Electrochemical Characterization of Central Action Tricyclic Drugs by Voltammetric Techniques and Density Functional Theory Calculations
Abstract
1. Introduction
2. Results and Discussions
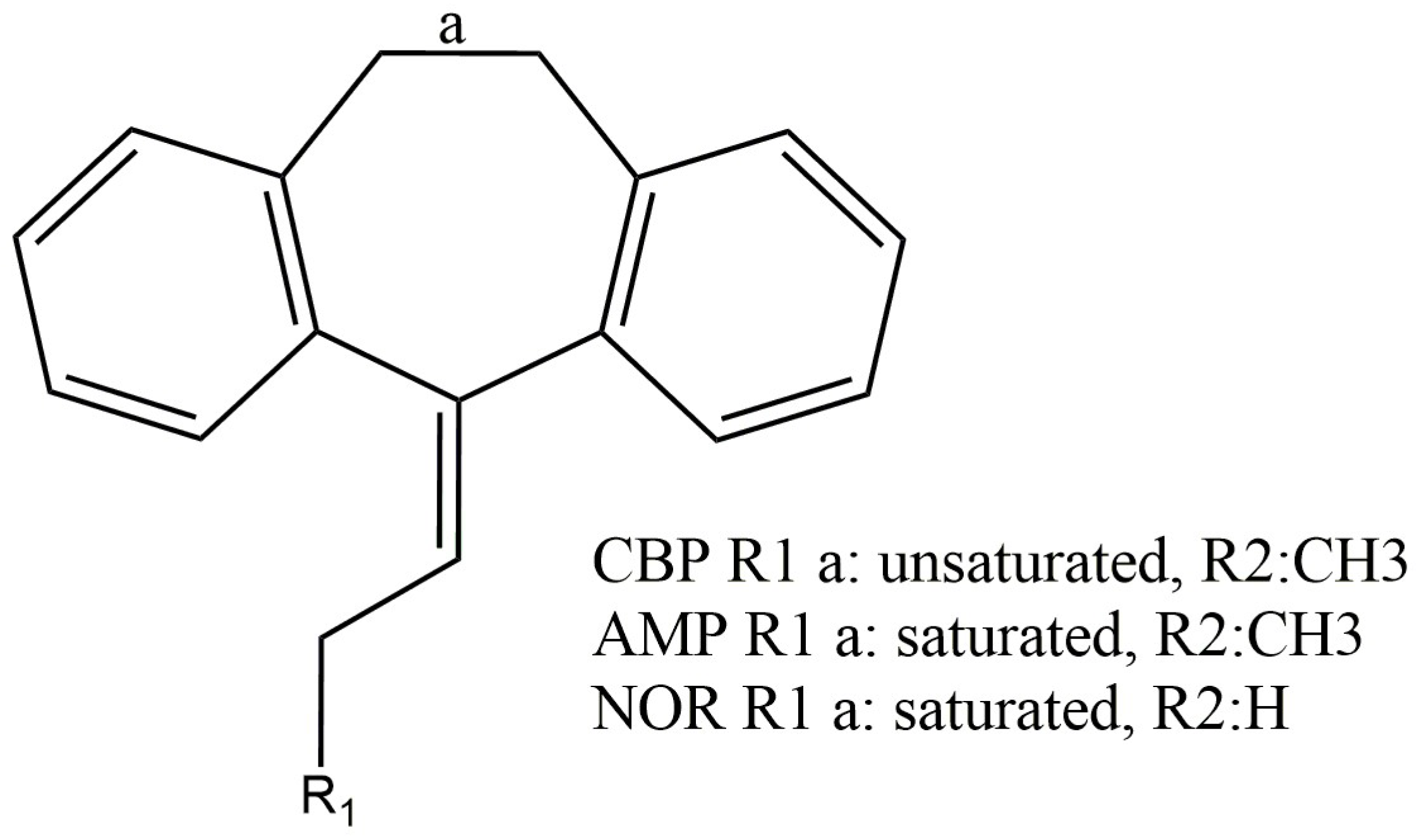
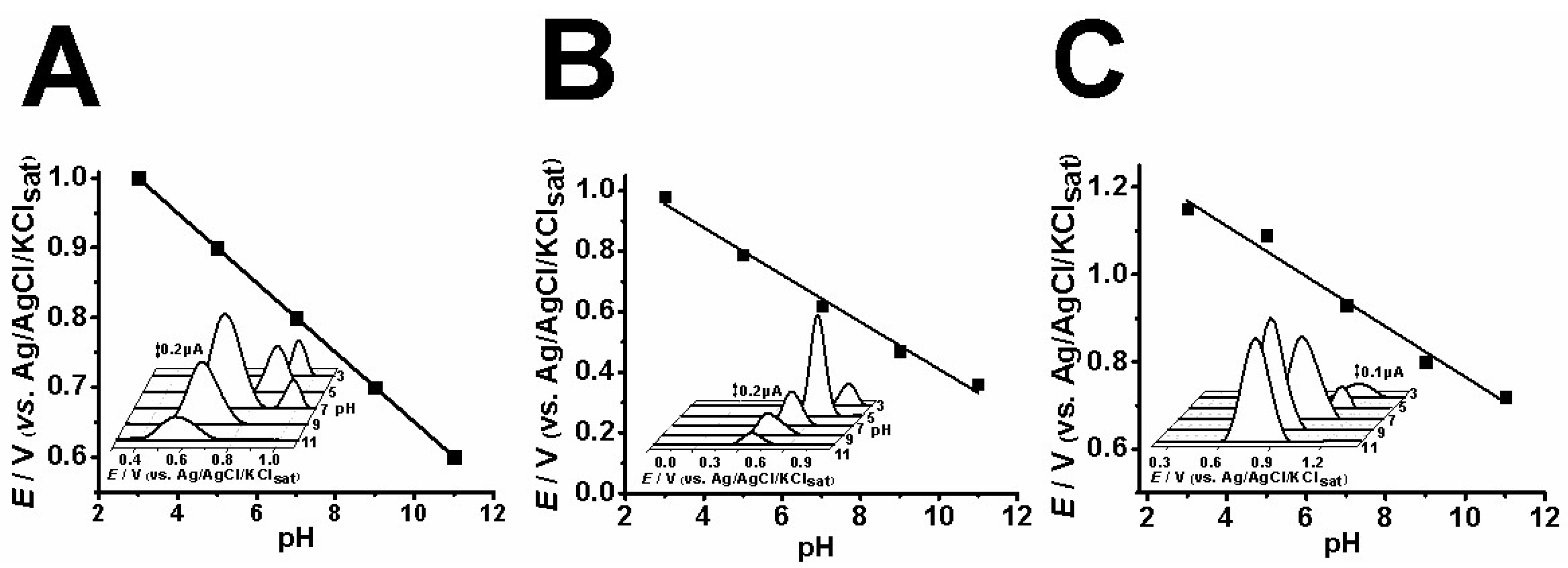
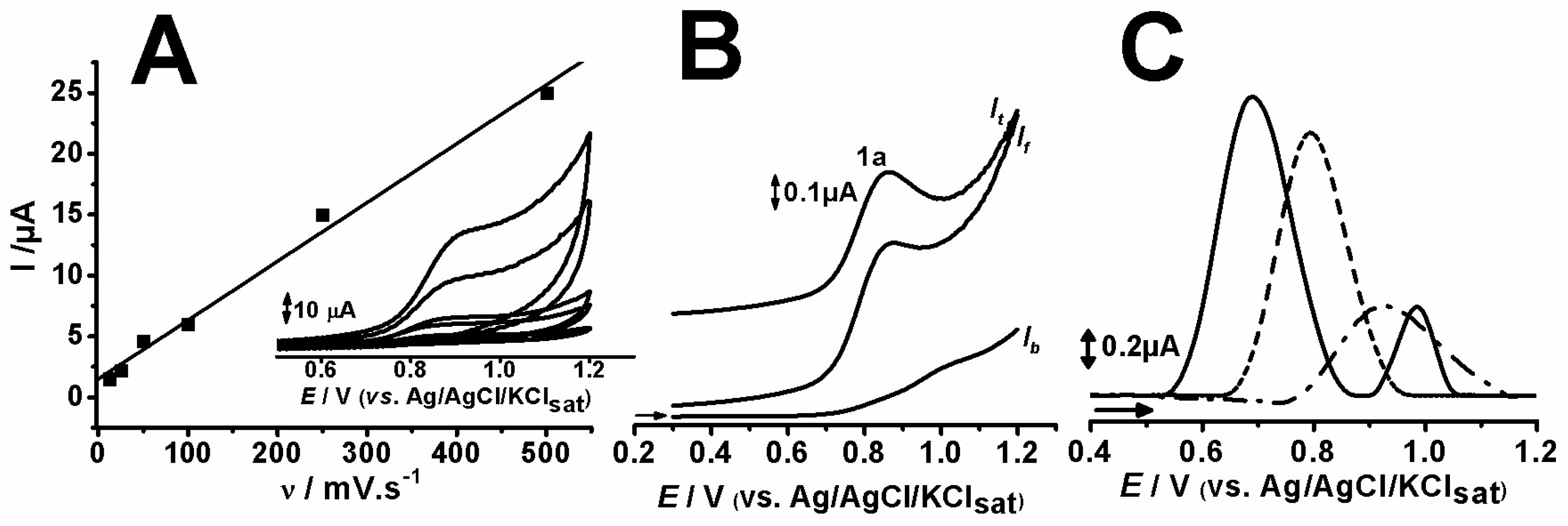
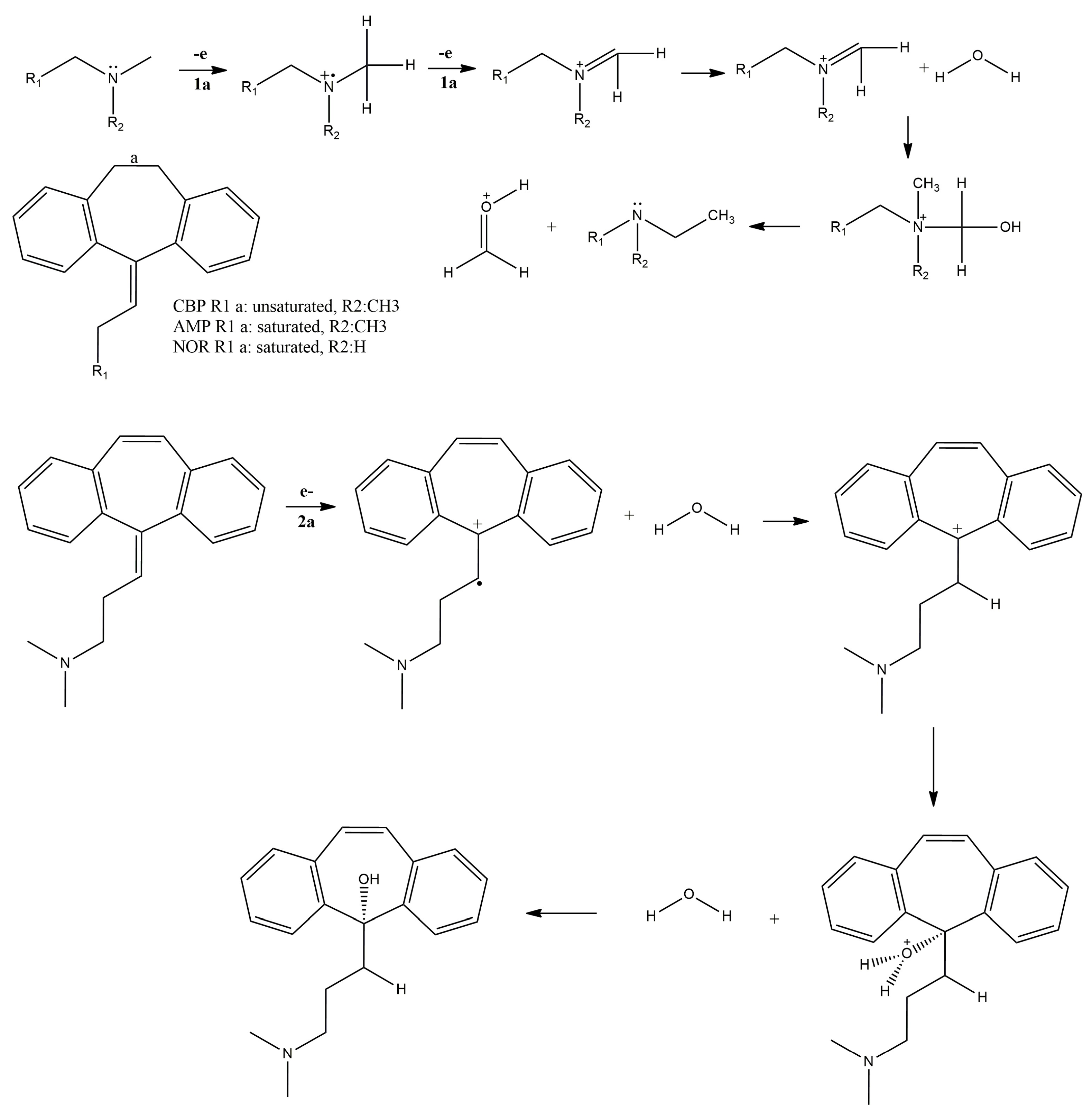
2.1. Electrochemical Assays
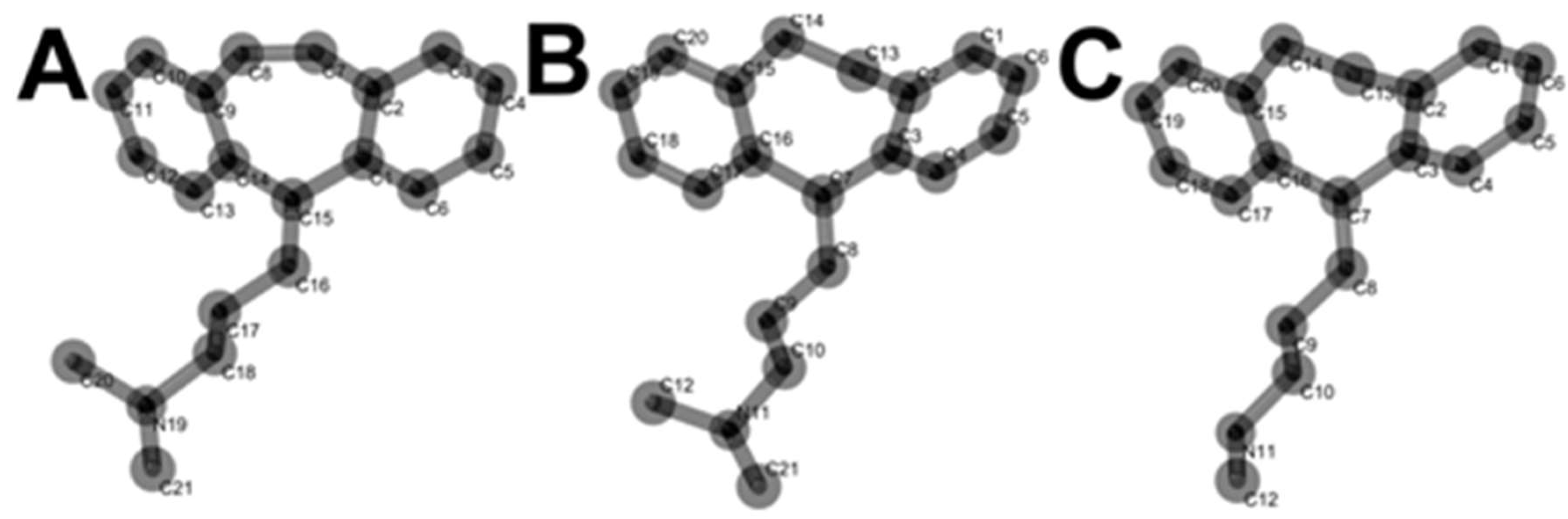
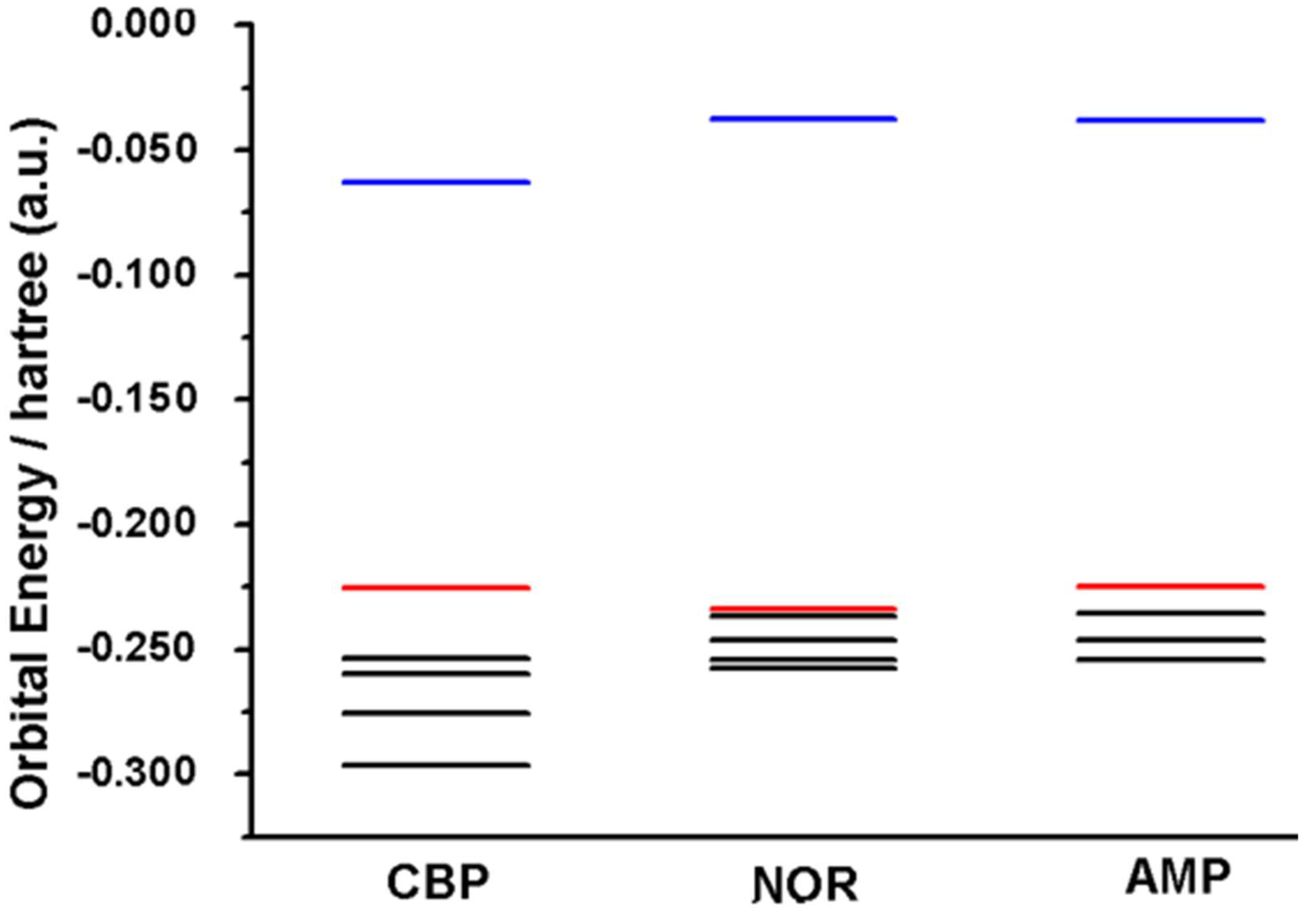
2.2. Molecular Electronic Structure Calculations
3. Materials and Methods
3.1. Reagents, Samples and Solutions
3.2. Electrochemical Assays
3.3. Molecular Electronic Structure Calculations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, Y.W.; Macdonald, J.B.; Nelson, S.A.; Sekulic, A. Treatment of vismodegib-associated muscle cramps with cyclobenzaprine: A retrospective review. J. Am. Acad. Dermatol. 2017, 77, 1170–1172. [Google Scholar] [CrossRef] [PubMed]
- De Santana Santos, T.; Calazans, A.C.M.; Martins-Filho, P.R.S.; Da Silva, L.C.F.; De Oliveira, E.; Silva, E.D.; Gomes, A.C.A. Evaluation of the muscle relaxant cyclobenzaprine after third-molar extraction. J. Am. Dent. Assoc. 2011, 142, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.T.; Xue, R.; Wang, X.; Zhao, S.W.; An, L.; Li, Y.F.; Zhang, Y.Z.; Li, S. Network-based drug repositioning: A novel strategy for discovering potential antidepressants and their mode of action. Eur. Neuropsychopharmacol. 2018, 28, 1137–1150. [Google Scholar] [CrossRef] [PubMed]
