Advances in CXCR7 Modulators
Abstract
1. Introduction
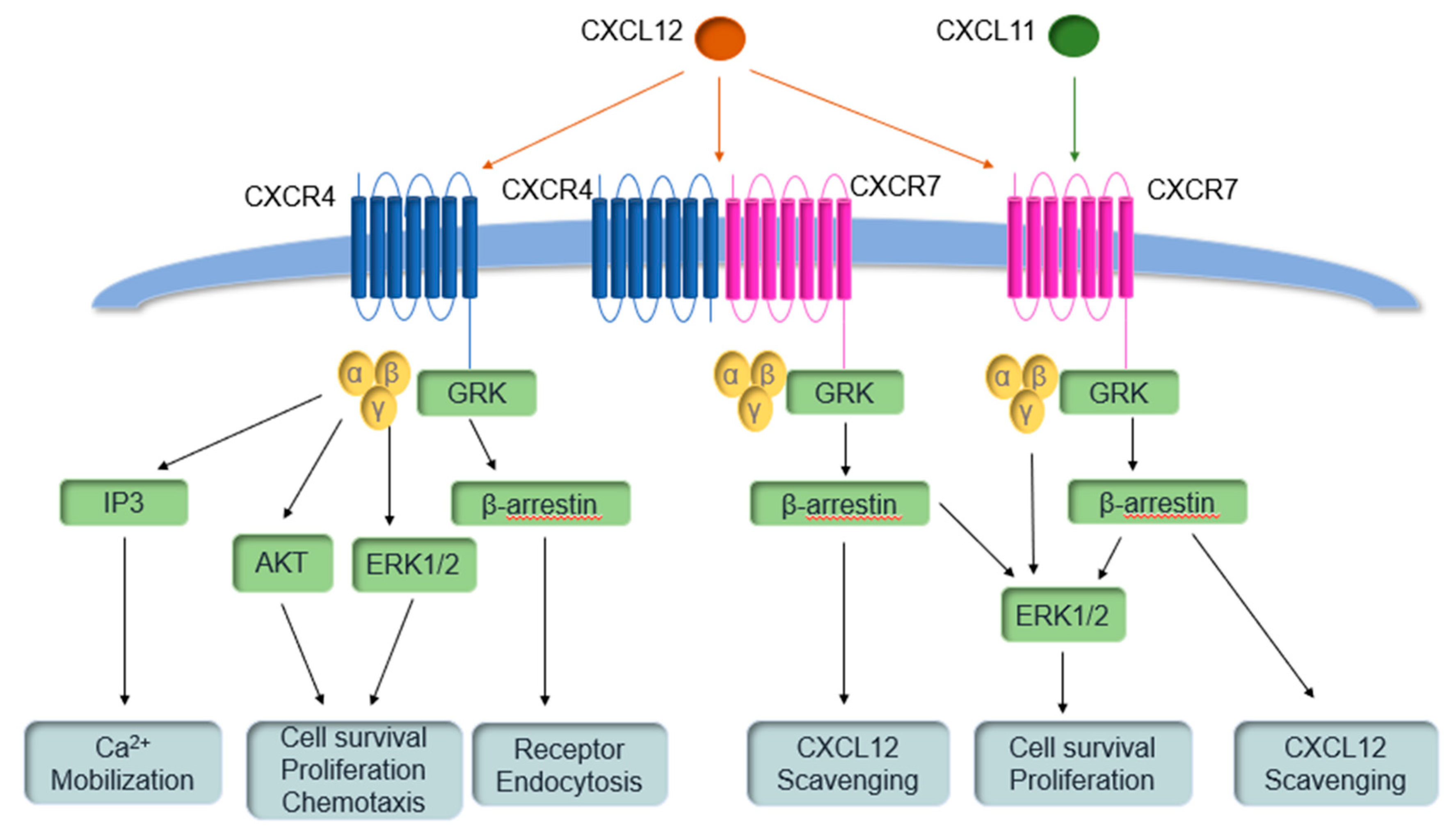
2. The Physiological Roles of CXCR7
2.1. The Role of CXCR7 in Neurological Conditions
2.2. The Role of CXCR7 in Inflammation
2.3. The Role of CXCR7 in Cancers
2.4. The Role of CXCR7 in Other Disease States
3. CXCR7 Modulators
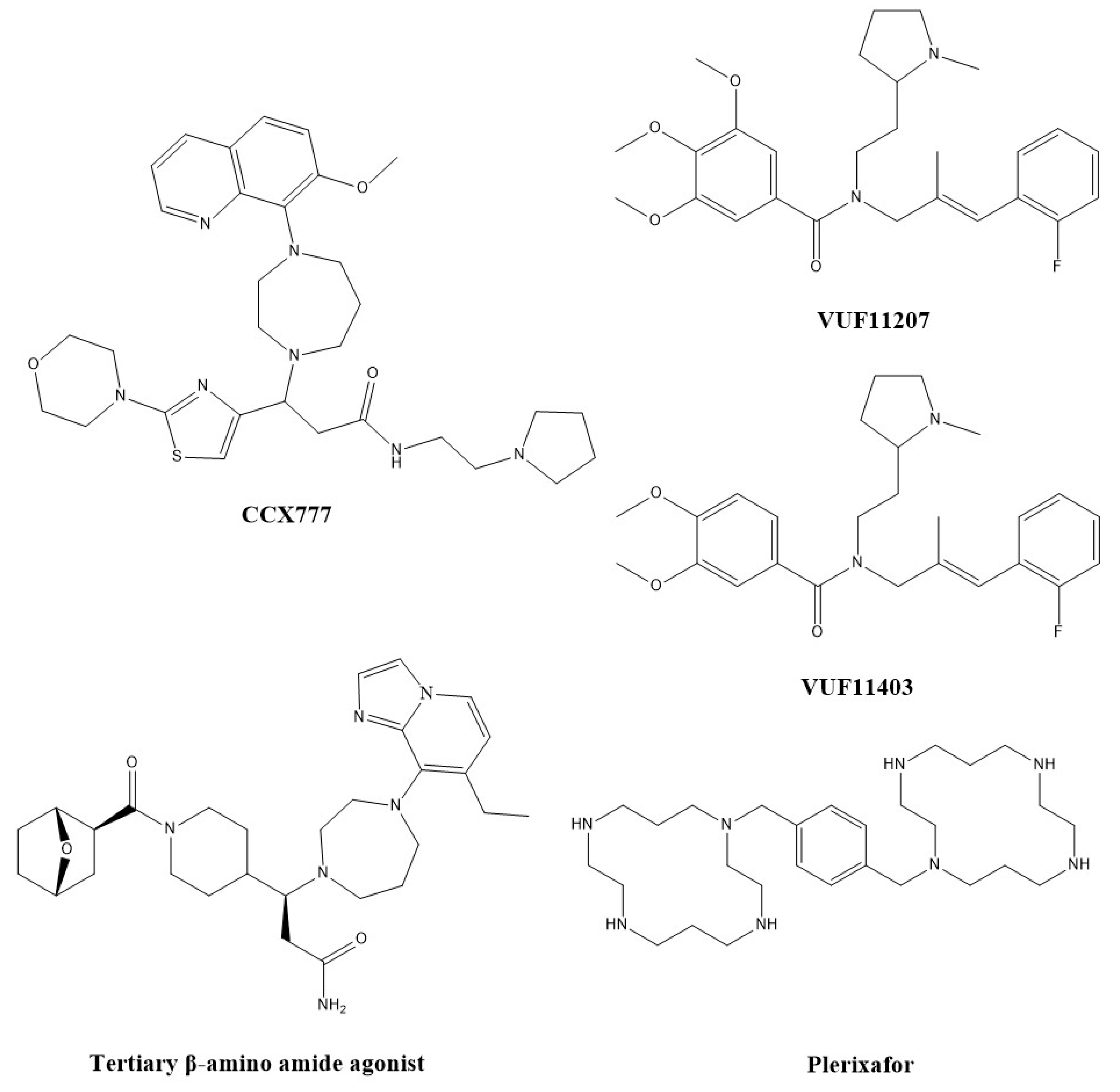
3.1. Small Molecule Modulators
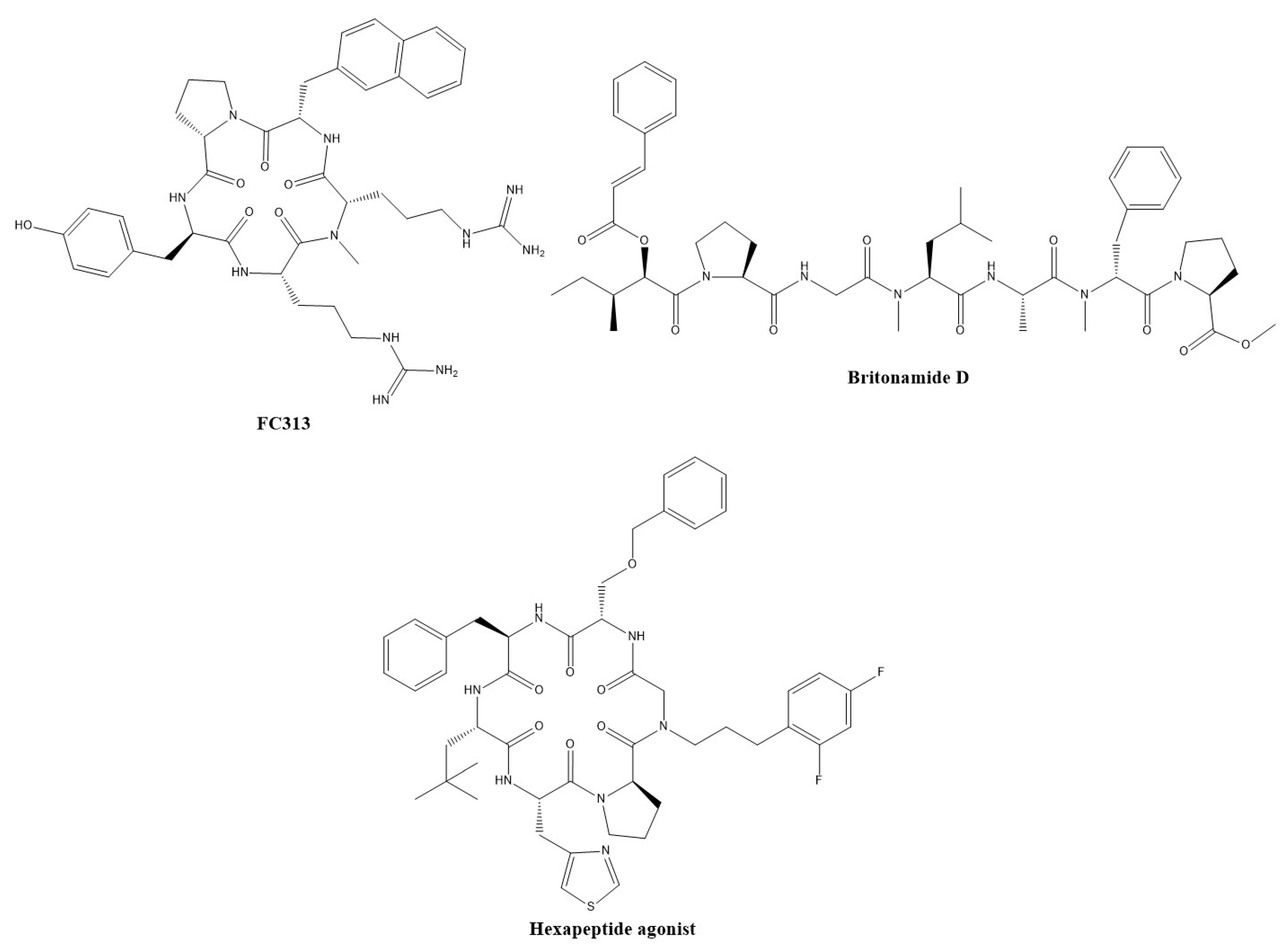
3.2. Peptide-Based Modulators
4. Why Agonists are Producing Hypothesized Antagonist Results
5. Conclusions
Funding
Conflicts of Interest
References
- Balabanian, K.; Lagane, B.; Infantino, S.; Chow, K.Y.C.; Harriague, J.; Moepps, B.; Arenzana-Seisdedos, F.; Thelen, M.; Bachelerie, F. The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J. Biol. Chem. 2005, 280, 35760–35766. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.M.; Summers, B.C.; Wang, Y.; Melikian, A.; Berahovich, R.; Miao, Z.; Penfold, M.E.; Sunshine, M.J.; Littman, D.R.; Kuo, C.J.; et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J. Exp. Med. 2006, 203, 2201–2213. [Google Scholar] [CrossRef] [PubMed]
- Monnier, J.; Boissan, M.; L’Helgoualc’h, A.; Lacombe, M.; Turlin, B.; Zucman-Rossi, J.; Théret, N.; Piquet-Pellorce, C.; Samson, M. CXCR7 is up-regulated in human and murine hepatocellular carcinoma and is specifically expressed by endothelial cells. Eur. J. Cancer 2012, 48, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Broxmeyer, H.E.; Cooper, S.; Kohli, L.; Hangoc, G.; Lee, Y.; Mantel, C.; Clapp, D.W.; Kim, C.H. Transgenic expression of stromal cell-derived factor-1/CXC chemokine ligand 12 enhances myeloid progenitor cell survival/antiapoptosis in vitro in response to growth factor withdrawal and enhances myelopoiesis in vivo. J. Immunol. 2003, 170, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Lataillade, J.J.; Clay, D.; Dupuy, C.; Rigal, S.; Jasmin, C.; Bourin, P.; Le Bousse-Kerdilès, M.C. Chemokine SDF-1 enhances circulating CD34+ cell proliferation in synergy with cytokines: Possible role in progenitor survival. Blood 2000, 95, 756–768. [Google Scholar] [CrossRef] [PubMed]
- Bhaloo, S.I.; Wu, Y.; Le Bras, A.; Yu, B.; Gu, W.; Xie, Y.; Deng, J.; Wang, Z.; Zhang, Z.; Kong, D.; et al. Binding of dickkopf-3 to CXCR7 enhances vascular progenitor cell migration and degradable graft regeneration. Circ. Res. 2018, 123, 451–466. [Google Scholar] [CrossRef] [PubMed]
- Szpakowska, M.; Dupuis, N.; Baragli, A.; Counson, M.; Hanson, J.; Piette, J.; Chevigné, A. Human herpesvirus 8-encoded chemokine vCCL2/vMIP-II is an agonist of the atypical chemokine receptor ACKR3/CXCR7. Biochem. Pharmacol. 2016, 114, 14–21. [Google Scholar] [CrossRef]
- Klein, K.R.; Karpinich, N.O.; Espenschied, S.T.; Wilcockson, H.H.; Dunworth, W.P.; Hoopes, S.L.; Kushner, E.J.; Bautch, V.L.; Caron, K.M. Decoy receptor CXCR7 modulates adrenomedullin-mediated cardiac and lymphatic vascular development. Dev. Cell. 2014, 30, 528–540. [Google Scholar] [CrossRef]
- Ikeda, Y.; Kumagai, H.; Skach, A.; Sato, M.; Yanagisawa, M. Modulation of circadian glucorticoid oscillation via adrenal opioid-CXCR7 signaling alters emotional behavior. Cell 2013, 155, 1323–1336. [Google Scholar] [CrossRef]
- Chatterjee, M.; Borst, O.; Walker, B.; Fotinos, A.; Vogel, S.; Seizer, P.; Mack, A.; Alampour-Rajabi, S.; Rath, D.; Geisler, T.; et al. Macrophage migration inhibitory factor limits activation-induced apoptosis of platelets via CXCR7-dependent Akt signaling. Circ. Res. 2014, 115, 939–949. [Google Scholar] [CrossRef]
- Rajagopal, S.; Kim, J.; Ahn, S.; Craig, S.; Lam, C.M.; Gerard, N.P.; Gerard, C.; Lefkowitz, R.J. β-arrestin but not G protein mediated signaling by the “decoy” receptor CXCR7. Proc. Natl. Acad. Sci. USA 2010, 107, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Hattermann, K.; Held-Feindt, J.; Lucius, R.; Müerköster, S.S.; Penforld, M.E.T.; Schall, T.J.; Mentlein, R. The Chemokine Receptor CXCR7 is Highly Expressed in Human Glioma Cells and Mediates Antiapoptic Effects. Cancer Res. 2010, 70, 3299–3308. [Google Scholar] [CrossRef] [PubMed]
