A Mechanism of Isoorientin-Induced Apoptosis and Migration Inhibition in Gastric Cancer AGS Cells
Abstract
1. Introduction
2. Results
2.1. Isoorientin Reduces the Viabilities of Gastric Cancer Cells
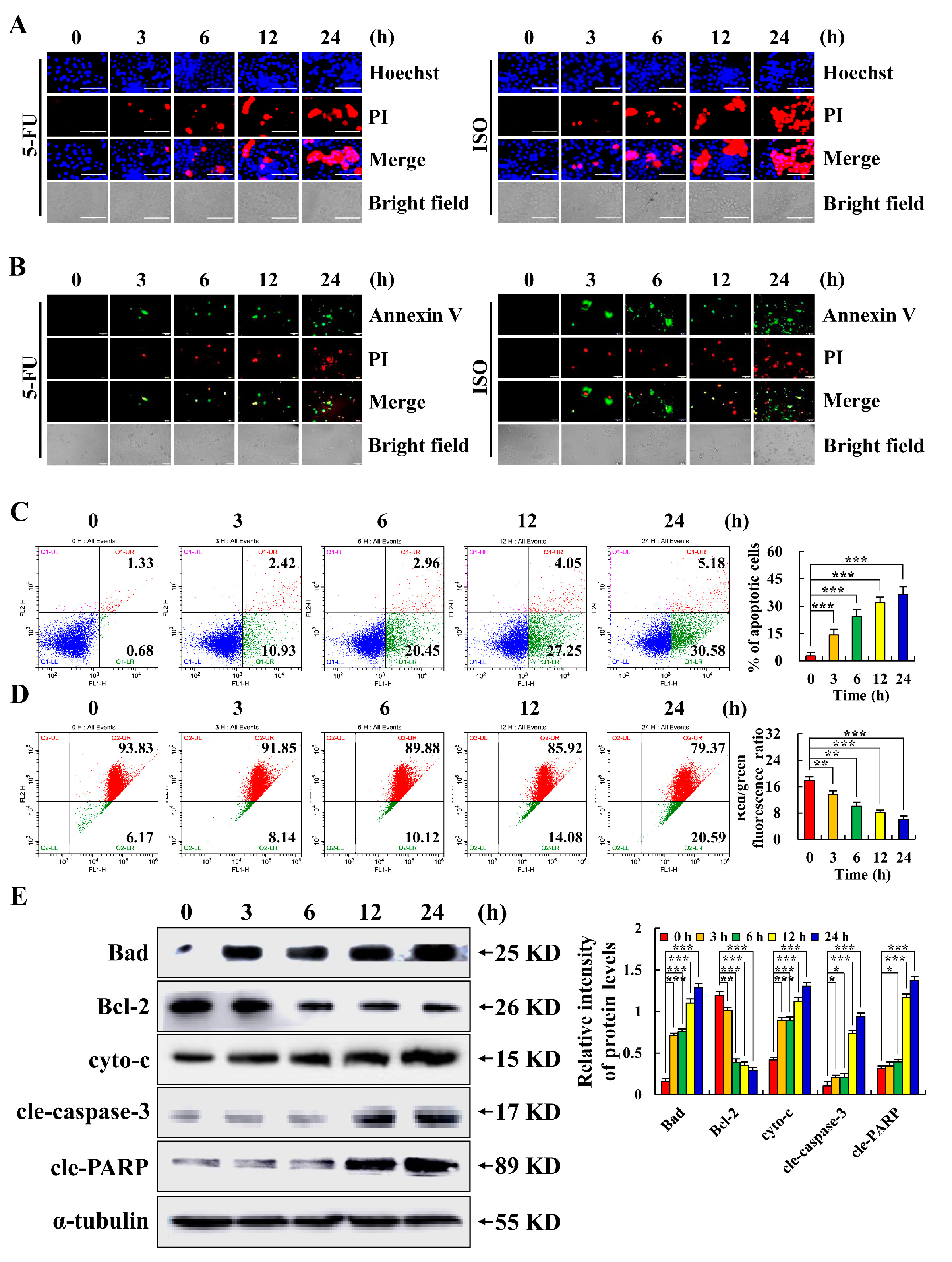
2.2. Isoorientin Induces AGS Cell Apoptosis
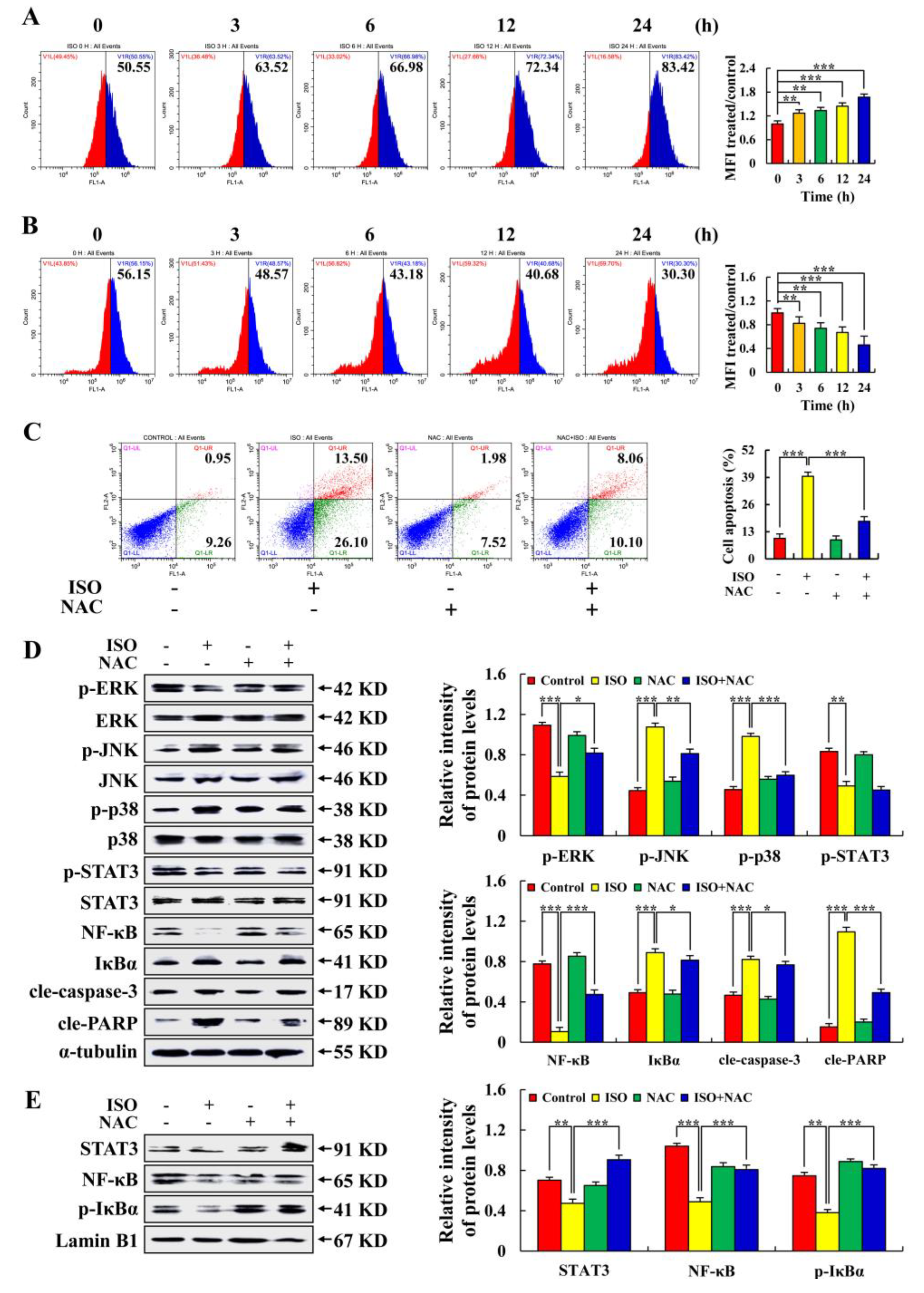
2.3. Isoorientin Regulates MAPK/STAT3/NF–κB Signaling Pathways
2.4. Isoorientin Regulates the MAPK/STAT3/NF–κB Signaling Pathways That Are Mediated by ROS
2.5. Isoorientin Arrested the AGS Cell Cycle at the G2/M Phase
2.6. Isoorientin Inhibited AGS Cell Migration via the AKT/GSK–3β/β–catenin Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Viability Analyses
4.3. Cell Apoptosis Analysis
4.4. Mitochondrial Membrane Potential Analysis
4.5. Extraction of Nucleoproteins
4.6. Western Blot Analysis
4.7. Measurement of ROS Generation
4.8. Cell Cycle Analysis
4.9. Cell Migration Analyses
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shan, C.; Zhang, Y.; Hao, X.; Gao, J.; Chen, X.; Wang, K. Biogenesis, functions and clinical significance of circRNAs in gastric cancer. Mol. Cancer 2019, 18, 136. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.M.; Zheng, R.S.; Zhang, S.W.; Zeng, H.M.; Chen, R.; Sun, K.X.; Gu, X.Y.; Wei, W.W.; He, J. Epidemiological characteristics of gastric cancer in China, 2015. Zhonghua Liu Xing Bing Xue Za Zhi 2019, 40, 1517–1521. [Google Scholar] [PubMed]
- Chen, W.Q.; Zheng, R.S.; Zhang, S.W.; Zeng, H.; Xia, C.; Zuo, T.; Yang, Z.; Zou, X.; He, J. Analysis of cancer incidence and mortality in elderly population in China, 2013. Zhonghua Liu Xing Bing Xue Za Zhi 2017, 39, 60–66. [Google Scholar]
