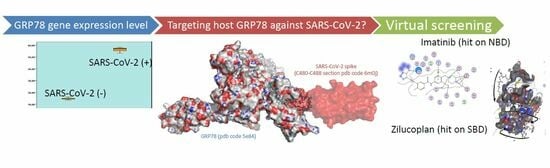
Preliminary Virtual Screening Studies to Identify GRP78 Inhibitors Which May Interfere with SARS-CoV-2 Infection
Abstract
1. Introduction
2. Results
2.1. GRP78 Gene Expression Studies
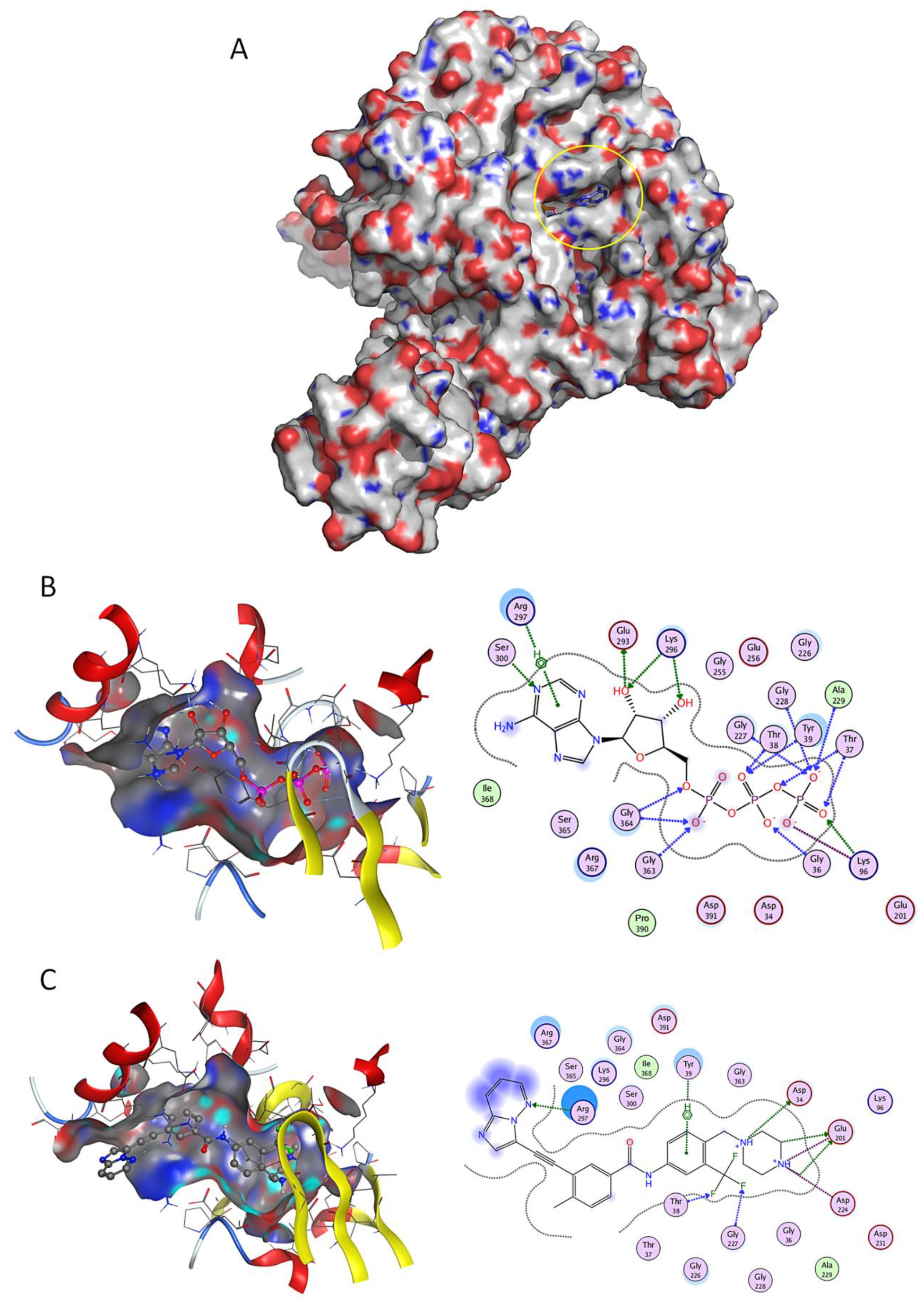
2.2. Virtual Screening Studies
3. Discussion
4. Materials and Methods
4.1. Gene Expression Studies
4.1.1. Patients
4.1.2. Gene Expression Analysis
4.1.3. Statistical Analysis
4.2. Molecular Docking Virtual Screening
4.2.1. Preparation of Receptor and Ligands
4.2.2. Molecular Docking
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 16, 69. [Google Scholar] [CrossRef]
- Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e6. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef]
- Shang, J.; Ye, G.; Shi, K.; Wan, Y.; Luo, C.; Aihara, H.; Geng, Q.; Auerbach, A.; Li, F. Structural basis of receptor recognition by SARS-CoV-2. Nature 2020, 581, 221–224. [Google Scholar] [CrossRef]
- Wan, Y.; Shang, J.; Graham, R.; Baric, R.S.; Li, F. Receptor Recognition by the Novel Coronavirus from Wuhan: An Analysis Based on Decade-Long Structural Studies of SARS Coronavirus. J. Virol. 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, M.S.; Zamzami, M.A.; Choudhry, H.; Murtaza, B.N.; Kazmi, I.; Ahmad, H.; Shakoori, A. Origin, Potential Therapeutic Targets and Treatment for Coronavirus Disease (COVID-19). Pathogens 2020, 9, 307. [Google Scholar] [CrossRef] [PubMed]
