Ketamine and Ceftriaxone-Induced Alterations in Glutamate Levels Do Not Impact the Specific Binding of Metabotropic Glutamate Receptor Subtype 5 Radioligand [18F]PSS232 in the Rat Brain
Abstract
1. Introduction
2. Results
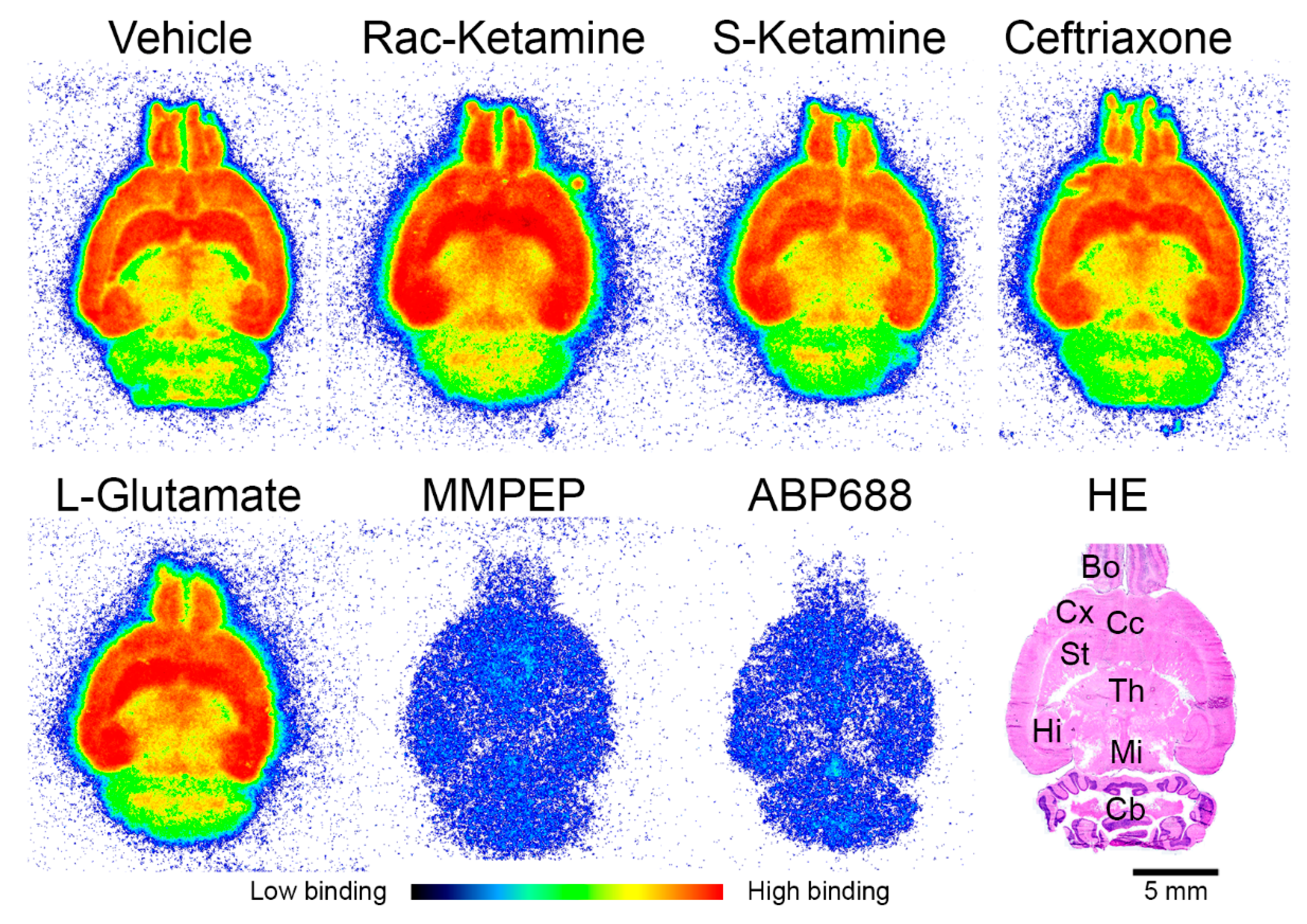
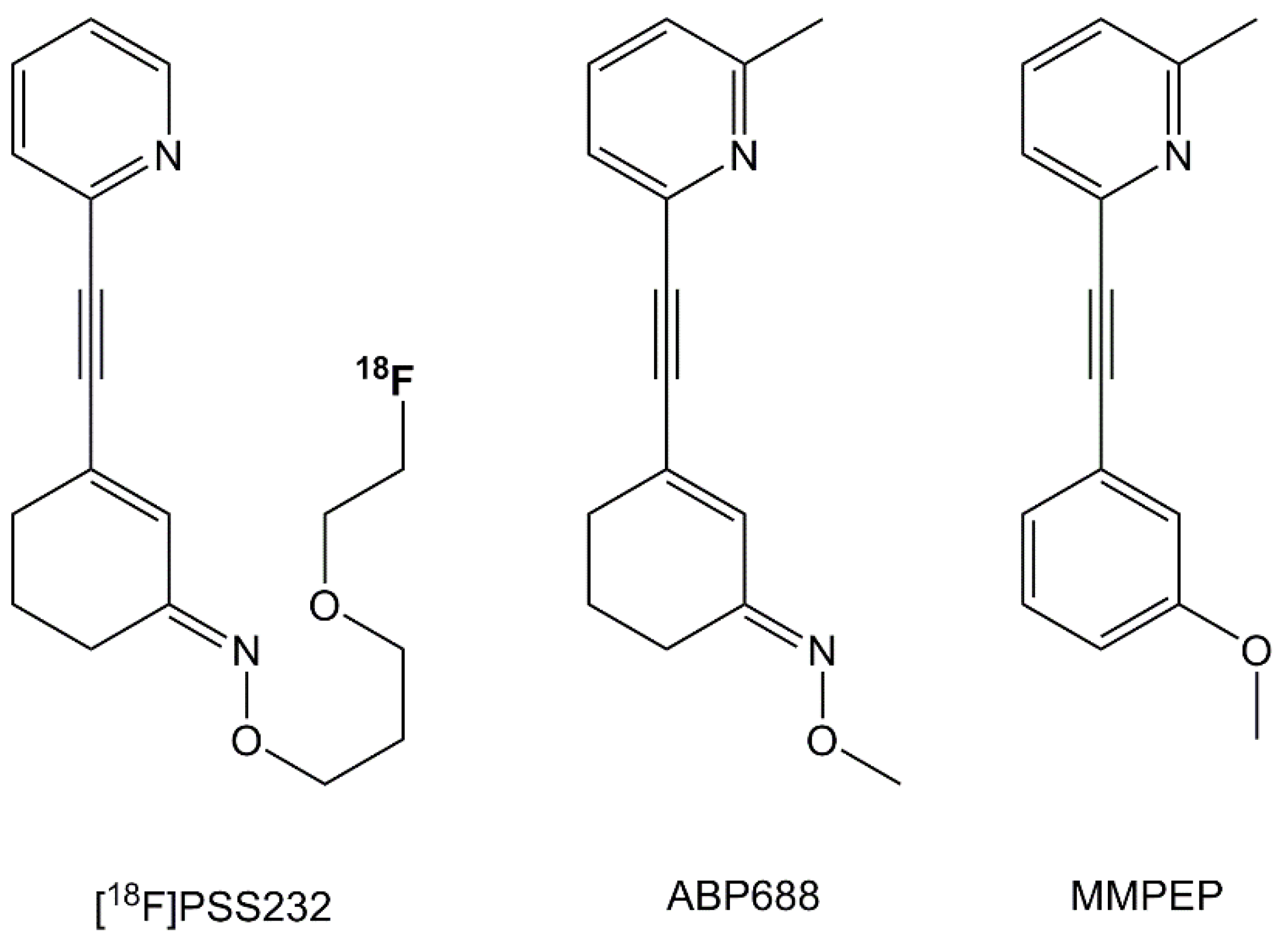
2.1. In Vitro Effects of Racemic Ketamine, S-Ketamine or Ceftriaxone on [18F]PSS232 Binding
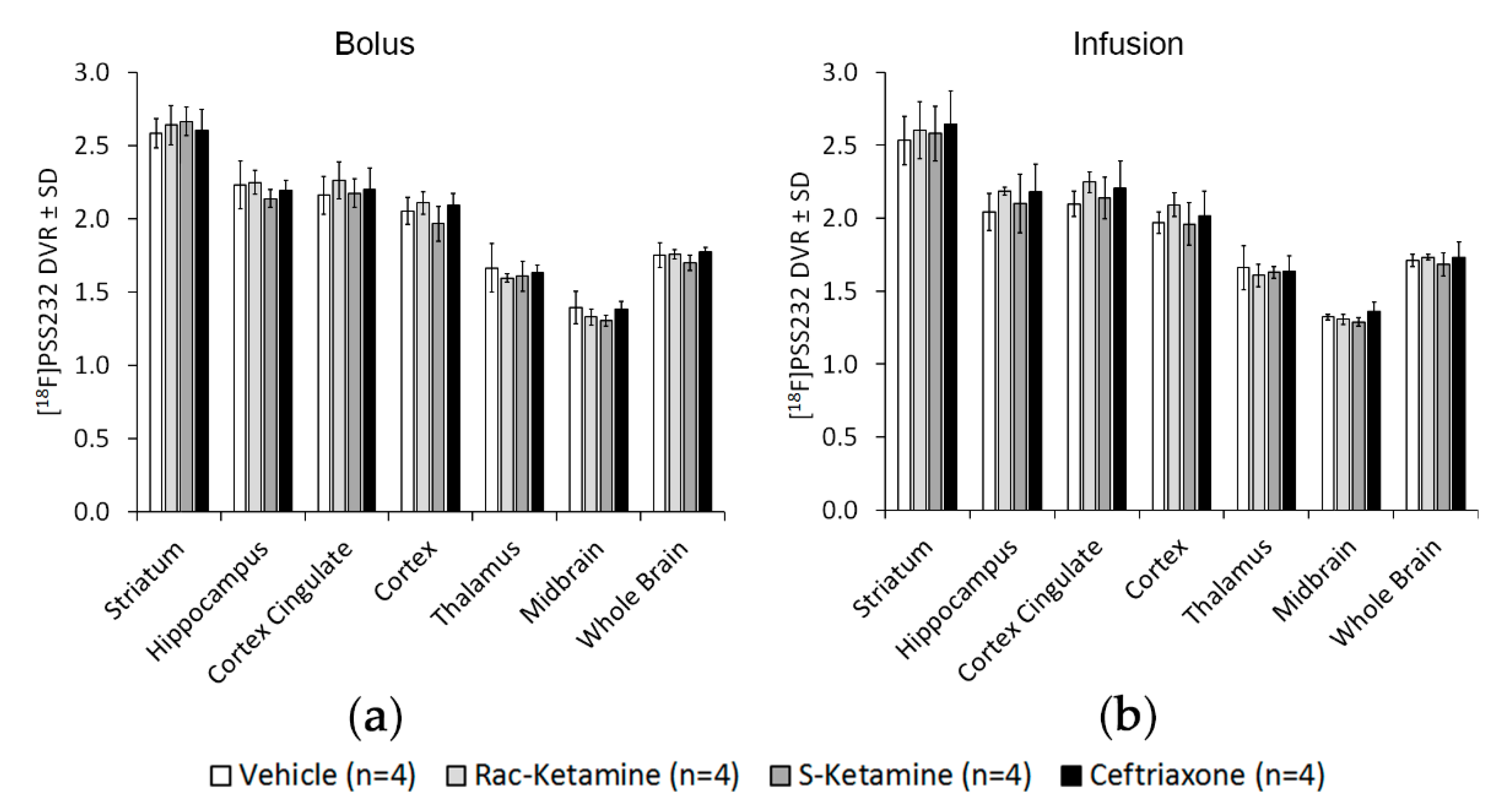
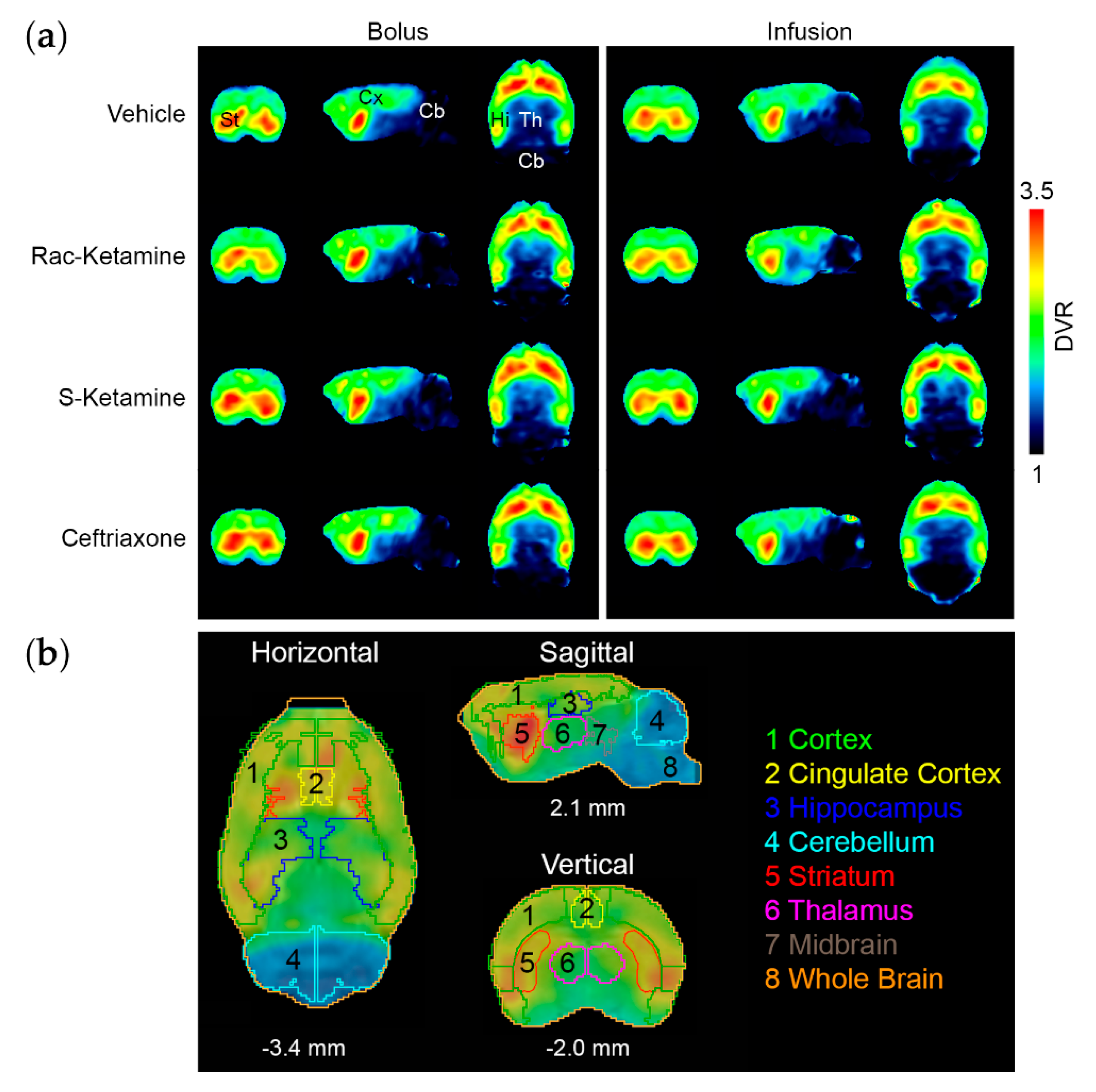
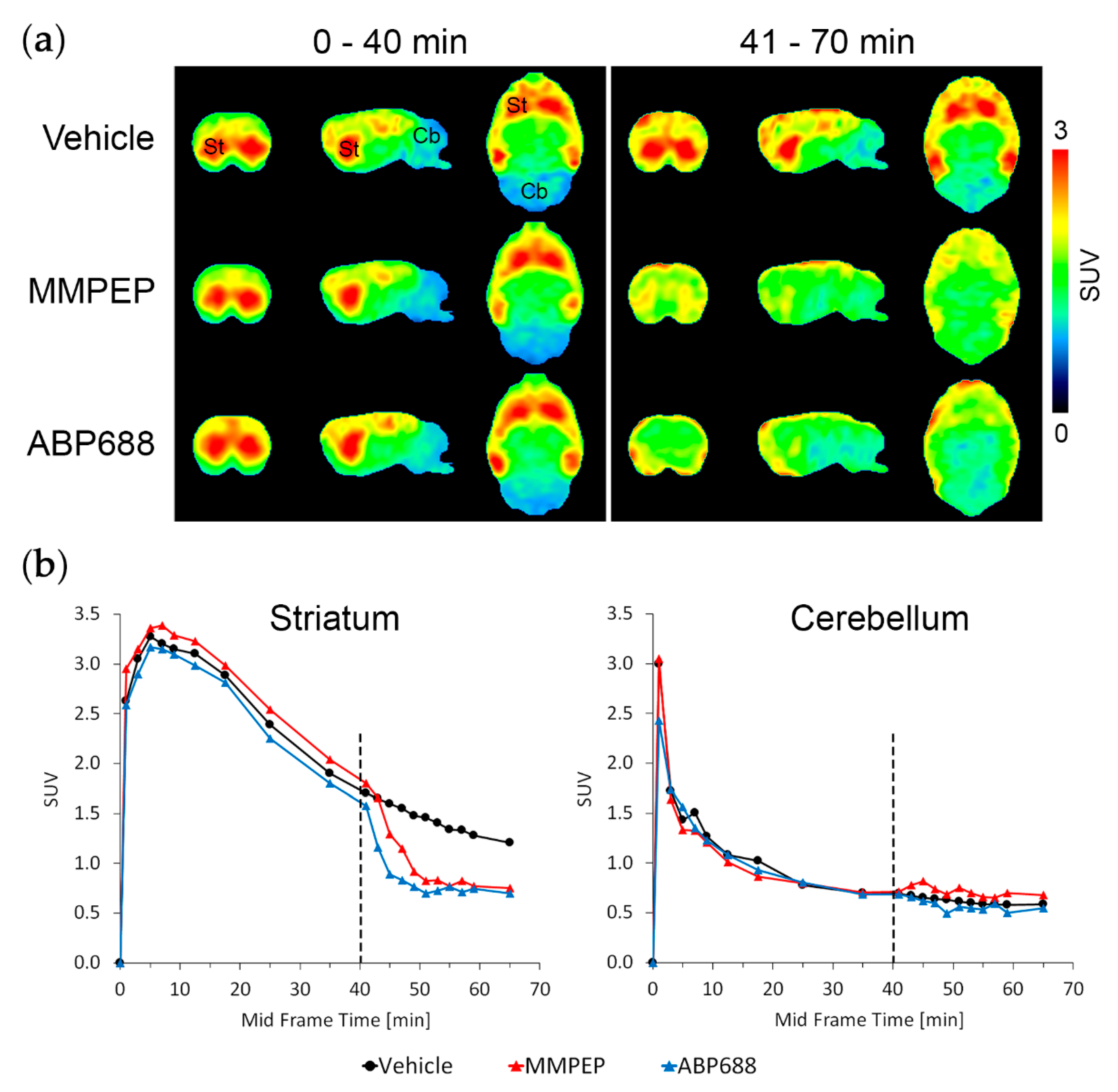
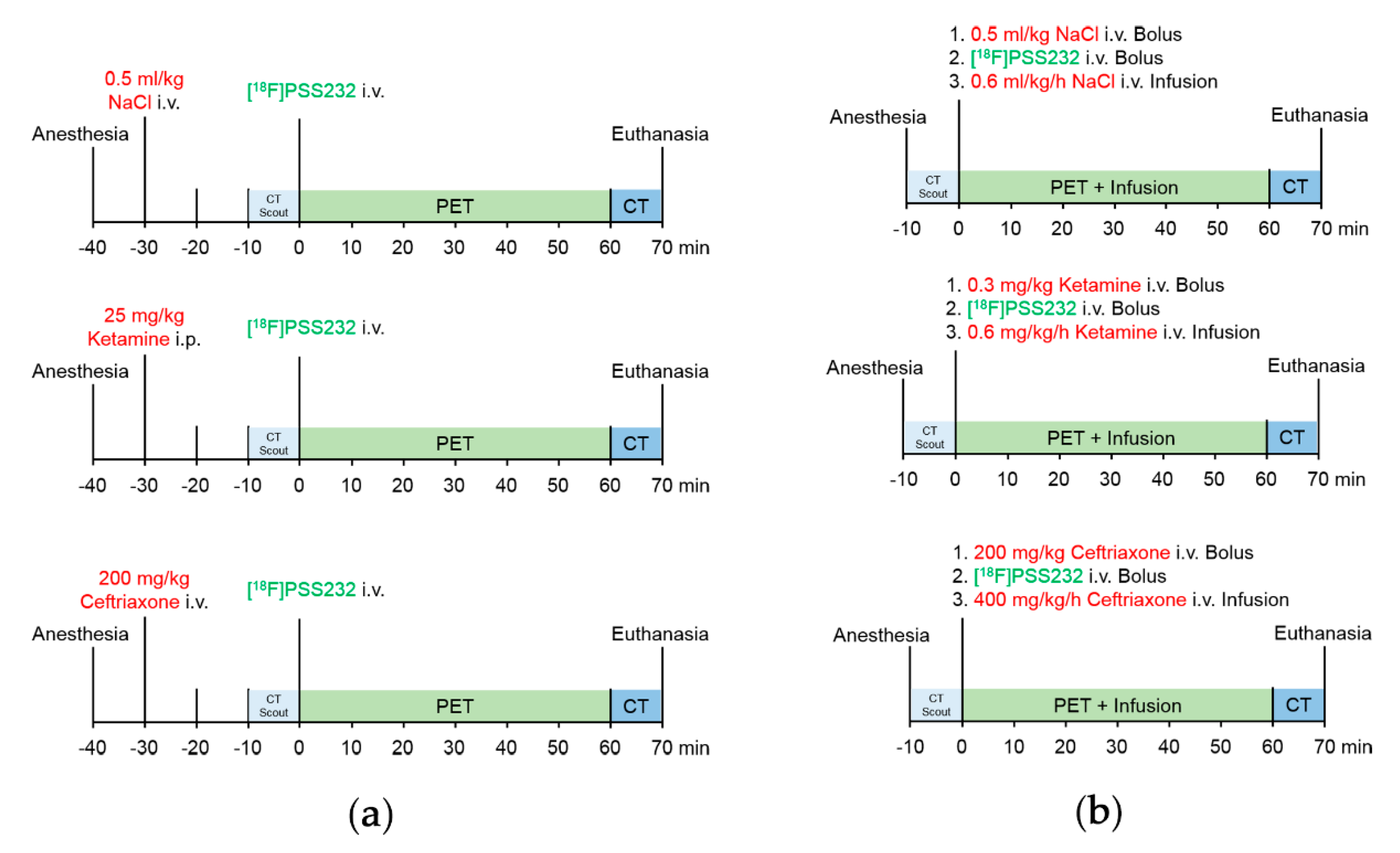
2.2. In Vivo Effects of Racemic Ketamine, S-Ketamine or Ceftriaxone on [18F]PSS232 Binding
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Radiosynthesis and Pharmaceuticals
4.3. In Vitro Autoradiography
4.4. In Vivo PET Imaging
4.5. Bolus and Infusion Protocol
4.6. In Vivo Displacement Study
4.7. Image Data Reconstruction, Analysis and Calculation of Distribution Volume Ratios (DVRs) as Well as Standardized Uptake Values (SUVs)
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pin, J.P.; Duvoisin, R. The metabotropic glutamate receptors: Structure and functions. Neuropharmacology 1995, 34, 1–26. [Google Scholar] [CrossRef]
- Danbolt, N.C. Glutamate uptake. Prog. Neurobiol. 2001, 65, 1–105. [Google Scholar] [CrossRef]
- Okubo, Y.; Sekiya, H.; Namiki, S.; Sakamoto, H.; Iinuma, S.; Yamasaki, M.; Watanabe, M.; Hirose, K.; Iino, M. Imaging extrasynaptic glutamate dynamics in the brain. Proc. Natl. Acad. Sci. USA 2010, 107, 6526–6531. [Google Scholar] [CrossRef] [PubMed]