- De Macêdo, I.Y.L.; Garcia, L.F.; Menegatti, R.; Guimarães, F.F.; Lião, L.M.; de Carvalho, F.S.; Torres Pio dos Santos, W.; Verly, R.M.; Arotiba, O.A.; de Souza Gil, E. Electrochemical characterizations of darbufelone, a di-tert-butylphenol derivative, by voltammetric techniques and density functional theory calculations. Electrochim. Acta 2018, 268, 462–468. [Google Scholar] [CrossRef]
- de Siqueira Leite, K.C.; Garcia, L.F.; Sanz, G.; Colmati, F.; de Souza, A.R.; da Costa Batista, D.; Menegatti, R.; de Souza Gil, E.; Luque, R. Electrochemical characterization of a novel nimesulide anti-inflammatory drug analog: LQFM-091. J. Electroanal. Chem. 2018, 818, 92–96. [Google Scholar] [CrossRef]
- Thomaz, D.V.; de S. Leite, K.C.; Moreno, E.K.G.; Garcia, L.F.; Alecrim, M.F.; Macêdo, I.Y.L.; Caetano, M.P.; de Carvalho, M.F.; Machado, F.B.; de Gil, E.S. Electrochemical study of commercial black tea samples. Int. J. Electrochem. Sci. 2018, 13, 5433–5439. [Google Scholar]
- Torres, S.; Brown, R.; Szucs, R.; Hawkins, J.M.; Zelesky, T.; Scrivens, G.; Pettman, A.; Taylor, M.R. The application of electrochemistry to pharmaceutical stability testing—Comparison with in silico prediction and chemical forced degradation approaches. J. Pharm. Biomed. Anal. 2015, 115, 487–501. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Xia, Z.N.; Qin, L.T.; Liu, S.S. Prediction of blood-brain partitioning: A model based on molecular electronegativity distance vector descriptors. J. Mol. Graph. Model. 2010, 29, 214–220. [Google Scholar] [CrossRef]
- Verstraelen, T.; Bultinck, P. Can the electronegativity equalization method predict spectroscopic properties? Acta Part A Mol. Biomol. Spectrosc. 2015, 136, 76–80. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision, A.02; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Hasanzadeh, N.; Nori-Shargh, D. Correlations between hardness, electronegativity, anomeric effect associated with electron delocalizations and electrostatic interactions in 1,4,5,8-tetraoxadecalin and its analogs containing S and Se atoms. Comput. Theor. Chem. 2015, 1051, 1–9. [Google Scholar] [CrossRef]
- Jmol: An Open-Source Java Viewer for Chemical Structures in 3D. 2009. Available online: http://jmol.sourceforge.net/ (accessed on 22 January 2019).
- Enache, T.A.; Oliveira-Brett, A.M. Phenol and para-substituted phenols electrochemical oxidation pathways. J. Electroanal. Chem. 2011, 655, 9–16. [Google Scholar] [CrossRef]
- Ban, Z.; Kätelhön, E.; Compton, R.G. Voltammetry of porous layers: Staircase vs. analog voltammetry. J. Electroanal. Chem. 2016, 776, 25–33. [Google Scholar] [CrossRef]
