An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L.
Abstract
1. Introduction
2. Chemical Composition
2.1. Carotenoids
2.2. Terpenoids
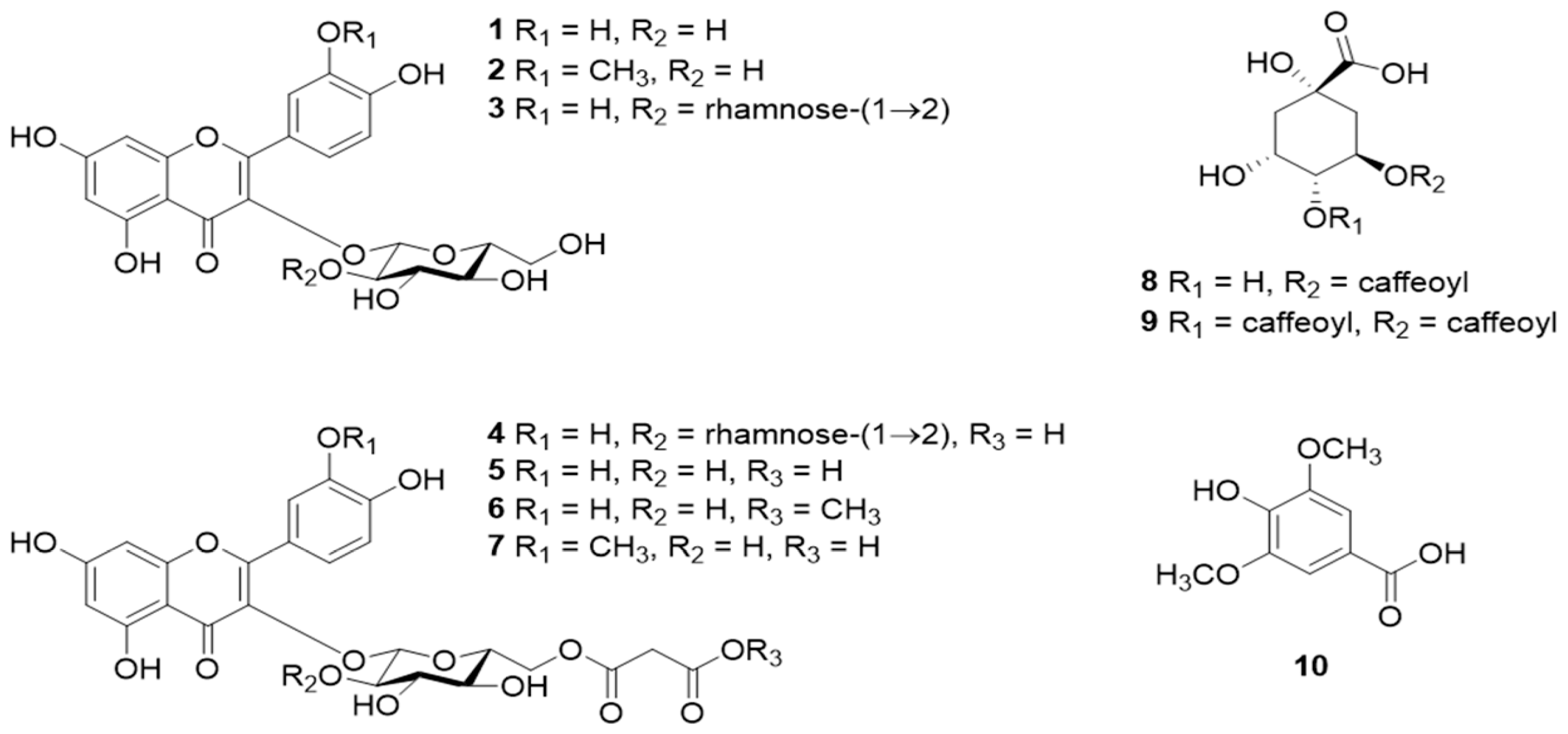
2.3. Flavonoids
2.4. Coumarins
2.5. Phenolic Acids
2.6. Quinones
2.7. Amino Acids
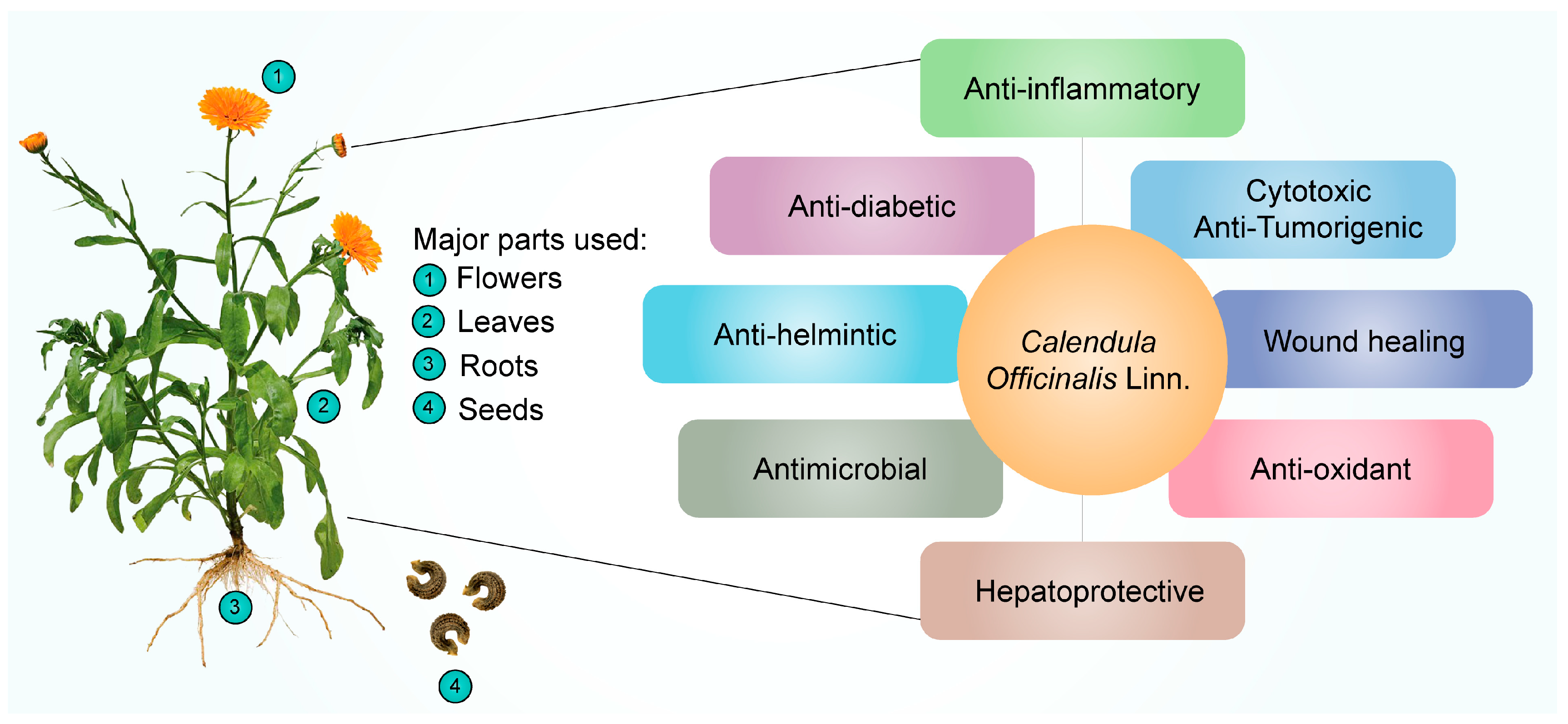
3. Therapeutic Applications of Calendula officinalis
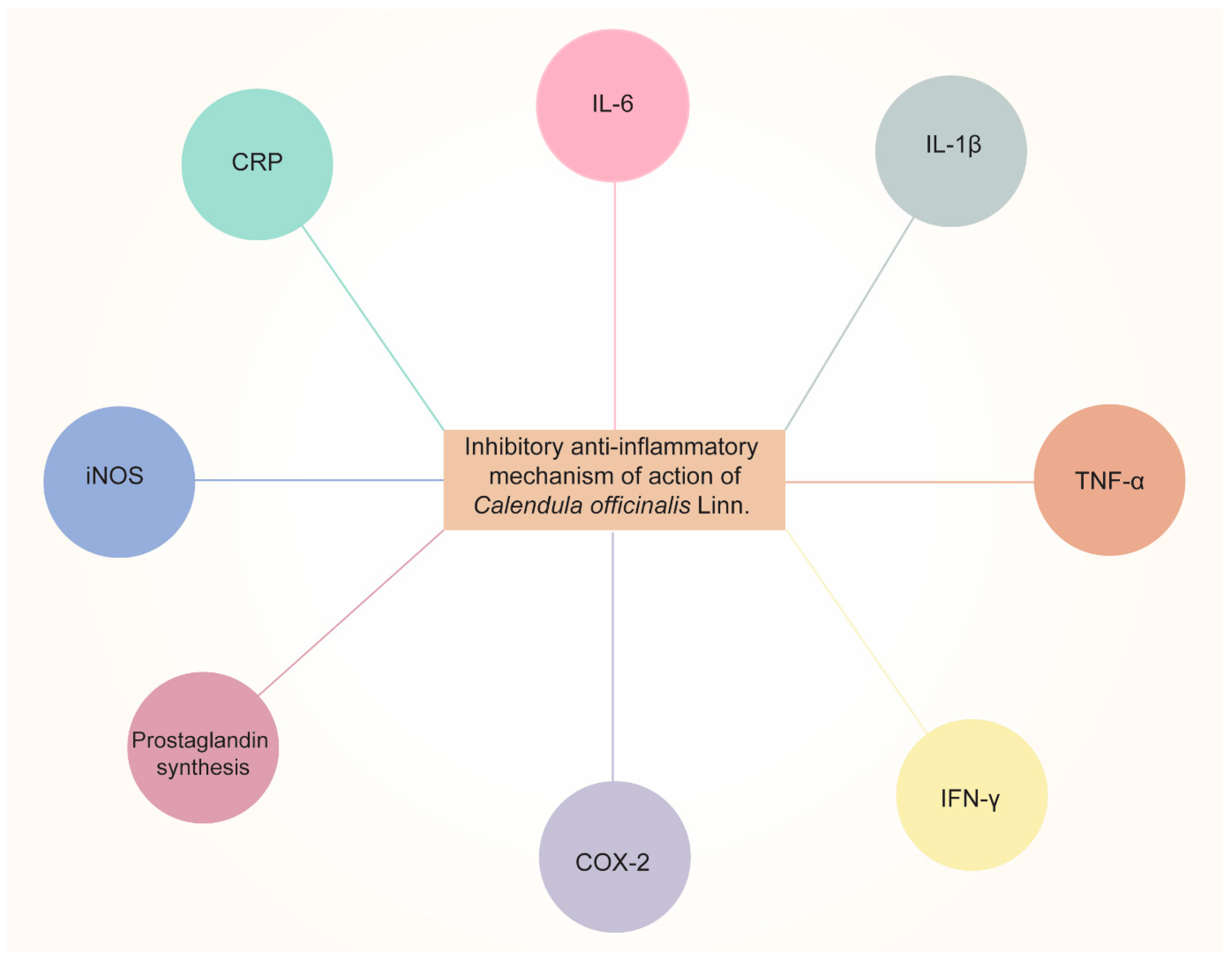
3.1. Anti-Inflammatory
3.2. Antioxidant Activity
3.3. Cytotoxic and Anti-Tumor Activity
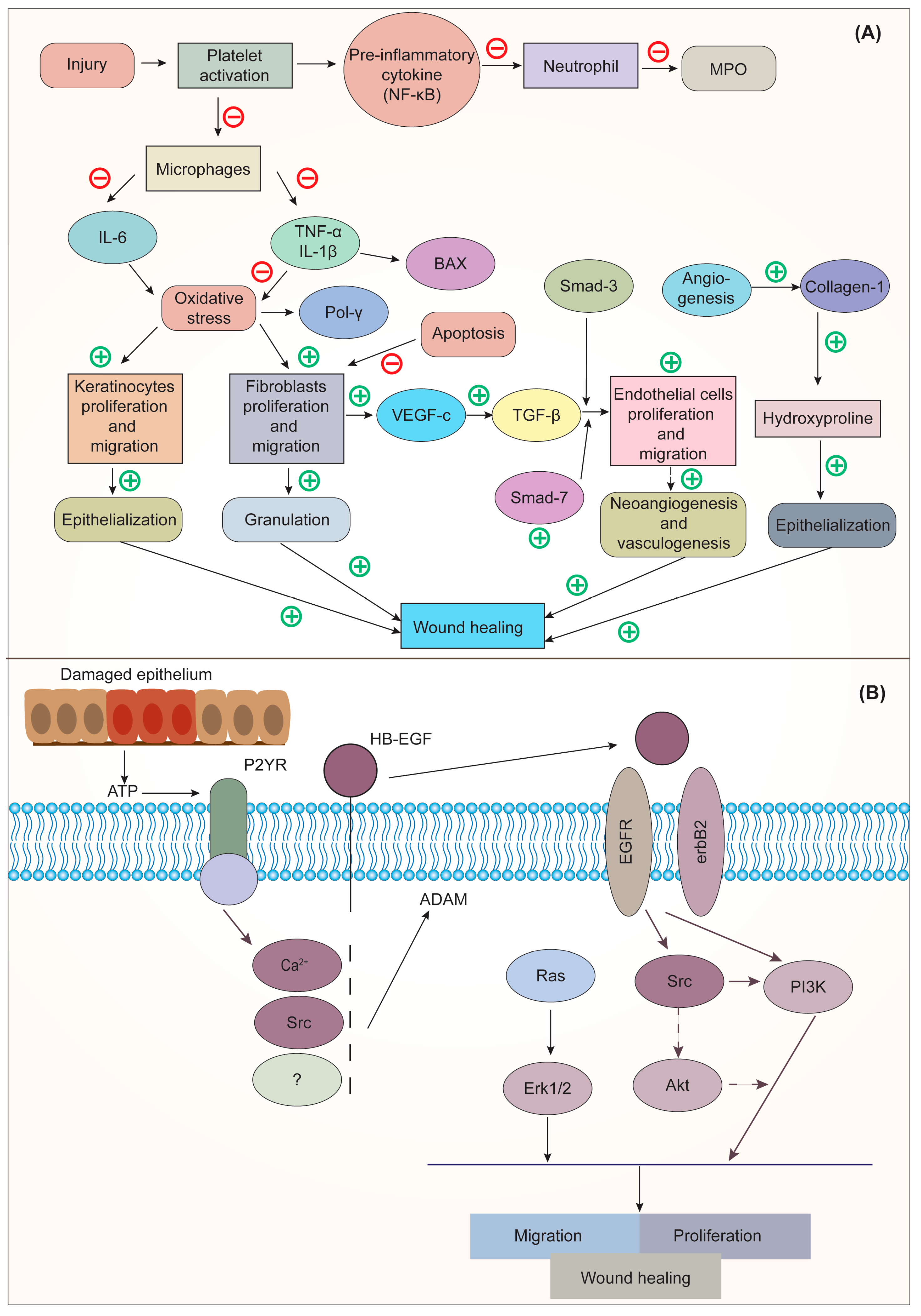
3.4. Wound-Healing Activity
3.5. Hepatoprotective Activity
3.6. Anthelmintic Activity
3.7. Antimicrobial Activity
3.7.1. Antibacterial Activity
3.7.2. Antiprotozoal Activity
3.7.3. Antifungal Activity
4. Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Knoess, W.; Wiesner, J. The Globalization of Traditional Medicines: Perspectives Related to the European Union Regulatory Environment. Engineering 2019, 5, 22–31. [Google Scholar] [CrossRef]
- Galucio, N.C.d.R.; Moysés, D.d.A.; Pina, J.R.S.; Marinho, P.S.B.; Gomes Júnior, P.C.; Cruz, J.N.; Vale, V.V.; Khayat, A.S.; Marinho, A.M.d.R. Antiproliferative, Genotoxic Activities and Quantification of Extracts and Cucurbitacin B Obtained from Luffa operculata (L.) Cogn. Arab. J. Chem. 2022, 15, 103589. [Google Scholar] [CrossRef]
- Chandorkar, N.; Tambe, S.; Amin, P.; Madankar, C. A Systematic and Comprehensive Review on Current Understanding of the Pharmacological Actions, Molecular Mechanisms, and Clinical Implications of the Genus Eucalyptus. Phytomed. Plus 2021, 1, 100089. [Google Scholar] [CrossRef]
- Rout, S.; Tambe, S.; Deshmukh, R.K.; Mali, S.; Cruz, J.; Srivastav, P.P.; Amin, P.D.; Gaikwad, K.K.; Andrade, E.H.D.A.; de Oliveira, M.S. Recent Trends in the Application of Essential Oils: The next Generation of Food Preservation and Food Packaging. Trends Food Sci. Technol. 2022, 129, 421–439. [Google Scholar] [CrossRef]
- Muzammil, S.; Neves Cruz, J.; Mumtaz, R.; Rasul, I.; Hayat, S.; Khan, M.A.; Khan, A.M.; Ijaz, M.U.; Lima, R.R.; Zubair, M. Effects of Drying Temperature and Solvents on In Vitro Diabetic Wound Healing Potential of Moringa Oleifera Leaf Extracts. Molecules 2023, 28, 710. [Google Scholar] [CrossRef]
- Savic Gajic, I.M.; Savic, I.M.; Skrba, M.; Dosić, A.; Vujadinovic, D. Food Additive Based on the Encapsulated Pot Marigold (Calendula officinalis L.) Flowers Extract in Calcium Alginate Microparticles. J. Food Process. Preserv. 2022, 46, e15792. [Google Scholar] [CrossRef]
- Pedram Rad, Z.; Mokhtari, J.; Abbasi, M. Preparation and Characterization of Calendula officinalis-Loaded PCL/Gum Arabic Nanocomposite Scaffolds for Wound Healing Applications. Iran. Polym. J. 2019, 28, 51–63. [Google Scholar] [CrossRef]
- Fallahi, M.; Mohammadi, A.; Miri, S.M. The Natural Variation in Six Populations of Calendula officinalis L.: A Karyotype Study. J. Genet. 2020, 6, 34–40. [Google Scholar] [CrossRef]
- Basch, E.; Bent, S.; Foppa, I.; Haskmi, S.; Kroll, D.; Mele, M.; Szapary, P.; Ulbricht, C.; Vora, M.; Yong, S. Marigold (Calendula officinalis L.): An Evidence-Based Systematic Review by the Natural Standard Research Collaboration. J. Herb. Pharm. 2006, 6, 135–159. [Google Scholar] [CrossRef]
- Gu, J.; Aidy, A.; Goorani, S. Anti-Human Lung Adenocarcinoma, Cytotoxicity, and Antioxidant Potentials of Copper Nanoparticles Green-Synthesized by Calendula officinalis. J. Exp. Nanosci. 2022, 17, 285–296. [Google Scholar] [CrossRef]
- Schneider, C. Traumeel – an Emerging Option to Nonsteroidal Anti-Inflammatory Drugs in the Management of Acute Musculoskeletal Injuries. Int. J. Gen. Med. 2011, 4, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Ashwlayan, V.D.; Kumar, A.; Verma, M.; Garg, V.K.; Gupta, S. Therapeutic Potential of Calendula officinalis. Pharm. Pharmacol. Int. J. 2018, 6, 1. [Google Scholar] [CrossRef]
- Almeida, V.M.; Dias, Ê.R.; Souza, B.C.; Cruz, J.N.; Santos, C.B.R.; Leite, F.H.A.; Queiroz, R.F.; Branco, A. Methoxylated Flavonols from Vellozia Dasypus Seub Ethyl Acetate Active Myeloperoxidase Extract: In Vitro and in Silico Assays. J. Biomol. Struct. Dyn 2022, 40, 7574–7583. [Google Scholar] [CrossRef] [PubMed]
- Arora, D.; Rani, A.; Sharma, A. A Review on Phytochemistry and Ethnopharmacological Aspects of Genus Calendula. Pharmacogn. Rev. 2013, 7, 179–187. [Google Scholar] [CrossRef]
- Rego, C.M.A.; Francisco, A.F.; Boeno, C.N.; Paloschi, M.V.; Lopes, J.A.; Silva, M.D.S.; Santana, H.M.; Serrath, S.N.; Rodrigues, J.E.; Lemos, C.T.L.; et al. Inflammasome NLRP3 Activation Induced by Convulxin, a C-Type Lectin-like Isolated from Crotalus Durissus Terrificus Snake Venom. Sci. Rep. 2022, 12, 1428. [Google Scholar] [CrossRef] [PubMed]
- Ercetin, T.; Senol, F.S.; Erdogan Orhan, I.; Toker, G. Comparative Assessment of Antioxidant and Cholinesterase Inhibitory Properties of the Marigold Extracts from Calendula arvensis L. and Calendula officinalis L. Ind. Crop. Prod. 2012, 36, 203–208. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Castro, S.; Paiva, J.; Santos, C.; Silveira, P. Taxonomic Revision of the Genus Calendula (Asteraceae) in the Iberian Peninsula and the Balearic Islands. Phytotaxa 2018, 352, 1–91. [Google Scholar] [CrossRef]
