Acute and Reproductive Toxicity Evaluation of Ormona® SI and Ormona® RC—Two New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—In Adult Zebrafish
Abstract
1. Introduction
2. Results
2.1. Acute Toxicity
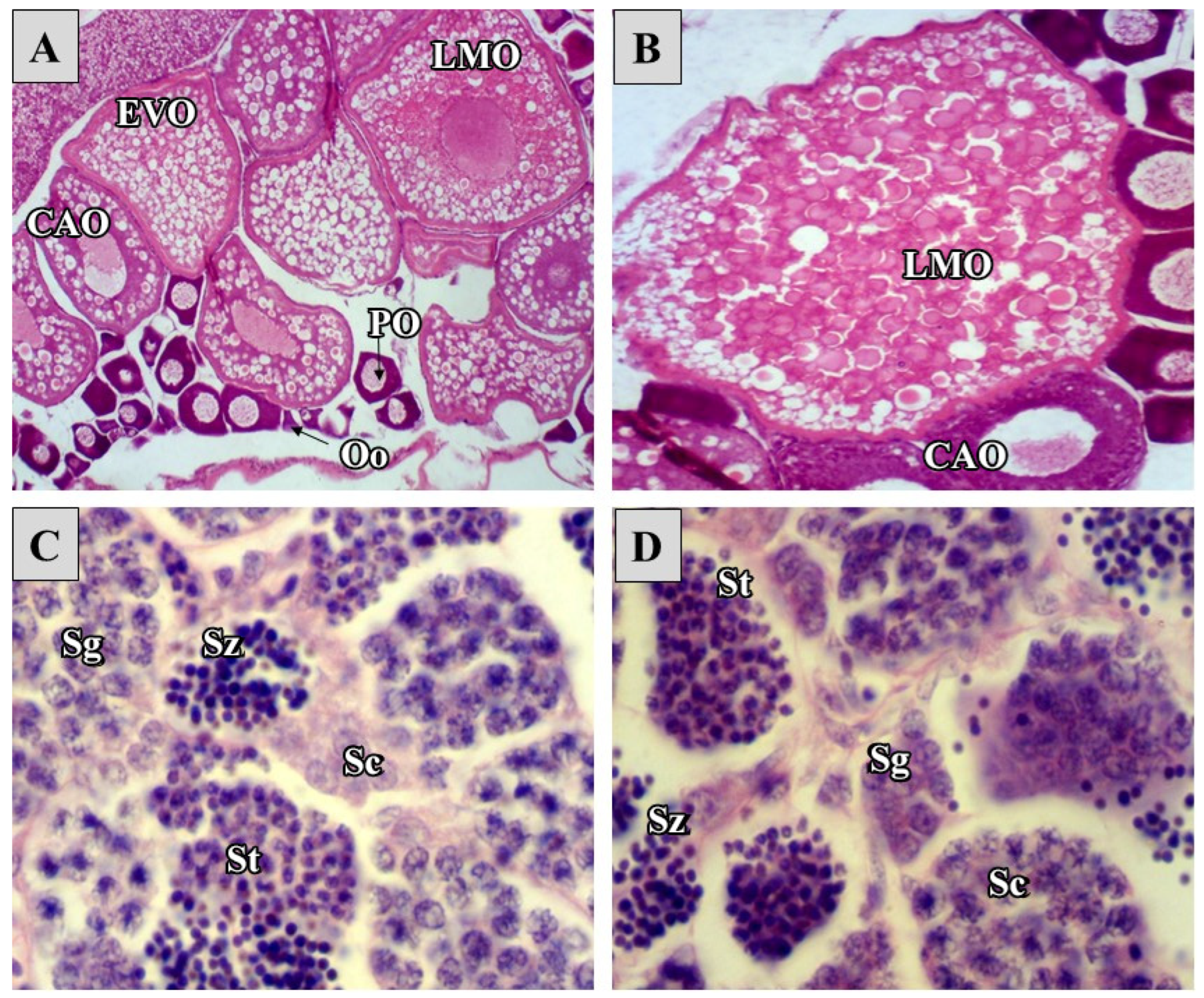
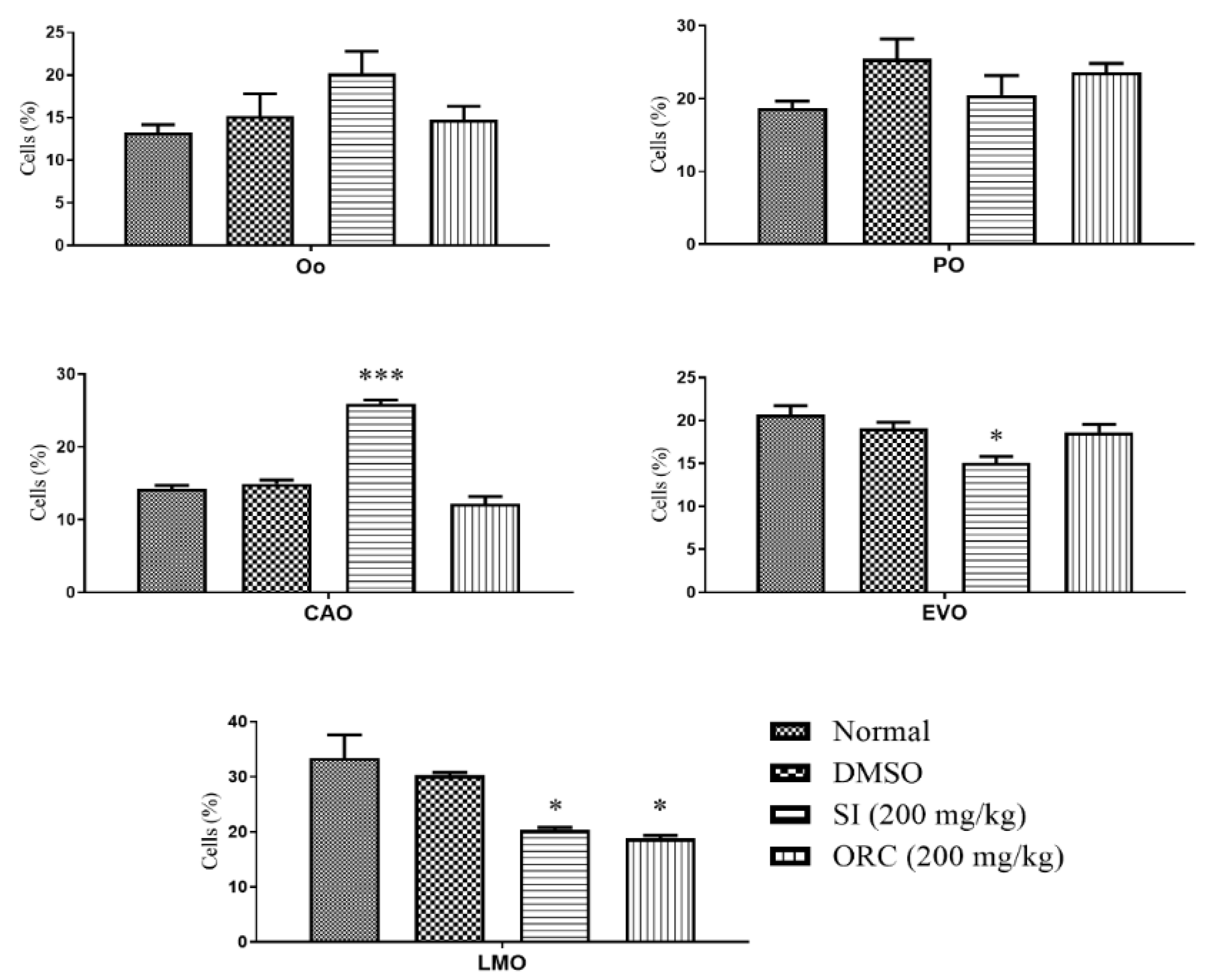
2.2. Reproductive Toxicity
3. Discussion
4. Materials and Methods
4.1. Material Test
4.2. Animals
4.3. Oral Acute Toxicity
4.4. Histopathology
4.5. Reproductive Toxicity
4.6. Parental Gonad Histopathology
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rodrigues, A.P.S.; da Silva Barbosa, R.; Pereira, A.C.M.; Batista, M.A.; Sales, P.F.; Ferreira, A.M.; Colares, N.N.D.; da Silva, H.R.; Soares, M.O.D.S.; da Silva Hage-Melim, L.I.; et al. Ormona® SI and Ormona® RC—New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—Decrease High-Fat Diet-Induced Dyslipidemia in Wistar Rats. Nutraceuticals 2022, 2, 311–322. [Google Scholar] [CrossRef]
- Chen, L.R.; Chen, K.H. Utilization of isoflavones in soybeans for women with menopausal syndrome: An overview. Int. J. Mol. Sci. 2021, 22, 3212. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, L.A.; Chedraui, P.A.; Morocho, N.; Ross, S.; San Miguel, G. The effect of red clover isoflavones on menopausal symptoms, lipids and vaginal cytology in menopausal women: A randomized, double-blind, placebo-controlled study. Gynecol. Endocrinol. 2005, 21, 257–264. [Google Scholar] [CrossRef]
- Kanadys, W.; Baranska, A.; Jedrych, M.; Religioni, U.; Janiszewska, M. Effects of red clover (Trifolium pratense) isoflavones on the lipid profile of perimenopausal and postmenopausal women—A systematic review and meta-analysis. Maturitas 2020, 132, 7–16. [Google Scholar] [CrossRef]
- Lambert, M.N.T.; Thorup, A.C.; Hansen, E.S.S.; Jeppesen, P.B. Combined Red Clover isoflavones and probiotics potently reduce menopausal vasomotor symptoms. PLoS ONE 2017, 12, e0176590. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.J.; Howell, A.; Evans, D.G.; McCloskey, E.V.; Ashley, S.; Greenhalgh, R.; Affen, J.; Flook, L.A.; Tidy, A. Red clover isoflavones are safe and well tolerated in women with a family history of breast cancer. Menopause Int. 2008, 14, 6–12. [Google Scholar] [CrossRef]
