Clinical-Based vs. Model-Based Adaptive Dosing Strategy: Retrospective Comparison in Real-World mRCC Patients Treated with Sunitinib
Abstract
1. Introduction
2. Results
2.1. Patients and Treatments
2.2. Clinical Outcome
2.3. Clinical-Based Dose Tailoring and Treatment Discontinuation
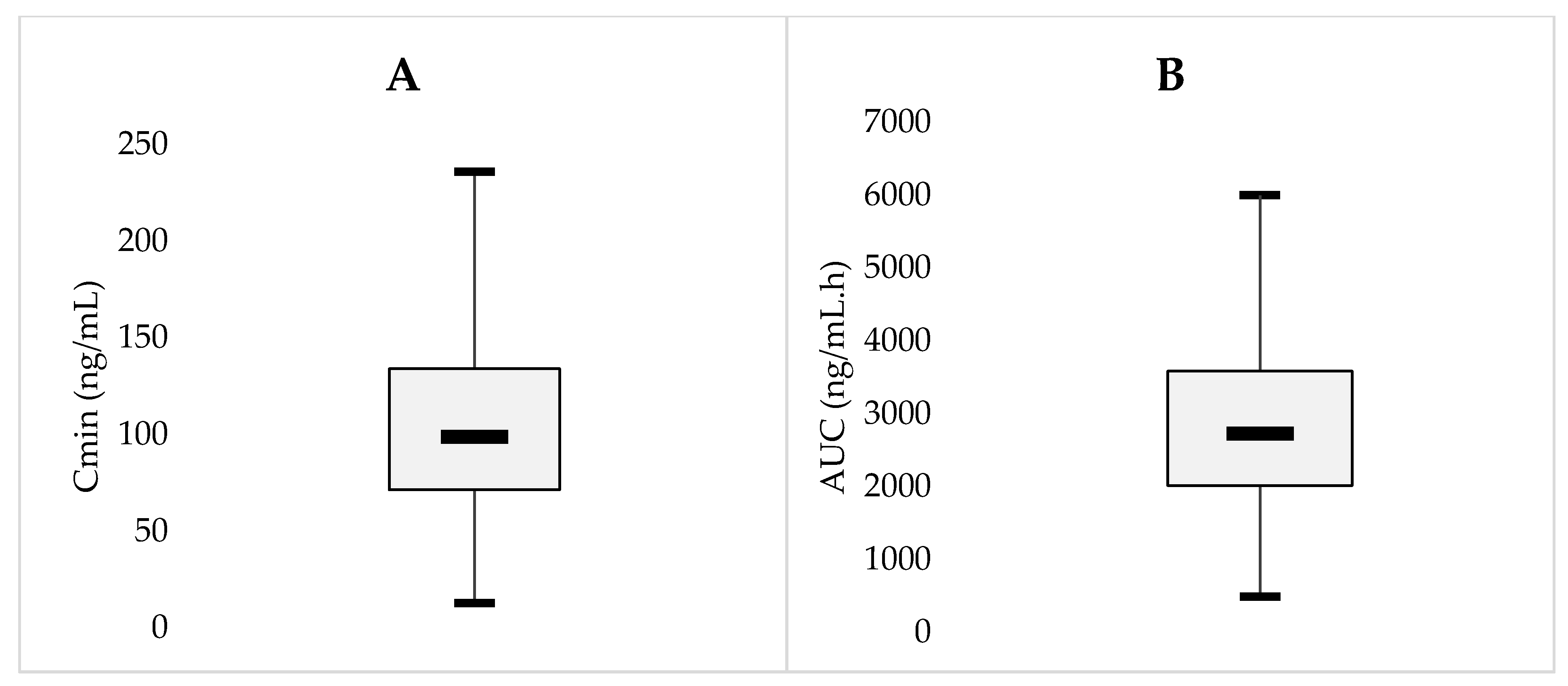
2.4. Model Simulations for Trough Concentrations and AUC
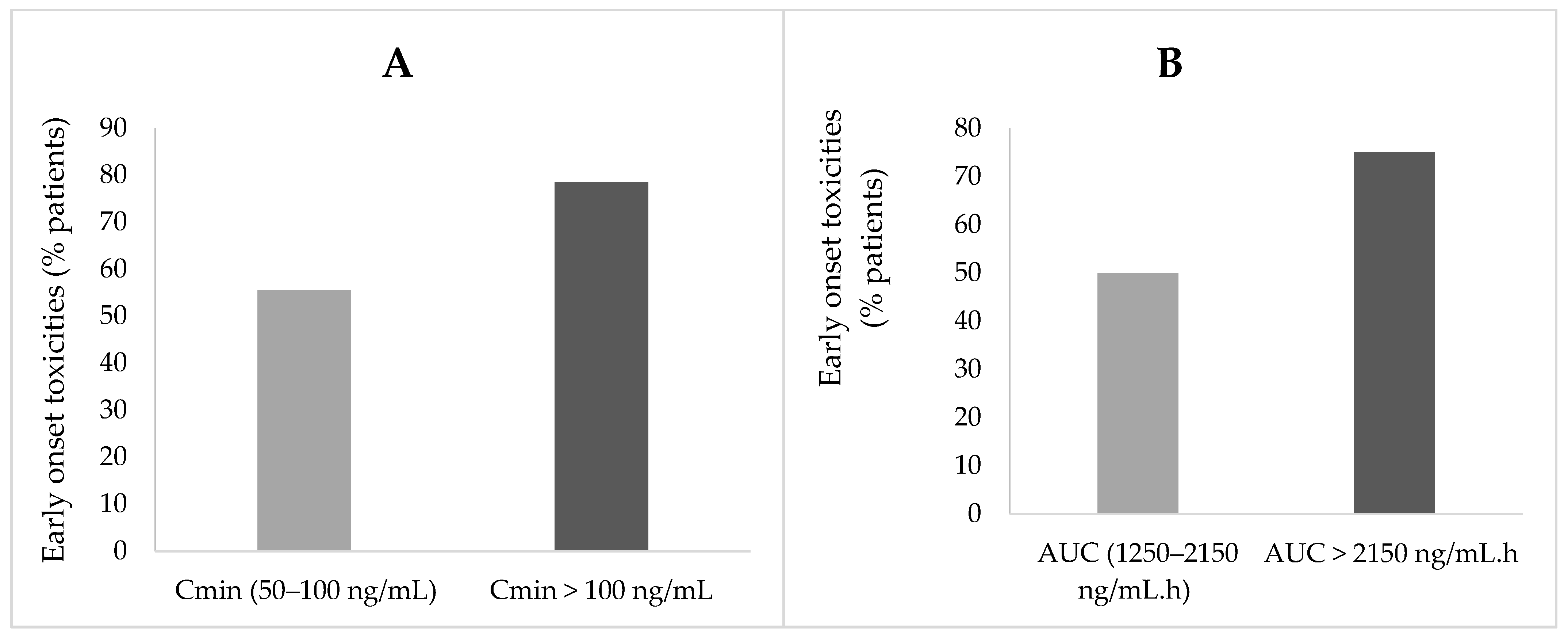
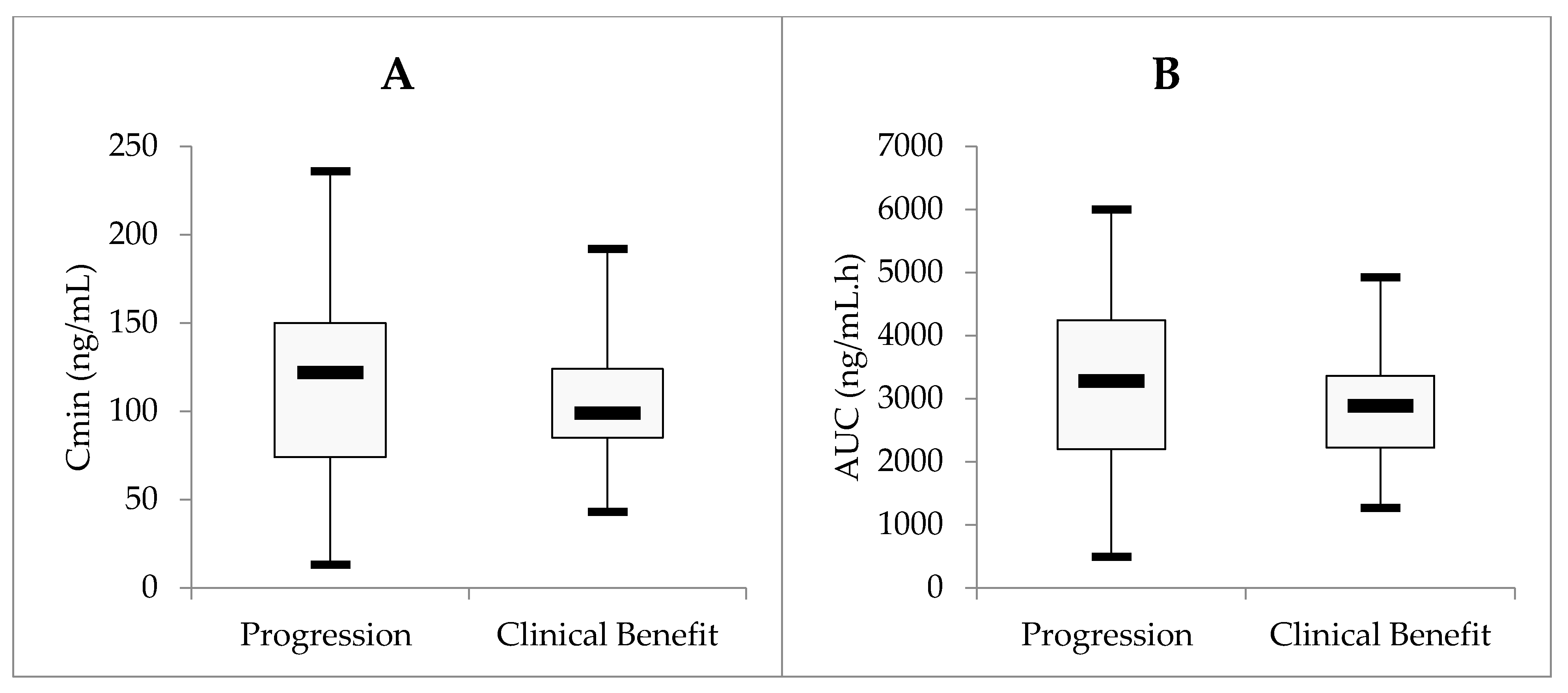
2.5. Sunitinib Exposure and Clinical Outcome
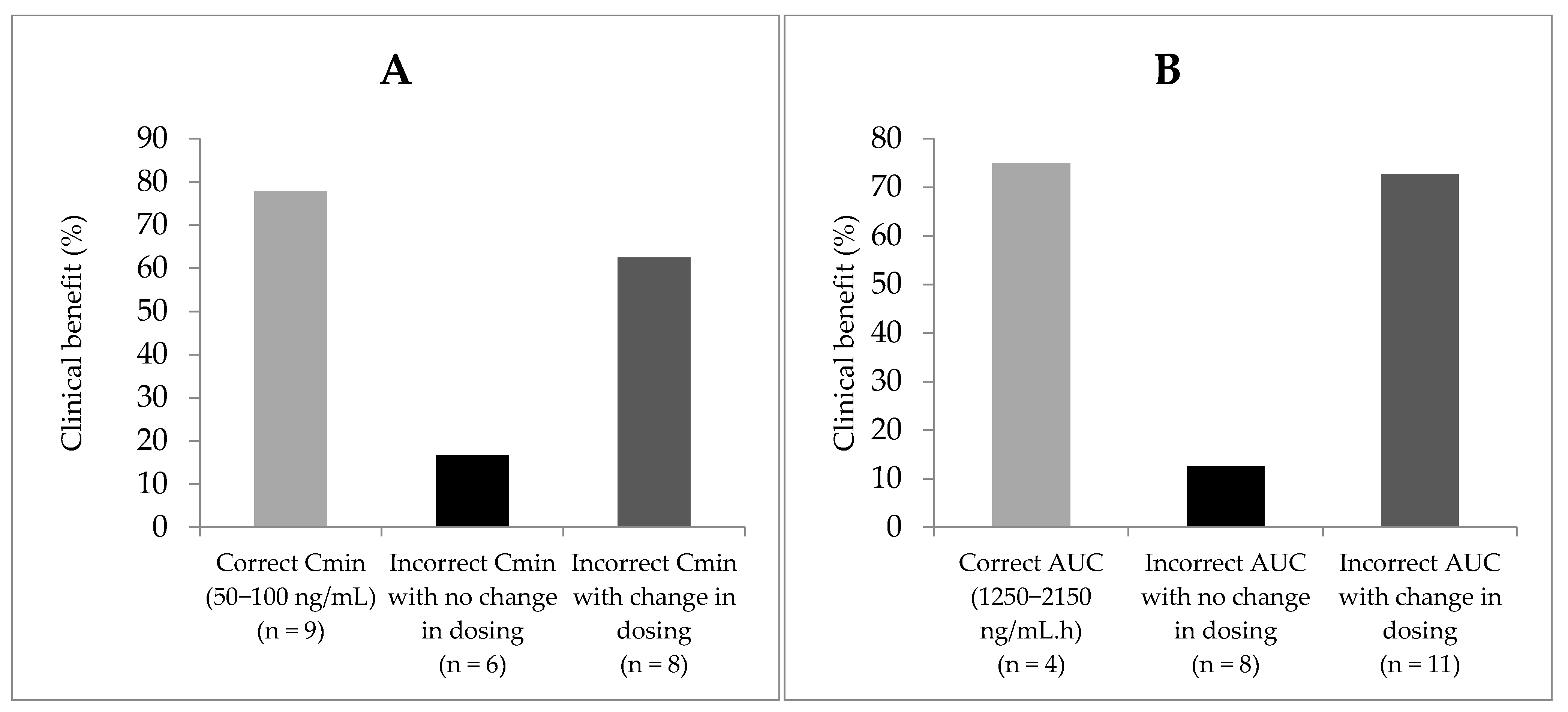
2.6. Efficacy of Sunitinib Depending on Empirical Change in Dosing
2.7. Model-Based Dosing Recommendations
2.8. Retrospective Comparison between Empirical and Model-Based Change in Dosing
3. Discussion
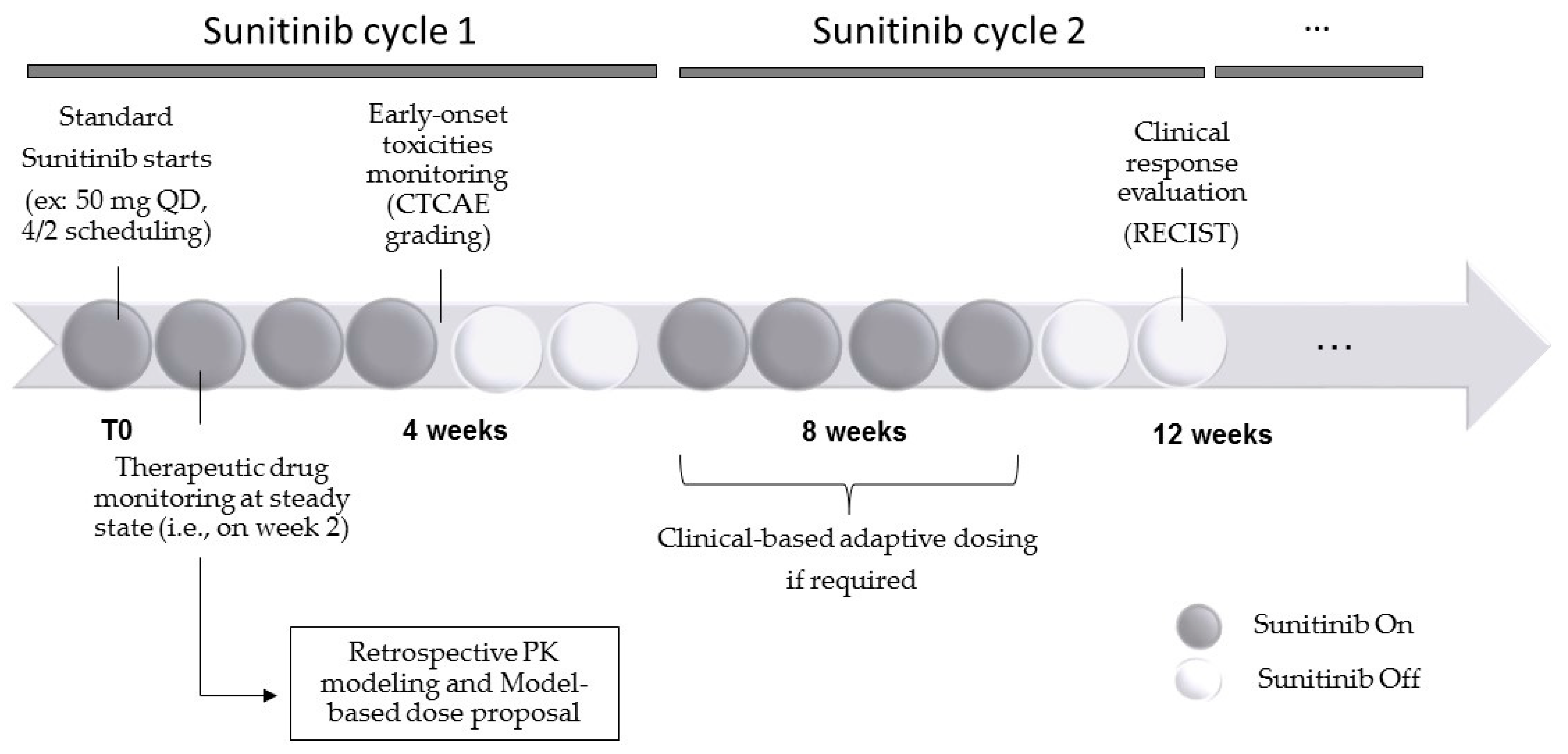
4. Materials and Methods
4.1. Patients
4.2. Measurement of Plasma Concentration of Sunitinib and N-Desethyl Sunitinib
4.3. Sunitinib Dosing and Adaptive Dosing
4.4. Sampling for Therapeutic Drug Monitoring
4.5. PK/PD Modeling
4.6. Clinical Endpoints
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mendel, D.B.; Laird, A.D.; Xin, X.; Louie, S.G.; Christensen, J.G.; Li, G.; Schreck, R.E.; Abrams, T.J.; Ngai, T.J.; Lee, L.B.; et al. In Vivo Antitumor Activity of SU11248, a Novel Tyrosine Kinase Inhibitor Targeting Vascular Endothelial Growth Factor and Platelet-Derived Growth Factor Receptors: Determination of a Pharmacokinetic/Pharmacodynamic Relationship. Clin. Cancer Res. 2003, 9, 327–337. [Google Scholar]