- Sanacora, G.; Zarate, C.A.; Krystal, J.H.; Manji, H.K. Targeting the glutamatergic system to develop novel, improved therapeutics for mood disorders. Nat. Rev. Drug Discov. 2008, 7, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Olney, J.W.; Farber, N.B. Glutamate receptor dysfunction and schizophrenia. Arch. Gen. Psychiatry 1995, 52, 998–1007. [Google Scholar] [CrossRef] [PubMed]
- Chase, T.N.; Oh, J.D. Striatal dopamine- and glutamate-mediated dysregulation in experimental parkinsonism. Trends Neurosci. 2000, 23, S86–S91. [Google Scholar] [CrossRef]
- Bruno, V.; Ksiazek, I.; Battaglia, G.; Lukic, S.; Leonhardt, T.; Sauer, D.; Gasparini, F.; Kuhn, R.; Nicoletti, F.; Flor, P.J. Selective blockade of metabotropic glutamate receptor subtype 5 is neuroprotective. Neuropharmacology 2000, 39, 2223–2230. [Google Scholar] [CrossRef]
- Chiamulera, C.; Epping-Jordan, M.P.; Zocchi, A.; Marcon, C.; Cottiny, C.; Tacconi, S.; Corsi, M.; Orzi, F.; Conquet, F. Reinforcing and locomotor stimulant effects of cocaine are absent in mGluR5 null mutant mice. Nat. Neurosci. 2001, 4, 873–874. [Google Scholar] [CrossRef] [PubMed]
- Ametamey, S.M.; Kessler, L.J.; Honer, M.; Wyss, M.T.; Buck, A.; Hintermann, S.; Auberson, Y.P.; Gasparini, F.; Schubiger, P.A. Radiosynthesis and preclinical evaluation of 11C-ABP688 as a probe for imaging the metabotropic glutamate receptor subtype 5. J. Nucl. Med. 2006, 47, 698–705. [Google Scholar] [PubMed]
