannabidiol for a Plethora of Therapeutic Opportunities. Molecules 2022, 27, 6070. [Google Scholar] [CrossRef]
- Huestis, M.A. Pharmacokinetics and Metabolism of the Plant Cannabinoids, Δ 9-Tetrahydrocannibinol, Cannabidiol and Cannabinol. In Cannabinoids. Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2005; pp. 657–690. [Google Scholar]
- Zendulka, O.; Dovrtělová, G.; Nosková, K.; Turjap, M.; Šulcová, A.; Hanuš, L.; Juřica, J. Cannabinoids and Cytochrome P450 Interactions. Curr. Drug Metab. 2016, 17, 206–226. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration Marinol (Dronabinol) Technical Sheet. Nda 18-651/s-021. 2004, 3. Available online: https://digitalcommons.wcl.american.edu/cgi/viewcontent.cgi?article=2087&context=aulr (accessed on 21 March 2023).
- Harvey, D.J.; Samara, E.; Mechoulam, R. Urinary metabolites of cannabidiol in dog, rat and man and their identification by gas chromatography—Mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 1991, 562, 299–322. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.; Yamaori, S.; Takeda, S.; Yamamoto, I.; Watanabe, K. Identification of cytochrome P450 enzymes responsible for metabolism of cannabidiol by human liver microsomes. Life Sci. 2011, 89, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Geffrey, A.L.; Pollack, S.F.; Bruno, P.L.; Thiele, E.A. Drug-drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia 2015, 56, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- Flannery, L.E.; Kerr, D.M.; Finn, D.P.; Roche, M. FAAH inhibition attenuates TLR3-mediated hyperthermia, nociceptive- and anxiety-like behaviour in female rats. Behav. Brain Res. 2018, 353, 11–20. [Google Scholar] [CrossRef]
- Zanfirescu, A.; Nitulescu, G.; Mihai, D.P.; Nitulescu, G.M. Identifying FAAH Inhibitors as New Therapeutic Options for the Treatment of Chronic Pain through Drug Repurposing. Pharmaceuticals 2021, 15, 38. [Google Scholar] [CrossRef]
- Hill, M.N.; Kumar, S.A.; Filipski, S.B.; Iverson, M.; Stuhr, K.L.; Keith, J.M.; Cravatt, B.F.; Hillard, C.J.; Chattarji, S.; McEwen, B.S. Disruption of fatty acid amide hydrolase activity prevents the effects of chronic stress on anxiety and amygdalar microstructure. Mol. Psychiatry 2013, 18, 1125–1135. [Google Scholar] [CrossRef]
- Schmidt, M.E.; Liebowitz, M.R.; Stein, M.B.; Grunfeld, J.; Van Hove, I.; Simmons, W.K.; Van Der Ark, P.; Palmer, J.A.; Saad, Z.S.; Pemberton, D.J.; et al. The effects of inhibition of fatty acid amide hydrolase (FAAH) by JNJ-42165279 in social anxiety disorder: A double-blind, randomized, placebo-controlled proof-of-concept study. Neuropsychopharmacology 2021, 46, 1004–1010. [Google Scholar] [CrossRef]
- Pacifici, R.; Marchei, E.; Salvatore, F.; Guandalini, L.; Busardò, F.P.; Pichini, S. Evaluation of long-term stability of cannabinoids in standardized preparations of cannabis flowering tops and cannabis oil by ultra-high-performance liquid chromatography tandem mass spectrometry. Clin. Chem. Lab. Med. 2018, 56, 94–96. [Google Scholar] [CrossRef]
- Fairbairn, J.W.; Liebmann, J.A.; Rowan, M.G. The stability of cannabis and its preparations on storage. J. Pharm. Pharmacol. 2011, 28, 1–7. [Google Scholar] [CrossRef]
- Agurell, S.; Carlsson, S.; Lindgren, J.E.; Ohlsson, A.; Gillespie, H.; Hollister, L. Interactions ofΔ 11-tetrahydrocannabinol with cannabinol and cannabidiol following oral administration in man. Assay of cannabinol and cannabidiol by mass fragmentographywith cannabinol and cannabidiol following oral administration in man. Assay of cannabinol and cannabidiol by mass fragmentography. Experientia 1981, 37, 1090–1092. [Google Scholar] [CrossRef] [PubMed]