- Medical Economics Company. PDR for Herbal Medicines; Medical Economics Co.: Montvale, NJ, USA, 2000; ISBN 1563633612. [Google Scholar]
- R Silva, E.J.; Gonçalves, E.S.; Aguiar, F.; Evêncio, L.B.; A Lyra, M.M.; Cristina C Coelho, M.O.; do Carmo A Fraga, M.C.; Wanderley, A.G.; Gonçalves Wanderley, A.; Rego, M. Toxicological Studies on Hydroalcohol Extract of Calendula officinalis L. Phytother. Res. 2007, 21, 332–336. [Google Scholar] [CrossRef]
- Pedram Rad, Z.; Mokhtari, J.; Abbasi, M. Calendula officinalis Extract/PCL/Zein/Gum Arabic Nanofibrous Bio-Composite Scaffolds via Suspension, Two-Nozzle and Multilayer Electrospinning for Skin Tissue Engineering. Int. J. Biol. Macromol. 2019, 135, 530–543. [Google Scholar] [CrossRef]
- Lima, A.D.M.; Siqueira, A.S.; Möller, M.L.S.; de Souza, R.C.; Cruz, J.N.; Lima, A.R.J.; da Silva, R.C.; Aguiar, D.C.F.; Junior, J.L.D.S.G.V.; Gonçalves, E.C. In Silico Improvement of the Cyanobacterial Lectin Microvirin and Mannose Interaction. J. Biomol. Struct. Dyn. 2022, 40, 1064–1073. [Google Scholar] [CrossRef]
- Chamansara, R.; Rashidfarokhi, R.; Sakallı, E.A.; Öztinen, N.; Koşar, M. Comparison of Commercial Calendula officinalis L. Samples with Pharmacopeial Drug: Antiradical Activities and Chemical Profiles. J. Res. Pharm. 2022, 26, 809–819. [Google Scholar] [CrossRef]
- Janarny, G.; Ranaweera, K.K.D.S.; Gunathilake, K.D.P.P. Antioxidant Activities of Hydro-Methanolic Extracts of Sri Lankan Edible Flowers. Biocatal. Agric. Biotechnol. 2021, 35, 102081. [Google Scholar] [CrossRef]
- Neukirch, H.; D’Ambrosio, M.; Dalla Via, J.; Guerriero, A. Simultaneous Quantitative Determination of Eight Triterpenoid Monoesters from Flowers of 10 Varieties of Calendula officinalis L. and Characterisation of a New Triterpenoid Monoester. Phytochem. Anal. 2004, 15, 30–35. [Google Scholar] [CrossRef]
- Kishimoto, S.; Maoka, T.; Sumitomo, K.; Ohmiya, A. Analysis of Carotenoid Composition in Petals of Calendula (Calendula officinalis L.). Biosci. Biotechnol. Biochem. 2005, 69, 2122–2128. [Google Scholar] [CrossRef]
- Muley, B.P.; Khadabadi, S.S.; Banarase, N.B. Phytochemical Constituents and Pharmacological Activities of Calendula officinalis Linn (Asteraceae): A Review. Trop. J. Pharm. Res. 2009, 8, 455–465. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Akobirshoeva, A.; Zilfikarov, I.N.; Vennos, C. Isorhamnetin and Quercetin Derivatives as Anti-Acetylcholinesterase Principles of Marigold (Calendula officinalis) Flowers and Preparations. Int. J. Mol. Sci. 2017, 18, 1685. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Murakami, T.; Kishi, A.; Kageura, T.; Matsuda, H. Medicinal Flowers. III. Marigold. (1): Hypoglycemic, Gastric Emptying Inhibitory, and Gastroprotective Principles and New Oleanane-Type Triterpene Oligoglycosides, Calendasaponins A, B, C, and D, from Egyptian Calendula officinalis. Chem. Pharm. Bull. 2001, 49, 863–870. [Google Scholar] [CrossRef]
- John, R.; Jan, N. Calendula officinalis—An Important Medicinal Plant with Potential Biological Properties. Proc. Indian Natl. Sci. Acad. 2017, 83, 769–787. [Google Scholar] [CrossRef]
- Khalil, M.Y.; Naguib, N.Y.; Sherbeny, S.E. A Comparative Study on the Productivity and Chemical Constituents of Various Sources and Species of Calendula Plants as Affected by Two Foliar Fertilizers Cultivation and Production of Medicinal and Aromatic Plants Department. J. Appl. Sci. Res. 2005, 1, 176–189. [Google Scholar]
- Paolini, J.; Barboni, T.; Desjobert, J.M.; Djabou, N.; Muselli, A.; Costa, J. Chemical Composition, Intraspecies Variation and Seasonal Variation in Essential Oils of Calendula arvensis L. Biochem. Syst. Ecol. 2010, 38, 865–874. [Google Scholar] [CrossRef]
- Bisset, N.G.; Wichtl, M. Herbal Drugs and Phytopharmaceuticals; Medpharm GmbH Scientific Publishers: Stuttgart, Germany; CRC Press: Boca Raton, FL, USA, 1994; pp. 91–95. [Google Scholar]
- Raal, A.; Orav, A.; Nesterovitsch, J.; Maidla, K. Analysis of Carotenoids, Flavonoids and Essential Oil of Calendula officinalis Cultivars Growing in Estonia. Nat. Prod. Commun. 2016, 11, 1934578X1601100831. [Google Scholar] [CrossRef]
- Sahingil, D. GC/MS-Olfactometric Characterization of the Volatile Compounds, Determination Antimicrobial and Antioxidant Activity of Essential Oil from Flowers of Calendula (Calendula officinalis L.). J. Essent. Oil Bear. Plants 2019, 22, 1571–1580. [Google Scholar] [CrossRef]
- Afolayan, A.J. Variation in the Essential Oil Composition of Calendula officinalis L. by Omobola Okoh Dissertation Submitted in Satisfaction of the Requirements for the Degree of Master of Science (Chemistry) in the Faculty of Science and Agriculture University of Fort Hare Supervisors. Ph.D. Thesis, University of Fort Hare, Alice, South Africa, 2008. [Google Scholar]
