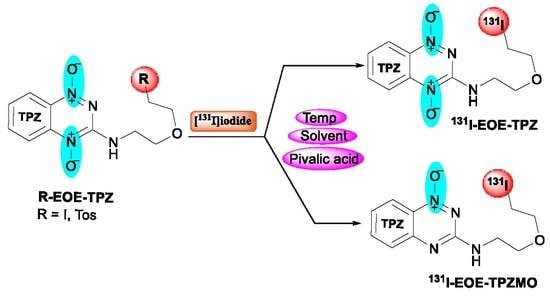
Development of [131I]I-EOE-TPZ and [131I]I-EOE-TPZMO: Novel Tirapazamine (TPZ)-Based Radioiodinated Pharmaceuticals for Application in Theranostic Management of Hypoxia
Abstract
1. Introduction
2. Results
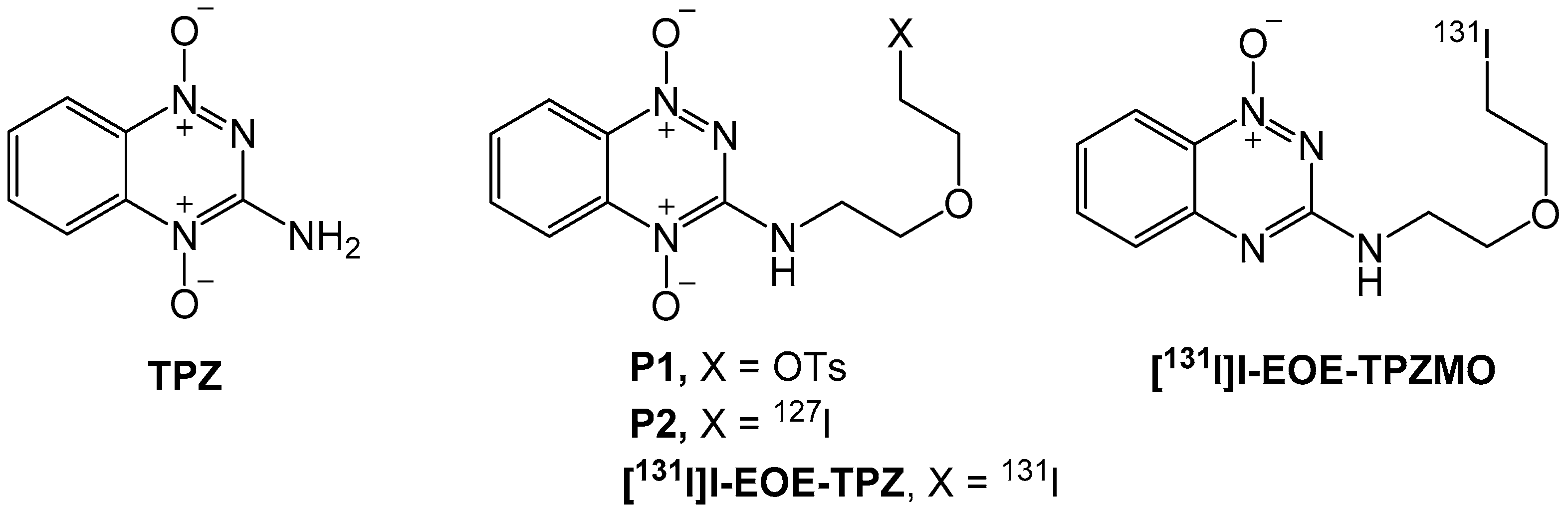
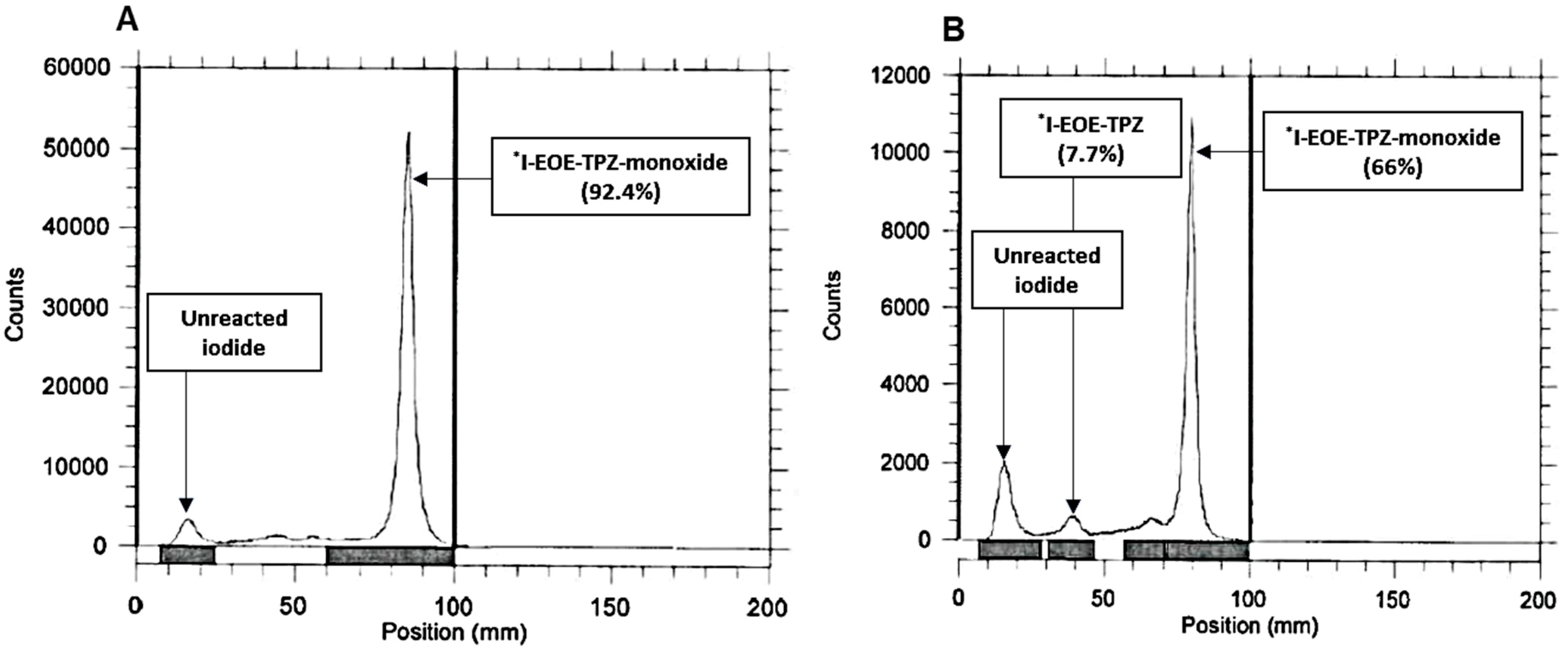
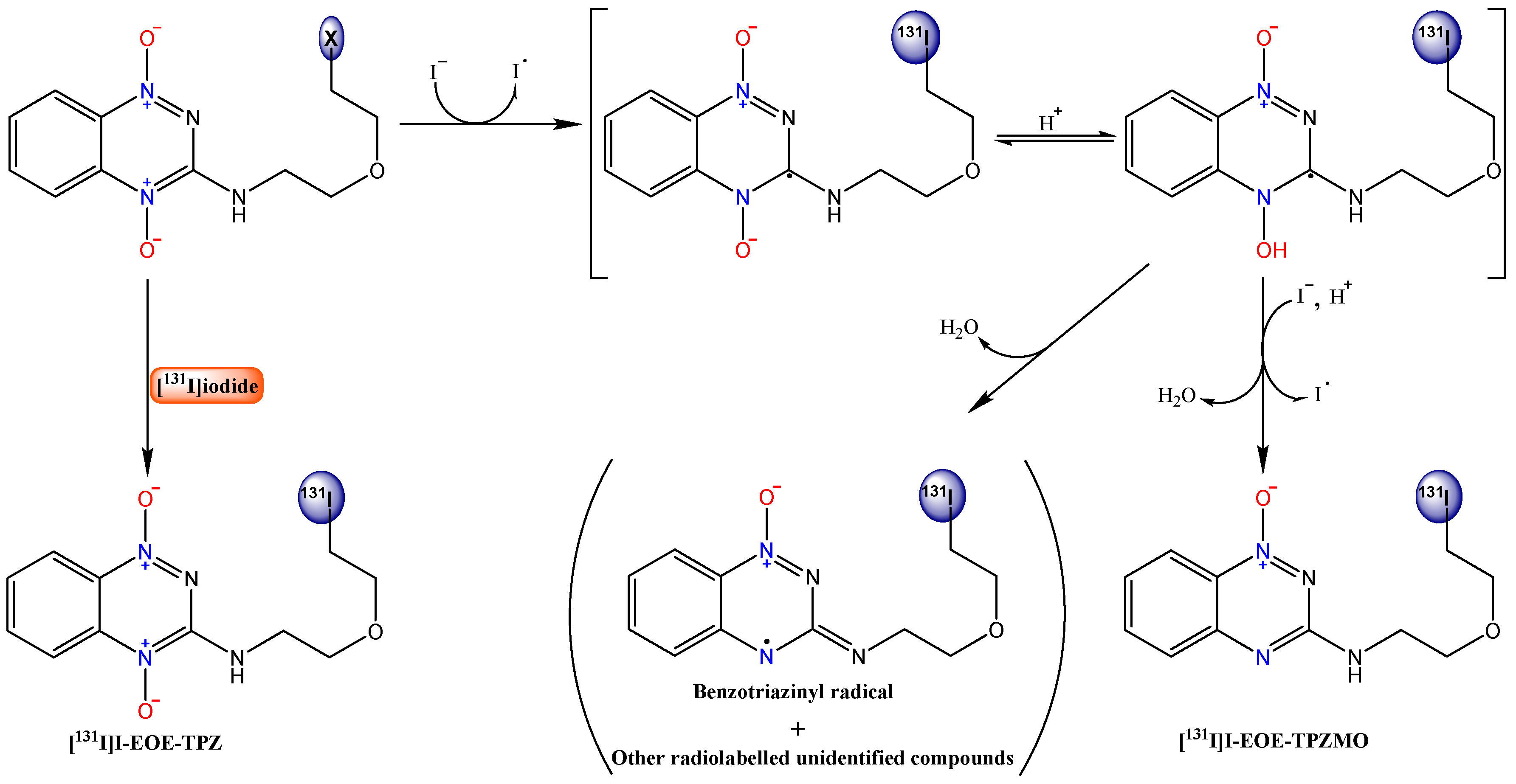
2.1. Nucleophilic Radioiodination of P1
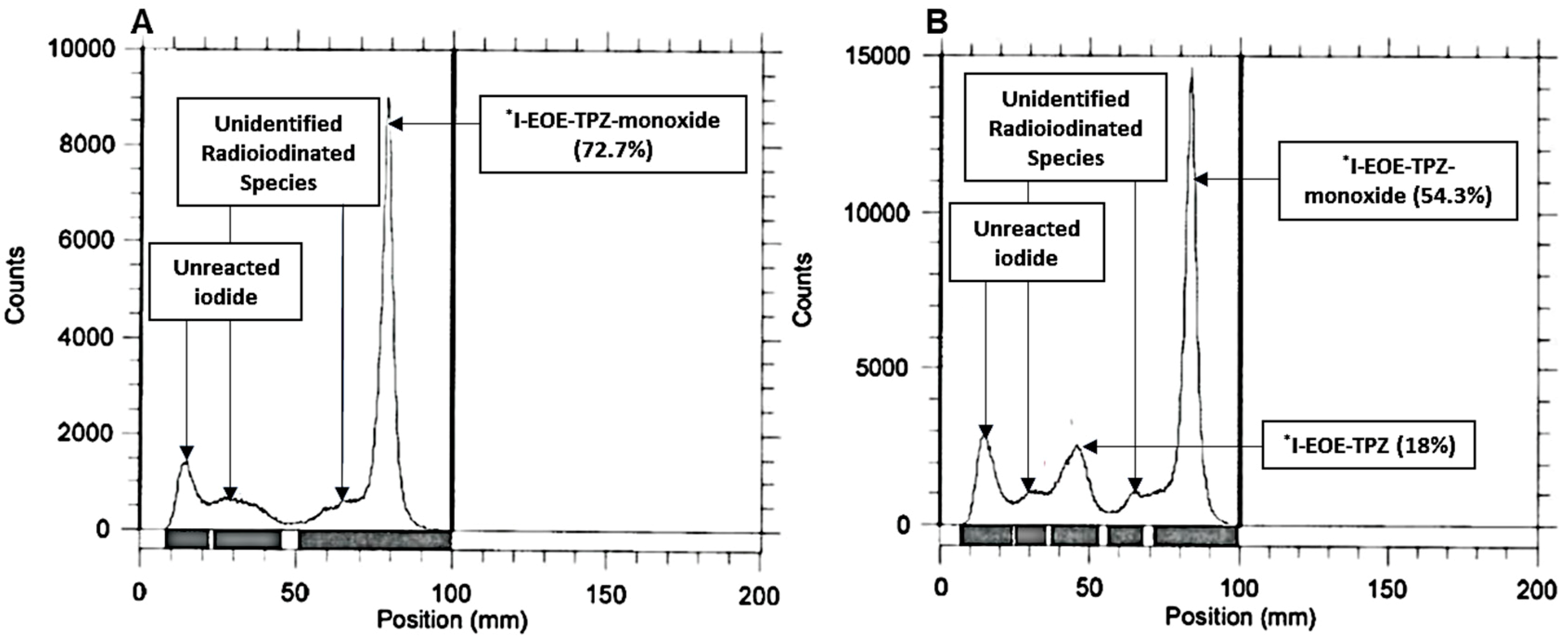
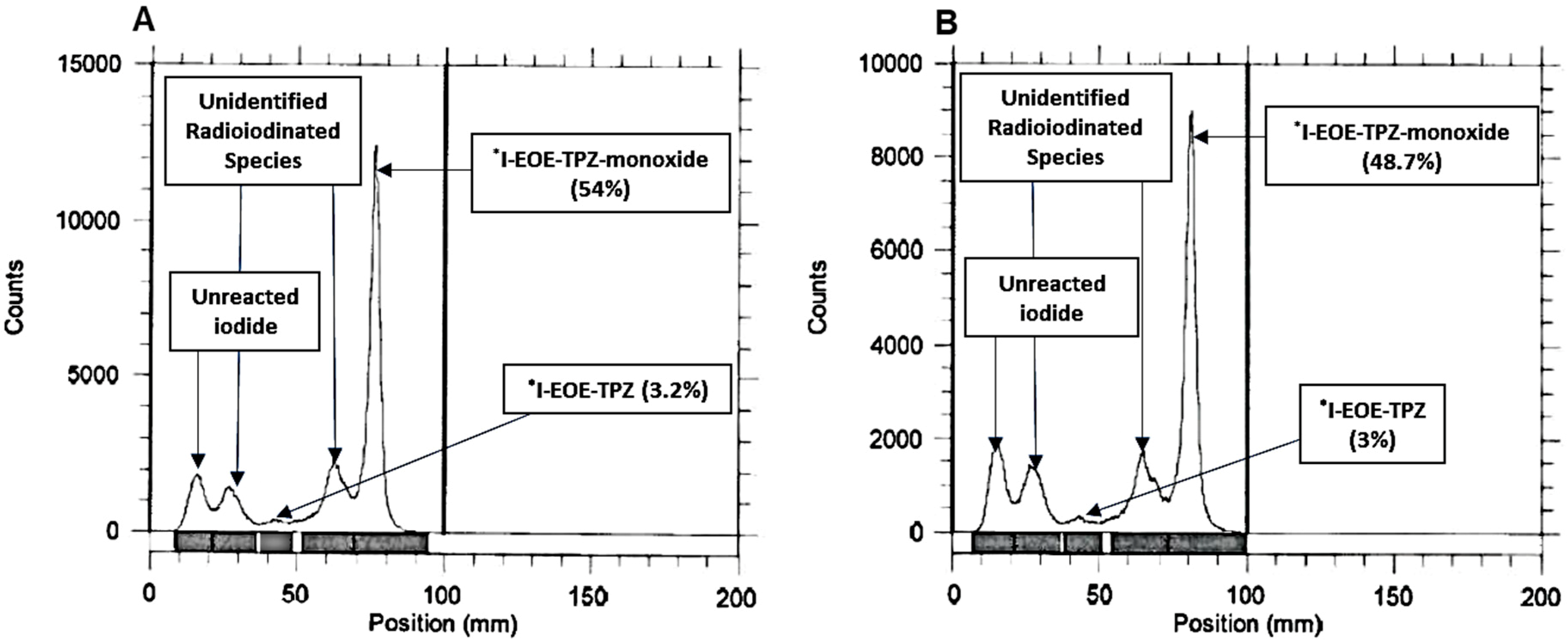
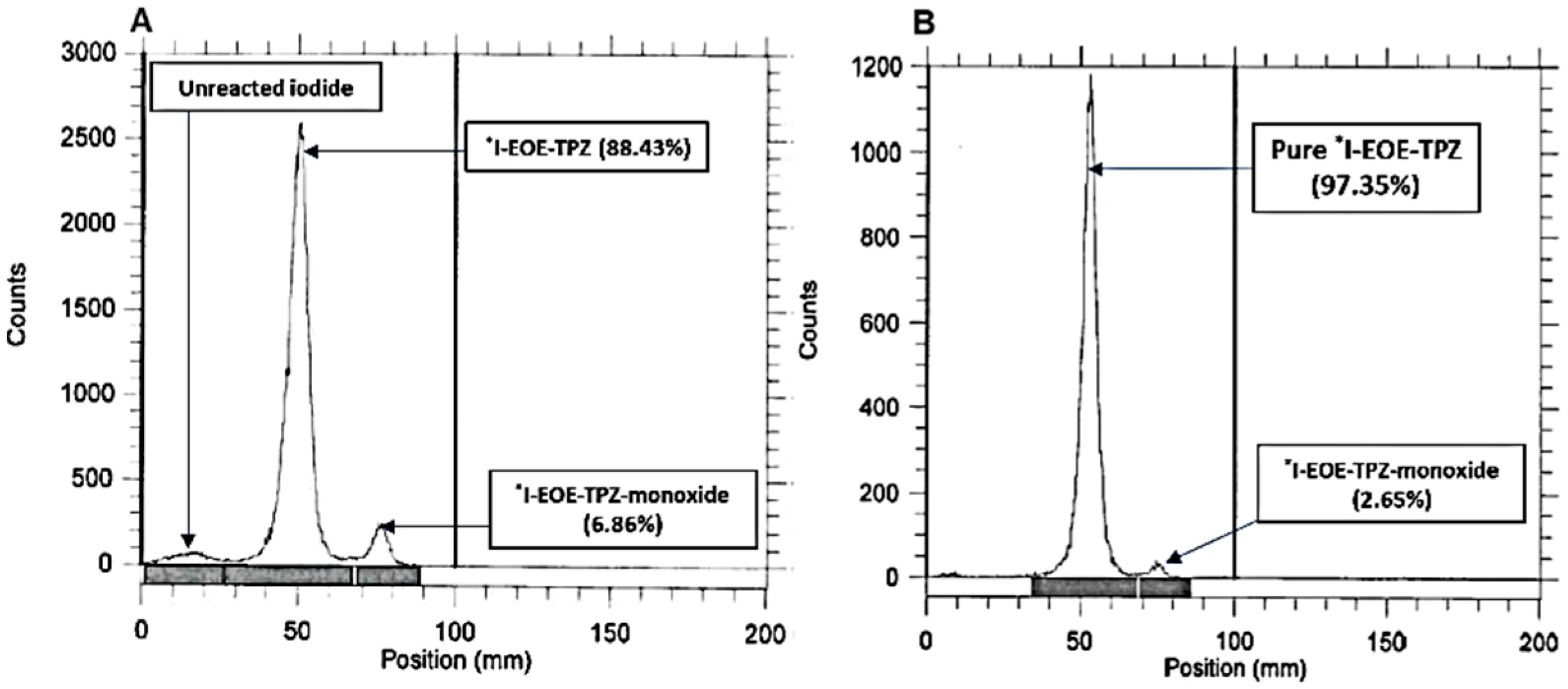
2.2. Isotope Exchange Radioiodination of P2
3. Discussion
4. Experimental
4.1. Materials
4.2. Methods
4.2.1. Nucleophilic Radioiodination of Tos-EOE-TPZ (Precursor P1)
4.2.2. Halogen Isotope Exchange Radioiodination of I-EOE-TPZ (Precursor P2)
4.3. Cartridge-Based Purification
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vaupel, P.; Mayer, A. Hypoxia in cancer: Significance and impact on clinical outcome. Cancer Metast. Rev. 2007, 26, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.T.; Boss, M.K.; Dewhirst, M.W. Imaging tumor hypoxia to advance radiation oncology. Antioxid. Redox Signal. 2014, 21, 313–337. [Google Scholar] [CrossRef] [PubMed]
