Clopidogrel Pharmacokinetics in Malaysian Population Groups: The Impact of Inter-Ethnic Variability
Abstract
1. Introduction
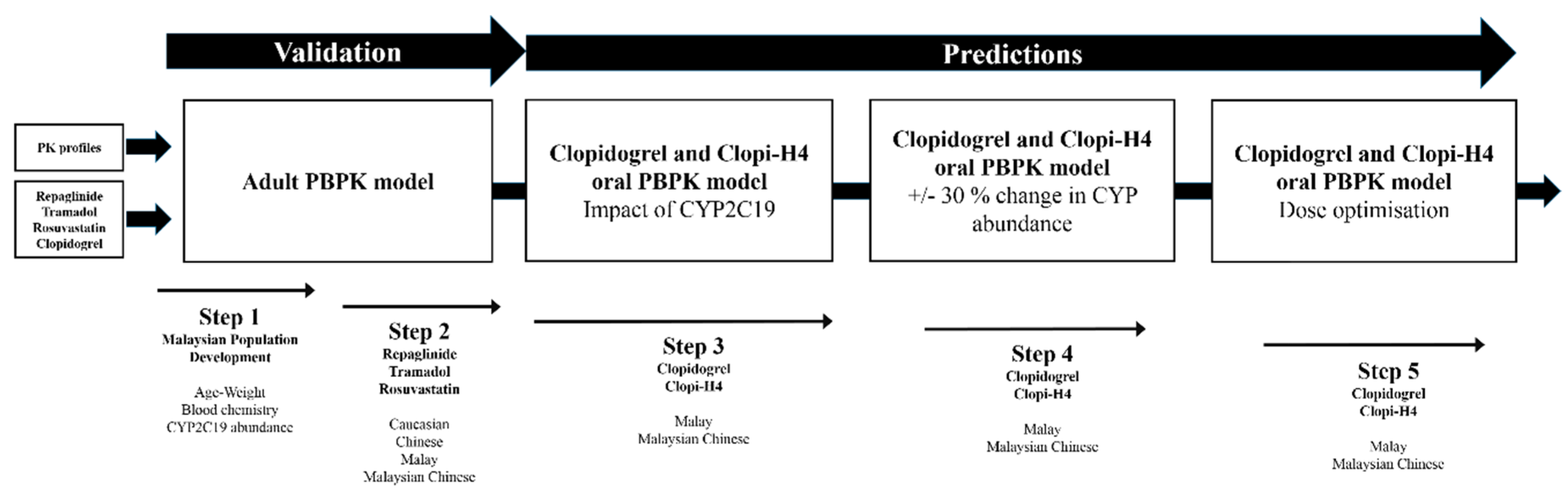
2. Results
2.1. Step 1: Malaysian Population Group Development
2.1.1. The National Cardiovascular Disease Database
