Development of (4-Phenylamino)quinazoline Alkylthiourea Derivatives as Novel NF-κB Inhibitors
Abstract
:1. Introduction
2. Results and Discussion
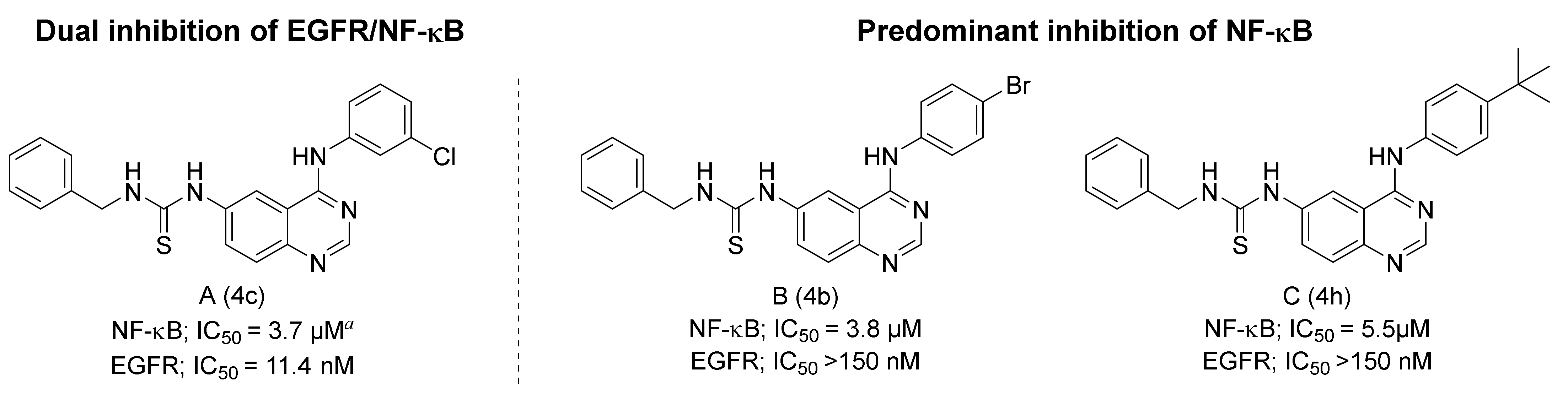
2.1. Compound Design
2.2. Chemistry
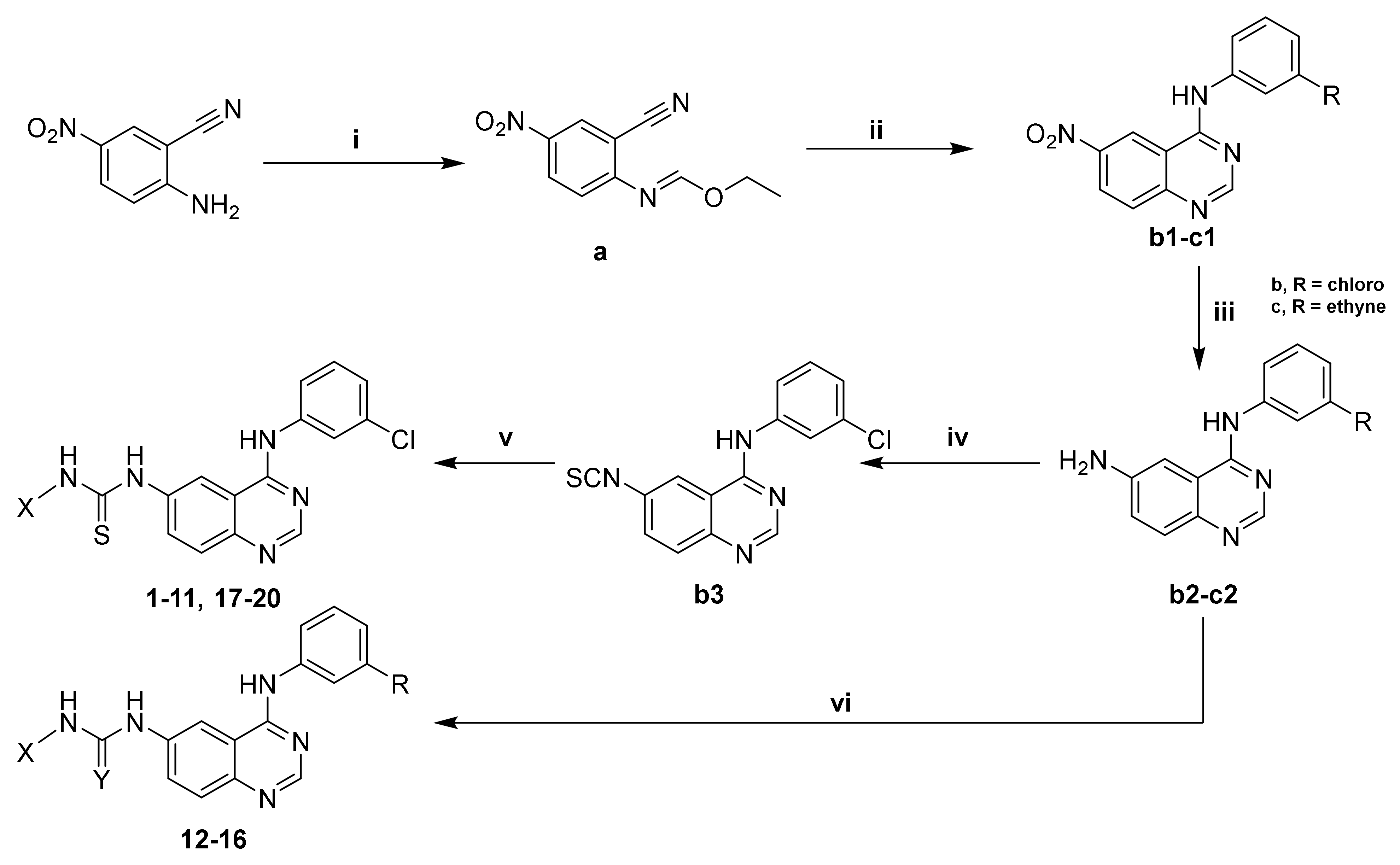
| Cpd. | X | Cpd. | X | R | Y |
| 1 |  | 11 | 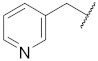 | - | - |
| 2 | 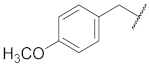 | 12 | 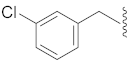 | Cl | O |
| 3 | 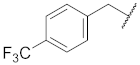 | 13 | 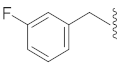 | Cl | O |
| 4 | 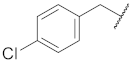 | 14 | 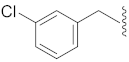 | 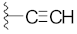 | O |
| 5 | 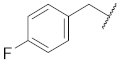 | 15 | 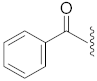 | Cl | O |
| 6 | 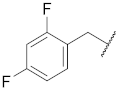 | 16 | 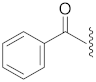 | Cl | S |
| 7 | 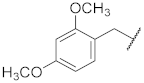 | 17 | 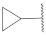 | - | - |
| 8 | 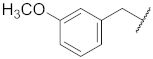 | 18 | 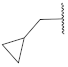 | - | - |
| 9 | 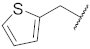 | 19 | 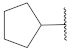 | - | - |
| 10 | 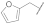 | 20 |  | - | - |
2.3. Structure–Activity Relationships (SAR)
2.3.1. Benzylthiourea Quinazoline Derivatives
2.3.2. Alkylthiourea Quinazoline Derivatives
2.4. Evaluation of the Anti-Inflammatory Activities in Macrophage-like dTHP−1 Cells
2.5. Investigation of the Compounds’ Mode of Action in dTHP−1 Cells
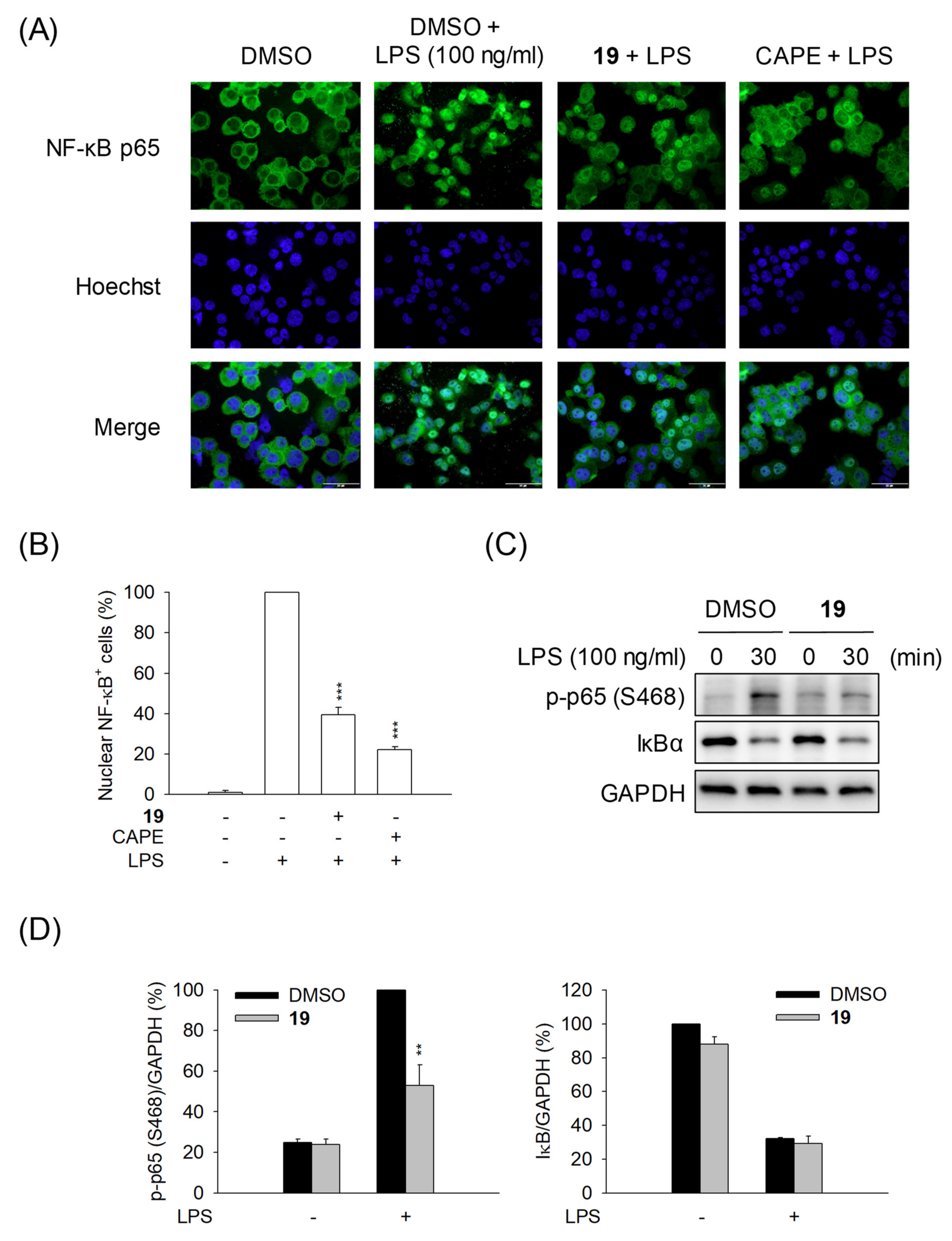
2.5.1. The Alkylthiourea Quinazoline Derivative 19 Inhibits the Translocation of NF-κB to the Nucleus
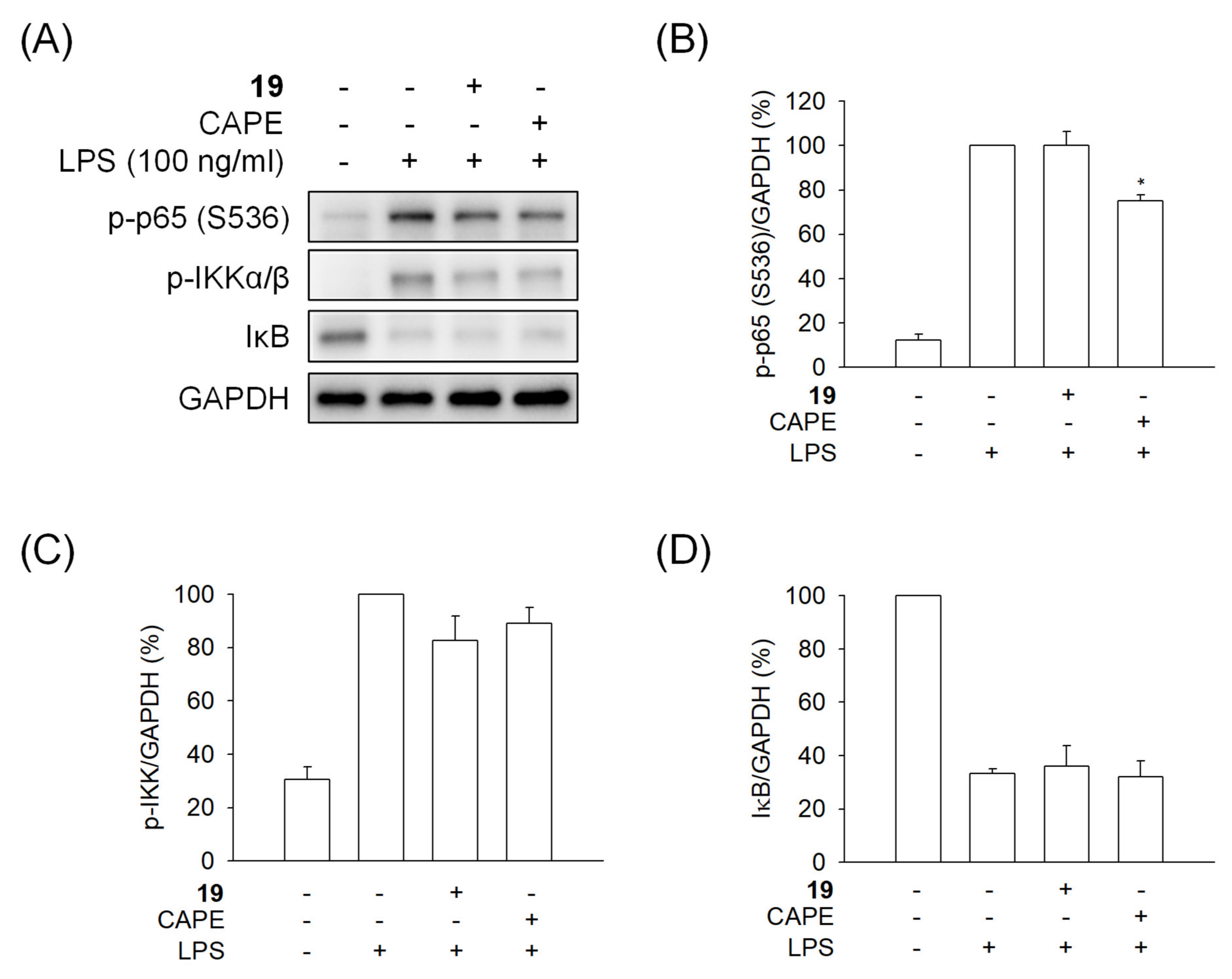
2.5.2. Compound 19 Does Not Inhibit IκB Degradation and NF-κB Release
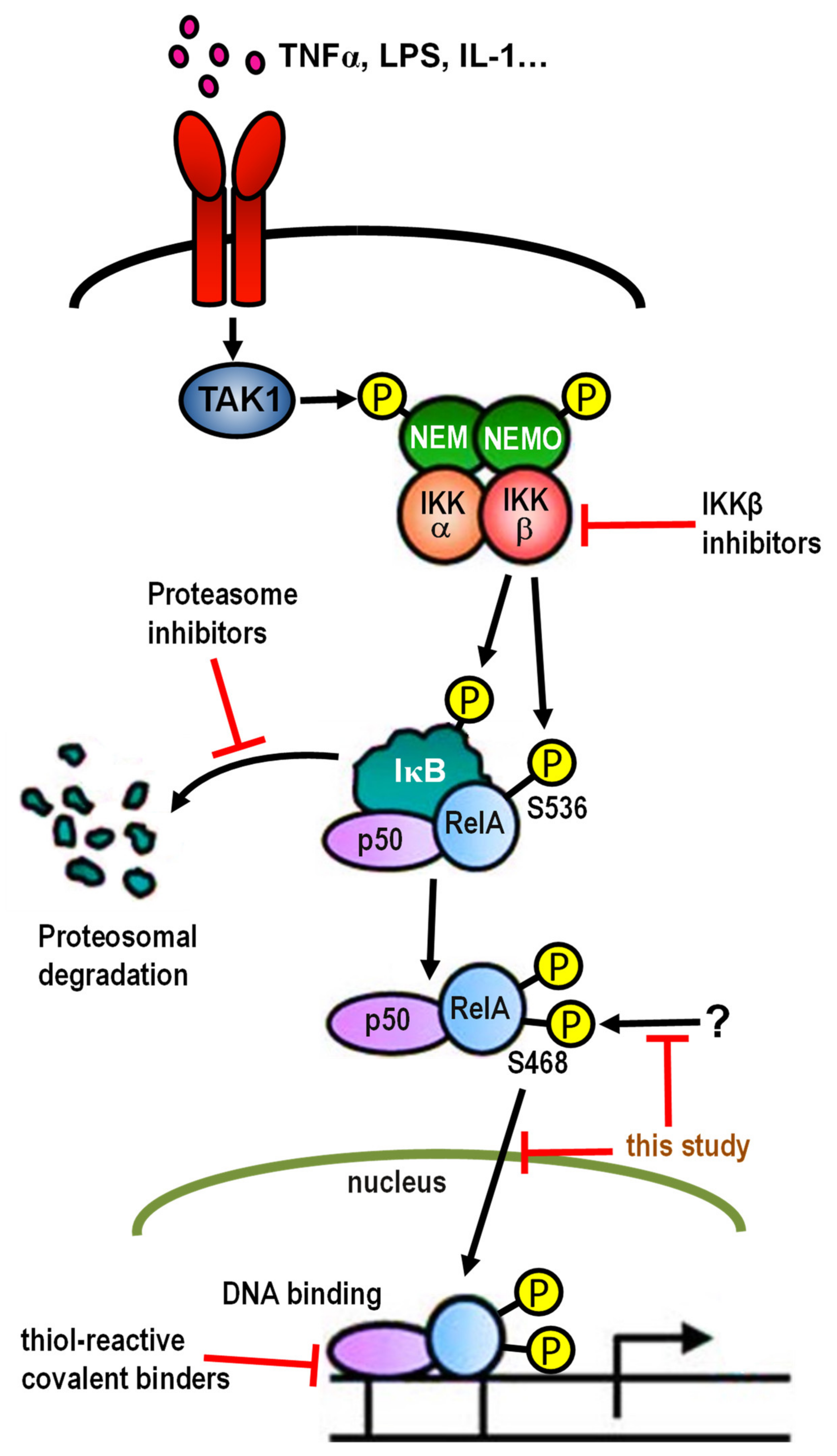
2.5.3. Compound 19 Inhibits the Phosphorylation of p65-Ser468
2.5.4. Compound 19 Has No Effect on the Kinases Involved in NF-κB Activation
3. Materials and Methods
3.1. Chemistry
General Synthetic Procedures and Experimental Details
3.2. Biological Methods
3.2.1. Cell Culture
3.2.2. NF-κB Luciferase Reporter Assay
3.2.3. Cell Viability
3.2.4. ELISA Assay
3.2.5. Immunofluorescent Staining
3.2.6. Western Blotting
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Zhu, Y.; Xian, X.; Wang, Z.; Bi, Y.; Chen, Q.; Han, X.; Tang, D.; Chen, R. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules 2018, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Guiteras, R.; Flaquer, M.; Cruzado, J.M. Macrophage in Chronic Kidney Disease. Clin. Kidney J. 2016, 9, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Nakae, J.; Watanabe, N.; Kikuchi, T.; Tateya, S.; Tamori, Y.; Kaneko, M.; Abe, T.; Onodera, M.; Itoh, H. Colonic Pro-Inflammatory Macrophages Cause Insulin Resistance in an Intestinal Ccl2/Ccr2-Dependent Manner. Cell Metab. 