- Eichler, M.; Spinedi, L.; Unfer-Grauwiler, S.; Bodmer, M.; Surber, C.; Luedi, M.; Drewe, J. Heat Exposure of Cannabis sativa Extracts Affects the Pharmacokinetic and Metabolic Profile in Healthy Male Subjects. Planta Med. 2012, 78, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Control. Release 2014, 190, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Cuevas, G.; Martin-Fardon, R.; Kerr, T.M.; Stouffer, D.G.; Parsons, L.H.; Hammell, D.C.; Banks, S.L.; Stinchcomb, A.L.; Weiss, F. Unique treatment potential of cannabidiol for the prevention of relapse to drug use: Preclinical proof of principle. Neuropsychopharmacology 2018, 43, 2036–2045. [Google Scholar] [CrossRef]
- Liput, D.J.; Hammell, D.C.; Stinchcomb, A.L.; Nixon, K. Transdermal delivery of cannabidiol attenuates binge alcohol-induced neurodegeneration in a rodent model of an alcohol use disorder. Pharmacol. Biochem. Behav. 2013, 111, 120–127. [Google Scholar] [CrossRef]
- Stinchcomb, A.; Golinski, M.; Hammell, D.; Alltranz, H.J. Products of Tetrahydrocannabinol, Compositions Comprising Prodrugs of Tetrahydrocannabinol and Methods of Using the Same. Patent US20090143462, 26 July 2019. [Google Scholar]
- Doroudian, A.; Frankham, P. Biphasix Cannabinoid Delivery. Patent WO2018213932, 25 May 2018. [Google Scholar]
- Gutterman, D.; Sebree, T.; Smith, T.; Messenheimer, J. Synthetic Transdermal Cannabidiol for the Treatment of Focal Epilepsy in Adults. Patent US20190083388, 19 March 2019. [Google Scholar]
- Bonn-Miller, M.; Tich, N.; Gutterman, D.; Messenheimer, J.; Sebree, T. Treatment of Fragile X Syndrome with Cannabidiol. Patent US10213390, 26 February 2019. [Google Scholar]
- ME, S. Formulations of Argan Oil and Cannabidiol for Treating Inflammatory Disorders Including Arthritis. WO2017160923A121, 21 September 2017. [Google Scholar]
- Cairns, E.A.; Baldridge, W.H.; Kelly, M.E.M. The Endocannabinoid System as a Therapeutic Target in Glaucoma. Neural Plast. 2016, 2016, 9364091. [Google Scholar] [CrossRef]
- Adelli, G.R.; Bhagav, P.; Taskar, P.; Hingorani, T.; Pettaway, S.; Gul, W.; ElSohly, M.A.; Repka, M.A.; Majumdar, S. Development of a Δ9-Tetrahydrocannabinol Amino Acid-Dicarboxylate Prodrug With Improved Ocular Bioavailability. Investig. Opthalmology Vis. Sci. 2017, 58, 2167. [Google Scholar] [CrossRef]
- Radwan-Pragłowska, J.; Janus, Ł.; Piątkowski, M.; Sierakowska, A.; Szajna, E.; Matýsek, D.; Bogdał, D. Development of Stimuli-Responsive Chitosan/ZnO NPs Transdermal Systems for Controlled Cannabidiol Delivery. Polymers 2021, 13, 211. [Google Scholar] [CrossRef]
- Park, C.; Zuo, J.; Somayaji, V.; Lee, B.-J.; Löbenberg, R. Development of a novel cannabinoid-loaded microemulsion towards an improved stability and transdermal delivery. Int. J. Pharm. 2021, 604, 120766. [Google Scholar] [CrossRef]
- Sharkawy, A.; Silva, A.M.; Rodrigues, F.; Barreiro, F.; Rodrigues, A. Pickering emulsions stabilized with chitosan/collagen peptides nanoparticles as green topical delivery vehicles for cannabidiol (CBD). Colloids Surf. A Physicochem. Eng. Asp. 2021, 631, 127677. [Google Scholar] [CrossRef]
- Wright, S.; Wilkhu, J. Oral Cannabinoid Formulations. Patent WO2018150182, 23 August 2018. [Google Scholar]
