New IMB16-4 Nanoparticles Improved Oral Bioavailability and Enhanced Anti-Hepatic Fibrosis on Rats
Abstract
1. Introduction
2. Results and Discussion
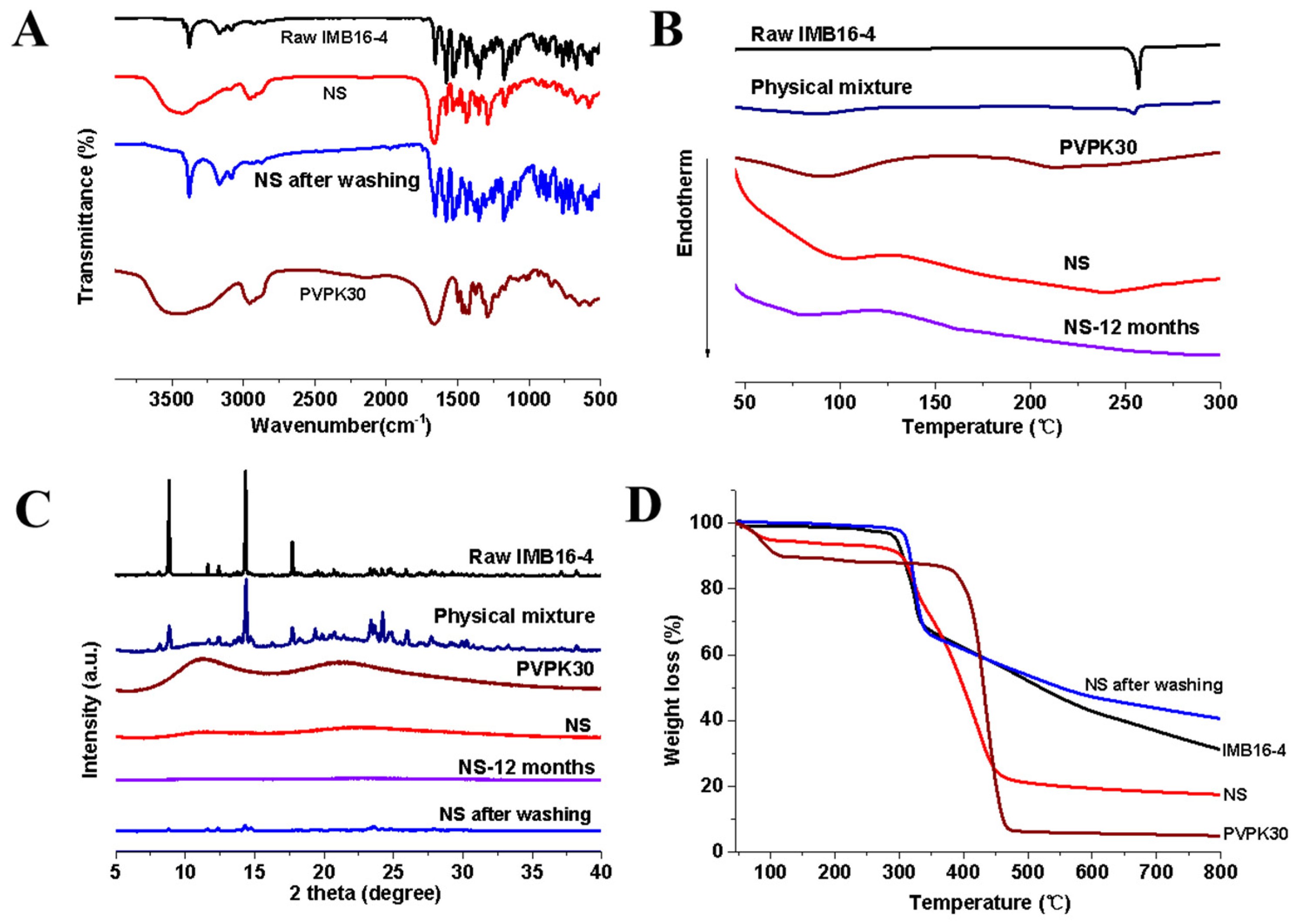
2.1. The Morphology and the Storage Stability of IMB16-4 Nanoparticles
2.2. Characterization of IMB16-4 Nanoparticles
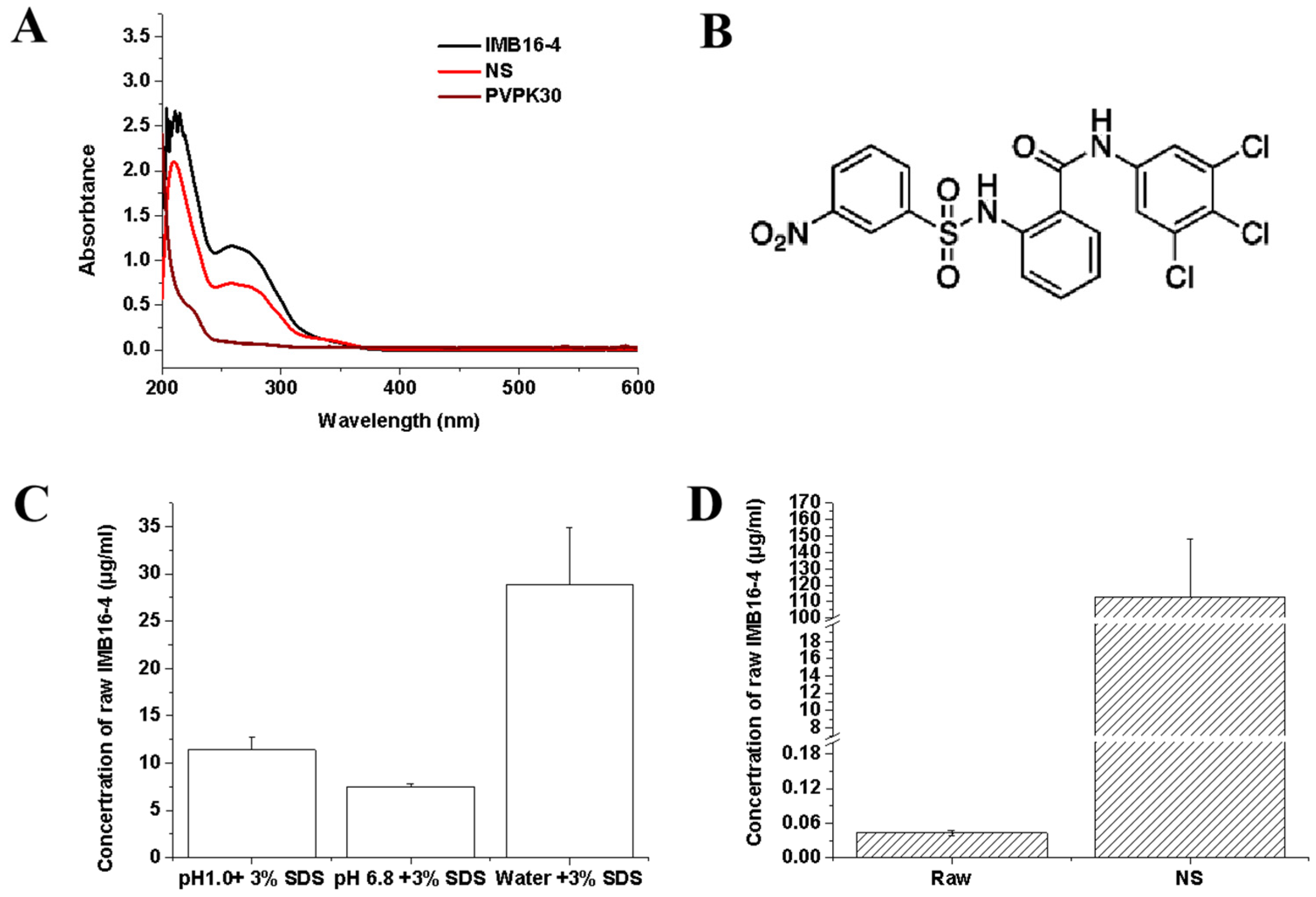
2.3. Estimation of IMB16-4 Content in the Nanoparticles by HPLC Analysis
2.4. The Equilibrium Concentrations
2.5. In Vitro Dissolution Study of IMB16-4 Nanoparticles
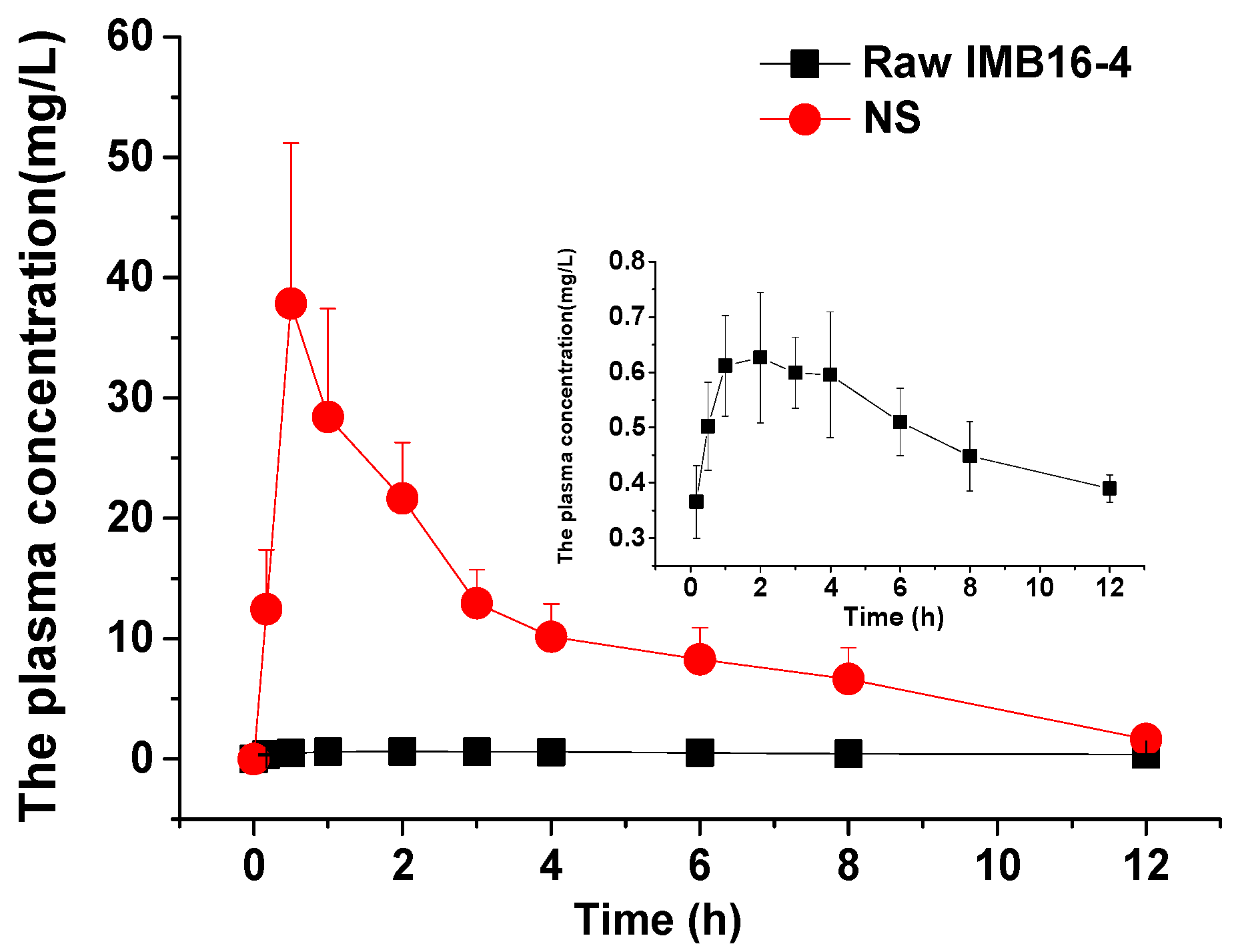
2.6. Nanoparticles Increase In Vivo Absorption and Liver Accumulation of IMB16-4
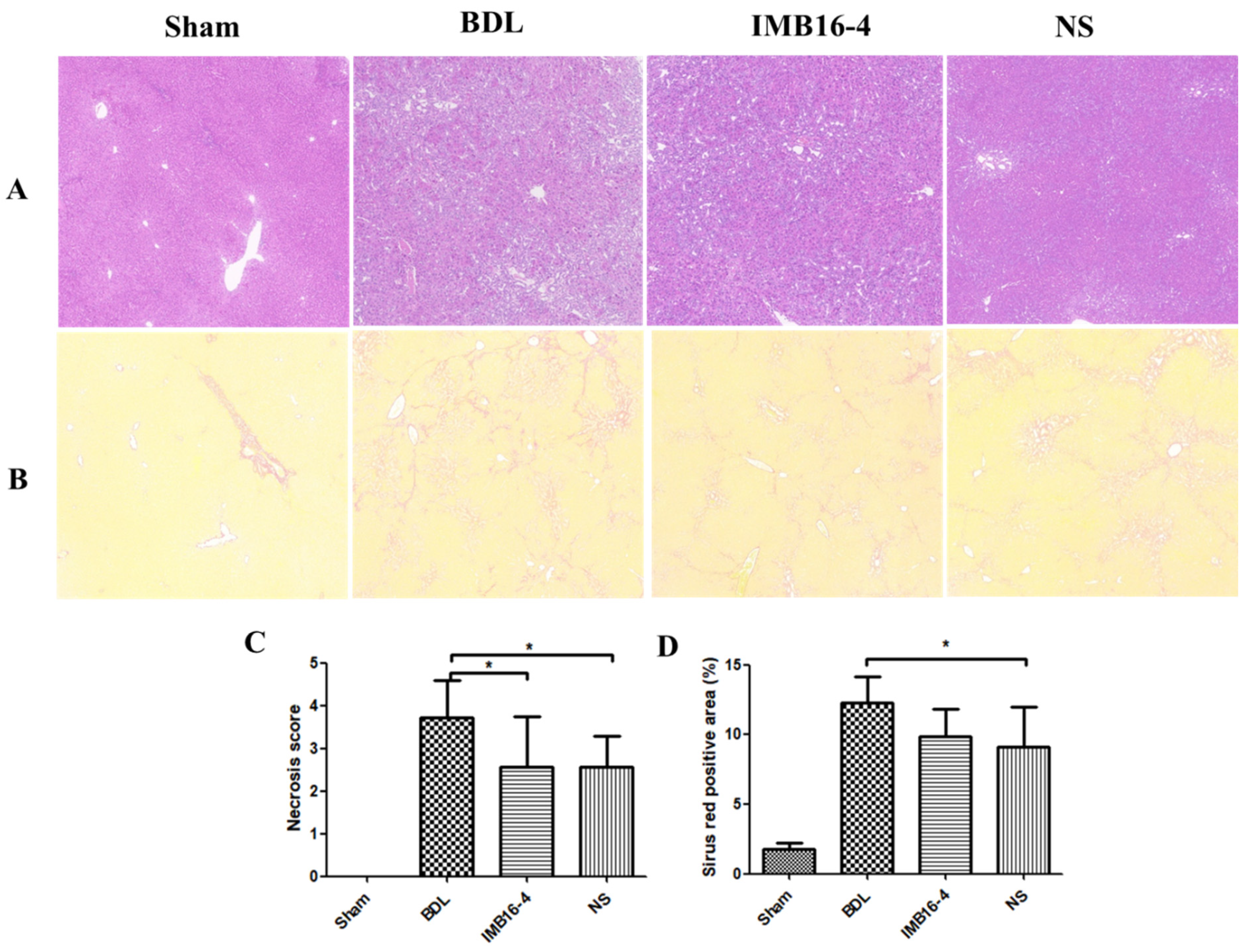
2.7. IMB16-4 Nanoparticles Attenuated BDL-Induced Hepatic Fibrosis in Rats
2.8. IMB16-4 Nanoparticles Improved the Liver Function
2.9. Effects of IMB16-4 Nanoparticles on Fibrotic Protein Marker in Liver
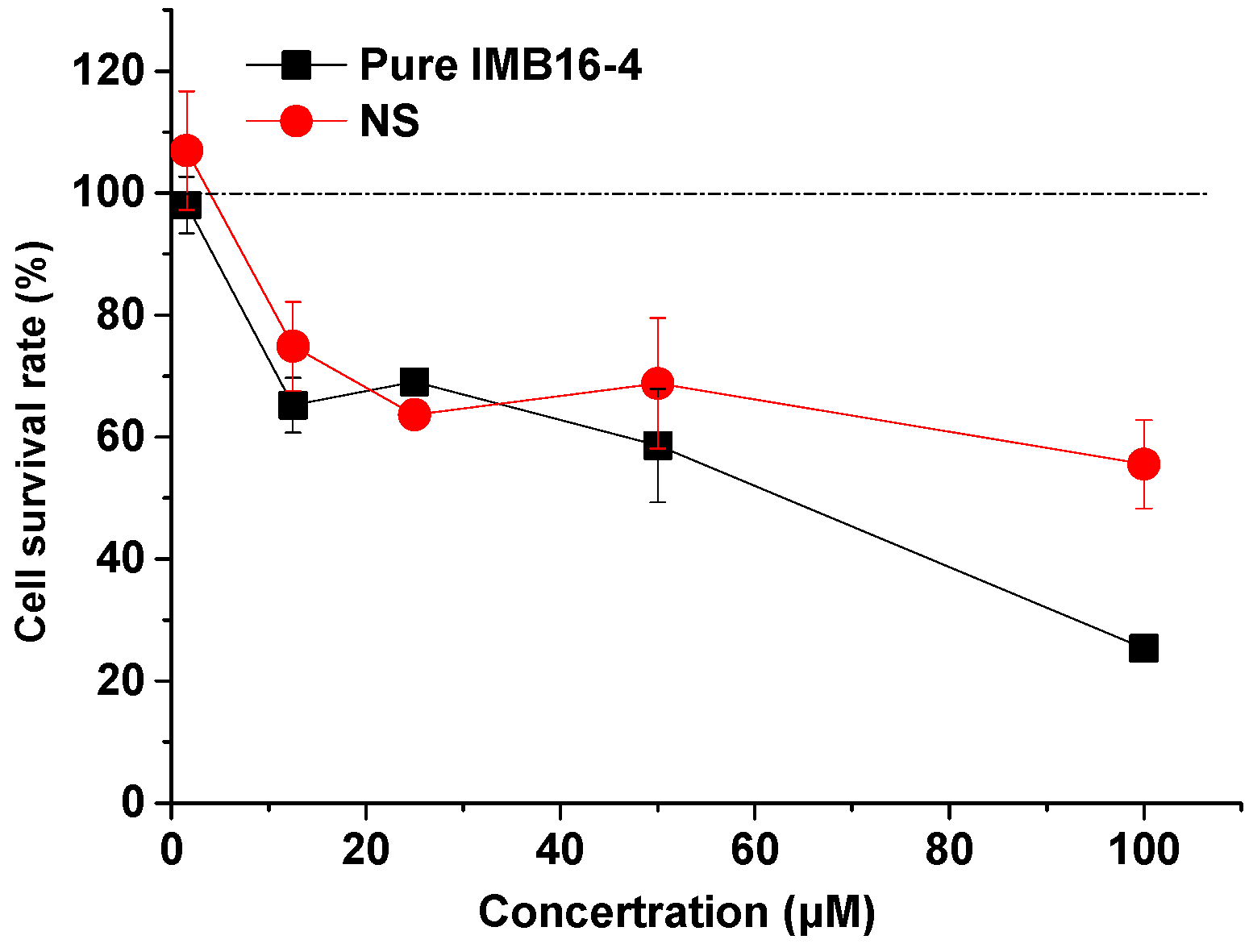
2.10. Cytotoxicity of NS on Human Hepatic Stellate Cells (LX-2 cells)
3. Materials and Methods
3.1. Materials
3.2. Preparation of IMB16-4 Nanoparticles
3.3. Long Term Stability Test of IMB16-4 Nanoparticles
3.4. Physicochemical Characterization of IMB16-4 Nanoparticles
3.5. Drug Loading Analysis by HPLC and TGA
3.6. Equilibrium Concentration Determination
3.7. In Vitro Release
3.8. Pharmacokinetics Study
3.9. Pharmacodynamics Study
3.9.1. Bile Duct Ligation (BDL) Surgery in Rats
3.9.2. Serum Biochemical Parameters
3.9.3. Histological Analysis of Liver Tissue
3.9.4. Western Blot Analysis