- Matias Pereira, A.C.; de Oliveira Carvalho, H.; Gonçalves, D.E.S.; Picanço, K.R.T.; de Lima Teixeira dos Santos, A.V.T.; da Silva, H.R.; Braga, F.S.; Bezerra, R.M.; de Sousa Nunes, A.; Nazima, M.T.S.T.; et al. Co-treatment of purified annatto oil (Bixa orellana L.) and its granules (chronic®) improves the blood lipid profile and bone protective effects of testosterone in the orchiectomy-induced osteoporosis in wistar rats. Molecules 2021, 26, 4720. [Google Scholar] [CrossRef]
- Daud, Z.A.M.; Tubie, B.; Sheyman, M.; Osia, R.; Adams, J.; Tubie, S.; Khosla, P. Vitamin E tocotrienol supplementation improves lipid profiles in chronic hemodialysis patients. Vasc. Health Risk Manag. 2013, 9, 747–761. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Sami, S.A.; Salser, W.A.; Khan, F.A. Dose-dependent suppression of serum cholesterol by tocotrienol-rich fraction (TRF25) of rice bran in hypercholesterolemic humans. Atherosclerosis 2002, 161, 199–207. [Google Scholar] [CrossRef]
- Shen, C.L.; Wang, S.; Yang, S.; Tomison, M.D.; Abbasi, M.; Hao, L.; Scott, S.; Khan, M.S.; Romero, A.W.; Felton, C.K.; et al. A 12-week evaluation of annatto tocotrienol supplementation for postmenopausal women: Safety, quality of life, body composition, physical activity, and nutrient intake. BMC Complement. Altern. Med. 2018, 18, 198. [Google Scholar] [CrossRef]
- Shen, C.L.; Yang, S.; Tomison, M.D.; Romero, A.W.; Felton, C.K.; Mo, H. Tocotrienol supplementation suppressed bone resorption and oxidative stress in postmenopausal osteopenic women: A 12-week randomized double-blinded placebo-controlled trial. Osteoporos. Int. 2018, 29, 881–891. [Google Scholar] [CrossRef] [PubMed]
- Fallah, A.A.; Sarmast, E.; Fatehi, P.; Jafari, T. Impact of dietary anthocyanins on systemic and vascular inflammation: Systematic review and meta-analysis on randomised clinical trials. Food Chem. Toxicol. 2020, 135, 110922. [Google Scholar] [CrossRef]
- Liu, C.; Sun, J.; Lu, Y.; Bo, Y. Effects of anthocyanin on serum lipids in dyslipidemia patients: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0162089. [Google Scholar] [CrossRef] [PubMed]
- Bambino, K.; Chu, J. Zebrafish in Toxicology and Environmental Health, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; Volume 124. [Google Scholar]