- Challapalli, A.; Carroll, L.; Aboagye, E.O. Molecular mechanisms of hypoxia in cancer. Clin. Transl. Imaging 2017, 5, 225–253. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, J.Y. Targeting tumor adaption to chronic hypoxia: Implications for drug resistance, and how it can be overcome. Int. J. Mol. Sci. 2017, 18, 1854. [Google Scholar]
- Fleming, I.N.; Manavaki, R.; Blower, P.J.; West, C.; Williams, K.J.; Harris, A.L.; Domarkas, J.; Lord, S.; Baldry, C.; Gilbert, F.J. Imaging tumor hypoxia with positron emission tomography. Br. J. Cancer 2015, 112, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.R.W.; Hay, M.P.M. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 2011, 11, 393–410. [Google Scholar] [CrossRef] [PubMed]
- Epel, B.; Halpern, H.J. In Vivo pO2 Imaging of tumors: Oxymetry with very low-frequency electron paramagnetic resonance. Methods Enzymol. 2015, 564, 501–527. [Google Scholar] [PubMed]
- Bernsen, M.R.; Kooiman, K.; Segbers, M.; van Leeuwen, F.W.; de Jong, M. Biomarkers in preclinical cancer imaging. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 579–596. [Google Scholar] [CrossRef] [PubMed]
- Martelli, C.; Lo Dico, A.; Diceglie, C.; Lucignani, G.; Ottobrini, L. Optical imaging probes in oncology. Oncotarget 2016, 7, 48753–48787. [Google Scholar] [CrossRef] [PubMed]
