Impact of Selected Small-Molecule Kinase Inhibitors on Lipid Membranes
Abstract
1. Introduction
2. Results
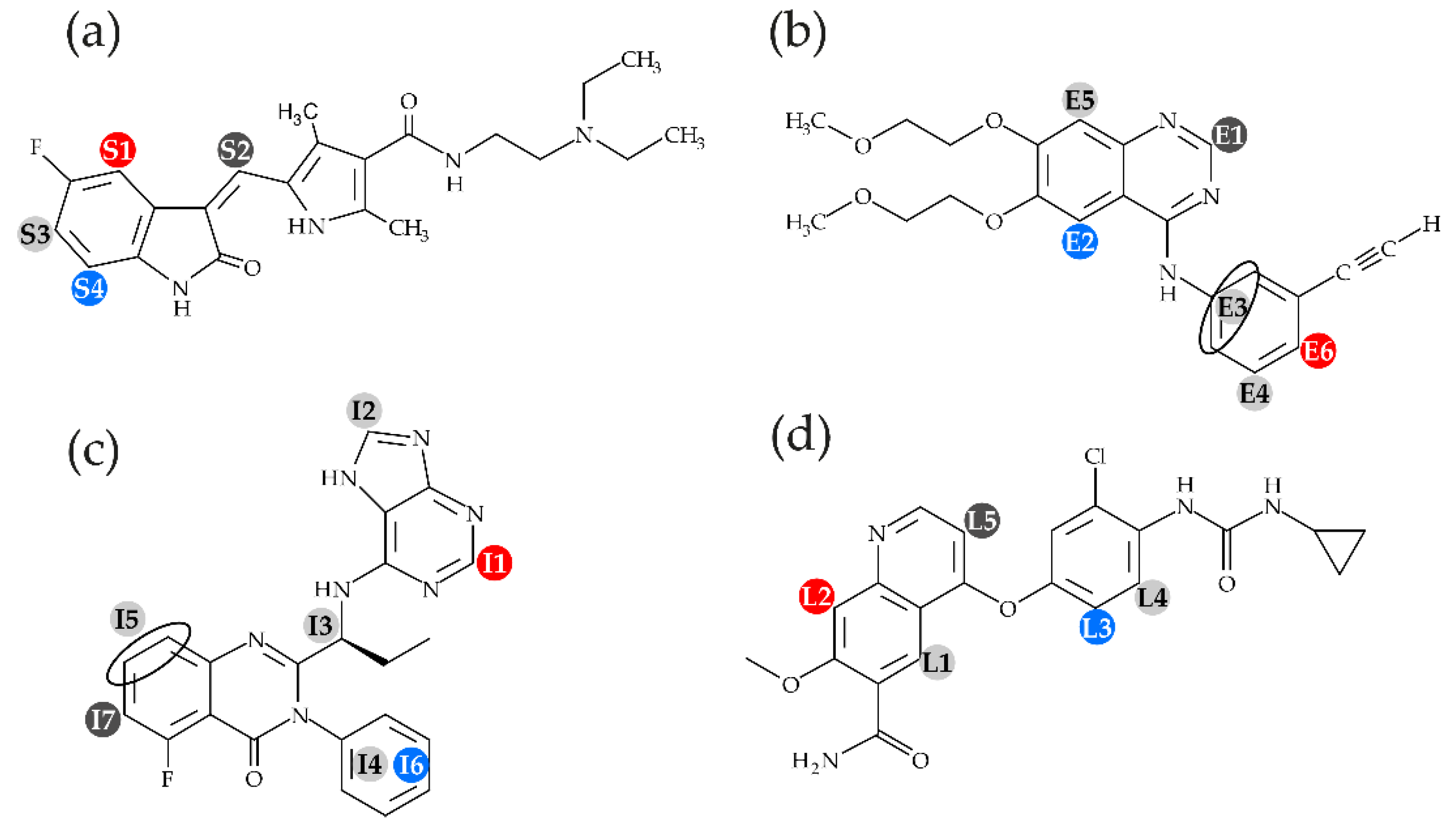
2.1. Localization of Kinase Inhibitors in the Membrane
2.2. Influence of Kinase Inhibitors on Headgroup and Acyl Chain Behavior of the Phospholipid Membrane
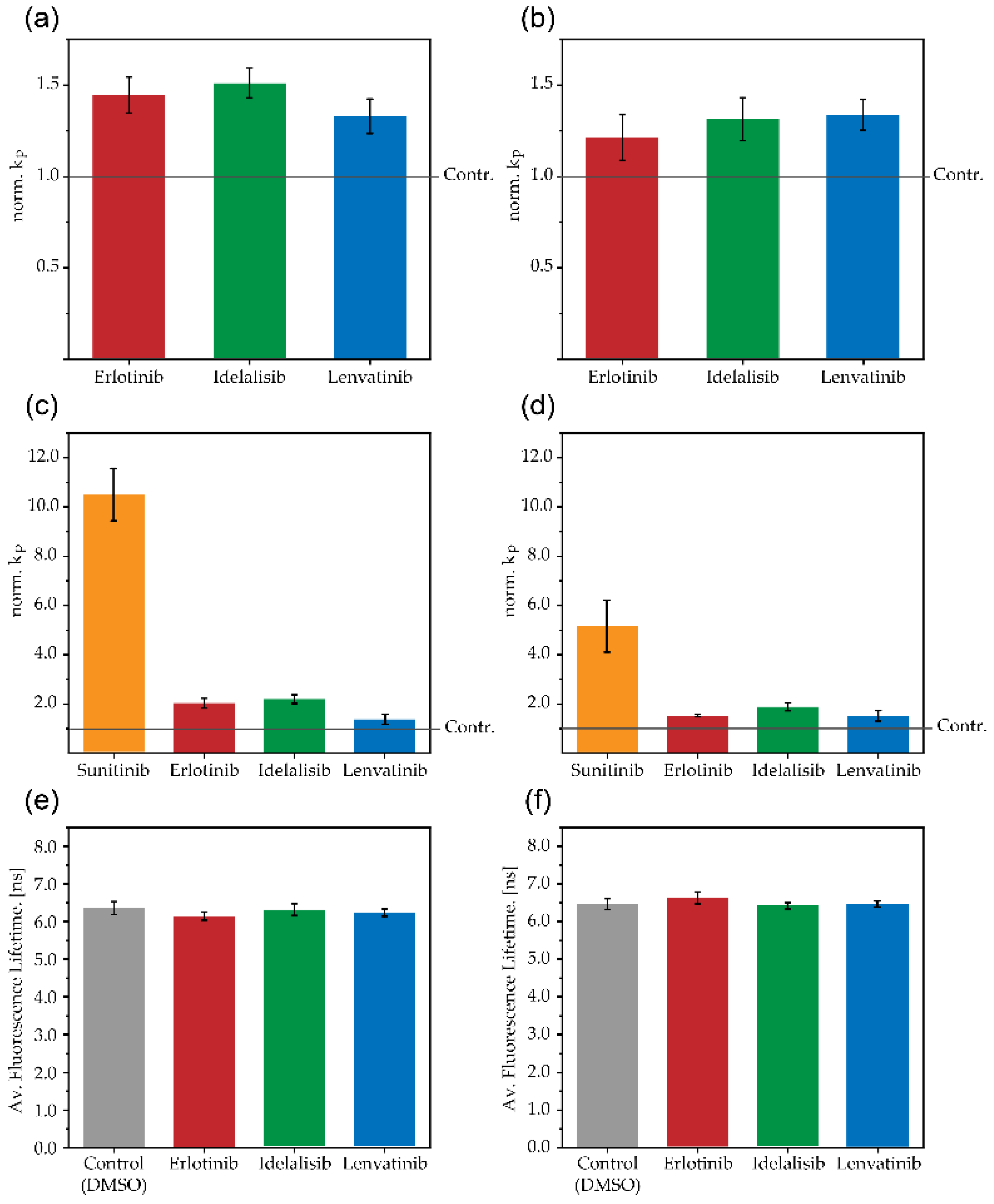
2.3. Influence of Kinase Inhibitors on Transmembrane Permeation of Anions
2.4. Influence of Kinase Inhibitors on Fluorescence Lifetime of NBD Labelled Vesicles
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Preparation of LUVs
4.3. Sample Preparation for NMR Experiments
4.4. H NMR and 31P NMR Spectroscopy
4.5. H MAS NMR Spectroscopy
4.6. Measurement of Membrane Permeation of Polar Molecules
4.7. Measurement of Fluorescence Lifetime
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Haralampiev, I.; Alonso de Armiño, D.J.; Luck, M.; Fischer, M.; Abel, T.; Huster, D.; Di Lella, S.; Scheidt, H.A.; Müller, P. Interaction of the small-molecule kinase inhibitors tofacitinib and lapatinib with membranes. Biochim. Biophys. Acta 2020, 1862, 183414. [Google Scholar] [CrossRef]
- Stebbing, J.; Phelan, A.; Griffin, I.; Tucker, C.; Oechsle, O.; Smith, D.; Richardson, P. COVID-19: Combining antiviral and anti-inflammatory treatments. Lancet Infect. Dis. 2020, 20, 400–402. [Google Scholar] [CrossRef]
- Fischer, M.; Luck, M.; Werle, M.; Scheidt, H.A.; Müller, P. Binding of the small-molecule kinase inhibitor ruxolitinib to membranes does not disturb membrane integrity. Biochem. Biophys. Rep. 2020, 24, 100838. [Google Scholar] [CrossRef]
- Bareschino, M.A.; Schettino, C.; Troiani, T.; Martinelli, E.; Morgillo, F.; Ciardiello, F. Erlotinib in cancer treatment. Ann. Oncol. 2007, 18, vi35–vi41. [Google Scholar] [CrossRef]
- Rosell, R.; Carcereny, E.; Gervais, R.; Vergnenegre, A.; Massuti, B.; Felip, E.; Palmero, R.; Garcia-Gomez, R.; Pallares, C.; Sanchez, J.M.; et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): A multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012, 13, 239–246. [Google Scholar] [CrossRef]
- Scagliotti, G.V.; Krzakowski, M.; Szczesna, A.; Strausz, J.; Makhson, A.; Reck, M.; Wierzbicki, R.F.; Albert, I.; Thomas, M.; Miziara, J.E.A.; et al. Sunitinib plus erlotinib versus placebo plus erlotinib in patients with previously treated advanced non–small-cell lung cancer: A phase III trial. J. Clin. Oncol. 2012, 30, 2070–2078. [Google Scholar] [CrossRef] [PubMed]