- He, J.H.; Gao, J.M.; Huang, C.J.; Li, C.Q. Zebrafish models for assessing developmental and reproductive toxicity. Neurotoxicol. Teratol. 2014, 42, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Horzmann, K.A.; Freeman, J.L. Making waves: New developments in toxicology with the zebrafish. Toxicol. Sci. 2018, 163, 5–12. [Google Scholar] [CrossRef]
- Takashima, F.; Hibiya, T. An Atlas of Fish Histology: Normal and Pathological Features, 2nd ed.; Kodansha Ltd.: Tokyo, Japan; Kodansha Limited: Tokyo, Japan, 1994; Volume 195. [Google Scholar]
- Carvalho, J.C.T.; Keita, H.; Santana, G.R.; de Souza, G.C.; dos Santos, I.V.F.; Amado, J.R.R.; Kourouma, A.; Prada, A.L.; de Oliveira Carvalho, H.; Silva, M.L. Effects of Bothrops alternatus venom in zebrafish: A histopathological study. Inflammopharmacology 2018, 26, 273–284. [Google Scholar] [CrossRef]
- De Souza, G.C.; Matias Pereira, A.C.; Viana, M.D.; Ferreira, A.M.; Da Silva, I.D.R.; De Oliveira, M.M.R.; Barbosa, W.L.R.; Silva, L.B.; Ferreira, I.M.; Dos Santos, C.B.R.; et al. Acmella oleracea (L.) R. K. Jansen Reproductive Toxicity in Zebrafish: An In Vivo and in silico Assessment. Evid.-Based Complement. Altern. Med. 2019, 2019, 1237301. [Google Scholar] [CrossRef]
- Silva, H.R.D.; Assis, D.d.C.d.; Prada, A.L.; Silva, J.O.C.; Sousa, M.B.d.; Ferreira, A.M.; Amado, J.R.R.; Carvalho, H.d.O.; Santos, A.V.T.d.L.T.d.; Carvalho, J.C.T. Obtaining and characterization of anthocyanins from Euterpe oleracea (açaí) dry extract for nutraceutical and food preparations. Rev. Bras. Farmacogn. 2019, 29, 677–685. [Google Scholar] [CrossRef]
- Borges, R.S.; Keita, H.; Ortiz, B.L.S.; dos Santos Sampaio, T.I.; Ferreira, I.M.; Lima, E.S.; de Jesus Amazonas da Silva, M.; Fernandes, C.P.; de Faria Mota Oliveira, A.E.M.; da Conceição, E.C.; et al. Anti-inflammatory activity of nanoemulsions of essential oil from Rosmarinus officinalis L.: In vitro and in zebrafish studies. Inflammopharmacology 2018, 26, 1057–1080. [Google Scholar] [CrossRef]
- Alvarez-Pellitero, P.; Sitja-Bobadilla, A. Pathology of Myxosporea in marine fish culture. Dis. Aquat. Org. 1993, 17, 229–238. [Google Scholar] [CrossRef]