3.10. Cytotoxicity Assays
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El-Mezayen, N.S.; El-Hadidy, W.F.; El-Refaie, W.M.; Shalaby, T.I.; Khattab, M.M.; El-Khatib, A.S. Oral vitamin-A-coupled valsartan nanomedicine: High hepatic stellate cell receptors accessibility and prolonged enterohepatic residence. J. Control. Release 2018, 283, 32–44. [Google Scholar] [CrossRef]
- Ramakrishna, G.; Rastogi, A.; Trehanpati, N.; Sen, B.; Khosla, R.; Sarin, S.K. From Cirrhosis to Hepatocellular Carcinoma: New Molecular Insights on Inflammation and Cellular Senescence. Liver Cancer 2013, 2, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Iredale, J.P.; Campana, L. Regression of Liver Fibrosis. Semin. Liver Dis. 2017, 37, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.L.; Sheppard, D.; Duffield, J.S.; Violette, S. Therapy for Fibrotic Diseases: Nearing the Starting Line. Sci. Transl. Med. 2013, 5, 167sr1. [Google Scholar] [CrossRef] [PubMed]
- Altamirano, J.; Bataller, R. Alcoholic liver disease: Pathogenesis and new targets for therapy. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-Y.; Yuan, W.-G.; He, P.; Lei, J.-H.; Wang, C.-X. Liver fibrosis and hepatic stellate cells: Etiology, pathological hallmarks and therapeutic targets. World J. Gastroenterol. 2016, 22, 10512–10522. [Google Scholar] [CrossRef]
- Taimr, P. Fibrogenesis, hepatic stellate cells and the immune system. Falk Symp. 2004, 135, 36–41. [Google Scholar]