- Pilleron, S.; Sarfati, D.; Janssen-Heijnen, M.; Vignat, J.; Ferlay, J.; Bray, F.; Soerjomataram, I. Global cancer incidence in older adults, 2012 and 2035: A population-based study. Int. J. Cancer 2019, 144, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.D.; Syn, N.L.; Moehler, M.; Grothe, W.; Yong, W.P.; Tai, B.C.; Ho, J.; Unverzagt, S. Chemotherapy for advanced gastric cancer. Cochrane Database Syst. Rev. 2017, 8, CD004064. [Google Scholar] [CrossRef]
- Créhange, G.; Huguet, F.; Quero, L.; N’Guyen, T.V.; Mirabel, X.; Lacornerie, T. Radiotherapy in cancers of the oesophagus, the gastric cardia and the stomach. Cancer Radiothérapie 2016, 20, S161–S168. [Google Scholar] [CrossRef]
- Jiao, L.; Dong, C.; Liu, J.; Chen, Z.; Zhang, L.; Xu, J.; Shen, X.; Che, J.; Yang, Y.; Huang, H.; et al. Effects of Chinese Medicine as Adjunct Medication for Adjuvant Chemotherapy Treatments of Non-Small Cell Lung Cancer Patients. Sci. Rep. 2017, 7, 46524. [Google Scholar] [CrossRef]
- Guo, H.; Liu, J.X.; Li, H.; Baak, J.P.A. In Metastatic Non-small cell Lung Cancer Platinum-Based Treated Patients, Herbal Treatment Improves the Quality of Life. A Prospective Randomized Controlled Clinical Trial. Front. Pharmacol. 2017, 8, 454. [Google Scholar] [CrossRef]
- Li, A.; Qu, X.; Li, Z.; Qu, J.; Song, N.; Ma, Y.; Liu, Y. Secreted protein acidic and rich in cysteine antagonizes bufalin-induced apoptosis in gastric cancer cells. Mol. Med. Rep. 2015, 12, 2926–2932. [Google Scholar] [CrossRef][Green Version]
- Peng, J.; Fan, G.; Hong, Z.; Chai, Y.; Wu, Y. Preparative separation of isovitexin and isoorientin from Patrinia villosa Juss by high-speed counter-current chromatography. J. Chromatogr. A 2005, 1074, 111–115. [Google Scholar] [CrossRef]