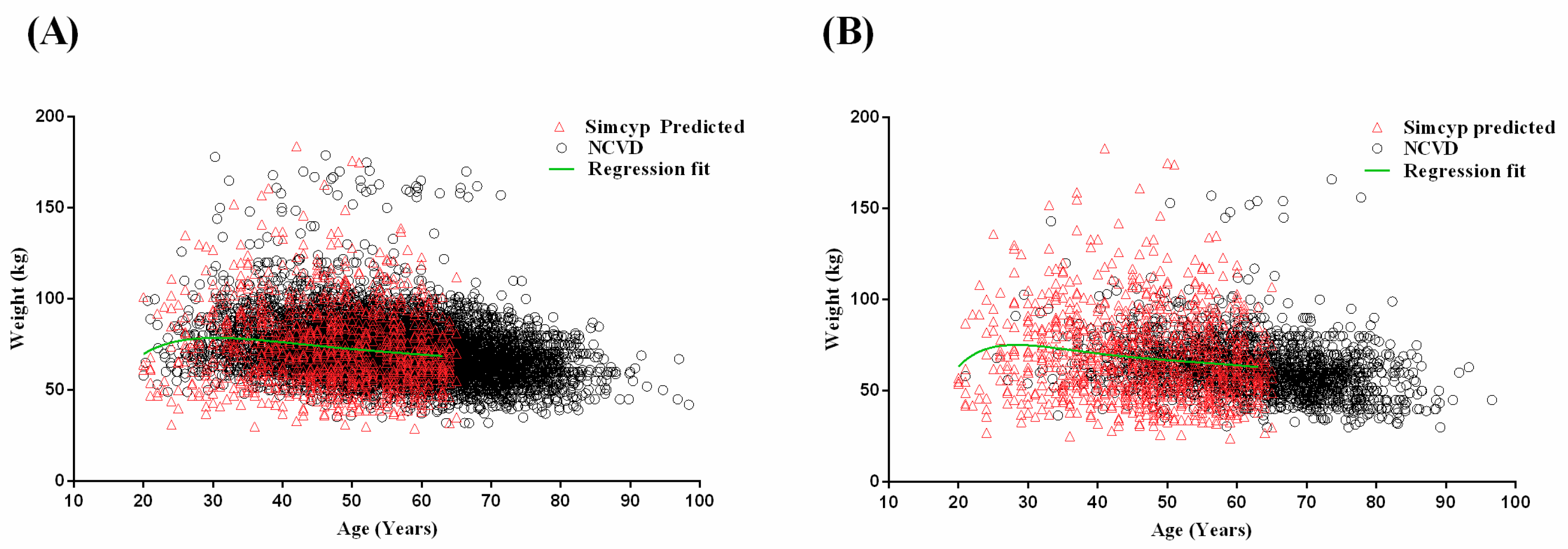
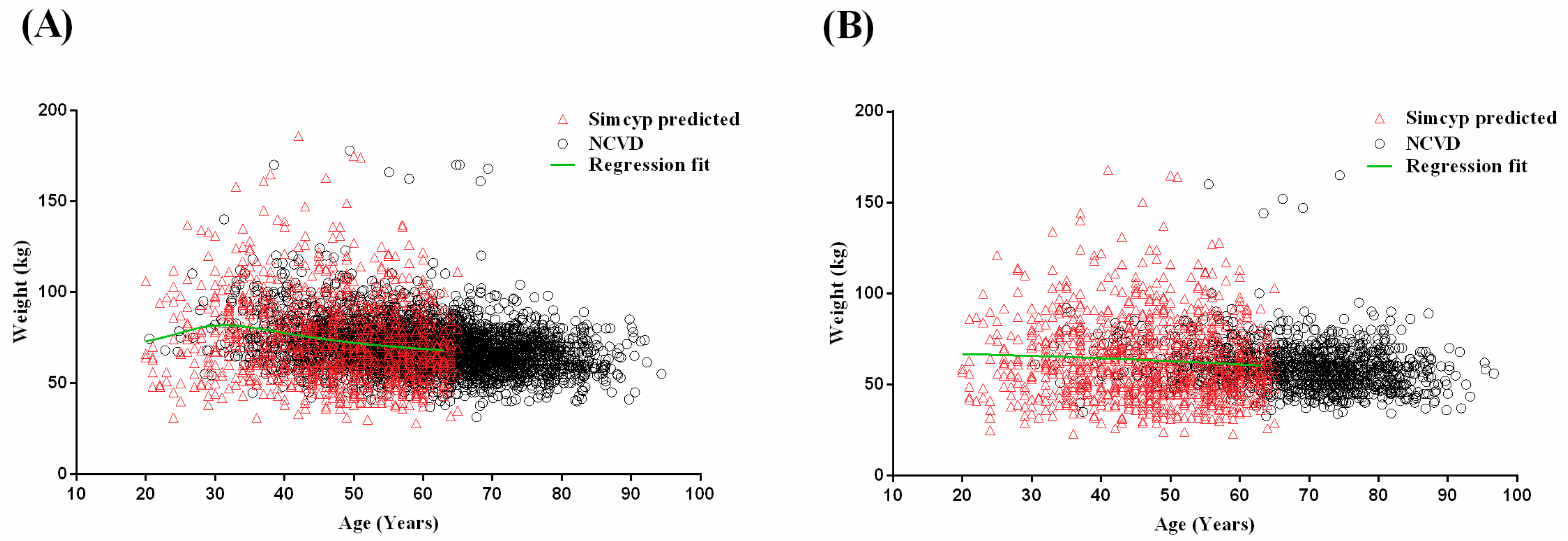
2.1.2. Development of Age–Weight Relationships