Role of Citicoline in the Management of Traumatic Brain Injury
Abstract
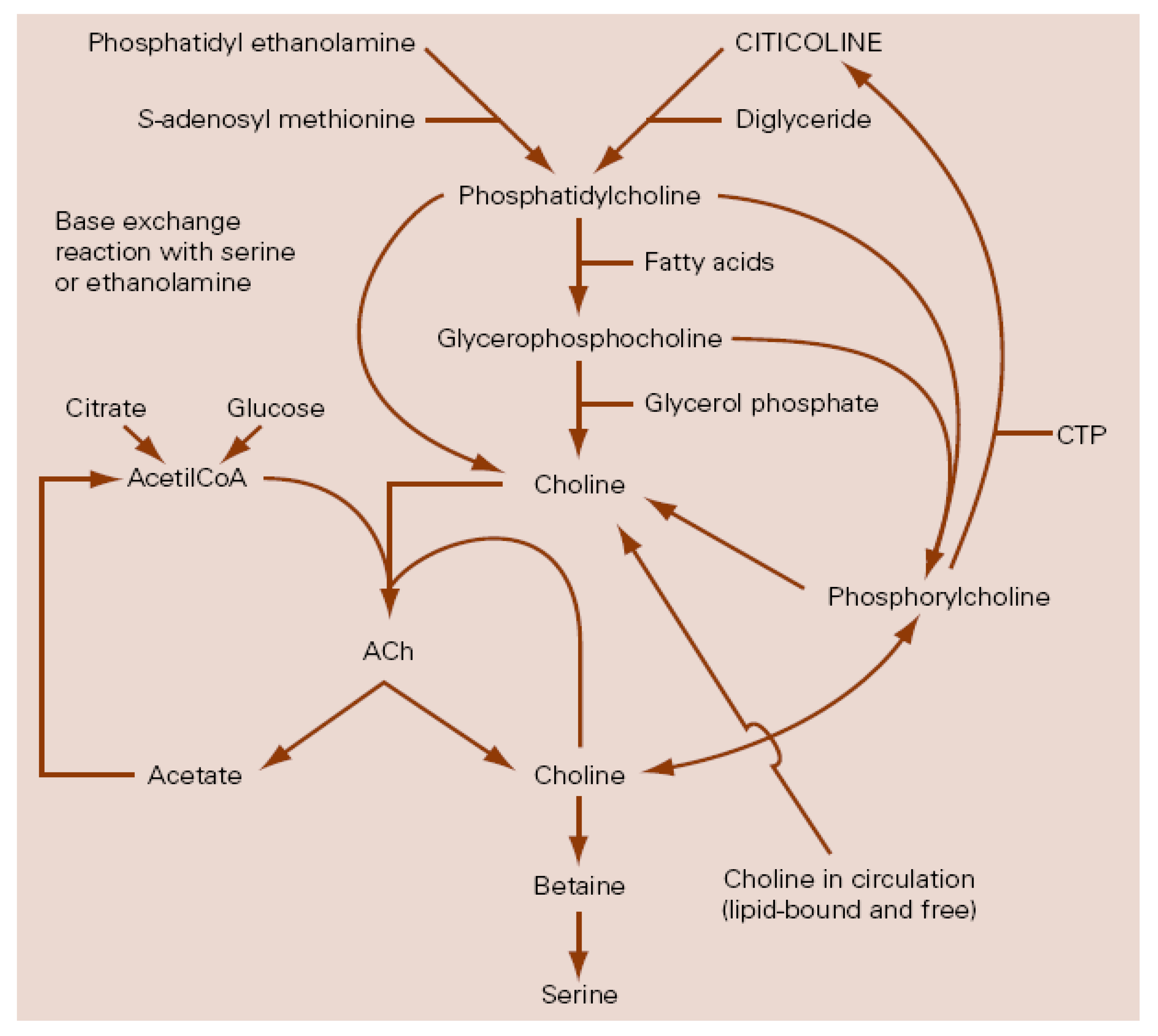
1. Introduction
2. Experimental Data
| Author | Year | Study Design | Experimental Model | Insult | Dose | Main Results |
|---|---|---|---|---|---|---|
| Tsuchida et al. [59] | 1967 | Comparative study | Rats | Cold injury | 100µc 3H-CDP-choline IP | Significant incorporation of the labelled drug in the affected areas of the brain |
| Kondo [58] | 1968 | Comparative study | Male cats (2.5–4 Kg) | Epidural compression | 15–20 mg/Kg intracarotid | Significant increase of survival rates |
| Boismare et al. [60] | 1977 | Comparative study | Rats | Whiplash injury | 20 mg/Kg IP | Significant prevention on catecholamines changes in brain and suppression of behavioral disorders |
| Cohadon F. et al. [62] | 1979 | Comparative study | Rabbits (~2.5 Kg) | Cryogenic lesion | 20 mg/Kg/4 d IV (starting 24 h after injury) | Significant restoration of the activity of the mitochondrial ATPase and of the membranous Na+/K+-ATPase. Acceleration of the reabsorption of brain edema. |
| Roda J.E. [66] | 1980 | Comparative study | Wistar Rats | Cryogenic lesion | 6 mg/Kg/12 h IP Starting 24 h before the lesion and continued until sacrifice | Significant reduction of the extravasation of blue Evans |
| Cats | Cryogenic lesion | 15 mg/Kg/12 h IP Starting 24 h before the lesion or 2 h after the lesion and continued until sacrifice | Significant reduction of the extravasation of blue Evans. Better results when administered before the lesion | |||
| Algate et al. [56] | 1983 | Comparative study | Male cats (2.65–3.65 Kg) | Epidural compression | 0.5 g/Kg/5 d PO | Significant increase in resistance to effects of mechanical compression |
| Ogashiwa M. et al. [48] | 1985 | Comparative randomized study | Mice | Mechanical impact | 1–250 mg/Kg IV | Significant dose-effect on duration of coma |
| Kitazaki T. et al. [54] | 1985 | Comparative study | Rats | N/A a | 1–10 mM | Dose-dependent inhibition of activity of PLA2 b |
| Clendenon et al. [61] | 1985 | Comparative study | Mongrel dogs (8–12 Kg) | Impact injury at spinal cord | 100–200 mg/Kg IV | Prevention of the decrease of Mg2+-dependent ATPase activity |
| Lafuente J.V. et al. [63]. | 1986 | Comparative randomized study | Male cats | Ultraviolet-induced brain edema | 20 mg/Kg IV | Significant acceleration of the reabsorption of brain edema |
| Majem X. et al. [65] | 1986 | Comparative study | Male rats (180–200 g) | Cryogenic lesion | 1 g/Kg/8 d PO | Significant increase of theta activity and decrease of delta activity on EEG |
| Arrigoni E. et al. [51] | 1987 | Comparative randomized study | Female rabbits (2.0–2.5 Kg) | Cryogenic lesion | 200 mg/Kg PO | Dose-dependent complete inhibition of PLA2 b activation |
| Cervós-Navarro J. et al. [64] | 1990 | Comparative randomized study | Mongrel cats (2.5–4.5 Kg) | Ultraviolet-induced brain edema | 100 mg/Kg IV (3 doses in 24 h) | Significant acceleration of the reabsorption of brain edema |
| Dixon C.E. et al. [69] | 1997 | Comparative study | Adult male Sprague Dawley rats (250–275 g) | Cortical impact injury | 100 mg/Kg/18 d | Significant reductions on cognitive deficits and increase of extracellular acetylcholine levels |
| Plataras C. et al. [70] | 2000 | Comparative study | Albino Wistar rats | Incubation of homogenates of whole brain | 0.1–1 mM | Stimulation of brain acetylcholinesterase and Na+/K+-ATPase |
| Başkaya M.K. at al. [71] | 2000 | Comparative study | Sprague Dawley rats (250–300 g) | Controlled cortical impact | 50–400 mg/Kg IP 2 times after injury | Significant dose-dependent reduction of brain edema and blood−brain barrier disruption |
| Dempsey R.J. et al. [72] | 2003 | Comparative study | Adult male Sprague Dawley rats (250–280 g) | Controlled cortical impact | 100–400 mg/Kg IP | Significant decrease of hippocampal neuronal death, cortical contusion volume, and neurological dysfunction |
| Menku A. et al. [73] | 2010 | Comparative study | Male Swiss albino rats (200–250 g) | Blunt trauma | 250 mg/Kg IP | Significant reduction of Malonyldialdehyde levels with citicoline alone or in combination with propofol |
| Qian K. et al. [75] | 2014 | Comparative randomized study | Adult male Sprague Dawley rats (290–330 g) | 250 mg/Kg IV 30 min and 4 h after | Marked reduction of brain edema and blood−brain barrier permeability, enhancement of the activities of superoxide dismutase and the levels of glutathione, reduction of the levels of malondialdehydeand lactic acid. Reduction of axonal damage and neuronal death | |
| Schmidt K. et al. [67] | 2015 | Comparative randomized study | Male Wistar rats | Endotoxemia induced by Lipopolysaccharide injection | 100 mg/Kg IV | Significant reduction of microvascular permeability |
| Hernekamp J.F. et al. [68] | 2015 | Comparative randomized study | Adult male Wistar rats (250–300 g) | Burn edema | 100 mg/Kg IV | Significant reduction of macromolecular efflux and reduction of leukocyte activation |