- Ferancová, A.; Korgová, E.; Mikó, R.; Labuda, J. Determination of tricyclic antidepressants using a carbon paste electrode modified with β-cyclodextrin. J. Electroanal. Chem. 2000, 492, 74–77. [Google Scholar] [CrossRef]
- Ivandini, T.A.; Sarada, B.V.; Terashima, C.; Rao, T.N.; Tryk, D.A.; Ishiguro, H.; Kubota, Y.; Fujishima, A. Electrochemical detection of tricyclic antidepressant drugs by HPLC using highly boron-doped diamond electrodes. J. Electroanal. Chem. 2002, 521, 117–126. [Google Scholar] [CrossRef]
- Wang, J.; Golden, T.; Ozsoz, M.; Lu, Z. Sensitive and selective voltammetric measurements of tricyclic antidepressants using lipid-coated electrodes. J. Electroanal. Chem. 1990, 298, 217–226. [Google Scholar] [CrossRef]
- Uchida, Y.; Kätelhön, E.; Compton, R.G. Cyclic voltammetry with non-triangular waveforms: Electrochemically irreversible and quasi-reversible systems. J. Electroanal. Chem. 2018, 810, 135–144. [Google Scholar] [CrossRef]
- Harada, R.; Hosomi, T.; Masai, H.; Terao, J. Reversible and stable redox behavior of a Pt (II) bis(dithiobenzoate)-type complex attributed to rotaxane-based stabilization. Tetrahedron Lett. 2018, 59, 2930–2933. [Google Scholar] [CrossRef]
- De Toledo, R.A.; Santos, M.C.; Suffredini, H.B.; Homem-de-Mello, P.; Honorio, K.M.; Mazo, L.H. DFT and electrochemical studies on nortriptyline oxidation sites. J. Mol. Modeling 2009, 15, 945–952. [Google Scholar] [CrossRef]