- Khalid, K.A.; EL-Ghorab, A.H. The Effect of Presowing Low Temperature on Essential Oil Content and Chemical Composition of Calendula officinalis. J. Essent. Oil Bear. Plants 2013, 9, 32–41. [Google Scholar] [CrossRef]
- Al-Rifai, A. Identification and Evaluation of In-Vitro Antioxidant Phenolic Compounds from the Calendula Tripterocarpa Rupr. S. Afr. J. Bot. 2018, 116, 238–244. [Google Scholar] [CrossRef]
- Al-Saleem, M.S.; Awaad, A.S.; Alothman, M.R.; Alqasoumi, S.I. Phytochemical Standardization and Biological Activities of Certain Desert Plants Growing in Saudi Arabia. Saudi Pharm. J. 2017, 26, 198–204. [Google Scholar] [CrossRef]
- Zitterl-Eglseer, K.; Sosa, S.; Jurenitsch, J.; Schubert-Zsilavecz, M.; della Loggia, R.; Tubaro, A.; Bertoldi, M.; Franz, C. Anti-Oedematous Activities of the Main Triterpendiol Esters of Marigold (Calendula officinalis L.). J. Ethnopharmacol. 1997, 57, 139–144. [Google Scholar] [CrossRef]
- Wojciechowski, Z.; Jelonkiewicz-Konador, A.; Tomaszewski, M.; Jankowski, J.; Kasprzyk, Z. The Structure of Glycosides of Oleanolic Acid Isolated from the Roots of Calendula officinalis. Phytochemistry 1971, 10, 1121–1124. [Google Scholar] [CrossRef]
- Vecherko, L.P.; Sviridov, A.F.; Zinkevich, E.P.; Kogan, L.M. The Structure of Calendulosides C and D from the Roots of Calendula officinalis. Chem. Nat. Compd. 1975, 11, 379–384. [Google Scholar] [CrossRef]
- Ukiya, M.; Akihisa, T.; Yasukawa, K.; Tokuda, H.; Suzuki, T.; Kimura, Y. Anti-Inflammatory, Anti-Tumor-Promoting, and Cytotoxic Activities of Constituents of Marigold (Calendula officinalis) Flowers. J. Nat. Prod. 2006, 69, 1692–1696. [Google Scholar] [CrossRef]
- Naved, T.; Ansari, S.H.; Mukhtar, H.M.; Ali, M. New Triterpenic Esters of Oleanene-Series from the Flowers of Calendula officinalis Linn. ChemInform 2005, 36, 1088–1091. [Google Scholar] [CrossRef]
- Kurkin, V.A.; Sharova, O.v. Flavonoids from Calendula officinalis Flowers. Chem. Nat. Compd. 2007, 43, 216–217. [Google Scholar] [CrossRef]
- Vidal-ollivier, E.; Balansard, G.; Faure, R.; Babadjamian, A. Revised Structures of Triterpenoid Saponins from the Flowers of Calendula officinalis. J. Nat. Prod. 1989, 52, 1156–1159. [Google Scholar] [CrossRef]
- Derkach, A.I.; Komissarenko, N.F.; Chernobai, V.T. Coumarins of the Inflorescences of Calendula officinalis and Helichrysum Arenarium. Chem. Nat. Compd. 1987, 22, 722–723. [Google Scholar] [CrossRef]
- Crabas, N.; Marongiu, B.; Piras, A.; Pivetta, T.; Porcedda, S. Extraction, Separation and Isolation of Volatiles and Dyes from Calendula officinalis L. and Aloysia triphylla (L’Her.) Britton by Supercritical CO2. J. Essent. Oil Res. 2011, 15, 272–277. [Google Scholar] [CrossRef]
- Khalid, K.; SILVA, D.A.; Teixeira, J.A. Biology of Calendula officinalis Linn.: Focus on Pharmacology, Biological Activities and Agronomic Practices. Med. Aromat. Plant Sci. Biotechnol. 2012, 6, 12–27. [Google Scholar]
- Janiszowska, W.; Michalski, W.; Kasprzyk, Z. Polyprenyl Quinones and α-Tocopherol in Calendula officinalis. Phytochemistry 1976, 15, 125–127. [Google Scholar] [CrossRef]
- Iatsyno, A.I.; Belova, L.F.; Lipkina, G.S.; Sokolov, S.I.; Trutneva, E.A. Pharmacology of Calenduloside B, a New Triterpene Glycoside from the Roots of Calendula officinalis. Farm. Toksikol. 1978, 41, 556–560. [Google Scholar]
- Goodwin, T.W. Studies in Carotenogenesis. 13. The Carotenoids of the Flower Petals of Calendula officinalis. Biochem. J. 1954, 58, 90. [Google Scholar] [CrossRef] [PubMed]
- Bakó, E.; Deli, J.; Tóth, G. HPLC Study on the Carotenoid Composition of Calendula Products. J. Biochem. Biophys. Methods 2002, 53, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Preethi, K.; Kuttan, R. Hepato and Reno Protective Action of Calendula officinalis L. Flower Extract. Indian J. Exp. Biol. 2009, 47, 163–180. [Google Scholar] [PubMed]
- Preethi, K.C.; Kuttan, G.; Kuttan, R. Antioxidant Potential of an Extract of Calendula officinalis. Flowers in Vitro. and in Vivo. Pharm. Biol. 2008, 44, 691–697. [Google Scholar] [CrossRef]
- Murillo, A.G.; Hu, S.; Fernandez, M.L. Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin. Antioxidants 2019, 8, 390. [Google Scholar] [CrossRef]
- Patil, K.; Sanjay, C.; Doggalli, N.; Devi, K.R.; Harshitha, N. A Review of Calendula officinalis Magic in Science. J. Clin. Diagn. Res. 2022, 16, ZE23–ZE27. [Google Scholar] [CrossRef]
- Selvaraj, S.; Fathima, N.N. Fenugreek Incorporated Silk Fibroin Nanofibers—A Potential Antioxidant Scaffold for Enhanced Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 5916–5926. [Google Scholar] [CrossRef] [PubMed]
- Khairnar, M.S.; Pawar, B.; Marawar, P.P.; Mani, A. Evaluation of Calendula officinalis as an Anti-Plaque and Anti-Gingivitis Agent. J. Indian Soc. Periodontol. 2013, 17, 741. [Google Scholar] [CrossRef] [PubMed]
- Saini, P.; Al-Shibani, N.; Sun, J.; Zhang, W.; Song, F.; Gregson, K.S.; Windsor, L.J. Effects of Calendula officinalis on Human Gingival Fibroblasts. Homeopathy 2012, 101, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kumari, K. An overview on Calendula officinalis L.: (pot marigold). J. Adv. Sci. Res. 2021, 12, 13–18. [Google Scholar] [CrossRef]