- Motzer, R.J.; Hutson, T.E.; Tomczak, P.; Michaelson, M.D.; Bukowski, R.M.; Rixe, O.; Oudard, S.; Negrier, S.; Szczylik, C.; Kim, S.T.; et al. Sunitinib versus Interferon Alfa in Metastatic Renal-Cell Carcinoma. N. Engl. J. Med. 2007, 356, 115–124. [Google Scholar] [CrossRef]
- Faivre, S.; Delbaldo, C.; Vera, K.; Robert, C.; Lozahic, S.; Lassau, N.; Bello, C.; Deprimo, S.; Brega, N.; Massimini, G.; et al. Safety, Pharmacokinetic, and Antitumor Activity of SU11248, a Novel Oral Multitarget Tyrosine Kinase Inhibitor, in Patients with Cancer. J. Clin. Oncol. 2006, 24, 25–35. [Google Scholar] [CrossRef]
- Motzer, R.J.; Hutson, T.E.; Cella, D.; Reeves, J.; Hawkins, R.; Guo, J.; Nathan, P.; Staehler, M.; de Souza, P.; Merchan, J.R.; et al. Pazopanib versus Sunitinib in Metastatic Renal-Cell Carcinoma. N. Engl. J. Med. 2013, 369, 722–731. [Google Scholar] [CrossRef]
- Takasaki, S.; Kawasaki, Y.; Kikuchi, M.; Tanaka, M.; Suzuka, M.; Noda, A.; Sato, Y.; Yamashita, S.; Mitsuzuka, K.; Saito, H.; et al. Relationships between Sunitinib Plasma Concentration and Clinical Outcomes in Japanese Patients with Metastatic Renal Cell Carcinoma. Int. J. Clin. Oncol. 2018, 23, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.L.; Kim, M.K.; Park, I.; Ahn, J.