2016, 24, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Falconer, J.; Murphy, A.N.; Young, S.P.; Clark, A.R.; Tiziani, S.; Guma, M.; Buckley, C.D. Review: Synovial Cell Metabolism and Chronic Inflammation in Rheumatoid Arthritis. Arthritis Rheumatol. 2018, 70, 984–999. [Google Scholar] [CrossRef]
- Rainsford, K. Profile and Mechanisms of Gastrointestinal and Other Side Effects of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Am. J. Med. 1999, 107, 27–35. [Google Scholar] [CrossRef]
- Riley, T.R.; George, M.D. Risk for Infections with Glucocorticoids and DMARDs in Patients with Rheumatoid Arthritis. RMD Open 2021, 7, e001235. [Google Scholar] [CrossRef]
- Aithal, G.P. Hepatotoxicity Related to Antirheumatic Drugs. Nat. Rev. Rheumatol. 2011, 7, 139–150. [Google Scholar] [CrossRef]
- Fardet, L.; Fève, B. Systemic Glucocorticoid Therapy: A Review of Its Metabolic and Cardiovascular Adverse Events. Drugs 2014, 74, 1731–1745. [Google Scholar] [CrossRef]
- Oray, M.; Abu Samra, K.; Ebrahimiadib, N.; Meese, H.; Foster, C.S. Long-Term Side Effects of Glucocorticoids. Expert Opin. Drug Saf. 2016, 15, 457–465. [Google Scholar] [CrossRef]
- Mohan, N.; Edwards, E.T.; Cupps, T.R.; Oliverio, P.J.; Sandberg, G.; Crayton, H.; Richert, J.R.; Siegel, J.N. Demyelination Occurring during Anti-Tumor Necrosis Factor? Therapy for Inflammatory Arthritides. Arthritis Rheum. 2001, 44, 2862–2869. [Google Scholar] [CrossRef]
- Kemanetzoglou, E.; Andreadou, E. CNS Demyelination with TNF-α Blockers. Curr. Neurol. Neurosci. Rep. 2017, 17, 36. [Google Scholar] [CrossRef]
- Atiqi, S.; Hooijberg, F.; Loeff, F.C.; Rispens, T.; Wolbink, G.J. Immunogenicity of TNF-Inhibitors. Front. Immunol. 2020, 11, 312. [Google Scholar] [CrossRef]
- Roda, G.; Jharap, B.; Neeraj, N.; Colombel, J.-F. Loss of Response to Anti-TNFs: Definition, Epidemiology, and Management. Clin. Transl. Gastroenterol. 2016, 7, e135. [Google Scholar] [CrossRef]
- Kalden, J.R.; Schulze-Koops, H. Immunogenicity and Loss of Response to TNF Inhibitors: Implications for Rheumatoid Arthritis Treatment. Nat. Rev. Rheumatol. 2017, 13, 707–718. [Google Scholar] [CrossRef]
- Christopher, J.A.; Avitabile, B.G.; Bamborough, P.; Champigny, A.C.; Cutler, G.J.; Dyos, S.L.; Grace, K.G.; Kerns, J.K.; Kitson, J.D.; Mellor, G.W.; et al. The Discovery of 2-Amino-3,5-Diarylbenzamide Inhibitors of IKK-α and IKK-β Kinases. Bioorg. Med. Chem. Lett. 2007, 17, 3972–3977. [Google Scholar] [CrossRef]
- Moarbess, G.; Guichou, J.-F.; Paniagua-Gayraud, S.; Chouchou, A.; Marcadet, O.; Leroy, F.; Ruédas, R.; Cuq, P.; Deleuze-Masquéfa, C.; Bonnet, P.-A. New IKK Inhibitors: Synthesis of New Imidazo [1, 2-a] Quinoxaline Derivatives Using Microwave Assistance and Biological Evaluation as IKK Inhibitors. Eur. J. Med. Chem. 2016, 115, 268–274. [Google Scholar] [CrossRef]