- DeLorenzo, C.; DellaGioia, N.; Bloch, M.; Sanacora, G.; Nabulsi, N.; Abdallah, C.; Yang, J.; Wen, R.; Mann, J.J.; Krystal, J.H.; et al. In vivo ketamine-induced changes in [11c]ABP688 binding to metabotropic glutamate receptor subtype 5. Biol. Psychiatry 2015, 77, 266–275. [Google Scholar] [CrossRef] [PubMed]
- DeLorenzo, C.; Sovago, J.; Gardus, J.; Xu, J.; Yang, J.; Behrje, R.; Kumar, J.S.; Devanand, D.P.; Pelton, G.H.; Mathis, C.A.; et al. Characterization of brain mGluR5 binding in a pilot study of late-life major depressive disorder using positron emission tomography and [11c]ABP688. Transl. Psychiatry 2015, 5, e693. [Google Scholar] [CrossRef] [PubMed]
- Esterlis, I.; DellaGioia, N.; Pietrzak, R.H.; Matuskey, D.; Nabulsi, N.; Abdallah, C.G.; Yang, J.; Pittenger, C.; Sanacora, G.; Krystal, J.H.; et al. Ketamine-induced reduction in mGluR5 availability is associated with an antidepressant response: An [11c]ABP688 and pet imaging study in depression. Mol. Psychiatry 2018, 23, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Miyake, N.; Skinbjerg, M.; Easwaramoorthy, B.; Kumar, D.; Girgis, R.R.; Xu, X.; Slifstein, M.; Abi-Dargham, A. Imaging changes in glutamate transmission in vivo with the metabotropic glutamate receptor 5 tracer [11c]ABP688 and n-acetylcysteine challenge. Biol. Psychiatry 2011, 69, 822–824. [Google Scholar] [CrossRef] [PubMed]