-H.; Lee, D.H.; Ryoo, H.M.; Song, C.; Hong, B.; Hong, J.H.; Ahn, H. RandomizEd Phase II Trial of Sunitinib Four Weeks on and Two Weeks off versus Two Weeks on and One Week off in Metastatic Clear-Cell Type REnal Cell Carcinoma: RESTORE Trial. Ann. Oncol. 2015, 26, 2300–2305. [Google Scholar] [CrossRef] [PubMed]
- Cabel, L.; Blanchet, B.; Thomas-Schoemann, A.; Huillard, O.; Bellesoeur, A.; Cessot, A.; Giroux, J.; Boudou-Rouquette, P.; Coriat, R.; Vidal, M.; et al. Drug Monitoring of Sunitinib in Patients with Advanced Solid Tumors: A Monocentric Observational French Study. Fundam. Clin. Pharmacol. 2018, 32, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Yeap, S.; Clements, A.; Balakrishnar, B.; Wong, M.; Gurney, H. Evidence for Therapeutic Drug Monitoring of Targeted Anticancer Therapies. J. Clin. Oncol. 2012, 30, 4017–4025. [Google Scholar] [CrossRef]
- Groenland, S.L.; Mathijssen, R.H.J.; Beijnen, J.H.; Huitema, A.D.R.; Steeghs, N. Individualized Dosing of Oral Targeted Therapies in Oncology Is Crucial in the Era of Precision Medicine. Eur. J. Clin. Pharmacol. 2019, 75, 1309–1318. [Google Scholar] [CrossRef]
- Widmer, N.; Bardin, C.; Chatelut, E.; Paci, A.; Beijnen, J.; Levêque, D.; Veal, G.; Astier, A. Review of Therapeutic Drug Monitoring of Anticancer Drugs Part Two--Targeted Therapies. Eur. J. Cancer 2014, 50, 2020–2036. [Google Scholar] [CrossRef]