- Rodrigues, A.S.; Nunes, M.J.; Lopes, A.; Silva, J.N.; Ciríaco, L.; Pacheco, M.J. Electrodegradation of naphthalenic amines: Influence of the relative position of the substituent groups, anode material and electrolyte on the degradation products and kinetics. Chemosphere 2018, 205, 433–442. [Google Scholar] [CrossRef]
- Shabbir, M.; Akhter, Z.; Ahmad, I.; Ahmed, S.; Bolte, M.; McKee, V. Synthesis and bioelectrochemical behavior of aromatic amines. Bioorg. Chem. 2017, 75, 224–234. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, D.; Zhao, Z. Study of degradation mechanisms of cyclobenzaprine by LC-MS/MS. Anal. Methods 2014, 6, 2320–2330. [Google Scholar] [CrossRef]
- Darwish, H.W.; Naguib, I.A.; Darwish, I.A. Five modified classical least squares based models for stability indicating analysis of cyclobenzaprine HCl with its major degradation products: A comparative study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 204, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Harde, M.T.; Wankhede, S.B.; Chaudhari, P.D. A validated inherent stability indicating HPTLC method for estimation of cyclobenzaprine hydrochloride in tablets and use of MS–QTOF in characterization of its alkaline stress degradation product. Bull. Fac. Pharm. Cairo Univ. 2016, 54, 145–156. [Google Scholar] [CrossRef][Green Version]
- Wang, J.; Bonakdar, M.; Morgan, C. Voltammetric measurement of tricyclic antidepressants following interfacial accumulation at carbon electrodes. Anal. Methods 1986, 58, 1024–1028. [Google Scholar]