- Lohani, A.; Mishra, A.K.; Verma, A. Cosmeceutical Potential of Geranium and Calendula Essential Oil: Determination of Antioxidant Activity and in Vitro Sun Protection Factor. J. Cosmet. Dermatol. 2019, 18, 550–557. [Google Scholar] [CrossRef]
- Rajwinder, K.; Singh, R.; Lalit; Jaswinder, K. Rajat Evaluation of the Antidepressant Like Activity of Ethanolic Extract of Calendula officinalis Using Rodent Models (Wistar Rat) of Depression. Curr. Psychopharmacol. 2020, 9, 58–67. [Google Scholar] [CrossRef]
- Zhang, N.; Ying, M.D.; Wu, Y.P.; Zhou, Z.H.; Ye, Z.M.; Li, H.; Lin, D.S. Hyperoside, a Flavonoid Compound, Inhibits Proliferation and Stimulates Osteogenic Differentiation of Human Osteosarcoma Cells. PLoS ONE 2014, 9, e98973. [Google Scholar] [CrossRef]
- Hsia, C.W.; Lin, K.C.; Lee, T.Y.; Hsia, C.H.; Chou, D.S.; Jayakumar, T.; Velusamy, M.; Chang, C.C.; Sheu, J.R. Esculetin, a Coumarin Derivative, Prevents Thrombosis: Inhibitory Signaling on PLCγ2–PKC–AKT Activation in Human Platelets. Int. J. Mol. Sci. 2019, 20, 2731. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Cruz-Martins, N.; López-Jornet, P.; Lopez, E.P.F.; Harun, N.; Yeskaliyeva, B.; Beyatli, A.; Sytar, O.; Shaheen, S.; Sharopov, F.; et al. Natural Coumarins: Exploring the Pharmacological Complexity and Underlying Molecular Mechanisms. Oxidative Med. Cell. Longev. 2021, 2021, 6492346. [Google Scholar] [CrossRef] [PubMed]
- Butnariu, M.; Coradini, C.Z. Evaluation of Biologically Active Compounds from Calendula officinalis Flowers Using Spectrophotometry. Chem. Cent. J. 2012, 6, 35. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic Acid (CGA): A Pharmacological Review and Call for Further Research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Ye, Y.; Cai, T.; Li, S.; Liu, X. Anti-Fatigue Activity of the Polysaccharides Isolated from Ribes Stenocarpum Maxim. J. Funct. Foods 2022, 89, 104947. [Google Scholar] [CrossRef]
- Cetkovic, G.; Djilas, S.; Canadanovic-Brunet, J.; Tumbas, V. Thin-Layer Chromatography Analysis and Scavenging Activity of Marigold (Calendula officinalis L.) Extracts. Acta Period. Technol. 2003, 34, 93–102. [Google Scholar] [CrossRef]
- Garaiova, M.; Hua, Q.; Holic, R. Heterologous Production of Calendic Acid Naturally Found in Calendula officinalis by Recombinant Fission Yeast. J. Agric. Food Chem. 2022, 71, 8. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Noguchi, R.; Ota, T.; Abe, M.; Miyashita, K.; Kawada, T. Cytotoxic Effect of Conjugated Trienoic Fatty Acids on Mouse Tumor and Human Monocytic Leukemia Cells. Lipids 2001, 36, 477–482. [Google Scholar] [CrossRef]
- Dulf, F.V.; Pamfil, D.; Baciu, A.D.; Pintea, A. Fatty Acid Composition of Lipids in Pot Marigold (Calendula officinalis L.) Seed Genotypes. Chem. Cent. J. 2013, 7, 8. [Google Scholar] [CrossRef]
- Pizza, C.; Zhong-Liang, Z.; Tommasi, N. Plant Metabolites Triterpenoid Saponins from Calendula Arvensis. J. Nat. Prod. 1987, 50, 927–931. [Google Scholar] [CrossRef]
- Tagousop, C.N.; Tamokou, J.D.D.; Feugap, L.D.T.; Harakat, D.; Voutquenne-Nazabadioko, L.; Ngnokam, D.; Tagousop, C.N.; Tamokou, J.-D.-D.; Feugap, L.D.T.; Harakat, D.; et al. New Hemisynthetic Oleanane Saponin with Antimicrobial Activities. Adv. Biol. Chem. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Vecherko, L.P.; Sviridov, A.F.; Zinkevich, E.P.; Kogan, L.M. Structures of Calendulosides G and H from the Roots of Calendula officinalis. Chem. Nat. Compd. 1974, 10, 548–549. [Google Scholar] [CrossRef]
- Szakiel, A.; Ruszkowski, D.; Grudniak, A.; Kurek, A.; Wolska, K.I.; Doligalska, M.; Janiszowska, W. Antibacterial and Antiparasitic Activity of Oleanolic Acid and Its Glycosides Isolated from Marigold (Calendula officinalis). Planta Med. 2008, 74, 1709–1715. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, B.; Khan, R.A.; Al-Howiriny, T.A.; Al-Rehaily, A.J. Osteosaponins 1 and 2: Two New Saponin Glycosides from Osteospermum vaillantii. Nat. Prod. Res. 2010, 24, 1258–1267. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Ollivier, E.; Balansard, G.; Babadjamian, A.; Faure, R.; Vincent, E.J.; Chemli, R.; Boukef, K. Two-Dimensional NMR Studies of Triterpenoid Glycosides. II)-1H NMR Assignment of Arvensoside A and B, Calenduloside C and D. Spectrosc. Lett. 1989, 22, 579–584. [Google Scholar] [CrossRef]
- Zaki, A.A.; Qiu, L. Machaerinic Acid 3-O-β-D-Glucuronopyranoside from Calendula officinalis. Nat. Prod. Res. 2019, 34, 2938–2944. [Google Scholar] [CrossRef]
- El-Najjar, N.; Gali-Muhtasib, H.; Ketola, R.A.; Vuorela, P.; Urtti, A.; Vuorela, H. The Chemical and Biological Activities of Quinones: Overview and Implications in Analytical Detection. Phytochem. Rev. 2011, 10, 353–370. [Google Scholar] [CrossRef]
- Baskaran Kaviena Pharmacological Activities of Calendula officinalis. Int. J. Sci. Res. 2017, 6, 43–47.
