Evaluation of Radiolabeled Girentuximab In Vitro and In Vivo
Abstract
1. Introduction
2. Results
2.1. Conjugation of p-SCN-βn-DOTA to cG250
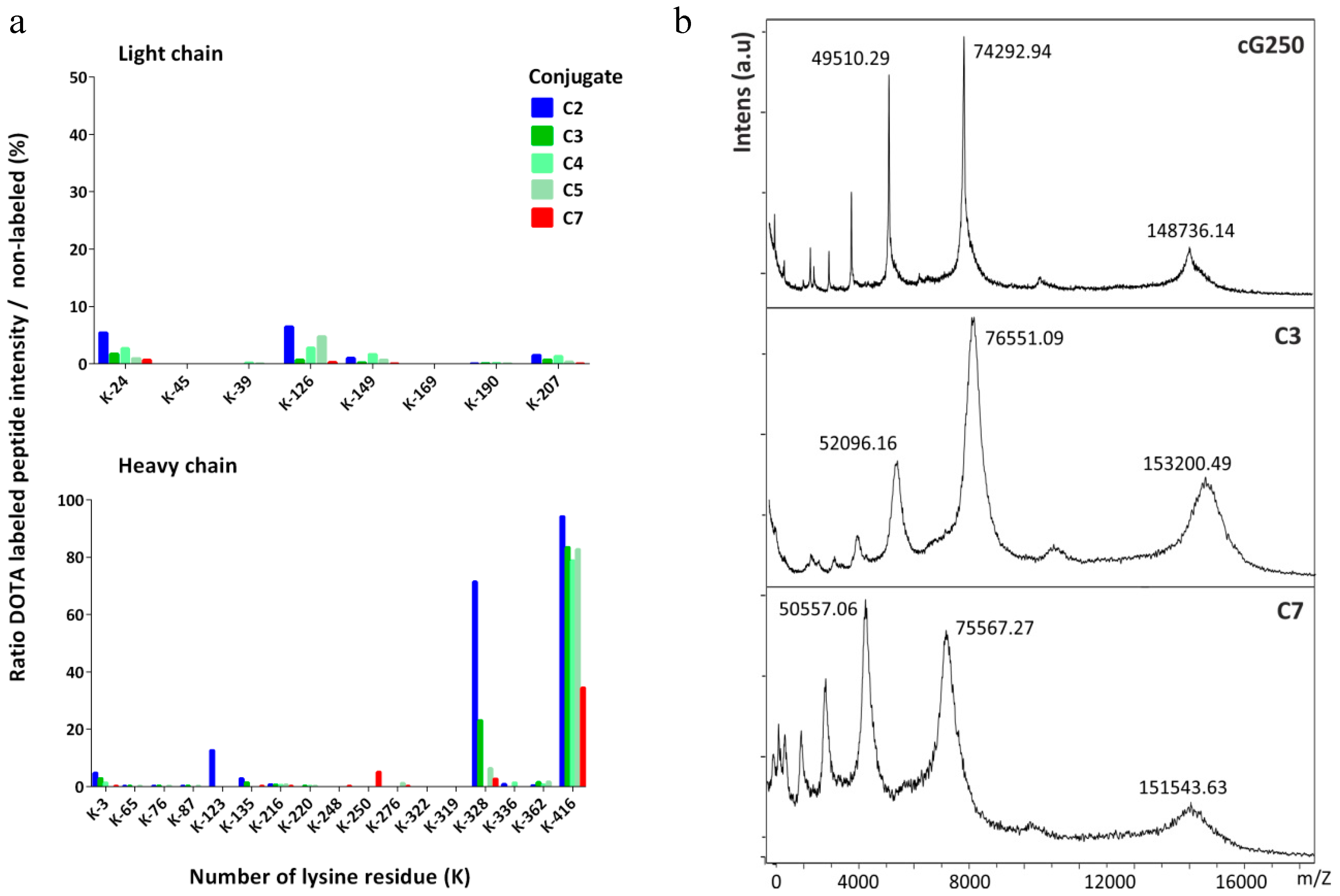
2.2. Identification of DOTA Modification Sites
2.3. Ratio of DOTA Molecules Per Molecule of Antibody
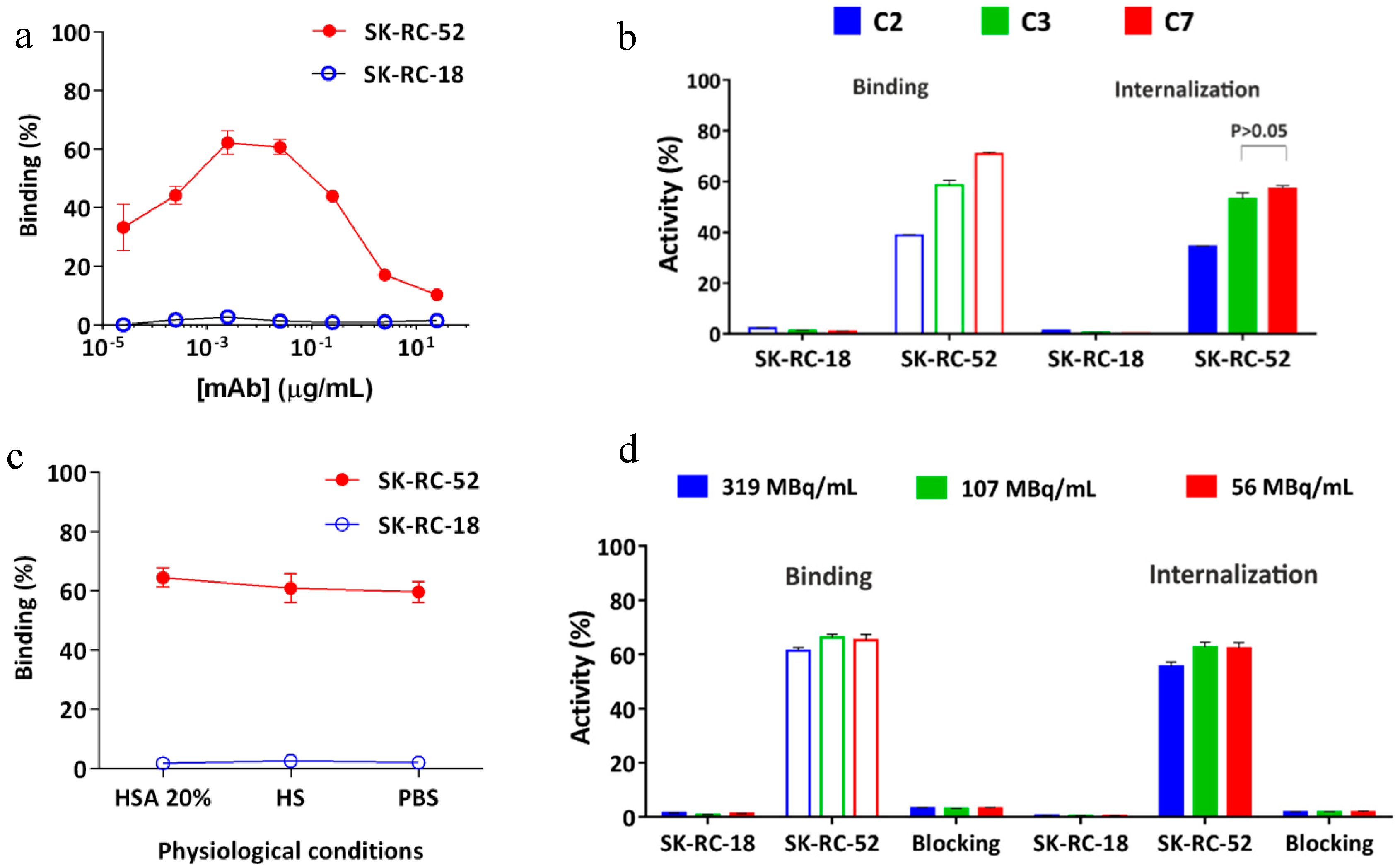
2.4. In Vitro Characterization of the Conjugates
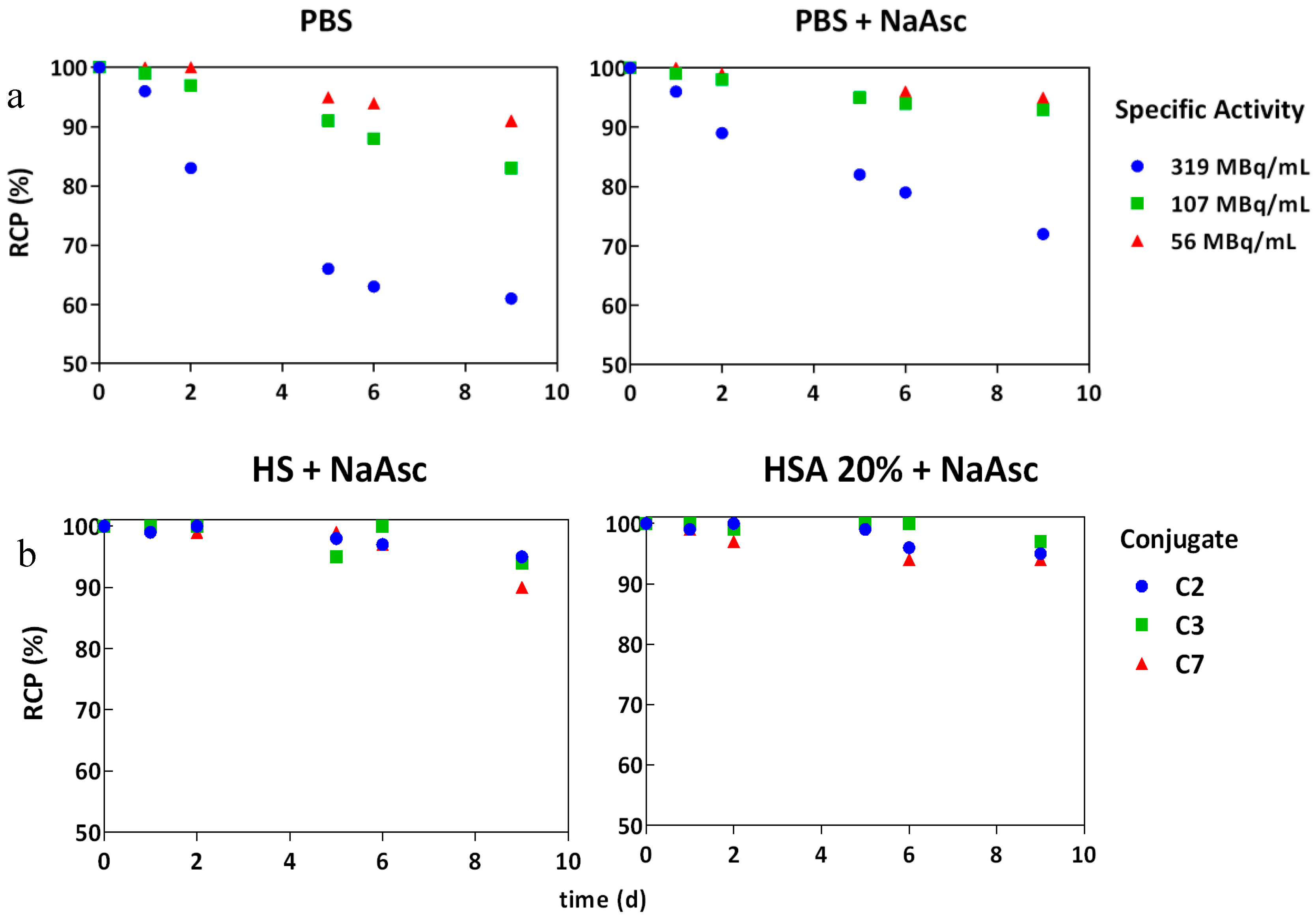
2.5. Radiochemical Purity and Influence of Sodium Ascorbate on the Stability In Vitro
2.6. In Vitro Radioimmunoreactivity of the Conjugates Labeled with 177Lu
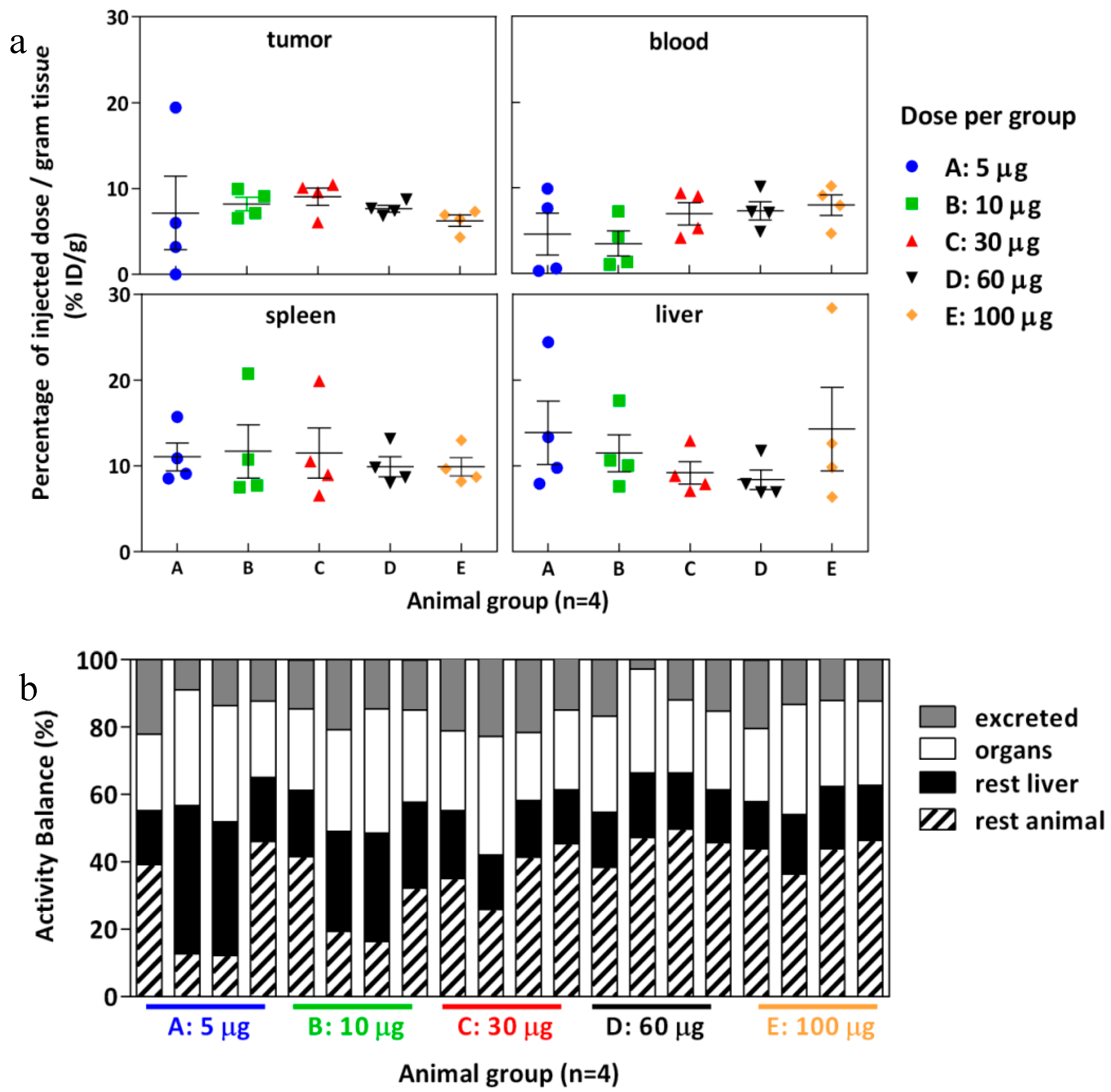
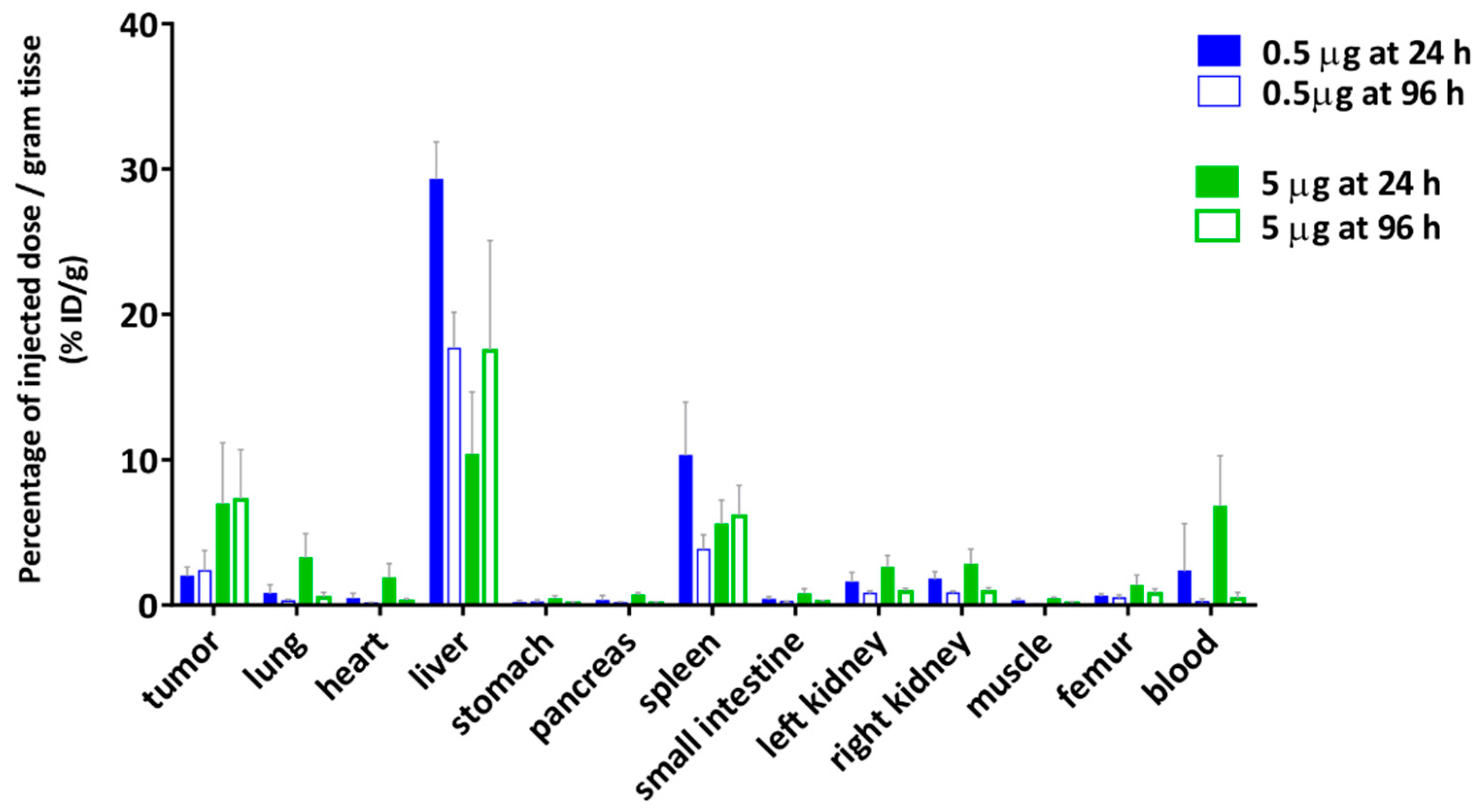
2.7. Biodistribution
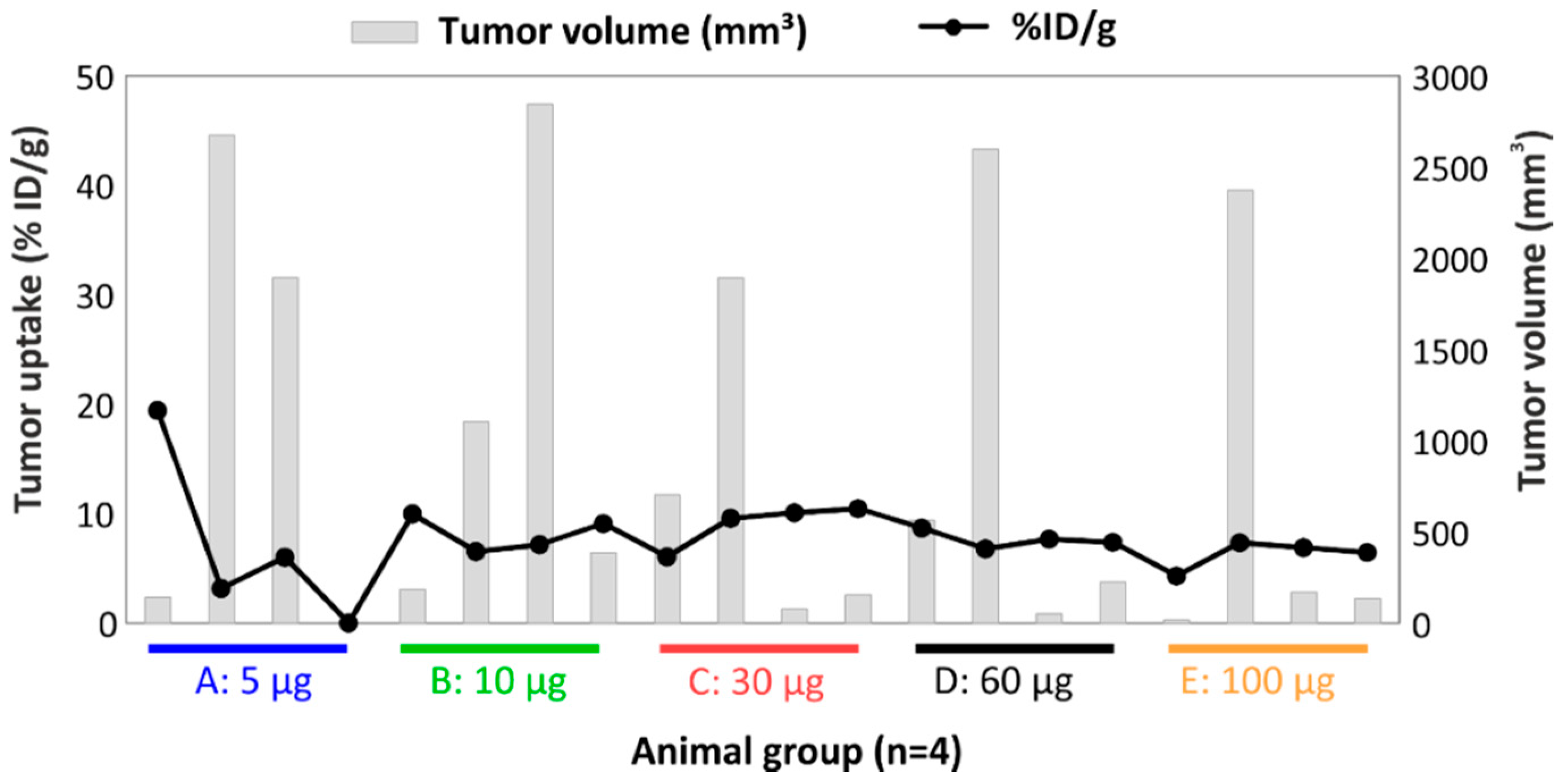
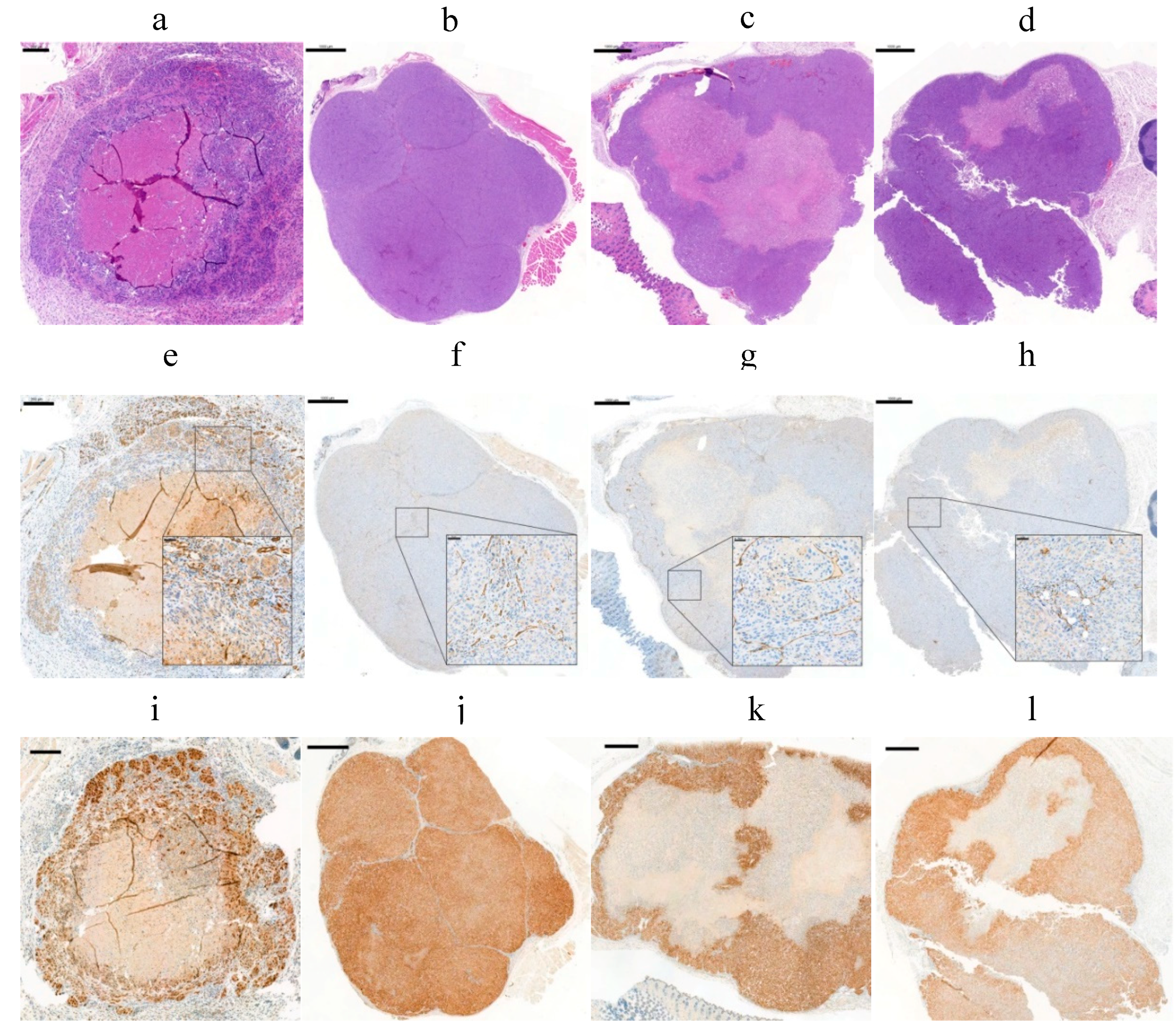
2.8. Effect of Tumor Volume, Hypoxia, and Necrosis on cG250 Tumor Retention
3. Discussion
4. Materials and Methods
4.1. Preparation of cG250 Conjugates
4.2. Characterization by Mass Spectrometry
4.3. Cell Culture
4.4. Flow Cytometric Analysis
4.5. Radiolabeling, Quality Control, and Radiostability
4.6. Radioactivity Binding Assay
4.7. Animals
4.8. Immunohistochemical Analysis (IHC)
4.9. Biodistribution
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Ab | antibody |
| ADC | antibody drug conjugate |
| Asn, N | asparagine amino residues |
| BM | biomolecule |
| BFC | bifunctional chelator |
| BSA | bovine serum albumin |
| CAIX | carbonic anhydrase IX |
| cG250 | monoclonal antibody anti-CAIX, girentuximab |
| Cys, C | cysteine amino residues |
| CDR | complementarity-determining regions |
| CHCA | hydroxycinnamic acid |
| CD31 | cluster of differentiation 31, endotelial cells marker |
| DOTA | tetraxetan or 1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid |
| DOTA(SCN) | p-SCN-Bn-DOTA or S-2-(4-Isothiocyanatobenzyl)-1,4,7,10-tetraazacyclododecane tetraacetic acid |
| DTPA | pentetic acid or diethylenetriaminepentaacetic acid |