- Burke, J.R.; Pattoli, M.A.; Gregor, K.R.; Brassil, P.J.; MacMaster, J.F.; McIntyre, K.W.; Yang, X.; Iotzova, V.S.; Clarke, W.; Strnad, J. BMS-345541 Is a Highly Selective Inhibitor of IκB Kinase That Binds at an Allosteric Site of the Enzyme and Blocks NF-ΚB-Dependent Transcription in Mice. J. Biol. Chem. 2003, 278, 1450–1456. [Google Scholar] [CrossRef]
- Liddle, J.; Bamborough, P.; Barker, M.D.; Campos, S.; Cousins, R.P.C.; Cutler, G.J.; Hobbs, H.; Holmes, D.S.; Ioannou, C.; Mellor, G.W.; et al. 4-Phenyl-7-Azaindoles as Potent and Selective IKK2 Inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 2504–2508. [Google Scholar] [CrossRef]
- Elkamhawy, A.; Kim, N.Y.; Hassan, A.H.E.; Park, J.; Yang, J.-E.; Oh, K.-S.; Lee, B.H.; Lee, M.Y.; Shin, K.J.; Lee, K.-T. Design, Synthesis and Biological Evaluation of Novel Thiazolidinedione Derivatives as Irreversible Allosteric IKK-β Modulators. Eur. J. Med. Chem. 2018, 157, 691–704. [Google Scholar] [CrossRef]
- Elkamhawy, A.; Kim, N.Y.; Hassan, A.H.E.; Park, J.; Paik, S.; Yang, J.-E.; Oh, K.-S.; Lee, B.H.; Lee, M.Y.; Shin, K.J. Thiazolidine-2, 4-Dione-Based Irreversible Allosteric IKK-β Kinase Inhibitors: Optimization into in Vivo Active Anti-Inflammatory Agents. Eur. J. Med. Chem. 2020, 188, 111955. [Google Scholar] [CrossRef]
- Prescott, J.A.; Cook, S.J. Targeting IKKβ in Cancer: Challenges and Opportunities for the Therapeutic Utilisation of IKKβ Inhibitors. Cells 2018, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Jaspars, M.; Dicato, M.; Diederich, M. Marine Natural Products as Targeted Modulators of the Transcription Factor NF-ΚB. Biochem. Pharmacol. 2008, 75, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Lyß, G.; Knorre, A.; Schmidt, T.J.; Pahl, H.L.; Merfort, I. The Anti-Inflammatory Sesquiterpene Lactone Helenalin Inhibits the Transcription Factor NF-ΚB by Directly Targeting P65. J. Biol. Chem. 1998, 273, 33508–33516. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, D.J. Marine Pharmacology. Antonie van Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2000, 77, 135–145. [Google Scholar] [CrossRef]
- Aggarwal, B.B. Targeting Inflammation-Induced Obesity and Metabolic Diseases by Curcumin and Other Nutraceuticals. Annu. Rev. Nutr. 2010, 30, 173–199. [Google Scholar] [CrossRef]
- Xie, Y.-L.; Chu, J.-G.; Jian, X.-M.; Dong, J.-Z.; Wang, L.-P.; Li, G.-X.; Yang, N.-B. Curcumin Attenuates Lipopolysaccharide/d-Galactosamine-Induced Acute Liver Injury by Activating Nrf2 Nuclear Translocation and Inhibiting NF-KB Activation. Biomed. Pharmacother. 2017, 91, 70–77. [Google Scholar] [CrossRef]
