New Method for the Monitoring of Antidepressants in Oral Fluid Using Dried Spot Sampling
Abstract
1. Introduction
2. Results and Discussion
2.1. Cross-Contribution Evaluation
2.2. Optimization of the Extraction Procedure
2.3. Validation Procedure
2.3.1. Selectivity
2.3.2. Calibration Curves and Limits
2.3.3. Intra-Day, Inter-Day, and Intermediate Precision and Accuracy
2.3.4. Extraction Recovery
2.3.5. Stability
2.3.6. Dilution Integrity
2.3.7. Method Applicability
3. Materials and Methods
3.1. Reagents and Standards
3.2. Biological Specimens
3.3. Sample Preparation
3.4. Gas Chromatographic and Mass Spectrometric Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. New WHO Guidelines to Improve the Physical Health of People with Severe Mental Disorders. Available online: https://www.who.int/mental_health/en/ (accessed on 20 December 2018).
- Boa-Sorte, N.; Galvão-Castro, A.V.; Borba, D.; Lima, R.B.N.D.C.; Galvão-Castro, B. HAM/TSP and major depression: The role of age. Braz. J. Infect. Dis. 2015, 19, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Dubovsky, S.L.; Ghosh, B.M.; Serotte, J.C.; Cranwell, V. Psychotic depression: Diagnosis, differential diagnosis, and treatment. Psychother. Psychosom. 2020, 90, 160–177. [Google Scholar] [CrossRef]
- Ridley, M.; Rao, G.; Schilbach, F.; Patel, V. Poverty, depression, and anxiety: Causal evidence and mechanisms. Science 2020, 370, 0214. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Petersen, K.S.; Hibbeln, J.R.; Hurley, D.; Kolick, V.; Peoples, S.; Rodriguez, N.; Woodward-Lopez, G. Nutrition and behavioral health disorders: Depression and anxiety. Nutr. Rev. 2021, 79, 247–260. [Google Scholar] [CrossRef]
- Soares, S.; Barroso, M.; Gallardo, E. A review of current bioanalytical approaches in sample pretreatment techniques for the determination of antidepressants in biological specimens. Rev. Anal. Chem. 2021, 40, 12–32. [Google Scholar] [CrossRef]
- Spielmans, G.I.; Berman, M.I.; Linardatos, E.; Rosenlicht, N.Z.; Perry, A.; Tsai, A. Adjunctive atypical antipsychotic treatment for major depressive disorder: A meta-analysis of depression, quality of life, and safety outcomes. PLoS Med. 2013, 10, e1001403. [Google Scholar] [CrossRef]
- Coulter, C.; Taruc, M.; Tuyay, J.; Moore, C. Antidepressant drugs in oral fluid using liquid chromatography-tandem mass spectrometry. J. Anal. Toxicol. 2010, 34, 64–72. [Google Scholar] [CrossRef][Green Version]
- Magni, L.R.; Purgato, M.; Gastaldon, C.; Papola, D.; Furukawa, A.T.; Cipriani, A.; Barbui, C. Fluoxetine versus other types of pharmacotherapy for depression. Cochrane Database Syst. Rev. 2013, 004185. [Google Scholar] [CrossRef] [PubMed]
- Juan, H.; Zhiling, Z.; Huande, L. Simultaneous determination of fluoxetine, citalopram, paroxetine, venlafaxine in plasma by high performance liquid chromatography–electrospray ionization mass spectrometry (HPLC–MS/ESI). J. Chromatogr. B 2005, 820, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; He, M.; Chen, B.; Hu, B. Hollow fiber based liquid–liquid–liquid microextraction combined with sweeping micellar electrokinetic chromatography for the sensitive determination of second-generation antidepressants in human fluids. Analyst 2015, 140, 1662–1671. [Google Scholar] [CrossRef]