- Duda, D.G.; Kozin, S.V.; Kirkpatrick, N.D.; Xu, L.; Fukumura, D.; Jain, R.K. CXCL12 (SDF1α)-CXCR4/CXCR7 pathway inhibition: An emerging sensitizer for anticancer therapies? Clin. Cancer Res. 2011, 17, 2074–2080. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Arya, R.K.; Trivedi, A.K.; Sanyal, S.; Baral, R.; Dormond, O.; Briscoe, D.M.; Datta, D. Chemokine receptor trio: CXCR3, CXCR4 and CXCR7 crosstalk via CXCL11 and CXCL12. Cytokine Growth Factor Rev. 2013, 24, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Levoye, A.; Balabanian, K.; Baleux, F.; Bachelerie, F.; Lagane, B. CXCR7 heterodimerizes with CXCR4 and regulates CXCL12-mediated G protein signaling. Blood 2009, 113, 6085–6093. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shlozawa, Y.; Wang, J.; Wang, Y.; Jung, Y.; Plenta, K.J.; Mehra, R.; Loberg, R.; Talchman, R.S. The role of CXCR7/RDC1 as a chemokine receptor for CXCL12/SDF-1 in prostate cancer. J. Biol. Chem. 2008, 283, 4283–4294. [Google Scholar] [CrossRef]
- Luker, K.E.; Gupta, M.; Luker, G.D. Imaging chemokine receptor dimerization with firefly luciferase complementation. FASEB J. 2009, 23, 823–834. [Google Scholar] [CrossRef]
- Thelen, M.; Thelen, S. CXCR7, CXCR4 and CXCL12: An eccentric trio? J. Neuroimmunol. 2008, 198, 9–13. [Google Scholar] [CrossRef]
- Infantino, S.; Moepps, B.; Thelen, M. Expression and regulation of the orphan receptor RDC1 and its putative ligand in human dendritic and B cells. J. Immunol. 2006, 176, 2197–2207. [Google Scholar] [CrossRef]
- Sierro, F.; Biben, C.; Martinez-Munoz, L.; Mellado, M.; Ransohoff, R.M.; Li, M.; Woel, B.; Leung, H.; Groom, J.; Batten, M.; et al. Disrupted cardiac development but normal hematopoiesis in mice deficient in the second CXCL12/SDF-1 receptor, CXCR7. Proc. Natl. Acad. Sci. USA 2007, 104, 14759–14764. [Google Scholar] [CrossRef]
- Dambly-Chaudière, C.; Cubedo, N.; Ghysen, A. Control of cell migration in the development of the posterior lateral line: Antagonistic interactions between the chemokine receptors CXCR4 and CXCR7/RDC1. BMC Dev. Biol. 2007, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Boldajipor, B.; Mahabaleshwar, H.; Kardash, E.; Reichman-Fried, M.; Blaser, H.; Minina, S.; Wilson, D.; Xu, Q.; Raz, E. Control of chemokine-guided cell migration by ligand sequestration. Cell 2008, 132, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Nibbs, R.J.B.; Graham, G.J. Immune regulation by atypical chemokine receptors. Nat. Rev. Immunol. 2013, 13, 815–829. [Google Scholar] [CrossRef] [PubMed]
- Mazzinghi, B.; Ronconi, E.; Lazzeri, E.; Sagrinati, C.; Ballerini, L.; Angelotti, M.L.; Parente, E.; Mancina, R.; Netti, G.S.; Becherucci, F.; et al. Essential but differential role for CXCR4 and CXCR7 in the therapeutic homing of human renal progenitor cells. J. Exp. Med. 2008, 205, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Valentin, G.; Haas, P.; Gilmour, D. The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr. Biol. 2007, 17, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Donà, E.; Barry, J.D.; Valentin, G.; Quirin, C.; Khmelinskii, A.; Kunze, A.; Durdu, S.; Newton, L.R.; Fernandez-Minan, A.; Huber, W.; et al. Directional tissue migration through a self-generated chemokine gradient. Nature 2013, 503, 285–289. [Google Scholar] [CrossRef]
- Wang, Y.; Li, G.; Stanco, A.; Long, J.E.; Crawford, D.; Potter, G.B.; Pleasure, S.J.; Behrens, T.; Rubenstein, J.L. CXCR4 and CXCR7 have distinct functions in regulating interneuron migration. Neuron 2011, 69, 61–76. [Google Scholar] [CrossRef]
- Sánchez-Alcañiz, J.A.; Haege, S.; Mueller, W.; Pla, R.; Mackay, F.; Schulz, S.; López-Bendito, G.; Stumm, R.; Marín, O. Cxcr7 controls neuronal migration by regulating chemokine responsiveness. Neuron 2011, 69, 77–90. [Google Scholar] [CrossRef]
- Bakondi, B.; Shimada, I.; Peterson, B.; Spees, J. SDF-1α secreted by human CD133-derived multipotent stromal cells promotes neural progenitor cell survival through CXCR7. Stem Cells Dev. 2011, 20, 1021–1029. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, J.; Li, Y.; Yang, G. Roles of chemokine CXCL12 and its receptors in ischemic stroke. Curr. Drug Targets 2012, 13, 166–172. [Google Scholar] [CrossRef]