| DTT | dithiothreitol |
| DCB | Department of Chemistry and Biochemistry, University of Bern, Switzerland |
| DBMR | Department for Biomedical Research, University of Bern, Switzerland |
| d | day |
| EDTA | ethylenediaminetetraacetic acid |
| ESI | electrospray ionization |
| FDR | false discovery rate |
| FFPE | formalin-fixed paraffin-embedded |
| FACS | fluorescence-activated cell sorting applied in flow cytometry |
| FCS | fetal calf serum |
| FT | Fourier transform |
| Fac | formic acid |
| Gln, Q | glutamine amino residues |
| HC | heavy chain of immunoglobulin |
| HIF-1 | hypoxia-inducible factor 1 |
| HSA | human serum albumin |
| HS | human serum |
| HNSCC | head and neck squamous cell carcinomas (HNSCC) |
| h | hour |
| HPLC | high-performance liquid chromatography |
| Ig | immunoglobulin |
| IC50 | concentration of an inhibitor where the response (or binding) is reduced by half |
| IHC | immunohistochemistry |
| ICC | immunocytochemistry |
| ITLC | instant thin layer chromatography |
| ID | injected dose |
| i.v. | intravenously |
| LET | linear energy transfer |
| LC | light chain of immunoglobulin |
| LC | liquid chromatography |
| Lys, K | lysine amino residues |
| LRC | Laboratory of Radiochemistry, Paul Scherrer Institute, Villigen PSI, Switzerland |
| mAb | monoclonal antibody |
| MBq | megabecquerel |
| Met, M | methionine amino residues |
| MALDI | matrix assisted laser desorption/ionization–mass spectrometry |
| MS | mass spectrometry |
| MW | molecular weight or mass |
| mgf | mascot generic file format |
| MSKCC | Memorial Sloan Kettering Cancer Centre, New York, USA |
| min | minute |
| NaAsc | sodium ascorbate |
| NaCl | saline solution |
| NHL | non-Hodgkin’s lymphoma |
| NHS | N-hydroxysuccinimide |
| OCT | optimum cutting temperature |
| PKM | pharmacokinetic modifying linker |
| PK | pharmacokinetics |
| PET | positron emission tomography |