- Chandra, P.; Kishore, K.; Ghosh, A.K. Evaluation of Antacid Capacity and Antiulcer Activity of Calendula officinalis L. in Experimental Rats. Orient. Pharm. Exp. Med. 2015, 15, 277–285. [Google Scholar] [CrossRef]
- Abasova, R.L.; Aslanov, S.M.; Mamedova, M.É. Amino Acids of Calendula officinalis. Chem. Nat. Compd. 1995, 30, 641. [Google Scholar] [CrossRef]
- Kadowaki, W.; Miyata, R.; Fujinami, M.; Sato, Y.; Kumazawa, S. Catechol-O-Methyltransferase Inhibitors from Calendula officinalis Leaf. Molecules 2023, 28, 1333. [Google Scholar] [CrossRef] [PubMed]
- Ak, G.; Zengin, G.; Ceylan, R.; Fawzi Mahomoodally, M.; Jugreet, S.; Mollica, A.; Stefanucci, A. Chemical Composition and Biological Activities of Essential Oils from Calendula officinalis L. Flowers and Leaves. Flavour. Fragr. J. 2021, 36, 554–563. [Google Scholar] [CrossRef]
- Ak, G.; Zengin, G.; Sinan, K.I.; Mahomoodally, M.F.; Picot-Allain, M.C.N.; Cakir, O.; Bensari, S.; Yilmaz, M.A.; Gallo, M.; Montesano, D. A Comparative Bio-Evaluation and Chemical Profiles of Calendula officinalis L. Extracts Prepared via Different Extraction Techniques. Appl. Sci. 2020, 10, 5920. [Google Scholar] [CrossRef]
- Mishra, A.K.; Mishra, A.; Pragya; Chattopadhyay, P. Screening of Acute and Sub-Chronic Dermal Toxicity of Calendula officinalis L Essential Oil. Regul. Toxicol. Pharmacol. 2018, 98, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Nicolaus, C.; Junghanns, S.; Hartmann, A.; Murillo, R.; Ganzera, M.; Merfort, I. In Vitro Studies to Evaluate the Wound Healing Properties of Calendula officinalis Extracts. J. Ethnopharmacol. 2017, 196, 94–103. [Google Scholar] [CrossRef]
- Khursheed, A.; Devender, P.; Shahid, A.; Vandana Arora, S.; Alok, B.; Alam Professor, K. Evaluation of Anthelmintic Activity of Calendula officinalis Flowers Extract. J. Drug Deliv. Ther. 2021, 11, 48–50. [Google Scholar] [CrossRef]
- Slavov, A.; Ognyanov, M.; Vasileva, I. Pectic Polysaccharides Extracted from Pot Marigold (Calendula officinalis) Industrial Waste. Food Hydrocoll. 2020, 101, 105545. [Google Scholar] [CrossRef]
- Bedoya, L.C.R.; López, K.G.; Pareja, N.M. Extraction of Metabolites from Calendula officinalis and Evaluation of Their Colorant and Antibacterial Capacity. Rev. Colomb. Biotecnol. 2020, 22, 60–69. [Google Scholar] [CrossRef]
- Gazim, Z.C.; Rezende, C.M.; Fraga, S.R.; Dias Filho, B.P.; Nakamura, C.V.; Cortez, D.A.G. Analysis of the Essential Oils from Calendula officinalis Growing in Brazil Using Three Different Extraction Procedures. Rev. Bras. Ciênc. Farm. 2008, 44, 391–395. [Google Scholar] [CrossRef]
- García-Risco, M.R.; Mouhid, L.; Salas-Pérez, L.; López-Padilla, A.; Santoyo, S.; Jaime, L.; Ramírez de Molina, A.; Reglero, G.; Fornari, T. Biological Activities of Asteraceae (Achillea millefolium and Calendula officinalis) and Lamiaceae (Melissa officinalis and Origanum majorana) Plant Extracts. Plant Foods Hum. Nutr. 2017, 72, 96–102. [Google Scholar] [CrossRef]
- López-Padilla, A.; Ruiz-Rodriguez, A.; Reglero, G.; Fornari, T. Supercritical Carbon Dioxide Extraction of Calendula officinalis: Kinetic Modeling and Scaling up Study. J. Supercrit. Fluids 2017, 130, 292–300. [Google Scholar] [CrossRef]
- Jiménez-Medina, E.; Garcia-Lora, A.; Paco, L.; Algarra, I.; Collado, A.; Garrido, F. A New Extract of the Plant Calendula officinalis Produces a Dual in Vitro Effect: Cytotoxic Anti-Tumor Activity and Lymphocyte Activation. BMC Cancer 2006, 6, 119. [Google Scholar] [CrossRef] [PubMed]
- Balciunaitiene, A.; Puzeryte, V.; Radenkovs, V.; Krasnova, I.; Memvanga, P.B.; Viskelis, P.; Streimikyte, P.; Viskelis, J. Sustainabl–Green Synthesis of Silver Nanoparticles Using Aqueous Hyssopus Officinalis and Calendula officinalis Extracts and Their Antioxidant and Antibacterial Activities. Molecules 2022, 27, 7700. [Google Scholar] [CrossRef]
- Vinola, S.M.J.; Sekar, M.; Renganathan, S.K.; Dhiraviam, S. Comparative Evaluation of Calendula officinalis and 2% Chlorhexidine against Enterococcus Faecalis and Candida Albicans. J. Interdiscip. Dent. 2022, 11, 119. [Google Scholar] [CrossRef]
- Garrido-Suárez, B.B.; Garrido, G.; Menéndez, A.B.; Merino, N.; Valdés, O.; de la Paz, N.; Romero, A.; Delgado, L.; Fernández, M.D.; Piñeros, O.; et al. Topical Calendula officinalis L. Inhibits Inflammatory Pain through Antioxidant, Anti-Inflammatory and Peripheral Opioid Mechanisms. J. Integr. Med. 2022, 21, 34–46. [Google Scholar] [CrossRef]
- Deka, B.; Bhattacharjee, B.; Shakya, A.; Ikbal, A.M.A.; Goswami, C.; Sarma, S. Mechanism of Action of Wound Healing Activity of Calendula officinalis: A Comprehensive Review. Pharm. Biosci. J. 2021, 9, 28–44. [Google Scholar] [CrossRef]
- Hao, W.; Jia, Y.; Wang, C.; Wang, X. Preparation, Chemical Characterization and Determination of the Antioxidant, Cytotoxicity and Therapeutic Effects of Gold Nanoparticles Green-Synthesized by Calendula officinalis Flower Extract in Diabetes-Induced Cardiac Dysfunction in Rat. Inorg. Chem. Commun. 2022, 144, 109931. [Google Scholar] [CrossRef]
