A Focus on Abuse/Misuse and Withdrawal Issues with Selective Serotonin Reuptake Inhibitors (SSRIs): Analysis of Both the European EMA and the US FAERS Pharmacovigilance Databases
Abstract
1. Introduction
1.1. Abusing with an AD
1.2. AD and SSRI-Associated Withdrawal Issues
1.3. AD and SSRI-Associated Withdrawal Issues; Post-Marketing Evidence
2. Results
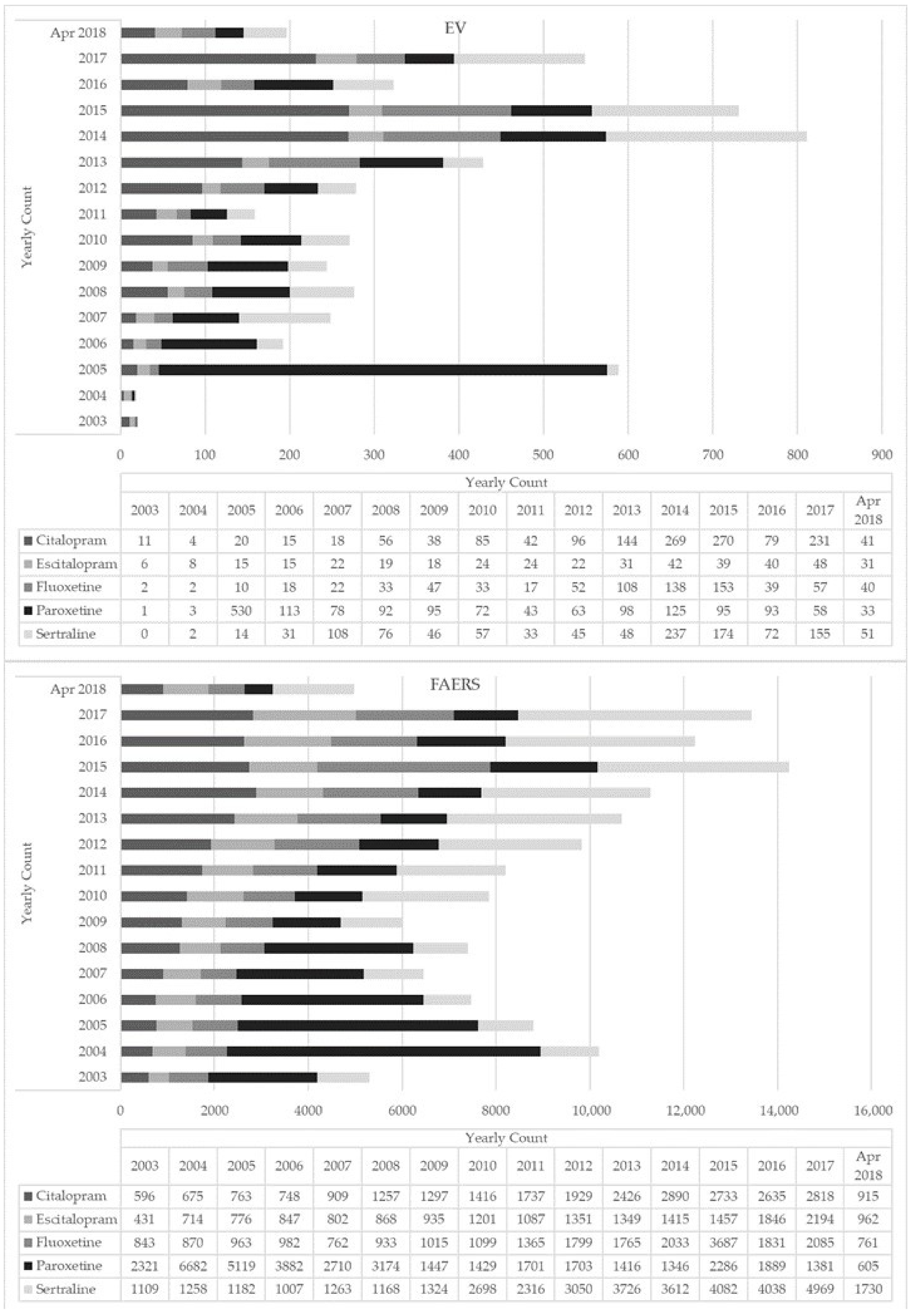
2.1. EMA Dataset
2.2. FAERS Dataset
3. Discussion
3.1. Comparison between the Two Datasets
3.2. SSRIs Abusing Issues; Differences between the Molecules Examined
3.3. SSRIs’ Dependence and Withdrawal Issues; Clinical and Theoretical Considerations
3.4. The “Denominator” Issue; Focus on SSRIs’ Prescription Data
3.5. Limitations
4. Materials and Methods
4.1. Data Sources
4.2. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sullivan, M.A. Abuse and misuse of antidepressants. Subst. Abus. Rehabil. 2014, 5, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Kataoka, Y.; Ostinelli, E.G.; Cipriani, A.; Furukawa, T.A. National Prescription Patterns of Antidepressants in the Treatment of Adults with Major Depression in the US between 1996 and 2015: A Population Representative Survey Based Analysis. Front. Psychiatry 2020, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Milani, S.A.; Raji, M.A.; Chen, L.; Kuo, Y. Trends in the Use of Benzodiazepines, Z-Hypnotics, and Serotonergic Drugs among US Women and Men before and during the COVID-19 Pandemic. JAMA Netw. Open 2021, 4, e2131012. [Google Scholar] [CrossRef] [PubMed]
- Trends, C. MEPS HC-213: 2019 Prescribed Medicines August 2021. Available online: https://meps.ahrq.gov/data_stats/download_data/pufs/h213i/h213idoc.shtml (accessed on 28 February 2022).