- Natarajan, K.; Singh, S.; Burke, T.R.; Grunberger, D.; Aggarwal, B.B. Caffeic Acid Phenethyl Ester Is a Potent and Specific Inhibitor of Activation of Nuclear Transcription Factor NF-Kappa B. Proc. Natl. Acad. Sci. USA 1996, 93, 9090–9095. [Google Scholar] [CrossRef]
- Lataifeh, A.R.; Nusair, A. Fatal Pulmonary Toxicity Due to Carfilzomib (KyprolisTM). J. Oncol. Pharm. Pract. 2016, 22, 720–724. [Google Scholar] [CrossRef]
- Arastu-Kapur, S.; Anderl, J.L.; Kraus, M.; Parlati, F.; Shenk, K.D.; Lee, S.J.; Muchamuel, T.; Bennett, M.K.; Driessen, C.; Ball, A.J. Nonproteasomal Targets of the Proteasome Inhibitors Bortezomib and Carfilzomib: A Link to Clinical Adverse Events. Clin. Cancer Res. 2011, 17, 2734–2743. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Roussou, M.; Gavriatopoulou, M.; Psimenou, E.; Ziogas, D.; Eleutherakis-Papaiakovou, E.; Fotiou, D.; Migkou, M.; Kanellias, N.; Panagiotidis, I. Cardiac and Renal Complications of Carfilzomib in Patients with Multiple Myeloma. Blood Adv. 2017, 1, 449–454. [Google Scholar] [CrossRef]
- Atallah-Yunes, S.A.; Soe, M.H. Drug-Induced Thrombotic Microangiopathy Due to Cumulative Toxicity of Ixazomib. Case Rep. Hematol. 2018, 2018, 7063145. [Google Scholar] [CrossRef]
- Ramadass, V.; Vaiyapuri, T.; Tergaonkar, V. Small Molecule NF-ΚB Pathway Inhibitors in Clinic. Int. J. Mol. Sci. 2020, 21, 5164. [Google Scholar] [CrossRef]
- Holcmann, M.; Sibilia, M. Mechanisms Underlying Skin Disorders Induced by EGFR Inhibitors. Mol. Cell. Oncol. 2015, 2, e1004969. [Google Scholar] [CrossRef]
- Lacouture, M.E. Mechanisms of Cutaneous Toxicities to EGFR Inhibitors. Nat. Rev. Cancer 2006, 6, 803–812. [Google Scholar] [CrossRef]
- Hamed, M.M.; Darwish, S.S.; Herrmann, J.; Abadi, A.H.; Engel, M. First Bispecific Inhibitors of the Epidermal Growth Factor Receptor Kinase and the NF-ΚB Activity as Novel Anticancer Agents. J. Med. Chem. 2017, 60, 2853–2868. [Google Scholar] [CrossRef]
- Darwish, S.S.; Abdel-Halim, M.; Salah, M.; Abadi, A.H.; Becker, W.; Engel, M. Development of Novel 2,4-Bispyridyl Thiophene–Based Compounds as Highly Potent and Selective Dyrk1A Inhibitors. Part I: Benzamide and Benzylamide Derivatives. Eur. J. Med. Chem. 2018, 157, 1031–1050. [Google Scholar] [CrossRef]
- Darwish, S.S.; Abdel-Halim, M.; ElHady, A.K.; Salah, M.; Abadi, A.H.; Becker, W.; Engel, M. Development of Novel Amide–Derivatized 2,4-Bispyridyl Thiophenes as Highly Potent and Selective Dyrk1A Inhibitors. Part II: Identification of the Cyclopropylamide Moiety as a Key Modification. Eur. J. Med. Chem. 2018, 158, 270–285. [Google Scholar] [CrossRef]
- Chanput, W.; Mes, J.J.; Wichers, H.J. THP-1 Cell Line: An in Vitro Cell Model for Immune Modulation Approach. Int. Immunopharmacol. 2014, 23, 37–45. [Google Scholar] [CrossRef]