- Panahi, Y.; Sharif, M.R.; Sharif, A.; Beiraghdar, F.; Zahiri, Z.; Amirchoopani, G.; Marzony, E.T.; Sahebkar, A. A Randomized Comparative Trial on the Therapeutic Efficacy of Topical Aloe Vera and Calendula officinalis on Diaper Dermatitis in Children. Sci. World J. 2012, 2012, 810234. [Google Scholar] [CrossRef]
- Pommier, P.; Gomez, F.; Sunyach, M.P.; D’Hombres, A.; Carrie, C.; Montbarbon, X. Phase III Randomized Trial of Calendula officinalis Compared with Trolamine for the Prevention of Acute Dermatitis during Irradiation for Breast Cancer. J. Clin. Oncol. 2004, 22, 1447–1453. [Google Scholar] [CrossRef]
- Singh, M.; Bagewadi, A. Comparison of Effectiveness of Calendula officinalis Extract Gel with Lycopene Gel for Treatment of Tobacco-Induced Homogeneous Leukoplakia: A Randomized Clinical Trial. Int. J. Pharm. Investig. 2017, 7, 88. [Google Scholar] [CrossRef]
- Babaee, N.; Moslemi, D.; Khalilpour, M.; Vejdani, F.; Moghadamnia, Y.; Bijani, A.; Baradaran, M.; Kazemi, M.T.; Khalilpour, A.; Pouramir, M.; et al. Antioxidant Capacity of Calendula officinalis Flowers Extract and Prevention of Radiation Induced Oropharyngeal Mucositis in Patients with Head and Neck Cancers: A Randomized Controlled Clinical Study. DARU J. Pharm. Sci. 2013, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Giostri, G.S.; Novak, E.M.; Buzzi, M.; Guarita-Souza, L.C. Treatment of Acute Wounds in Hand with Calendula officinalis L.: A Randomized Trial. Tissue Barriers 2021, 10, 1994822. [Google Scholar] [CrossRef]
- Buzzi, M.; De Freitas, F.; De Barros Winter, M. Therapeutic Effectiveness of a Calendula officinalis Extract in Venous Leg Ulcer Healing. J. Wound Care 2016, 25, 732–739. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, C.; Di Stadio, A.; Vitale, S.; Saccone, G.; De Angelis, M.C.; Zizolfi, B.; Di Spiezio Sardo, A. Use of Calendula Ointment after Episiotomy: A Randomized Clinical Trial. J. Matern. Fetal Neonatal Med. 2020, 35, 1860–1864. [Google Scholar] [CrossRef] [PubMed]
- Saffari, E.; Mohammad-Alizadeh-Charandabi, S.; Adibpour, M.; Mirghafourvand, M.; Javadzadeh, Y. Comparing the Effects of Calendula officinalis and Clotrimazole on Vaginal Candidiasis: A Randomized Controlled Trial. Women Health 2016, 57, 1145–1160. [Google Scholar] [CrossRef]
- Pazhohideh, Z.; Mohammadi, S.; Bahrami, N.; Mojab, F.; Abedi, P.; Maraghi, E. The Effect of Calendula officinalis versus Metronidazole on Bacterial Vaginosis in Women: A Double-Blind Randomized Controlled Trial. J. Adv. Pharm. Technol. Res. 2018, 9, 19. [Google Scholar] [CrossRef]
- Fonseca, Y.M.; Catini, C.D.; Vicentini, F.T.M.C.; Nomizo, A.; Gerlach, R.F.; Fonseca, M.J.V. Protective Effect of Calendula officinalis Extract against UVB-Induced Oxidative Stress in Skin: Evaluation of Reduced Glutathione Levels and Matrix Metalloproteinase Secretion. J. Ethnopharmacol. 2010, 127, 596–601. [Google Scholar] [CrossRef]
- Hamburger, M.; Adler, S.; Baumann, D.; Förg, A.; Weinreich, B. Preparative Purification of the Major Anti-Inflammatory Triterpenoid Esters from Marigold (Calendula officinalis). Fitoterapia 2003, 74, 328–338. [Google Scholar] [CrossRef]
- Silva, D.; Ferreira, M.S.; Sousa-Lobo, J.M.; Cruz, M.T.; Almeida, I.F. Anti-Inflammatory Activity of Calendula officinalis L. Flower Extract. Cosmetics 2021, 8, 31. [Google Scholar] [CrossRef]
- Dennis Bilavendran, J.; Manikandan, A.; Thangarasu, P.; Sivakumar, K. Synthesis and Discovery of Pyrazolo-Pyridine Analogs as Inflammation Medications through pro- and Anti-Inflammatory Cytokine and COX-2 Inhibition Assessments. Bioorg. Chem. 2020, 94, 103484. [Google Scholar] [CrossRef]
- Kiaei, N.; Hajimohammadi, R.; Hosseini, M. Investigation of the Anti-Inflammatory Properties of Calendula Nanoemulsion on Skin Cells. Bioinspired Biomim. Nanobiomater. 2018, 7, 228–237. [Google Scholar] [CrossRef]
- Cook, N.C.; Samman, S. Flavonoids--Chemistry, Metabolism, Cardioprotective Effects, and Dietary Sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Younes, M.; Siegers, C.P. Inhibitory Action of Some Flavonoids on Enhanced Spontaneous Lipid Peroxidation Following Glutathione Depletion. Planta Med. 1981, 43, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Jacobo-Velázquez, D.A.; Cisneros-Zevallos, L. Correlations of Antioxidant Activity against Phenolic Content Revisited: A New Approach in Data Analysis for Food and Medicinal Plants. J. Food Sci. 2009, 74, R107–R113. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Despande, B. Antioxidant Activity in the Leaves and Petals of Calendula officinalis Linn. Asian Pac. J. Health Sci. 2022, 9, 130–132. [Google Scholar] [CrossRef]
- Elias, R.; De Méo, M.; Vidal-Ollivier, E.; Laget, M.; Balansard, G.; Dumenil, G. Antimutagenic Activity of Some Saponins Isolated from Calendula officinalis L., C. arvensis L. and Hedera helix L. Mutagenesis 1990, 5, 327–332. [Google Scholar] [CrossRef]