- Poluzzi, E.; Piccinni, C.; Sangiorgi, E.; Clo, M.; Tarricone, I.; Menchetti, M.; De Ponti, F. Trend in SSRI-SNRI antidepressants prescription over a 6-year period and predictors of poor adherence. Eur. J. Clin. Pharmacol. 2013, 69, 2095–2101. [Google Scholar] [CrossRef]
- Abbing-Karahagopian, V.; Huerta, C.; Souverein, P.C.; De Abajo, F.; Leufkens, H.G.M.; Slattery, J.; Alvarez, Y.; Miret, M.; Gil, M.; Oliva, B.; et al. Antidepressant prescribing in five European countries: Application of common definitions to assess the prevalence, clinical observations, and methodological implications. Eur. J. Clin. Pharmacol. 2014, 70, 849–857. [Google Scholar] [CrossRef]
- Bauer, M.; Monz, B.U.; Montejo, A.L.; Quail, D.; Dantchev, N.; Demyttenaere, K.; Garcia-Cebrian, A.; Grassi, L.; Perahia, D.G.; Reed, C.; et al. Prescribing patterns of antidepressants in Europe: Results from the Factors Influencing Depression Endpoints Research (FINDER) study. Eur. Psychiatry 2008, 23, 66–73. [Google Scholar] [CrossRef]
- Lewer, D.; O’Reilly, C.; Mojtabai, R.; Evans-Lacko, S. Antidepressant use in 27 European countries: Associations with sociodemographic, cultural and economic factors. Br. J. Psychiatry 2015, 207, 221–226. [Google Scholar] [CrossRef]
- Taylor, S.; Annand, F.; Burkinshaw, P.; Greaves, F.; Kelleher, M.; Knight, J.; Perkins, C.; Tran, A.; White, M.; Marsden, J.D. Dependence and Withdrawal Associated with Some Prescribed Medicines. An Evidence Review; Public Health England: London, UK, 2019; pp. 89–92. Available online: Publi-cHealthEngland%0Ahttps://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/829777/PHE_PMR_report.pdf (accessed on 28 February 2022).
- Carvalho, A.F.; Sharma, S.; Brunoni, A.R. The Safety, Tolerability and Risks Associated with the Use of Newer Generation Antidepressant Drugs: A Critical Review of the Literature. Psychother. Psychosom. 2016, 85, 270–288. [Google Scholar] [CrossRef]
- Massabki, I.; Abi-Jaoude, E. Selective serotonin reuptake inhibitor ‘discontinuation syndrome’ or withdrawal. Br. J. Psychiatry 2021, 218, 168–171. [Google Scholar] [CrossRef]
- Fava, G.A.; Gatti, A.; Belaise, C.; Guidi, J.; Offidani, E. Withdrawal symptoms after selective serotonin reuptake inhibitor discontinuation: A systematic review. Psychother. Psychosom. 2015, 84, 72–81. [Google Scholar] [CrossRef]
- Hernandez, S.H.; Nelson, L.S. Prescription Drug Abuse: Insight Into the Epidemic. Clin. Pharmacol. Ther. 2009, 88, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Dawson, D.A.; Stinson, F.S.; Hasin, D.S.; Ruan, W.J.; Saha, T.D.; Smith, S.M.; Goldstein, R.B.; Grant, B.F. Prevalence, correlates, and comorbidity of nonmedical prescription drug use and drug use disorders in the United States: Results of the National Epidemiologic Survey on Alcohol and Related Conditions. J. Clin. Psychiatry 2006, 67, 1062–1073. [Google Scholar] [CrossRef] [PubMed]
- Chiappini, S.; Schifano, F. What about “pharming”? Issues regarding the misuse of prescription and over-the-counter drugs. Brain Sci. 2020, 10, 736. [Google Scholar] [CrossRef]
- Schifano, F.; Chiappini, S.; Corkery, J.M.; Guirguis, A. Abuse of prescription drugs in the context of novel psychoactive substances (NPS): A systematic review. Brain Sci. 2018, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Chiappini, S.; Guirguis, A.; Corkery, J.M.; Schifano, F. Misuse of prescription and over-the-counter drugs to obtain illicit highs: How pharmacists can prevent abuse. Pharm. J. 2020, 305, 7943. [Google Scholar]
- Evans, S.J.; Waller, P.C.; Davis, S. Use of proportional reporting ratios (PRRs) for signal generation from spontaneous adverse drug reaction reports. Pharmacoepidemiol. Drug Saf. 2001, 10, 483–486. [Google Scholar] [CrossRef]
- Prahlow, J.A.; Landrum, J.E. Amitriptyline abuse and misuse. Am. J. Forensic Med. Pathol. 2005, 26, 86–88. [Google Scholar] [CrossRef]
- Sein Anand, J.; Chodorowski, Z.; Habrat, B. Recreational amitriptyline abuse. Przegląd Lek. 2005, 62, 397–398. [Google Scholar]
- Schifano, F.; Chiappini, S. Is there a potential of misuse for venlafaxine and bupropion? Front. Pharmacol. 2018, 9, 239. [Google Scholar] [CrossRef]
- Stall, N.; Godwin, J.; Juurlink, D. Five things to know about: Bupropion abuse and overdose. CMAJ 2014, 186, 1015. [Google Scholar] [CrossRef]
- Stassinos, G.L.; Klein-Schwartz, W. Bupropion ‘abuse’ reported to us poison centers. J. Addict. Med. 2016, 10, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Francesconi, G.; Orsolini, L.; Papanti, D.; Corkery, J.M.; Schifano, F. Venlafaxine as the ‘baby ecstasy’? Literature overview and analysis of web-based misusers’ experiences. Hum. Psychopharmacol. 2015, 30, 255–261. [Google Scholar] [CrossRef]
- Singh, A.N.; Catalan, J. Rave drug (ecstasy) and selective serotonin reuptake inhibitor anti-depressants. Indian J. Psychiatry 2000, 42, 195–197. [Google Scholar] [PubMed]
- Davies, J.; Read, J. Addictive Behaviors a systematic review into the incidence, severity and duration of antidepressant withdrawal effects: Are guidelines evidence-based? Addict. Behav. 2019, 97, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Marsden, J.; White, M.; Annand, F.; Burkinshaw, P.; Carville, S.; Eastwood, B.; Kelleher, M.; Knight, J.; O’Connor, R.; Tran, A.; et al. Medicines associated with dependence or withdrawal: A mixed-methods public health review and national database study in England. Lancet Psychiatry 2019, 6, 935–950. [Google Scholar] [CrossRef]