| PBS | phosphate buffer saline |
| ppm | parts per million (chemical shift) |
| PSI | Paul Scherrer Institute, Villigen PSI, Switzerland |
| PKI | Institute of Pharmacology, University of Bern, Switzerland |
| RIT | radioimmunotherapy |
| RCCs | renal cell carcinomas |
| RCP | radiochemical purity |
| Rf | retardation factor |
| RPMI | Roswell Park Memorial Institute medium |
| RPM | revolutions per minute |
| SEC | size exclusion chromatography |
| SDS-PAGE | sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| SCN | isothiocyanate |
| SG | silica gel |
| t1/2 | half-life |
| Tyr,Y | tyrosine amino residues |
| TOF | time of flight |
| TFA | trifluoroacetic acid |
| TRU | Translational Research Unit, University of Bern, Switzerland |
| Tris | hydroxymethylaminomethane |
| UV | ultraviolet |
References
- Fleuren, E.D.; Versleijen-Jonkers, Y.M.; Heskamp, S.; van Herpen, C.M.; Oyen, W.J.; van der Graaf, W.T.; Boerman, O.C. Theranostic applications of antibodies in oncology. Mol. Oncol. 2014, 8, 799–812. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, A.H.; van Eerd, J.E.; Frielink, C.; Oosterwijk, E.; Oyen, W.J.; Corstens, F.H.; Boerman, O.C. Optimization of radioimmunotherapy of renal cell carcinoma: Labeling of monoclonal antibody cG250 with 131I, 90Y, 177Lu, or 186Re. J. Nucl. Med. 2004, 45, 327–337. [Google Scholar] [PubMed]
- Thakral, P.; Singla, S.; Yadav, M.P.; Vasisht, A.; Sharma, A.; Gupta, S.K.; Bal, C.S.; Malhotra, A. An approach for conjugation of (177) Lu-DOTA-SCN-Rituximab (BioSim) & its evaluation for radioimmunotherapy of relapsed & refractory B-cell non Hodgkins lymphoma patients. Indian J. Med. Res. 2014, 139, 544–554. [Google Scholar] [PubMed]