- Cruz-Orgeno, L.; Holman, D.W.; Dorsey, D.; Zhou, L.; Zhang, P.; Wright, M.; McCandless, E.E.; Patel, J.R.; Luker, G.D.; Littman, D.R.; et al. CXCR7 influences leukocyte entry into the CNS parenchyma by controlling abluminal CXCL12 abundance during autoimmunity. J. Exp. Med. 2011, 208, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Puchert, M.; Pelkner, F.; Stein, G.; Angelov, D.N.; Boltze, J.; Wagner, D.C.; Odoardi, F.; Flügel, A.; Streit, W.J.; Engele, J. Astrocytic expression of the CXCL12 receptor, CXCR7/ACKR3 is a hallmark of the diseased, but not developing CNS. Mol. Cell. Neurosci. 2017, 85, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Yu, X.; Deng, J.; Ou, S.; Liu, X.; Wang, T.; Liu, Y.; Yang, J.; Tan, C.; Yuan, J.; et al. CXCR7 regulates epileptic seizures by controlling the synaptic activity of hippocampal granule cells. Cell Death Dis. 2019, 10, 825. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Penfold, M.E.T.; Matsuda, A.; Ohyanagi, N.; Kaneko, K.; Miyabe, Y.; Matsumoto, K.; Schall, T.J.; Miyasaka, N.; Nanki, T. Pathogenic role of CXCR7 in rheumatoid arthritis. Arthritis Rheum. 2010, 62, 3211–3220. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.F.; Ansari, M.A.; Nadeem, A.; Bakheet, S.A.; Al-Ayadhi, L.Y.; Attia, S.M. Upregulation of peripheral CXC and CC chemokine receptor expression on CD4+ T cells is associated with immune dysregulation in children with autism. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 81, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, M.; Rath, D.; Schlotterbeck, J.; Rheinlaender, J.; Walker-Allgaier, B.; Alnaggar, N.; Zdanyte, M.; Müller, I.; Borst, O.; Geisler, T.; et al. Regulation of oxidized platelet lipidome: Implications for coronary artery disease. Eur. Heart J. 2017, 38, 1993–2005. [Google Scholar] [CrossRef]
- Gerbauer, F.; Tachezy, M.; Effenberger, K.; von Loga, K.; Zander, H.; Marx, A.; Kaifi, J.T.; Sauter, G.; Izbicki, J.R.; Bockhorn, M. Prognostic impact of CXCR4 and CXCR7 expression in pancreatic adenocarcinoma. J. Surg. Oncol. 2011, 104, 140–145. [Google Scholar] [CrossRef]
- Yu, Y.; Li, H.; Xue, B.; Jiang, X.; Huang, K.; Ge, J.; Zhang, H.; Chen, B. SDF-1/CXCR7 axis enhances ovarian cancer cell invasion by MMP-9 expression through p38 MAPK pathway. DNA Cell Biol. 2014, 33, 543–549. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Li, Z.; Zhang, Z.; Zhang, Y. Chemokine receptor 7 targets the vascular endothelial growth factor vis the AKT/ERK pathway to regulate angiogenesis in colon cancer. Cancer Med. 2019, 8, 5327–5340. [Google Scholar] [CrossRef]
- Al-Toub, M.; Almohawes, M.; Vishnubalaji, R.; Alfayez, M.; Aldahmash, A.; Kassem, M.; Alajez, N.M. CXCR7 signaling promotes breast cancer survival in response to mesenchymal stromal stem cell-derived factors. Cell Death Discov. 2019, 5, 87. [Google Scholar] [CrossRef]
- Miao, Z.; Luker, K.E.; Summers, B.C.; Berahovich, R.; Bhojani, M.S.; Rehemtulla, A.; Kleer, C.G.; Essner, J.J.; Nasevicius, A.; Luker, G.D.; et al. CXCR7 (RDC1) promotes breast and lung tumor growth in vivo and is expressed on tumor-associated vasculature. Proc. Natl. Acad. Sci. USA 2007, 104, 15735–15740. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, N.; Soda, Y.; Kanbe, K.; Liu, H.; Mukai, R.; Kitamura, T.; Hoshino, H. A putative G protein-coupled receptor, RDC1, is a novel coreceptor for human and simian immunodeficiency viruses. J. Virol. 2000, 74, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.C.; Li, M.X.; Wang, X.Y.; Cheng, X.; Wang, Y.; Xiao, T.; Jolkkonen, J.; Zhao, C.S.; Zhao, S.S. Effects of CXCR7-neutralizing antibody on neurogenesis in the hippocampal dentate gyrus and cognitive function in the chronic phase of cerebal ischemia. Neural Regen. Res. 2020, 15, 1079–1085. [Google Scholar] [PubMed]
- Bao, J.; Zhu, J.; Luo, S.; Cheng, Y.; Zhou, S. CXCR7 suppression modulates microglial chemotaxis to ameliorate experimentally-induced autoimmune encephalomyelitis. Biochem. Biophys. Res. Commun. 2016, 469, 1–7. [Google Scholar] [CrossRef] [PubMed]