- Berm, E.; Paardekooper, J.; Brummel-Mulder, E.; Hak, E.; Wilffert, B.; Maring, J. A simple dried blood spot method for therapeutic drug monitoring of the tricyclic antidepressants amitriptyline, nortriptyline, imipramine, clomipramine, and their active metabolites using LC-MS/MS. Talanta 2015, 134, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Wille, S.; Maudens, K.; Van Peteghem, C.H.; Lambert, W.E. Development of a solid phase extraction for 13 ‘new’ generation antidepressants and their active metabolites for gas chromatographic–mass spectrometric analysis. J. Chromatogr. A 2005, 1098, 19–29. [Google Scholar] [CrossRef]
- Hiemke, C.; Pfuhlmann, B. Therapeutic drug monitoring for antidepressant drug treatment. Curr. Pharm. Des. 2012, 18, 5818–5827. [Google Scholar] [CrossRef]
- Wille, S.M.R.; Cooreman, S.G.; Neels, H.M.; Lambert, W.E.E. Relevant issues in the monitoring and the toxicology of antidepressants. Crit. Rev. Clin. Lab. Sci. 2008, 45, 25–89. [Google Scholar] [CrossRef] [PubMed]
- Moreno, A.M.J.; Navas, M.J.; Asuero, A.G. HPLC-DAD determination of CNS-acting drugs in human blood, plasma, and serum. Crit. Rev. Anal. Chem. 2013, 44, 68–106. [Google Scholar] [CrossRef]
- Degreef, M.; van Nuijs, A.L.; Maudens, K.E. Validation of a simple, fast liquid chromatography-tandem mass spectrometry method for the simultaneous quantification of 40 antidepressant drugs or their metabolites in plasma. Clin. Chim. Acta 2018, 485, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Rosado, T.; Gonçalves, A.; Martinho, A.; Alves, G.; Duarte, A.P.; Domingues, F.; Silvestre, S.; Granadeiro, L.A.T.B.; Oliveira, V.; Leitão, C.; et al. Simultaneous quantification of antidepressants and metabolites in urine and plasma samples by GC–MS for therapeutic drug monitoring. Chromatographia 2017, 80, 301–328. [Google Scholar] [CrossRef]
- Kall, A.M.; Rohde, M.; Jørgensen, M. Quantitative determination of the antidepressant vortioxetine and its major human metabolite in plasma. Bioanalysis 2015, 7, 2881–2894. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.S.; Borg, D.; Stripp, R. Developing and validating a fast and accurate method to quantify 18 antidepressants in oral fluid samples using SPE and LC–MS-MS. J. Anal. Toxicol. 2020, 44, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Majda, A.; Mrochem, K.; Wietecha-Posłuszny, R.; Zapotoczny, S.; Zawadzki, M. Fast and efficient analyses of the post-mortem human blood and bone marrow using DI-SPME/LC-TOFMS method for forensic medicine purposes. Talanta 2020, 209, 120533. [Google Scholar] [CrossRef] [PubMed]
- Amoli, H.S.; Yamini, Y.; Darmani, H. Polyoxomolybdate 368 /polyaniline nanocomposite as a novel fiber for solid-phase microextraction of antidepressant drugs in biological samples. J. Sep. Sci. 2020, 43, 2636–2645. [Google Scholar] [CrossRef] [PubMed]