- Wilkhu, J.; Bender, J. GW Research Limited. Cannabinoid Formulations. Patent WO2018002665, 4 January 2018. [Google Scholar]
- Wilkhu, J.; Johan, B. Oral Pharmaceutical Formulation Comprising Cannabinoids and Poloxamer. Patent WO2019135077A1, 11 July 2019. [Google Scholar]
- Hershberger, P.; Arlen, P. Cannabinoid Conjugate Molecules. Patent WO2020263888A1, 30 December 2020. [Google Scholar]
- Patrician, A.; Versic-Bratincevic, M.; Mijacika, T.; Banic, I.; Marendic, M.; Sutlović, D.; Dujić, Ž.; Ainslie, P.N. Examination of a New Delivery Approach for Oral Cannabidiol in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Pharmacokinetics Study. Adv. Ther. 2019, 36, 3196–3210. [Google Scholar] [CrossRef]
- Macleman, P.D.; Mavlianov, M. Solid Self-Emulsifying Pharmaceutical Compositions. WO2020024009A1, 06 February 2020. [Google Scholar]
- Mavlianov, M.; Macleman, P.D.R. Free-Flowing Powder Compositions. WO2020024011A1, 06 February 2020. [Google Scholar]
- Anastassov, G.; Changoer, L. Chewing Gum Composition Comprising Cannabinoids and Opioid Agonists and/or Antagonists. Patent WO2018075665A1, 26 April 2018. [Google Scholar]
- Williams, N.N.B.; Ewell, T.R.; Abbotts, K.S.S.; Harms, K.J.; Woelfel, K.A.; Dooley, G.P.; Weir, T.L.; Bell, C. Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability. Pharmaceuticals 2021, 14, 35. [Google Scholar] [CrossRef] [PubMed]
- Knaub, K.; Sartorius, T.; Dharsono, T.; Wacker, R.; Wilhelm, M.; Schön, C. A Novel Self-Emulsifying Drug Delivery System (SEDDS) Based on VESIsorb® Formulation Technology Improving the Oral Bioavailability of Cannabidiol in Healthy Subjects. Molecules 2019, 24, 2967. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, P.; Avidan-Shlomovich, S.; Shoken, D.; Salzman, A.L. Cannabinoid Derivatives and Conjugates and Uses Thereof. Patent WO2019159168A1, 22 August 2019. [Google Scholar]
- NCT03773796; Nabilone for Non-Motor Symptoms in Parkinson’s Disease: An Open-Label Study to Evaluate Longterm Safety and Efficacy. U.S. National Library of Medicine: Bethesda, MD, USA, 2021.
- Viernstein, H.; Toegel, S.; Schueller, R. Fast Disintegrating Compositions Comprising Nabilone and Randomly Methylated β-Cyclodextrin. Patent WO2012069591, 31 May 2012. [Google Scholar]
- Kingsley, K.; Lee, S.; Greenbaum, E. Cannabinoid Formulations with Improved Solubility. Patent US20190030170, 31 January 2019. [Google Scholar]
- Temtsin-Krayz, G.; Glozman, S.; Kazhdan, P. Pharmaceutical Compositions for Transmucosal Delivery. WO2017072774A1, 04 May 2017. [Google Scholar]
- Bryson, N.; Sharma, A.C. Nasal Cannabidiol Compositions. WO2017208072A2, 11 January 2018. [Google Scholar]
- Matarazzo, A.P.; Elisei, L.M.S.; Carvalho, F.C.; Bonfílio, R.; Ruela, A.L.M.; Galdino, G.; Pereira, G.R. Mucoadhesive nanostructured lipid carriers as a cannabidiol nasal delivery system for the treatment of neuropathic pain. Eur. J. Pharm. Sci. 2021, 159, 105698. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A.; Ben Shabo, R.; Halamish, H.M. Cannabidiol-Loaded Mixed Polymeric Micelles of Chitosan/Poly(Vinyl Alcohol) and Poly(Methyl Methacrylate) for Trans-Corneal Delivery. Pharmaceutics 2021, 13, 2142. [Google Scholar] [CrossRef] [PubMed]
- Moqejwa, T.; Marimuthu, T.; Kondiah, P.P.D.; Choonara, Y.E. Development of Stable Nano-Sized Transfersomes as a Rectal Colloid for Enhanced Delivery of Cannabidiol. Pharmaceutics 2022, 14, 703. [Google Scholar] [CrossRef]