- Sandiego, C.M.; Nabulsi, N.; Lin, S.F.; Labaree, D.; Najafzadeh, S.; Huang, Y.; Cosgrove, K.; Carson, R.E. Studies of the metabotropic glutamate receptor 5 radioligand [11c]ABP688 with n-acetylcysteine challenge in rhesus monkeys. Synapse 2013, 67, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, E.R.; Parent, M.J.; Leuzy, A.; Aliaga, A.; Aliaga, A.; Moquin, L.; Schirrmacher, E.S.; Soucy, J.P.; Skelin, I.; Gratton, A.; et al. Imaging in vivo glutamate fluctuations with [11c]ABP688: a GLT-1 challenge with ceftriaxone. J. Cereb. Blood Flow Metab. 2015, 35, 1169–1174. [Google Scholar] [CrossRef] [PubMed]
- Wyckhuys, T.; Verhaeghe, J.; Wyffels, L.; Langlois, X.; Schmidt, M.; Stroobants, S.; Staelens, S. N-acetylcysteine- and mk-801-induced changes in glutamate levels do not affect in vivo binding of metabotropic glutamate 5 receptor radioligand [11c]ABP688 in rat brain. J. Nucl. Med. 2013, 54, 1954–1961. [Google Scholar] [CrossRef] [PubMed]
- Rathinam, M.L.; Watts, L.T.; Stark, A.A.; Mahimainathan, L.; Stewart, J.; Schenker, S.; Henderson, G.I. Astrocyte control of fetal cortical neuron glutathione homeostasis: Up-regulation by ethanol. J. Neurochem. 2006, 96, 1289–1300. [Google Scholar] [CrossRef] [PubMed]