- Marasca, C.; Protti, M.; Mandrioli, R.; Atti, A.R.; Armirotti, A.; Cavalli, A.; De Ronchi, D.; Mercolini, L. Whole blood and oral fluid microsampling for the monitoring of patients under treatment with antidepressant drugs. J. Pharm. Biomed. Anal. 2020, 188, 113384. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, A.M.A.; Fernández, P.; Fernández, A.M.; Carro, A.M.; Lorenzo, R.A. Microextraction by packed sorbent followed by ultra high performance liquid chromatography for the fast extraction and determination of six antidepressants in urine. J. Sep. Sci. 2019, 42, 2053–2061. [Google Scholar] [CrossRef]
- Magalhães, P.; Alves, G.; Llerena, A.; Falcão, A. Therapeutic drug monitoring of Fluoxetine, Norfluoxetine and Paroxetine: A new tool based on Microextraction by Packed Sorbent Coupled to Liquid Chromatography. J. Anal. Toxicol. 2017, 41, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Karami, M.; Yamini, Y. On-disc electromembrane extraction-dispersive liquid-liquid microextraction: A fast and effective method for extraction and determination of ionic target analytes from complex biofluids by GC/MS. Anal. Chim. Acta 2020, 1105, 95–104. [Google Scholar] [CrossRef]
- De Boeck, M.; Dubrulle, L.; Dehaen, W.; Tytgat, J.; Cuypers, E. Fast and easy extraction of antidepressants from whole blood using ionic liquids as extraction solvent. Talanta 2018, 180, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Mohebbi, A.; Farajzadeh, M.A.; Yaripour, S.; Mogaddam, M.R.A. Determination of tricyclic antidepressants in human urine samples by the three-step sample pretreatment followed by HPLC-UV analysis: An efficient analytical method for further pharmacokinetic and forensic studies. EXCLI J. 2018, 17, 952–963. [Google Scholar] [CrossRef]
- Farajzadeh, M.A.; Abbaspour, M. Development of new extraction method based on liquid-liquid-liquid extraction followed by dispersive liquid-liquid microextraction for extraction of three tricyclic antidepressants in plasma samples. Biomed. Chromatogr. 2018, 32, e4251. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zheng, S.; Le, J.; Qian, Z.; Zhang, R.; Hong, Z.; Chai, Y. Ultrasound-assisted low-density solvent dispersive liquid–liquid microextraction for the simultaneous determination of 12 new antidepressants and 2 antipsychotics in whole blood by gas chromatography–mass spectrometry. J. Pharm. Biomed. Anal. 2017, 142, 19–27. [Google Scholar] [CrossRef]
- Cabarcos-Fernández, P.; Tabernero-Duque, M.J.; Álvarez-Freire, I.; Bermejo-Barrera, A.M. Determination of seven antidepressants in Pericardial fluid by means of dispersive liquid–Liquid Microextraction and gas Chromatography–Mass Spectrometry. J. Anal. Toxicol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Oenning, A.; Birk, L.; Eller, S.; Oliveira, T.; Merib, J.; Carasek, E. A green and low-cost method employing switchable hydrophilicity solvent for the simultaneous determination of antidepressants in human urine by gas chromatography—Mass spectrometry detection. J. Chromatogr. B 2020, 1143, 122069. [Google Scholar] [CrossRef] [PubMed]