- Winfield, J.M.; Payne, G.S.; Weller, A.; deSouza, N.M. DCE-MRI, DW-MRI, and MRS in cancer: Challenges and advantages of implementing qualitative and quantitative multi-parametric imaging in the clinic. Top. Magn. Reson. Imaging 2016, 25, 245–254. [Google Scholar] [CrossRef]
- Cabral, P.; Cerecetto, H. Radiopharmaceuticals in tumor hypoxia imaging: A review focused on medicinal chemistry aspects. Anticancer Agents Med. Chem. 2017, 17, 318–332. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Bacchu, V.; Wiebe, L.I. The chemistry and radiochemistry of hypoxia-specific, radiohalogenated nitroaromatic imaging probes. Semin. Nucl. Med. 2015, 45, 122–135. [Google Scholar] [CrossRef] [PubMed]
- Ricardo, C.L.; Kumar, P.; Wiebe, L.I. Bifunctional metal-nitroimidazole complexes for hypoxia theranosis in cancer. J. Diagn. Imaging Ther. 2015, 2, 103–158. [Google Scholar] [CrossRef]
- Brown, J.M. SR 4233 (Tirapazamine): A new anticancer drug exploiting hypoxia in solid tumors. Br. J. Cancer 1993, 67, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Robbins, R.F.; Schofield, K. Polyazabicyclic compounds. Part II. Further derivatives of benzo-1:2:4-triazine. J. Chem. Soc. 1957. [Google Scholar] [CrossRef]