- Tapfumaneyi, P.; Imran, M.; Mohammed, Y.; Roberts, M.S. Recent advances and future prospective of topical and transdermal delivery systems. Front. Drug Deliv. 2022, 2, 957732. [Google Scholar] [CrossRef]
- Sinai, A.; Turner, Z.; Baruch, Y. Cannabis-Based Extracts and Topical Formulations for Use in Skin Disorders. US20180042890A1, 21 June 2017. [Google Scholar]
- Hossain, S. Use of Topical Formulations of Cannabinoids in the Treatment of Epidermolysis Bullosa and Related Connective Tissue Disorders. WO2017190249A1, 4 May 2017. [Google Scholar]
- Lindholst, C. Long term stability of cannabis resin and cannabis extracts. Aust. J. Forensic Sci. 2010, 42, 181–190. [Google Scholar] [CrossRef]
- Hammell, D.C.; Zhang, L.P.; Ma, F.; Abshire, S.M.; McIlwrath, S.L.; Stinchcomb, A.L.; Westlund, K.N. Transdermal cannabidiol reduces inflammation and pain-related behaviours in a rat model of arthritis. Eur. J. Pain 2016, 20, 936–948. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Potential of Nanoparticles as Permeation Enhancers and Targeted Delivery Options for Skin: Advantages and Disadvantages. Drug Des. Devel. Ther. 2020, 14, 3271–3289. [Google Scholar] [CrossRef]
- Danielsson, I.; Lindman, B. The definition of microemulsion. Colloids Surf. 1981, 3, 391–392. [Google Scholar] [CrossRef]
- Shukla, T.; Upmanyu, N.; Agrawal, M.; Saraf, S.; Saraf, S.; Alexander, A. Biomedical applications of microemulsion through dermal and transdermal route. Biomed. Pharmacother. 2018, 108, 1477–1494. [Google Scholar] [CrossRef] [PubMed]
- Egbaria, K.; Ramachandran, C.; Weiner, N. Topical Delivery of Ciclosporin: Evaluation of Various Formulations Using in vitro Diffusion Studies in Hairless Mouse Skin. Skin Pharmacol. Physiol. 1990, 3, 21–28. [Google Scholar] [CrossRef] [PubMed]
- El Maghraby, G.M.M.; Williams, A.C.; Barry, B.W. Can drug-bearing liposomes penetrate intact skin? J. Pharm. Pharmacol. 2010, 58, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Shilo-Benjamini, Y.; Cern, A.; Zilbersheid, D.; Hod, A.; Lavy, E.; Barasch, D.; Barenholz, Y. A Case Report of Subcutaneously Injected Liposomal Cannabidiol Formulation Used as a Compassion Therapy for Pain Management in a Dog. Front. Vet. Sci. 2022, 9, 892306. [Google Scholar] [CrossRef]
- Choi, M.J.; Maibach, H.I. Liposomes and Niosomes as Topical Drug Delivery Systems. Skin Pharmacol. Physiol. 2005, 18, 209–219. [Google Scholar] [CrossRef]
- Pokharkar, V.B.; Mendiratta, C.; Kyadarkunte, A.Y.; Bhosale, S.H.; Barhate, G.A. Skin delivery aspects of benzoyl peroxide-loaded solid lipid nanoparticles for acne treatment. Ther. Deliv. 2014, 5, 635–652. [Google Scholar] [CrossRef]
- Fang, J.-Y.; Fang, C.-L.; Liu, C.-H.; Su, Y.-H. Lipid nanoparticles as vehicles for topical psoralen delivery: Solid lipid nanoparticles (SLN) versus nanostructured lipid carriers (NLC). Eur. J. Pharm. Biopharm. 2008, 70, 633–640. [Google Scholar] [CrossRef]
- Ricci, M.; Puglia, C.; Bonina, F.; Di Giovanni, C.; Giovagnoli, S.; Rossi, C. Evaluation of Indomethacin Percutaneous Absorption from Nanostructured Lipid Carriers (NLC): In Vitro and In Vivo Studies. J. Pharm. Sci. 2005, 94, 1149–1159. [Google Scholar] [CrossRef]
- Garcês, A.; Amaral, M.H.; Sousa Lobo, J.M.; Silva, A.C. Formulations based on solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for cutaneous use: A review. Eur. J. Pharm. Sci. 2018, 112, 159–167. [Google Scholar] [CrossRef]