- Iacobucci, G. NHS prescribed record number of antidepressants last year. BMJ 2019, 364, l1508. [Google Scholar] [CrossRef]
- Fava, G.A.; Belaise, C. Discontinuing Antidepressant Drugs: Lesson from a Failed Trial and Extensive Clinical Experience. Psychother. Psychosom. 2018, 87, 257–267. [Google Scholar] [CrossRef]
- Guy, A.; Brown, M.; Lewis, S.; Horowitz, M. The ‘patient voice’: Patients who experience antidepressant withdrawal symptoms are often dismissed, or misdiagnosed with relapse, or a new medical condition. Ther. Adv. Psychopharmacol. 2020, 10. [Google Scholar] [CrossRef]
- NICE. Depression in Adults: Recognition and Management Clinical Guideline. 2009. Available online: https://www.nice.org.uk/guidance/cg90 (accessed on 28 February 2022).
- Read, J. Addictive Behaviors How common and severe are withdrawal effects from, and addiction to, antidepressants? The experiences of a large international sample of patients. Addict. Behav. 2020, 102, 106157. [Google Scholar] [CrossRef]
- Read, J.; Renton, J.; Harrop, C.; Geekie, J.; Dowrick, C. A survey of UK general practitioners about depression, antidepressants and withdrawal: Implementing the 2019 Public Health England report. Ther. Adv. Psychopharmacol. 2020, 10. [Google Scholar] [CrossRef]
- Fava, G.A.; Offidani, E. The mechanisms of tolerance in antidepressant action. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 1593–1602. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.H.; Jacques, P.; Breland-Noble, A.M.; Cuijpers, P.; Sciences, M.; Amsterdam, V.U.; Forrest, S.J.; Charles, F.R., III; Arthur, M.N.; Laura, H.M.; et al. Clinical Practice Guideline for the Treatment of Depression Across Three Age Cohorts American Psychological Association Guideline Development Panel for the Treatment of Depressive Disorders; American Psychological Association: Washington, DC, USA, 2019; pp. 1–213. Available online: https://www.apa.org/depression-guideline/guideline.pdf (accessed on 28 February 2022).
- Schifano, F. Coming off Prescribed Psychotropic Medications: Insights from Their Use as Recreational Drugs. Psychother. Psychosom. 2020, 89, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Young, A.H.; Currie, A.; Ashton, C.H. Antidepressant withdrawal syndrome. Br. J. Psychiatry 1997, 170, 288. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fava, G.A.; Grandi, S. Withdrawal syndromes after paroxetine and sertraline discontinuation. J. Clin. Psychopharmacol. 1995, 15, 374–375. [Google Scholar] [CrossRef]
- Blayac, J.P.; Hillaire-Buys, D.; Peyrière, H. La pharmacovigilance des nouveaux antidépresseurs: Évaluation des troubles neuro-psychocomportementaux [Pharmacovigilance of new antidepressants: Evaluation of neuro-psychobehavioral disorders]. Therapie 1997, 52, 117–122. [Google Scholar] [PubMed]
- Price, J.S.; Waller, P.C.; Wood, S.M.; Mackay, A.V.P. A comparison of the post-marketing safety of four selective serotonin re-uptake inhibitors including the investigation of symptoms occurring on withdrawal. Br. J. Clin. Pharmacol. 1996, 42, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Medawar, C.; Herxheimer, A.; Bell, A.; Jofre, S. Paroxetine, Panorama and user reporting of ADRs: Consumer intelligence matters in clinical practice and post-marketing drug surveillance. Int. J. Risk Saf. Med. 2002, 15, 161–169. [Google Scholar]
- Nomura, K.; Hinomura, Y.; Kawaguchi, G.; Matsushita, Y.; Marui, H.; Anzai, T.; Hashiguchi, M.; Mochizuki, M.; Takahashi, K. Effect of database profile variation on drug safety assessment: An analysis of spontaneous adverse event reports of Japanese cases. Drug Des. Dev. Ther. 2015, 9, 3031–3041. [Google Scholar] [CrossRef]
- Fujiwara, M.; Kawasaki, Y.; Yamada, H. A Pharmacovigilance Approach for Post-Marketing in Japan Using the Japanese Adverse Drug Event Report (JADER) Database and Association Analysis. PLoS ONE 2016, 11, e0154425. [Google Scholar]
- Kawahara, A. Future perspectives for pharmacovigilance in Japan. J. Health Sci. 2009, 55, 593–600. [Google Scholar] [CrossRef][Green Version]
- Davies, J.; Cooper, R.E.; Moncrieff, J.; Montagu, L.; Rae, T.; Parhi, M. The costs incurred by the NHS in England due to the unnecessary prescribing of dependency-forming medications. Addict. Behav. 2021, 125, 107143. [Google Scholar] [CrossRef] [PubMed]
- Brandt, J.; Janzen, D.; Alessi-Severini, S.; Singer, A.; Chateau, D.; Enns, M.; Leong, C. Risk of long-term benzodiazepine and Z-drug use following the first prescription among community-dwelling adults with anxiety/mood and sleep disorders: A retrospective cohort study. BMJ Open 2021, 11, e046916. [Google Scholar] [CrossRef] [PubMed]
- Jann, M.; Kennedy, W.K.; Lopez, G. Benzodiazepines: A major component in unintentional prescription drug overdoses with opioid analgesics. J. Pharm. Pract. 2014, 27, 5–16. [Google Scholar] [CrossRef]
- Paulozzi, L.J.; Strickler, G.K.; Kreiner, P.W.; Koris, C.M. Controlled Substance Prescribing Patterns-Prescription Behavior Surveillance System, Eight States, 2013; Morbidity and Mortality Weekly Report (MMWR) Surveillance Summaries; Centers for Disease Control & Prevention (CDC): Washington, DC, USA, October 2015; Volume 64, pp. 1–14.