- Kisseleva, T.; Brenner, D. Role of hepatic stellate cells in fibrogenesis and the reversal of fibrosis. J. Gastroenterol. Hepatol. 2007, 22, S73–S78. [Google Scholar] [CrossRef]
- Inoue, T.; Jackson, E.K. Strong antiproliferative effects of baicalein in cultured rat hepatic stellate cells. Eur. J. Pharmacol. 1999, 378, 129–135. [Google Scholar] [CrossRef]
- Benyon, R.C.; Iredale, J.P.; Goddard, S.; Winwood, P.J.; Arthur, M.J. Expression of tissue inhibitor of metalloproteinases 1 and 2 is increased in fibrotic human liver. Gastroenterology 1996, 110, 821–831. [Google Scholar] [CrossRef]
- Iredale, J.P.; Benyon, R.C.; Pickering, J.; McCullen, M.; Northrop, M.; Pawley, S.; Hovell, C.; Arthur, M.J. Mechanisms of spontaneous resolution of rat liver fibrosis. Hepatic stellate cell apoptosis and reduced hepatic expression of metalloproteinase inhibitors. J. Clin. Investig. 1998, 102, 538–549. [Google Scholar] [CrossRef]
- Murphy, F.; Waung, J.; Collins, J.; Arthur, M.J.; Nagase, H.; Mann, D.; Benyon, R.C.; Iredale, J.P. N-Cadherin cleavage during activated hepatic stellate cell apoptosis is inhibited by tissue inhibitor of metalloproteinase-1. Comp. Hepatol 2004, 3, S8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murphy, F.R.; Issa, R.; Zhou, X.; Ratnarajah, S.; Nagase, H.; Arthur, M.J.; Benyon, C.; Iredale, J.P. Inhibition of apoptosis of activated hepatic stellate cells by tissue inhibitor of metalloproteinase-1 is mediated via effects on matrix metalloproteinase inhibition: Implications for reversibility of liver fibrosis. J. Biol. Chem. 2002, 277, 11069–11076. [Google Scholar] [CrossRef] [PubMed]