- Mason, J.C.; Tennant, G. Heterocyclic N-oxides. Part VI. Synthesis and nuclear magnetic resonance spectra of 3-aminobenzo-1,2,4-triazines and their mono- and di-N-oxides. J. Chem. Soc. B 1970, 911–916. [Google Scholar] [CrossRef]
- Zeman, E.M.; Brown, J.M.; Lemmon, M.J.; Hirst, V.K.; Lee, W.W. SR 4233: A new bioreductive agent with high selective toxicity for hypoxic mammalian cells. Int. J. Radiat. Oncol. Biol. Phys. 1986, 12, 1239–1242. [Google Scholar] [CrossRef]
- Zeman, E.M.; Hirst, V.K.; Lemmon, M.J.; Brown, J.M. Enhancement of radiation-induced tumor cell killing by the hypoxic cell toxin SR 4233. Radiother. Oncol. 1988, 12, 209–218. [Google Scholar] [CrossRef]
- Chopra, S.; Koolpe, G.A.; Tambo-Ong, A.A.; Matsuyama, K.N.; Ryan, K.J.; Tran, T.B.; Doppalapudi, R.S.; Riccio, E.S.; Iyer, L.V.; Green, C.E.; et al. Discovery and optimization of benzotriazine di-N-oxides targeting replicating and nonreplicating Mycobacterium tuberculosis. J. Med. Chem. 2012, 55, 6047–6060. [Google Scholar] [CrossRef]
- Xia, Q.; Zhang, L.; Zhang, J.; Sheng, R.; Yang, B.; He, Q.; Hu, Y. Synthesis, hypoxia-selective cytotoxicity of new 3-amino-1,2,4-benzotriazine-1,4-dioxide derivatives. Eur. J. Med. Chem. 2011, 46, 919–926. [Google Scholar] [CrossRef]