- Demlová, R.; Turjap, M.; Peš, O.; Kostolanská, K.; Juřica, J. Therapeutic Drug Monitoring of Sunitinib in Gastrointestinal Stromal Tumors and Metastatic Renal Cell Carcinoma in Adults—A Review. Ther. Drug Monit. 2020, 42, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Houk, B.E.; Bello, C.L.; Kang, D.; Amantea, M. A Population Pharmacokinetic Meta-Analysis of Sunitinib Malate (SU11248) and Its Primary Metabolite (SU12662) in Healthy Volunteers and Oncology Patients. Clin. Cancer Res. 2009, 15, 2497–2506. [Google Scholar] [CrossRef] [PubMed]
- Bertolaso, P.; Gross-Goupil, M.; Molimard, M.; Cochin, V.; Ravaud, A.; Daste, A. Drug Interaction With Sunitinib and the Evidence of Therapeutic Drug Monitoring: A Case Report and Review of the Literature. Clin. Genitourin. Cancer 2017, 15, e885–e887. [Google Scholar] [CrossRef]
- Diekstra, M.H.; Fritsch, A.; Kanefendt, F.; Swen, J.J.; Moes, D.; Sörgel, F.; Kinzig, M.; Stelzer, C.; Schindele, D.; Gauler, T.; et al. Population Modeling Integrating Pharmacokinetics, Pharmacodynamics, Pharmacogenetics, and Clinical Outcome in Patients With Sunitinib-Treated Cancer. CPT Pharmacometr. Syst. Pharmacol. 2017, 6, 604–613. [Google Scholar] [CrossRef]