- Almond, S.-A.M.; Warren, M.J.; Shealy, K.M.; Threatt, T.B.; Ward, E.D. A Systematic Review of the Efficacy and Safety of Over-the-Counter Medications Used in Older People for the Treatment of Primary insomnia. Sr. Care Pharm. 2021, 36, 83–92. [Google Scholar] [CrossRef]
- Schifano, F.; Chiappini, S.; Miuli, A.; Mosca, A.; Santovito, M.C.; Corkery, J.M.; Guirguis, A.; Pettorruso, M.; Di Giannantonio, M.; Martinotti, G. Focus on Over-the-Counter Drugs’ Misuse: A Systematic Review on Antihistamines, Cough Medicines, and Decongestants. Front. Psychiatry. 2021, 12, 458. [Google Scholar] [CrossRef] [PubMed]
- Schifano, F.; Napoletano, F.; Chiappini, S.; Guirguis, A.; Corkery, J.M.; Bonaccorso, S.; Ricciardi, A.; Scherbaum, N.; Vento, A. New/emerging psychoactive substances and associated psychopathological consequences. Psychol. Med. 2021, 51, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Stahl Stephen, G.M. Stahl’s Essential Psychopharmacology: Prescriber’s Guide, 6th ed.; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Mccance-Katz, E.F. Webcast Slides for the 2019 National Survey on Drug Use and Health. 2020. Available online: https://www.samhsa.gov/data/sites/default/files/reports/rpt29392/Assistant-Secretary-nsduh2019_presentation/Assistant-Secretary-nsduh2019_presentation.pdf (accessed on 28 February 2022).
- DeVido, J.J.; Weiss, R.D. Treatment of the depressed alcoholic patient. Curr. Psychiatry Rep. 2012, 14, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Agabio, R.; Trogu, E.; Pani, P.P. Antidepressants for the treatment of people with co-occurring depression and alcohol dependence. Cochrane Database Syst. Rev. 2018, 2018, CD008581. [Google Scholar] [CrossRef]
- Kurtz, S.P.; Margolin, Z.R.; Wogenstahl, K. The diversion of nonscheduled psychoactive prescription medications in the United States, 2002 to 2017. Pharmacoepidemiol. Drug Saf. 2019, 28, 700–706. [Google Scholar] [CrossRef]
- Thom, R.P.; Alexander, J.L.; Baron, D.; Garakani, A.; Gross, L.; Pine, J.H.; Radhakrishnan, R.; Slaby, A.; Sumner, C.R. Selective Serotonin Reuptake Inhibitors: How Long Is Long Enough? J. Psychiatr. Pract. 2021, 27, 361–371. [Google Scholar] [CrossRef]
- Pollock, B.G. Expert Opinion on Pharmacotherapy Citalopram: A comprehensive review. Expert Opin. Pharmacother. 2001, 2, 681–698. [Google Scholar] [CrossRef] [PubMed]
- Glassman, A.H. Citalopram toxicity. Lancet (Lond. Engl.) 1997, 350, 818. [Google Scholar] [CrossRef]
- Lau, G.T.; Horowitz, B.Z. Sertraline overdose. Acad Emerg. Med. 1996, 3, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Kecskemeti, V. Cardiovascular side effects of new antidepressants and antipsychotics: New drugs, old concerns? Curr. Pharm. Des. 2004, 10, 2463–2475. [Google Scholar] [CrossRef]
- Di Chiara, G. A motivational learning hypothesis of the role of mesolimbic dopamine in compulsive drug use. J. Psychopharmacol. 1998, 12, 54–67. [Google Scholar] [CrossRef]
- Ashton, A.H. SSRIs, Drug Withdrawal and Abuse: Problem of Treatment? In Selective Serotonin Reuptake Inhibitors (SSRIs): Past, Present and Future; University of Newcastle-upon-Tyne: Newcastle, UK, 1999; pp. 65–80. [Google Scholar]
- European Monitoring Centre for Drugs and Drug Addiction. Non-Medical Use of Medicines: Health and Social Responses. 2021. Available online: https://www.emcdda.europa.eu/publications/mini-guides/non-medical-use-of-medicines-health-and-social-responses_en (accessed on 28 February 2022).