| Jacotte-Simancas A. et al. [74] | 2015 | Comparative randomized study | Male Sprague Dawley albino rats (~250 g) | Controlled cortical impact injury | 200 mg/Kg IP starting 4 h after surgery, and thereafter daily until completing five injections | Significant improvement of memory deficits and reduction of interhemispheric differences in the volume of the hippocampal formation |
| Gan D, et al., [76] | 2020 | Comparative randomized study | Zebrafish larvae | In vivo TBI zebrafish model | 2.5 mg/mL for the drug incubation | Activation of microglia, reduction of neuronal apoptosis and promotion of neuronal proliferation |
3. Clinical Experiences of Patients with Traumatic Brain Injuries
3.1. Clinical Studies on Mild, Complicated to Severe Head Injuries
| Authors | Year | n | Severity | Type of Study | Control | Time Window | Schedule of Treatment | Follow-up | Main Results |
|---|---|---|---|---|---|---|---|---|---|
| Misbach et al. [87] | 1978 | 80 | Moderate to severe | Double blind RCT a | Placebo | NA b | 300 mg/d/14 d IV | 14 d | Better recovery rate (GCS c) |
| Espagno et al. [90] | 1979 | 46 | Severe | Double blind RCT a | Placebo | NA b | 250 mg/d/5 d IV + 250 mg/d/15 d IM | 30 d | Better recovery of consciousness |
| Carcasonne & LeTourneau [91] | 1979 | 43 | Moderate to severe (children) | Double blind RCT a | Placebo | NA b | NAb | 20 d | Faster recovery from coma |
| Richer & Cohadon [92] | 1980 | 60 | Severe | Double blind RCT a | Placebo | 24 h | 750 mg/d IV (6 d) + IM (14 d) | 90 d | More independent patients (clinical evaluation) |
| Lecuire & Duplay [93] | 1982 | 25 | Moderate to severe | Double blind RCT a | Meclophenoxate | 24 h | 750 mg/d/10 d IV | 10 d | More patients with a favorable outcome |
| Lecuire & Duplay [94] | 1982 | 154 | Moderate to severe | Open study | Bibliographic data | 24 h | 750 mg/d/10 d IV + 250 mg/d/10 d IM | 20 d | Significant improvement of survival and resolution of neurological deficits and consciousness troubles |
| Cohadon et al. [96] | 1982 | 60 | Severe | Double blind RCT a | Placebo | 24 h | 750 mg/d IV (6 d) + IM (20 d) | 120 d | More independent patients (~GOS d) |
| Lecuire [95] | 1985 | 40 | Moderate to severe | Double blind RCT a | Piracetam | 24 h | 750 mg/d/10 d IV | 10 d | Global result favorable to citicoline (p < 0.01) |
| Deleuze et al. [97] | 1985 | 11 | Severe | Open study | None | 24 h | 500 md IV single dose | 4 d | Significant decrease of lactate and lactate/pyruvate ratio in CSF e |
| De Blas et al. [100] | 1986 | 100 | Moderate to severe | Open RCT a | Control | 24 h | 200–400 mg/8 h IV or IM in the acute phase, followed by 100–200 mg/8 h PO during follow-up | 180 d | Reduction of coma and neurological and psychological sequelae |
| Ragguenneau & Jarrige [101] | 1988 | 921 | Severe | Cohort study | Control | 24 h | 500–750 mg/d/20 d IV | 180 d | More independent patients (~GOS d) |
| Calatayud Maldonado et al. [102] | 1991 | 216 | Moderate to severe | Single blind RCT a | Control | 24 h | 3–4 g/d/4 d IV + 2 g/d/26 d PO | 90 d | More independent patients (GOS d) Decreased hospital stay |
| Lozano [103] | 1991 | 78 | Severe | Single blind RCT a | Control | 24 h | 3–6 g/d/14 d IV | 90 d | Trend to have more independent patients (GOS d) Reduction of brain edema (CTscan) Decreased hospital stay |
| Lazowski et al. [104] | 2003 | 28 | RCT a | Placebo | NA b | NA b | 30 d | GCS 21 is significantly correlated with GOS 30 (r = 0.68; p < 0.01) showing the protective effect of citicoline | |
| Hinev et al. [105] | 2007 | 8 | Severe | Open | None | 36 h | 1 g/d/5–7 d IV | NA b | 80% of patients recovered from neurological symptoms and un-consciousness |
| Krishna et al. [106] | 2012 | 100 | Moderate to severe | Single blind RCT a | Placebo | 24 h | 2 g/d/60 d PO | 90 d | Earlier rate of recovery, less duration of stay, early gaining of full consciousness and relief from cognitive symptoms |
| Zafonte et al. [108] | 2012 | 1213 | Mild, complicated, moderate and severe | Double blind RCT a | Placebo | 24 h | 2 g/d/90 d PO or enteral | 180 d | No differences on the TBI-Clinical Trials Network Core Battery |
| El Reweny et al. [109] | 2012 | 40 | Severe | Open RCT a | Control | NA b | 1 g/d/14 d IV | NA b | Trend to improve the outcome |
| Salehpour et al. [114] | 2013 | 40 | Severe with diffuse axonal injury | Single blind RCT a | Control | 24 h | 2 g/d/12 d IV | 12 d | Reduction of MDA plasma levels |
| Shokouhi et al. [116] | 2014 | 58 | Severe with diffuse axonal injury | Double blind RCT a | Control | 24 h | 2 g/d/15 d IV | 15 d | Increased plasma levels of fetuin-A and matrix Gla-protein |
| Salehpour et al. [115] | 2015 | 40 | Severe with diffuse axonal injury | Single blind RCT a | Control | 24 h | 2 g/d/15 d IV | 15 d | Reduction of MDA plasma levels No differences on GCS c |
| Varadaraju et al. [110] | 2017 | 60 | Mild to moderate | Open RCT a | Citicoline + Cerebrolysin | NAb | 2 g stat followed by 500 mg IV/PO twice daily continued for 3 months. | 180 d | The association had better outcome (GOS) than patients treated with citicoline alone |
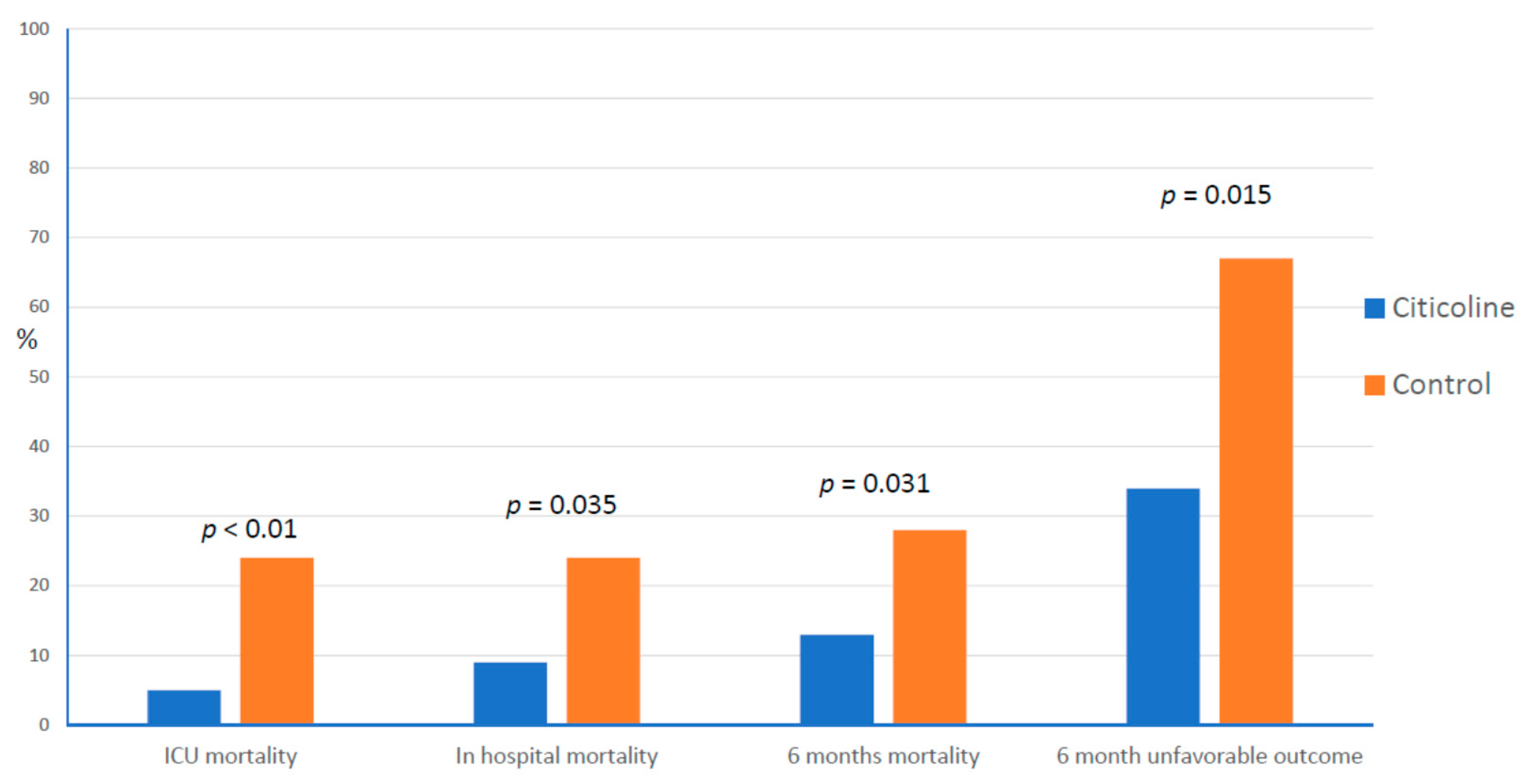
| Trimmel et al. [112] | 2018 | 134 | Moderate to severe | Retrospective matched pair analysis | Control | 24–48 h | 3 g/d/21 d IV | 180 d | Reduction of the rates of mortality Reduction of the rates of unfavorable outcome (GOS) |