- De Klerck, B.; Geboes, L.; Hatse, S.; Kelchtermans, H.; Meyvis, Y.; Vermeire, K.; Bridger, G.; Billiau, A.; Schols, D.; Matthys, P. Pro-inflammatory properties of stromal cell-derived factor-1 (CXCL12) in collagen-induced arthritis. Arthritis Res. Ther. 2005, 7, 1208–1220. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Huang, P.H.; Syu, F.S.; Hsieh, C.H.; Chang, S.L.; Lu, J.; Chen, H.C. Critical involvement of atypical chemokine receptor CXCR7 in allergic airway inflammation. Immunology 2018, 154, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Ngamsri, K.C.; Müller, A.; Bösmüller, H.; Gamper-Tsigaras, J.; Reutershan, J.; Konrad, F.M. The pivotal role of CXCR7 in stabilization of the pulmonary epithelial barrier in acute pulmonary inflammation. J. Immunol. 2017, 198, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Tourniaire, F.; Romier-Crouzet, B.; Lee, J.H.; Marcotorchino, J.; Gouranton, E.; Salles, J.; Malezet, C.; Astier, J.; Darmon, P.; Blouin, E.; et al. Chemokine expression in inflamed adipose tissue is mainly mediated by NF-kappaB. PLoS ONE 2013, 8, e66515. [Google Scholar] [CrossRef]
- Peng, H.; Zhang, H.; Zhu, H. Blocking CXCR7-mediated adipose tissue macrophages chemotaxis attenuates insulin resistance and inflammation in obesity. Biochem. Biophys. Res. Commun. 2016, 479, 649–655. [Google Scholar] [CrossRef]
- Yates, T.J.; Knapp, J.; Gosalbez, M.; Lokeshwar, S.D.; Gomez, C.S.; Benitez, A.; Ekwenna, O.O.; Young, E.E.; Manoharan, M.; Lokeshwar, V.B. C-X-C chemokine receptor 7: A functionally associated molecular marker for bladder cancer. Cancer 2013, 119, 61–71. [Google Scholar] [CrossRef]
- Desnoyer, A.; Dupin, N.; Assoumou, L.; Carlotti, A.; Gaudin, F.; Deback, C.; Peytavin, G.; Marcelin, A.G.; Boué, F.; Balabanian, K.; et al. Expression pattern of the CXCL12/CXCR4-CXCR7 trio in Kaposi sarcoma skin lesions. Br. J. Dermatol. 2016, 175, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Schrevel, M.; Karim, R.; ter Haar, N.T.; van der Burg, S.H.; Trimbos, J.B.; Fleuren, G.J.; Gorter, A.; Jordanova, E.S. CXCR7 expression is associated with disease-free and disease-specific survival in cervical cancer patients. Br. J. Cancer 2012, 106, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Wang, F.; Cui, S.; Gao, Z.; Qu, X. CXCR7/CXCR4 heterodimer-induced histone demethylation: A new mechanism of colorectal tumorigenesis. Oncogene 2018, 38, 1560–1575. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Wang, W.; Yan, J.; Xiao, L.; Yang, L. Prognostic significance of CXCR7 in cancer patients: A meta-analysis. Cancer Cell Int. 2018, 18, 212. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhang, P.; Qin, G.; Ren, F.; Zheng, Y.; Qiao, Y.; Sun, T.; Zhang, Y. Role of CXCR7 as a common predictor for prognosis in solid tumors: A meta-analysis. J. Cancer 2018, 9, 3138–3148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Qin, W.; Zhao, C.; Long, J.; Li, M. CXCR7, a prognostic biomarker in cervical squamous cell carcinoma, may be a screening index for treatment options at stages IB1 and IIA1. Cancer Manag. Res. 2019, 11, 10287–10296. [Google Scholar] [CrossRef]
- Li, N.; Xu, H.; Ou, Y.; Feng, Z.; Zhang, Q.; Zhu, Q.; Cai, Z. LPS-induced CXCR7 expression promotes gastric cancer proliferation and migration via the TLR4/MD-2 pathway. Diagn. Pathol. 2019, 14, 3. [Google Scholar] [CrossRef]
- Kim, N.; Ryu, H.; Kim, S.; Joo, M.; Jeon, H.J.; Lee, M.W.; Song, I.C.; Kim, M.N.; Kim, J.M.; Lee, H.J. CCXR7 promotes migration and invasion in head and neck squamous cell carcinoma by upregulating TGF-β1/Smad2/3 signaling. Sci. Rep. 2019, 9, 18100. [Google Scholar] [CrossRef]
- Hui, Y.; Li, Y.; Jing, Y.; Feng, J.Q.; Ding, Y. miRNA-101 acts as a tumor suppressor in oral squamous cell carcinoma by targeting CX chemokine receptor 7. Am. J. Transl. Res. 2016, 8, 4902–4911. [Google Scholar]
- Zhang, S.; Hong, Z.; Li, Q.; Lei, J.; Huang, H.; Liu, Q. Effect of MicroRNA-218 on the viability, apoptosis and invasion of renal cell carcinoma cells under hypoxia by targeted downregulation of CXCR7 expression. Biomed. Pharmacother. 2016, 80, 213–219. [Google Scholar] [CrossRef]
- Zhou, S.M.; Zhang, F.; Chen, X.B.; Jun, C.M.; Jing, X.; Wei, D.X.; Xia, Y.; Zhou, Y.B.; Xiao, X.Q.; Jia, R.Q.; et al. miR-100 suppresses the proliferation and tumor growth of esophageal squamous cancer cells via targeting CXCR7. Oncol. Rep. 2016, 35, 3453–3459. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ye, Y.; Long, P.; Zhao, S.; Zhang, L.; A, Y. Silencing of CXCR4 and CXCR7 expression by RNA interference suppresses human endometrial carcinoma growth in vivo. Am. J. Transl. Res. 2017, 9, 1896–1904. [Google Scholar] [PubMed]