- Bosshart, H.; Heinzelmann, M. THP-1 Cells as a Model for Human Monocytes. Ann. Transl. Med. 2016, 4, 438. [Google Scholar] [CrossRef]
- Maeß, M.B.; Wittig, B.; Cignarella, A.; Lorkowski, S. Reduced PMA Enhances the Responsiveness of Transfected THP-1 Macrophages to Polarizing Stimuli. J. Immunol. Methods 2014, 402, 76–81. [Google Scholar] [CrossRef]
- Liu, X.; Yin, S.; Chen, Y.; Wu, Y.; Zheng, W.; Dong, H.; Bai, Y.; Qin, Y.; Li, J.; Feng, S. LPS-induced Proinflammatory Cytokine Expression in Human Airway Epithelial Cells and Macrophages via NF-κB, STAT3 or AP-1 Activation. Mol. Med. Rep. 2018, 17, 5484–5491. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.M.; Temkin, V.; Shi, B.; Pagliari, L.; Daniel, S.; Ferran, C.; Pope, R.M. TNFα-Induced Macrophage Death via Caspase-Dependent and Independent Pathways. Apoptosis 2009, 14, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Giridharan, S.; Srinivasan, M. Mechanisms of NF-ΚB P65 and Strategies for Therapeutic Manipulation. J. Inflamm. Res. 2018, 11, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, C.Y.; Barberi, T.J.; Ghosh, P.; Longo, D.L. Phosphorylation of RelA/P65 on Serine 536 Defines an IκBα-Independent NF-ΚB Pathway. J. Biol. Chem. 2005, 280, 34538–34547. [Google Scholar] [CrossRef]
- Mattioli, I.; Geng, H.; Sebald, A.; Hodel, M.; Bucher, C.; Kracht, M.; Schmitz, M.L. Inducible Phosphorylation of NF-ΚB P65 at Serine 468 by T Cell Costimulation Is Mediated by IKKϵ. J. Biol. Chem. 2006, 281, 6175–6183. [Google Scholar] [CrossRef]
- Christian, F.; Smith, E.L.; Carmody, R.J. The Regulation of NF-ΚB Subunits by Phosphorylation. Cells 2016, 5, 12. [Google Scholar] [CrossRef]
- Renner, F.; Moreno, R.; Schmitz, M.L. SUMOylation-Dependent Localization of IKKɛ in PML Nuclear Bodies Is Essential for Protection against DNA-Damage-Triggered Cell Death. Mol. Cell 2010, 37, 503–515. [Google Scholar] [CrossRef]
- Kaur, S.; Bansal, Y.; Kumar, R.; Bansal, G. A Panoramic Review of IL-6: Structure, Pathophysiological Roles and Inhibitors. Bioorg. Med. Chem. 2020, 28, 115327. [Google Scholar] [CrossRef]
- Pope, R.M. Apoptosis as a Therapeutic Tool in Rheumatoid Arthritis. Nat. Rev. Immunol. 2002, 2, 527–535. [Google Scholar] [CrossRef]
- Mahida, Y.R. The Key Role of Macrophages in the Immunopathogenesis of Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2000, 6, 21–33. [Google Scholar] [CrossRef]
- Sarzi-Puttini, P.; Atzeni, F.; Doria, A.; Iaccarino, L.; Turiel, M. Tumor Necrosis Factor-a, Biologic Agents and Cardiovascular Risk. Lupus 2005, 14, 780–784. [Google Scholar] [CrossRef]
- Martinet, W.; Coornaert, I.; Puylaert, P.; De Meyer, G.R.Y. Macrophage Death as a Pharmacological Target in Atherosclerosis. Front. Pharmacol. 2019, 10, 306. [Google Scholar] [CrossRef]