- Ikejima, K.; Okumura, K.; Kon, K.; Takei, Y.; Sato, N. Role of adipocytokines in hepatic fibrogenesis. J. Gastroenterol. Hepatol. 2007, 22 (Suppl. 1), S87–S92. [Google Scholar] [CrossRef] [PubMed]
- Derynck, R.; Zhang, Y.E. Smad-dependent and Smad-independent pathways in TGF-beta family signaling. Nature 2003, 425, 577–584. [Google Scholar] [CrossRef]
- Yi, C.; Shen, Z.; Stemmer-Rachamimov, A.; Dawany, N.; Troutman, S.; Showe, L.C.; Liu, Q.; Shimono, A.; Sudol, M.; Holmgren, L.; et al. The p130 isoform of angiomotin is required for Yap-mediated hepatic epithelial cell proliferation and tumorigenesis. Sci. Signal. 2013, 6, ra77. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, P.; Henderson, N.C. Antifibrotics in chronic liver disease: Tractable targets and translational challenges. Lancet Gastroenterol. Hepatol. 2016, 1, 328–340. [Google Scholar] [CrossRef]
- Sinha, B.; Müller, R.H.; Möschwitzer, J.P. Bottom-up approaches for preparing drug nanocrystals: Formulations and factors affecting particle size. Int. J. Pharm. 2013, 453, 126–141. [Google Scholar] [CrossRef]
- van Eerdenbrugh, B.; van den Mooter, G.; Augustijns, P. Top-down production of drug nanocrystals: Nanosuspension stabilization, miniaturization and transformation into solid products. Int. J. Pharm. 2008, 364, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Ain-Ai, A.; Gupta, P.K. Effect of arginine hydrochloride and hydroxypropyl cellulose as stabilizers on the physical stability of high drug loading nanosuspensions of a poorly soluble compound. Int. J. Pharm. 2008, 351, 282–288. [Google Scholar] [CrossRef]