- Wu, K.; Cui, L.; Yang, Y.; Zhao, J.; Zhu, D.; Liu, D.; Zhang, C.; Qi, Y.; Li, X.; Li, W.; et al. Silencing of CXCR2 and CXCR7 protects against esophageal cancer. Am. J. Transl. Res. 2016, 8, 3398–3408. [Google Scholar] [PubMed]
- Liu, Y.; Carson-Walter, E.; Walter, K.A. Targeting chemokine receptor CXCR7 inhibits glioma cell proliferation and mobility. Anticancer Res. 2015, 35, 53–64. [Google Scholar] [PubMed]
- Salazar, N.; Carlson, J.C.; Huang, K.; Zheng, Y.; Oderup, C.; Gross, J.; Jang, A.D.; Burke, T.M.; Lewén, S.; Scholz, A.; et al. A chimeric antibody against ACKR3/CXCR7 in combination with TMZ activates immune responses and extends survival in mouse GBM models. Mol. Ther. 2018, 26, 1354–1365. [Google Scholar] [CrossRef] [PubMed]
- Maussang, D.; Mujic-Delic, A.; Descamps, F.J.; Stortelers, C.; Vanlandschoot, P.; Stigter-van Walsum, M.; Vischer, H.F.; van Roy, M.; Vosjan, M.; Gonzalez-Pajuelo, M.; et al. Llama-derived single variable domains (nanobodies) directed against chemokine receptor CXCR7 reduce head and neck cancer cell growth in vivo. J. Biol. Chem. 2013, 288, 29562–29572. [Google Scholar] [CrossRef]
- Mir, H.; Kapur, N.; Singh, R.; Sonpavde, G.; Lillard Jr, J.W.; Singh, S. Andrographolide inhibits prostate cancer by targeting cell cycle regulators, CXCR3 and CXCR7 chemokine receptors. Cell Cycle 2016, 15, 819–826. [Google Scholar] [CrossRef]
- Pluchino, N.; Mamillapalli, R.; Moridi, I.; Tal, R.; Taylor, H.S. G-Protein-coupled receptor CXCR7 is overexpressed in human and murine endometrosis. Reprod. Sci. 2018, 25, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Guan, S.; Zhou, J. CXCR7 attenuates the TFG-β-unduced endothelial-to-mesenchymal transition and pulmonary fibrosis. Mol. Biosyst. 2017, 13, 2116–2124. [Google Scholar] [CrossRef]
- Ma, W.; Liu, Y.; Ellison, N.; Shen, J. Induction of C-X-C chemokine receptor type 7 (CXCR7) switches stromal cell-derived factor-1 (SDF-1) signaling and phagocytic activity in macrophages linked to atherosclerosis. J. Biol. Chem. 2013, 288, 15481–15494. [Google Scholar] [CrossRef]
- Ma, W.; Liu, Y.; Wang, C.; Zhang, L.; Crocker, L.; Shen, J. Atorvastatin inhibits CXCR7 induction to reduce macrophage migration. Biochem. Pharmacol. 2014, 89, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Zhu, Z.; Li, D.; Xu, R.; Wang, T.; Liu, K. Pioglitazone suppresses CXCR7 expression to inhibit human macrophage chemotaxis through peroxisome proliferator-activated receptor γ. Biochemistry 2015, 54, 6806–6814. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Brown, M.; Sengupta, R.; Penfold, M.E.; Meucci, O. CXCR7 protein expression in human adult brain and differentiated neurons. PLoS ONE 2011, 6, e20680. [Google Scholar] [CrossRef] [PubMed]
- Walters, M.J.; Ebsworth, K.; Berahovich, R.D.; Penfold, M.E.T.; Liu, S.-C.; Al Omran, R.; Kioi, M.; Chernikova, S.B.; Tseng, D.; Mulkearns-Hubert, E.E.; et al. Inhibition of CXCR7 extends survival following irradiation of brain tumours in mice and rats. Br. J. Cancer 2014, 110, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhu, M.; Penfold, M.E.; Koenen, R.R.; Thiemann, A.; Heyll, K.; Akhtar, S.; Koyadan, S.; Wu, Z.; Gremse, F.; et al. Activation of CXCR7 limits atherosclerosis and improves hyperlipidemia by increasing cholesterol uptake in adipose tissue. Circulation 2014, 129, 1244–1253. [Google Scholar] [CrossRef] [PubMed]
- Pluchino, N.; Mamillapalli, R.; Shaikh, S.; Habata, S.; Tal, A.; Gaye, M.; Taylor, H.S. CXCR4 or CXCR7 antagonists treat endometriosis by reducing bone marrow cell trafficking. J. Cell. Mol. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zabel, B.A.; Wang, Y.; Lewen, S.; Berahovich, R.D.; Penfold, M.E.; Zhang, P.; Powers, J.; Summers, B.C.; Miao, Z.; Zhao, B.; et al. Elucidation of CXCR7-mediated signaling events and inhibition of CXCR4-mediated tumor cell transendothelial migration by CXCR7 ligands. J. Immunol. 2009, 183, 3204–3211. [Google Scholar] [CrossRef]
- Uto-Konomi, A.; McKibben, B.; Wirtz, J.; Sato, Y.; Takano, A.; Nanki, T.; Suzuki, S. CXCR7 agonists inhibit the function of CXCL12 by down-regulation of CXCR4. Biochem. Biophys. Res. Commun. 2013, 431, 772–776. [Google Scholar] [CrossRef]
- Gustavsson, M.; Wang, L.; van Gils, N.; Stephens, B.S.; Zhang, P.; Schall, T.J.; Yang, S.; Abagyan, R.; Chance, M.R.; Kufareva, I.; et al. Structural basis of ligand interaction with atypical chemokine receptor 3. Nat. Commun. 2017, 8, 14135. [Google Scholar] [CrossRef]