- Shereen, M.A.; Khan, S.; Kazmi, A.; Bashir, N.; Siddique, R. COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. J. Adv. Res. 2020, 24, 91–98. [Google Scholar] [CrossRef]
- Versteeg, G.A.; van de Nes, P.S.; Bredenbeek, P.J.; Spaan, W.J. The Coronavirus Spike Protein Induces Endoplasmic Reticulum Stress and Upregulation of Intracellular Chemokine mRNA Concentrations. J. Virol. 2007, 81, 10981–10990. [Google Scholar] [CrossRef] [PubMed]
- Quinones, Q.J.; de Ridder, G.; Pizzo, S.V. GRP78: A chaperone with diverse roles beyond the endoplasmic reticulum. Histol. Histopathol. 2008, 23, 1409–1416. [Google Scholar] [PubMed]
- Zhang, Y.; Liu, R.; Ni, M.; Gill, P.; Lee, A.S. Cell Surface Relocalization of the Endoplasmic Reticulum Chaperone and Unfolded Protein Response Regulator GRP78/BiP. J. Boil. Chem. 2010, 285, 15065–15075. [Google Scholar] [CrossRef]
- Ibrahim, I.M.; Abdelmalek, D.H.; ElFiky, A.A. GRP78: A cell’s response to stress. Life Sci. 2019, 226, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.M.; Abdelmalek, D.H.; Elshahat, M.E.; ElFiky, A.A. COVID-19 spike-host cell receptor GRP78 binding site prediction. J. Infect. 2020, 80, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; Tsai, Y. Patent Application Publication: SRC inhibitor to block cell surface GRP78 expression 2019. Available online: http://www.freepatentsonline.com/y2019/0076431.html (accessed on 25 June 2020).