- Suckow, R.F.; Cooper, T.B. Simultaneous determination of imipramine, desipramine, and their 2-hydroxy etabolites in plasma by ion-pair reversed-phase high-performance liquid chromatography with amperometric detection. J. Pharm. Sci. 1981, 70, 257–261. [Google Scholar]
- Gil, E.S.; Kubota, L.T. Electrochemical properties of Doyle catalyst immobilized on carbon paste in the presence of DNA. Bioelectrochem. Bioenerg. 2000, 51, 145–149. [Google Scholar] [CrossRef]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Munir, A.; Bozal-Palabiyik, B.; Khan, A.; Shah, A.; Uslu, B. A novel electrochemical method for the detection of oxymetazoline drug based on MWCNTs and TiO2 nanoparticles. J. Electroanal. Chem. 2019, 844, 58–65. [Google Scholar] [CrossRef]
- Thomaz, D.V.; de Aguiar Filho, A.M.; de Macêdo, I.Y.L.; Rodrigues, E.S.B.; Gil, E.S. Predictive modelling to study the electrochemical behaviour of PdO, TiO2 and perovskite-type LaFeO3 modified carbon paste electrodes. Path Sci. 2018, 5, 4001–4007. [Google Scholar] [CrossRef]
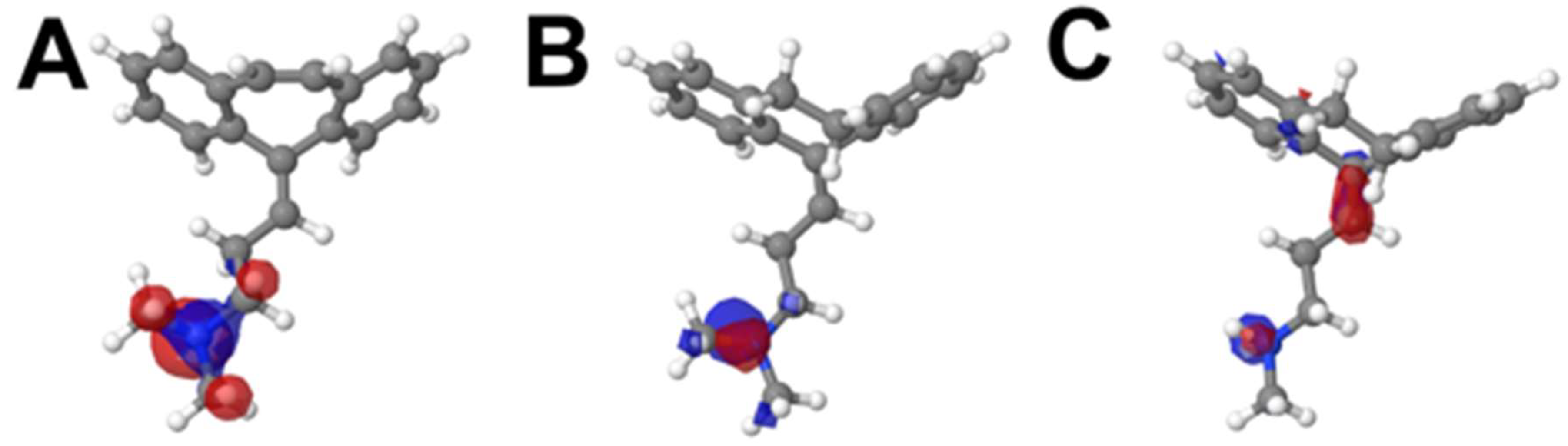
| CBP | AMP | NOR | |||
|---|---|---|---|---|---|
| Atom Number | Mulliken Charge | Atom Number | Mulliken Charge | Atom Number | Mulliken Charge |
| C1 | −0.042401 | C1 | −0.112381 | C1 | −0.119449 |
| C2 | 0.039541 | C2 | 0.442957 | C2 | 0.447457 |
| C3 | −0.027008 | C3 | 0.409320 | C3 | 0.398639 |