- Rothstein, J.D.; Patel, S.; Regan, M.R.; Haenggeli, C.; Huang, Y.H.; Bergles, D.E.; Jin, L.; Dykes Hoberg, M.; Vidensky, S.; Chung, D.S.; et al. Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 2005, 433, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.Y.; Xu, J.; Zhang, M.; Wang, D.; Li, L.; Li, W.B. Ceftriaxone modulates uptake activity of glial glutamate transporter-1 against global brain ischemia in rats. J. Neurochem. 2015, 132, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Milicevic Sephton, S.; Müller Herde, A.; Mu, L.; Keller, C.; Rudisuhli, S.; Auberson, Y.; Schibli, R.; Krämer, S.D.; Ametamey, S.M. Preclinical evaluation and test-retest studies of [18F]PSS232, a novel radioligand for targeting metabotropic glutamate receptor 5 (mGlu5). Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Müller Herde, A.; Keller, C.; Milicevic Sephton, S.; Mu, L.; Schibli, R.; Ametamey, S.M.; Krämer, S.D. Quantitative positron emission tomography of mGluR5 in rat brain with [18F]PSS232 at minimal invasiveness and reduced model complexity. J. Neurochem. 2015, 133, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Lorrain, D.S.; Baccei, C.S.; Bristow, L.J.; Anderson, J.J.; Varney, M.A. Effects of ketamine and N-methyl-d-aspartate on glutamate and dopamine release in the rat prefrontal cortex: Modulation by a group II selective metabotropic glutamate receptor agonist LY379268. Neuroscience 2003, 117, 697–706. [Google Scholar] [CrossRef]