- Colletti, L.M.; Remick, D.G.; Burtch, G.D.; Kunkel, S.L.; Strieter, R.M.; Campbell, D.A. Role of Tumor Necrosis Factor-Alpha in the Pathophysiologic Alterations after Hepatic Ischemia/Reperfusion Injury in the Rat. J. Clin. Investig. 1990, 85, 1936–1943. [Google Scholar] [CrossRef]
- Gonzalez-Amaro, R.; Garcia-Monzon, C.; Garcia-Buey, L.; Moreno-Otero, R.; Alonso, J.; Yagüe, E.; Pivel, J.P.; Lopez-Cabrera, M.; Fernandez-Ruiz, E.; Sanchez-Madrid, F. Induction of Tumor Necrosis Factor Alpha Production by Human Hepatocytes in Chronic Viral Hepatitis. J. Exp. Med. 1994, 179, 841–848. [Google Scholar] [CrossRef]
- Velazquez, R.; Meechoovet, B.; Ow, A.; Foley, C.; Shaw, A.; Smith, B.; Oddo, S.; Hulme, C.; Dunckley, T. Chronic Dyrk1 Inhibition Delays the Onset of AD-Like Pathology in 3xTg-AD Mice. Mol. Neurobiol. 2019, 56, 8364–8375. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, N.; Diao, Z.; Chen, Y.; Zhang, Y. Therapeutic Potential of TNFα Inhibitors in Chronic Inflammatory Disorders: Past and Future. Genes Dis. 2021, 8, 38–47. [Google Scholar] [CrossRef]
- Olona, A.; Hateley, C.; Guerrero, A.; Ko, J.; Johnson, M.R.; Anand, P.K.; Thomas, D.; Gil, J.; Behmoaras, J. Cardiac Glycosides Cause Cytotoxicity in Human Macrophages and Ameliorate White Adipose Tissue Homeostasis. Br. J. Pharmacol. 2021, 179, 1874–1886. [Google Scholar] [CrossRef]
- Chu, F.; Shi, M.; Lang, Y.; Chao, Z.; Jin, T.; Cui, L.; Zhu, J. Adoptive Transfer of Immunomodulatory M2 Macrophages Suppresses Experimental Autoimmune Encephalomyelitis in C57BL/6 Mice via Blockading NF-κB Pathway. Clin. Exp. Immunol. 2021, 204, 199–211. [Google Scholar] [CrossRef]
- Martinet, W.; Verheye, S.; De Meyer, G.R.Y. Selective Depletion of Macrophages in Atherosclerotic Plaques via Macrophage-Specific Initiation of Cell Death. Trends Cardiovasc. Med. 2007, 17, 69–75. [Google Scholar] [CrossRef]
- Gorina, R.; Font-Nieves, M.; Márquez-Kisinousky, L.; Santalucia, T.; Planas, A.M. Astrocyte TLR4 Activation Induces a Proinflammatory Environment through the Interplay between MyD88-dependent NFκB Signaling, MAPK, and Jak1/Stat1 Pathways. Glia 2011, 59, 242–255. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, S.H.; Shin, S.Y.; Lee, Y.H. Clozapine Reduces Toll-like Receptor 4/NF-ΚB-Mediated Inflammatory Responses through Inhibition of Calcium/Calmodulin-Dependent Akt Activation in Microglia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2018, 81, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Gong, R.; Rifai, A.; Ge, Y.; Chen, S.; Dworkin, L.D. Hepatocyte Growth Factor Suppresses Proinflammatory NFκB Activation through GSK3β Inactivation in Renal Tubular Epithelial Cells. J. Biol. Chem. 2008, 283, 7401–7410. [Google Scholar] [CrossRef] [PubMed]


| Cpd. | X | a,b NF-κB Luciferase Activity | c Cell Viability (%) | Recombinant EGFR Kinase Activity (%inh at 150 nM) | ||
