Matrix Metalloproteinase in Abdominal Aortic Aneurysm and Aortic Dissection
Abstract
1. Introduction
2. Epidemiology of AAA and AD
3. Cellular and Molecular Pathophysiology of AAA and AD
4. MMP and TIMPs Overview
5. Clinical and GWAS Studies: MMPs in AAA and AD
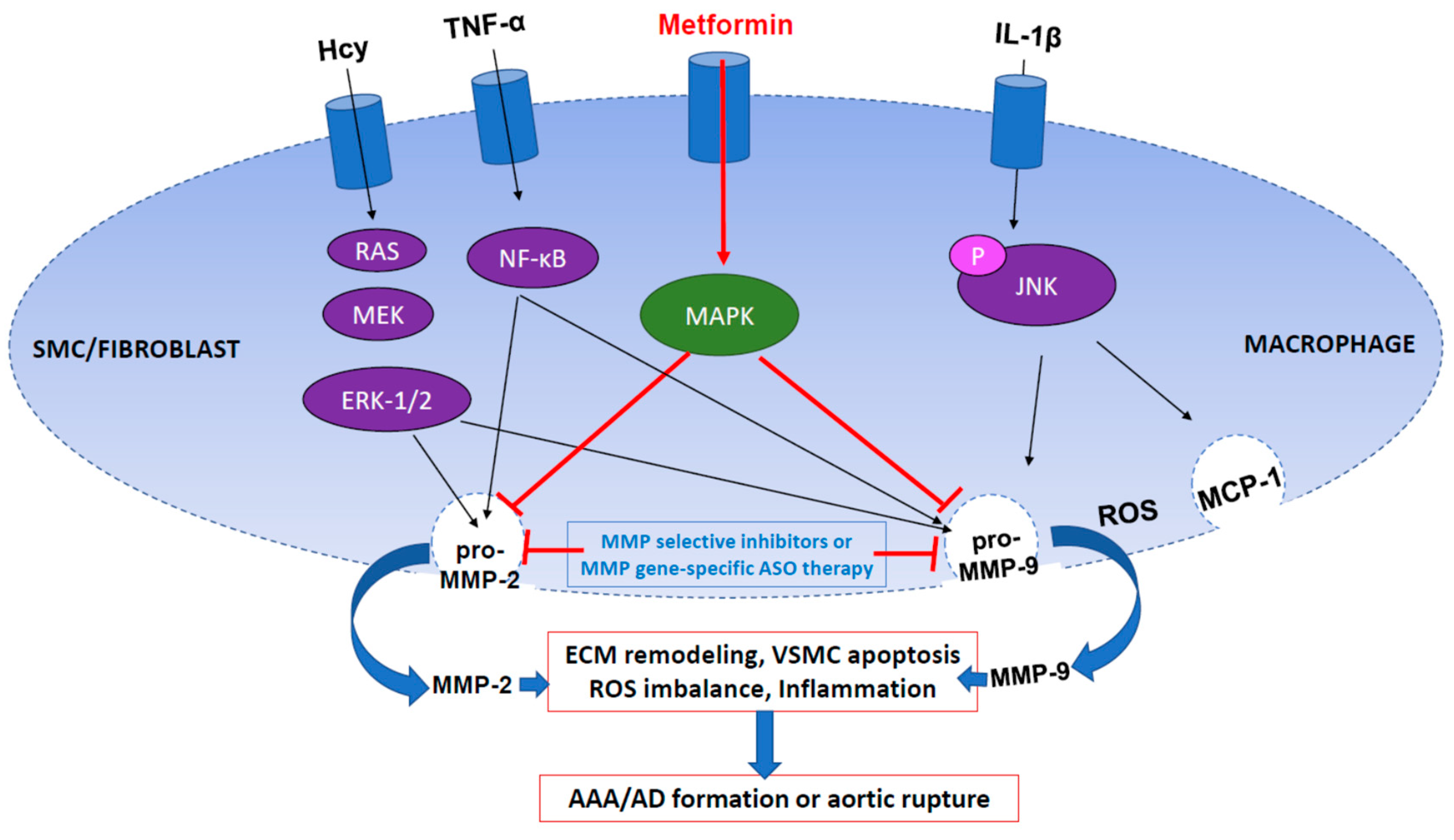
6. MMP2 and MMP9 in AAA and AD
7. Other MMPs or TIMPS in AAA and AD
8. Potential Applications of MMP Inhibition
9. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Halpern, V.J.; Kline, R.G.; D’Angelo, A.J.; Cohen, J.R. Factors that affect the survival rate of patients with ruptured abdominal aortic aneurysms. J. Vasc. Surg. 1997, 26, 939–945. [Google Scholar] [CrossRef]
- Goossens, D.; Schepens, M.; Hamerlijnck, R.; Hartman, M.; Suttorp, M.J.; Koomen, E.; Vermeulen, F. Predictors of hospital mortality in type A aortic dissections: A retrospective analysis of 148 consecutive surgical patients. Cardiovasc. Surg. 1998, 6, 76–80. [Google Scholar] [CrossRef]
- Juang, D.; Braverman Alan, C.; Eagle, K. Aortic Dissection. Circulation 2008, 118, e507–e510. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Qamar, A.; Sharma, V.; Sharma, A. Abdominal aortic aneurysm: A comprehensive review. Exp. Clin. Cardiol. 2011, 16, 11–15. [Google Scholar] [PubMed]
- Rabkin, S.W. The Role Matrix Metalloproteinases in the Production of Aortic Aneurysm. Prog. Mol. Biol. Transl. Sci. 2017, 147, 239–265. [Google Scholar] [CrossRef]
- Alcorn, H.G.; Wolfson, S.K., Jr.; Sutton-Tyrrell, K.; Kuller, L.H.; O’Leary, D. Risk factors for abdominal aortic aneurysms in older adults enrolled in The Cardiovascular Health Study. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 963–970. [Google Scholar] [CrossRef]
- Von Allmen, R.S.; Powell, J.T. The management of ruptured abdominal aortic aneurysms: Screening for abdominal aortic aneurysm and incidence of rupture. J. Cardiovasc. Surg. 2012, 53, 69–76. [Google Scholar]
- Simoni, G.; Pastorino, C.; Perrone, R.; Ardia, A.; Gianrossi, R.; De Cian, F.; Cittadini, G., Jr.; Baiardi, A.; Bachi, V. Screening for abdominal aortic aneurysms and associated risk factors in a general population. Eur. J. Vasc. Endovasc. Surg. 1995, 10, 207–210. [Google Scholar] [CrossRef]
- Guirguis, E.M.; Barber, G.G. The natural history of abdominal aortic aneurysms. Am. J. Surg. 1991, 162, 481–483. [Google Scholar] [CrossRef]
- Signorelli, M.; Tosini, S.; Gaeta, A.; De Nale, A.; Malacrida, G.; Tealdi, D.G. Aneurysms of the abdominal aorta in elderly patients. Minerva Chir. 1991, 46, 947–951. [Google Scholar]
- Jamrozik, K.; Norman, P.E.; Spencer, C.A.; Parsons, R.W.; Tuohy, R.; Lawrence-Brown, M.M.; Dickinson, J.A. Screening for abdominal aortic aneurysm: Lessons from a population-based study. Med. J. Aust. 2000, 173, 345–350. [Google Scholar] [PubMed]
- Lederle, F.A.; Johnson, G.R.; Wilson, S.E.; Chute, E.P.; Littooy, F.N.; Bandyk, D.; Krupski, W.C.; Barone, G.W.; Acher, C.W.; Ballard, D.J. Prevalence and associations of abdominal aortic aneurysm detected through screening. Aneurysm Detection and Management (ADAM) Veterans Affairs Cooperative Study Group. Ann. Intern. Med. 1997, 126, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Bonaa, K.H.; Jacobsen, B.K.; Bjork, L.; Solberg, S. Prevalence of and risk factors for abdominal aortic aneurysms in a population-based study: The Tromso Study. Am. J. Epidemiol. 2001, 154, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Buck, D.B.; van Herwaarden, J.A.; Schermerhorn, M.L.; Moll, F.L. Endovascular treatment of abdominal aortic aneurysms. Nat. Rev. Cardiol. 2014, 11, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Kanagasabay, R.; Gajraj, H.; Pointon, L.; Scott, R.A. Co-morbidity in patients with abdominal aortic aneurysm. J. Med. Screen. 1996, 3, 208–210. [Google Scholar] [CrossRef] [PubMed]
- Pleumeekers, H.J.; Hoes, A.W.; van der Does, E.; van Urk, H.; Hofman, A.; de Jong, P.T.; Grobbee, D.E. Aneurysms of the abdominal aorta in older adults. The Rotterdam Study. Am. J. Epidemiol. 1995, 142, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Moll, F.L.; Powell, J.T.; Fraedrich, G.; Verzini, F.; Haulon, S.; Waltham, M.; van Herwaarden, J.A.; Holt, P.J.; van Keulen, J.W.; Rantner, B.; et al. Management of abdominal aortic aneurysms clinical practice guidelines of the European society for vascular surgery. Eur. J. Vasc. Endovasc. Surg. 2011, 41 (Suppl. 1), S1–S58. [Google Scholar] [CrossRef] [PubMed]
- Karthikesalingam, A.; Holt, P.J.; Vidal-Diez, A.; Ozdemir, B.A.; Poloniecki, J.D.; Hinchliffe, R.J.; Thompson, M.M. Mortality from ruptured abdominal aortic aneurysms: Clinical lessons from a comparison of outcomes in England and the USA. Lancet 2014, 383, 963–969. [Google Scholar] [CrossRef]
- Jacomelli, J.; Summers, L.; Stevenson, A.; Lees, T.; Earnshaw, J.J. Update on the prevention of death from ruptured abdominal aortic aneurysm. J. Med. Screen. 2017, 24, 166–168. [Google Scholar] [CrossRef]
- Roy, J.; Labruto, F.; Beckman, M.O.; Danielson, J.; Johansson, G.; Swedenborg, J. Bleeding into the intraluminal thrombus in abdominal aortic aneurysms is associated with rupture. J. Vasc. Surg. 2008, 48, 1108–1113. [Google Scholar] [CrossRef]