- Moghaddam, B.; Adams, B.; Verma, A.; Daly, D. Activation of glutamatergic neurotransmission by ketamine: A novel step in the pathway from NMDA receptor blockade to dopaminergic and cognitive disruptions associated with the prefrontal cortex. J. Neurosci. 1997, 17, 2921–2927. [Google Scholar] [CrossRef] [PubMed]
- Doenicke, A.; Kugler, J.; Mayer, M.; Angster, R.; Hoffmann, P. Ketamine racemate or S-(+)-ketamine and midazolam. The effect on vigilance, efficacy and subjective findings. Anaesthesist 1992, 41, 610–618. [Google Scholar] [PubMed]
- Domino, E.F. Taming the ketamine tiger. Anesthesiology 2010, 113, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Himmelseher, S.; Pfenninger, E. The clinical use of S-(+)-ketamine—A determination of its place. Anasthesiol. Intensivmed. Notfallmed. Schmerzther. 1998, 33, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Alagarsamy, S.; Marino, M.J.; Rouse, S.T.; Gereau, R.W.t.; Heinemann, S.F.; Conn, P.J. Activation of NMDA receptors reverses desensitization of mGluR5 in native and recombinant systems. Nat. Neurosci. 1999, 2, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Homayoun, H.; Moghaddam, B. Bursting of prefrontal cortex neurons in awake rats is regulated by metabotropic glutamate 5 (mGlu5) receptors: Rate-dependent influence and interaction with NMDA receptors. Cereb. Cortex 2006, 16, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.P.; Edelstein, S.J. Allosteric mechanisms of signal transduction. Science 2005, 308, 1424–1428. [Google Scholar] [CrossRef] [PubMed]