- Wedler, J.; Daubitz, T.; Schlotterbeck, G.; Butterweck, V. In vitro anti-inflammatory and wound-healing potential of a Phyllostachys edulis leaf extract--identification of isoorientin as an active compound. Planta Med. 2014, 80, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, I.C.V.; Kaluđerović, G.N.; de Oliveira, P.F.; Guimarães, D.O.; Quaresma, C.H.; Porzel, A.; Muzitano, M.F.; Wessjohann, L.A.; Leal, I.C.R. Apoptosis Caused by Triterpenes and Phytosterols and Antioxidant Activity of an Enriched Flavonoid Extract from Passiflora mucronata. Anticancer Agents Med. Chem. 2018, 18, 1405–1416. [Google Scholar] [CrossRef] [PubMed]
- Anilkumar, K.; Reddy, G.V.; Azad, R.; Yarla, N.S.; Dharmapuri, G.; Srivastava, A.; Kamal, M.A.; Pallu, R. Evaluation of Anti-Inflammatory Properties of Isoorientin Isolated from Tubers of Pueraria tuberosa. Oxidative Med. Cell. Longev. 2017, 2017, 5498054. [Google Scholar] [CrossRef] [PubMed]
- Mykhailenko, O.; Petrikaite, V.; Korinek, M.; Chang, F.R.; El-Shazly, M.; Yen, C.H.; Bezruk, I.; Chen, B.H.; Hsieh, C.F.; Lytkin, D.; et al. Pharmacological Potential and Chemical Composition of Crocus sativus Leaf Extracts. Molecules 2021, 27, 10. [Google Scholar] [CrossRef]
- Aljubiri, S.M.; Mahgoub, S.A.; Almansour, A.I.; Shaaban, M.; Shaker, K.H. Isolation of diverse bioactive compounds from Euphorbia balsamifera: Cytotoxicity and antibacterial activity studies. Saudi J. Biol. Sci. 2021, 28, 417–426. [Google Scholar] [CrossRef]
- Bai, F.; Huang, Q.; Wei, J.; Lv, S.; Chen, Y.; Liang, C.; Wei, L.; Lu, Z.; Lin, X. Gypsophila elegans isoorientin-2″-O-α-l-arabinopyranosyl ameliorates porcine serum-induced immune liver fibrosis by inhibiting NF-κB signaling pathway and suppressing HSC activation. Int. Immunopharmacol. 2018, 54, 60–67. [Google Scholar] [CrossRef]