| C4 | 0.010317 | C4 | −0.221850 | C4 | −0.235249 |
| C5 | 0.010578 | C5 | −0.158772 | C5 | −0.143385 |
| C6 | 0.016292 | C6 | −0.316322 | C6 | −0.302634 |
| C7 | −0.027150 | C7 | 0.996592 | C7 | 0.994888 |
| C8 | −0.016568 | C8 | −0.127010 | C8 | −0.050367 |
| C9 | 0.050069 | C9 | 0.080692 | C9 | −0.001748 |
| C10 | −0.032538 | C10 | −0.584003 | C10 | −0.490114 |
| C11 | 0.015741 | N11 | 0.173777 | N11 | 0.121114 |
| C12 | 0.010368 | C12 | −0.020840 | C12 | 0.005466 |
| C13 | 0.021583 | C13 | 0.057774 | C13 | 0.036809 |
| C14 | −0.054836 | C14 | −0.253322 | C14 | −0.238812 |
| C15 | −0.066515 | C15 | −0.088678 | C15 | −0.129595 |
| C16 | 0.112695 | C16 | 0.311043 | C16 | 0.321218 |
| C17 | −0.019144 | C17 | −0.196833 | C17 | −0.204345 |
| C18 | 0.142342 | C18 | −0.174628 | C18 | −0.128244 |
| N19 | −0.416923 | C19 | −0.415570 | C19 | −0.424846 |
| C20 | 0.139806 | C20 | 0.184025 | C20 | 0.143199 |
| C21 | 0.133751 | C21 | 0.014030 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, E.S.B.; de Macêdo, I.Y.L.; da Silva Lima, L.L.; Thomaz, D.V.; da Cunha, C.E.P.; Teles de Oliveira, M.; Ballaminut, N.; Alecrim, M.F.; Ferreira de Carvalho, M.; Isecke, B.G.; et al. Electrochemical Characterization of Central Action Tricyclic Drugs by Voltammetric Techniques and Density Functional Theory Calculations. Pharmaceuticals 2019, 12, 116. https://doi.org/10.3390/ph12030116