| Ahmadi et al. [113] | 2020 | 30 | Severe | Double blind RCT a | Control | NA b | 1–3 g/d/14 d IV | 30 d | According to protocol: significant dose-dependent effect on outcome (GOS) |
| According to article: no positive effect |
3.2. Clinical Studies on Mild Head Injuries
3.3. Clinical Studies on Cognitive Disorders Associated to TBI
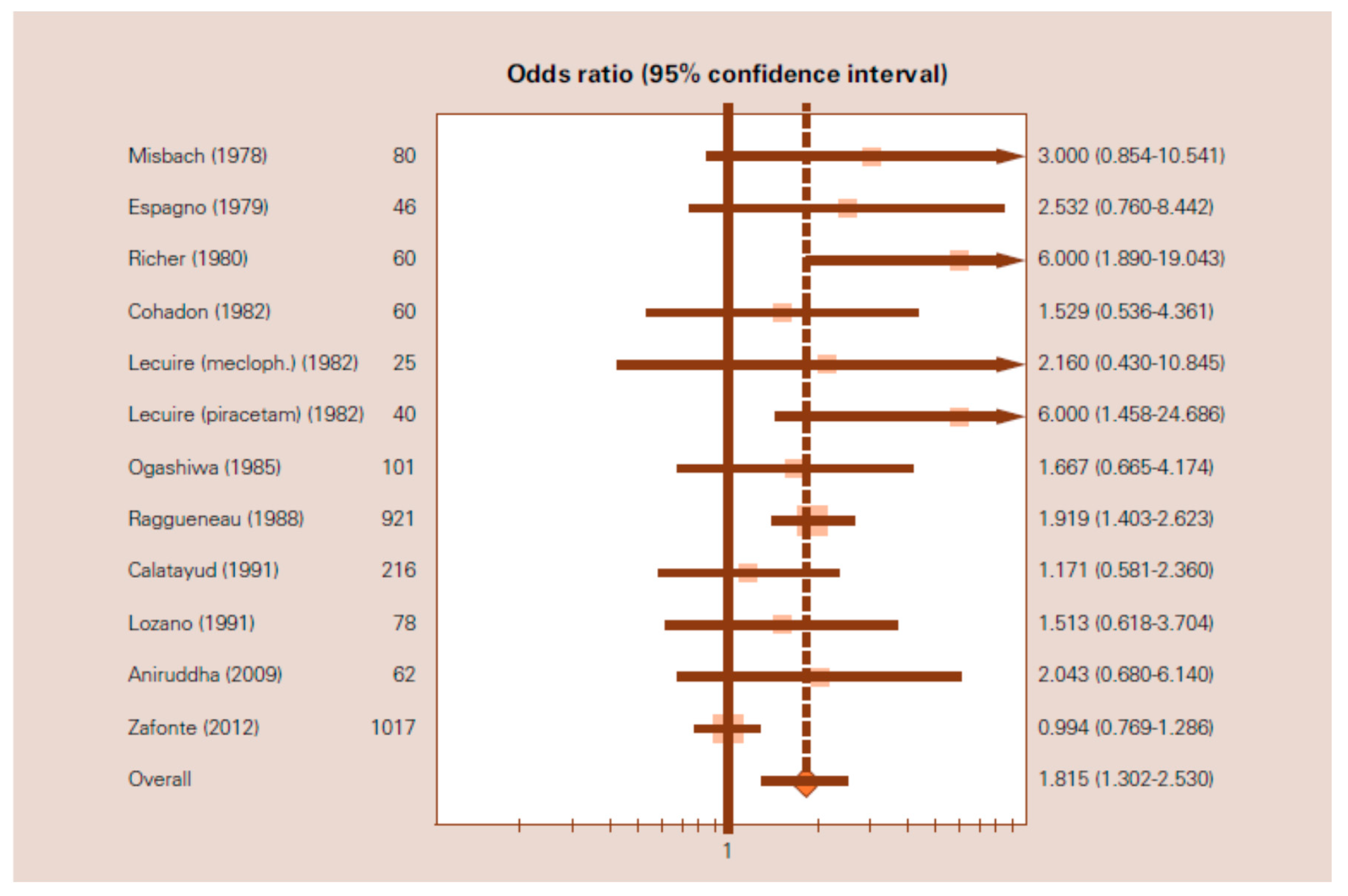
3.4. Meta-Analysis on the Effects of Citicoline in the Management of TBI
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators. Global, regional, and national burden of traumatic brain injury and spinal cord injury, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 56–87. [Google Scholar] [CrossRef]
- James, S.L.; Lucchesi, L.R.; Bisignano, C.; Castle, C.D.; Dingels, Z.V.; Fox, J.T.; Hamilton, E.B.; Liu, Z.; McCracken, D.; Nixon, M.R.; et al. Morbidity and mortality from road injuries: Results from the Global Burden of Disease Study 2017. Inj. Prev. 2020, 26 (Suppl. 1), i46–i56. [Google Scholar] [CrossRef] [PubMed]
- Bogoslovsky, T.; Wilson, D.; Chen, Y.; Hanlon, D.; Gill, J.; Jeromin, A.; Song, L.; Moore, C.; Gong, Y.; Kenney, K.; et al. Increases of Plasma Levels of Glial Fibrillary Acidic Protein, Tau, and Amyloid β up to 90 Days after Traumatic Brain Injury. J. Neurotrauma. 2017, 34, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Stocchetti, N.; Carbonara, M.; Citerio, G.; Ercole, A.; Skrifvars, M.B.; Smielewski, P.; Zoerle, T.; Menon, D.K. Severe traumatic brain injury: Targeted management in the intensive care unit. Lancet Neurol. 2017, 16, 452–464. [Google Scholar] [CrossRef]
- Khellaf, A.; Khan, D.Z.; Helmy, A. Recent advances in traumatic brain injury. J. Neurol. 2019, 266, 2878–2889. [Google Scholar] [CrossRef]
- Capizzi, A.; Woo, J.; Verduzco-Gutierrez, M. Traumatic Brain Injury: An Overview of Epidemiology, Pathophysiology, and Medical Management. Med. Clin. N. Am. 2020, 104, 213–238. [Google Scholar] [CrossRef]
- Vella, M.A.; Crandall, M.L.; Patel, M.B. Acute Management of Traumatic Brain Injury. Surg. Clin. N. Am. 2017, 97, 1015–1030. [Google Scholar] [CrossRef]
- Ng, S.Y.; Lee, A.Y.W. Traumatic Brain Injuries: Pathophysiology and Potential Therapeutic Targets. Front. Cell. Neurosci. 2019, 13, 528. [Google Scholar] [CrossRef]
- Stocker, R.A. Intensive Care in Traumatic Brain Injury Including Multi-Modal Monitoring and Neuroprotection. Med. Sci. 2019, 7, 37. [Google Scholar] [CrossRef]
- Hosomi, S.; Ohnishi, M.; Ogura, H.; Shimazu, T. Traumatic brain injury-related inflammatory projection: Beyond local inflammatory responses. Acute Med. Surg. 2020, 7, e520. [Google Scholar] [CrossRef]
- Jha, R.M.; Kochanek, P.M.; Simard, J.M. Pathophysiology and treatment of cerebral edema in traumatic brain injury. Neuropharmacology 2019, 145 Pt B, 230–246. [Google Scholar] [CrossRef]
- Jarrahi, A.; Braun, M.; Ahluwalia, M.; Gupta, R.V.; Wilson, M.; Munie, S.; Ahluwalia, P.; Vender, J.R.; Vale, F.L.; Dhandapani, K.M.; et al. Revisiting Traumatic Brain Injury: From Molecular Mechanisms to Therapeutic Interventions. Biomedicines 2020, 8, 389. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Skolnick, B.; Narayan, R.K. Neuroprotection Trials in Traumatic Brain Injury. Curr. Neurol. Neurosci. Rep. 2016, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Mallah, K.; Couch, C.; Borucki, D.M.; Toutonji, A.; Alshareef, M.; Tomlinson, S. Anti-inflammatory and Neuroprotective Agents in Clinical Trials for CNS Disease and Injury: Where Do We Go from Here? Front. Immunol. 2020, 11, 2021. [Google Scholar] [CrossRef]
- Sakai, S.; Shichita, T. Inflammation and neural repair after ischemic brain injury. Neurochem. Int. 2019, 130, 104316. [Google Scholar] [CrossRef]
- Crupi, R.; Cordaro, M.; Cuzzocrea, S.; Impellizzeri, D. Management of Traumatic Brain Injury: From Present to Future. Antioxidants 2020, 9, 297. [Google Scholar] [CrossRef]
- Boismare, F. Souffrance cérébrale: Comportement et neurotransmetteurs sur des modeles expérimentaux. In Symposium Internacional Souffrance Cérébrale et Précurseurs des Phospholipides; Laboratoires Cassenne-Takeda: Paris, France, 1980. [Google Scholar]
- Cohadon, F.; Rigoulet, M.; Averet, M.; Arrigoni, E. Membrane damage in acute brain trauma. Nouv. Presse Med. 1989, 10, 147–155. [Google Scholar] [CrossRef]
- Cohadon, F.; Rigoulet, M.; Guérin, B.; Vandendriessche, M. L’activité membranaire dans la souffrance cérébrale. Altérations des ATPases membranaires dans l’edème cérébral vasogénique. Restauration par un précurseur des phospholipides. In Symposium Internacional Souffrance Cérébrale et Précurseurs des Phospholipides; Laboratoires Cassenne-Takeda: Paris, France, 1980. [Google Scholar]
- Cohadon, F. Physiopathologie des edèmes cérébraux. Rev. Neurol. 1987, 143, 3–20. [Google Scholar]
- Rigoulet, M.; Guérin, B.; Cohadon, F.; Vandendriessche, M. Unilateral brain injury in the rabbit; reversible and irreversible damage of the membranal ATPases. J. Neurochem. 1979, 32, 535–541. [Google Scholar] [CrossRef]