for Malaysian Populations
2.2. Step 2: Adult Simulations: Validation with Repaglinide, Tramadol and Rosuvastatin
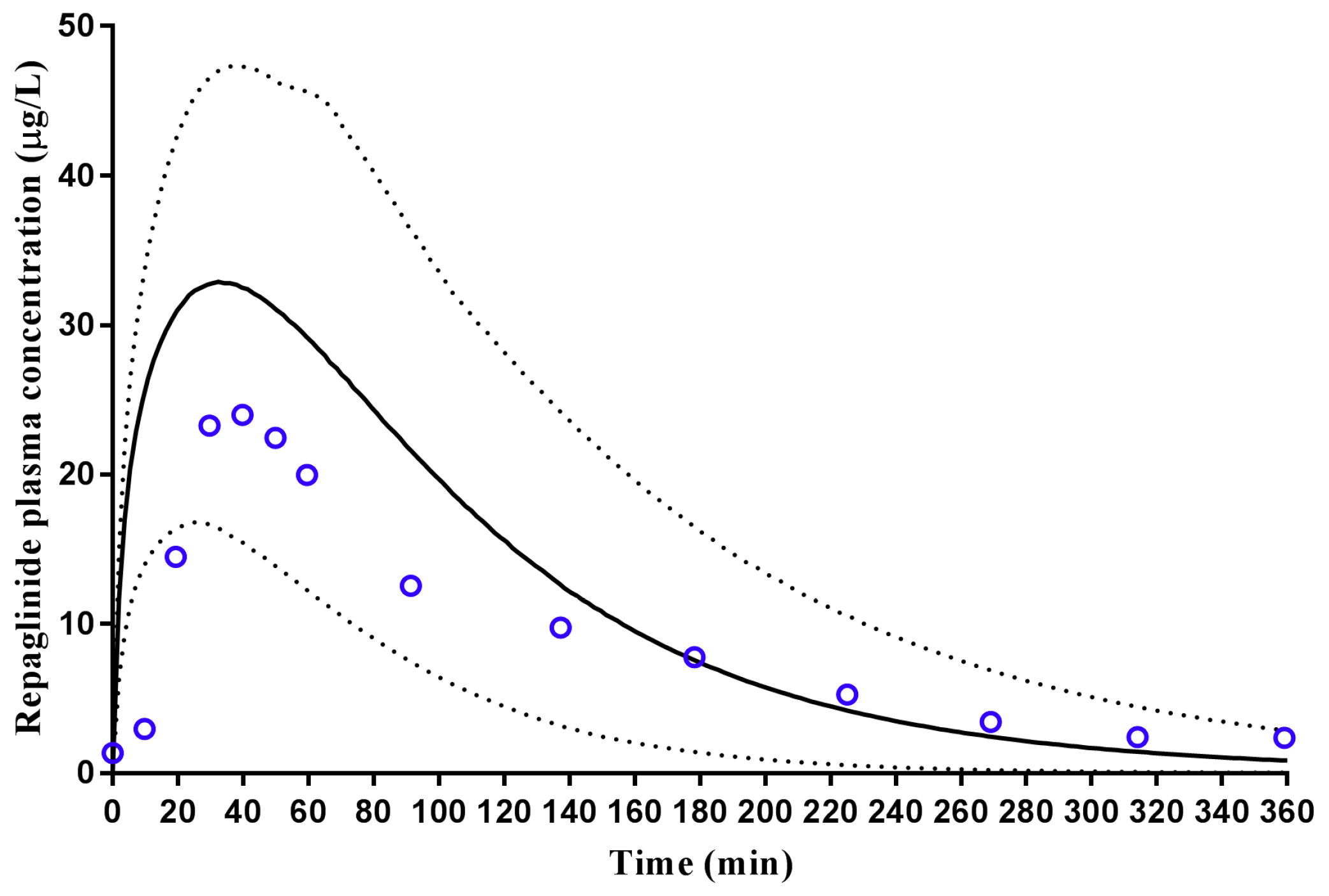