- Demetri, G.D.; van Oosterom, A.T.; Garrett, C.R.; Blackstein, M.E.; Shah, M.H.; Verweij, J.; McArthur, G.; Judson, I.R.; Heinrich, M.C.; Morgan, J.A.; et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: A randomised controlled trial. Lancet 2006, 368, 1329–1338. [Google Scholar] [CrossRef]
- Raymond, E.; Dahan, L.; Raoul, J.-L.; Bang, Y.-J.; Borbath, I.; Lombard-Bohas, C.; Valle, J.; Metrakos, P.; Smith, D.; Vinik, A.; et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 2011, 364, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; Habra, M.A. Lenvatinib: Role in thyroid cancer and other solid tumors. Cancer Treat. Rev. 2016, 42, 47–55. [Google Scholar] [CrossRef]
- Akinleye, A.; Avvaru, P.; Furqan, M.; Song, Y.; Liu, D. Phosphatidylinositol 3-kinase (PI3K) inhibitors as cancer therapeutics. J. Hematol. Oncol. 2013, 6, 88. [Google Scholar] [CrossRef]
- Furman, R.R.; Sharman, J.P.; Coutre, S.E.; Cheson, B.D.; Pagel, J.M.; Hillmen, P.; Barrientos, J.C.; Zelenetz, A.D.; Kipps, T.J.; Flinn, I.; et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 2014, 370, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Graf, S.A.; Gopal, A.K. Idelalisib for the treatment of non-Hodgkin lymphoma. Expert Opin. Pharmacother. 2016, 17, 265–274. [Google Scholar] [CrossRef]
- Scheidt, H.A.; Huster, D. The Interaction of small molecules with phospholipid membranes studied by 1H NOESY NMR under Magic-Angle Spinning. Acta Pharmacol. Sin. 2008, 29, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Haralampiev, I.; Scheidt, H.A.; Abel, T.; Luckner, M.; Herrmann, A.; Huster, D.; Müller, P. The interaction of sorafenib and regorafenib with membranes is modulated by their lipid composition. Biochim. Biophys. Acta 2016, 1858, 2871–2881. [Google Scholar] [CrossRef]
- Petrache, H.I.; Dodd, S.W.; Brown, M.F. Area per lipid and acyl length distributions in fluid phosphatidylcholines determined by (2)H NMR spectroscopy. Biophys. J. 2000, 79, 3172–3192. [Google Scholar] [CrossRef]
- Nowak-Sliwinska, P.; Weiss, A.; van Beijnum, J.R.; Wong, T.J.; Kilarski, W.W.; Szewczyk, G.; Verheul, H.M.W.; Sarna, T.; van den Bergh, H.; Griffioen, A.W. Photoactivation of lysosomally sequestered sunitinib after angiostatic treatment causes vascular occlusion and enhances tumor growth inhibition. Cell Death Dis. 2015, 6, e1641. [Google Scholar] [CrossRef]