- Torrens, M.; Fonseca, F.; Mateu, G.; Farré, M. Efficacy of antidepressants in substance use disorders with and without comorbid depression. A systematic review and meta-analysis. Drug Alcohol. Depend. 2005, 78, 1–22. [Google Scholar] [CrossRef]
- Cheeta, S.; Schifano, F.; Oyefeso, A.; Webb, L.; Ghodse, A.H. Antidepressant-related deaths and antidepressant prescriptions in England and Wales, 1998–2000. Br. J. Psychiatry 2004, 184, 41–47. [Google Scholar] [CrossRef]
- Jokela, M.; Virtanen, M.; David Batty, G.; Kivimaki, M. Research letter. Pharmaceutical Overdose Deaths, United States, 2010. JAMA Psychiatry 2016, 73, 87–88. [Google Scholar] [CrossRef]
- Ferner, R.E.; Easton, C.; Cox, A.R. Deaths from Medicines: A Systematic Analysis of Coroners’ Reports to Prevent Future Deaths. Drug Saf. 2018, 41, 103–110. [Google Scholar] [CrossRef]
- Pilgrim, J.L.; Gerostamoulos, D.; Drummer, O.H. Deaths involving serotonergic drugs. Forensic Sci. Int. 2010, 198, 110–117. [Google Scholar] [CrossRef]
- Jauhar, S.; Hayes, J.; Goodwin, G.M.; Baldwin, D.S.; Cowen, P.J.; Nutt, D.J. Antidepressants, withdrawal, and addiction; where are we now? J. Psychopharmacol. 2019, 33, 655–659. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Read, J.; Hengartner, M.P.; Cosci, F.; Fava, G.; Chouinard, G.; van Os, J.; Nardi, A.; Gøtzsche, P.; Groot, P.; et al. Clinical guidelines on antidepressant withdrawal urgently need updating. BMJ 2019, 365, l2238. [Google Scholar] [CrossRef] [PubMed]
- Iacobucci, G. NEWS NICE updates antidepressant guidelines to reflect severity and length of withdrawal symptoms. BMJ 2019, 367, l6103. [Google Scholar] [CrossRef] [PubMed]
- Henssler, J.; Heinz, A.; Brandt, L.; Bschor, T. Antidepressant Withdrawal and Rebound Phenomena. Dtsch. Ärzteblatt Int. 2019, 116, 355–361. [Google Scholar] [CrossRef]
- Medawar, C.; Herxheimer, A. A comparison of adverse drug reaction reports from professionals and users, relating to risk of dependence and suicidal behaviour with paroxetine. Int. J. Risk Saf. Med. 2003, 16, 5–19. [Google Scholar]
- Marken, P.A.; Stuart Munro, J. Selecting a selective serotonin reuptake inhibitor: Clinically important distinguishing features. Prim. Care Companion J. Clin. Psychiatry 2000, 2, 205–210. [Google Scholar] [CrossRef]
- Pierre, J.M. Abuse of psychiatric medications: Not just stimulants and benzodiazepines misuse or abuse. Curr. Psychiatry 2019, 18, 11. [Google Scholar]
- Schatzberg, A.F.; Haddad, P.; Kaplan, E.M.; Lejoyeux, M.; Rosenbaum, J.F.; Young, A.H.; Zajecka, J. Serotonin reuptake inhibitor discontinuation syndrome: A hypothetical definition. Discontinuation Consensus panel. J. Clin. Psychiatry 1997, 58 (Suppl. 7), 5–10. [Google Scholar]
- Nielsen, M.; Hansen, E.H.; Gøtzsche, P.C. What is the difference between dependence and withdrawal reactions? A comparison of benzodiazepines and selective serotonin re-uptake inhibitors. Addiction 2012, 107, 900–908. [Google Scholar] [CrossRef]
- Balon, R.; Silberman, E.K.; Starcevic, V.; Cosci, F.; Freire, R.C.; Nardi, A.E.; Rickels, K.; Shader, R. Benzodiazepines, antidepressants and addiction: A plea for conceptual rigor and consistency. J. Psychopharmacol. 2019, 33, 1467–1470. [Google Scholar] [CrossRef]
- Fava, G.A.; Cosci, F.; Offidani, E.; Guidi, J. Behavioral Toxicity Revisited: Iatrogenic Comorbidity in Psychiatric Evaluation and Treatment. J. Clin. Psychopharmacol. 2016, 36, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Cosci, F.; Chouinard, G. Acute and Persistent Withdrawal Syndromes Following Discontinuation of Psychotropic Medications. Psychother Psychosom 2020, 89, 283–306. [Google Scholar] [CrossRef] [PubMed]
- Fava, G.A. May antidepressant drugs worsen the conditions they are supposed to treat? The clinical foundations of the oppositional model of tolerance. Ther. Adv. Psychopharmacol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Heinz, A.; Daedelow, L.S.; Wackerhagen, C.; Di Chiara, G. Addiction theory matters—Why there is no dependence on caffeine or antidepressant medication. Addict. Biol. 2020, 25, e12735. [Google Scholar] [CrossRef] [PubMed]
- Postigo, R.; Brosch, S.; Slattery, J.; van Haren, A.; Dogné, J.-M.; Kurz, X.; Candore, G.; Domergue, F.; Arlett, P. EudraVigilance Medicines Safety Database: Publicly Accessible Data for Research and Public Health Protection. Drug Saf. 2018, 41, 665–675. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Reporting and Learning Systems for Medication Errors: The Role of Pharmacovigilance Centres; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Alomar, M.; Tawfiq, A.M.; Hassan, N.; Palaian, S. Post marketing surveillance of suspected adverse drug reactions through spontaneous reporting: Current status, challenges and the future. Ther. Adv. Drug Saf. 2020, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.D.; Andrews, E.B. Pharmacovigilance. In Pharmacovigilance: Second Edition, 2nd ed.; John Wiley & Sons Ltd.: Chichester, UK, 2007. [Google Scholar]
- Brody, D.J.; Gu, Q. Antidepressant Use among Adults: United States, 2015–2018. NCHS Data Brief 2020, 377, 1–8. [Google Scholar]
- Heald, A.H.; Stedman, M.; Davies, M.; Livingston, M.; Taylor, D.; Gadsby, R. Antidepressant Prescribing in England: Patterns and Costs. Prim. Care Companion CNS Disord. 2020, 22, 26631. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration (FDA). FDA Adverse Event Reporting System (FAERS) Public Dashboard; U.S. Food & Drug Administration: Silver Spring, MD, USA, 2021; p. 19. Available online: https://www.fda.gov/drugs/questions-and-answers-fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-public-dashboard (accessed on 28 February 2022).