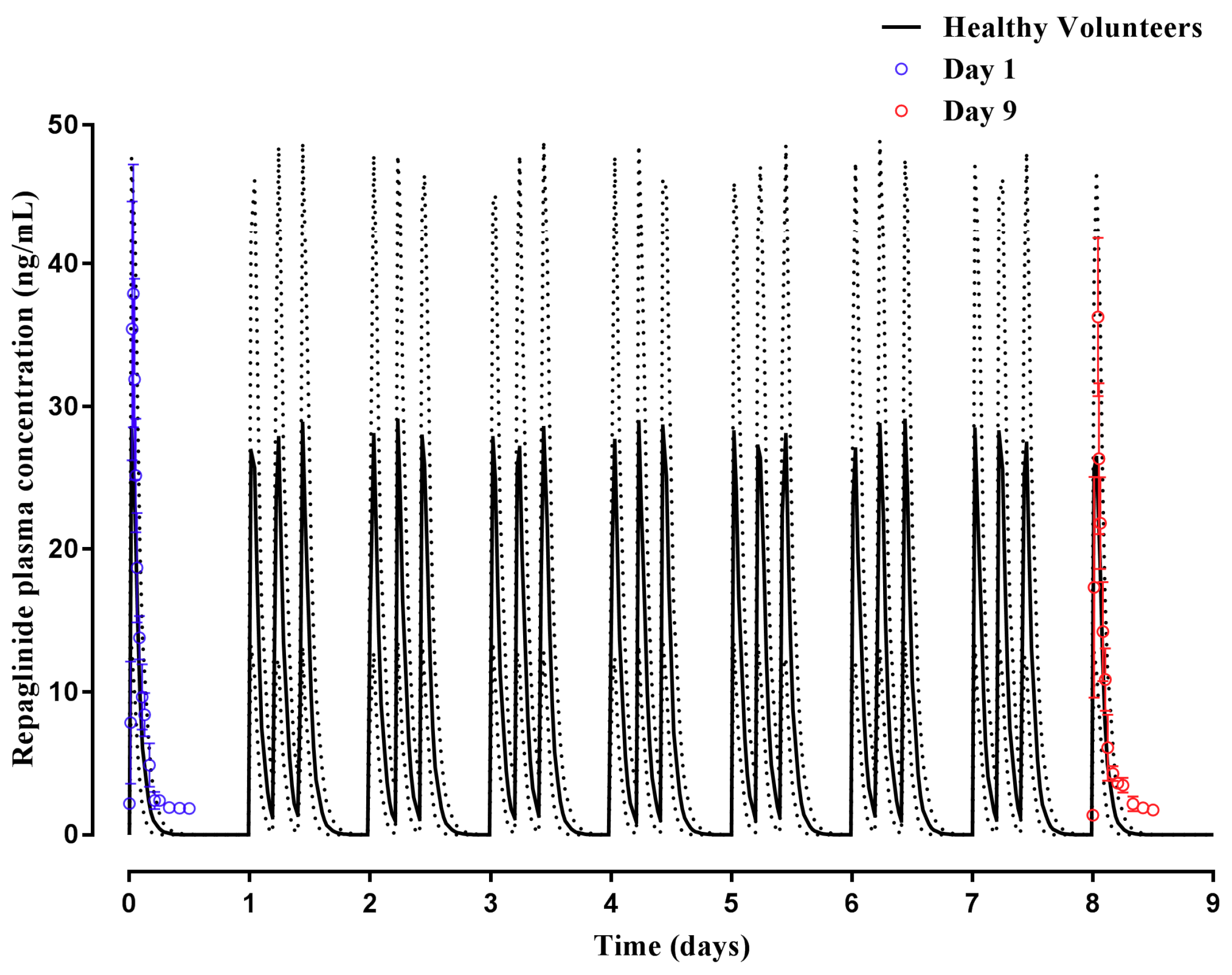
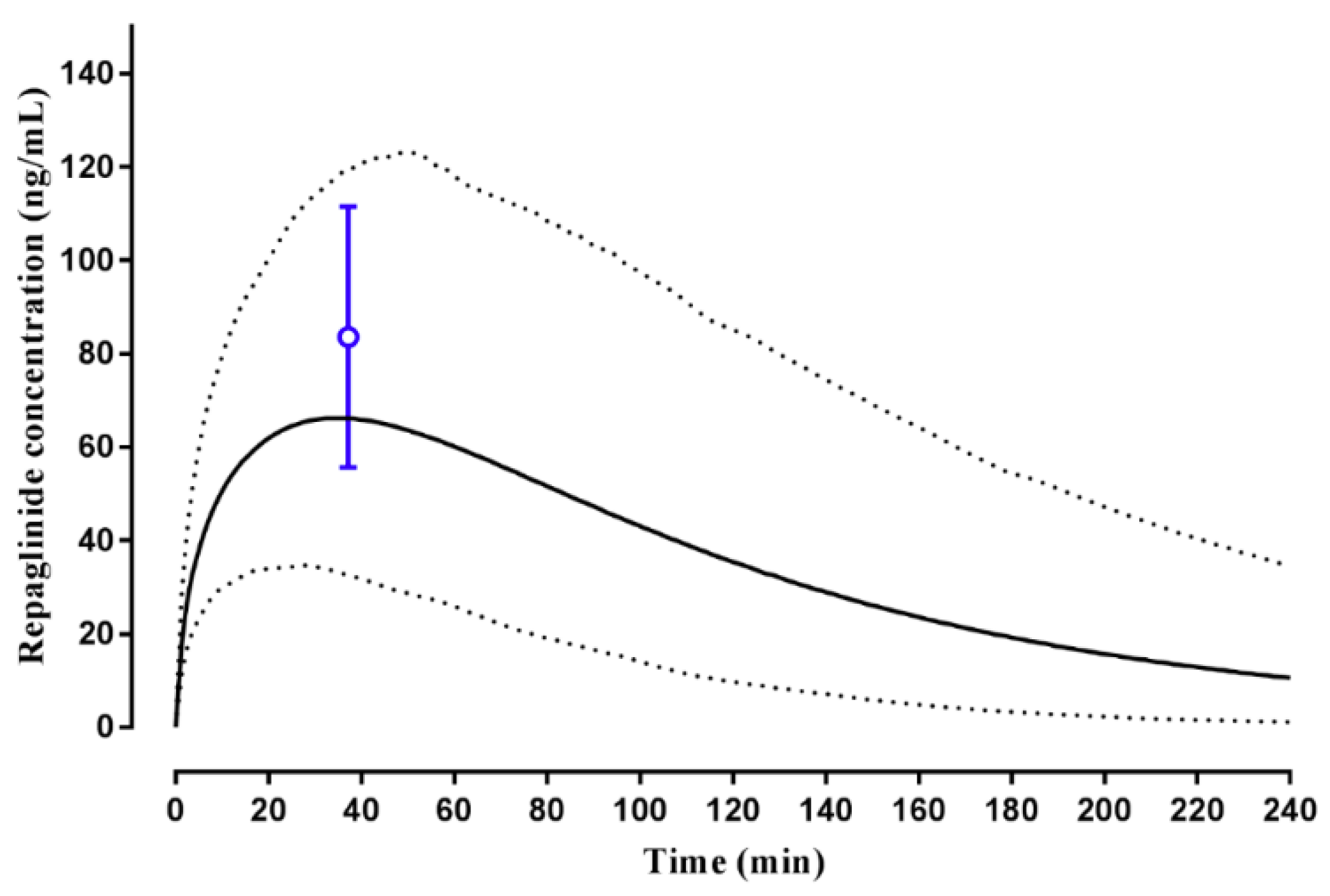
2.2.1. Repaglinide
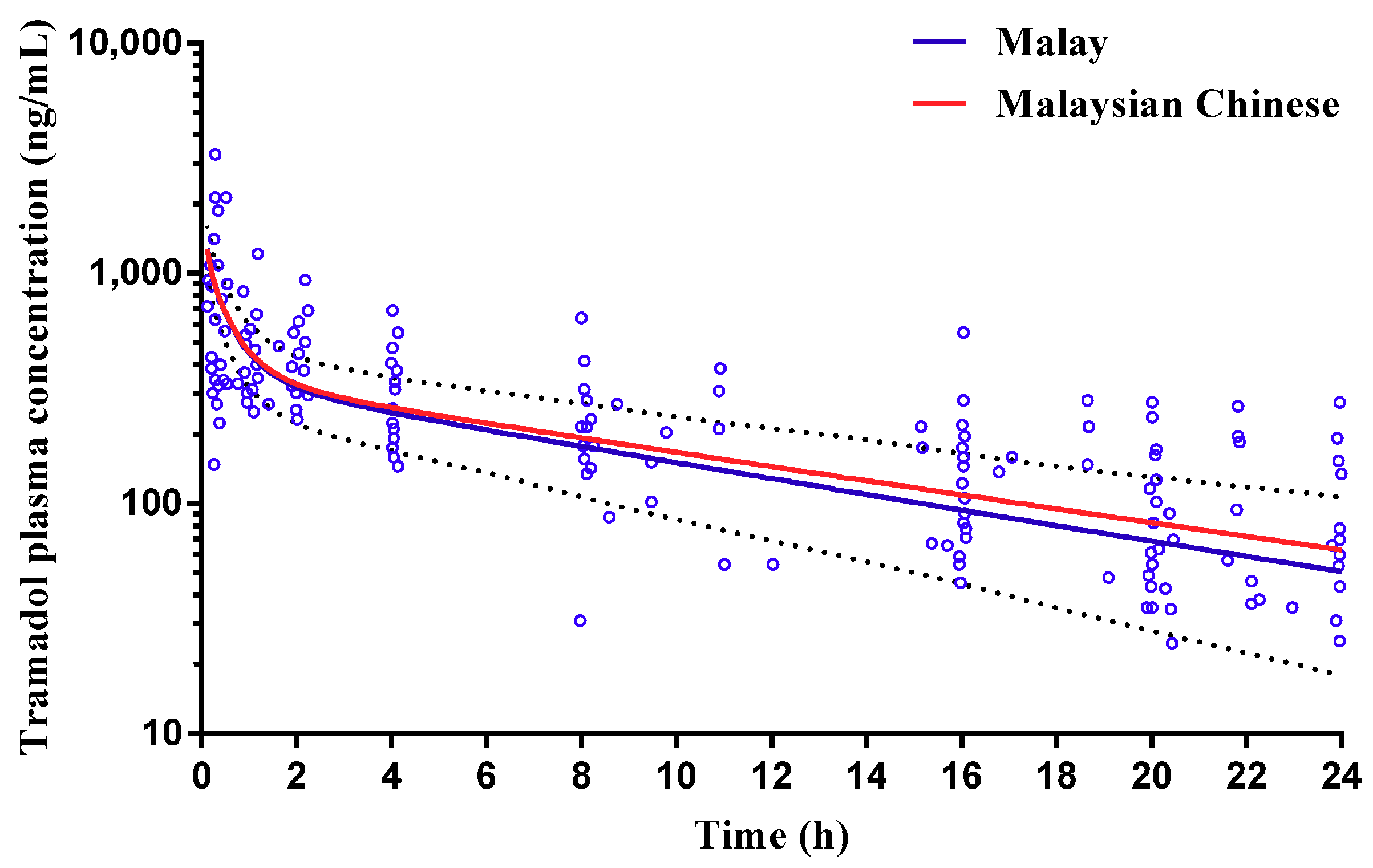