- Hedeshi, M.H.; Rezvani, O.; Bagheri, H. Silane–based modified papers and their extractive phase roles in a microfluidic platform. Anal. Chim. Acta 2020, 1128, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Salamat, Q.; Yamini, Y.; Moradi, M.; Farahani, A.; Feizi, N. Extraction of antidepressant drugs in biological samples using alkanol-based nano structured supramolecular solvent microextraction followed by gas chromatography with mass spectrometric analysis. J. Sep. Sci. 2019, 42, 1620–1628. [Google Scholar] [CrossRef]
- Mohebbi, A.; Farajzadeh, M.A.; Nemati, M.; Sarhangi, N.; Mogaddam, M.R.A. Development of green sodium sulfate-induced solidification of floating organic droplets–dispersive liquid phase microextraction method: Application to extraction of four antidepressants. Biomed. Chromatogr. 2019, 33, e4642. [Google Scholar] [CrossRef]
- Mohebbi, A.; Yaripour, S.; Farajzadeh, M.A.; Mogaddam, M.R.A. Combination of dispersive solid phase extraction and deep eutectic solvent–based air–assisted liquid–liquid microextraction followed by gas chromatography–mass spectrometry as an efficient analytical method for the quantification of some tricyclic antidepressant drugs in biological fluids. J. Chromatogr. A 2018, 1571, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Ríos-Gómez, J.; Fresco-Cala, B.; García-Valverde, M.T.; Lucena, R.; Cardenas, S. Carbon Nanohorn Suprastructures on a Paper Support as a Sorptive Phase. Molecules 2018, 23, 1252. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, P.K.; Tapadia, K. Pharmacokinetic determination and analysis of nortriptyline based on GC–MS coupled with hollow-fiber drop-to-drop solvent microextraction technique. Bioanalysis 2018, 10, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Zhang, C.; Wang, L.; Wang, K.; Kang, W.; Lian, K.; Li, H. Determination of nine mental drugs in human plasma using solid-phase supported liquid-liquid extraction and HPLC-MS/MS. Microchem. J. 2021, 160, 105647. [Google Scholar] [CrossRef]
- Moretti, M.; Freni, F.; Valentini, B.; Vignali, C.; Groppi, A.; Visonà, S.D.; Osculati, A.M.M.; Morini, L. Determination of antidepressants and antipsychotics in Dried Blood Spots (DBSs) collected from post-mortem samples and evaluation of the stability over a three-month period. Molecules 2019, 24, 3636. [Google Scholar] [CrossRef] [PubMed]
- Ask, K.S.; Lid, M.; Øiestad, E.L.; Pedersen-Bjergaard, S.; Gjelstad, A. Liquid-phase microextraction in 96-well plates—Calibration and accurate quantification of pharmaceuticals in human plasma samples. J. Chromatogr. A 2019, 1602, 117–123. [Google Scholar] [CrossRef]
- Ask, K.S.; Øiestad, E.L.; Pedersen-Bjergaard, S.; Gjelstad, A. Dried blood spots and parallel artificial liquid membrane extraction–A simple combination of microsampling and microextraction. Anal. Chim. Acta 2018, 1009, 56–64. [Google Scholar] [CrossRef]