- Bastos, A.E.P.; Scolari, S.; Stöckl, M.; de Almeida, R.F.M. Chapter three—Applications of fluorescence lifetime spectroscopy and imaging to lipid domains In Vivo. Meth. Enzymol. 2012, 504, 57–81. [Google Scholar]
- Huster, D.; Müller, P.; Arnold, K.; Herrmann, A. Dynamics of membrane penetration of the fluorescent 7-nitrobenz-2-oxa-1,3-diazol-4-yl (NBD) group attached to an acyl chain of phosphatidylcholine. Biophys. J. 2001, 80, 822–831. [Google Scholar] [CrossRef]
- Loura, L.M.S.; Ramalho, J.P.P. Location and dynamics of acyl chain NBD-labeled phosphatidylcholine (NBD-PC) in DPPC bilayers. A molecular dynamics and time-resolved fluorescence anisotropy study. Biochim. Biophys. Acta 2007, 1768, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Raghuraman, H.; Dasgupta, S.; Chattopadhyay, A. Organization and dynamics of N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)-labeled lipids: A fluorescence approach. Chem. Phys. Lipids 2004, 127, 91–101. [Google Scholar] [CrossRef]
- Dias, M.; Hadgraft, J.; Lane, M.E. Influence of membrane–solvent–solute interactions on solute permeation in model membranes. Int. J. Pharm. 2007, 336, 108–114. [Google Scholar] [CrossRef]
- Roskoski, R., Jr. Classification of small molecule protein kinase inhibitors based upon the structures of their drug-enzyme complexes. Pharmacol. Res. 2016, 103, 26–48. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R. Properties of FDA-approved small molecule phosphatidylinositol 3-kinase inhibitors prescribed for the treatment of malignancies. Pharmacol. Res. 2021, 168, 105579. [Google Scholar] [CrossRef]
- Alves, A.C.; Ribeiro, D.; Nunes, C.; Reis, S. Biophysics in cancer: The relevance of drug-membrane interaction studies. Biochim. Biophys. Acta 2016, 1858, 2231–2244. [Google Scholar] [CrossRef]
- Porta, C.; Paglino, C.; Imarisio, I.; Bonomi, L. Uncovering Pandora’s vase: The growing problem of new toxicities from novel anticancer agents. The case of sorafenib and sunitinib. Clin. Exp. Med. 2007, 7, 127–134. [Google Scholar] [CrossRef] [PubMed]
- McMullen, C.J.; Chalmers, S.; Wood, R.; Cunningham, M.R.; Currie, S. Sunitinib and imatinib display differential cardiotoxicity in adult rat cardiac fibroblasts that involves a role for calcium/calmodulin dependent protein kinase II. Front. Cardiovasc. Med. 2020, 7, 630480. [Google Scholar] [CrossRef]