- Czemplik, M.; Mierziak, J.; Szopa, J.; Kulma, A. Flavonoid C-glucosides Derived from Flax Straw Extracts Reduce Human Breast Cancer Cell Growth In vitro and Induce Apoptosis. Front. Pharmacol. 2016, 7, 282. [Google Scholar] [CrossRef]
- Liu, S.C.; Huang, C.S.; Huang, C.M.; Hsieh, M.S.; Huang, M.S.; Fong, I.H.; Yeh, C.T.; Lin, C.C. Isoorientin inhibits epithelial-to-mesenchymal properties and cancer stem-cell-like features in oral squamous cell carcinoma by blocking Wnt/β-catenin/STAT3 axis. Toxicol. Appl. Pharmacol. 2021, 424, 115581. [Google Scholar] [CrossRef]
- Lin, X.; Wei, J.; Chen, Y.; He, P.; Lin, J.; Tan, S.; Nie, J.; Lu, S.; He, M.; Lu, Z.; et al. Isoorientin from Gypsophila elegans induces apoptosis in liver cancer cells via mitochondrial-mediated pathway. J. Ethnopharmacol. 2016, 187, 187–194. [Google Scholar] [CrossRef]
- Huang, D.; Jin, L.; Li, Z.; Wu, J.; Zhang, N.; Zhou, D.; Ni, X.; Hou, T. Isoorientin triggers apoptosis of hepatoblastoma by inducing DNA double-strand breaks and suppressing homologous recombination repair. Biomed. Pharmacother. 2018, 101, 719–728. [Google Scholar] [CrossRef]
- Yuan, Z.F.; Tang, Y.M.; Xu, X.J.; Li, S.S.; Zhang, J.Y. 10-Hydroxycamptothecin induces apoptosis in human neuroblastoma SMS-KCNR cells through p53, cytochrome c and caspase 3 pathways. Neoplasma 2016, 63, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Sinha, K.; Das, J.; Pal, P.B.; Sil, P.C. Oxidative stress: The mitochondria-dependent and mitochondria-independent pathways of apoptosis. Arch. Toxicol. 2013, 87, 1157–1180. [Google Scholar] [CrossRef] [PubMed]