- Pinto, M.A.L.; de Souza, I.D.; Queiroz, M.E.C. Determination of drugs in plasma samples by disposable pipette extraction with C18-BSA phase and liquid chromatography–tandem mass spectrometry. J. Pharm. Biomed. Anal. 2017, 139, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Fahimirad, B.; Rajabi, M.; Elhampour, A. A rapid and simple extraction of anti-depressant drugs by effervescent salt-assisted dispersive magnetic micro solid-phase extraction method using new adsorbent Fe3O4@SiO2@N3. Anal. Chim. Acta 2019, 1047, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Cala, B.F.; Mompó-Roselló, Ó.; Simó-Alfonso, E.F.; Cárdenas, S.; Herrero-Martínez, J.M. Carbon nanotube-modified monolithic polymethacrylate pipette tips for (micro)solid-phase extraction of antidepressants from urine samples. Microchim. Acta 2018, 185, 127. [Google Scholar] [CrossRef] [PubMed]
- Safari, M.; Shahlaei, M.; Yamini, Y.; Shakorian, M.; Arkan, E. Magnetic framework composite as sorbent for magnetic solid phase extraction coupled with high performance liquid chromatography for simultaneous extraction and determination of tricyclic antidepressants. Anal. Chim. Acta 2018, 1034, 204–213. [Google Scholar] [CrossRef]
- Cai, J.; Zhu, G.-T.; He, X.-M.; Zhang, Z.; Wang, R.-Q.; Feng, Y.-Q. Polyoxometalate incorporated polymer monolith microextraction for highly selective extraction of antidepressants in undiluted urine. Talanta 2017, 170, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, F.; Hadjmohammadi, M.R.; Aghaie, A.B. Ultrasound-assisted dispersive magnetic solid phase extraction based on amino-functionalized Fe3O4 adsorbent for recovery of clomipramine from human plasma and its determination by high performance liquid chromatography: Optimization by experimental design. J. Chromatogr. B 2017, 1063, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Zilfidou, E.; Kabir, A.; Furton, K.G.; Samanidou, V. An improved fabric phase sorptive extraction method for the determination of five selected antidepressant drug residues in human blood serum prior to high performance liquid chromatography with diode array detection. J. Chromatogr. B 2019, 1125, 121720. [Google Scholar] [CrossRef] [PubMed]
- Lioupi, A.; Kabir, A.; Furton, K.G.; Samanidou, V. Fabric phase sorptive extraction for the isolation of five common antidepressants from human urine prior to HPLC-DAD analysis. J. Chromatogr. B 2019, 1118–1119, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, M.; Chamsaz, M.; Aghamohammadhasan, M.; Shams, A. Ultrasonic assisted magnetic dispersive solid phase microextraction for pre concentration of serotonin–norepinephrine reuptake inhibitor drugs. Anal. Biochem. 2018, 551, 7–18. [Google Scholar] [CrossRef]
- Behpour, M.; Nojavan, S.; Asadi, S.; Shokri, A. Combination of gel-electromembrane extraction with switchable hydrophilicity solvent-based homogeneous liquid-liquid microextraction followed by gas chromatography for the extraction and determination of antidepressants in human serum, breast milk and wastewater. J. Chromatogr. A 2020, 1621, 461041. [Google Scholar] [CrossRef] [PubMed]
- Nojavan, S.; Shaghaghi, H.; Rahmani, T.; Shokri, A.; Nasiri-Aghdam, M. Combination of electromembrane extraction and electro-assisted liquid-liquid microextraction: A tandem sample preparation method. J. Chromatogr. A 2018, 1563, 20–27. [Google Scholar] [CrossRef]