- Goksøyr, A. Use of cytochrome P450 1A (CYP1A) in fish as a biomarker of aquatic pollution. Arch. Toxicol. Suppl. 1995, 17, 80–95. [Google Scholar] [CrossRef] [PubMed]
- Vliegenthart, A.D.B.; Tucker, C.S.; Del Pozo, J.; Dear, J.W. Zebrafish as model organisms for studying drug-induced liver injury. Br. J. Clin. Pharmacol. 2014, 78, 1217–1227. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Y.; Pan, M.H.; Ho, C.T. Anti-obesity molecular mechanism of soy isoflavones: Weaving the way to new therapeutic routes. Food Funct. 2017, 8, 3831–3846. [Google Scholar] [CrossRef]
- Thompson Coon, J.; Pittler, M.H.; Ernst, E. Trifolium pratense isoflavones in the treatment of menopausal hot flushes: A systematic review and meta-analysis. Phytomedicine 2007, 14, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Cederroth, C.R.; Zimmermann, C.; Nef, S. Soy, phytoestrogens and their impact on reproductive health. Mol. Cell. Endocrinol. 2012, 355, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, H.K.; de Oliveira, J.M.; Langoni de Freitas, G.B. Isoflavones alter male and female fertility in different development windows. Biomed. Pharmacother. 2021, 140, 111448. [Google Scholar] [CrossRef] [PubMed]
- Ji, K.; Hong, S.; Kho, Y.; Choi, K. Effects of bisphenol S exposure on endocrine functions and reproduction of zebrafish. Environ. Sci. Technol. 2013, 47, 8793–8800. [Google Scholar] [CrossRef]
- Van den Belt, K.; Verheyen, R.; Witters, H. Reproductive effects of ethynylestradiol and 4t-octylphenol on the zebrafish (Danio rerio). Arch. Environ. Contam. Toxicol. 2001, 41, 458–467. [Google Scholar] [CrossRef]
- Messina, M. Soy foods, isoflavones, and the health of postmenopausal women. Am. J. Clin. Nutr. 2014, 100, 423S–430S. [Google Scholar] [CrossRef]
- OECD. Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure; OECD Guidelines for the Testing of Chemicals, Section 4; OECD: Paris, France, 2008; ISBN 9789264071049. [Google Scholar]
- Leary, S.; Pharmaceuticals, F.; Ridge, H.; Underwood, W.; Anthony, R.; Cartner, S.; Grandin, T.; Collins, F.; Greenacre, C.; Gwaltney-brant, S.; et al. AVMA Guidelines for the Euthanasia of Animals: 2020 Edition. Available online: https://www.avma.org/sites/default/files/2020-02/Guidelines-on-Euthanasia-2020.pdf (accessed on 20 October 2022).