- Witjmans, M.; Maussang, D.; Sirci, F.; Scholten, D.J.; Canals, M.; Mujić-Delić, A.; Chong, M.; Chatalic, K.L.S.; Custers, H.; Janssen, E.; et al. Synthesis, modeling and functional activity of substituted styrene-amides as small-molecule CXCR7 agonists. Eur. J. Med. Chem. 2012, 51, 184–192. [Google Scholar]
- Chen, X.; Fan, P.; Gleason, M.M.; Jean, J.C.; Li, L.; McMahon, J.P.; Powers, J.; Zeng, Y.; Zhang, P. Diazepane Derivatives as CXCR7 Modulators and Their Preparation, Pharmaceutical Compositions and Use in the Treatment of Diseases. Patent WO2010054006A1, 14 May 2010. [Google Scholar]
- Menhaji-Klotz, E.; Hesp, K.D.; Londregan, A.T.; Kalgutkar, A.S.; Piotrowski, D.W.; Boehm, M.; Song, K.; Ryder, T.; Beaumont, K.; Jones, R.M.; et al. Discovery of a novel small-molecule modulator of C-X-C chemokine receptor type 7 as a treatment for cardiac fibrosis. J. Med. Chem. 2018, 61, 3685–3696. [Google Scholar] [CrossRef] [PubMed]
- Redjal, N.; Chan, J.A.; Segal, R.A.; Kung, A.L. CXCR4 inhibition synergizes with cytotoxic chemotherapy in gliomas. Clin. Cancer Res. 2006, 12, 6765–6771. [Google Scholar] [CrossRef] [PubMed]
- Kalatskaya, I.; Berchiche, Y.A.; Gravel, S.; Limberg, B.J.; Rosenbaum, J.S.; Heveker, N. AMD3100 is a CXCR7 ligand with allosteric agonist properties. Mol. Pharmacol. 2009, 75, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, Y.; Oishi, S.; Kubo, T.; Tanahara, N.; Fujii, N.; Furuya, T. Optimized method of G-protein-coupled receptor homology modeling: Its application to the discovery of novel CXCR7 ligands. J. Med. Chem. 2013, 56, 4236–4251. [Google Scholar] [CrossRef] [PubMed]
- Oishi, S.; Kuroyanagi, T.; Kubo, T.; Montpas, N.; Yoshikawa, Y.; Misu, R.; Kobayashi, Y.; Ohno, H.; Heveker, N.; Furuya, T.; et al. Development of novel CXC chemokine receptor 7 (CXCR7) ligands: Selectivity switch from CXCR4 antagonists with a cyclic pentapeptide scaffold. J. Med. Chem. 2015, 58, 5218–5225. [Google Scholar] [CrossRef]
- Fujii, N.; Oishi, S.; Hiramatsu, K.; Araki, T.; Ueda, S.; Tamamura, H.; Otaka, A.; Kusano, S.; Terakubo, S.; Nakashima, H.; et al. Molecular-size reduction of a potent CXCR4-chemokine antagonist using orthogonal combination of conformation- and sequence-based libraries. Angew. Chem. Int. Ed. 2003, 42, 3251–3253. [Google Scholar] [CrossRef]
- Sekiguchi, H.; Kuroyanagi, T.; Rhainds, D.; Kobayashi, K.; Kobayashi, Y.; Ohno, H.; Heveker, N.; Akaji, K.; Fujii, N.; Oishi, S. Structure-activity relationship study of cyclic pentapeptide ligands for atypical chemokine receptor 3 (ACKR3). J. Med. Chem. 2018, 61, 3745–3751. [Google Scholar] [CrossRef]
- Al-Awadhi, F.H.; Gao, B.; Rezaei, M.A.; Kwan, J.C.; Li, C.; Ye, T.; Paul, V.J.; Luesch, H. Discovery, synthesis, pharmacological profiling, and biological characterization of brintonamides A-E, novel dual protease and GPCR modulators from a marine cyanobacterium. J. Med. Chem. 2018, 61, 6364–6378. [Google Scholar] [CrossRef]
- Ehrlich, A.; Ray, P.; Luker, K.E.; Lolis, E.J.; Luker, G.D. Allosteric peptide regulators of chemokine receptors CXCR4 and CXCR7. Biochem. Pharmacol. 2013, 86, 1263–1271. [Google Scholar] [CrossRef][Green Version]
- Boehm, M.; Beaumont, K.; Jones, R.; Kalgutkar, A.S.; Zhang, L.; Atkinson, K.; Bai, G.; Brown, J.A.; Eng, H.; Goetz, G.H.; et al. Discovery of potent and orally bioavailable macrocyclic peptide-peptoid hybrid CXCR7 modulators. J. Med. Chem. 2017, 60, 9653–9663. [Google Scholar] [CrossRef] [PubMed]
- Gombert, F.O.; Lederer, A.; Obrecht, D.; Romagnoli, B.; Bisang, C.; Ludin, C. Template-Fixed Peptidomimetics with CXCR7 Modulating Activity. Patent WO2011095220, 8 November 2011. [Google Scholar]
- Gravel, S.; Malouf, C.; Boulais, P.E.; Berchiche, Y.A.; Oishi, S.; Fujii, N.; Leduc, R.; Sinnett, D.; Heveker, N. The peptidomimetic CXCR4 antagonist TC14012 recruits β-arrestin to CXCR7: Roles of receptor domains. J. Biol. Chem. 2010, 285, 37939–37943. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.; Hartmann, T.; Krome, M.; Rawluk, J.; Tamamura, H.; Fujii, N.; Kipps, T.J.; Burger, J.A. Small peptide inhibitors of the CXCR4 chemokine receptor (CD184) antagonize the activation, migration, and antiapoptotic responses of CXCL12 in chronic lymphocytic leukemia B cells. Blood 2005, 106, 1824–1830. [Google Scholar] [CrossRef] [PubMed]