- Secades, J.J.; Lozano, R. Traumatismos Craneoencefálicos: Revisión Fisiopatológica y Terapéutica. Aportaciones de la Citicolina; Excerpta Medica: Amsterdam, The Netherlands, 1991. [Google Scholar]
- Homayoun, P.; Parkins, N.E.; Soblosky, J.; Carey, M.E.; Rodriguez de Turco, E.B.; Bazan, N.G. Cortical impact injury in rats promotes a rapid and sustained increase in polyunsaturated free fatty acids and diacylglycerols. Neurochem. Res. 2000, 25, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Alberghina, M.; Giuffrida, A.M. Effect of hypoxia on the incorporation of [2-3H] glycerol and [1-14C]-palmitate into lipids of various brain regions. J. Neurosci. Res. 1981, 6, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, W.J.; Dorman, R.V.; Horrocks, L.A. Effects of ischemia and diglycerides on ethanolamine and choline phosphotransferase activities from rat brain. Neurochem. Pathol. 1983, 1, 225–234. [Google Scholar]
- Goldberg, W.J.; Dorman, R.V.; Dabrowiecki, Z.; Horrocks, L.A. The effects of ischemia and CDPamines on Na+,K+-ATPase and acetylcholinesterase activities in rat brain. Neurochem. Pathol. 1985, 3, 237–248. [Google Scholar]
- Han, M.; Sun, T.; Chen, H.; Han, M.; Wang, D. Potential sphingosine-1-phosphate-related therapeutic targets in the treatment of cerebral ischemia reperfusion injury. Life Sci. 2020, 249, 117542. [Google Scholar] [CrossRef]
- Solovieva, E.Y.; Farrahova, K.I.; Karneev, A.N.; Chipova, D.T. Phospholipids metabolism disorders in acute stroke. Zh. Nevrol. Psikhiatr. Im. S. S. Korsakova. 2016, 116, 104–112. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L.; Sun, W.; Pei, L.L.; Tian, M.; Liang, J.; Liu, X.; Zhang, R.; Fang, H.; Wu, J.; et al. Changes of Metabolites in Acute Ischemic Stroke and Its Subtypes. Front. Neurosci. 2021, 14, 580929. [Google Scholar] [CrossRef]
- Bazan, N.G. Docosanoids and elovanoids from omega-3 fatty acids are pro-homeostatic modulators of inflammatory responses, cell damage and neuroprotection. Mol. Aspects Med. 2018, 64, 18–33. [Google Scholar] [CrossRef]
- Tayebati, S.K.; Marucci, G.; Santinelli, C.; Buccioni, M.; Amenta, F. Choline-Containing Phospholipids: Structure-Activity Relationships Versus Therapeutic Applications. Curr. Med. Chem. 2015, 22, 4328–4340. [Google Scholar] [CrossRef] [PubMed]
- Chomova, M.; Zitnanova, I. Look into brain energy crisis and membrane pathophysiology in ischemia and reperfusion. Stress 2016, 19, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.Y.; Miao, H.; Cheng, X.L.; Wei, F. Lipidomics: Novel insight into the biochemical mechanism of lipid metabolism and dysregulation-associated disease. Chem. Biol. Interact. 2015, 240, 220–238. [Google Scholar] [CrossRef]
- Bogie, J.F.J.; Haidar, M.; Kooij, G.; Hendriks, J.J.A. Fatty acid metabolism in the progression and resolution of CNS disorders. Adv. Drug Deliv. Rev. 2020, 159, 198–213. [Google Scholar] [CrossRef]
- Espinós, C.; Galindo, M.I.; García-Gimeno, M.A.; Ibáñez-Cabellos, J.S.; Martínez-Rubio, D.; Millán, J.M.; Rodrigo, R.; Sanz, P.; Seco-Cervera, M.; Sevilla, T.; et al. Oxidative Stress, a Crossroad between Rare Diseases and Neurodegeneration. Antioxidants 2020, 9, 313. [Google Scholar] [CrossRef]
- Mizuma, A.; Yenari, M.A. Clinical perspectives on ischemic stroke. Exp. Neurol. 2021, 338, 113599. [Google Scholar] [CrossRef] [PubMed]
- Zweifler, R.M. Membrane stabilizer: Citicoline. Curr. Med. Res. Opin. 2002, 18 (Suppl. 2), S14–S17. [Google Scholar] [CrossRef] [PubMed]
- Poddar, J.; Pradhan, M.; Ganguly, G.; Chakrabarti, S. Biochemical deficits and cognitive decline in brain aging: Intervention by dietary supplements. J. Chem. Neuroanat. 2019, 95, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Linnerbauer, M.; Rothhammer, V. Protective Functions of Reactive Astrocytes Following Central Nervous System Insult. Front. Immunol. 2020, 11, 573256. [Google Scholar] [CrossRef] [PubMed]
- Saver, J.L. Targeting the brain: Neuroprotection and neurorestoration in ischemic stroke. Pharmacotherapy 2010, 30, 62S–69S. [Google Scholar] [CrossRef] [PubMed]
- Jasielski, P.; Piędel, F.; Piwek, M.; Rocka, A.; Petit, V.; Rejdak, K. Application of Citicoline in Neurological Disorders: A Systematic Review. Nutrients 2020, 12, 3113. [Google Scholar] [CrossRef]
- Horrocks, L.A.; Dorman, R.V. Prevention by CDP-choline and CDPethanolamine of lipid changes during brain ischemia. In Novel Biochemical, Pharmacological and Clinical Aspects of Cytidinediphosphocholine; Zappia, V., Kennedy, E.P., Nilsson, B.I., Galletti, P., Eds.; Elsevier Science Publishing: Amsterdam, The Netherlands, 1985; pp. 205–215. [Google Scholar]
- Le Poncin-Lafitte, M.; Dutertem, D.; Lageron, A.; Rapin, J.R. CDP-choline et accident cérébral expérimental d’origine vasculaire. Agressologie 1986, 27, 413–416. [Google Scholar] [PubMed]
- Mykita, S.; Golly, F.; Dreyfus, H.; Freysz, L.; Massarelli, R. Effect of CDP-choline on hypocapnic neurons in culture. J. Neurochem. 1986, 47, 223–231. [Google Scholar] [CrossRef]
- Yasuhara, M.; Naito, H. Characteristic actions of CDP-choline on the central nervous system. Curr. Ther. Res. Clin. Exp. 1974, 16, 346–374. [Google Scholar] [PubMed]