- Mofazzeli, F.; Shirvan, H.A.; Mohammadi, F. Extraction and determination of tricyclic antidepressants in real samples using air-dispersed liquid–liquid microextraction prior to gas chromatography and flame ionization detection. J. Sep. Sci. 2018, 41, 4340–4347. [Google Scholar] [CrossRef]
- He, X.; Sun, T.; Wang, L.; Jiang, X. Pipette-tip micro-solid phase extraction based on melamine-foam@polydopamine followed by ultra-high-performance liquid chromatography–quadrupole–time-of-flight mass spectrometry for detection of psychotropic drugs in human serum. J. Chromatogr. B 2021, 1163, 122499. [Google Scholar] [CrossRef]
- Huestis, M.A.; Verstraete, A.; Kwong, T.C.; Morland, J.; Vincent, M.J.; De La Torre, R. Oral fluid testing: Promises and pitfalls. Clin. Chem. 2011, 57, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, E.; Barroso, M.; Queiroz, J. Current technologies and considerations for drug bioanalysis in oral fluid. Bioanalysis 2009, 1, 637–667. [Google Scholar] [CrossRef]
- Gallardo, E.; Queiroz, J.A. The role of alternative specimens in toxicological analysis. Biomed. Chromatogr. 2008, 22, 795–821. [Google Scholar] [CrossRef]
- Neumann, J.; Beck, O.; Dahmen, N.; Böttcher, M. Potential of oral fluid as a clinical specimen for compliance monitoring of psychopharmacotherapy. Ther. Drug Monit. 2018, 40, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Abu-Rabie, P.; Spooner, N. Dried matrix spot direct analysis: Evaluating the robustness of a direct elution technique for use in quantitative bioanalysis. Bioanalysis 2011, 3, 2769–2781. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rehim, A.; Abdel-Rehim, M. Dried saliva spot as a sampling technique for saliva samples. Biomed. Chromatogr. 2014, 28, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.; Rosado, T.; Barroso, M.; Gallardo, E. Determination of antiepileptic drugs using dried saliva spots. J. Anal. Toxicol. 2019, 43, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Prata, M.; Vaz, C.; Rosado, T.; Restolho, J.; Barroso, M.; Araújo, A.R.T.S.; Gallardo, E. Determination of methadone and EDDP in oral fluid using the dried saliva spots sampling approach and gas chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2019, 411, 2177–2187. [Google Scholar] [CrossRef] [PubMed]
- Caramelo, D.; Rosado, T.; Oliveira, V.; Rodilla, J.M.L.; Rocha, P.; Barroso, M.; Gallardo, E. Determination of antipsychotic drugs in oral fluid using dried saliva spots by gas chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2019, 411, 6141–6153. [Google Scholar] [CrossRef] [PubMed]
- Elmongy, H.; Abdel-Rehim, M. Saliva as an alternative specimen to plasma for drug bioanalysis: A review. TrAC Trends Anal. Chem. 2016, 83, 70–79. [Google Scholar] [CrossRef]
- Cheng, P.-S.; Fu, C.-Y.; Lee, C.-H.; Liu, C.; Chien, C.-S. GC–MS quantification of ketamine, norketamine, and dehydronorketamine in urine specimens and comparative study using ELISA as the preliminary test methodology. J. Chromatogr. B 2007, 852, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Simões, S.S.; Ajenjo, A.C.; Dias, M.J. Dried blood spots combined to an UPLC–MS/MS method for the simultaneous determination of drugs of abuse in forensic toxicology. J. Pharm. Biomed. Anal. 2018, 147, 634–644. [Google Scholar] [CrossRef]