- Montpas, N.; Cabana, J.; St-Onge, G.; Gravel, S.; Morin, G.; Kuroyanagi, T.; Lavigne, P.; Fujii, N.; Oishi, S.; Heveker, N. Mode of binding of the cyclic agonist peptide TC14012 to CXCR7: Identification of receptor and compound determinants. Biochemistry 2015, 54, 1505–1515. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Lis, R.; Ginsberg, M.; Chavez, D.; Shido, K.; Rabbany, S.Y.; Fong, G.; Sakmar, T.P.; Rafii, S.; Ding, B. Targeting of the pulmonary capillary vascular niche promotes lung alveolar repair and ameliorates fibrosis. Nat. Med. 2016, 22, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Dai, X.; Chen, J.; Epstein, P.N.; Cai, L.; Wintergerst, K.A.; Qian, Y.; Tan, Y. CXCR7 agonist TC14012 improves angiogenic function of endothelial progenitor cells in diabetic limb ischemia. Diabetes 2018, 67 (Suppl. 1). [Google Scholar] [CrossRef]
- Melikian, A.; Burns, J.; Mcmaster, B.E.; Schall, T.; Wright, J.J. Inhibitors of Human Tumor-Expressed CCXCKR2. Patent WO2004058705, 15 July 2004. [Google Scholar]
- Burns, J.; Summers, B.; Wang, Y.; Howard, M.; Schall, T.; Miao, Z. Methods and Compositions for Modulating Angiogenesis. Patent WO2005074645, 18 August 2005. [Google Scholar]
- Melikian, A.; Wright, J.J.K. Substituted Arylamides. U.S. Patent 20060074071, 6 April 2006. [Google Scholar]
- Leleti, M.R.; Thomas, W.D.; Zhang, P.; Pennell, A.M.K. CXCR4 Modulators. Patent WO2007115232, 11 October 2007. [Google Scholar]
- Melikian, A.; Wright, J.J.; Krasinski, A.; Hu, C.; Novack, A. Substituted Quinolones and Methods of Use. Patent WO2007059108, 24 May 2007. [Google Scholar]
- Melikian, A.; Wright, J.J.K.; Krasinski, A. Substituted N-Cinnamyl Benzamides. Patent WO2007002842, 4 January 2007. [Google Scholar]
- Thomas, W.D.; Leleti, M.R.; Pennell, A.M.K. CXCR4 Modulators. Patent WO2007115231, 11 October 2007. [Google Scholar]
- Clark, M.P.; Lockwood, M.A.; Wagner, F.F.; Natchus, M.G.; Doroh, B.C. Chemokine Receptor Modulators. Patent WO2008112156, 18 September 2008. [Google Scholar]
- Fretz, H.; Guerry, P.; Kimmerlin, T.; Lehembre, F.; Pothier, J.; Siendt, H.; Valdenaire, A. CXCR7 Receptor Modulators. Patent WO2014191929, 4 December 2014. [Google Scholar]
- Fan, J.; Krasinski, A.; Lange, C.W.; Lui, R.M.; McMahon, J.P.; Powers, J.P.; Zeng, Y.; Zhang, P. CXCR7 Antagonists. Patent WO2014085490, 5 June 2014. [Google Scholar]
- Chen, X.; Fan, P.; Gleason, M.M.; Jaen, J.C.; Li, L.; McMahon, J.P.; Powers, J.; Zeng, Y. Modulators of CXCR7. Patent WO2010054006, 14 May 2010. [Google Scholar]
- Gombert, F.O.; Lederer, A.; Löwe, R.; Obrecht, D.; Romagnoli, B.; Zimmermann, J.; Patel, K. Template-Fixed Peptidomimetics with CXCR7 Modulating Activity. Patent WO2011095607, 11 August 2011. [Google Scholar]
- Gombert, F.O.; Lederer, A.; Obrecht, D.; Romagnoli, B.; Löwe, R.; Zimmermann, J. Template-Fixed Peptidomimetics with CXCR7 Modulating Activity. Patent WO2011095218, 11 August 2011. [Google Scholar]
- Fretz, H.; Gude, M.; Guerry, P.; Kimmerlin, T.; Lehembre, F.; Pfeifer, T.; Valdenaire, A. 1-[M-carboxamido(hetero)aryl-methyl]-piperidine-4-carboxamide Derivatives. U.S. Patent 20130345199, 26 December 2013. [Google Scholar]
- Muller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001, 410, 50–56. [Google Scholar] [CrossRef]
- Kijima, T.; Maulik, G.; Ma, P.C.; Tibaldi, E.V.; Turner, R.E.; Rollins, B.; Sattler, M.; Johnson, B.E.; Salgia, R. Regulation of cellular proliferation, cytoskeletal function, and signal transduction through CXCR4 and c-Kit in small cell lung cancer cells. Cancer Res. 2002, 62, 6304–6311. [Google Scholar]
- Nanki, T.; Hayashida, K.; El-Gabalawy, H.S.; Suson, S.; Shi, K.; Girschick, H.J.; Yavuz, S.; Lipsky, P.E. Stromal cell-derived factor-1-CXC chemokine receptor 4 interactions play a central role in CD4+ T cell accumulation in rheumatoid arthritis synovium. J. Immunol. 2000, 165, 6590–6598. [Google Scholar] [CrossRef]
- Grassi, F.; Cristino, S.; Toneguzzi, S.; Piacentini, A.; Facchini, A.; Lisignoli, G. CXCL12 chemokine up-regulates bone resorption and MMP-9 release by human osteoclasts: CXCL12 levels are increased in synovial and bone tissue of rheumatoid arthritis patients. J. Cell. Physiol. 2004, 199, 244–251. [Google Scholar] [CrossRef]