- Nakagami, H.; Morishita, R.; Yamamoto, K.; Taniyama, Y.; Aoki, M.; Matsumoto, K.; Nakamura, T.; Kaneda, Y.; Horiuchi, M.; Ogihara, T. Mitogenic and antiapoptotic actions of hepatocyte growth factor through ERK, STAT3, and AKT in endothelial cells. Hypertension 2001, 37, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zou, L.; Yao, Q.; Zhang, Y.; Gan, L.; Tang, L. Silencing DEK downregulates cervical cancer tumorigenesis and metastasis via the DEK/p-Ser9-GSK-3β/p-Tyr216-GSK-3β/β-catenin axis. Oncol. Rep. 2017, 38, 1035–1042. [Google Scholar] [CrossRef]
- Yan, Z.; Lai, Z.; Lin, J. Anticancer Properties of Traditional Chinese Medicine. Comb. Chem. High Throughput Screen. 2017, 20, 423–429. [Google Scholar] [CrossRef]
- Wang, X.; Fang, G.; Pang, Y. Chinese Medicines in the Treatment of Prostate Cancer: From Formulas to Extracts and Compounds. Nutrients 2018, 10, 283. [Google Scholar] [CrossRef]
- Wu, X.; Chung, V.C.H.; Lu, P.; Poon, S.K.; Hui, E.P.; Lau, A.Y.L.; Balneaves, L.G.; Wong, S.Y.S.; Wu, J.C.Y. Chinese Herbal Medicine for Improving Quality of Life Among Nonsmall Cell Lung Cancer Patients: Overview of Systematic Reviews and Network Meta-Analysis. Medicine 2016, 95, e2410. [Google Scholar] [CrossRef]
- Ślefarska-Wolak, D.; Heinzle, C.; Leiherer, A.; Ager, C.; Muendlein, A.; Mezmale, L.; Leja, M.; Corvalan, A.H.; Drexel, H.; Królicka, A.; et al. Volatilomic Signatures of AGS and SNU-1 Gastric Cancer Cell Lines. Molecules 2022, 27, 4012. [Google Scholar] [CrossRef]