- Damaraju, V.L.; Kuzma, M.; Cass, C.E.; Putman, C.T.; Sawyer, M.B. Multitargeted kinase inhibitors imatinib, sorafenib and sunitinib perturb energy metabolism and cause cytotoxicity to cultured C2C12 skeletal muscle derived myotubes. Biochem. Pharmacol. 2018, 155, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R. Phosphatidylinositol 3 kinase δ inhibitors: Present and future. Cancer J. 2019, 25, 394–400. [Google Scholar] [CrossRef]
- Paech, F.; Bouitbir, J.; Krähenbühl, S. Hepatocellular toxicity associated with tyrosine kinase inhibitors: Mitochondrial damage and inhibition of glycolysis. Front. Pharmacol. 2017, 8, 367. [Google Scholar] [CrossRef]
- Peters, G.H.; Bywater, R.P. Influence of a lipid interface on protein dynamics in a fungal lipase. Biophys. J. 2001, 81, 3052–3065. [Google Scholar] [CrossRef][Green Version]
- Rujiviphat, J.; Meglei, G.; Rubinstein, J.L.; McQuibban, G.A. Phospholipid association is essential for dynamin-related protein Mgm1 to function in mitochondrial membrane fusion. J. Biol. Chem. 2009, 284, 28682–28686. [Google Scholar] [CrossRef] [PubMed]
- Nasr, M.L.; Shi, X.; Bowman, A.L.; Johnson, M.; Zvonok, N.; Janero, D.R.; Vemuri, V.K.; Wales, T.E.; Engen, J.R.; Makriyannis, A. Membrane phospholipid bilayer as a determinant of monoacylglycerol lipase kinetic profile and conformational repertoire. Prot. Sci. 2013, 22, 774–787. [Google Scholar] [CrossRef]
- Zhou, Q.; Lv, H.; Mazloom, A.R.; Xu, H.; Ma’ayan, A.; Gallo, J.M. Activation of alternate prosurvival pathways accounts for acquired sunitinib resistance in U87MG glioma xenografts. J. Pharmacol. Exp. Ther. 2012, 343, 509–519. [Google Scholar] [CrossRef]
- Saleeb, R.M.; Farag, M.; Lichner, Z.; Brimo, F.; Bartlett, J.; Bjarnason, G.; Finelli, A.; Rontondo, F.; Downes, M.R.; Yousef, G.M. Modulating ATP binding cassette transporters in papillary renal cell carcinoma type 2 enhances its response to targeted molecular therapy. Mol. Oncol. 2018, 12, 1673–1688. [Google Scholar] [CrossRef] [PubMed]
- Elmeliegy, M.A.; Carcaboso, A.M.; Tagen, M.; Bai, F.; Stewart, C.F. Role of ATP-binding cassette and solute carrier transporters in erlotinib CNS penetration and intracellular accumulation. Clin. Cancer Res. 2011, 17, 89–99. [Google Scholar] [CrossRef]