- Hay, M.P.; Hicks, K.O.; Pchalek, K.; Lee, H.H.; Blaser, A.; Pruijn, F.B.; Anderson, A.F.; Shinde, S.S.; Wilson, W.R.; Denny, W.A. Tricyclic [1,2,4]Triazine 1,4-Dioxides as hypoxia selective cytotoxins. J. Med. Chem. 2008, 51, 6853–6865. [Google Scholar] [CrossRef] [PubMed]
- Hay, M.P.; Gamage, S.A.; Kovacs, M.S.; Pruijn, F.B.; Anderson, R.F.; Patterson, A.V.; Wilson, W.R.; Brown, M.; Denny, W.A. Structure−activity relationships of 1,2,4-benzotriazine 1,4-dioxides as hypoxia-selective analogues of tirapazamine. J. Med. Chem. 2003, 46, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Elsaidi, H.R.H.; Yang, X.-H.; Ahmadi, F.; Weinfeld, M.; Wiebe, L.I.; Kumar, P. Putative electron-affinic radiosensitizers and markers of hypoxic tissue: Synthesis and preliminary in vitro biological characterization of C3-amino-substituted benzotriazine dioxides. Eur. J. Med. Chem. 2018. submitted. [Google Scholar]
- Yin, J.; Glaser, R.; Gates, K.S. On the reaction mechanism of tirapazamine reduction chemistry: Unimolecular N–OH homolysis, stepwise dehydration, or triazene ring-opening. Chem. Res. Toxicol. 