- Schindler, E.; Amantea, M.A.; Karlsson, M.O.; Friberg, L.E. PK-PD Modeling of Individual Lesion FDG-PET Response to Predict Overall Survival in Patients with Sunitinib-Treated Gastrointestinal Stromal Tumor. CPT Pharmacometr. Syst. Pharmacol. 2016, 5, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Narjoz, C.; Cessot, A.; Thomas-Schoemann, A.; Golmard, J.L.; Huillard, O.; Boudou-Rouquette, P.; Behouche, A.; Taieb, F.; Durand, J.P.; Dauphin, A.; et al. Role of the Lean Body Mass and of Pharmacogenetic Variants on the Pharmacokinetics and Pharmacodynamics of Sunitinib in Cancer Patients. Investig. New Drugs 2015, 33, 257–268. [Google Scholar] [CrossRef]
- Gotta, V.; Widmer, N.; Decosterd, L.A.; Chalandon, Y.; Heim, D.; Gregor, M.; Benz, R.; Leoncini-Franscini, L.; Baerlocher, G.M.; Duchosal, M.A.; et al. Clinical Usefulness of Therapeutic Concentration Monitoring for Imatinib Dosage Individualization: Results from a Randomized Controlled Trial. Cancer Chemother. Pharmacol. 2014, 74, 1307–1319. [Google Scholar] [CrossRef]
- Bardin, C.; Veal, G.; Paci, A.; Chatelut, E.; Astier, A.; Levêque, D.; Widmer, N.; Beijnen, J. Therapeutic Drug Monitoring in Cancer--Are We Missing a Trick? Eur. J. Cancer 2014, 50, 2005–2009. [Google Scholar] [CrossRef]
- Mueller-Schoell, A.; Groenland, S.L.; Scherf-Clavel, O.; van Dyk, M.; Huisinga, W.; Michelet, R.; Jaehde, U.; Steeghs, N.; Huitema, A.D.R.; Kloft, C. Therapeutic Drug Monitoring of Oral Targeted Antineoplastic Drugs. Eur. J. Clin. Pharmacol. 2021, 77, 441–464. [Google Scholar] [CrossRef] [PubMed]