- Gu, X.L.; Liu, L.; Lu, X.D.; Liu, Z.R. Serum CXCL12 levels as a novel predictor of future stroke recurrence in patients with acute ischemic stroke. Mol. Neurobiol. 2016, 53, 2807–2814. [Google Scholar] [CrossRef]
- Robin, A.M.; Zhang, Z.G.; Wang, L.; Zhang, R.L.; Katakowski, M.; Zhang, L.; Wang, Y.; Zhang, C.; Chopp, M. Stromal cell-derived factor 1alpha mediates neural progenitor cell motility after focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2006, 26, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Krumbholz, M.; Theil, D.; Cepok, S.; Hemmer, B.; Kivisäkk, P.; Ransohoff, R.M.; Hofbauer, M.; Farina, C.; Derfuss, T.; Hartle, C.; et al. Chemokines in multiple sclerosis: CXCL12 and CXCL13 up-regulation is differentially linked to CNS immune cell recruitment. Brain 2006, 129, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.J.; Wu, W.Q.; Ying, G.R.; Fu, Q.Y.; Xiong, K. Serum CXCL12 concentration in patients with severe traumatic brain injury are associated with mortality. Clin. Chim. Acta 2016, 454, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Liu, N.; Yi, B.; Zhang, X.; Gao, B.B.; Zhang, Y.; Xu, R.; Li, X.; Dai, Y. Transplanted hUCB-MSCs migrated to the damaged area by SDF-1/CXCR4 signaling to promote functional recovery after traumatic brain injury in rats. Neurol. Res. 2015, 37, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Liu, J.; Huan, Y.; Zhang, C. Intracranial injection of recombinant stromal-derived factor-1 alpha (SDF-1α) attenuates traumatic brain injury in rats. Inflamm. Res. 2014, 63, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Laske, C.; Stellos, K.; Eschweiler, G.W.; Leyhe, T.; Gawaz, M. Decreased CXCL12 (SDF-1) plasma levels in early Alzheimer’s disease: A contribution to a deficient hematopoietic brain support? J. Alzheimers Dis. 2008, 15, 451–460. [Google Scholar] [CrossRef]
- Zabel, B.A.; Lewen, S.; Berahovich, R.D.; Jaen, J.C.; Schall, T.J. The novel chemokine receptor CXCR7 regulates trans-endothelial migration of cancer cells. Mol. Cancer 2011, 10, 73. [Google Scholar] [CrossRef]
- Zhong, C.; Wang, J.; Li, B.; Xiang, H.; Ultsch, M.; Coons, M.; Wong, T.; Chiang, N.Y.; Clark, S.; Clark, R.; et al. Development and preclinical characterization of a humanized antibody targeting CXCL12. Clin. Cancer Res. 2013, 19, 4433–4445. [Google Scholar] [CrossRef]
- Regenass, P.; Abboud, D.; Daubeuf, F.; Lehalle, C.; Gizzi, P.; Riché, S.; Hachet-Haas, M.; Rohmer, F.; Gasparik, V.; Boeglin, D.; et al. Discovery of a locally and orally active CXCL12 neutraligand (LIT-927) with anti-inflammatory effect in a murine model of allergic airway hypereosinophilia. J. Med. Chem. 2018, 61, 7671–7686. [Google Scholar] [CrossRef]
- Berahovich, R.D.; Penfold, M.E.; Schall, T.J. Nonspecific CXCR7 antibodies. Immunol. Lett. 2010, 133, 112–114. [Google Scholar] [CrossRef]
- Hartmann, T.N.; Grabovsky, V.; Pasvolsky, R.; Shulman, Z.; Buss, E.C.; Spiegel, A.; Nagler, A.; Lapidot, T.; Thelen, M.; Alon, R. A crosstalk between intracellular CXCR7 and CXCR4 involved in rapid CXCL12-triggered integrin activation but not in chemokine-triggered motility of human T lymphocytes and CD34+ cells. J. Leukoc. Biol. 2008, 84, 1130–1140. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Carson-Walter, E.B.; Cooper, A.; Winans, B.N.; Johnson, M.D.; Walter, K.A. Vascular gene expression patters are conserved in primary and metastatic brain tumors. J. Neurooncol. 2010, 99, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Salmaggi, A.; Maderna, E.; Calatozzolo, C.; Gaviani, P.; Canazza, A.; Milanesi, I.; Silvani, A.; DiMeco, F.; Carbone, A.; Pollo, B. CXCL12, CXCR4 and CXCR7 expression in brain metastases. Cancer Biol. Ther. 2009, 8, 1608–1614. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lounsbury, N. Advances in CXCR7 Modulators. Pharmaceuticals 2020, 13, 33. https://doi.org/10.3390/ph13020033
Lounsbury N. Advances in CXCR7 Modulators. Pharmaceuticals. 2020; 13(2):33. https://doi.org/10.3390/ph13020033
Chicago/Turabian StyleLounsbury, Nicole. 2020. "Advances in CXCR7 Modulators" Pharmaceuticals 13, no. 2: 33. https://doi.org/10.3390/ph13020033
APA StyleLounsbury, N. (2020). Advances in CXCR7 Modulators. Pharmaceuticals, 13(2), 33. https://doi.org/10.3390/ph13020033