- Yasuhara, M.; Naito, H.; Tachibana, Y.; Yasuhara, A. An electrophysiological study on the effects of CDP-choline in the central nervous system. In Novel Biochemical, Pharmacological and Clinical Aspects of Cytidinediphosphocholine; Zappia, V., Kennedy, E.P., Nilsson, B.I., Galletti, P., Eds.; Elsevier Science Publishing: Amsterdam, The Netherlands, 1985; pp. 259–274. [Google Scholar]
- Martí-Viaño, J.L.; Selles, J.; Orts, A.; Marco, J.; Vega, F.; Espluges, J. Antagonismo del coma barbitúrico mediante productos alertizantes. Estudio experimental. Rev. Esp. Anest. Reanim. 1978, 25, 21–28. [Google Scholar]
- Ogashiwa, M.; Sano, K.; Manaka, S.; Kitamura, K.; Kagawa, M.; Takeuchi, K. Effectiveness of CDP-choline on disturbance of consciousness (DOC): 1. An experimental study of concussive head injury in mice. 2. A controlled trial in patients with DOC. In Novel Biochemical, Pharmacological and Clinical Aspects of Cytidinediphosphocholine; Zappia, V., Kennedy, E.P., Nilsson, B.I., Galletti, P., Eds.; Elsevier Science Publishing: Amsterdam, The Netherlands, 1985; pp. 317–327. [Google Scholar]
- Watanabe, S.; Kono, S.; Nakashima, Y.; Mitsunobu, K.; Otsuki, S. Effects of various cerebral metabolic activators on glucose metabolism of brain. Folia Psychiat. Neurol. Jpn. 1975, 29, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Alberghina, M.; Giuffrida-Stella, A.M. Changes of phospholipid metabolizing and lysosomal enzymes in hypoglossal nucleus and ventral horn motoneurons during regeneration of craniospinal nerves. J. Neurochem. 1988, 51, 15–20. [Google Scholar] [CrossRef]
- Arrigoni, E.; Averet, N.; Cohadon, F. Effects of CDP-choline on phospholipase A2 and cholinephosphotransferase activities following a cryogenic brain injury in the rabbit. Biochem. Pharmacol. 1987, 36, 3697–3700. [Google Scholar] [CrossRef]
- Freysz, L.; Golly, F.; Mykita, S.; Avola, R.; Dreyfus, H.; Massarelli, R. Metabolism of neuronal cell culture: Modifications induced by CDP-choline. In Novel Biochemical, Pharmacological and Clinical Aspects of Cytidinediphosphocholine; Zappia, V., Kennedy, E.P., Nilsson, B.I., Galletti, P., Eds.; Elsevier Science Publishing: Amsterdam, The Netherlands, 1985; pp. 117–125. [Google Scholar]
- Massarelli, R.; Mozzi, R.; Golly, F.; Hattori, H.; Dainous, F.; Kanfer, J.N.; Freysz, L. Synthesis de novo of choline, production of choline from phospholipids, and effects of CDP-choline on nerve cell survival. Fidia Res. Ser. 1986, 4, 273–281. [Google Scholar]
- Kitazaki, T.; Ohta, Y.; Tsuda, M. Inhibition of membrane associated phospholipase A2 by CDP-choline. Jpn. Pharmacol. Ther. 1985, 13, 159–164. [Google Scholar]
- Farooqui, A.A.; Litsky, M.L.; Farooqui, T.; Horrocks, L.A.. Inhibitors of intracellular phospholipase A2 activity: Their neurochemical effects and therapeutical importance for neurological disorders. Brain Res. Bull. 1999, 49, 139–153. [Google Scholar] [CrossRef]
- Algate, D.R.; Beard, D.J.; Sacristán, A.; Ortíz, J.A.; Davies, J.E. Study on the effects of oral administration of CDP-choline on EEG changes and lethality induced by epidural compression in the anesthetised cat. Arzneimittelforschung 1983, 33, 1013–1016. [Google Scholar]
- Hayaishi, O.; Ozawa, K.; Araki, C.; Ishii, S.; Kondo, Y. Biochemical studies of head injury and brain edema. Jpn. J. Med. Prog. 1961, 48, 519–539. [Google Scholar]
- Kondo, Y. Experimental study of the therapeutic use of cytidine nucleotids for brain injury. Nihon Geka Hokan. 1968, 32, 489–505. [Google Scholar]
- Tsuchida, T.; Nagai, M.; Hoshino, T.; Kamano, S.; Miyake, H. Treatment of head injuries with intermediate substances of metabolic cycle of brain. II. Basic study on metabolism of cytidine diphosphate choline. Brain Nerve 1967, 19, 1041–1045. [Google Scholar] [PubMed]
- Boismare, F.; Le Poncin, M.; Le François, J.; Hacpille, L.; Marchand, J.C. Étude des effets de l’administration de cytidinediphosphocholine sur les consèquences hémodynamiques, fonctionelles et biochimiques du traumatisme crâniocervical chez le rat. Thérapie 1977, 32, 345–354. [Google Scholar] [PubMed]
- Clendenon, N.R.; Palayoor, S.T.; Gordon, W.A. Influence of CDP-choline on ATPase activity in acute experimental spinal cord trauma. In Novel Biochemical, Pharmacological and Clinical Aspects of Cytidinediphosphocholine; Zappia, V., Kennedy, E.P., Nilsson, B.I., Galletti, P., Eds.; Elsevier Science Publishing: Amsterdam, The Netherlands, 1985; pp. 275–284. [Google Scholar]
- Cohadon, F.; Rigoulet, M.; Guérin, B.; Vandendriessche, M. Edème cérébral vasogénique. Altérations des ATPases membranaires. Restauration par un précurseur des phospholipides. Nouv. Presse Med. 1979, 8, 1589–1591. [Google Scholar]
- Lafuente, J.V.; Cervós-Navarro, J. Estudio por microgavimetría del efecto de la CDP-colina en el edema cerebral experimental inducido por radiaciones ultravioletas. Med. Clin. 1986, 87 (Suppl. 1), 5–8. [Google Scholar]
- Cervós-Navarro, J.; Lafuente, J.V. Effect of cytidine diphosphate choline on ultraviolet-induced brain edema. Adv. Neurol. 1990, 52, 421–429. [Google Scholar] [PubMed]