- Li, X.; Yuan, H.; Zhang, C.; Chen, W.; Cheng, W.; Chen, X.; Ye, X. Preparation and in-vitro/in-vivo evaluation of curcumin nanosuspension with solubility enhancement. J. Pharm. Pharmacol. 2016, 68, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, Y.; Zhang, L.; Wang, Q.; Zhang, D. Stability of nanosuspensions in drug delivery. J. Control. Release 2013, 172, 1126–1141. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Hua, S.; Tian, Y.; Liu, J. Chemical and physical chitosan hydrogels as prospective carriers for drug delivery: A review. J. Mater. Chem. B 2020, 8, 10050–10064. [Google Scholar] [CrossRef] [PubMed]
- Bandi, S.P.; Bhatnagar, S.; Venuganti, V.V.K. Advanced materials for drug delivery across mucosal barriers. Acta Biomater. 2021, 119, 13–29. [Google Scholar] [CrossRef]
- Chithrani, D.B. Nanoparticles for improved therapeutics and imaging in cancer therapy. Recent Patents Nanotechnol. 2010, 4, 171–180. [Google Scholar] [CrossRef]
- Du, J.; Feng, B.; Dong, Y.; Zhao, M.; Yang, X.-D. Vanadium coordination compounds loaded on graphene quantum dots (GQDs) exhibit improved pharmaceutical properties and enhanced anti-diabetic effects. Nanoscale 2020, 12, 9219–9230. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Dubbu, S.; Kumari, N.; Kumar, A.; Lim, J.; Kim, S.; Lee, I.S. Magnetothermia-Induced Catalytic Hollow Nanoreactor for Bioorthogonal Organic Synthesis in Living Cells. Nano Lett. 2020, 20, 6981–6988. [Google Scholar] [CrossRef] [PubMed]