- Rosa, S.G.V.; Santos, W.C. Clinical trials on drug repositioning for COVID-19 treatment. Rev. Panam. Salud Pública 2020, 44, e40-13. [Google Scholar] [CrossRef] [PubMed]
- Serafin, M.B.; Bottega, A.; Foletto, V.S.; da Rosa, T.F.; Hörner, A.; Hörner, R. Drug repositioning is an alternative for the treatment of coronavirus COVID-19. Int. J. Antimicrob. Agents 2020, 105969. [Google Scholar] [CrossRef] [PubMed]
- Guy, R.K.; di Paola, R.S.; Romanelli, F.; Dutch, R.E. Rapid repurposing of drugs for COVID-19. Science 2020, 368, 829–830. [Google Scholar] [CrossRef]
- Huang, M.; Tang, T.; Pang, P.; Li, M.; Ma, R.; Lu, J.; Shu, J.; You, Y.; Chen, B.; Liang, J.; et al. Treating COVID-19 with Chloroquine. J. Mol. Cell Boil. 2020, 12, 322–325. [Google Scholar] [CrossRef]
- Mehra, M.R.; Ruschitzka, F.; Patel, A.N. Retraction—Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: A multinational registry analysis. Lancet 2020, 10. [Google Scholar] [CrossRef]
- Hughes, S.J.; Antoshchenko, T.; Chen, Y.; Lü, H.; Pizarro, J.C.; Park, H.-W. Probing the ATP Site of GRP78 with Nucleotide Triphosphate Analogs. PLoS ONE 2016, 11, e0154862. [Google Scholar] [CrossRef]
- Koseler, A.; Sabirli, R.; Goren, T.; Turkçuer, I.; Kurt, O. Endoplasmic Reticulum Stress Markers in SARS-COV-2 Infection and Pneumonia: Case-Control Study. In Vivo 2020, 34, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Barabutis, N. Unfolded Protein Response in Acute Respiratory Distress Syndrome. Lung 2019, 197, 827–828. [Google Scholar] [CrossRef]
- Bromberg, Z.; Deutschman, C.S.; Weiss, Y.G. Heat shock protein 70 and the acute respiratory distress syndrome. J. Anesthesia 2005, 19, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Haider, T.; Simader, E.; Glück, O.; Ankersmit, H.J.; Heinz, T.; Hajdu, S.; Negrin, L.L. Systemic release of heat-shock protein 27 and 70 following severe trauma. Sci. Rep. 2019, 9, 9595. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, J.A.; Tremblay, B.J.-M.; Mansfield, M.J.; Woody, O.; Lobb, B.; Banerjee, A.; Chandiramohan, A.; Tiessen, N.; Dvorkin-Gheva, A.; Revill, S.; et al. Gene Expression and in Situ Protein Profiling of Candidate SARS-CoV-2 Receptors in Human Airway Epithelial Cells and Lung Tissue. bioRxiv. 2020. Available online: https://www.biorxiv.org/content/10.1101/2020.04.07.030742v2 (accessed on 25 June 2020).
- Chen, H.-H.; Chen, C.-C.; Lin, Y.-S.; Chang, P.-C.; Lu, Z.-Y.; Lin, C.-F.; Chen, C.-L.; Chang, C.-P. AR-12 suppresses dengue virus replication by down-regulation of PI3K/AKT and GRP78. Antivir. Res. 2017, 142, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Nain, M.; Mukherjee, S.; Karmakar, S.P.; Paton, A.W.; Paton, J.C.; Abdin, M.Z.; Basu, A.; Kalia, M.; Vrati, S. GRP78 Is an Important Host Factor for Japanese Encephalitis Virus Entry and Replication in Mammalian Cells. J. Virol. 2017, 91, e02274-16. [Google Scholar] [CrossRef]
- Reid, S.P.; Shurtleff, A.C.; Costantino, J.A.; Tritsch, S.R.; Retterer, C.; Spurgers, K.B.; Bavari, S. HSPA5 is an essential host factor for Ebola virus infection. Antivir. Res. 2014, 109, 171–174. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2017, 46, D1074–D1082. [Google Scholar] [CrossRef]
- Wishart, D.S.; Knox, C.; Guo, A.C.; Cheng, D.; Shrivastava, S.; Tzur, D.; Gautam, B.; Hassanali, M. DrugBank: A knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2007, 36, D901–D906. [Google Scholar] [CrossRef]
- Ermakova, S.P.; Kang, B.S.; Choi, B.Y.; Schuster, T.F.; Ma, W.-Y.; Bode, A.M.; Dong, Z. Epigallocatechin Gallate Overcomes Resistance to Etoposide-Induced Cell Death by Targeting the Molecular Chaperone Glucose-Regulated Protein 78. Cancer Res. 2006, 66, 9260–9269. [Google Scholar] [CrossRef]
- Dyall, J.; Coleman, C.; Hart, B.; Venkataraman, T.; Holbrook, M.R.; Kindrachuk, J.; Johnson, R.F.; Olinger, G.; Jahrling, P.B.; Laidlaw, M.; et al. Repurposing of Clinically Developed Drugs for Treatment of Middle East Respiratory Syndrome Coronavirus Infection. Antimicrob. Agents Chemother. 2014, 58, 4885–4893. [Google Scholar] [CrossRef]
- Versailles-Hospital. A Randomized Non-Comparative Phase 2 Pilot Study Testing the Value of Imatinib Mesylate as an Early Treatment of Covid-19 Disease in Aged Hospitalized Patients; NCT04357613, France. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04357613 (accessed on 25 June 2020).