Rodrigues ESB, de Macêdo IYL, da Silva Lima LL, Thomaz DV, da Cunha CEP, Teles de Oliveira M, Ballaminut N, Alecrim MF, Ferreira de Carvalho M, Isecke BG, et al. Electrochemical Characterization of Central Action Tricyclic Drugs by Voltammetric Techniques and Density Functional Theory Calculations. Pharmaceuticals. 2019; 12(3):116. https://doi.org/10.3390/ph12030116
Chicago/Turabian StyleRodrigues, Edson Silvio Batista, Isaac Yves Lopes de Macêdo, Larissa Lesley da Silva Lima, Douglas Vieira Thomaz, Carlos Eduardo Peixoto da Cunha, Mayk Teles de Oliveira, Nara Ballaminut, Morgana Fernandes Alecrim, Murilo Ferreira de Carvalho, Bruna Guimarães Isecke, and et al. 2019. "Electrochemical Characterization of Central Action Tricyclic Drugs by Voltammetric Techniques and Density Functional Theory Calculations" Pharmaceuticals 12, no. 3: 116. https://doi.org/10.3390/ph12030116
APA StyleRodrigues, E. S. B., de Macêdo, I. Y. L., da Silva Lima, L. L., Thomaz, D. V., da Cunha, C. E. P., Teles de Oliveira, M., Ballaminut, N., Alecrim, M. F., Ferreira de Carvalho, M., Isecke, B. G., Carneiro de Siqueira Leite, K., Machado, F. B., Guimarães, F. F., Menegatti, R., Somerset, V., & de Souza Gil, E. (2019). Electrochemical Characterization of Central Action Tricyclic Drugs by Voltammetric Techniques and Density Functional Theory Calculations. Pharmaceuticals, 12(3), 116. https://doi.org/10.3390/ph12030116