- Majem, X.; Bidón-Chanal, A.; Vilá-Badó, J. Estudio de los efectos del tratamiento oral con CDP-colina sobre los cambios inducidos por el edema encefálico experimental en el electroencefalograma de la rata no anestesiada. Med. Clin. 1986, 87 (Suppl. 1), 23–25. [Google Scholar]
- Roda, J.E. Répartition macro et microscopique d’un oedème cérébral vasogenique experimental. In Symposium International: Souffrance Cérébrale et Précurseurs des Phospholipides; Laboratoires Cassenne-Takeda: Paris, France, 1980. [Google Scholar]
- Schmidt, K.; Hernekamp, J.F.; Doerr, M.; Zivkovic, A.R.; Brenner, T.; Walther, A.; Weigand, M.A.; Hofer, S. Cytidine-5-diphosphocholine reduces microvascular permeability during experimental endotoxemia. BMC Anesthesiol. 2015, 15, 114. [Google Scholar] [CrossRef] [PubMed]
- Hernekamp, J.F.; Hu, S.X.; Schmidt, V.J.; Vogelpohl, J.; Kneser, U.; Kremer, T. Influence of Cdp-Choline Administration on Early Burn Edema in Rats. Ann. Plast. Surg. 2015, 75, 388–392. [Google Scholar] [CrossRef]
- Dixon, C.E.; Ma, X.; Marion, D.W. Effects of CDP-choline treatment on neurobehavioral deficits after TBI and on hippocampal and neocortical acetylcholine release. J. Neurotrauma 1997, 14, 161–169. [Google Scholar] [CrossRef]
- Plataras, C.; Tsakiris, S.; Angelogianni, P. Effect of CDP-choline on brain acetylcholinesterase and Na(+), K(+)-ATPase in adult rats. Clin. Biochem. 2000, 33, 351–357. [Google Scholar] [CrossRef]
- Başkaya, M.K.; Doğan, A.; Rao, A.M.; Dempsey, R.J. Neuroprotective effects of citicoline on brain edema and blood-brain barrier breakdown after traumatic brain injury. J. Neurosurg. 2000, 92, 448–452. [Google Scholar] [CrossRef]
- Dempsey, R.J.; Raghavendra Rao, V.L. Cytidinediphosphocholine treatment to decrease traumatic brain injury-induced hippocampal neuronal death, cortical contusion volume, and neurological dysfunction in rats. J. Neurosurg. 2003, 98, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Menku, A.; Ogden, M.; Saraymen, R. The protective effects of propofol and citicoline combination in experimental head injury in rats. Turk Neurosurg. 2010, 20, 57–62. [Google Scholar] [PubMed]
- Jacotte-Simancas, A.; Costa-Miserachs, D.; Coll-Andreu, M.; Torras-Garcia, M.; Borlongan, C.V.; Portell-Cortés, I. Effects of voluntary physical exercise, citicoline, and combined treatment on object recognition memory, neurogenesis, and neuroprotection after traumatic brain injury in rats. J. Neurotrauma 2015, 32, 739–751. [Google Scholar] [CrossRef]
- Qian, K.; Gu, Y.; Zhao, Y.; Li, Z.; Sun, M. Citicoline protects brain against closed head injury in rats through suppressing oxidative stress and calpain over-activation. Neurochem. Res. 2014, 39, 1206–1218. [Google Scholar] [CrossRef] [PubMed]
- Gan, D.; Wu, S.; Chen, B.; Zhang, J. Application of the Zebrafish Traumatic Brain Injury Model in Assessing Cerebral Inflammation. Zebrafish 2020, 17, 73–82. [Google Scholar] [CrossRef]
- Cakir, E.; Usul, H.; Peksoylu, B.; Sayin, O.C.; Alver, A.; Topbas, M.; Baykal, S.; Kuzeyli, K. Effects of citicoline on experimental spinal cord injury. J. Clin. Neurosci. 2005, 12, 923–926. [Google Scholar] [CrossRef]
- Yücel, N.; Cayli, S.R.; Ateş, O.; Karadağ, N.; Firat, S.; Turköz, Y. Evaluation of the neuroprotective effects of citicoline after experimental spinal cord injury: Improved behavioral and neuroanatomical recovery. Neurochem. Res. 2006, 31, 767–775. [Google Scholar] [CrossRef]
- Coskun, C.; Avci, B.; Ocak, N.; Yalcin, M.; Dirican, M.; Savci, V. Effect of repeatedly given CDP-choline on cardiovascular and tissue injury in spinal shock conditions: Investigation of the acute phase. J. Pharm. Pharmacol. 2010, 62, 497–506. [Google Scholar] [CrossRef]
- Turkkan, A.; Alkan, T.; Goren, B.; Kocaeli, H.; Akar, E.; Korfali, E. Citicoline and postconditioning provides neuroprotection in a rat model of ischemic spinal cord injury. Acta Neurochir. 2010, 152, 1033–1042. [Google Scholar] [CrossRef]
- Galletti, P.; De Rosa, M.; Cotticelli, M.G.; Morana, A.; Vaccaro, R.; Zappia, V. Biochemical rationale for the use of CDPcholine in traumatic brain injury: Pharmacokinetics of the orally administered drug. J. Neurol. Sci. 1991, 103, S19–S25. [Google Scholar] [CrossRef]
- Qureshi, I.; Endres, J.R. Citicoline: A novel therapeutic agent with neuroprotective, neuromodulatory, and neuroregenerative properties. Nat. Med. J. 2010, 2, 11–25. [Google Scholar]
- Saver, J.L. Target brain: Neuroprotection and neurorestoration in ischemic stroke. Rev. Neurol. Dis. 2010, 7 (Suppl. 1), s14–s21. [Google Scholar]
- Secades, J.J. Citicoline: Pharmacological and clinical review, 2016 update. Rev. Neurol. 2016, 63, S1–S73. [Google Scholar]
- Alvarez-Sabín, J.; Román, G.C. The role of citicoline in neuroprotection and neurorepair in ischemic stroke. Brain Sci. 2013, 3, 1395–1414. [Google Scholar] [CrossRef]
- Moriyama, M.; Tsukumo, T.; Nakagawa, Y. Effects of CDPcholine on head injury. Gendai No Rinsho 1967, 1, 114–120. [Google Scholar]
- Misbach, J.; Andradi, S.; Harahap, T.P.; Soemargo, S.; Markam, S. Double blind trial of Nicholin (CDP-choline) on the patients with severe head injury. In Biannual Meeting of Neurology, Psychiatry and Neurosurgery; Surabaya, Indonesia, 1978. [Google Scholar]