- Martin, S.; Lamb, H.K.; Brady, C.; Lefkove, B.; Bonner, M.Y.; Thompson, P.; E Lovat, P.; Arbiser, J.L.; Hawkins, A.R.; Redfern, C.P. Inducing apoptosis of cancer cells using small-molecule plant compounds that bind to GRP78. Br. J. Cancer 2013, 109, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Kosakowska-Cholody, T.; Lin, J.; Srideshikan, S.M.; Scheffer, L.; I Tarasova, N.; Acharya, J. HKH40A downregulates GRP78/BiP expression in cancer cells. Cell Death Dis. 2014, 5, e1240. [Google Scholar] [CrossRef] [PubMed]
- Asling, J.; Morrison, J.; Mutsaers, A. Targeting HSP70 and GRP78 in canine osteosarcoma cells in combination with doxorubicin chemotherapy. Cell Stress Chaperon 2016, 21, 1065–1076. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Wang, Z.; Peng, C.; You, J.; Shen, J.; Han, S.; Chen, J. Dietary compound isoliquiritigenin targets GRP78 to chemosensitize breast cancer stem cells via β-catenin/ABCG2 signaling. Carcinogenesis 2014, 35, 2544–2554. [Google Scholar] [CrossRef]
- Wilhelm, S.M.; Adnane, L.; Newell, P.; Villanueva, A.; Llovet, J.M.; Lynch, M. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol. Cancer Ther. 2008, 7, 3129–3140. [Google Scholar] [CrossRef]
- Nagano, T.; Tachihara, M.; Nishimura, Y. Dacomitinib, a second-generation irreversible epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) to treat non-small cell lung cancer. Drugs Today 2019, 55, 231–236. [Google Scholar] [CrossRef]
- Paranjpe, R.; Basatneh, D.; Tao, G.; de Angelis, C.; Noormohammed, S.; Ekinci, E.; Abughosh, S.; Ghose, R.; Trivedi, M.V. Neratinib in HER2-Positive Breast Cancer Patients. Ann. Pharmacother. 2019, 53, 612–620. [Google Scholar] [CrossRef]
- Tan, F.H.; Putoczki, T.L.; Stylli, S.S.; Luwor, R. Ponatinib: A novel multi-tyrosine kinase inhibitor against human malignancies. OncoTargets Ther. 2019, 12, 635–645. [Google Scholar] [CrossRef]
- Langdon, S.P.; Kay, C.; Um, I.H.; Dodds, M.; Muir, M.; Sellar, G.; Kan, J.; Gourley, C.; Harrison, D.J. Evaluation of the dual mTOR/PI3K inhibitors Gedatolisib (PF-05212384) and PF-04691502 against ovarian cancer xenograft models. Sci. Rep. 2019, 9, 18742–18749. [Google Scholar] [CrossRef] [PubMed]
- Meulenbeld, H.; Mathijssen, R.H.; Verweij, J.; de Wit, R.; de Jonge, M.J. Danusertib, an aurora kinase inhibitor. Expert Opin. Investig. Drugs 2012, 21, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, L.D.; Aranda, R.; Lee, M.; Tovar, E.A.; Essenburg, C.; Madaj, Z.; Chiang, H.; Briere, D.; Hallin, J.; Lopez-Casas, P.P.; et al. Glesatinib Exhibits Antitumor Activity in Lung Cancer Models and Patients HarboringMETExon 14 Mutations and Overcomes Mutation-mediated Resistance to Type I MET Inhibitors in Nonclinical Models. Clin. Cancer Res. 2017, 23, 6661–6672. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.; Normanno, N. Tivozanib, a pan-VEGFR tyrosine kinase inhibitor for the potential treatment of solid tumors. IDrugs Investig. Drugs J. 2010, 13, 636–645. [Google Scholar]