- Gado, F.; Ferrisi, R.; Polini, B.; Mohamed, K.A.; Ricardi, C.; Lucarini, E.; Carpi, S.; Domenichini, F.; Stevenson, L.A.; Rapposelli, S.; et al. Design, Synthesis, and Biological Activity of New CB2 Receptor Ligands: From Orthosteric and Allosteric Modulators to Dualsteric/Bitopic Ligands. J. Med. Chem. 2022, 65, 9918–9938. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhu, L.; Li, R.; Pang, L.; Zhu, S.; Ma, J.; Du, L.; Jin, Y. Electroporation-enhanced transdermal drug delivery: Effects of logP, pKa, solubility and penetration time. Eur. J. Pharm. Sci. 2020, 151, 105410. [Google Scholar] [CrossRef] [PubMed]
- Uritu, C.M.; Calin, M.; Maier, S.S.; Cojocaru, C.; Nicolescu, A.; Peptanariu, D.; Constantinescu, C.A.; Stan, D.; Barboiu, M.; Pinteala, M. Flexible cyclic siloxane core enhances the transfection efficiency of polyethylenimine-based non-viral gene vectors. J. Mater. Chem. B 2015, 3, 8250–8267. [Google Scholar] [CrossRef] [PubMed]
- N’Da, D. Prodrug Strategies for Enhancing the Percutaneous Absorption of Drugs. Molecules 2014, 19, 20780–20807. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.B.; Martin, G.P.; Jones, S.A.; Akomeah, F.K. Dermal and Transdermal Drug Delivery Systems: Current and Future Prospects. Drug Deliv. 2006, 13, 175–187. [Google Scholar] [CrossRef]
- Reddy, T.S.; Zomer, R.; Mantri, N. Nanoformulations as a strategy to overcome the delivery limitations of cannabinoids. Phyther. Res. 2023, 37, 1526–1538. [Google Scholar] [CrossRef]
- Kuznetsova, D.A.; Vasilieva, E.A.; Kuznetsov, D.M.; Lenina, O.A.; Filippov, S.K.; Petrov, K.A.; Zakharova, L.Y.; Sinyashin, O.G. Enhancement of the Transdermal Delivery of Nonsteroidal Anti-inflammatory Drugs Using Liposomes Containing Cationic Surfactants. ACS Omega 2022, 7, 25741–25750. [Google Scholar] [CrossRef]
- Ogunsola, O.A.; Kraeling, M.E.; Zhong, S.; Pochan, D.J.; Bronaugh, R.L.; Raghavan, S.R. Structural analysis of “flexible” liposome formulations: New insights into the skin-penetrating ability of soft nanostructures. Soft Matter 2012, 8, 10226. [Google Scholar] [CrossRef]
- Kumar, A. Transferosome: A Recent Approach for Transdermal Drug Delivery. J. Drug Deliv. Ther. 2018, 8, 100–104. [Google Scholar] [CrossRef]
- Sermsaksasithorn, P.; Asawanonda, P.; Phutrakool, P.; Ondee, T.; Payungporn, S.; Pongpirul, K.; Hirankarn, N.; Medicine, S.; Management, C.D.; Botany, P.; et al. Efficacy and Safety of Cannabis Transdermal Patch for Alleviating Psoriasis Symptoms: Protocol for a Randomized Controlled Trial (CanPatch). medRxiv 2023, 1–15. [Google Scholar] [CrossRef]
- Bunge, A.; Vecchia, B. Evaluating the Transdermal Permeability of Chemicals. In Transdermal Drug Delivery Systems; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
| Phytocannabinoid | LogP Octanol/Water | Chemical Structure |
|---|---|---|
| Cannabidiolic acid (CBDA) | 4.86 [154] | 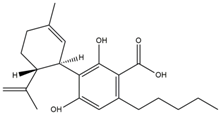 |
| Cannabidiol (CBD) | 5.79 [155] | 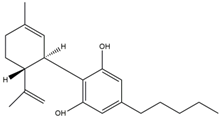 |
| Cannabinodiol (CBND) | 6.00 [156] | 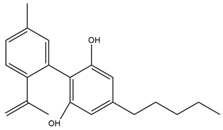 |
| Δ9-trans-tetrahidrocannabinol (THC) | 7.26 [157] | 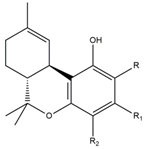 |
| Δ8-trans-tetrahidrocannabinol (Δ8-THC) | 7.4 [156] | 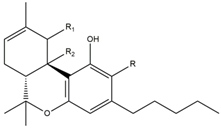 |
| Cannabigerol (CBG) | 7.47 [158] |  |
| Cannabigerolic acid (CBGA) | 8.31 [158] | 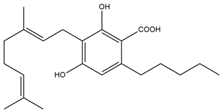 |
| Tetrahydrocannabinolic acid (THCA) | 8.41 [158] | 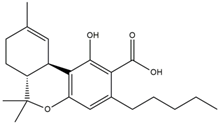 |
| Route of Absorption | Pharmaceutical Form Type | Disease | Observations | Reference |