- Hu, S.; Chen, Z.; Franke, R.; Orwick, S.; Zhao, M.; Rudek, M.A.; Sparreboom, A.; Baker, S.D. Interaction of the multikinase inhibitors sorafenib and sunitinib with solute carriers and ATP-binding cassette transporters. Clin. Cancer Res. 2009, 15, 6062–6069. [Google Scholar] [CrossRef]
- Sugawara, S.; Takayanagi, M.; Honda, S.; Tatsuta, T.; Fujii, Y.; Ozeki, Y.; Ito, J.; Sato, M.; Hosono, A.M. Catfish egg lectin affects influx and efflux rates of sunitinib in human cervical carcinoma HeLa cells. Glycobiology 2020, 30, 802–816. [Google Scholar] [CrossRef]
- Zimmerman, E.I.; Hu, S.; Roberts, J.L.; Gibson, A.A.; Orwick, S.J.; Li, L.; Sparreboom, A.; Baker, S.D. Contribution of OATP1B1 and OATP1B3 to the disposition of sorafenib and sorafenib-glucuronide. Clin. Cancer Res. 2013, 19, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Kameswaran, M.; Pandey, U.; Prabhash, K.; Sarma, H.D.; Dash, A. (68)Ga labeled Erlotinib: A novel PET probe for imaging EGFR over-expressing tumors. Bioorg. Med. Chem. Lett. 2017, 27, 4552–4557. [Google Scholar] [CrossRef]
- Bauer, M.; Matsuda, A.; Wulkersdorfer, B.; Philippe, C.; Traxl, A.; Özvegy-Laczka, C.; Stanek, J.; Nics, L.; Klebermass, E.M.; Poschner, S.; et al. Influence of OATPs on hepatic disposition of erlotinib measured with positron emission tomography. Clin. Pharmacol. Ther. 2018, 104, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Altintas, I.; Heukers, R.; van der Meel, R.; Lacombe, M.; Amidi, M.; van Bergen En Henegouwen, P.M.; Hennink, W.E.; Schiffelers, R.M.; Kok, R.J. Nanobody-albumin nanoparticles (NANAPs) for the delivery of a multikinase inhibitor 17864 to EGFR overexpressing tumor cells. J. Control. Release 2013, 165, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Dora, C.P.; Kushwah, V.; Katiyar, S.S.; Kumar, P.; Pillay, V.; Suresh, S.; Jain, S. Improved oral bioavailability and therapeutic efficacy of erlotinib through molecular complexation with phospholipid. Int. J. Pharm. 2017, 534, 1–13. [Google Scholar] [CrossRef]
- Shi, J.F.; Sun, M.G.; Li, X.Y.; Zhao, Y.; Ju, R.J.; Mu, L.M.; Yan, Y.; Li, X.T.; Zeng, F.; Lu, W.L. A combination of targeted sunitinib liposomes and targeted vinorelbine liposomes for treating invasive breast cancer. J. Biomed. Nanotechnol. 2015, 11, 1568–1582. [Google Scholar] [CrossRef]