- Sakamoto, A.; Nagai, R.; Saito, K.; Imai, Y.; Takahashi, M.; Hosoya, Y.; Takeda, N.; Hirano, K.; Koike, K.; Enomoto, Y.; et al. Idiopathic retroperitoneal fibrosis, inflammatory aortic aneurysm, and inflammatory pericarditis--retrospective analysis of 11 case histories. J. Cardiol. 2012, 59, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Chaikof, E.L.; Dalman, R.L.; Eskandari, M.K.; Jackson, B.M.; Lee, W.A.; Mansour, M.A.; Mastracci, T.M.; Mell, M.; Murad, M.H.; Nguyen, L.L.; et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J. Vasc. Surg. 2018, 67, 2–77. [Google Scholar] [CrossRef] [PubMed]
- Filardo, G.; Powell, J.T.; Martinez, M.A.-M.; Ballard, D.J. Surgery for small asymptomatic abdominal aortic aneurysms. Cochrane Database Syst. Rev. 2012, 3, CD001835. [Google Scholar] [CrossRef]
- Sweeting, M.J.; Thompson, S.G.; Brown, L.C.; Powell, J.T. Meta-analysis of individual patient data to examine factors affecting growth and rupture of small abdominal aortic aneurysms. Br. J. Surg. 2012, 99, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Wiersema, A.M.; Jongkind, V.; Bruijninckx, C.M.; Reijnen, M.M.; Vos, J.A.; van Delden, O.M.; Zeebregts, C.J.; Moll, F.L. Prophylactic perioperative anti-thrombotics in open and endovascular abdominal aortic aneurysm (AAA) surgery: A systematic review. Eur. J. Vasc. Endovasc. Surg. 2012, 44, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Wanhainen, A.; Verzini, F.; Van Herzeele, I.; Allaire, E.; Bown, M.; Cohnert, T.; Dick, F.; van Herwaarden, J.; Karkos, C.; Koelemay, M.; et al. Editor’s Choice – European Society for Vascular Surgery (ESVS) 2019 Clinical Practice Guidelines on the Management of Abdominal Aorto-iliac Artery Aneurysms. Eur. J. Vasc. Endovasc. Surg. 2019, 57, 8–93. [Google Scholar] [CrossRef] [PubMed]
- Ricco, J.B.; Cau, J.; Biancari, F.; Desvergnes, M.; Lefort, N.; Belmonte, R.; Schneider, F. Outcome After Open and Laparoscopic Aortic Surgery in Matched Cohorts Using Propensity Score Matching. Eur. J. Vasc. Endovasc. Surg. 2016, 52, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Curci, J.A.; Petrinec, D.; Liao, S.; Golub, L.M.; Thompson, R.W. Pharmacologic suppression of experimental abdominal aortic aneurysms: Acomparison of doxycycline and four chemically modified tetracyclines. J. Vasc. Surg. 1998, 28, 1082–1093. [Google Scholar] [CrossRef]
- Meijer, C.A.; Stijnen, T.; Wasser, M.N.; Hamming, J.F.; van Bockel, J.H.; Lindeman, J.H. Doxycycline for stabilization of abdominal aortic aneurysms: A randomized trial. Ann. Intern. Med. 2013, 159, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Kurosawa, K.; Matsumura, J.S.; Yamanouchi, D. Current status of medical treatment for abdominal aortic aneurysm. Circ. J. 2013, 77, 2860–2866. [Google Scholar] [CrossRef] [PubMed]
- Hackam, D.G.; Thiruchelvam, D.; Redelmeier, D.A. Angiotensin-converting enzyme inhibitors and aortic rupture: A population-based case-control study. Lancet 2006, 368, 659–665. [Google Scholar] [CrossRef]
- Sweeting, M.J.; Thompson, S.G.; Brown, L.C.; Greenhalgh, R.M.; Powell, J.T. Use of angiotensin converting enzyme inhibitors is associated with increased growth rate of abdominal aortic aneurysms. J. Vasc. Surg. 2010, 52, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Lysgaard Poulsen, J.; Stubbe, J.; Lindholt, J.S. Animal Models Used to Explore Abdominal Aortic Aneurysms: A Systematic Review. Eur. J. Vasc. Endovasc. Surg. 2016, 52, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Powell, J.T.; Muller, B.R.; Greenhalgh, R.M. Acute phase proteins in patients with abdominal aortic aneurysms. J. Cardiovasc. Surg. 1987, 28, 528–530. [Google Scholar]
- Vainas, T.; Lubbers, T.; Stassen, F.R.; Herngreen, S.B.; van Dieijen-Visser, M.P.; Bruggeman, C.A.; Kitslaar, P.J.; Schurink, G.W. Serum C-reactive protein level is associated with abdominal aortic aneurysm size and may be produced by aneurysmal tissue. Circulation 2003, 107, 1103–1105. [Google Scholar] [CrossRef] [PubMed]
- Dale, M.A.; Ruhlman, M.K.; Baxter, B.T. Inflammatory cell phenotypes in AAAs: Their role and potential as targets for therapy. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1746–1755. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Zhou, X.L.; Li, J.J.; Hui, R.T. Inflammatory response is associated with aortic dissection. Ageing Res. Rev. 2009, 8, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Tambyraja, A.L.; Dawson, R.; Valenti, D.; Murie, J.A.; Chalmers, R.T. Systemic inflammation and repair of abdominal aortic aneurysm. World J. Surg. 2007, 31, 1210–1214. [Google Scholar] [CrossRef] [PubMed]
- Berge, C.; Hagen, A.I.; Myhre, H.O.; Dahl, T. Preoperative White Blood Cell Count in Patients with Abdominal Aortic Aneurysms and Its Relation to Survival following Surgery. Ann. Vasc. Surg. 2017, 41, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.I.; Bloor, K.; Williams, G.; Gillie, I. Inflammatory aneurysms of the abdominal aorta. Br. J. Surg. 1972, 59, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, D.B.; Grand, D.J.; Freischlag, J.A. Inflammatory abdominal aortic aneurysm. JAMA 2007, 297, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Eliason, J.L.; Hannawa, K.K.; Ailawadi, G.; Sinha, I.; Ford, J.W.; Deogracias, M.P.; Roelofs, K.J.; Woodrum, D.T.; Ennis, T.L.; Henke, P.K.; et al. Neutrophil depletion inhibits experimental abdominal aortic aneurysm formation. Circulation 2005, 112, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.R.; Parikh, S.; Grella, L.; Sarfati, I.; Corbie, G.; Danna, D.; Wise, L. Role of the neutrophil in abdominal aortic aneurysm development. Cardiovasc. Surg. 1993, 1, 373–376. [Google Scholar] [PubMed]