- Westerdijk, K.; Desar, I.M.E.; Steeghs, N.; van der Graaf, W.T.A.; van Erp, N.P.; Dutch Pharmacology and Oncology Group (DPOG). Imatinib, Sunitinib and Pazopanib: From Flat-Fixed Dosing towards a Pharmacokinetically Guided Personalized Dose. Br. J. Clin. Pharmacol. 2020, 86, 258–273. [Google Scholar] [CrossRef]
- Noda, S.; Otsuji, T.; Baba, M.; Yoshida, T.; Kageyama, S.; Okamoto, K.; Okada, Y.; Kawauchi, A.; Onishi, H.; Hira, D.; et al. Assessment of Sunitinib-Induced Toxicities and Clinical Outcomes Based on Therapeutic Drug Monitoring of Sunitinib for Patients With Renal Cell Carcinoma. Clin. Genitourin. Cancer 2015, 13, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Guideline-Bioanalytical-Method-Validation_en.Pdf. Available online: https://www.ema.europa.eu/en/bioanalytical-method-validation (accessed on 1 October 2020).
- Lavielle, M.; Ribba, B. Enhanced Method for Diagnosing Pharmacometric Models: Random Sampling from Conditional Distributions. Pharm. Res. 2016, 33, 2979–2988. [Google Scholar] [CrossRef] [PubMed]

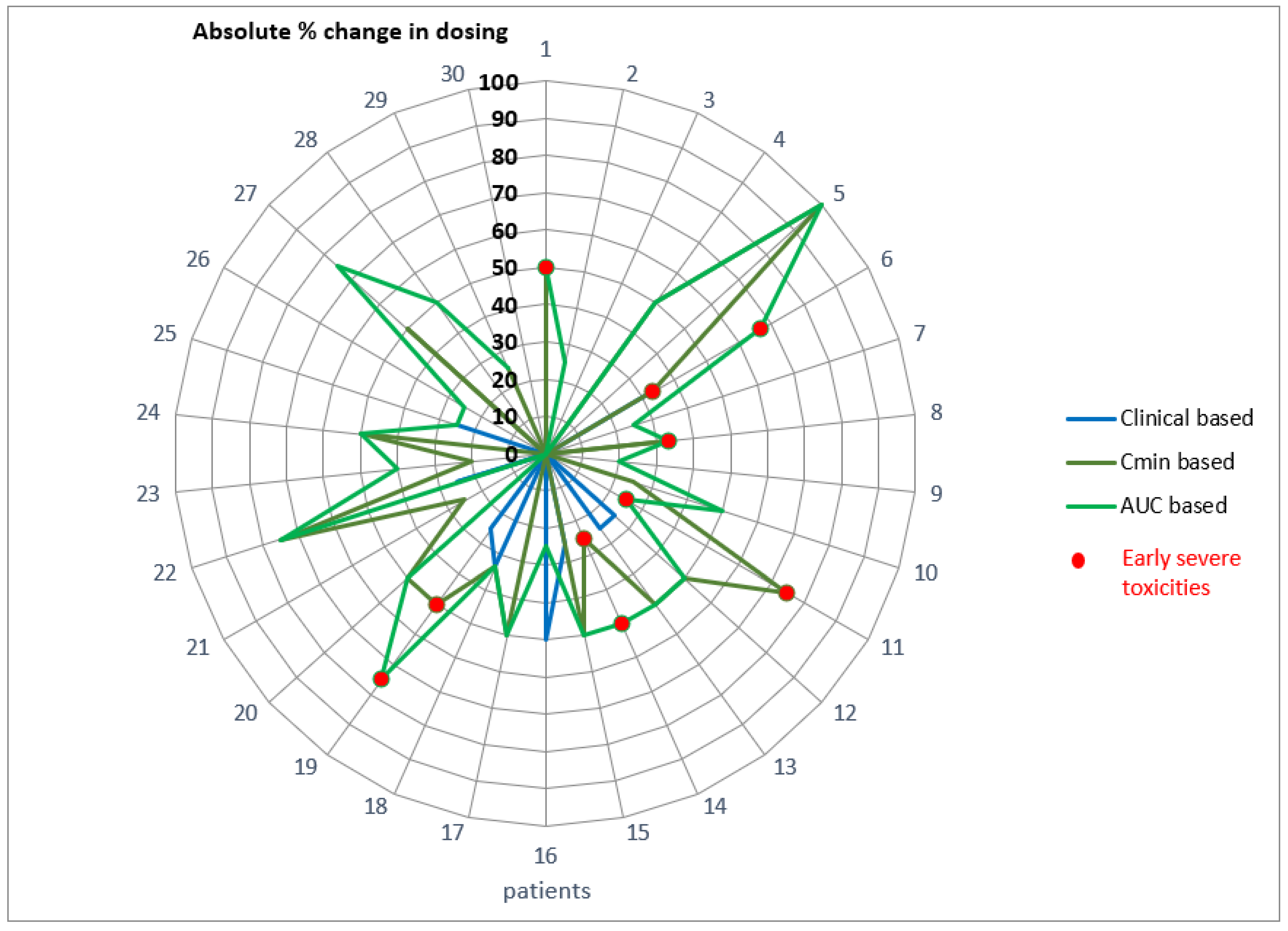
| No. (%) | |
|---|---|
| Characteristic | Patients (n = 31) |
| Age, years | |
| Median | 62 |
| Range | 26–87 |
| Sex | |
| Male | 26 (83.9) |
| Female | 5 (16.1) |
| Body weight (kg) | |
| Median | 78 |
| Range | 46–160 |
| BSA (m2) | |
| Mean | 1.9 |
| Standard deviation | ±0.2 |
| BMI (kg/m2) | |
| Mean | 25.6 |