- Li, F.; Mei, H.; Gao, Y.; Xie, X.; Nie, H.; Li, T.; Zhang, H.; Jia, L. Co-delivery of oxygen and erlotinib by aptamer-modified liposomal complexes to reverse hypoxia-induced drug resistance in lung cancer. Biomaterials 2017, 145, 56–71. [Google Scholar] [CrossRef]
- Lakkadwala, S.; Dos Santos Rodrigues, B.; Sun, C.; Singh, J. Biodistribution of TAT or QLPVM coupled to receptor targeted liposomes for delivery of anticancer therapeutics to brain In Vitro and In Vivo. Nanomed. Nanotechnol. Biol. Med. 2020, 23, 102112. [Google Scholar] [CrossRef]
- Saber, M.M.; Bahrainian, S.; Dinarvand, R.; Atyabi, F. Targeted drug delivery of Sunitinib Malate to tumor blood vessels by cRGD-chiotosan-gold nanoparticles. Int. J. Pharm. 2017, 517, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Lakkadwala, S.; Singh, J. Co-delivery of doxorubicin and erlotinib through liposomal nanoparticles for glioblastoma tumor regression using an In Vitro brain tumor model. Colloids Surf. B 2019, 173, 27–35. [Google Scholar] [CrossRef]
- Dora, C.P.; Trotta, F.; Kushwah, V.; Devasari, N.; Singh, C.; Suresh, S.; Jain, S. Potential of erlotinib cyclodextrin nanosponge complex to enhance solubility, dissolution rate, in vitro cytotoxicity and oral bioavailability. Carbohydr. Polym. 2016, 137, 339–349. [Google Scholar] [CrossRef]
- Fellmann, P.; Zachowski, A.; Devaux, P.F. Synthesis and use of spin-labeled lipids for studies of the transmembrane movement of phospholipids. In Biomembrane Protocols: II. Architecture and Function; Graham, J.M., Higgins, J.A., Eds.; Humana Press Inc.: Totowa, NJ, USA, 1994; pp. 161–175. [Google Scholar]
- Mayer, L.D.; Hope, M.J.; Cullis, R.P.; Janoff, A.S. Solute distributions and trapping efficiencies observed in freeze-thawed multilamellar vesicles. Biochim. Biophys. Acta 1985, 817, 193–196. [Google Scholar] [CrossRef]
- Davis, J.H.; Jeffrey, K.R.; Bloom, M.; Valic, M.I.; Higgs, T.P. Quadrupolar echo deuteron magnetic resonance spectroscopy in ordered hydrocarbon chains. Chem. Phys. Lett. 1976, 42, 390–394. [Google Scholar] [CrossRef]
- Sternin, E.; Bloom, M.; Mackay, A.L. De-pake-ing of NMR spectra. J. Magn. Reson. 1983, 55, 274–282. [Google Scholar] [CrossRef]
- Lafleur, M.; Fine, B.; Sternin, E.; Cullis, P.R.; Bloom, M. Smoothed orientational order profile of lipid bilayers by 2H-nuclear magnetic resonance. Biophys. J. 1989, 56, 1037–1041. [Google Scholar] [CrossRef]