|---|---|---|---|---|---|---|
| Inh % | IC50 (μM) | HEK293 | A549 | |||
| Ad | 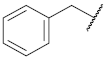 | 94.6 ± 1.5 ** | 0.79 | 72.4 ± 3.9 *** | 81.7 ± 1.2 | 84.8 |
| 1 | 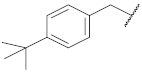 | 41.6 ± 3.8 *** | >10 | 73.9 ± 4.6 ** | 91.1 ± 3.4 | 42 |
| 2 | 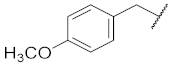 | 94.6 ± 0.1 *** | 1.7 | 100.9 ± 7.4 | 95.4 ± 1.9 | 67 |
| 3 | 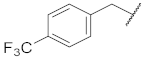 | 46.5 ± 4.9 *** | >10 | 68.9 ± 6.4 ** | 73.4 ± 4.3 | 70 |
| 4 | 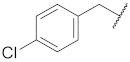 | 31.9 ± 9.6 ** | >10 | 95.9 ± 3.4 | 98.1 ± 1.9 | 56 |
| 5 |  | 79.1 ± 0.8 *** | 1.9 | 67.0 ± 6.1 ** | 65.3 ± 1.1 | 72 |
| 6 | 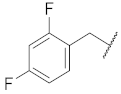 | 78.4 ± 2.7 ** | 1.6 | 85.5 ± 6.4 * | 75.5 ± 2.9 | 68 |
| 7 | 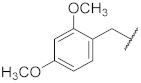 | 27.1 ± 3.4 *** | >10 | 85.9 ± 2.5 * | 99.5 ± 1.4 | 48 |
| 8 |  | 76.1 ± 2.6 *** | 2.3 | 100.2 ± 6.9 | 101.1 ± 2.3 | 40 |
| 9 | 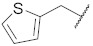 | 98.0 ± 0.4 *** | 2.8 | 13.9 ± 0.7 *** | 50.7 ± 2.4 | 69.8 |
| 10 | 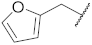 | 76.1 ± 2.3 *** | 4.3 | 71.7 ± 3.1 *** | 74.4 ± 11.7 | 79.1 |
| 11 |  | 44.7 ± 5.8 ** | >10 | 62.8 ± 4.0 *** | 98.9 ± 4.1 | 66 |
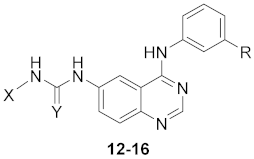
| Cpd. | R | X | Y | a,b NF-κB Luciferase Activity | c Cell Viability (%) | Recombinant EGFR Kinase Activity (%inh at 150 nM) | ||
|---|---|---|---|---|---|---|---|---|
| Inh % | IC50 (μM) | HEK293 | A549 | |||||
| 12 | Cl | 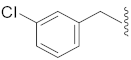 | O | 42.7 ± 9.3 * | >10 | 113.6 ± 3.2 | 100.7 ± 4.2 | 34.6 |
| 13 | Cl | 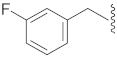 | O | 53.3 ± 10.0 ** | 8.1 | 95.5 ± 6.1 | 103.5 ± 5.0 | 23.3 |
| 14 | 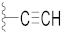 | 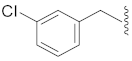 | O | 58.5 ± 0.9 *** | 8.0 | 110.2 ± 2.7 | 98.6 ± 3.0 | 44.5 |
| 15 | Cl | 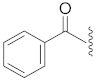 | O | 48.3 ± 9.3 * | >10 | 113.8 ± 2.6 | 100.1 ± 4.5 | 26 |
| 16 | Cl | 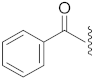 | S | 58.2 ± 5.2 *** | 7.0 | 101.5 ± 4.0 | 102.3 ± 1.6 | 48.9 |
| Cpd. | X | a, b NF-κB Luciferase Activity | c Cell Viability (%) | Recombinant EGFR Kinase Activity (%inh at 150 nM) | ||
|---|---|---|---|---|---|---|
| Inh % | IC50 (μM) | HEK293 | A549 | |||
| 17 | 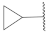 | 47.0 ± 5.3 *** | >10 | 103.0 ± 8.4 | 102.6 ± 0.2 | 36.5 |
| 18 | 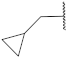 | 89.9 ± 1.5 *** | 2.8 | 75.7 ± 3.8 | 79.1 ± 0.6 | 59 |
| 19 | 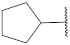 | 92.5 ± 0.8 *** | 2.3 | 118.5 ± 11.4 | 99.9 ± 1.6 | 23 |
| 20 | 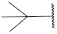 | 75.7 ± 3.1 ** | 2.0 | 99.7 ± 8.0 | 98.1 ± 3.1 | 1.5 |
| Cpd. | TNFα Production | IL-6 Production | ||||