- European Medicines Agency. Module VI–Collection, Management and Submission of Reports of Suspected Adverse Reactions to Medicinal Products (Rev 2). Guidel Good Pharmacovigil Pract. 2017. Revision 2 July. pp. 1–144. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Regulatory_and_procedural_guideline/2017/08/WC500232767.pdf (accessed on 28 February 2022).
- Prescription Cost Analysis (PCA) Data 2021. Available online: https://www.nhsbsa.nhs.uk/prescription-data/dispensing-data/prescription-cost-analysis-pca-data (accessed on 28 February 2022).
- Lalji, H.M.; McGrogan, A.; Bailey, S.J. An analysis of antidepressant prescribing trends in England 2015–2019. J. Affect. Disord. Rep. 2021, 6, 100205. [Google Scholar] [CrossRef]
- National Center for Health Statistics. National Health and Nutrition Examination Survey (NHANES). Available online: https://www.cdc.gov/nchs/nhanes/index.htm (accessed on 28 February 2022).
- Hales, C.M.; Kit, B.K.; Gu, Q.; Ogden, C.L. Trends in Prescription Medication Use Among Children and Adolescents-United States, 1999–2014. JAMA 2018, 319, 2009–2020. [Google Scholar] [CrossRef]
- Kantor, E.D.; Rehm, C.D.; Haas, J.S.; Chan, A.T.; Giovannucci, E.L. Trends in Prescription Drug Use Among Adults in the United States From 1999–2012. JAMA 2015, 314, 1818–1831. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency (EMA). Note for Guidance–EudraVigilance Human–Processing of Safety Messages and Individual Case Safety Reports (ICSRs). Available online: https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/note-guidance-eudravigilance-human-processing-safety-messages-individual-case-safety-reports-icsrs_en.pdf (accessed on 28 February 2022).
- Vogel, U.; van Stekelenborg, J.; Dreyfus, B.; Garg, A.; Habib, M.; Hosain, R.; Wisniewski, A. Investigating Overlap in Signals from EVDAS, FAERS, and VigiBase®. Drug Saf. 2020, 43, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Pariente, A.; Gregoire, F.; Fourrier-Reglat, A.; Haramburu, F.; Moore, N. Impact of safety alerts on measures of disproportionality in spontaneous reporting databases. Drug Saf. 2007, 30, 891–898. [Google Scholar] [CrossRef] [PubMed]
- ICH. MedDRA® Term Selection: Points to Consider. ICH-Endorsed Guide for MedDRA Users; Release 4. (Version 17.1); London, UK, 2014; pp. 1–49. Available online: https://meddra.org/sites/default/files/guidance/file/9491-1700_termselptc_r4.7_mar2014.pdf (accessed on 28 February 2022).
- Vickers-Smith, R.; Sun, J.; Charnigo, R.J.; Lofwall, M.R.; Walsh, S.L.; Havens, J.R. Gabapentin drug misuse signals: A pharmacovigilance assessment using the FDA adverse event reporting system. Drug Alcohol Depend. 2020, 206, 107709. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.; Poncet, A. PhViD: An R package for PharmacoVigilance Signal Detection, R package version 1.0.8; 2016. Available online: https://cran.r-project.org/web/packages/PhViD/PhViD.pdf (accessed on 28 February 2022).
- Subeesh, V.; Maheswari, E.; Saraswathy, G.R.; Swaroop, A.M.; Minnikanti, S.S. A comparative study of data mining algorithms used for signal detection in FDA AERS database. J. Young Pharm. 2018, 10, 444–449. [Google Scholar] [CrossRef]
- Poluzzi, E.; Raschi, E.; Piccinni, C.; De, F. Data Mining Techniques in Pharmacovigilance: Analysis of the Publicly Accessible FDA Adverse Event Reporting System (AERS). In Data Mining Applications in Engineering and Medicine; InTechOpen: Rijeka, Croatia, 2012; Available online: http://www.intechopen.com/books/data-mining-applications-in-engineering-and-medicine/data-mining-techniques-in-pharmacovigilance-analysis-of-the-publicly-accessible-fda-adverse-event-re (accessed on 28 February 2022).