- Meher, A.K.; Spinosa, M.; Davis, J.P.; Pope, N.; Laubach, V.E.; Su, G.; Serbulea, V.; Leitinger, N.; Ailawadi, G.; Upchurch, G.R., Jr. Novel Role of IL (Interleukin)-1beta in Neutrophil Extracellular Trap Formation and Abdominal Aortic Aneurysms. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ruiz, I. NETs are involved in AAA. Nat. Rev. Cardiol. 2018, 15, 257. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, T.; Shimizu-Hirota, R.; Shimoda, M.; Adachi, T.; Shimizu, H.; Weiss Stephen, J.; Itoh, H.; Hori, S.; Aikawa, N.; Okada, Y. Neutrophil-Derived Matrix Metalloproteinase 9 Triggers Acute Aortic Dissection. Circulation 2012, 126, 3070–3080. [Google Scholar] [CrossRef] [PubMed]
- Raffort, J.; Lareyre, F.; Clément, M.; Hassen-Khodja, R.; Chinetti, G.; Mallat, Z. Monocytes and macrophages in abdominal aortic aneurysm. Nat. Rev. Cardiol. 2017, 14, 457. [Google Scholar] [CrossRef] [PubMed]
- Juvonen, J.; Surcel, H.M.; Satta, J.; Teppo, A.M.; Bloigu, A.; Syrjala, H.; Airaksinen, J.; Leinonen, M.; Saikku, P.; Juvonen, T. Elevated circulating levels of inflammatory cytokines in patients with abdominal aortic aneurysm. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2843–2847. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, L.; Bergqvist, D.; Lindback, J.; Parsson, H. Expansion of small-diameter abdominal aortic aneurysms is not reflected by the release of inflammatory mediators IL-6, MMP-9 and CRP in plasma. Eur. J. Vasc. Endovasc. Surg. 2009, 37, 420–424. [Google Scholar] [CrossRef]
- Boytard, L.; Spear, R.; Chinetti-Gbaguidi, G.; Acosta-Martin, A.E.; Vanhoutte, J.; Lamblin, N.; Staels, B.; Amouyel, P.; Haulon, S.; Pinet, F. Role of proinflammatory CD68(+) mannose receptor(-) macrophages in peroxiredoxin-1 expression and in abdominal aortic aneurysms in humans. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 431–438. [Google Scholar] [CrossRef]
- Hasan, D.; Chalouhi, N.; Jabbour, P.; Hashimoto, T. Macrophage imbalance (M1 vs. M2) and upregulation of mast cells in wall of ruptured human cerebral aneurysms: Preliminary results. J. Neuroinflamm. 2012, 9, 222. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Zhao, Y.; Prall, A.; Greiner, T.C.; Baxter, B.T. Key roles of CD4+ T cells and IFN-gamma in the development of abdominal aortic aneurysms in a murine model. J. Immunol. 2004, 172, 2607–2612. [Google Scholar] [CrossRef] [PubMed]
- King Victoria, L.; Lin Alexander, Y.; Kristo, F.; Anderson Thomas, J.T.; Ahluwalia, N.; Hardy Gregory, J.; Owens, A.P.; Howatt Deborah, A.; Shen, D.; Tager Andrew, M.; et al. Interferon-γ and the Interferon-Inducible Chemokine CXCL10 Protect Against Aneurysm Formation and Rupture. Circulation 2009, 119, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Shichiri, M.; Libby, P.; Lee, R.T.; Mitchell, R.N. Th2-predominant inflammation and blockade of IFN-gamma signaling induce aneurysms in allografted aortas. J. Clin. Investig. 2004, 114, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Schönbeck, U.; Sukhova, G.K.; Gerdes, N.; Libby, P. T(H)2 predominant immune responses prevail in human abdominal aortic aneurysm. Am. J. Pathol. 2002, 161, 499–506. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y. B lymphocytes in abdominal aortic aneurysms. Atherosclerosis 2015, 242, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Furusho, A.; Aoki, H.; Ohno-Urabe, S.; Nishihara, M.; Hirakata, S.; Nishida, N.; Ito, S.; Hayashi, M.; Imaizumi, T.; Hiromatsu, S.; et al. Involvement of B Cells, Immunoglobulins, and Syk in the Pathogenesis of Abdominal Aortic Aneurysm. J. Am. Heart Assoc. 2018, 7, e007750. [Google Scholar] [CrossRef]
- Dong, Y.-Q.; Lu, C.-W.; Zhang, L.; Yang, J.; Hameed, W.; Chen, W. Toll-like receptor 4 signaling promotes invasion of hepatocellular carcinoma cells through MKK4/JNK pathway. Mol. Immunol. 2015, 68, 671–683. [Google Scholar] [CrossRef]
- Lai, C.-H.; Wang, K.-C.; Lee, F.-T.; Tsai, H.-W.; Ma, C.-Y.; Cheng, T.-L.; Chang, B.-I.; Yang, Y.-J.; Shi, G.-Y.; Wu, H.-L. Toll-Like Receptor 4 Is Essential in the Development of Abdominal Aortic Aneurysm. PLoS ONE 2016, 11, e0146565. [Google Scholar] [CrossRef]
- Qin, Z.; Bagley, J.; Sukhova, G.; Baur, W.E.; Park, H.-J.; Beasley, D.; Libby, P.; Zhang, Y.; Galper, J.B. Angiotensin II-induced TLR4 mediated abdominal aortic aneurysm in apolipoprotein E knockout mice is dependent on STAT3. J. Mol. Cell. Cardiol. 2015, 87, 160–170. [Google Scholar] [CrossRef]
- Li, H.; Bai, S.; Ao, Q.; Wang, X.; Tian, X.; Li, X.; Tong, H.; Hou, W.; Fan, J. Modulation of Immune-Inflammatory Responses in Abdominal Aortic Aneurysm: Emerging Molecular Targets. J. Immunol. Res. 2018, 2018, 7213760. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Nagasawa, A.; Kudo, J.; Onoda, M.; Morikage, N.; Furutani, A.; Aoki, H.; Hamano, K. Inhibitory Effect of Statins on Inflammation-Related Pathways in Human Abdominal Aortic Aneurysm Tissue. Int. J. Mol. Sci. 2015, 16, 11213–11228. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.Z.; Cao, Q.A.; Li, Z.Z.; Liu, L.P.; Zhang, Z.; Zhu, Y.J.; Chu, G.; Dai, Q.Y. SP600125 Attenuates Nicotine-Related Aortic Aneurysm Formation by Inhibiting Matrix Metalloproteinase Production and CC Chemokine-Mediated Macrophage Migration. Mediat. Inflamm. 2016, 2016, 9142425. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Li, X.; Yan, Y.-W.; Tian, X.-H.; Hou, W.-J.; Tong, H.; Bai, S.-L. Curcumin attenuates rat thoracic aortic aneurysm formation by inhibition of the c-Jun N-terminal kinase pathway and apoptosis. Nutrition 2012, 28, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Cui, B.; Zhang, X.; Fu, X.; Yan, J.; Wang, X.; Lv, X.; Chen, Z.; Hu, Z. Antagonism of toll-like receptor 2 attenuates the formation and progression of abdominal aortic aneurysm. Acta Pharm. Sin. B 2015, 5, 176–187. [Google Scholar] [CrossRef]