- Cabello, N.; Gandia, J.; Bertarelli, D.C.; Watanabe, M.; Lluis, C.; Franco, R.; Ferre, S.; Lujan, R.; Ciruela, F. Metabotropic glutamate type 5, dopamine D2 and adenosine A2a receptors form higher-order oligomers in living cells. J. Neurochem. 2009, 109, 1497–1507. [Google Scholar] [CrossRef] [PubMed]
- Canela, L.; Fernandez-Duenas, V.; Albergaria, C.; Watanabe, M.; Lluis, C.; Mallol, J.; Canela, E.I.; Franco, R.; Lujan, R.; Ciruela, F. The association of metabotropic glutamate receptor type 5 with the neuronal Ca2+-binding protein 2 modulates receptor function. J. Neurochem. 2009, 111, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Seneca, N.; Finnema, S.J.; Farde, L.; Gulyas, B.; Wikstrom, H.V.; Halldin, C.; Innis, R.B. Effect of amphetamine on dopamine D2 receptor binding in nonhuman primate brain: A comparison of the agonist radioligand [11c]MNPA and antagonist [11c]raclopride. Synapse 2006, 59, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.A.; McCormick, P.; Kapur, S.; Willeit, M.; Garcia, A.; Hussey, D.; Houle, S.; Seeman, P.; Ginovart, N. Radiosynthesis and evaluation of [11c]-(+)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-naphtho[1,2-b][1,4]oxazin-9-ol as a potential radiotracer for in vivo imaging of the dopamine D2 high-affinity state with positron emission tomography. J. Med. Chem. 2005, 48, 4153–4160. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Müller Herde, A.; Boss, S.D.; He, Y.; Schibli, R.; Mu, L.; Ametamey, S.M. Ketamine and Ceftriaxone-Induced Alterations in Glutamate Levels Do Not Impact the Specific Binding of Metabotropic Glutamate Receptor Subtype 5 Radioligand [18F]PSS232 in the Rat Brain. Pharmaceuticals 2018, 11, 83. https://doi.org/10.3390/ph11030083
Müller Herde A, Boss SD, He Y, Schibli R, Mu L, Ametamey SM. Ketamine and Ceftriaxone-Induced Alterations in Glutamate Levels Do Not Impact the Specific Binding of Metabotropic Glutamate Receptor Subtype 5 Radioligand [18F]PSS232 in the Rat Brain. Pharmaceuticals. 2018; 11(3):83. https://doi.org/10.3390/ph11030083
Chicago/Turabian StyleMüller Herde, Adrienne, Silvan D. Boss, Yingfang He, Roger Schibli, Linjing Mu, and Simon M. Ametamey. 2018. "Ketamine and Ceftriaxone-Induced Alterations in Glutamate Levels Do Not Impact the Specific Binding of Metabotropic Glutamate Receptor Subtype 5 Radioligand [18F]PSS232 in the Rat Brain" Pharmaceuticals 11, no. 3: 83. https://doi.org/10.3390/ph11030083
APA StyleMüller Herde, A., Boss, S. D., He, Y., Schibli, R., Mu, L., & Ametamey, S. M. (2018). Ketamine and Ceftriaxone-Induced Alterations in Glutamate Levels Do Not Impact the Specific Binding of Metabotropic Glutamate Receptor Subtype 5 Radioligand [18F]PSS232 in the Rat Brain. Pharmaceuticals, 11(3), 83. https://doi.org/10.3390/ph11030083