- Kennel, S.J.; Brechbiel, M.W.; Milenic, D.E.; Schlom, J.; Mirzadeh, S. Actinium-225 conjugates of MAb CC49 and humanized delta CH2CC49. Cancer Biother. Radiopharm. 2002, 17, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Zalutsky, M.R.; Reardon, D.A.; Akabani, G.; Coleman, R.E.; Friedman, A.H.; Friedman, H.S.; McLendon, R.E.; Wong, T.Z.; Bigner, D.D. Clinical experience with alpha-particle emitting 211At: Treatment of recurrent brain tumor patients with 211At-labeled chimeric antitenascin monoclonal antibody 81C6. J. Nucl. Med. 2008, 49, 30–38. [Google Scholar] [CrossRef] [PubMed]
- White, C.A. Rituxan immunotherapy and zevalin radioimmunotherapy in the treatment of non-Hodgkin’s lymphoma. Curr. Pharm. Biotechnol. 2003, 4, 221–238. [Google Scholar] [CrossRef] [PubMed]
- Witzig, T.E.; Wiseman, G.A.; Maurer, M.J.; Habermann, T.M.; Micallef, I.N.; Nowakowski, G.S.; Ansell, S.M.; Colgan, J.P.; Inwards, D.J.; Porrata, L.F.; et al. A phase I trial of immunostimulatory CpG 7909 oligodeoxynucleotide and 90 yttrium ibritumomab tiuxetan radioimmunotherapy for relapsed B-cell non-Hodgkin lymphoma. Am. J. Hematol. 2013, 88, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Shadman, M.; Li, H.; Rimsza, L.; Leonard, J.P.; Kaminski, M.S.; Braziel, R.M.; Spier, C.M.; Gopal, A.K.; Maloney, D.G.; Cheson, B.D.; et al. Continued Excellent Outcomes in Previously Untreated Patients With Follicular Lymphoma After Treatment With CHOP Plus Rituximab or CHOP Plus 131I-Tositumomab: Long-Term Follow-Up of Phase III Randomized Study SWOG-S0016. J. Clin. Oncol. 2018, 36, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Ray Banerejee, S.; Kumar, V.; Lisok, A.; Plyku, D.; Novakova, Z.; Wharram, B.; Brummet, M.; Barinka, C.; Hobbs, R.F.; Pomper, M.G. Evaluation of (111)In-DOTA-5D3, a Surrogate SPECT Imaging Agent for Radioimmunotherapy of Prostate-Specific Membrane Antigen. J. Nucl. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Stillebroer, A.B.; Oosterwijk, E.; Oyen, W.J.; Mulders, P.F.; Boerman, O.C. Radiolabeled antibodies in renal cell carcinoma. Cancer Imaging 2007, 7, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Diamantis, N.; Banerji, U. Antibody-drug conjugates—An emerging class of cancer treatment. Br. J. Cancer 2016, 114, 362. Available online: https://www.nature.com/articles/bjc2015435#supplementary-information (accessed on 1 October 2018). [CrossRef] [PubMed]
- Reichert, J.M.; Valge-Archer, V.E. Development trends for monoclonal antibody cancer therapeutics. Nat. Rev. Drug Discov. 2007, 6, 349. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, R.M.; Goldenberg, D.M. Perspectives on Cancer Therapy with Radiolabeled Monoclonal Antibodies. J. Nucl. Med. 2005, 46, 115S–127S. [Google Scholar] [PubMed]
- Sands, H.; Jones, P.L.; Shah, S.A.; Palme, D.; Vessella, R.L.; Gallagher, B.M. Correlation of vascular permeability and blood flow with monoclonal antibody uptake by human Clouser and renal cell xenografts. Cancer Res. 1988, 48, 188–193. [Google Scholar] [PubMed]
- Chakrabarti, M.C.; Le, N.; Paik, C.H.; De Graff, W.G.; Carrasquillo, J.A. Prevention of radiolysis of monoclonal antibody during labeling. J. Nucl. Med. 1996, 37, 1384–1388. [Google Scholar] [PubMed]
- Macey, D.J.; Meredith, R.F. A strategy to reduce red marrow dose for intraperitoneal radioimmunotherapy. Clin. Cancer Res. 1999, 5, 3044s–3047s. [Google Scholar] [PubMed]
- Read, E.D.; Eu, P.; Little, P.J.; Piva, T.J. The status of radioimmunotherapy in CD20+ non-Hodgkin’s lymphoma. Target Oncol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Wilder, R.B.; DeNardo, G.L.; DeNardo, S.J. Radioimmunotherapy: Recent results and future directions. J. Clin. Oncol. 1996, 14, 1383–1400. [Google Scholar] [CrossRef] [PubMed]
- IAEA. Comparative Evaluation of Therapeutic Radiopharmaceuticals; International Atomic Energy Agency: Vienna, Austria, 2007; Available online: https://www-pub.iaea.org/MTCD/publications/PDF/TRS458_web.pdf (accessed on 28 November 2018).