- Yang, Y.; Ma, Z.; Yang, G.; Wan, J.; Li, G.; Du, L.; Lu, P. Alginate oligosaccharide indirectly affects toll-like receptor signaling via the inhibition of microRNA-29b in aneurysm patients after endovascular aortic repair. Drug Des. Dev. Ther. 2017, 11, 2565–2579. [Google Scholar] [CrossRef]
- Shang, T.; Ran, F.; Qiao, Q.; Liu, Z.; Liu, C.J. Tanshinone IIA attenuates elastase-induced AAA in rats via inhibition of MyD88-dependent TLR-4 signaling. Vasa 2014, 43, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Pirianov, G.; Torsney, E.; Howe, F.; Cockerill, G.W. Rosiglitazone negatively regulates c-Jun N-terminal kinase and toll-like receptor 4 proinflammatory signalling during initiation of experimental aortic aneurysms. Atherosclerosis 2012, 225, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Hasegawa, Y.; Ishigaki, Y.; Yamada, T.; Gao, J.; Imai, J.; Uno, K.; Kaneko, K.; Ogihara, T.; Shimosawa, T.; et al. Importance of endothelial NF-kappaB signalling in vascular remodelling and aortic aneurysm formation. Cardiovasc. Res. 2013, 97, 106–114. [Google Scholar] [CrossRef]
- Xiong, W.; Mactaggart, J.; Knispel, R.; Worth, J.; Zhu, Z.; Li, Y.; Sun, Y.; Baxter, B.T.; Johanning, J. Inhibition of reactive oxygen species attenuates aneurysm formation in a murine model. Atherosclerosis 2009, 202, 128–134. [Google Scholar] [CrossRef]
- Satoh, K.; Nigro, P.; Berk, B.C. Oxidative stress and vascular smooth muscle cell growth: A mechanistic linkage by cyclophilin A. Antioxid. Redox Signal. 2010, 12, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Griendling, K.K.; Ushio-Fukai, M. Redox control of vascular smooth muscle proliferation. J. Lab. Clin. Med. 1998, 132, 9–15. [Google Scholar] [CrossRef]
- Dobrin, P.B.; Mrkvicka, R. Failure of elastin or collagen as possible critical connective tissue alterations underlying aneurysmal dilatation. Cardiovasc. Surg. 1994, 2, 484–488. [Google Scholar] [PubMed]
- Deng, G.G.; Martin-McNulty, B.; Sukovich, D.A.; Freay, A.; Halks-Miller, M.; Thinnes, T.; Loskutoff, D.J.; Carmeliet, P.; Dole, W.P.; Wang, Y.X. Urokinase-type plasminogen activator plays a critical role in angiotensin II-induced abdominal aortic aneurysm. Circ. Res. 2003, 92, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Satta, J.; Haukipuro, K.; Kairaluoma, M.I.; Juvonen, T. Aminoterminal propeptide of type III procollagen in the follow-up of patients with abdominal aortic aneurysms. J. Vasc. Surg. 1997, 25, 909–915. [Google Scholar] [CrossRef]
- Thompson, R.W.; Parks, W.C. Role of matrix metalloproteinases in abdominal aortic aneurysms. Ann. N. Y. Acad. Sci. 1996, 800, 157–174. [Google Scholar] [CrossRef] [PubMed]
- Aziz, F.; Kuivaniemi, H. Role of matrix metalloproteinase inhibitors in preventing abdominal aortic aneurysm. Ann. Vasc. Surg. 2007, 21, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Rundhaug, J.E. Matrix metalloproteinases and angiogenesis. J. Cell. Mol. Med. 2005, 9, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011, 278, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Sun, M.; Sader, S. Matrix metalloproteinases in cardiovascular disease. Can. J. Cardiol. 2006, 22 (Suppl. B), 25B–30B. [Google Scholar] [CrossRef]
- Zhang, X.; Shen, Y.H.; LeMaire, S.A. Thoracic aortic dissection: Are matrix metalloproteinases involved? Vascular 2009, 17, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Osenkowski, P.; Meroueh, S.O.; Pavel, D.; Mobashery, S.; Fridman, R. Mutational and structural analyses of the hinge region of membrane type 1-matrix metalloproteinase and enzyme processing. J. Biol. Chem. 2005, 280, 26160–26168. [Google Scholar] [CrossRef] [PubMed]
- Nagase, H.; Visse, R.; Murphy, G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc. Res. 2006, 69, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Freestone, T.; Turner, R.J.; Coady, A.; Higman, D.J.; Greenhalgh, R.M.; Powell, J.T. Inflammation and matrix metalloproteinases in the enlarging abdominal aortic aneurysm. Arterioscler. Thromb. Vasc. Biol. 1995, 15, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Tamarina, N.A.; McMillan, W.D.; Shively, V.P.; Pearce, W.H. Expression of matrix metalloproteinases and their inhibitors in aneurysms and normal aorta. Surgery 1997, 122, 264–271. [Google Scholar] [CrossRef]
- Davis, V.; Persidskaia, R.; Baca-Regen, L.; Itoh, Y.; Nagase, H.; Persidsky, Y.; Ghorpade, A.; Baxter, B.T. Matrix metalloproteinase-2 production and its binding to the matrix are increased in abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1625–1633. [Google Scholar] [CrossRef]
- Gretarsdottir, S.; Baas, A.F.; Thorleifsson, G.; Holm, H.; den Heijer, M.; de Vries, J.P.; Kranendonk, S.E.; Zeebregts, C.J.; van Sterkenburg, S.M.; Geelkerken, R.H.; et al. Genome-wide association study identifies a sequence variant within the DAB2IP gene conferring susceptibility to abdominal aortic aneurysm. Nat. Genet. 2010, 42, 692–697. [Google Scholar] [CrossRef]