|---|---|---|---|---|---|---|
| Inh % (7.5 μM) | Inh % (15 μM) | IC50 (μM) | Inh % (7.5 μM) | Inh % (15 μM) | IC50 (μM) | |
| 2 | 32.5 ± 8.5 *** | 60.4 ± 1.8 *** | 10.2 ± 0.27 | 76.2 ± 13.3 ** | 87.5 ± 6.8 *** | 1.2 ± 0.12 |
| 6 | 61.1 ± 5.8 ** | 76.6 ± 3.8 *** | 4.4 ± 0.85 | 97.6 ± 0.6 *** | 98.2 ± 1.3 *** | 0.36 ± 0.07 |
| 17 | 19.0 ± 8.0 | 62.0 ± 4.2 ** | 12.3 ± 1.0 | 86.0 ± 2.3 ** | 95.8 ± 0.33 *** | 0.72 ± 0.17 |
| 18 | 31.0 ± 4.5 ** | 65.9 ± 7.9 ** | 11.5 ± 1.7 | 78.9 ± 14.8 ** | 89.4 ± 7.0 *** | 1.1 ± 0.36 |
| 19 | 82.5 ± 5.6 *** | 91.5 ± 2.5 *** | 4.0 ± 0.20 | 92.7 ± 5.5 *** | 98.3 ± 0.28 *** | 0.84 ± 0.12 |
| 20 | 57.4 ± 7.2 *** | 89.9 ± 0.88 *** | 5.1 ± 0.43 | 91.8 ± 3.5 *** | 98.7 ± 1.2 *** | 0.94 ± 0.11 |
| 7 | 37.0 ± 4.9 * | 50.3 ± 5.5 * | 11.9 ± 0.70 | 71.0 ± 3.4 ** | 84.0 ± 0.82 *** | 1.2 ± 0.16 |
| CAPE | ND | ND | 11.4 ± 0.21 | ND | ND | 1.1 ± 0.36 |
| Cpd. | 7.5 μM | 5 μM | ||
|---|---|---|---|---|
| - | LPS | - | LPS | |
| 2 | 99.2 ± 1.5 | 57.5 ± 1.4 *** | 94.2 ± 1.6 * | 78.8 ± 5.3 * |
| 6 | 62.0 ± 14.5 | 6.2 ± 0.12 *** | 43.6 ± 9.0 ** | 5.0 ± 0.35 *** |
| 17 | 94.3 ± 1.9 * | 84.3 ± 2.1 ** | 84.5 ± 4.8 * | 62.0 ± 3.4 *** |
| 18 | 82.7 ± 10.3 | 34.9 ± 1.6 *** | 87.6 ± 2.5 ** | 11.2 ± 3.3 *** |
| 19 | 60.3 ± 0.85 *** | 6.7 ± 0.14 *** | 30.0 ± 1.3 *** | 4.9 ± 0.38 *** |
| 20 | 78.0 ± 0.09 *** | 32.5 ± 0.89 *** | 42.5 ± 10.6 ** | 12.5 ± 0.23 *** |
| 7 | 94.4 ± 0.61 *** | 94.7 ± 1.2 ** | 96.8 ± 4.2 | 91.9 ± 4.2 |
| CAPE (10 μM) | 97.8 ± 0.41 ** | 52.2 ± 9.7 ** | ND | ND |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Darwish, S.S.; Chen, P.-J.; Hamed, M.M.; Wagdy, R.A.; Chen, S.-H.; Abadi, A.H.; Abdel-Halim, M.; Hwang, T.-L.; Engel, M. Development of (4-Phenylamino)quinazoline Alkylthiourea Derivatives as Novel NF-κB Inhibitors. Pharmaceuticals 2022, 15, 778. https://doi.org/10.3390/ph15070778
Darwish SS, Chen P-J, Hamed MM, Wagdy RA, Chen S-H, Abadi AH, Abdel-Halim M, Hwang T-L, Engel M. Development of (4-Phenylamino)quinazoline Alkylthiourea Derivatives as Novel NF-κB Inhibitors. Pharmaceuticals. 2022; 15(7):778. https://doi.org/10.3390/ph15070778
Chicago/Turabian StyleDarwish, Sarah S., Po-Jen Chen, Mostafa M. Hamed, Reem A. Wagdy, Shun-Hua Chen, Ashraf H. Abadi, Mohammad Abdel-Halim, Tsong-Long Hwang, and Matthias Engel. 2022. "Development of (4-Phenylamino)quinazoline Alkylthiourea Derivatives as Novel NF-κB Inhibitors" Pharmaceuticals 15, no. 7: 778. https://doi.org/10.3390/ph15070778
APA StyleDarwish, S. S., Chen, P.-J., Hamed, M. M., Wagdy, R. A., Chen, S.-H., Abadi, A. H., Abdel-Halim, M., Hwang, T.-L., & Engel, M. (2022). Development of (4-Phenylamino)quinazoline Alkylthiourea Derivatives as Novel NF-κB Inhibitors. Pharmaceuticals, 15(7), 778. https://doi.org/10.3390/ph15070778