- Ayuso, J.L.; Saiz, J. Efecto protector del citidín-5-difosfato de colina sobre el defecto mnésico post-electrochoque. Munch. Med. Wochenschr. 1977, 119, 53–59. [Google Scholar]
- De la Herrán, J.; Cortina, C.; Salazar, J.; de Monge, F. Utilización del citidín difosfato de colina en lesiones encefálicas graves. Actas Luso Esp. Neurol. Psiquiatr. Ciencias Afines. 1978, 6, 3–12. [Google Scholar]
- Espagno, J.; Trémoulet, M.; Gigaud, M.; Espagno, C. Étude de l’action de la CDP-choline dans les troubles de la vigilance post-traumatique. Vie Médicale 1979, 3, 195–196. [Google Scholar]
- Carcasonne, M.; LeTourneau, J.N. Étude en double insu du réxort en neurotraumatologie infantile. Vie Médicale 1979, 12, 1007. [Google Scholar]
- Richer, E.; Cohadon, F. Essai thérapeutique d’un précurseur des phospholipides sur le traitement des comas traumatiques. In Symposium International: Souffrance cérébrale et Précurseurs des Phospholipides; Laboratoires Cassenne-Takeda: Paris, France, 1980. [Google Scholar]
- Lecuire, J.; Duplay, J. Sperimentazione in doppio cieco della citicolina versus meclofenossato in pazienti colpiti da trauma cranico. G. Ital. Ric. Clin. Ter. 1982, 3, 51–55. [Google Scholar]
- Lecuire, J.; Duplay, J. Sperimentazione della citicolina in un camipone di 154 traumatizzati cranici. G. Ital. Ric. Clin. Ter. 1982, 3, 61–67. [Google Scholar]
- Lecuire, J. Traumatismes crâniens: Étude comparative piracetam-CDP-choline. C. R. Ther. Pharmacol. Clin. 1985, 3, 3–7. [Google Scholar]
- Cohadon, F.; Richer, E.; Poletto, B. Etude d’un precurseur des phospholipides dans le traitement des comas traumatiques graves (A precursor of phospholipids in the treatment of severe traumatic comas). Neurochirurgie 1982, 28, 287–290. [Google Scholar] [PubMed]
- Deleuze, R.; Huguenard, P.; Laborit, G.; Roujas, F. Effets de la CDP-choline sur le rapport lactates/pyruvates dans le LCR en cas de souffrance cérébrale grave. C. R. Ther. 1985, 4, 11–18. [Google Scholar]
- Ogashiwa, M.; Takeuchi, K.; Hara, M.; Tanaka, Y.; Okada, J. Studies on the intrathecal pharmacotherapy. Part I: CDP-choline. Int. J. Clin. Pharmacol. Biopharm. 1975, 12, 327–335. [Google Scholar]
- Ogashiwa, M.; Takeuchi, K. Intrathecal pharmacotherapy in coma. Acta Neurochir. 1976, 34, 37–44. [Google Scholar] [CrossRef]
- De Blas, A.; Martínez-Cubells, J.; Hernando, C. Valoración de la efectividad de la citicolina en el tratamiento de los traumatismos craneoencefálicos. Med. Clin. 1986, 87 (Suppl. 1), 41–44. [Google Scholar]
- Ragguenneau, J.L.; Jarrige, B. Enquête nationale sur les suites des traumatismes crâniens graves: Analyse des 219 traumatismes traités par CDP-choline. Agressologie 1988, 29, 439–443. [Google Scholar]
- Calatayud Maldonado, V.; Calatayud Pérez, J.B.; Aso Escario, J. Effects of CDP-choline on the recovery of patients with head injury. J. Neurol. Sci. 1991, 103, S15–S18. [Google Scholar] [CrossRef]
- Lozano, R. CDP-choline in the treatment of cranio-encephalic traumata. J. Neurol. Sci. 1991, 103, S43–S47. [Google Scholar] [CrossRef]
- Lazowski, T.; Kierul, K.; Bartnicki, M.; Mayzner-Zawadzka, E.; Toczylowska, B.; Ryba, M.; Lewandowski, Z. Effects of citicoline treatment in patients with isolated head trauma: A randomized trial. Critical Care 2003, 7 (Suppl. 2), 78. [Google Scholar] [CrossRef]
- Hinev, S.; Tzoneva, D.; Ljubenova, K.; Tzvetkov, V.; Dimitrov, G.; Stefanov, I. Neuroprotection as component of complex therapy in patients with severe head trauma and cerebrovascular diseases. Anaesthesiol. Intensive Care 2007, 34, 19–24. [Google Scholar]
- Krishna, D.; Chaurasia, I.D.; Jethwani, U. Role of citicoline in traumatic brain injury: A randomized controlled study. IJPMR 2012, 2, 1–5. [Google Scholar]
- Zafonte, R.; Friedewald, W.T.; Lee, S.M.; Levin, B.; Diaz-Arrastia, R.; Ansel, B.; Eisenberg, H.; Timmons, S.D.; Temkin, N.; Novack, T.; et al. The citicoline brain injury treatment (COBRIT) trial: Design and methods. J. Neurotrauma 2009, 26, 2207–2216. [Google Scholar] [CrossRef] [PubMed]
- Zafonte, R.D.; Bagiella, E.; Ansel, B.M.; Novack, T.A.; Friedewald, W.T.; Hesdorffer, D.C.; Timmons, S.D.; Jallo, J.; Eisenberg, H.; Hart, T.; et al. Effect of citicoline on functional and cognitive status among patients with traumatic brain injury: Citicoline Brain Injury Treatment Trial (COBRIT). JAMA 2012, 308, 1993–2000. [Google Scholar] [CrossRef]
- El Reweny, E.M.; Okasha, A.; Hafez, A. The neuroprotective effect of citicholine (CDP choline) in patients with traumatic brain injury. In Proceedings of the 25th ESICM Annual Congress, Lisbon, Portugal, 13–17 October 2012. [Google Scholar]
- Varadaraju, D.N.; Ananthakishan, A. Effect of cerebroprotein hydrolysate with citicoline versus citicoline alone in the initial management of head injury and its clinical outcome a prospective randomised comparative study. J. Evid. Based Med. Healthc. 2017, 4, 2835–2837. [Google Scholar]
- Titov, I.I.; Voloshinsky, O.V.; Martin, A.Y.; Vintonyak, I.V.; Nestor, I.I. Evaluation of neuroprotectoral therapy efficiency in TBI. Pain Anesth. Intensive Care 2018, 3, 61–68. [Google Scholar]