- Scientific Working Group for Forensic Toxicology. Scientific Working Group for Forensic Toxicology (SWGTOX) standard practices for method validation in forensic toxicology. J. Anal. Toxicol. 2013, 37, 452–474. [Google Scholar] [CrossRef]
- World Anti-doping Agency. International Standard for Laboratories: Identification Criteria for Qualitative Assays Incorporating Column Chromatography and Mass Spectrometry; World Anti-doping Agency: Montreal, QC, Canada, 2010. [Google Scholar]
- De Castro, A.; Concheiro, M.; Quintela, O.; Cruz, A.; López-Rivadulla, M. LC–MS/MS method for the determination of nine antidepressants and some of their main metabolites in oral fluid and plasma: Study of correlation between venlafaxine concentrations in both matrices. J. Pharm. Biomed. Anal. 2008, 48, 183–193. [Google Scholar] [CrossRef]





| Analytes | Weight | Linear Range (ng/mL) | Linearity | R2 a | LOD/LLOQ (ng/mL) | |
|---|---|---|---|---|---|---|
| Slope a | Intercept a | |||||
| FLX | 1/× | 100–500 | 0.0022 ± 0.0006 | −0.0923 ± 0.0839 | 0.9965 ± 0.0022 | 100 |
| VLX | 1/× | 100–500 | 0.0079 ± 0.0059 | 0.1559 ± 0.1499 | 0.9935 ± 0.0028 | 100 |
| DVLX | 1/× | 50–500 | 0.0083 ± 0.0038 | −0.0450 ± 0.0888 | 0.9954 ± 0.0025 | 50 |
| CIT | 1/× | 20–200 | 0.0021 ± 0.0006 | −0.0085 ± 0.0110 | 0.9935 ± 0.0040 | 20 |
| SRT | 1/× | 40–250 | 0.0003 ± 0.0003 | −0.0023 ± 0.0024 | 0.9964 ± 0.0032 | 40 |
| PXT | 1/× | 10–100 | 0.0041 ± 0.0029 | −0.0247 ± 0.0227 | 0.9959 ± 0.0013 | 10 |
| Analytes | Spiked (ng/mL) | Measured a (ng/mL) | CV (%) | RE (%) |
|---|---|---|---|---|
| FLX | 125 | 133.65 ± 13.57 | 10.15 | 6.92 |
| 275 | 298.07 ± 18.17 | 6.09 | 8.39 | |
| 450 | 468.57 ± 58.50 | 12.48 | 4.13 | |
| VLX | 125 | 126.05 ± 20.47 | 16.24 | 0.84 |
| 275 | 280.38 ± 42.13 | 15.03 | 1.96 | |
| 450 | 434.36 ± 51.96 | 11.96 | 3.47 | |
| DVLX | 62.5 | 66.57 ± 10.17 | 15.28 | 6.51 |
| 137.5 | 141.81 ± 21.86 | 15.42 | 3.14 | |
| 450 | 469.84 ± 54.61 | 11.62 | 4.41 | |
| CIT | 25 | 26.73 ± 3.29 | 12.30 | 6.94 |
| 55 | 49.14 ± 5.87 | 11.94 | −10.65 | |
| 180 | 183.10 ± 27.42 | 14.97 | 1.72 | |
| SRT | 50 | 49.73 ± 8.61 | 17.31 | −0.54 |
| 110 | 108.69 ± 13.43 | 12.35 | −1.19 | |
| 225 | 234.13 ± 33.14 | 14.15 | 4.06 | |
| PXT | 12.5 | 14.18 ± 0.74 | 5.19 | 13.43 |
| 27.5 | 26.42 ± 1.59 | 6.01 | −3.94 | |
| 90 | 100.20 ± 4.33 | 4.33 | 11.33 |
| Analytes | Spiked (ng/mL) | Inter-Day (n = 3) | Intra-Day (n = 6) | ||
|---|---|---|---|---|---|
| CV (%) | RE (%) | CV (%) | RE (%) | ||
| FLX | 100 | 1.86 | 2.44 | 8.83 | 7.89 |
| 200 | 4.19 | −0.55 | - | - | |
| 250 | 5.61 | −2.34 | - | - | |
| 300 | 2.88 | −1.30 | 4.00 | −4.83 | |
| 400 | 1.18 | 0.35 | - | - | |
| 500 | 0.75 | 1.40 | 6.31 | −5.39 | |
| VLX | 100 | 5.04 | 0.62 | 14.64 | −0.95 |
| 200 | 9.63 | −1.77 | - | - | |
| 250 | 3.94 | 1.06 | - | - | |
| 300 | 2.59 | −0.38 | 5.23 | −12.99 | |
| 400 | 2.83 | 0.42 | - | - | |
| 500 | 3.10 | −0.22 | 3.23 | −8.39 | |
| DVLX | 50 | 7.65 | 6.34 | 8.98 | −2.52 |