- Poleksic, V.; Mitrovic-Tutundzic, V. Fish gills as a monitor of sublethal and chronic effects of pollution. In Sublethal and Chronic Effects of Pollutants on Freshwater Fish; Müller, L., Ed.; Fishing New Books Ltd.: Farnham, UK; Oxford, UK, 1994; pp. 339–352. [Google Scholar]
- OECD. Test No. 236: Fish Embryo Acute Toxicity (FET) Test. In OECD Guideline for the Testing of Chemicals Section 2; OECD: Paris, France, 2013; pp. 1–22. [Google Scholar]
- Johnson, R.; Wolf, J.; Braunbeck, T. OECD Guidance Document for the Diagnosis of Endocrine-Related Histopathology of Fish Gonads; OECD: Paris, France, 2009. [Google Scholar]





| Group (n = 5) | Stage I | Stage II | Stage III | Death | |
|---|---|---|---|---|---|
| SI | 2000 mg/kg | 1/3 | 0/2 | 3/4 | 2 |
| 1000 mg/kg | 1/3 | 0/2 | 2/4 | 2 | |
| 500 mg/kg | 1/3 | 0/2 | 0/4 | 0 | |
| RC | 2000 mg/kg | 1/3 | 0/2 | 2/4 | 2 |
| 1000 mg/kg | 1/3 | 0/2 | 2/4 | 2 | |
| 500 mg/kg | 1/3 | 0/2 | 0/4 | 0 | |
| Control (DMSO) | 1/3 | 0/2 | 0/4 | 0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Rocha, C.F.; Flexa, C.d.N.N.; de Souza, G.C.; Pereira, A.C.M.; Carvalho, H.d.O.; do Nascimento, A.L.; de Jesus Vasconcelos, N.J.P.; da Silva, H.R.; Carvalho, J.C.T. Acute and Reproductive Toxicity Evaluation of Ormona® SI and Ormona® RC—Two New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—In Adult Zebrafish. Pharmaceuticals 2022, 15, 1434. https://doi.org/10.3390/ph15111434
da Rocha CF, Flexa CdNN, de Souza GC, Pereira ACM, Carvalho HdO, do Nascimento AL, de Jesus Vasconcelos NJP, da Silva HR, Carvalho JCT. Acute and Reproductive Toxicity Evaluation of Ormona® SI and Ormona® RC—Two New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—In Adult Zebrafish. Pharmaceuticals. 2022; 15(11):1434. https://doi.org/10.3390/ph15111434
Chicago/Turabian Styleda Rocha, Clarice Flexa, Camila de Nazaré Nunes Flexa, Gisele Custodio de Souza, Arlindo César Matias Pereira, Helison de Oliveira Carvalho, Aline Lopes do Nascimento, Natasha Juliana Perdigão de Jesus Vasconcelos, Heitor Ribeiro da Silva, and José Carlos Tavares Carvalho. 2022. "Acute and Reproductive Toxicity Evaluation of Ormona® SI and Ormona® RC—Two New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—In Adult Zebrafish" Pharmaceuticals 15, no. 11: 1434. https://doi.org/10.3390/ph15111434
APA Styleda Rocha, C. F., Flexa, C. d. N. N., de Souza, G. C., Pereira, A. C. M., Carvalho, H. d. O., do Nascimento, A. L., de Jesus Vasconcelos, N. J. P., da Silva, H. R., & Carvalho, J. C. T. (2022). Acute and Reproductive Toxicity Evaluation of Ormona® SI and Ormona® RC—Two New Nutraceuticals with Geranylgeraniol, Tocotrienols, Anthocyanins, and Isoflavones—In Adult Zebrafish. Pharmaceuticals, 15(11), 1434. https://doi.org/10.3390/ph15111434