- Bradley, D.T.; Hughes, A.E.; Badger, S.A.; Jones, G.T.; Harrison, S.C.; Wright, B.J.; Bumpstead, S.; Baas, A.F.; Gretarsdottir, S.; Burnand, K.; et al. A variant in LDLR is associated with abdominal aortic aneurysm. Circ. Cardiovasc. Genet. 2013, 6, 498–504. [Google Scholar] [CrossRef]
- Bown, M.J.; Jones, G.T.; Harrison, S.C.; Wright, B.J.; Bumpstead, S.; Baas, A.F.; Gretarsdottir, S.; Badger, S.A.; Bradley, D.T.; Burnand, K.; et al. Abdominal aortic aneurysm is associated with a variant in low-density lipoprotein receptor-related protein 1. Am. J. Hum. Genet. 2011, 89, 619–627. [Google Scholar] [CrossRef]
- Jones, G.T.; Bown, M.J.; Gretarsdottir, S.; Romaine, S.P.; Helgadottir, A.; Yu, G.; Tromp, G.; Norman, P.E.; Jin, C.; Baas, A.F.; et al. A sequence variant associated with sortilin-1 (SORT1) on 1p13.3 is independently associated with abdominal aortic aneurysm. Hum. Mol. Genet. 2013, 22, 2941–2947. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.C.; Smith, A.J.; Jones, G.T.; Swerdlow, D.I.; Rampuri, R.; Bown, M.J.; Folkersen, L.; Baas, A.F.; de Borst, G.J.; Blankensteijn, J.D.; et al. Interleukin-6 receptor pathways in abdominal aortic aneurysm. Eur. Heart J. 2013, 34, 3707–3716. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.M.; Strong, A.; Tohyama, J.; Jin, X.; Morales, C.R.; Billheimer, J.; Millar, J.; Kruth, H.; Rader, D.J. Macrophage sortilin promotes LDL uptake, foam cell formation, and atherosclerosis. Circ. Res. 2015, 116, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Fuster, J.J.; Walsh, K. The good, the bad, and the ugly of interleukin-6 signaling. EMBO J. 2014, 33, 1425–1427. [Google Scholar] [CrossRef] [PubMed]
- Chasman, D.I.; Lawler, P.R. Understanding AAA Pathobiology: A GWAS Leads the Way. Circ. Res. 2017, 120, 259–261. [Google Scholar] [CrossRef]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat. Rev. Mol. Cell Biol. 2007, 8, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.W.; Holmes, D.R.; Mertens, R.A.; Liao, S.; Botney, M.D.; Mecham, R.P.; Welgus, H.G.; Parks, W.C. Production and localization of 92-kilodalton gelatinase in abdominal aortic aneurysms. An elastolytic metalloproteinase expressed by aneurysm-infiltrating macrophages. J. Clin. Investig. 1995, 96, 318–326. [Google Scholar] [CrossRef]
- Longo, G.M.; Xiong, W.; Greiner, T.C.; Zhao, Y.; Fiotti, N.; Baxter, B.T. Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J. Clin. Investig. 2002, 110, 625–632. [Google Scholar] [CrossRef]
- Wang, Y.; Krishna, S.; Walker, P.J.; Norman, P.; Golledge, J. Transforming growth factor-beta and abdominal aortic aneurysms. Cardiovasc. Pathol. 2013, 22, 126–132. [Google Scholar] [CrossRef]
- Ignotz, R.A.; Massague, J. Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J. Biol. Chem. 1986, 261, 4337–4345. [Google Scholar]
- Keski-Oja, J.; Raghow, R.; Sawdey, M.; Loskutoff, D.J.; Postlethwaite, A.E.; Kang, A.H.; Moses, H.L. Regulation of mRNAs for type-1 plasminogen activator inhibitor, fibronectin, and type I procollagen by transforming growth factor-beta. Divergent responses in lung fibroblasts and carcinoma cells. J. Biol. Chem. 1988, 263, 3111–3115. [Google Scholar] [PubMed]
- Kwak, H.J.; Park, M.J.; Cho, H.; Park, C.M.; Moon, S.I.; Lee, H.C.; Park, I.C.; Kim, M.S.; Rhee, C.H.; Hong, S.I. Transforming growth factor-beta1 induces tissue inhibitor of metalloproteinase-1 expression via activation of extracellular signal-regulated kinase and Sp1 in human fibrosarcoma cells. Mol. Cancer Res. 2006, 4, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Fujii, K.; Akiyama, N.; Furutani, A.; Hoshii, Y.; Tanaka, N.; Ricci, R.; Ishihara, T.; et al. Regression of abdominal aortic aneurysm by inhibition of c-Jun N-terminal kinase. Nat. Med. 2005, 11, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; DiMusto, P.D.; Ehrlichman, L.K.; Sadiq, O.; McEvoy, B.; Futchko, J.S.; Henke, P.K.; Eliason, J.L.; Upchurch, G.R., Jr. The role of extracellular signal-related kinase during abdominal aortic aneurysm formation. J. Am. Coll. Surg. 2012, 215, 668–680. [Google Scholar] [CrossRef] [PubMed]
- Moshal, K.S.; Sen, U.; Tyagi, N.; Henderson, B.; Steed, M.; Ovechkin, A.V.; Tyagi, S.C. Regulation of homocysteine-induced MMP-9 by ERK1/2 pathway. Am. J. Physiol. Cell Physiol. 2006, 290, C883–C891. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Q.; Li, W.; Li, S.Q.; Li, J.; Li, Y.W.; Kong, S.X.; Liu, R.M.; Wang, S.M.; Lv, W.M. MCP-1 stimulates MMP-9 expression via ERK 1/2 and p38 MAPK signaling pathways in human aortic smooth muscle cells. Cell. Physiol. Biochem. 2014, 34, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Shen, L.; Gao, P.; Li, G.; He, Y.; Wang, M.; Zhou, H.; Yuan, H.; Jin, X.; Wu, X. Effect of AMPK signal pathway on pathogenesis of abdominal aortic aneurysms. Oncotarget 2017, 8, 92827–92840. [Google Scholar] [CrossRef] [PubMed]
- Radak, D.; Tanaskovic, S.; Katsiki, N.; Isenovic, E.R. Protective Role of Diabetes Mellitus on Abdominal Aortic Aneurysm Pathogenesis: Myth or Reality? Curr. Vasc. Pharmacol. 2016, 14, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Hinchliffe, R.J. Metformin and Abdominal Aortic Aneurysm. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 679–680. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shen, M.; Lee, J.; Basu, R.; Sakamuri, S.S.; Wang, X.; Fan, D.; Kassiri, Z. Divergent roles of matrix metalloproteinase 2 in pathogenesis of thoracic aortic aneurysm. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 888–898. [Google Scholar] [CrossRef] [PubMed]