- Huang, L.-T.; Ma, J.-T.; Zhang, S.-L.; Li, X.-H.; Sun, L.; Jing, W.; Zhao, J.-Z.; Wang, Y.-R.; Han, C.-B. Durable Clinical Response to Pyrotinib After Resistance to Prior Anti-HER2 Therapy for HER2-Positive Advanced Gastric Cancer: A Case Report. Front. Oncol. 2019, 9, 1453. [Google Scholar] [CrossRef]
- Verhagen, C.V.; de Haan, R.; Hageman, F.; Oostendorp, T.P.; Carli, A.L.; O’Connor, M.J.; Jonkers, J.; Verheij, M.; Brekel, M.W.V.D.; Vens, C. Extent of radiosensitization by the PARP inhibitor olaparib depends on its dose, the radiation dose and the integrity of the homologous recombination pathway of tumor cells. Radiother. Oncol. 2015, 116, 358–365. [Google Scholar] [CrossRef]
- Spinello, A.; Saltalamacchia, A.; Magistrato, A. Is the Rigidity of SARS-CoV-2 Spike Receptor-Binding Motif the Hallmark for Its Enhanced Infectivity? Insights from All-Atom Simulations. J. Phys. Chem. Lett. 2020, 11, 4785–4790. [Google Scholar] [CrossRef]
- Hati, S.; Bhattacharyay, S. Impact of Thiol-Disulfide Balance on the Binding of Covid-19 Spike Protein with Angiotensin Converting Enzyme 2 Receptor. bioRxiv. 23 June 2020. Available online: https://www.biorxiv.org/content/10.1101/2020.05.07.083147v1 (accessed on 25 June 2020). [CrossRef]
- Elfiky, A.A. Natural products may interfere with SARS-CoV-2 attachment to the host cell. J. Biomol. Struct. Dyn. 2020, 1–10. [Google Scholar] [CrossRef]
- Triantafilou, K.; Fradelizi, D.; Wilson, K.; Triantafilou, M. GRP78, a Coreceptor for Coxsackievirus A9, Interacts with Major Histocompatibility Complex Class I Molecules Which Mediate Virus Internalization. J. Virol. 2002, 76, 633–643. [Google Scholar] [CrossRef]
- Wu, Y.-P.; Chang, C.-M.; Hung, C.-Y.; Tsai, M.-C.; Schuyler, S.C.; Wang, R. Japanese encephalitis virus co-opts the ER-stress response protein GRP78 for viral infectivity. Virol. J. 2011, 8, 128. [Google Scholar] [CrossRef]
- Gurusinghe, K.R.D.S.N.S.; Mishra, A.; Mishra, S. Glucose-regulated protein 78 substrate-binding domain alters its conformation upon EGCG inhibitor binding to nucleotide-binding domain: Molecular dynamics studies. Sci. Rep. 2018, 8, 5487. [Google Scholar] [CrossRef]
- Macias, A.; Williamson, D.S.; Allen, N.; Borgognoni, J.; Clay, A.; Daniels, Z.; Dokurno, P.; Drysdale, M.J.; Francis, G.L.; Graham, C.J.; et al. Adenosine-Derived Inhibitors of 78 kDa Glucose Regulated Protein (Grp78) ATPase: Insights into Isoform Selectivity. J. Med. Chem. 2011, 54, 4034–4041. [Google Scholar] [CrossRef] [PubMed]
- Birukova, A.A.; Singleton, P.A.; Gawlak, G.; Tian, X.; Mirzapoiazova, T.; Mambetsariev, B.; Dubrovskyi, O.; Oskolkova, O.; Bochkov, V.N.; Birukov, K.G. GRP78 is a novel receptor initiating a vascular barrier protective response to oxidized phospholipids. Mol. Boil. Cell 2014, 25, 2006–2016. [Google Scholar] [CrossRef] [PubMed]