2.2.2. Tramadol
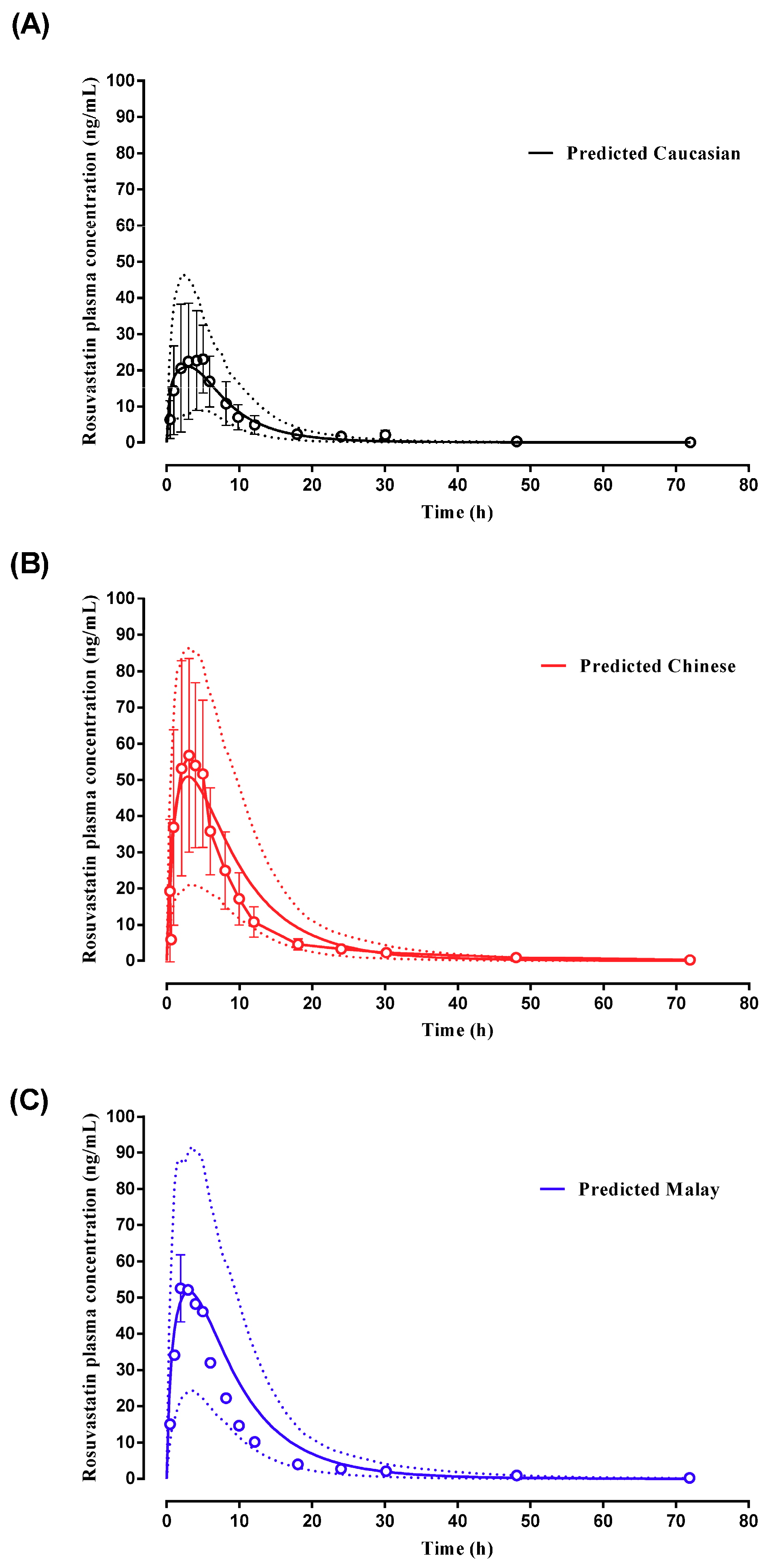
2.2.3. Rosuvastatin
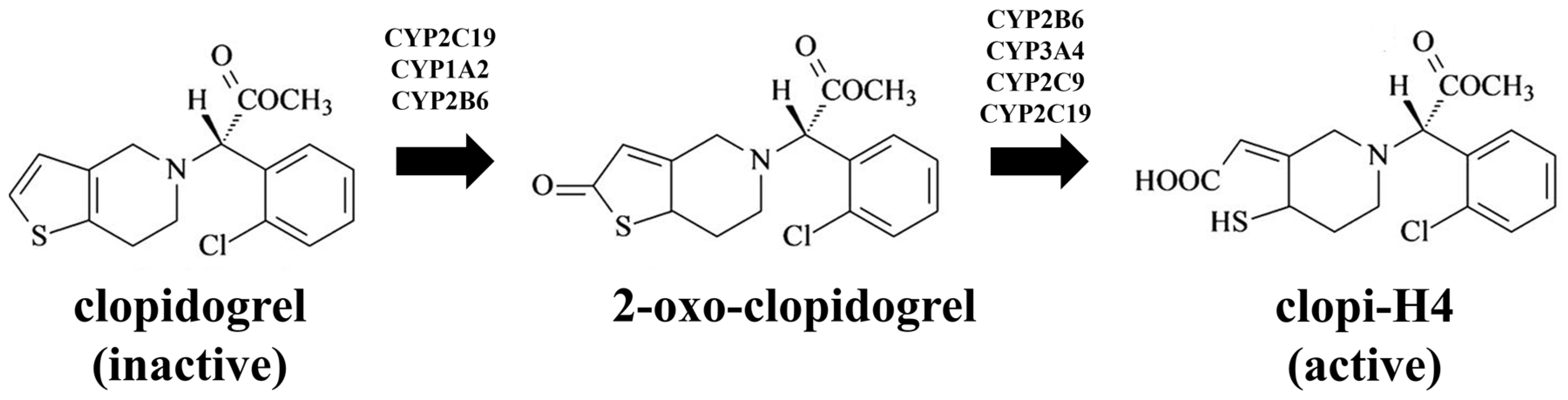