- Theruvath, T.P.; Jones, J.A.; Ikonomidis, J.S. Matrix metalloproteinases and descending aortic aneurysms: Parity, disparity, and switch. J. Card. Surg. 2012, 27, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Barbour, J.R.; Stroud, R.E.; Lowry, A.S.; Clark, L.L.; Leone, A.M.; Jones, J.A.; Spinale, F.G.; Ikonomidis, J.S. Temporal disparity in the induction of matrix metalloproteinases and tissue inhibitors of metalloproteinases after thoracic aortic aneurysm formation. J. Thorac. Cardiovasc. Surg. 2006, 132, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.A.; Barbour, J.R.; Lowry, A.S.; Bouges, S.; Beck, C.; McClister, D.M., Jr.; Mukherjee, R.; Ikonomidis, J.S. Spatiotemporal expression and localization of matrix metalloproteinas-9 in a murine model of thoracic aortic aneurysm. J. Vasc. Surg. 2006, 44, 1314–1321. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jones, J.A.; Zavadzkas, J.A.; Chang, E.I.; Sheats, N.; Koval, C.; Stroud, R.E.; Spinale, F.G.; Ikonomidis, J.S. Cellular phenotype transformation occurs during thoracic aortic aneurysm development. J. Thorac. Cardiovasc. Surg. 2010, 140, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Hussien, H.; Soekhoe, R.G.V.; Weber, E.; von der Thüsen, J.H.; Kleemann, R.; Mulder, A.; van Bockel, J.H.; Hanemaaijer, R.; Lindeman, J.H.N. Collagen degradation in the abdominal aneurysm: A conspiracy of matrix metalloproteinase and cysteine collagenases. Am. J. Pathol. 2007, 170, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, C.; Qin, Y. Cathepsins: A new culprit behind abdominal aortic aneurysm. Regen. Med. Res. 2013, 1, 5. [Google Scholar] [CrossRef][Green Version]
- Xiong, W.; Knispel, R.; Mactaggart, J.; Baxter, B.T. Effects of tissue inhibitor of metalloproteinase 2 deficiency on aneurysm formation. J. Vasc. Surg. 2006, 44, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Aoki, T.; Kataoka, H.; Moriwaki, T.; Nozaki, K.; Hashimoto, N. Role of TIMP-1 and TIMP-2 in the progression of cerebral aneurysms. Stroke 2007, 38, 2337–2345. [Google Scholar] [CrossRef]
- Nascimento Dda, C.; Durigan Rde, C.; Tibana, R.A.; Durigan, J.L.; Navalta, J.W.; Prestes, J. The response of matrix metalloproteinase-9 and -2 to exercise. Sports Med. 2015, 45, 269–278. [Google Scholar] [CrossRef]
- Sohail, A.; Marco, M.; Zhao, H.; Shi, Q.; Merriman, S.; Mobashery, S.; Fridman, R. Characterization of the dimerization interface of membrane type 4 (MT4)-matrix metalloproteinase. J. Biol. Chem. 2011, 286, 33178–33189. [Google Scholar] [CrossRef]
- Wilson, W.R.; Anderton, M.; Choke, E.C.; Dawson, J.; Loftus, I.M.; Thompson, M.M. Elevated plasma MMP1 and MMP9 are associated with abdominal aortic aneurysm rupture. Eur. J. Vasc. Endovasc. Surg. 2008, 35, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Vincenti, M.P.; Brinckerhoff, C.E. Transcriptional regulation of collagenase (MMP-1, MMP-13) genes in arthritis: Integration of complex signaling pathways for the recruitment of gene-specific transcription factors. Arthritis Res. 2002, 4, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Vianello, E.; Dozio, E.; Rigolini, R.; Marrocco-Trischitta, M.M.; Tacchini, L.; Trimarchi, S.; Corsi Romanelli, M.M. Acute phase of aortic dissection: A pilot study on CD40L, MPO, and MMP-1, -2, 9 and TIMP-1 circulating levels in elderly patients. Immun. Ageing 2016, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Hadi, T.; Boytard, L.; Silvestro, M.; Alebrahim, D.; Jacob, S.; Feinstein, J.; Barone, K.; Spiro, W.; Hutchison, S.; Simon, R.; et al. Macrophage-derived netrin-1 promotes abdominal aortic aneurysm formation by activating MMP3 in vascular smooth muscle cells. Nat. Commun. 2018, 9, 5022. [Google Scholar] [CrossRef] [PubMed]
- Lyon, C.A.; Williams, H.; Bianco, R.; Simmonds, S.J.; Brown, B.A.; Wadey, K.S.; Smith, F.C.T.; Johnson, J.L.; George, S.J. Aneurysm Severity is Increased by Combined Mmp-7 Deletion and N-cadherin Mimetic (EC4-Fc) Over-Expression. Sci. Rep. 2017, 7, 17342. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.R.; Anderton, M.; Schwalbe, E.C.; Jones, J.L.; Furness, P.N.; Bell, P.R.; Thompson, M.M. Matrix metalloproteinase-8 and -9 are increased at the site of abdominal aortic aneurysm rupture. Circulation 2006, 113, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.A.; Cox, J.H.; Bellac, C.L.; Doucet, A.; Starr, A.E.; Overall, C.M. Macrophage-specific metalloelastase (MMP-12) truncates and inactivates ELR+ CXC chemokines and generates CCL2, -7, -8, and -13 antagonists: Potential role of the macrophage in terminating polymorphonuclear leukocyte influx. Blood 2008, 112, 3455–3464. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, A.; Slater, S.C.; George, S.J. MMP-9 and -12 cause N-cadherin shedding and thereby beta-catenin signalling and vascular smooth muscle cell proliferation. Cardiovasc. Res. 2009, 81, 178–186. [Google Scholar] [CrossRef]
- Longo, G.M.; Buda, S.J.; Fiotta, N.; Xiong, W.; Griener, T.; Shapiro, S.; Baxter, B.T. MMP-12 has a role in abdominal aortic aneurysms in mice. Surgery 2005, 137, 457–462. [Google Scholar] [CrossRef]