- Trimmel, H.; Majdan, M.; Wodak, A.; Herzer, G.; Csomor, D.; Brazinova, A. Citicoline in severe traumatic brain injury: Indications for improved outcome: A retrospective matched pair analysis from 14 Austrian trauma centers. Wien. Klin. Wochenschr. 2018, 130, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, J.; Hoseinzadeh-Chahkandak, F.; Roobiyat, M.Y.; Pourbagher-Shahri, A.M.; Irankhah, S.; Rajabpour-Sanati, A. Comparison of two different doses of citicoline in patients with traumatic brain injury. J. Surg. Trauma 2020, 8, 8–15. [Google Scholar]
- Salehpour, F.; Shokouhi, G.; Shakeri, M.; Shimia, M.; Mahdkhah, A.; Baradaran, A.; Imani, M.T.; Mirzaee, F.; Azar, A.K.; Bazzazi, A.M.; et al. Neuroprotective effects of citicoline in diffuse axonal injuries. Adv. Biosci. Clin. Med. 2013, 1, 12–15. [Google Scholar]
- Salehpour, F.; Aghazade, J.; Mirzaee, F.; Mahdkhah, A. Citicoline in patients with traumatic brain injuries. EC Neurol. 2015, 2, 87–93. [Google Scholar]
- Shokouhi, G.; Haghjoo, A.G.; Sattarnezhad, N.; Asghari, M.; Sattarnezhad, A.; Asghari, A.; Pezeshki, A. Effects of citicoline on level of consciousness, serum level of fetuin-A and matrix Gla-protein (MGP) in trauma patients with diffuse axonal injury (DAI) and GCS ≤ 8. Ulus Travma Acil. Cerrahi. Derg. 2014, 20, 410–416. [Google Scholar] [CrossRef][Green Version]
- Levin, H.S. Treatment of postconcussional symptoms with CDP-choline. J. Neurol. Sci. 1991, 103, S39–S42. [Google Scholar] [CrossRef]
- Aniruddha, T.J.; Pillai, S.; Devi, B.I.; Sampath, S.; Chandramouli, B.A. Role of citicoline in the management of mild head injury. Indian J. Neurotrauma 2009, 6, 49–52. [Google Scholar]
- Jotwani, V.; Harmon, K.G. Postconcussion syndrome in athletes. Curr. Sports Med. Rep. 2010, 9, 21–26. [Google Scholar] [CrossRef]
- León-Carrión, J.; Domínguez-Roldán, J.M.; Murillo-Cabeza, F.; Domínguez-Morales, M.R.; Muñoz-Sánchez, M.A.; Forastero, P. Advances in the treatment of memory deficits after brain injury: The role of citicholine. In Proceedings of the 3rd World Congress on Brain Injury, Quebec City, QC, Canada, 12–17 June 1999. [Google Scholar]
- León-Carrión, J.; Domínguez-Roldán, J.M.; Murillo-Cabeza, F.; Domínguez-Morales, M.R.; Muñoz-Sánchez, M.A. Normalization of memory-related cerebral blood flow in severe traumatic brain injury patients and improvements of memory induced by citicholine (CDP-choline): The role of a pro-cognitive drug. In Proceedings of the International Conference on Recent Advances in Neurotraumatology, ICRAN’99, Taipei, Taiwan, 20–23 November 1999. [Google Scholar]
- León-Carrión, J.; Domínguez-Roldán, J.M.; Murillo-Cabeza, F.; Domínguez-Morales, M.R.; Muñoz-Sánchez, M.A. The role of citicholine in neuropsychological training after traumatic brain injury. Neurorehabilitation 2000, 14, 33–40. [Google Scholar] [CrossRef]
- Wortzel, H.S.; Arciniegas, D.B. Treatment of post-traumatic cognitive impairments. Curr. Treat. Options Neurol. 2012, 14, 493–508. [Google Scholar] [CrossRef]
- Spiers, P.A.; Hochanadel, G. Citicoline for traumatic brain injury: Report of two cases, including my own. J. Int. Neuropsychol. Soc. 1999, 5, 260–264. [Google Scholar] [CrossRef]
- Secades, J.J. Citicoline for the Treatment of Head Injury: A Systematic Review and Meta-analysis of Controlled Clinical Trials. J. Trauma. Treat 2014, 4, 227. [Google Scholar] [CrossRef]
- El Sayed, I.; Zaki, A.; Fayed, A.M.; Shehata, G.M.; Abdelmonem, S. A meta-analysis of the effect of different neuroprotective drugs in management of patients with traumatic brain injury. Neurosurg. Rev. 2018, 41, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, M.; Yanagi, M. Cytidinediphosphocholine (CDP-choline) for cognitive and behavioural disturbances associated with chronic cerebral disorders in the elderly. Cochrane Database Syst. Rev. 2005, 2, CD000269. [Google Scholar] [CrossRef] [PubMed]
- Mondello, S.; Hasan, A.; Shear, D.A. Editorial: Developing Successful Neuroprotective Treatments for TBI: Translational Approaches, Novel Directions, Opportunities and Challenges. Front. Neurol. 2019, 10, 1326. [Google Scholar] [CrossRef] [PubMed]
- Lerouet, D.; Marchand-Leroux, C.; Besson, V.C. Neuropharmacology in traumatic brain injury: From preclinical to clinical neuroprotection? Fundam. Clin. Pharmacol. 2021. [Google Scholar] [CrossRef]
- Rajguru, M.; Agrawal, A.; Sampath Kumar, N.S.; Amil Kumar, T. An overview of clinical and therapeutic implications of citicoline. Narayana Med. J. 2014, 3, 54–60. [Google Scholar]
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Secades, J.J. Role of Citicoline in the Management of Traumatic Brain Injury. Pharmaceuticals 2021, 14, 410. https://doi.org/10.3390/ph14050410
Secades JJ. Role of Citicoline in the Management of Traumatic Brain Injury. Pharmaceuticals. 2021; 14(5):410. https://doi.org/10.3390/ph14050410
Chicago/Turabian StyleSecades, Julio J. 2021. "Role of Citicoline in the Management of Traumatic Brain Injury" Pharmaceuticals 14, no. 5: 410. https://doi.org/10.3390/ph14050410
APA StyleSecades, J. J. (2021). Role of Citicoline in the Management of Traumatic Brain Injury. Pharmaceuticals, 14(5), 410. https://doi.org/10.3390/ph14050410