- Girona, J.; Rodríguez-Borjabad, C.; Ibarretxe, D.; Vallvé, J.-C.; Ferré, R.; Heras, M.; Rodríguez-Calvo, R.; Guaita-Esteruelas, S.; Martínez-Micaelo, N.; Plana, N.; et al. The Circulating GRP78/BiP Is a Marker of Metabolic Diseases and Atherosclerosis: Bringing Endoplasmic Reticulum Stress into the Clinical Scenario. J. Clin. Med. 2019, 8, 1793. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Feith, M.; Siewert, J.; Wester, H.-J.; Hoefler, H. Expression and clinical significance of Glucose Regulated Proteins GRP78 (BiP) and GRP94 (GP96) in human adenocarcinomas of the esophagus. BMC Cancer 2008, 8, 70. [Google Scholar] [CrossRef]
- Yang, J.; Nune, M.; Zong, Y.; Zhou, L.; Liu, Q. Close and Allosteric Opening of the Polypeptide-Binding Site in a Human Hsp70 Chaperone BiP. Structure 2015, 23, 2191–2203. [Google Scholar] [CrossRef]
- Vilar, S.; Cozza, G.; Moro, S. Medicinal chemistry, and the molecular operating environment (MOE): Application of QSAR and molecular docking to drug discovery. Curr. Top. Med. Chem. 2008, 8, 1555–1572. [Google Scholar] [CrossRef]

| Parameters | SARS-CoV-2 (-) Pneumonia (n = 24) Median (IQR) | SARS-CoV-2 (+) Pneumonia (n = 10) Median (IQR) | p-Value |
|---|---|---|---|
| Gender Male | 14 (41.17%) | 6 (60%) | p1 = 0.928 |
| Female | 10 (58.83%) | 4 (40%) | |
| Age | 52.5 (37–75.75) | 60.5 (36.5–72.5) | p2 = 0.838 |
| Fever (°C) | 36.85 (36.525–37.675) | 36.7 (36.27–37.35) | p2 = 0.589 |
| sPO2 | 95 (92.25–97) | 95 (93–97.25) | p2 = 0.926 |
| Sys. BP (mm/Hg) | 127 (110–140.75) | 128 (110–142.5) | p2 = 0.956 |
| Dias. BP (mm/Hg) | 80 (70.5–88.75) | 70 (61.5–80) | p2 = 0.056 |
| PSI Score | 76.5 (38.25–113) | 82.5 (51–118) | p2 = 0.642 |
| CURB-65 Score | 1 (0.5–2) | 1 (1–2) | p2 = 0.341 |
| Patient Groups | SARS-CoV-2(−) Pneumonia (n = 24) | SARS-CoV-2(+) Pneumonia (n = 10) | p-Value | ||
|---|---|---|---|---|---|
| Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | ||
| GRP-78 mRNA Levels | 14.7 ± 0.95 | 14.7 (14.26–15.25) | 56.41 ± 1.69 | 57.09 (54.09–57.69) | * 0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmeira, A.; Sousa, E.; Köseler, A.; Sabirli, R.; Gören, T.; Türkçüer, İ.; Kurt, Ö.; Pinto, M.M.; Vasconcelos, M.H. Preliminary Virtual Screening Studies to Identify GRP78 Inhibitors Which May Interfere with SARS-CoV-2 Infection. Pharmaceuticals 2020, 13, 132. https://doi.org/10.3390/ph13060132
Palmeira A, Sousa E, Köseler A, Sabirli R, Gören T, Türkçüer İ, Kurt Ö, Pinto MM, Vasconcelos MH. Preliminary Virtual Screening Studies to Identify GRP78 Inhibitors Which May Interfere with SARS-CoV-2 Infection. Pharmaceuticals. 2020; 13(6):132. https://doi.org/10.3390/ph13060132
Chicago/Turabian StylePalmeira, Andreia, Emília Sousa, Aylin Köseler, Ramazan Sabirli, Tarık Gören, İbrahim Türkçüer, Özgür Kurt, Madalena M. Pinto, and M. Helena Vasconcelos. 2020. "Preliminary Virtual Screening Studies to Identify GRP78 Inhibitors Which May Interfere with SARS-CoV-2 Infection" Pharmaceuticals 13, no. 6: 132. https://doi.org/10.3390/ph13060132
APA StylePalmeira, A., Sousa, E., Köseler, A., Sabirli, R., Gören, T., Türkçüer, İ., Kurt, Ö., Pinto, M. M., & Vasconcelos, M. H. (2020). Preliminary Virtual Screening Studies to Identify GRP78 Inhibitors Which May Interfere with SARS-CoV-2 Infection. Pharmaceuticals, 13(6), 132. https://doi.org/10.3390/ph13060132