- Curci, J.A.; Liao, S.; Huffman, M.D.; Shapiro, S.D.; Thompson, R.W. Expression and localization of macrophage elastase (matrix metalloproteinase-12) in abdominal aortic aneurysms. J. Clin. Investig. 1998, 102, 1900–1910. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, C.; Jia, L.; Chen, B.; Liu, L.; Sun, J.; Zhang, W.; You, B.; Li, Y.; Li, P.; et al. Interleukin-3 stimulates matrix metalloproteinase 12 production from macrophages promoting thoracic aortic aneurysm/dissection. Clin. Sci. 2018, 132, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.; Lee, J.K.; VanVickle, S.J.; Thompson, R.W. Expression of collagenase-3 (MMP-13) in human abdominal aortic aneurysms and vascular smooth muscle cells in culture. Biochem. Biophys. Res. Commun. 1999, 261, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, D.; Choi, J.C.; Minard, C.G.; Hou, X.; Coselli, J.S.; Shen, Y.H.; LeMaire, S.A. Matrix metalloproteinase levels in chronic thoracic aortic dissection. J. Surg. Res. 2014, 189, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Jackson, V.; Olsson, T.; Kurtovic, S.; Folkersen, L.; Paloschi, V.; Wagsater, D.; Franco-Cereceda, A.; Eriksson, P. Matrix metalloproteinase 14 and 19 expression is associated with thoracic aortic aneurysms. J. Thorac. Cardiovasc. Surg. 2012, 144, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Martin-Alonso, M.; Garcia-Redondo, A.B.; Guo, D.; Camafeita, E.; Martinez, F.; Alfranca, A.; Mendez-Barbero, N.; Pollan, A.; Sanchez-Camacho, C.; Denhardt, D.T.; et al. Deficiency of MMP17/MT4-MMP proteolytic activity predisposes to aortic aneurysm in mice. Circ. Res. 2015, 117, e13–e26. [Google Scholar] [CrossRef] [PubMed]
- Bumdelger, B.; Kokubo, H.; Kamata, R.; Fujii, M.; Ishida, M.; Ishida, T.; Yoshizumi, M. Induction of Timp1 in smooth muscle cells during development of abdominal aortic aneurysms. Hiroshima J. Med. Sci. 2013, 62, 63–67. [Google Scholar] [PubMed]
- Allaire, E.; Forough, R.; Clowes, M.; Starcher, B.; Clowes, A.W. Local overexpression of TIMP-1 prevents aortic aneurysm degeneration and rupture in a rat model. J. Clin. Investig. 1998, 102, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Ikonomidis, J.S.; Gibson, W.C.; Butler, J.E.; McClister, D.M.; Sweterlitsch, S.E.; Thompson, R.P.; Mukherjee, R.; Spinale, F.G. Effects of deletion of the tissue inhibitor of matrix metalloproteinases-1 gene on the progression of murine thoracic aortic aneurysms. Circulation 2004, 110, II268–II273. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, M.K.; Vijungco, J.D.; Flores, A.; Borensztajn, J.; Shively, V.; Pearce, W.H. Enhanced abdominal aortic aneurysm in TIMP-1-deficient mice. J. Surg. Res. 2005, 123, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Basu, R.; Fan, D.; Kandalam, V.; Lee, J.; Das, S.K.; Wang, X.; Baldwin, T.A.; Oudit, G.Y.; Kassiri, Z. Loss of Timp3 gene leads to abdominal aortic aneurysm formation in response to angiotensin II. J. Biol. Chem. 2012, 287, 44083–44096. [Google Scholar] [CrossRef]
- Leivonen, S.-K.; Lazaridis, K.; Decock, J.; Chantry, A.; Edwards, D.R.; Kähäri, V.-M. TGF-β-Elicited Induction of Tissue Inhibitor of Metalloproteinases (TIMP)-3 Expression in Fibroblasts Involves Complex Interplay between Smad3, p38α, and ERK1/2. PLoS ONE 2013, 8, e57474. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Qi, X.; Li, Z.; Zhang, W.; Li, C.; Ji, L.; Xu, K.; Zhong, H. TIMP-3 suppresses the proliferation and migration of SMCs from the aortic neck of atherosclerotic AAA in rabbits, via decreased MMP-2 and MMP-9 activity, and reduced TNF-α expression. Mol. Med. Rep. 2018, 18, 2061–2067. [Google Scholar] [CrossRef] [PubMed]
- Uitto, V.J.; Firth, J.D.; Nip, L.; Golub, L.M. Doxycycline and chemically modified tetracyclines inhibit gelatinase A (MMP-2) gene expression in human skin keratinocytes. Ann. N. Y. Acad. Sci. 1994, 732, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Pyo, R.; Lee, J.K.; Shipley, J.M.; Curci, J.A.; Mao, D.; Ziporin, S.J.; Ennis, T.L.; Shapiro, S.D.; Senior, R.M.; Thompson, R.W. Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J. Clin. Investig. 2000, 105, 1641–1649. [Google Scholar] [CrossRef]
- Prall, A.K.; Longo, G.M.; Mayhan, W.G.; Waltke, E.A.; Fleckten, B.; Thompson, R.W.; Baxter, B.T. Doxycycline in patients with abdominal aortic aneurysms and in mice: Comparison of serum levels and effect on aneurysm growth in mice. J. Vasc. Surg. 2002, 35, 923–929. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, J.; Li, D.; Chen, J.; Shen, X.; Xu, F.; Teng, F.; Deng, Y.; Ma, H.; Zhang, L.; et al. Salvianolic acid A, a matrix metalloproteinase-9 inhibitor of Salvia miltiorrhiza, attenuates aortic aneurysm formation in apolipoprotein E-deficient mice. Phytomedicine 2014, 21, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, H.; Aoka, Y.; Sakomura, Y.; Sakuta, A.; Aomi, S.; Ishizuka, N.; Hagiwara, N.; Kawana, M.; Kasanuki, H. A 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor, cerivastatin, suppresses production of matrix metalloproteinase-9 in human abdominal aortic aneurysm wall. J. Vasc. Surg. 2002, 36, 158–163. [Google Scholar] [CrossRef]
- Kalyanasundaram, A.; Elmore, J.R.; Manazer, J.R.; Golden, A.; Franklin, D.P.; Galt, S.W.; Zakhary, E.M.; Carey, D.J. Simvastatin suppresses experimental aortic aneurysm expansion. J. Vasc. Surg. 2006, 43, 117–124. [Google Scholar] [CrossRef]
- Boyle, J.R.; Loftus, I.M.; Goodall, S.; Crowther, M.; Bell, P.R.; Thompson, M.M. Amlodipine potentiates metalloproteinase activity and accelerates elastin degradation in a model of aneurysmal disease. Eur. J. Vasc. Endovasc. Surg. 1998, 16, 408–414. [Google Scholar] [CrossRef]