- Schuppan, D.; Ashfaq-Khan, M.; Yang, A.T.; Kim, Y.O. Liver fibrosis: Direct antifibrotic agents and targeted therapies. Matrix Biol. 2018, 68–69, 435–451. [Google Scholar] [CrossRef]
- Al-Kassas, R.; Bansal, M.; Shaw, J. Nanosizing techniques for improving bioavailability of drugs. J. Control. Release 2017, 260, 202–212. [Google Scholar] [CrossRef]
- Niu, X.; Wang, X.; Niu, B.; Li, G.; Yang, X.; Wang, Y.; Li, G. Novel IMB16-4 Compound Loaded into Silica Nanoparticles Exhibits Enhanced Oral Bioavailability and Increased Anti-Liver Fibrosis In Vitro. Molecules 2021, 26, 1545. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhi, Z.; Jiang, T.; Zhang, J.; Wang, Z.; Wang, S. Spherical mesoporous silica nanoparticles for loading and release of the poorly water-soluble drug telmisartan. J. Control. Release 2010, 145, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.; Rahman, Z.; Korang-Yeboah, M.; Khan, M.A. Development and validation of X-ray diffraction method for quan-titative determination of crystallinity in warfarin sodium products. Int. J. Pharm. 2015, 493, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Sever, A.Z.; Sever, M.; Gojkovic, S.; Krezic, I.; Zizek, H.; Malekinusic, D.; Durasin, T.; Drmic, D.; Vranes, H.; Dretar, V.; et al. Sikiric, Pentadecapeptide Bpc 157 Therapy in Bile Duct Ligated (Bdl) Rats. In Rats with Cirrhosis and Portal Hypertension, Bpc 157 May Be Useful Also in the Conditions of the Increased Intracranial Pressure and Vessels Obstruction. Gastroenterology 2020, 158, S502. [Google Scholar] [CrossRef]