- Van Puijenbroek, E.P.; Bate, A.; Leufkens, H.G.M.; Lindquist, M.; Orre, R.; Egberts, A.C.G. A comparison of measures of disproportionality for signal detection is spontaneous reporting systems for adverse drug reactions. Pharmacoepidemiol. Drug Saf. 2002, 11, 3–10. [Google Scholar] [CrossRef]
- Suling, M.; Pigeot, I. Signal detection and monitoring based on longitudinal healthcare data. Pharmaceutics 2012, 4, 607–640. [Google Scholar] [CrossRef]
- Ahmed, I.; Thiessard, F.; Miremont-Salam, G.; Haramburu, F.; Kreft-Jais, C.; Bgaud, B.; Tubert-Bitter, P. Early detection of pharmacovigilance signals with automated methods based on false discovery rates: A comparative study. Drug Saf. 2012, 35, 495–506. [Google Scholar] [CrossRef]
- Ahmed, I.; Dalmasso, C.; Haramburu, F.; Thiessard, F.; Broët, P.; Tubert-Bitter, P. False discovery rate estimation for frequentist pharmacovigilance signal detection methods. Biometrics 2010, 66, 301–309. [Google Scholar] [CrossRef]
- National Center for Health Statistics (U.S.). National Health and nutrition Examination Survey: Analytic guidelines, 1999–2010. Available online: https://www.cdc.gov/nchs/data/series/sr_02/sr02_161.pdf (accessed on 28 February 2022).
- National Health and Nutrition Examination Survey: Analytic Guidelines, 2011–2014 and 2015–2016. 14 December 2018. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/analyticguidelines/11-16-analytic-guidelines.pdf (accessed on 28 February 2022).
- National Health and Nutrition Examination Survey. NHANES Analytic Guidance and Brief Overview for the 2017-March 2020 Pre-Pandemic Data Files. Available online: https://wwwn.cdc.gov/nchs/nhanes/continuousnhanes/overviewbrief.aspx?cycle=2017-2020 (accessed on 28 February 2022).
| Citalopram | Escitalopram | Fluoxetine | Paroxetine | Sertraline | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| EMA | FAERS | EMA | FAERS | EMA | FAERS | EMA | FAERS | EMA | FAERS | |
| Individual Cases | 1419 | 25,744 | 404 | 18,235 | 771 | 22,793 | 1592 | 39,091 | 1149 | 38,532 |
| Mean Age in years (SD) | 42.6 (14.2) | 47.6 (21.5) | 43.3 (18.6) | 48.2 (22.5) | 43.1 (15.5) | 42.5 (20.6) | 41.4 (15.8) | 44.0 (23.5) | 41.6 (16.5) | 45.6 (22.0) |
| M/F (%) | 615/773 (44%/56%) | 8770/14,169 (38%/62%) | 138/244 (36%/64%) | 5988/10,920 (35%/65%) | 279/457 (38%/62%) | 6547/13,141 (33%/67%) | 554/959 (37%/63%) | 13,124/22,609 (37%/63%) | 493/606 (45%/55%) | 12,245/21,972 (36%/64%) |
| Most common psychiatric indications recorded for the index SSRI (%) | Depression (14.6) Drug abuse (3.2) Anxiety (2.6) | Depression (38.7) Anxiety (9.4) | Depression (51.0) Anxiety (12.9) Drug abuse (5.8) | Depression (50.5) Anxiety (15.3) | Depression (22.7) Drug abuse (4.1) Anxiety (3.4) | Depression (43.4) Anxiety (7.6) Obsessive-compulsive disorder (2.7) | Depression (38.1) Anxiety (14.8) Panic disorder/attack (6.5) | Depression (32.8) Anxiety (9.9) Generalised anxiety disorder (3.9) | Depression (34.5) Anxiety (7.5) | Depression (44.8) Anxiety (15.1) |
| ROA (%) | Oral (68.4) Parenteral * (0.8) T-placent (0.7) Inhalation (0.2) NA (29.9) | Oral (70.7) T-placent (8.3) Parenteral * (0.3) T-mam (0.2) NA (20.4) | Oral (79.7) T-placent (2.5) NA (16.6) | Oral (77.8) T-placent (6.3) T-mam (0.2) NA: (15.7) | Oral (61.9) Inhalation (1.9) T-placent (1.7) Parenteral * (1.2) NA (33.2) | Oral (57.4) T-placent (15.0) T-mam (0.3) Parenteral * (0.2) NA (27.2) | Oral (82.2) T-placent (1.4) Parenteral * (0.2) Inhalation (0.1) NA (16.0) | Oral (74.7) T-placent (10.3) Parenteral * (0.1) NA (15.1) | Oral (65.5) T-placent (2.2) Inhalation (0.5) Parenteral * (0.4) NA (30.9) | Oral (69.4) T-placent (11.7) T-mam (0.3) NA (15.7) |
| Therapeutic regimen (Mono/Poly) | 139 (10%)/ 1280 (90%) | 1502 (6%)/ 24,242 (94%) | 131 (32%)/ 273 (68%) | 1260 (7%)/ 16,975 (93%) | 76 (10%)/ 695 (90%) | 1072 (5%)/ 21,721 (95%) | 537 (34%)/ 1055 (66%) | 3586 (9%)/ 35,505 (91%) | 197 (17%)/ 952 (83%) | 3365 (9%)/ 35,167 (91%) |