- Dash, A.; Pillai, M.R.; Knapp, F.F., Jr. Production of (177)Lu for Targeted Radionuclide Therapy: Available Options. Nucl. Med. Mol. Imaging 2015, 49, 85–107. [Google Scholar] [CrossRef] [PubMed]
- Stimmel, J.B.; Kull, F.C. Samarium-153 and Lutetium-177 Chelation Properties of Selected Macrocyclic and Acyclic Ligands. Nucl. Med. Biol. 1998, 25, 117–125. [Google Scholar] [CrossRef]
- Yordanov, A.T.; Hens, M.; Pegram, C.; Bigner, D.D.; Zalutsky, M.R. Antitenascin antibody 81C6 armed with 177Lu: In vivo comparison of macrocyclic and acyclic ligands. Nucl. Med. Biol. 2007, 34, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.S.; Sun, X.; Jia, F.; Song, H.A.; Chen, Y.; Lewis, M.; Chong, H.-S. Synthesis and Preclinical Evaluation of Bifunctional Ligands for Improved Chelation Chemistry of 90Y and 177Lu for Targeted Radioimmunotherapy. Bioconjug. Chem. 2012, 23, 1775–1782. [Google Scholar] [CrossRef] [PubMed]
- Hens, M.; Vaidyanathan, G.; Zhao, X.G.; Bigner, D.D.; Zalutsky, M.R. Anti-EGFRvIII monoclonal antibody armed with 177Lu: In vivo comparison of macrocyclic and acyclic ligands. Nucl. Med. Biol. 2010, 37, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Edwards, D.S. Bifunctional chelators for therapeutic lanthanide radiopharmaceuticals. Bioconjug. Chem. 2001, 12, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Milenic, D.E.; Garmestani, K.; Chappell, L.L.; Dadachova, E.; Yordanov, A.; Ma, D.; Schlom, J.; Brechbiel, M.W. In vivo comparison of macrocyclic and acyclic ligands for radiolabeling of monoclonal antibodies with 177Lu for radioimmunotherapeutic applications. Nucl. Med. Biol. 2002, 29, 431–442. [Google Scholar] [CrossRef]
- Liu, S. The role of coordination chemistry in the development of target-specific radiopharmaceuticals. Chem. Soc. Rev. 2004, 33, 445–461. [Google Scholar] [CrossRef] [PubMed]
- Liu, S. Bifunctional coupling agents for radiolabeling of biomolecules and target-specific delivery of metallic radionuclides. Adv. Drug Deliv. Rev. 2008, 60, 1347–1370. [Google Scholar] [CrossRef] [PubMed]
- Grunberg, J.; Jeger, S.; Sarko, D.; Dennler, P.; Zimmermann, K.; Mier, W.; Schibli, R. DOTA-functionalized polylysine: A high number of DOTA chelates positively influences the biodistribution of enzymatic conjugated anti-tumor antibody chCE7agl. PLoS ONE 2013, 8, e60350. [Google Scholar] [CrossRef] [PubMed]
- Garnett, M.C. Targeted drug conjugates: Principles and progress. Adv. Drug Deliv. Rev. 2001, 53, 171–216. [Google Scholar] [CrossRef]
- Hermanson, G.T. Chapter 3—The Reactions of Bioconjugation. In Bioconjugate Techniques, 3rd ed.; Academic Press: Boston, MA, USA, 2013; pp. 229–258. [Google Scholar] [CrossRef]
- Rana, T.M.; Meares, C.F. N-terminal modification of immunoglobulin polypeptide chains tagged with isothiocyanato chelates. Bioconjug. Chem. 1990, 1, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Oosterwijk, E.; Ruiter, D.J.; Hoedemaeker, P.J.; Pauwels, E.K.; Jonas, U.; Zwartendijk, J.; Warnaar, S.O. Monoclonal antibody G 250 recognizes a determinant present in renal-cell carcinoma and absent from normal kidney. Int. J. Cancer 1986, 38, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Zatovicova, M.; Jelenska, L.; Hulikova, A.; Csaderova, L.; Ditte, Z.; Ditte, P.; Goliasova, T.; Pastorek, J.; Pastorekova, S. Carbonic anhydrase IX as an anticancer therapy target: Preclinical evaluation of internalizing monoclonal antibody directed to catalytic domain. Curr. Pharm. Des. 2010, 16, 3255–3263. [Google Scholar] [CrossRef] [PubMed]
- Oosterwijk-Wakka, J.C.; Boerman, O.C.; Mulders, P.F.; Oosterwijk, E. Application of monoclonal antibody G250 recognizing carbonic anhydrase IX in renal cell carcinoma. Int. J. Mol. Sci 2013, 14, 11402–11423. [Google Scholar] [CrossRef] [PubMed]
- Zatovicova, M.; Jelenska, L.; Hulikova, A.; Ditte, P.; Ditte, Z.; Csaderova, L.; Svastova, E.; Schmalix, W.; Boettger, V.; Bevan, P.; et al. Monoclonal antibody G250 targeting CA: Binding specificity, internalization and therapeutic effects in a non-renal cancer model. Int. J. Oncol. 2014, 45, 2455–2467. [Google Scholar] [CrossRef] [PubMed]
- Oosterwijk, E.; Ruiter, D.J.; Wakka, J.C.; Huiskens-van der Meij, J.W.; Jonas, U.; Fleuren, G.J.; Zwartendijk, J.; Hoedemaeker, P.; Warnaar, S.O. Immunohistochemical analysis of monoclonal antibodies to renal antigens. Application in the diagnosis of renal cell carcinoma. Am. J. Pathol. 1986, 123, 301–309. [Google Scholar] [PubMed]
- Ivanov, S.; Liao, S.-Y.; Ivanova, A.; Danilkovitch-Miagkova, A.; Tarasova, N.; Weirich, G.; Merrill, M.J.; Proescholdt, M.A.; Oldfield, E.H.; Lee, J.; et al. Expression of Hypoxia-Inducible Cell-Surface Transmembrane Carbonic Anhydrases in Human Cancer. Am. J. Pathol. 2001, 158, 905–919. [Google Scholar] [CrossRef]