- Chen, X.; Rateri, D.L.; Howatt, D.A.; Balakrishnan, A.; Moorleghen, J.J.; Morris, A.J.; Charnigo, R.; Cassis, L.A.; Daugherty, A. Amlodipine reduces AngII-induced aortic aneurysms and atherosclerosis in hypercholesterolemic mice. PLoS ONE 2013, 8, e81743. [Google Scholar] [CrossRef]
- Takahashi, K.; Matsumoto, Y.; Doe, Z.; Kanazawa, M.; Satoh, K.; Shimizu, T.; Sato, A.; Fukumoto, Y.; Shimokawa, H. Combination Therapy with Atorvastatin and Amlodipine Suppresses Angiotensin II-Induced Aortic Aneurysm Formation. PLoS ONE 2013, 8, e72558. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Van den Steen, P.E.; Sang, Q.-X.A.; Opdenakker, G. Matrix metalloproteinase inhibitors as therapy for inflammatory and vascular diseases. Nat. Rev. Drug Discov. 2007, 6, 480. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; Lapiere, C.M. Collagenolytic activity in amphibian tissues: A tissue culture assay. Proc. Natl. Acad. Sci. USA 1962, 48, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Miyagawa, S.; Fukushima, S.; Daimon, T.; Shirakawa, Y.; Kuratani, T.; Sawa, Y. Development of a prostacyclin-agonist–eluting aortic stent graft enhancing biological attachment to the aortic wall. J. Thorac. Cardiovasc. Surg. 2014, 148, 2325–2334. [Google Scholar] [CrossRef] [PubMed]
- Shirasu, T.; Koyama, H.; Miura, Y.; Hoshina, K.; Kataoka, K.; Watanabe, T. Nanoparticles Effectively Target Rapamycin Delivery to Sites of Experimental Aortic Aneurysm in Rats. PLoS ONE 2016, 11, e0157813. [Google Scholar] [CrossRef] [PubMed]
- Nosoudi, N.; Chowdhury, A.; Siclari, S.; Parasaram, V.; Karamched, S.; Vyavahare, N. Systemic Delivery of Nanoparticles Loaded with Pentagalloyl Glucose Protects Elastic Lamina and Prevents Abdominal Aortic Aneurysm in Rats. J. Cardiovasc. Transl. Res. 2016, 9, 445–455. [Google Scholar] [CrossRef]
- Nosoudi, N.; Chowdhury, A.; Siclari, S.; Karamched, S.; Parasaram, V.; Parrish, J.; Gerard, P.; Vyavahare, N. Reversal of Vascular Calcification and Aneurysms in a Rat Model Using Dual Targeted Therapy with EDTA- and PGG-Loaded Nanoparticles. Theranostics 2016, 6, 1975–1987. [Google Scholar] [CrossRef]
- Miyake, T.; Morishita, R. Pharmacological treatment of abdominal aortic aneurysm. Cardiovasc. Res. 2009, 83, 436–443. [Google Scholar] [CrossRef]
| Role within AAA, AD or Rupture | Substrate | Stimulus | Known Signaling Pathways | Ref. | |
|---|---|---|---|---|---|
| MMP-1 | Associated with increased rates of aneurysmal rupture and reduced survival, and aortic dissection | Collagen triple helix | TNF-α IL-1 | (MAPK family) JNK, ERK and p38 kinase-induced activation of MMP-1 | [121,122,123] |
| MMP-3 | Activity promotes AAA | Elastin | Netrin-1 | Netrin-1 binds neogenin-1 receptors on VSMCs to activate MMP3 | [124] |
| MMP-7 | Increased expression in AAA | N-cadherin | PI-3 kinase/Akt VSMC apoptosis and proliferation | [125] | |
| MMP-8 | Elevated expression in growing and rupture AAA, released by neutrophils | Collagen triple helix Cystatin C | Ox-LDL IL-1 | [115,126] | |
| MMP-12 | Promotes AAA growth by regulating leukocyte recruitment Promotes AD formation | Elastin CXC-chemokine ligand 2 and 3 IFNγ | IL-3 | MMP-12 cleaves N-cadherin, triggering ß-catenin signalling and VSMC proliferation | [127,128,129,130,131] |
| MMP-13 | Elevated expression in AAA sections and thoracic aortic dissection tissues, predominantly localised to VSMCs | Collagen triple helix | TNF-α IL-1 | (MAPK family) JNK, ERK and p38 kinase-induced activation of MMP-13 | [132,133] |
| MMP-14 (MT1-MMP) | Modest increase in tissues of ruptured AAA | Collagen triple helix | [134] | ||
| MMP-17 (MT4-MMP) | Inhibits AAA formation | Osteopontin in VSMCs | c-Jun N-terminal kinase signalling, VSMC maturation | [135] | |
| MMP-19 | Expression is associated with aneurysms | [134] | |||
| TIMP-1 | Increased levels in AAA Overexpression leads to ablation of AAA within experimental rodent model Deletion enhances aneurysm formation | MMP-1, MMP-9 and MMP-3 | TNF-α | Inhibits MMP-1, MMP-9 and MMP-3 | [97,136,137,138,139] |
| TIMP-2 | TIMP-2 promotes aortic growth through activation of MMP-2 in murine model of AAA Reduced expression of TIMP-2 in late stage cerebral aneurysm formation | MMP-2 | Regulates MMP-2 | [117,118] | |
| TIMP-3 | Increased expression in response to MMP over activity, with heightened expression in human AAA end stage tissues Loss of TIMP-3 leads to worsening of the AAA pathology Reduces VSMC proliferation and migration | MMP-2, MMP-9, TNF-α | TGF-ß | [140,141,142] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maguire, E.M.; Pearce, S.W.A.; Xiao, R.; Oo, A.Y.; Xiao, Q. Matrix Metalloproteinase in Abdominal Aortic Aneurysm and Aortic Dissection. Pharmaceuticals 2019, 12, 118. https://doi.org/10.3390/ph12030118
Maguire EM, Pearce SWA, Xiao R, Oo AY, Xiao Q. Matrix Metalloproteinase in Abdominal Aortic Aneurysm and Aortic Dissection. Pharmaceuticals. 2019; 12(3):118. https://doi.org/10.3390/ph12030118
Chicago/Turabian StyleMaguire, Eithne M., Stuart W. A. Pearce, Rui Xiao, Aung Y. Oo, and Qingzhong Xiao. 2019. "Matrix Metalloproteinase in Abdominal Aortic Aneurysm and Aortic Dissection" Pharmaceuticals 12, no. 3: 118. https://doi.org/10.3390/ph12030118
APA StyleMaguire, E. M., Pearce, S. W. A., Xiao, R., Oo, A. Y., & Xiao, Q. (2019). Matrix Metalloproteinase in Abdominal Aortic Aneurysm and Aortic Dissection. Pharmaceuticals, 12(3), 118. https://doi.org/10.3390/ph12030118