2012, 25, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.F.; Shinde, S.S.; Hay, M.P.; Gamage, S.A.; Denny, W.A. Activation of 3-amino-1,2,4-benzotriazine 1,4-dioxide antitumor agents to oxidizing species following their one-electron reduction. J. Am. Chem. Soc. 2003, 125, 748–756. [Google Scholar] [CrossRef]
- Siim, B.G.; Pruijn, F.B.; Sturman, J.R.; Hogg, A.; Hay, M.P.; Brown, J.M.; Wilson, W.R. Selective potentiation of the hypoxic cytotoxicity of tirapazamine by its 1-N-oxide metabolite SR 4317. Cancer Res. 2004, 64, 736–742. [Google Scholar] [CrossRef]
- Yoo, B.W.; Park, M.C. Mild and efficient deoxygenation of amine-N-oxides with MoCl5/NaI system. Synth. Commun. 2008, 38, 1646–1650. [Google Scholar] [CrossRef]
- Revuelta, J.; Cicchi, S.; Brandi, A. Samarium(II) iodide reduction of isoxazolidines. Tetrahedron Lett. 2004, 45, 8375–8377. [Google Scholar] [CrossRef]
- Singh, D.; Singh, V.; Rai, B.P. An efficient method for the reduction of cephalosporin sulfoxide. Asian J. Chem. 2007, 19, 5787–5789. [Google Scholar]
- Das, A.K.; Srivastav, M.; Layek, R.K.; Uddin, M.E.; Jung, D.; Kim, N.H.; Lee, J.H. Iodide-mediated room temperature reduction of graphene oxide: A rapid chemical route for the synthesis of a bifunctional electrocatalyst. J. Mater. Chem. A 2014, 2, 1332–1340. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, R. Some deoxygenation and reduction reactions with samarium diiodide. Synth. Commun. 1987, 17, 329–332. [Google Scholar] [CrossRef]