- Muselaers, C.H.; Boerman, O.C.; Oosterwijk, E.; Langenhuijsen, J.F.; Oyen, W.J.; Mulders, P.F. Indium-111-labeled girentuximab immunoSPECT as a diagnostic tool in clear cell renal cell carcinoma. Eur. Urol. 2013, 63, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Kaluz, S.; Kaluzová, M.; Liao, S.-Y.; Lerman, M.; Stanbridge, E.J. Transcriptional control of the tumor- and hypoxia-marker carbonic anhydrase 9: A one transcription factor (HIF-1) show? Biochim. Biophys. Acta (BBA) Rev. Cancer 2009, 1795, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, S.V.; Kuzmin, I.; Wei, M.-H.; Pack, S.; Geil, L.; Johnson, B.E.; Stanbridge, E.J.; Lerman, M.I. Down-regulation of transmembrane carbonic anhydrases in renal cell carcinoma cell lines by wild-type von Hippel-Lindau transgenes. Proc. Natl. Acad. Sci. USA 1998, 95, 12596–12601. [Google Scholar] [CrossRef] [PubMed]
- Grabmaier, K.; de Weijert, M.C.A.; Verhaegh, G.W.; Schalken, J.A.; Oosterwijk, E. Strict regulation of CAIXG250/MN by HIF-1α in clear cell renal cell carcinoma. Oncogene 2004, 23, 5624. [Google Scholar] [CrossRef] [PubMed]
- Chamie, K.; Donin, N.M.; Klopfer, P.; Bevan, P.; Fall, B.; Wilhelm, O.; Storkel, S.; Said, J.; Gambla, M.; Hawkins, R.E.; et al. Adjuvant Weekly Girentuximab Following Nephrectomy for High-Risk Renal Cell Carcinoma: The ARISER Randomized Clinical Trial. JAMA Oncol. 2017, 3, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Cheal, S.M.; Punzalan, B.; Doran, M.G.; Evans, M.J.; Osborne, J.R.; Lewis, J.S.; Zanzonico, P.; Larson, S.M. Pairwise comparison of 89Zr- and 124I-labeled cG250 based on positron emission tomography imaging and nonlinear immunokinetic modeling: In vivo carbonic anhydrase IX receptor binding and internalization in mouse xenografts of clear-cell renal cell carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Huizing, F.J.; Hoeben, B.A.W.; Franssen, G.; Lok, J.; Heskamp, S.; Oosterwijk, E.; Boerman, O.C.; Bussink, J. Preclinical validation of (111)In-girentuximab-F(ab’)2 as a tracer to image hypoxia related marker CAIX expression in head and neck cancer xenografts. Radiother. Oncol. 2017, 124, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Stillebroer, A.B.; Boerman, O.C.; Desar, I.M.; Boers-Sonderen, M.J.; van Herpen, C.M.; Langenhuijsen, J.F.; Smith-Jones, P.M.; Oosterwijk, E.; Oyen, W.J.; Mulders, P.F. Phase 1 radioimmunotherapy study with lutetium 177-labeled anti-carbonic anhydrase IX monoclonal antibody girentuximab in patients with advanced renal cell carcinoma. Eur. Urol. 2013, 64, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Muselaers, C.H.; Boers-Sonderen, M.J.; van Oostenbrugge, T.J.; Boerman, O.C.; Desar, I.M.; Stillebroer, A.B.; Mulder, S.F.; van Herpen, C.M.; Langenhuijsen, J.F.; Oosterwijk, E.; et al. Phase 2 Study of Lutetium 177-Labeled Anti-Carbonic Anhydrase IX Monoclonal Antibody Girentuximab in Patients with Advanced Renal Cell Carcinoma. Eur. Urol. 2016, 69, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Stillebroer, A.B.; Zegers, C.M.; Boerman, O.C.; Oosterwijk, E.; Mulders, P.F.; O’Donoghue, J.A.; Visser, E.P.; Oyen, W.J. Dosimetric analysis of 177Lu-cG250 radioimmunotherapy in renal cell carcinoma patients: Correlation with myelotoxicity and pretherapeutic absorbed dose predictions based on 111In-cG250 imaging. J. Nucl. Med. 2012, 53, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.; King, A.G.; Moreno, B.J.M. Methods for Generating Radioimmunoconjugates. U.S. Patents 9,603,954, 3 March 2017. [Google Scholar]
- Nakamura, T.; Kawai, Y.; Kitamoto, N.; Osawa, T.; Kato, Y. Covalent modification of lysine residues by allyl isothiocyanate in physiological conditions: Plausible transformation of isothiocyanate from thiol to amine. Chem. Res. Toxicol. 2009, 22, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Spicer, C.D.; Davis, B.G. Selective chemical protein modification. Nat. Commun. 2014, 5, 4740. [Google Scholar] [CrossRef] [PubMed]
- Forrer, F.; Chen, J.; Fani, M.; Powell, P.; Lohri, A.; Müller-Brand, J.; Moldenhauer, G.; Maecke, H.R. In vitro characterization of 177Lu-radiolabelled chimeric anti-CD20 monoclonal antibody and a preliminary dosimetry study. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Price, E.W.; Edwards, K.J.; Carnazza, K.E.; Carlin, S.D.; Zeglis, B.M.; Adam, M.J.; Orvig, C.; Lewis, J.S. A comparative evaluation of the chelators H4octapa and CHX-A″-DTPA with the therapeutic radiometal 90Y. Nucl. Med. Biol. 2016, 43, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Steffens, M.G.; Kranenborg, M.H.; Boerman, O.C.; Zegwaart-Hagemeier, N.E.; Debruyne, F.M.; Corstens, F.H.; Oosterwijk, E. Tumor retention of 186Re-MAG3, 111In-DTPA and 125I labeled monoclonal antibody G250 in nude mice with renal cell carcinoma xenografts. Cancer Biother. Radiopharm. 1998, 13, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Oosterwijk, E.; Bander, N.H.; Divgi, C.R.; Welt, S.; Wakka, J.C.; Finn, R.D.; Carswell, E.A.; Larson, S.M.; Warnaar, S.O.; Fleuren, G.J.; et al. Antibody localization in human renal cell carcinoma: A phase I study of monoclonal antibody G250. J. Clin. Oncol. 1993, 11, 738–750. [Google Scholar] [CrossRef] [PubMed]