- Xu, W.T.; Shen, G.N.; Li, T.Z.; Zhang, Y.; Zhang, T.; Xue, H.; Zuo, W.B.; Li, Y.N.; Zhang, D.J.; Jin, C.H. Isoorientin induces the apoptosis and cell cycle arrest of A549 human lung cancer cells via the ROS-regulated MAPK, STAT3 and NF-κB signaling pathways. Int. J. Oncol. 2020, 57, 550–561. [Google Scholar] [CrossRef]
- Yuan, L.; Wang, J.; Xiao, H.; Xiao, C.; Wang, Y.; Liu, X. Isoorientin induces apoptosis through mitochondrial dysfunction and inhibition of PI3K/Akt signaling pathway in HepG2 cancer cells. Toxicol. Appl. Pharmacol. 2012, 265, 83–92. [Google Scholar] [CrossRef]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qian, J.; Cao, J.; Wang, D.; Liu, C.; Yang, R.; Li, X.; Sun, C. Antioxidant Capacity, Anticancer Ability and Flavonoids Composition of 35 Citrus (Citrus reticulata Blanco) Varieties. Molecules 2017, 22, 1114. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Wang, J.; Wu, W.; Liu, Q.; Liu, X. Effect of isoorientin on intracellular antioxidant defence mechanisms in hepatoma and liver cell lines. Biomed. Pharmacother. 2016, 81, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.R.; Luo, Y.H.; Piao, X.J.; Zhang, Y.; Feng, Y.C.; Li, J.Q.; Xu, W.T.; Zhang, Y.; Zhang, T.; Wang, S.N.; et al. Mechanisms underlying isoliquiritigenin-induced apoptosis and cell cycle arrest via ROS-mediated MAPK/STAT3/NF-κB pathways in human hepatocellular carcinoma cells. Drug Dev. Res. 2019, 80, 461–470. [Google Scholar] [CrossRef]
- Ye, T.; Su, J.; Huang, C.; Yu, D.; Dai, S.; Huang, X.; Chen, B.; Zhou, M. Isoorientin induces apoptosis, decreases invasiveness, and downregulates VEGF secretion by activating AMPK signaling in pancreatic cancer cells. OncoTargets Ther. 2016, 9, 7481–7492. [Google Scholar] [CrossRef]
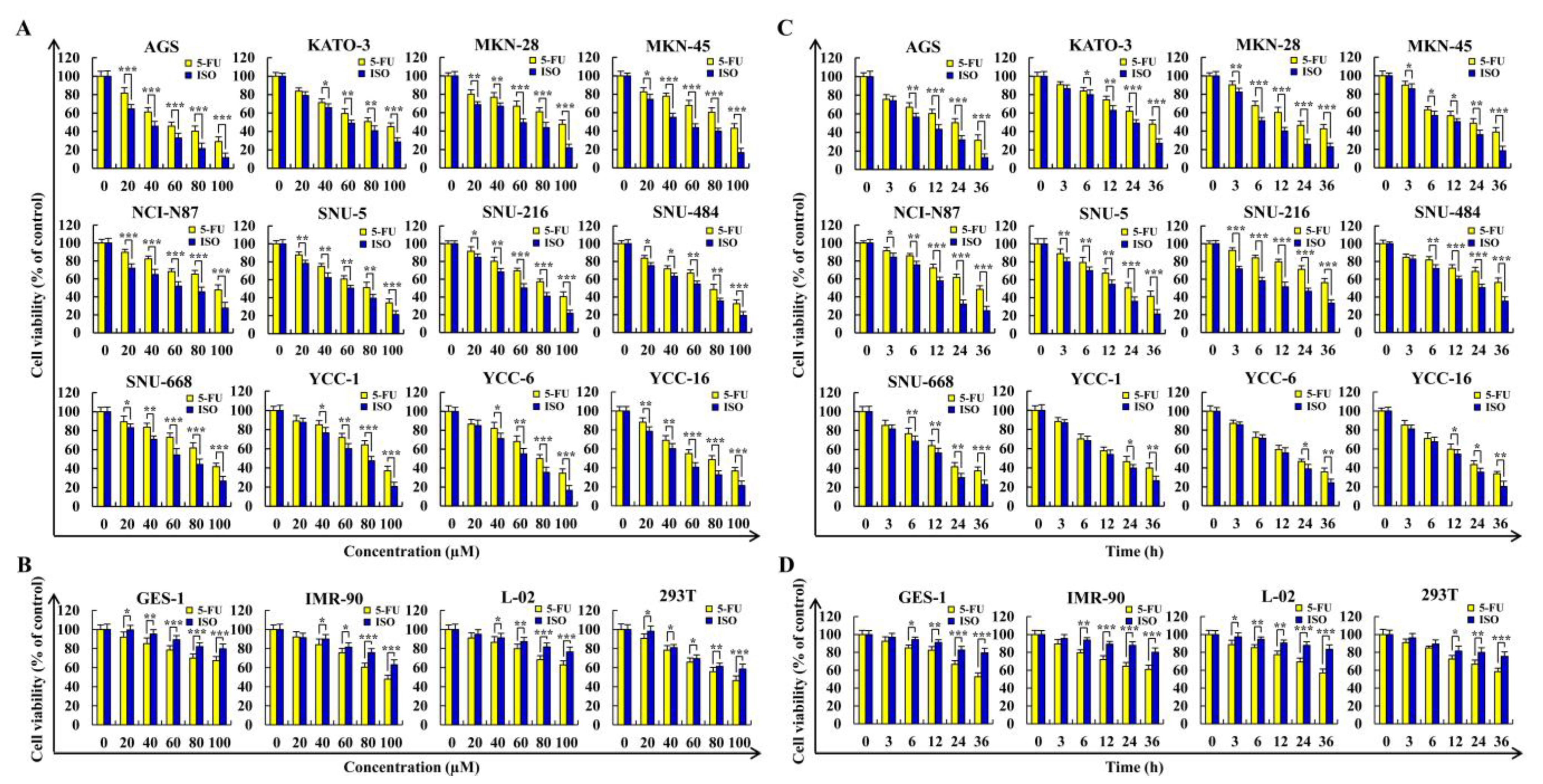




| Cell Line | 5–FU (µM) | ISO (µM) |
|---|---|---|