- Weichert, J.P.; Van Dort, M.E.; Groziak, M.P.; Counsell, R.E. Radioiodination via isotope exchange in pivalic acid. Int. J. Rad. Appl. Instrum. A 1986, 37, 907–913. [Google Scholar] [CrossRef]
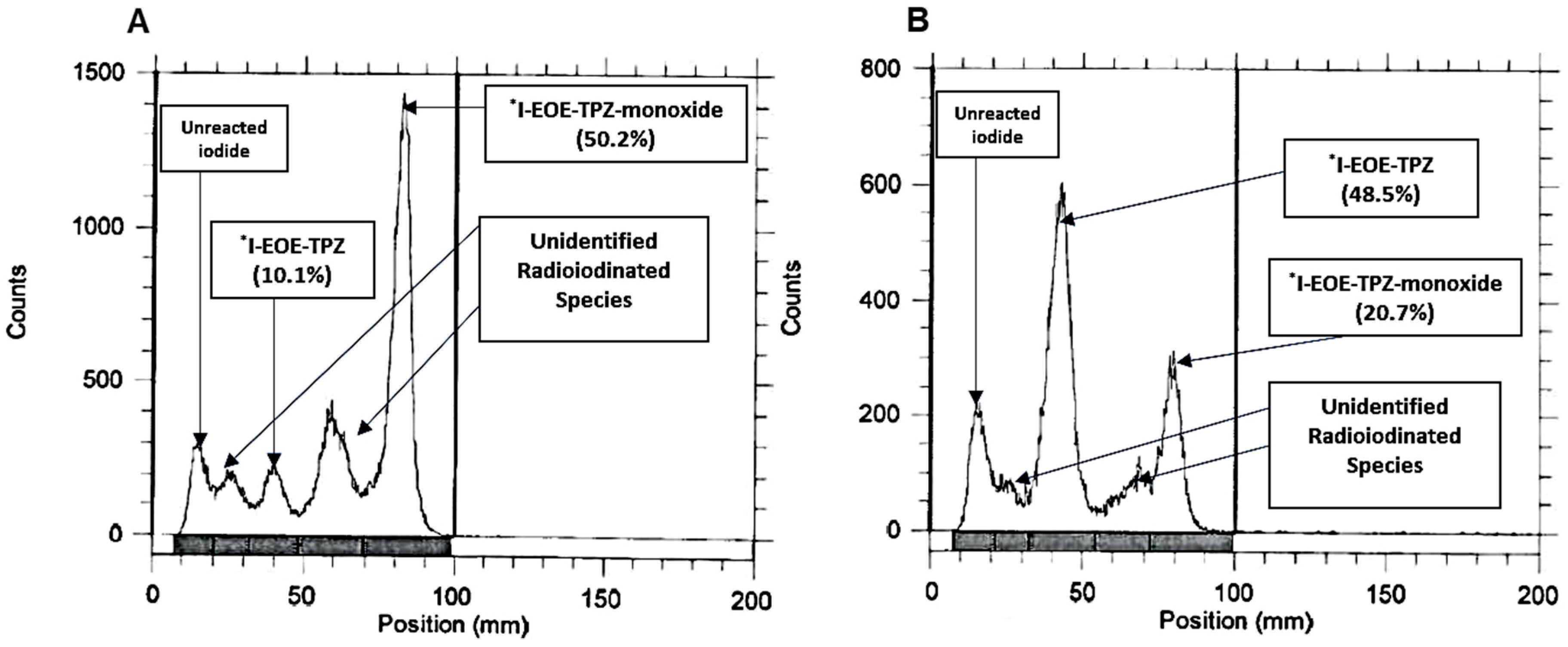
| Solvent | Temp (°C) | Time (min) | [131I]I-EOE-TPZ % of Total [131I] | [131I]I-EOE-TPZMO % of Total [131I] |
|---|---|---|---|---|
| ACN | 22 | 30 and 60 | 0, 0 | 0, 0 |
| ACN | 60 | 30 and 60 | 0, 0 | 17.2, 31.2 |
| ACN | 80 | 60 | 48.5 | 20.7 |
| ACN | 100 | 60 | 10.1 | 50.2 |
| DMF | 22 | 30 and 90 | 0, 0 | 0, 0 |
| DMF DMF DMF | 60 80 100 | 30 and 60 60 60 | 0, 0 24.6 7.8 | 0, 0 |
| 16.7 | ||||
| 34.2 |
| Solvent | Temp (°C) | Time (min) | Pivalic Acid (mg) | [131I]I-EOE-TPZ % of Total [131I] | [131I]I-EOE-TPZMO % of Total [131I] |
|---|---|---|---|---|---|
| DMF | 22 | 30, 60 | 0 | 0, 18 | 72.7, 54.3 |
| ACN | 22 | 30, 60 | 0 | 0, 0 | 34.7, 7.5 |
| ACN | 80 | 30 | 0 | 89 | 6.9 |
| ACN | 50 | 30, 60 | 3.5 | 0, 7.7 | 92.4, 66 |
| ACN | 22 | 30, 60, 90 | 3.5 | 3.2, 3.5, 3 | 54, 50, 48.7 |