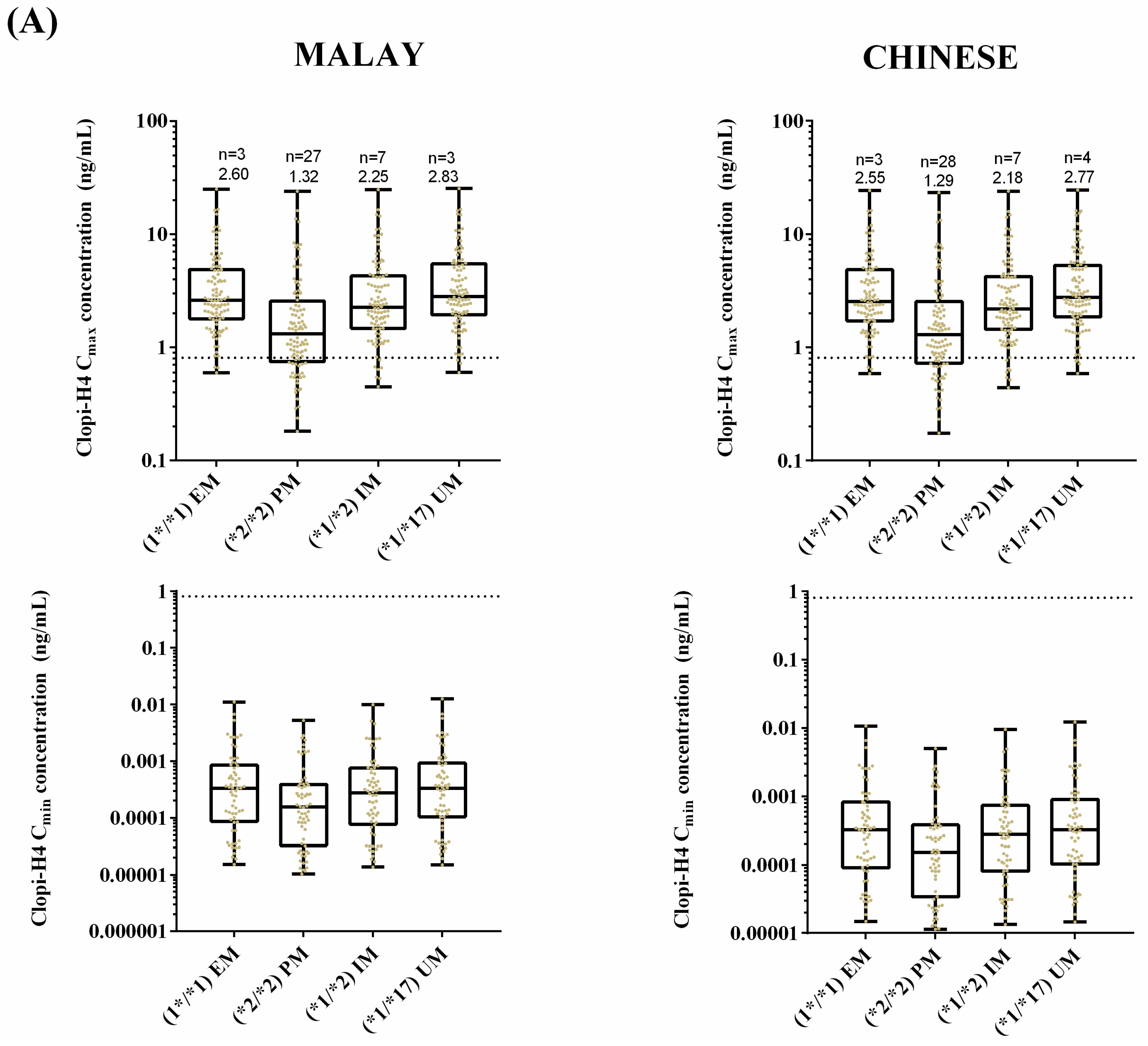
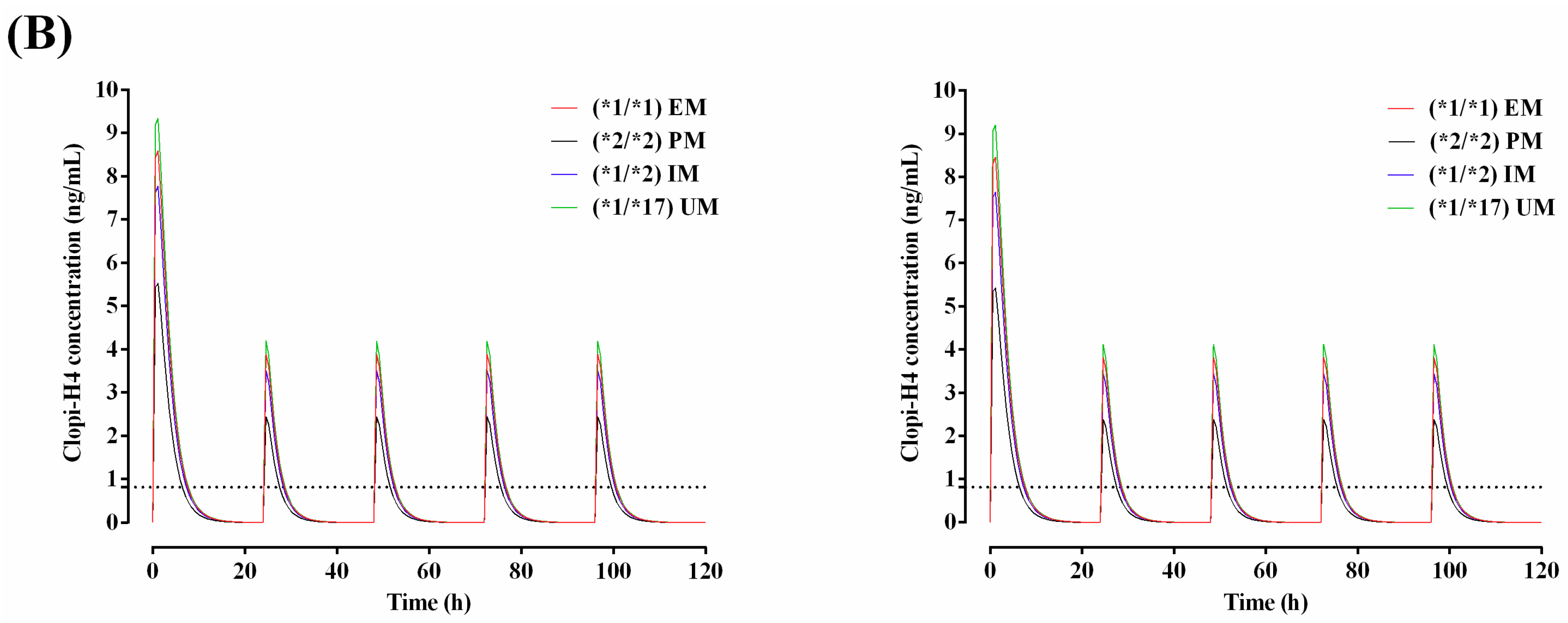
2.3. Step 3: Prediction of the Impact of CYP2C19 