- Cruceriu, D.; Balacescu, O.; Rakosy, E. Calendula officinalis: Potential Roles in Cancer Treatment and Palliative Care. Integr. Cancer Ther. 2018, 17, 1068–1078. [Google Scholar] [CrossRef]
- Hernández-Rosas, N.A.; García-Zebadúa, J.C.; Hernández-Delgado, N.; Torres-Castillo, S.; Figueroa-Arredondo, P.; Mora-Escobedo, R.; Hernández-Rosas, N.A.; García-Zebadúa, J.C.; Hernández-Delgado, N.; Torres-Castillo, S.; et al. Polyphenols Profile, Antioxidant Capacity, and in Vitro Cytotoxic Effect on Human Cancer Cell Lines of a Hydro-Alcoholic Extract from Calendula officinalis L. Petals. TIP Rev. Espec. Cienc. Quím.-Biol. 2018, 21, 54–64. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Arul, A.; Nayagam, J.; Natarajan, R. Wound Healing Potentials of Herbal Ointment Containing Calendula officinalis Linn. on the Alteration of Immunological Markers and Biochemical Parameters in Excision Wounded Animals. Clin. Phytosci. 2020, 6, 77. [Google Scholar] [CrossRef]
- Rathod, L.; Bhowmick, S.; Patel, P.; Sawant, K. Calendula Flower Extract Loaded Collagen Film Exhibits Superior Wound Healing Potential: Preparation, Evaluation, in-Vitro & in-Vivo Wound Healing Study. J. Drug Deliv. Sci. Technol. 2022, 72, 103363. [Google Scholar] [CrossRef]
- Jahdi, F.; Khabbaz, A.H.; Kashian, M.; Taghizadeh, M.; Haghani, H. The Impact of Calendula Ointment on Cesarean Wound Healing: A Randomized Controlled Clinical Trial. J. Fam. Med. Prim. Care 2018, 7, 893. [Google Scholar] [CrossRef]
- Bosley, R.; Leithauser, L.; Turner, M.; Gloster, H.M. The Efficacy of Second-Intention Healing in the Management of Defects on the Dorsal Surface of the Hands and Fingers after Mohs Micrographic Surgery. Dermatol. Surg. 2012, 38, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Pawan, V.; Rajinder, R.; Maninder, S.; Wazir, V.S. Kumar Pawan Attenuating Potential of Calendula officinalis on Biochemical and Antioxidant Parameters in Hepatotoxic Rats. Indian J Physiol. Pharmacol. 2017, 61, 398–410. [Google Scholar]
- Băieş, M.H.; Gherman, C.; Boros, Z.; Olah, D.; Vlase, A.M.; Cozma-Petruț, A.; Györke, A.; Miere, D.; Vlase, L.; Crișan, G.; et al. The Effects of Allium sativum L., Artemisia absinthium L., Cucurbita pepo L., Coriandrum sativum L., Satureja hortensis L. and Calendula officinalis L. on the Embryogenesis of Ascaris Suum Eggs during an In Vitro Experimental Study. Pathogens 2022, 11, 1065. [Google Scholar] [CrossRef] [PubMed]
- Boyko, O.; Brygadyrenko, V. Nematicidal Activity of Essential Oils of Medicinal Plants. Folia Oecologica 2021, 48, 42–48. [Google Scholar] [CrossRef]
- Rafał, I.G.; Króliczewski, B.J.; Górniak, I.; Bartoszewski, R.; Króliczewski, Á.J. Comprehensive Review of Antimicrobial Activities of Plant Flavonoids. Phytochem. Rev. 2018, 18, 241–272. [Google Scholar] [CrossRef]
- Manandhar, S.; Luitel, S.; Dahal, R.K. In Vitro Antimicrobial Activity of Some Medicinal Plants against Human Pathogenic Bacteria. J. Trop. Med. 2019, 2019, 1895340. [Google Scholar] [CrossRef]
- Djeussi, D.E.; Noumedem, J.A.K.; Seukep, J.A.; Fankam, A.G.; Voukeng, I.K.; Tankeo, S.B.; Nkuete, A.H.L.; Kuete, V. Antibacterial Activities of Selected Edible Plants Extracts against Multidrug-Resistant Gram-Negative Bacteria. BMC Complement. Altern. Med. 2013, 13, 164. [Google Scholar] [CrossRef]
- Duraipandiyan, V.; Ayyanar, M.; Ignacimuthu, S. Antimicrobial Activity of Some Ethnomedicinal Plants Used by Paliyar Tribe from Tamil Nadu, India. BMC Complement. Altern. Med. 2006, 6, 35. [Google Scholar] [CrossRef]
- Tambe, S.M.; Mali, S.; Amin, P.D.; Oliveira, M. Neuroprotective Potential of Cannabidiol: Molecular Mechanisms and Clinical Implications. J. Integr. Med. 2023, 3, 1–9. [Google Scholar] [CrossRef]
- Karnwal, A. In Vitro Antibacterial Activity of Hibiscus Rosa Sinensis, Chrysanthemum Indicum, and Calendula officinalis Flower Extracts against Gram Negative and Gram Positive Food Poisoning Bacteria. Adv. Tradit. Med. 2022, 22, 607–619. [Google Scholar] [CrossRef]
- Yalgi, V.S.; Bhat, K.G. Compare and Evaluate the Antibacterial Efficacy of Sodium Hypochlorite and Calendula officinalis against Streptococcus Mutans as a Root Canal Irrigating Solution: An in Vivo Study. J. Int. Oral. Health 2020, 12, 74. [Google Scholar] [CrossRef]
- Darekar, D.; Hate, M. Phytochemical Screening of Calendula officinalis (Linn.) Using Gas-Chromatography-Mass Spectroscopy with Potential Antibacterial Activity. J. Sci. Res. 2021, 65, 131–134. [Google Scholar] [CrossRef]
- Zulfiqer Shahen, M.; Mahmud, S.; Nahid Sohana, S.; Hasan Rony, M.; Abu Sayeed Imran, M.; Abdullah Al Maruf, M.; Afzal Ahmed Azim, M.; Monirul Islam, M.; Rokibul Islam, M.; Ekhlas Uddin, M.; et al. Effect of Antibiotic Susceptibility and Inhibitory Activity for the Control of Growth and Survival of Microorganisms of Extracts of Calendula officinalis. Eur. J. Med. Health Sci. 2019, 1, 1–9. [Google Scholar] [CrossRef]
- Removing Obstacles to Healthy Development: Report on Infectious Diseases. Available online: https://apps.who.int/iris/handle/10665/65847 (accessed on 25 March 2023).