| Standard deviation | ±7.2 |
| Histology | |
| Clear cell | 31 (100) |
| Metastasis | 27 (93.1) |
| Initial dosing | |
| 62.5 mg QD | 2 (6.4) |
| 50 mg QD | 25 (80.7) |
| 37.5 mg QD | 3 (9.7) |
| 25 mg QD | 0 (0) |
| 12.5 mg QD | 1 (3.2) |
| Schedule | |
| 4 weeks ON/2 weeks OFF | 29 (93.6) |
| 2 weeks ON/2 weeks OFF | 1 (3.2) |
| Other | 1 (3.2) |
| Patients (n = 27) | |||
|---|---|---|---|
| n (%) | |||
| Adverse Event | No Toxicity | Grade 1/2 | Grade 3 |
| Diarrhea | 2 (7.4) | 20 (74.1) | 5 (18.5) |
| Arterial hypertension | 8 (29.6) | 15 (55.6) | 4 (14.8) |
| Skin toxicity | 11 (40.7) | 14 (51.9) | 2 (7.4) |
| Headache, anosmia | 18 (66.7) | 9 (33.3) | 0 (0) |
| Fatigue | 8 (29.6) | 17 (63) | 2 (7.4) |
| Neutropenia | 24 (89.9) | 2 (7.4) | 1 (3.7) |
| A | Adequate Trough concentrations (50–100 ng/mL) | Abnormal trough concentrations with subsequent change in dosing | Abnormal trough concentrations without subsequent change in dosing |
| n | 9 | 8 | 6 |
| Clinical benefit n (%) | 7 (77.8) | 5 (62.5) | 1 (16.7) |
| B | Adequate AUC (1250–2150 ng/mL.h) | Abnormal AUC with subsequent change in dosing | Abnormal AUC without subsequent change in dosing |
| n | 4 | 11 | 8 |
| Clinical benefit n (%) | 3 (75) | 8 (72.7) | 1 (12.5) |
 : no change,
: no change,  : decrease,
: decrease,  : increase.
: increase.
 : no change,
: no change,  : decrease,
: decrease,  : increase.