|---|---|---|---|---|
| Transdermal | cannabinoids in a gel formulation using Transcutol® | increased penetration through the skin in various disease models | tested in vivo in two rat models of cocaine and alcohol addiction in the prevention of relapse to drug use | [179,180] |
| ZYN001—Synthetic D-glyceryl acid ester prodrug of THC | fibromyalgia and neuropathic pain | tested by Zynerba Pharmaceuticals Inc. (USA), failed to achieve the desired blood levels of THC | [181] | |
| THC encapsulated in MLVs for transdermal applications. | pain treatment | tested in vitro for skin penetration by Altum Pharmaceuticals Inc. (USA) | [182] | |
| gel containing CBD using Transcutol® | epilepsy, developmental and epileptic encephalopathy, fragile-X syndrome, and osteoarthritis | developed and tested in vitro by Zynerba Pharmaceuticals Inc. (USA) | [183,184] | |
| Transdermal | CBD and argan oil combination using Transcutol® to encapsulate CBD | pain and inflammation associated with rheumatic or arthritic inflammatory disorders | Shemanky et al. [97] reported the beneficial effects of this formulation in a group of ten volunteer patients. | [185] |
| THC prodrugs encapsulated using Transcutol® | patients with pathologically high intraocular pressure | tested by Zynerba Pharmaceuticals Inc. | [186,187] | |
| Transdermal | stimulus-responsive chitosan/ZnO nanoparticles for transdermal absorption of CBD | epilepsy | delivery system was tested in vitro for drug release properties and cytotoxicity | [188] |
| microemulsion containing THCA and CBDA | increased penetration through the skin in various disease models | tested in vitro for permeation improvement | [189] | |
| CBD emulsions stabilized with chitosan/collagen peptides nanoparticles | cosmetic applications | in vitro testing demonstrated that particle is able to penetrate the stratum corneum and diffuse into deeper layers of the skin | [190] | |
| Transmucosal (oral) | oral formulation containing one or more cannabinoid–micelle encapsulation | Alzheimer’s disease, neuropathic pain, Dravet syndrome, Lennox–Gastaut syndrome, myoclonic seizures, juvenile myoclonic epilepsy, refractory epilepsy, schizophrenia, juvenile spasms, tuberous sclerosis complex, West syndrome, anxiety | not tested in vivo | [191] |
| oral formulation containing one or more cannabinoids formulated as encapsulated micelles in the form of mucoadhesive gel, tablet, powder, liquid gel capsule, oral solution, granules, extrudates | tested in vivo on dogs by GW Research Limited (GB) | [192] | ||
| oral formulation comprising a combination of at least two cannabinoids (THC or analogies and CBD or analogues)—in the form of mucoadhesive gel, tablet, powder, liquid gel capsule, oral solution, granules, extrudates | pediatric epilepsy | tested in vivo on dogs by GW Research Limited (GB) | [193] | |
| CBD or another cannabinoid conjugates | cancer | tested by Diverse Biotech Inc. | [194] | |
| TurboCBD™ delivery technology capsules | increasing circulating CBD in various disease | pharmacokinetic profile in a double-blinded, placebo-controlled, cross-over study on 12 healthy volunteers | [195] | |
| Canemes®—solid self-emulsifying pharmaceutical compositions comprising CBD and THC combination | pain-relieving drug | not tested in vivo | [196,197] | |
| Canemes®—cannabinoids chewing gum containing CBD/ CBDV/ THC/ CBC/ CBG combined with other compounds. | multiple sclerosis-related pain and spasticity, Parkinson’s disease, dementia, restless leg syndrome, and post-herpetic neuralgia | not tested in vivo | [198] | |