| AGS | 56.27 ± 2.04 | 36.54 ± 1.93 |
| KATO-3 | 81.11 ± 1.28 | 60.28 ± 1.21 |
| MKN-28 | 98.12 ± 1.87 | 61.31 ± 2.42 |
| MKN-45 | 89.67 ± 2.26 | 49.77 ± 2.18 |
| NCI-N87 | 99.37 ± 1.98 | 68.16 ± 2.38 |
| SNU-5 | 80.97 ± 2.01 | 62.21 ± 1.73 |
| SNU-216 | 86.35 ± 2.84 | 59.57 ± 1.66 |
| SNU-484 | 78.38 ± 1.76 | 65.59 ± 2.44 |
| SNU-668 | 90.63 ± 1.57 | 71.74 ± 2.75 |
| YCC-1 | 91.37 ± 1.03 | 78.62 ± 1.24 |
| YCC-6 | 82.47 ± 1.63 | 66.83 ± 2.68 |
| YCC-16 | 80.16 ± 2.72 | 56.72 ± 2.62 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.; Xiu, Y.-H.; Xue, H.; Li, Y.-N.; Cao, J.-L.; Hou, W.-S.; Liu, J.; Cui, Y.-H.; Xu, T.; Wang, Y.; et al. A Mechanism of Isoorientin-Induced Apoptosis and Migration Inhibition in Gastric Cancer AGS Cells. Pharmaceuticals 2022, 15, 1541. https://doi.org/10.3390/ph15121541
Zhang T, Xiu Y-H, Xue H, Li Y-N, Cao J-L, Hou W-S, Liu J, Cui Y-H, Xu T, Wang Y, et al. A Mechanism of Isoorientin-Induced Apoptosis and Migration Inhibition in Gastric Cancer AGS Cells. Pharmaceuticals. 2022; 15(12):1541. https://doi.org/10.3390/ph15121541
Chicago/Turabian StyleZhang, Tong, Yun-Hong Xiu, Hui Xue, Yan-Nan Li, Jing-Long Cao, Wen-Shuang Hou, Jian Liu, Yu-He Cui, Ting Xu, Ying Wang, and et al. 2022. "A Mechanism of Isoorientin-Induced Apoptosis and Migration Inhibition in Gastric Cancer AGS Cells" Pharmaceuticals 15, no. 12: 1541. https://doi.org/10.3390/ph15121541
APA StyleZhang, T., Xiu, Y.-H., Xue, H., Li, Y.-N., Cao, J.-L., Hou, W.-S., Liu, J., Cui, Y.-H., Xu, T., Wang, Y., & Jin, C.-H. (2022). A Mechanism of Isoorientin-Induced Apoptosis and Migration Inhibition in Gastric Cancer AGS Cells. Pharmaceuticals, 15(12), 1541. https://doi.org/10.3390/ph15121541