- McIntyre, J.C.; Sleight, R.G. Fluorescence assay for phospholipid membrane asymmetry. Biochemistry 1991, 30, 11819–11827. [Google Scholar] [CrossRef]
- Pomorski, T.; Herrmann, A.; Zachowski, A.; Devaux, P.F.; Müller, P. Rapid determination of the transbilayer distribution of NBD-phospholipids in erythrocyte membranes with dithionite. Mol. Membr. Biol. 1994, 11, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Greube, A.; Müller, K.; Töpfer-Petersen, E.; Herrmann, A.; Müller, P. Influence of the bovine seminal plasma protein PDC-109 on the physical state of membranes. Biochemistry 2001, 40, 8326–8334. [Google Scholar] [CrossRef] [PubMed]
- Langner, M.; Hui, S.W. Dithionite penetration through phospholipid bilayers as a measure of defects in lipid molecular packing. Chem. Phys. Lipids 1993, 65, 23–30. [Google Scholar] [CrossRef]
- Tannert, A.; Töpfer-Petersen, E.; Herrmann, A.; Müller, K.; Müller, P. The lipid composition modulates the influence of the bovine seminal plasma protein PDC-109 on membrane stability. Biochemistry 2007, 46, 11621–11629. [Google Scholar] [CrossRef] [PubMed]


| 31P CSA (ppm) | Lc*[Å] | <S> | |
|---|---|---|---|
| POPC | 45 | 11.0 | 0.144 |
| +Erlotinib | 45 | 11.6 | 0.154 |
| +Lenvatinib | 44 | 11.2 | 0.147 |
| +Sunitinib | 47 | 10.6 | 0.130 |
| +Idelalisib | 44 | 10.8 | 0.136 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luck, M.; Fischer, M.; Werle, M.; Scheidt, H.A.; Müller, P. Impact of Selected Small-Molecule Kinase Inhibitors on Lipid Membranes. Pharmaceuticals 2021, 14, 746. https://doi.org/10.3390/ph14080746
Luck M, Fischer M, Werle M, Scheidt HA, Müller P. Impact of Selected Small-Molecule Kinase Inhibitors on Lipid Membranes. Pharmaceuticals. 2021; 14(8):746. https://doi.org/10.3390/ph14080746
Chicago/Turabian StyleLuck, Meike, Markus Fischer, Maximilian Werle, Holger A. Scheidt, and Peter Müller. 2021. "Impact of Selected Small-Molecule Kinase Inhibitors on Lipid Membranes" Pharmaceuticals 14, no. 8: 746. https://doi.org/10.3390/ph14080746
APA StyleLuck, M., Fischer, M., Werle, M., Scheidt, H. A., & Müller, P. (2021). Impact of Selected Small-Molecule Kinase Inhibitors on Lipid Membranes. Pharmaceuticals, 14(8), 746. https://doi.org/10.3390/ph14080746