| 100 | 8.56 | −3.73 | - | - | |
| 125 | 11.26 | −3.47 | - | - | |
| 300 | 2.79 | −1.63 | 11.63 | −12.04 | |
| 400 | 2.24 | 2.66 | - | - | |
| 500 | 2.01 | −0.17 | 3.35 | −3.89 | |
| CIT | 20 | 10.27 | 13.16 | 13.31 | 6.10 |
| 40 | 7.61 | −7.00 | - | - | |
| 50 | 8.82 | −8.42 | - | - | |
| 120 | 2.88 | 0.68 | 8.37 | −9.22 | |
| 160 | 1.10 | −0.98 | - | - | |
| 200 | 2.14 | 2.57 | 5.07 | −10.16 | |
| SRT | 40 | 3.75 | 5.23 | 7.76 | 1.75 |
| 80 | 2.57 | −6.29 | - | - | |
| 100 | 5.77 | −0.70 | - | - | |
| 150 | 1.83 | 0.83 | 3.85 | −10.15 | |
| 200 | 1.41 | −0.14 | - | - | |
| 250 | 1.62 | 1.07 | 5.19 | −9.56 | |
| PXT | 10 | 1.48 | 12.83 | 1.44 | 9.72 |
| 20 | 1.58 | −5.88 | - | - | |
| 25 | 3.78 | −9.05 | - | - | |
| 60 | 2.42 | −1.04 | 7.02 | −6.55 | |
| 80 | 0.95 | 1.80 | - | - | |
| 100 | 1.60 | 1.34 | 5.57 | 2.53 | |
| Analytes | Recoveries a (%) | ||
|---|---|---|---|
| Low-Spiked Concentration | Medium-Spiked Concentration | High-Spiked Concentration | |
| FLX | 21.68 ± 0.41 | 22.70 ± 1.20 | 23.29 ± 1.28 |
| VLX | 46.48 ± 4.44 | 29.58 ± 3.14 | 35.79 ± 1.91 |
| DVLX | 35.77 ± 2.45 | 33.88 ± 1.09 | 38.49 ± 3.21 |
| NFLX | 38.30 ± 7.61 | 13.03 ± 2.38 | 14.07 ± 2.59 |
| CIT | 40.78 ± 6.15 | 41.57 ± 7.18 | 35.25 ± 2.04 |
| SRT | 24.26 ± 2.70 | 21.56 ± 2.60 | 24.42 ± 0.55 |
| PXT | 21.53 ± 2.66 | 21.74 ± 1.91 | 20.47 ± 0.91 |
| Samples | Analytes | Concentration (ng/mL) |
|---|---|---|
| 1 | FLX/NFLX | 542.1/Detected—Identified |
| 2 | VLX/DVLX | 2033.4/701.7 |
| 3 | CIT | 32.2 |
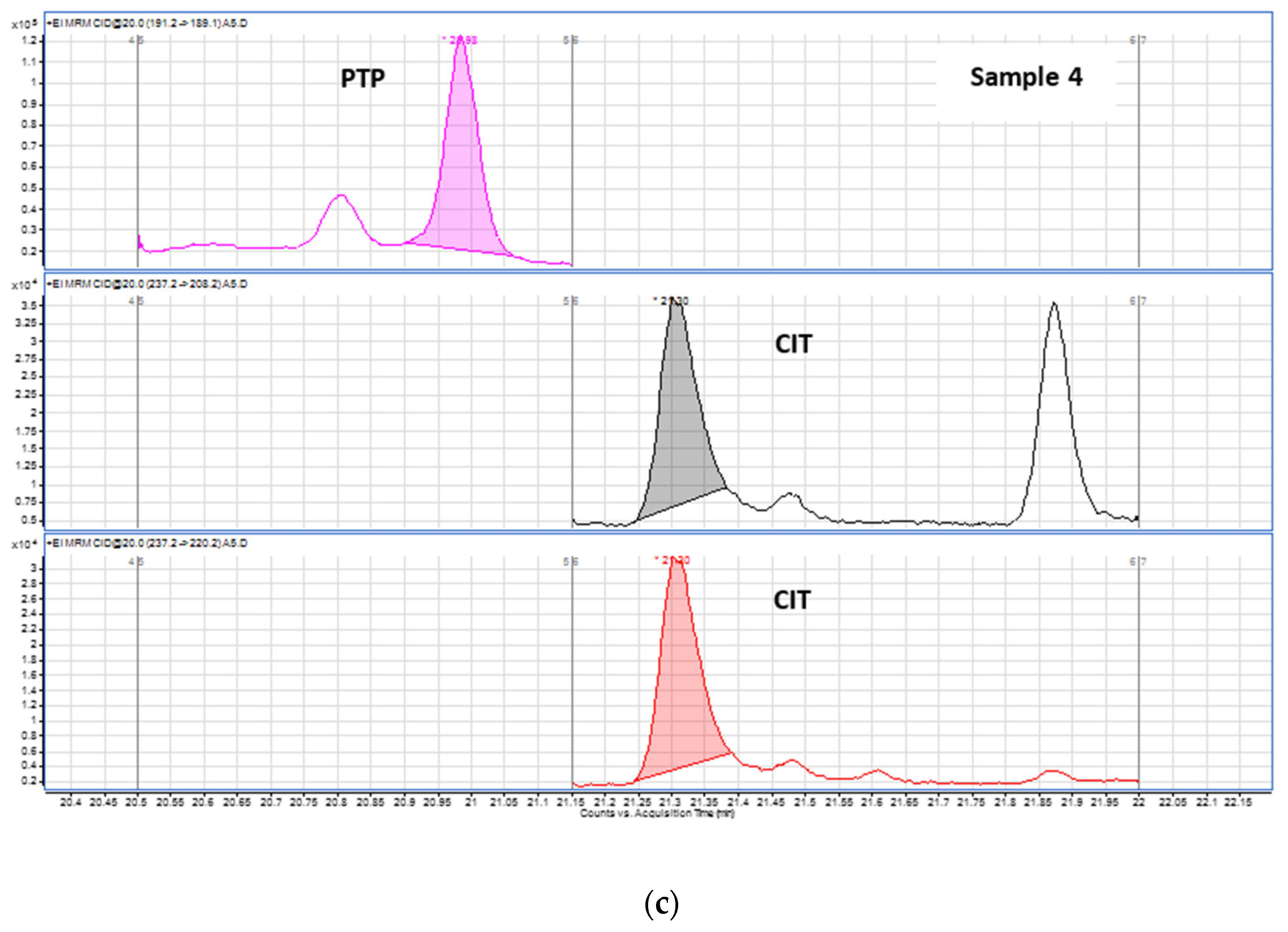
| 4 | CIT | 242.8 |
| 5 | VLX/DVLX | 136.0/356.8 |
| Analytes | Retention Time (min) | Quantifier Transition (m/z) | Qualifier Transition (m/z) | Collision Energy (eV) |
|---|---|---|---|---|
| FLX | 14.20 | 261.2–219.1 | 381.6–116.1 | 20 (20) |
| VLX | 15.86 | 134.1–119.1 | 178.8–134.1 | 10 (20) |
| DVLX | 16.86 | 191.7–177.1 | 391.0–273.3 | 10 (15) |
| NFLX b | 17.29 | 319.2–215.1 | 319.2–86.1 | 5 (15) |
| PTP a | 21.01 | 191.2–189.1 | - | 20 |
| CIT | 21.33 | 237.2–208.2 | 237.2–220.2 | 20 (20) |
| SRT | 23.27 | 346.8–189.2 | 333.0–73.1 | 5 (20) |
| PXT | 24.72 | 250.1–154.2 | 250.1–140.2 | 20 (15) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares, S.; Rosado, T.; Barroso, M.; Gallardo, E. New Method for the Monitoring of Antidepressants in Oral Fluid Using Dried Spot Sampling. Pharmaceuticals 2021, 14, 1284. https://doi.org/10.3390/ph14121284
Soares S, Rosado T, Barroso M, Gallardo E. New Method for the Monitoring of Antidepressants in Oral Fluid Using Dried Spot Sampling. Pharmaceuticals. 2021; 14(12):1284. https://doi.org/10.3390/ph14121284
Chicago/Turabian StyleSoares, Sofia, Tiago Rosado, Mário Barroso, and Eugenia Gallardo. 2021. "New Method for the Monitoring of Antidepressants in Oral Fluid Using Dried Spot Sampling" Pharmaceuticals 14, no. 12: 1284. https://doi.org/10.3390/ph14121284
APA StyleSoares, S., Rosado, T., Barroso, M., & Gallardo, E. (2021). New Method for the Monitoring of Antidepressants in Oral Fluid Using Dried Spot Sampling. Pharmaceuticals, 14(12), 1284. https://doi.org/10.3390/ph14121284