- Chen, L.X.; O’Brien, A.; Meng, F.Y.; Wu, N.; White, T.; Venter, J.; Francis, H.L.; Zhou, T.H.; Kyritsi, K.; Gaudio, E.; et al. Pinealectomy or Prolonged Light Exposure Exacerbates Biliary Proliferation and Senescence and Liver Fibrosis in Bile Duct Ligated (BDL) Rats Via Decreased Biliary Melatonin Synthesis. Hepatology 2018, 68, 830a–831a. [Google Scholar]
- Li, Y.; Liu, F.; Ding, F.; Chen, P.; Tang, M. Inhibition of liver fibrosis using vitamin A-coupled liposomes to deliver matrix metalloproteinase-2 siRNA in vitro. Mol. Med. Rep. 2015, 12, 3453–3461. [Google Scholar] [CrossRef]
- Ge, M.; Liu, H.; Zhang, Y.; Li, N.; Zhao, S.; Zhao, W.; Zhen, Y.; Yu, J.; He, H.; Shao, R.G. The anti-hepatic fibrosis effects of dihydrotanshinone I are mediated by disrupting the yes-associated protein and transcriptional enhancer factor D2 complex and stimulating autophagy. Br. J. Pharmacol. 2017, 174, 1147–1160. [Google Scholar] [CrossRef]



| Time (h) | 25 °C | 37 °C | ||
|---|---|---|---|---|
| Mean PS ± SD | PDI | Mean PS ± SD | PDI | |
| 0 | 112.4 ± 43.9 | 0.083 | 124.8 ± 40.0 | 0.073 |
| 2 | 118.0 ± 38.5 | 0.078 | 119.2 ± 41.2 | 0.078 |
| 4 | 119.8 ± 40.5 | 0.093 | 301.8 ± 61.8 | 0.373 |
| 6 | 117.7 ± 47.8 | 0.108 | 268.2 ± 172.7 | 0.606 |
| Samples | Mean PS ± SD | PDI | Mean ZP ± SD | Degree of Drug Loading (%) |
|---|---|---|---|---|
| NS | 164.3 ± 78.5 | 0.150 | −15.6 ± 6.7 | 34.9 |
| NS after one year | 174.3 ± 83.6 | 0.218 | −13.6 ± 5.8 | 33.8 |