| Transmucosal (oral) | Canemes®—CBD in the self-emulsifying delivery system | increases bioavailability in various disease | bioavailability tests in human | [199,200] |
| Canemes®—cannabinoid derivatives and conjugates | acute and chronic pain | not tested in vivo | [201] | |
| Canemes®—CBD cyclodextrins; cannabinoids with sulfo-alkyl-β-CD | pain, Parkinson’s disease | clinical trial | [202,203] | |
| BRCX014—CBD with poloxamer 407, carboxymethyl cellulose, and starch | various disease | not tested in vivo | [204] | |
| BRCX014—CBD sublingual formulation | cancer treatment | not tested in vivo | [108,205] | |
| BCT-521—Combination of CBD and THC | pain management with cancer patients, Fibromyalgia, symptom relief in patients with advanced cancer | not tested in vivo | [205] | |
| ZYN001—synthetic D-glyceric acid ester prodrugTHC encapsulations in multilayered lipid vesicles | fibromyalgia and neuropathic pain pain treatment | not tested in vivo | [182] | |
| Transmucosal (nasal) | nasal pharmaceutical product containing CBD plus other cannabinoids for topical application in the nasal cavity | schizophrenia | CBD+THC formulation tested on healthy volunteers | [206] |
| Transmucosal (pulmonary) | Canemes®—cannabinoid derivatives and conjugates | acute and chronic pain | not tested in vivo | [201] |
| CBD mucoadhesive nanostructured lipid carriers | neuropathic pain | tested in vivo on mice model of neuropathic pain produced a better antinociceptive effect than oral or nasal administration of simple CBD oil | [207] | |
| Canemes®—cannabinoid derivatives and conjugates | acute and chronic pain | not tested in vivo | [201] | |
| Trans-corneal | CBD-loaded mixed polymeric micelles of chitosan | degenerative and inflammatory disease of the eye | tested in vitro on human corneal epithelial cell line | [208] |
| Transmucosal (rectal) | CBD stable nanosized transfersomes as a rectal colloid | various diseases in which CBD has proven efficacy | tested in vivo for CBD release and nanoparticle delivery through the rectal membrane | [209] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filipiuc, S.-I.; Neagu, A.-N.; Uritu, C.M.; Tamba, B.-I.; Filipiuc, L.-E.; Tudorancea, I.M.; Boca, A.N.; Hâncu, M.F.; Porumb, V.; Bild, W. The Skin and Natural Cannabinoids–Topical and Transdermal Applications. Pharmaceuticals 2023, 16, 1049. https://doi.org/10.3390/ph16071049
Filipiuc S-I, Neagu A-N, Uritu CM, Tamba B-I, Filipiuc L-E, Tudorancea IM, Boca AN, Hâncu MF, Porumb V, Bild W. The Skin and Natural Cannabinoids–Topical and Transdermal Applications. Pharmaceuticals. 2023; 16(7):1049. https://doi.org/10.3390/ph16071049
Chicago/Turabian StyleFilipiuc, Silviu-Iulian, Anca-Narcisa Neagu, Cristina Mariana Uritu, Bogdan-Ionel Tamba, Leontina-Elena Filipiuc, Ivona Maria Tudorancea, Andreea Nicoleta Boca, Mădălina Florina Hâncu, Vlad Porumb, and Walther Bild. 2023. "The Skin and Natural Cannabinoids–Topical and Transdermal Applications" Pharmaceuticals 16, no. 7: 1049. https://doi.org/10.3390/ph16071049
APA StyleFilipiuc, S.-I., Neagu, A.-N., Uritu, C. M., Tamba, B.-I., Filipiuc, L.-E., Tudorancea, I. M., Boca, A. N., Hâncu, M. F., Porumb, V., & Bild, W. (2023). The Skin and Natural Cannabinoids–Topical and Transdermal Applications. Pharmaceuticals, 16(7), 1049. https://doi.org/10.3390/ph16071049