: increase.| Early-Onset Toxicities (n = 17 Patients) | |||
|---|---|---|---|
| Initial Dosing (mg) | Clinical-Based Dosing (% Change) | Cmin-Based Dosing (% Change) | AUC-Based Dosing (% Change) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50) 25 (−50) |
| 37.5 |  37.5 (0%) 37.5 (0%) |  25 (−33%) 25 (−33%) |  25 (−33%) 25 (−33%) |
| 62.5 |  50 (−20%) 50 (−20%) |  62.5 (0%) 62.5 (0%) |  50 (−20%) 50 (−20%) |
| 50 |  50 (0%) 50 (0%) |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |
| 50 |  50 (0%) 50 (0%) |  87.5 (+75%) 87.5 (+75%) |  62.5 (+25%) 62.5 (+25%) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  50 (0%) 50 (0%) |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  12.5 (−75%) 12.5 (−75%) |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| 50 |  50 (0%) 50 (0%) |  62.5 (+25%) 62.5 (+25%) |  50 (0%) 50 (0%) |
| 50 |  37.5 (−25%) 37.5 (−25%) |  12.5 (−75%) 12.5 (−75%) |  12.5 (−75%) 12.5 (−75%) |
| 62.5 |  62.5 (0%) 62.5 (0%) |  50 (−20%) 50 (−20%) |  37.5 (−40%) 37.5 (−40%) |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) |
| Initial Dosing | Trough Levels (Target: 50–100 ng/mL) | AUC (Target: 1250–2150 ng/mL.h) | Model-Based Recommendation | Clinical-Based Recommendation | Clinical Outcome | Early-Onset Toxicities |
|---|---|---|---|---|---|---|
| 50 mg | 13 | 491 | 100 mg | 50 mg | Progressive Disease | No |
| 50 mg | 43 | 1264 | 87.5 mg | 50 mg | Clinical Benefit | No |
 : no change,
: no change,  : decrease,
: decrease,  : increase.
: increase.
 : no change,
: no change,  : decrease,
: decrease,  : increase.
: increase.| Patients with Progressive Disease (n = 11) | ||||
|---|---|---|---|---|
| Initial Dosing (mg) | Clinical-Based Dosing (mg) | Cmin-Based Dosing (mg) | AUC-Based Dosing (mg) | Early Severe Toxicities |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) | yes |
| 50 |  50 (0%) 50 (0%) |  50 (0%) 50 (0%) |  37.5 (−25%) 37.5 (−25%) | no |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) | yes |
| 50 |  50 (0%) 50 (0%) |  100 (+100%) 100 (+100%) |  100 (+100%) 100 (+100%) | no |
| 62.5 |  50 (−20%) 50 (−20%) |  62.5 (0%) 62.5 (0%) |  50 (−20%) 50 (−20%) | yes |
| 50 |  37.5 (−25%) 37.5 (−25%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) | yes |
| 50 |  25 (−50%) 25 (−50%) |  50 (0%) 50 (0%) |  37.5 (−25%) 37.5 (−25%) | no |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) | no |
| 50 |  50 (0%) 50 (0%) |  25 (−50%) 25 (−50%) |  25 (−50%) 25 (−50%) | yes |
| 50 |  37.5 (−25%) 37.5 (−25%) |  12.5 (−75%) 12.5 (−75%) |  12.5 (−75%) 12.5 (−75%) | yes |
| 62.5 |  62.5 (0%) 62.5 (0%) |  50 (−20%) 50 (−20%) |  37.5 (−40%) 37.5 (−40%) | yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrer, F.; Chauvin, J.; DeVictor, B.; Lacarelle, B.; Deville, J.-L.; Ciccolini, J. Clinical-Based vs. Model-Based Adaptive Dosing Strategy: Retrospective Comparison in Real-World mRCC Patients Treated with Sunitinib. Pharmaceuticals 2021, 14, 494. https://doi.org/10.3390/ph14060494
Ferrer F, Chauvin J, DeVictor B, Lacarelle B, Deville J-L, Ciccolini J. Clinical-Based vs. Model-Based Adaptive Dosing Strategy: Retrospective Comparison in Real-World mRCC Patients Treated with Sunitinib. Pharmaceuticals. 2021; 14(6):494. https://doi.org/10.3390/ph14060494
Chicago/Turabian StyleFerrer, Florent, Jonathan Chauvin, Bénédicte DeVictor, Bruno Lacarelle, Jean-Laurent Deville, and Joseph Ciccolini. 2021. "Clinical-Based vs. Model-Based Adaptive Dosing Strategy: Retrospective Comparison in Real-World mRCC Patients Treated with Sunitinib" Pharmaceuticals 14, no. 6: 494. https://doi.org/10.3390/ph14060494
APA StyleFerrer, F., Chauvin, J., DeVictor, B., Lacarelle, B., Deville, J.-L., & Ciccolini, J. (2021). Clinical-Based vs. Model-Based Adaptive Dosing Strategy: Retrospective Comparison in Real-World mRCC Patients Treated with Sunitinib. Pharmaceuticals, 14(6), 494. https://doi.org/10.3390/ph14060494