| Most important concomitant prescription psychotropic drugs recorded (%) | ||||||||||
| Antidepressants | 17.5 | 19.2 | 10.4 | 17.3 | 18.8 | 20.2 | 9.0 | 11.0 | 13.4 | 13.6 |
| Antihistamines | 25.4 | 9.0 | 3.2 | 5.7 | 19.3 | 8.5 | 10.8 | 4.6 | 17.5 | 6.5 |
| Antipsychotics | 13.7 | 17.1 | 16.1 | 19.0 | 19.3 | 18.7 | 9.5 | 9.8 | 13.2 | 15.8 |
| Benzodiazepines ° | 36.9 | 22.8 | 28.7 | 23.8 | 43.3 | 20.4 | 27.6 | 17.2 | 29.9 | 16.4 |
| Gabapentinoids | 2.5 | 5.0 | 4.5 | 4.2 | 4.2 | 4.8 | 2.3 | 2.0 | 3.7 | 4.7 |
| Mood Stabilizers | 3.0 | 8.4 | 7.2 | 10.3 | 5.4 | 9.2 | 3.7 | 5.4 | 7.0 | 7.0 |
| Opioids | 59.7 | 22.7 | 10.9 | 11.0 | 43.3 | 16.4 | 16.0 | 7.1 | 40.7 | 11.1 |
| Z-Drugs | 3.4 | 6.3 | 4.5 | 6.8 | 9.3 | 5.6 | 8.2 | 4.8 | 5.6 | 4.5 |
| Most important concomitant recreational drugs recorded (%) | ||||||||||
| Alcohol | 13.5 | 4.3 | 2.7 | 1.7 | 14.8 | 3.1 | 3.5 | 1.4 | 7.1 | 1.3 |
| Amphetamines | 2.4 | 1.1 | 1.5 | 0.7 | 3.1 | 1.2 | 0.5 | 0.3 | 3.3 | 0.7 |
| Cannabis and Cannabinoids | 1.2 | 0.6 | 1.7 | 0.4 | 1.3 | 0.4 | 0.5 | 0.2 | 1.2 | 0.3 |
| Cocaine | 9.4 | 1.5 | 2.2 | 0.4 | 4.9 | 0.6 | 1.0 | 0.2 | 9.8 | 0.6 |
| Heroin | 0 | 1.8 | 0 | 0 | 0 | 0.5 | 0 | 0.1 | 0 | 0.5 |
| Ketamine | 0.2 | 0 | 0 | 0 | 0.4 | 0 | 0 | 0 | 0 | 0 |
| EMA | FAERS | ||||
|---|---|---|---|---|---|
| Drug (Total Cases) | Cases with Fatal Outcome Sex ° (%) and Mean Age (SD) | Percent of Drug-Specific Cases * | Drug (Total Cases) | Cases with Fatal Outcome Sex ° (%) and Mean Age (SD) | Percent of Drug- Specific Cases |
| Citalopram (1419) | 994 F: 49.6% Mean age: 42.0 yy (12.4) | 70.0% | Citalopram (25,744) | 7402 F: 50.0% Mean age: 45.7 yy (17.1) | 28.8% |
| Escitalopram (404) | 31 F: 54.8% Mean age: 40.4 yy (16.0) | 7.7% | Escitalopram (18,235) | 2293 F: 50.6% Mean age: 48.3 yy (20.7) | 12.6% |
| Fluoxetine (771) | 424 F: 55.7% Mean age: 44.3 yy (12.9) | 55.0% | Fluoxetine (22,793) | 4659 F: 53.8% Mean age: 44.1 yy (17.5) | 20.4% |
| Paroxetine (1592) | 271 F: 44.6% Mean age: 43.6 yy (12.0) | 17.0% | Paroxetine (39,091) | 3438 F: 45.2% Mean age: 48.8 yy (20.9) | 8.8% |
| Sertraline (1149) | 532 F: 41.0% Mean age: 41.8 yy (14.0) | 46.3% | Sertraline (38,532) | 4863 F: 45.2% Mean age 48.1 yy (20.8) | 12.6% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiappini, S.; Vickers-Smith, R.; Guirguis, A.; Corkery, J.M.; Martinotti, G.; Schifano, F. A Focus on Abuse/Misuse and Withdrawal Issues with Selective Serotonin Reuptake Inhibitors (SSRIs): Analysis of Both the European EMA and the US FAERS Pharmacovigilance Databases. Pharmaceuticals 2022, 15, 565. https://doi.org/10.3390/ph15050565
Chiappini S, Vickers-Smith R, Guirguis A, Corkery JM, Martinotti G, Schifano F. A Focus on Abuse/Misuse and Withdrawal Issues with Selective Serotonin Reuptake Inhibitors (SSRIs): Analysis of Both the European EMA and the US FAERS Pharmacovigilance Databases. Pharmaceuticals. 2022; 15(5):565. https://doi.org/10.3390/ph15050565
Chicago/Turabian StyleChiappini, Stefania, Rachel Vickers-Smith, Amira Guirguis, John Martin Corkery, Giovanni Martinotti, and Fabrizio Schifano. 2022. "A Focus on Abuse/Misuse and Withdrawal Issues with Selective Serotonin Reuptake Inhibitors (SSRIs): Analysis of Both the European EMA and the US FAERS Pharmacovigilance Databases" Pharmaceuticals 15, no. 5: 565. https://doi.org/10.3390/ph15050565
APA StyleChiappini, S., Vickers-Smith, R., Guirguis, A., Corkery, J. M., Martinotti, G., & Schifano, F. (2022). A Focus on Abuse/Misuse and Withdrawal Issues with Selective Serotonin Reuptake Inhibitors (SSRIs): Analysis of Both the European EMA and the US FAERS Pharmacovigilance Databases. Pharmaceuticals, 15(5), 565. https://doi.org/10.3390/ph15050565