| EtOH/ACN | 22 | 30, 60, 90 | 3.5 | 0, 0, 0 | 43, 42.4, 42.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsaidi, H.; Ahmadi, F.; Wiebe, L.I.; Kumar, P. Development of [131I]I-EOE-TPZ and [131I]I-EOE-TPZMO: Novel Tirapazamine (TPZ)-Based Radioiodinated Pharmaceuticals for Application in Theranostic Management of Hypoxia. Pharmaceuticals 2019, 12, 3. https://doi.org/10.3390/ph12010003
Elsaidi H, Ahmadi F, Wiebe LI, Kumar P. Development of [131I]I-EOE-TPZ and [131I]I-EOE-TPZMO: Novel Tirapazamine (TPZ)-Based Radioiodinated Pharmaceuticals for Application in Theranostic Management of Hypoxia. Pharmaceuticals. 2019; 12(1):3. https://doi.org/10.3390/ph12010003
Chicago/Turabian StyleElsaidi, Hassan, Fatemeh Ahmadi, Leonard I. Wiebe, and Piyush Kumar. 2019. "Development of [131I]I-EOE-TPZ and [131I]I-EOE-TPZMO: Novel Tirapazamine (TPZ)-Based Radioiodinated Pharmaceuticals for Application in Theranostic Management of Hypoxia" Pharmaceuticals 12, no. 1: 3. https://doi.org/10.3390/ph12010003
APA StyleElsaidi, H., Ahmadi, F., Wiebe, L. I., & Kumar, P. (2019). Development of [131I]I-EOE-TPZ and [131I]I-EOE-TPZMO: Novel Tirapazamine (TPZ)-Based Radioiodinated Pharmaceuticals for Application in Theranostic Management of Hypoxia. Pharmaceuticals, 12(1), 3. https://doi.org/10.3390/ph12010003