- Samra, R.M.; Maatooq, G.T.; Zaki, A.A. A New Antiprotozoal Compound from Calendula officinalis. Nat. Prod. Res. 2022, 36, 5747–5752. [Google Scholar] [CrossRef] [PubMed]
- Webster, D.; Taschereau, P.; Belland, R.J.; Sand, C.; Rennie, R.P. Antifungal Activity of Medicinal Plant Extracts; Preliminary Screening Studies. J. Ethnopharmacol. 2008, 115, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Martínez, L.G. Preclinical Vascular Activity of an Aqueous Extract from Flowers of Calendula officinalis. J. Pharm. Pharmacol. 2020, 8, 339–344. [Google Scholar] [CrossRef]
- Athaiban, M.A. Issue 6|Page 22–28 Maha A. Athaiban, Effect of Calendula officinalis Extract Against Streptozotocin Induced Diabetes in Male Rats ISSN (Online) 2249-6084 (Print) 2250-1029 EIJPPR. Int. J. Pharm. Phytopharm. Res. 2018, 8, 22–28. [Google Scholar]
- Ebrahimi, E.; Shirali, S.; Talaei, R. The Protective Effect of Marigold Hydroalcoholic Extract in STZ-Induced Diabetic Rats: Evaluation of Cardiac and Pancreatic Biomarkers in the Serum. J. Bot. 2016, 2016, 9803928. [Google Scholar] [CrossRef]
- Gharanjik, F.; Shojaeifard, M.B.; Karbalaei, N.; Nemati, M. The Effect of Hydroalcoholic Calendula officinalis Extract on Androgen-Induced Polycystic Ovary Syndrome Model in Female Rat. Biomed Res. Int. 2022, 2022, 7402598. [Google Scholar] [CrossRef]
- Pandey, P.; Despande, B.; Sarkar, A.K. A Pharmacological Study on the Effect of Calendula officinalis Leaf Extract on Excision Wound Healing in Swiss Albino Mice. Asian Pac. J. Health Sci. 2022, 9, 109–113. [Google Scholar] [CrossRef]
- Ghodoosi, S.; Namazi, Z.; Mashhadiabbas, F.; Bakhtiari, S.; Afraz, E.S. Efficacy of 10% Calendula officinalis Extract Gel for Oral Wound Healing in Rats. J. Biol. Stud. 2022, 5, 52–63. [Google Scholar]
- Rathod, L.; Bhowmick, S.; Patel, P.; Sawant, K. Calendula Flower Extract Loaded PVA Hydrogel Sheet for Wound Management: Optimization, Characterization and in-Vivo Study. J. Drug Deliv. Sci. Technol. 2022, 68, 103035. [Google Scholar] [CrossRef]
- Lohani, A.; Morganti, P. Age-Defying and Photoprotective Potential of Geranium/Calendula Essential Oil Encapsulated Vesicular Cream on Biochemical Parameters against UVB Radiation Induced Skin Aging in Rat. Cosmetics 2022, 9, 43. [Google Scholar] [CrossRef]
- Ali, E.M.; Abd El-Moaty, H. Antifungal Activity of Achillea santolina L. and Calendula officinalis L. Essential Oils and Their Constituents Against Fungal Infection of Liver as Complication of Cyclophosphamide Therapy. J. Essent. Oil Bear. Plants 2017, 20, 1030–1043. [Google Scholar] [CrossRef]
- Mali, S.N.; Tambe, S.; Pratap, A.P.; Cruz, J.N. Molecular Modeling Approaches to Investigate Essential Oils (Volatile compounds) Interacting with Molecular Targets. In Essential Oils: Applications and Trends in Food Science and Technology; Springer: Cham, Switzerland, 2022; pp. 417–442. [Google Scholar] [CrossRef]
| Species | Major Component | Percentage | References |
|---|---|---|---|
| Calendula suffruticosa | α-linolenic acid | 24.20 | [17,30] |
| Calendula arvensis | d-cadinene (Sesquiterpines) | 15.1 | [16,31] |
| CO | α-cadinol | 64 | [31,32,33,34,35,36] |
| Calendula stellata | linalool | 34.40 | [30] |
| Calendula tripterocarpa | Phenolic compounds | 11.22 | [37,38] |
| Plant part | Groups | Active Ingredients | Ref. |
|---|---|---|---|
| Flower | Terpenoids | ψ-taraxasteol, Lupeol | [39] |
| Erythrodiol | [40] | ||
| Calenduloside | [41] | ||
| Calendula glycoside A and B | [42] | ||
| Cornulacic acid acetate | [43] | ||
| Flavonoids | Calendoflavoside Isoquercitrin, rutin | [42] | |
| Isorhamnetin, Quercetin | [44] | ||
| Narcissin, Isorhamnetin-3-O-β-D glycoside | [45] | ||
| Coumarins | Scopoletin, umbelliferone, Esculetin | [46] | |
| Volatile oils | Oplopanone, Cubenol, methyl linoleate | [47] | |
| Limonene, nerolidol, palustron p-cymene, nonanal, Sabinene, carvacrol, α-pinene, t-muurolol, geraniol | [48] | ||
| Leaves | Quinones | α-tocopherol, plastoquinone, Phylloquinone, ubiquinone | [49] |
| Root | Terpenoid | Calenduloside B | [50] |
| Author and Year | Applicability | Outcomes | Reference |
|---|---|---|---|
| Panahi et al., 2012 | Diaper dermatitis | This study suggests that topical use of CO could be used effectively for the treatment of diaper dermatitis in infants. | [101] |
| Khairnar et al., 2013 | Dental plaque and gingival inflammation | The use of CO mouthwash was able to reduce dental plaque and gingivitis. | [58] |
| Pommier et al., 2004 | Acute dermatitis | Topical use of CO prevented acute dermatitis grade 2 or higher in breast cancer patients given radiation therapy. | [102] |
| Singh and Bagewadi, 2017 | Homogeneous leukoplakia | The use of CO extract gel was effective in reducing the size of the lesion. | [103] |
| Babaee et al., 2013 | Oropharyngeal mucositis | The use of CO extract gel was able to reduce the intensity of oropharyngeal mucositis in patients undergoing radiotherapy during treatment for head and neck cancer. | [104] |
| Giostri et al., 2022 | Acute wounds on hand | CO induced more rapid secondary intention healing in hand and finger wounds | [105] |
| Buzzi et al., 2016 | Venous leg ulcer healing | Patients with ulcers treated with CO extract had a significant 4-fold increase in percentage healing velocity per week, compared with the control group. | [106] |
| De Angelis et al., 2022 | Episiotomy | Women who used CO ointment after episiotomy had significantly lower pain level from the second day and during the entire follow-up. In addition, CO ointment also improves wound healing in terms of redness and edema. | [107] |
| Saffari et al., 2017 | Vaginal candidiasis | Treatment of vaginal candidiasis with CO vaginal lotion seems to be successful. | [108] |
| Pazhohideh et al., 2018 | Bacterial vaginosis | CO was used successfully and without any negative side effects to treat bacterial vaginosis in women of reproductive age. | [109] |
| Therapeutic Application | Model | Results/Clinical Outcomes | Ref |
|---|---|---|---|
| Cardiovascular | Wister rats | In endothelium-depleted rat aortic rings pre-contracted with 60 mmol/L of KCl, the experiment revealed that floral extract of CO caused a concentration-dependent relaxation. | [141] |
| Hepatoprotective | Albino rats | The results showed that feeding on CO had a protective effect on the liver against CCl4 and had improvement effect on liver. | [29] |
| Antidiabetic | Albino wister rats | Significant changes occurred in glucose and insulin level (135.32 ± 2.43 and 88.42 ± 2.17, respectively) at a dose of 200 mg/kg/day in a dose-dependent manner as compared to the control positive group level (211.76 ± 3.95 and 134.82 ± 2.95) (p < 0.05). | [142] |
| Antidiabetic | Wister rats | In diabetic rats, the results revealed that CO was able to normalize levels of creatine kinase (CK-MB and total CK), amylase, and lipase. This allowed for a reduction in the negative effects of diabetes. | [143] |
| Polycystic ovary syndrome | Sprague–Dawley rats | After 21 days, the results demonstrated that rats given CO subcutaneous injections of DHEA were successfully induced with PCOS condition. | [144] |
| Wound healing | Swiss albino mice | In a wound model including excision, mice that had been treated with an extract of the leaves of CO showed a significant reduction in both the wound area and the time it took to epithelize. | [145] |
| Oral wound healing | Wister rats | The application of 10% CO gel improves wound contraction and enhances healing. | [146] |
| Wound healing | Wister rats | The healing percent of the lesion area ranged from 7.69% to 87.01% with CO-flower-extract-loaded hydrogel sheet. | [147] |
| Age-defying and photoprotective | Albino rats | According to the findings, a pre-treatment with GEO/CEO-encapsulated vesicular cream formulations significantly reversed the detrimental biochemical alterations and protected the skin from the deteriorating effects of UVB radiation. | [148] |
| Antifungal | Swiss albino rats | According to the findings, CO essential oils and their combination provide a significant advantage in terms of lowering the risk of fungal infection after chemotherapy with cyclophosphamide. | [149] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahane, K.; Kshirsagar, M.; Tambe, S.; Jain, D.; Rout, S.; Ferreira, M.K.M.; Mali, S.; Amin, P.; Srivastav, P.P.; Cruz, J.; et al. An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L. Pharmaceuticals 2023, 16, 611. https://doi.org/10.3390/ph16040611
Shahane K, Kshirsagar M, Tambe S, Jain D, Rout S, Ferreira MKM, Mali S, Amin P, Srivastav PP, Cruz J, et al. An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L. Pharmaceuticals. 2023; 16(4):611. https://doi.org/10.3390/ph16040611
Chicago/Turabian StyleShahane, Kiran, Madhuri Kshirsagar, Srushti Tambe, Divya Jain, Srutee Rout, Maria Karolina Martins Ferreira, Suraj Mali, Purnima Amin, Prem Prakash Srivastav, Jorddy Cruz, and et al. 2023. "An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L." Pharmaceuticals 16, no. 4: 611. https://doi.org/10.3390/ph16040611
APA StyleShahane, K., Kshirsagar, M., Tambe, S., Jain, D., Rout, S., Ferreira, M. K. M., Mali, S., Amin, P., Srivastav, P. P., Cruz, J., & Lima, R. R. (2023). An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L. Pharmaceuticals, 16(4), 611. https://doi.org/10.3390/ph16040611