- Muselaers, C.H.; Oosterwijk, E.; Bos, D.L.; Oyen, W.J.; Mulders, P.F.; Boerman, O.C. Optimizing lutetium 177-anti-carbonic anhydrase IX radioimmunotherapy in an intraperitoneal clear cell renal cell carcinoma xenograft model. Mol. Imaging 2014, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Oosterwijk-Wakka, J.C.; de Weijert, M.C.; Franssen, G.M.; Leenders, W.P.; van der Laak, J.A.; Boerman, O.C.; Mulders, P.F.; Oosterwijk, E. Successful combination of sunitinib and girentuximab in two renal cell carcinoma animal models: A rationale for combination treatment of patients with advanced RCC. Neoplasia 2015, 17, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2010, 29, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Price, L.; Schorzman, A.; Storrie, M.; Piscitelli, J.; Razo, J.; Zamboni, W. Factors Affecting the Pharmacology of Antibody–Drug Conjugates. Antibodies 2018, 7, 10. [Google Scholar] [CrossRef]
- Durrbach, A.; Angevin, E.; Poncet, P.; Rouleau, M.; Chavanel, G.; Chapel, A.; Thierry, D.; Gorter, A.; Hirsch, R.; Charpentier, B.; et al. Antibody-mediated endocytosis of G250 tumor-associated antigen allows targeted gene transfer to human renal cell carcinoma in vitro. Cancer Gene Ther. 1999, 6, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Honarvar, H.; Garousi, J.; Gunneriusson, E.; Hoiden-Guthenberg, I.; Altai, M.; Widstrom, C.; Tolmachev, V.; Frejd, F.Y. Imaging of CAIX-expressing xenografts in vivo using 99mTc-HEHEHE-ZCAIX:1 affibody molecule. Int. J. Oncol. 2015, 46, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Roscher, M.; Hormann, I.; Leib, O.; Marx, S.; Moreno, J.; Miltner, E.; Friesen, C. Targeted alpha-therapy using [Bi-213]anti-CD20 as novel treatment option for radio- and chemoresistant non-Hodgkin lymphoma cells. Oncotarget 2013, 4, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Graf, F.; Fahrer, J.; Maus, S.; Morgenstern, A.; Bruchertseifer, F.; Venkatachalam, S.; Fottner, C.; Weber, M.M.; Huelsenbeck, J.; Schreckenberger, M.; et al. DNA Double Strand Breaks as Predictor of Efficacy of the Alpha-Particle Emitter Ac-225 and the Electron Emitter Lu-177 for Somatostatin Receptor Targeted Radiotherapy. PLoS ONE 2014, 9, e88239. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Candiano, G.; Bruschi, M.; Musante, L.; Santucci, L.; Ghiggeri, G.M.; Carnemolla, B.; Orecchia, P.; Zardi, L.; Righetti, P.G. Blue silver: A very sensitive colloidal Coomassie G-250 staining for proteome analysis. Electrophoresis 2004, 25, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- Gunasekera, K.; Wuthrich, D.; Braga-Lagache, S.; Heller, M.; Ochsenreiter, T. Proteome remodelling during development from blood to insect-form Trypanosoma brucei quantified by SILAC and mass spectrometry. BMC Genom. 2012, 13, 556. [Google Scholar] [CrossRef] [PubMed]
- Ozerdem, U.; Wojcik, E.M.; Barkan, G.A.; Duan, X.; Ersahin, C. A practical application of quantitative vascular image analysis in breast pathology. Pathol. Res. Pract. 2013, 209, 455–458. [Google Scholar] [CrossRef] [PubMed]
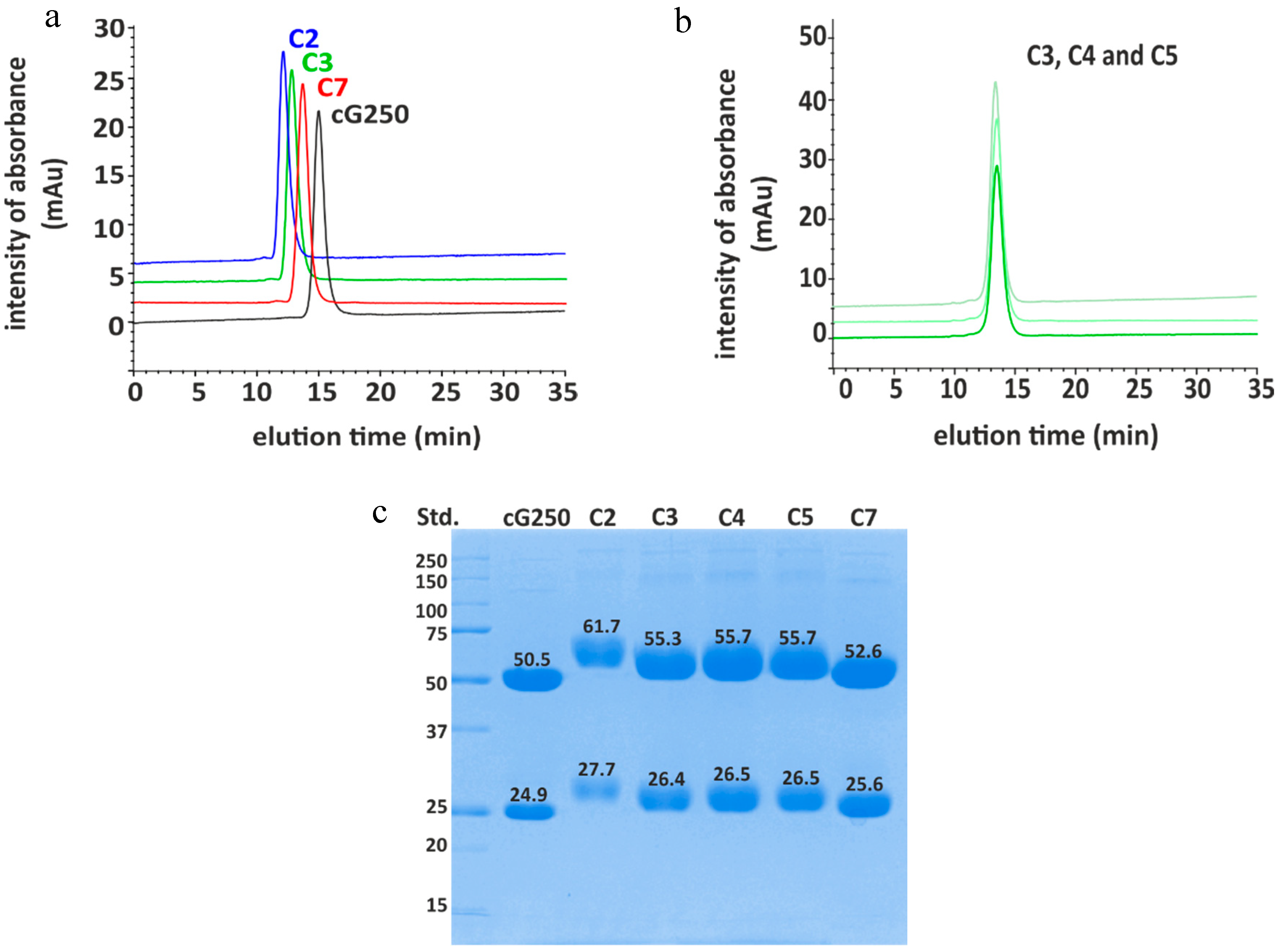

| Conjugate | MW (Da) | Ratio Non Reduced | Ratio Reduced | |
|---|---|---|---|---|
| HC | LC | |||
| C2 (90 min) | - | - | 12–23 1 | 6–7 1 |
| C3 (60 min) | 154,187.07 | 8–10 | 3–4 | 1–2 |
| C7 (30 min) | 151,543.63 | 5–6 | 1–2 | 1–2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basaco, T.; Pektor, S.; Bermudez, J.M.; Meneses, N.; Heller, M.; Galván, J.A.; Boligán, K.F.; Schürch, S.; Von Gunten, S.; Türler, A.; et al. Evaluation of Radiolabeled Girentuximab In Vitro and In Vivo. Pharmaceuticals 2018, 11, 132. https://doi.org/10.3390/ph11040132
Basaco T, Pektor S, Bermudez JM, Meneses N, Heller M, Galván JA, Boligán KF, Schürch S, Von Gunten S, Türler A, et al. Evaluation of Radiolabeled Girentuximab In Vitro and In Vivo. Pharmaceuticals. 2018; 11(4):132. https://doi.org/10.3390/ph11040132
Chicago/Turabian StyleBasaco, Tais, Stefanie Pektor, Josue M. Bermudez, Niurka Meneses, Manfred Heller, José A. Galván, Kayluz F. Boligán, Stefan Schürch, Stephan Von Gunten, Andreas Türler, and et al. 2018. "Evaluation of Radiolabeled Girentuximab In Vitro and In Vivo" Pharmaceuticals 11, no. 4: 132. https://doi.org/10.3390/ph11040132
APA StyleBasaco, T., Pektor, S., Bermudez, J. M., Meneses, N., Heller, M., Galván, J. A., Boligán, K. F., Schürch, S., Von Gunten, S., Türler, A., & Miederer, M. (2018). Evaluation of Radiolabeled Girentuximab In Vitro and In Vivo. Pharmaceuticals, 11(4), 132. https://doi.org/10.3390/ph11040132