| Pharmacokinetic Parameter | Pure IMB16-4 | NS |
|---|---|---|
| Cmax (mg/L) | 0.68 ± 0.11 | 46.98 ± 13.0 |
| Tmax (h) | 2.25 ± 1.33 | 1.17 ± 1.08 |
| AUC0~12h (mg/L h) | 5.95 ± 0.57 | 155.5 ± 70.5 |
| Sham | BDL | BDL-IMB16-4 | BDL-NS | |
|---|---|---|---|---|
| ALT (U L−1) | 39.71 ± 9.25 | 80.29 ± 12.80 ### | 78.86 ± 12.93 | 56.00 ± 15.46 * |
| AST (U L−1) | 128.86 ± 49.81 | 405.00 ± 57.07 ### | 317.43 ± 74.65 * | 297.71 ± 95.08 * |
| ALP (U L−1) | 191.14 ± 38.10 | 311.57 ± 65.91 ### | 328.00 ± 47.62 | 235.00 ± 36.97 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, X.; Wang, X.; Niu, B.; Wang, Y.; He, H.; Li, G. New IMB16-4 Nanoparticles Improved Oral Bioavailability and Enhanced Anti-Hepatic Fibrosis on Rats. Pharmaceuticals 2022, 15, 85. https://doi.org/10.3390/ph15010085
Niu X, Wang X, Niu B, Wang Y, He H, Li G. New IMB16-4 Nanoparticles Improved Oral Bioavailability and Enhanced Anti-Hepatic Fibrosis on Rats. Pharmaceuticals. 2022; 15(1):85. https://doi.org/10.3390/ph15010085
Chicago/Turabian StyleNiu, Xia, Xiaomei Wang, Bingyu Niu, Yucheng Wang, Hongwei He, and Guiling Li. 2022. "New IMB16-4 Nanoparticles Improved Oral Bioavailability and Enhanced Anti-Hepatic Fibrosis on Rats" Pharmaceuticals 15, no. 1: 85. https://doi.org/10.3390/ph15010085
APA StyleNiu, X., Wang, X., Niu, B., Wang, Y., He, H., & Li, G. (2022). New IMB16-4 Nanoparticles Improved Oral Bioavailability and Enhanced Anti-Hepatic Fibrosis on Rats. Pharmaceuticals, 15(1), 85. https://doi.org/10.3390/ph15010085