Polymorphisms on Clopidogrel Pharmacokinetics in Malaysians
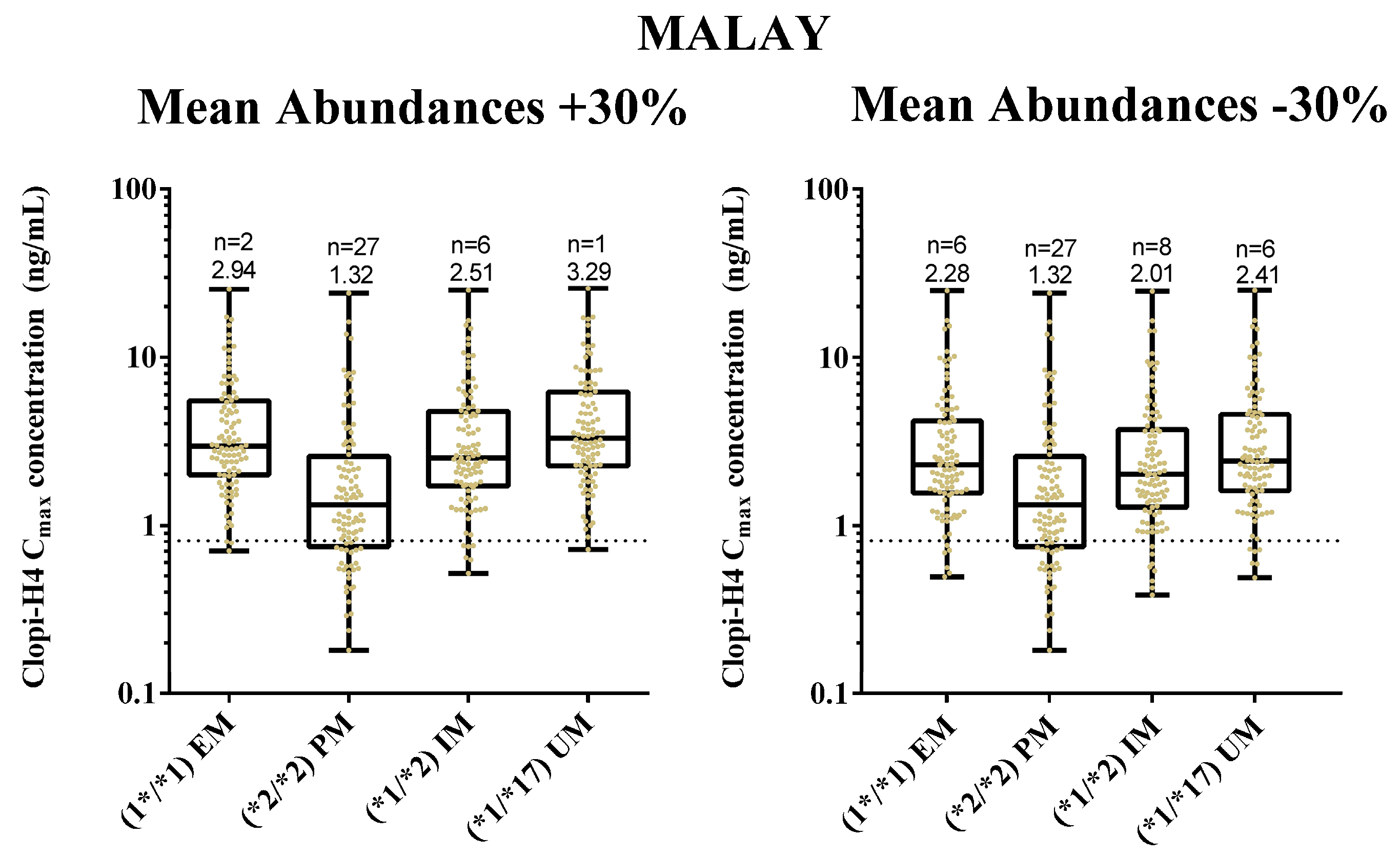
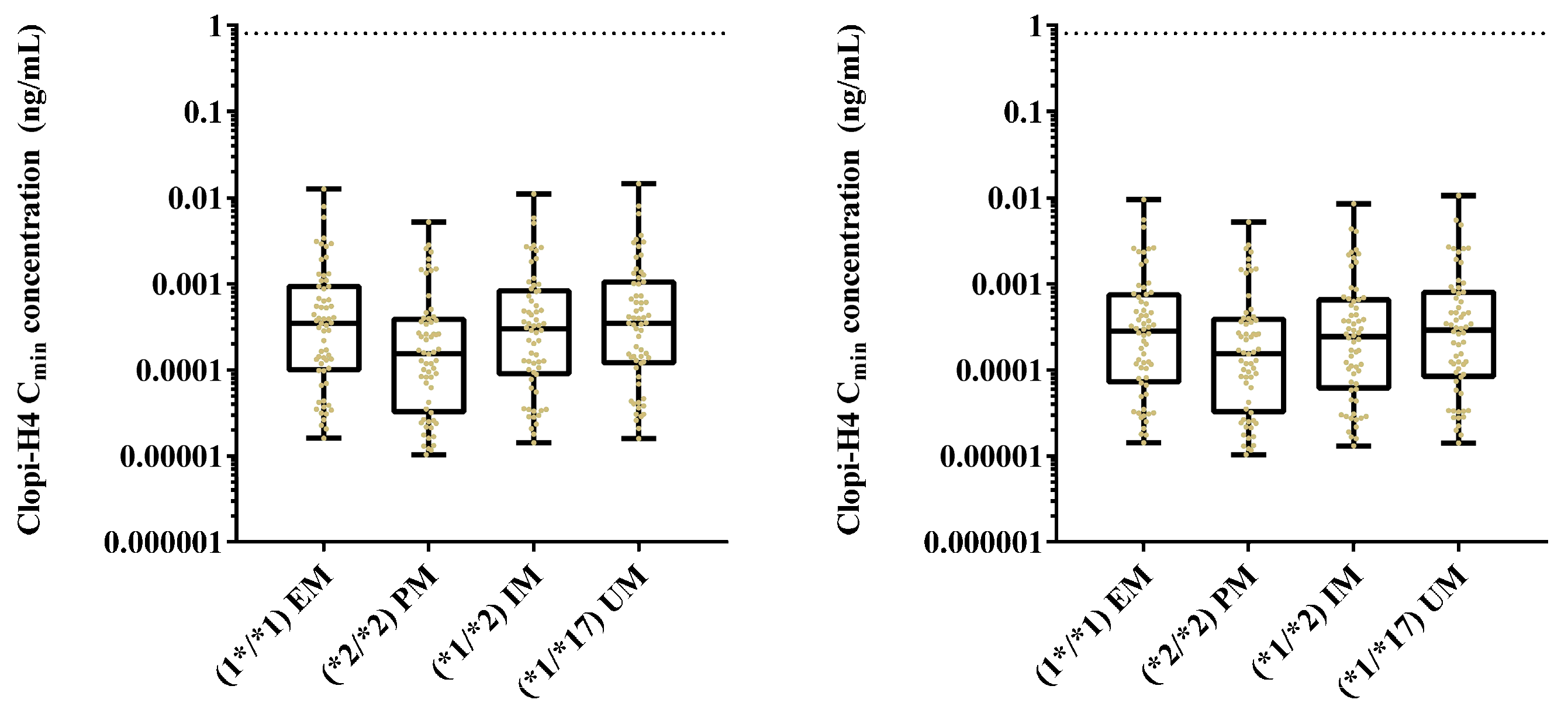
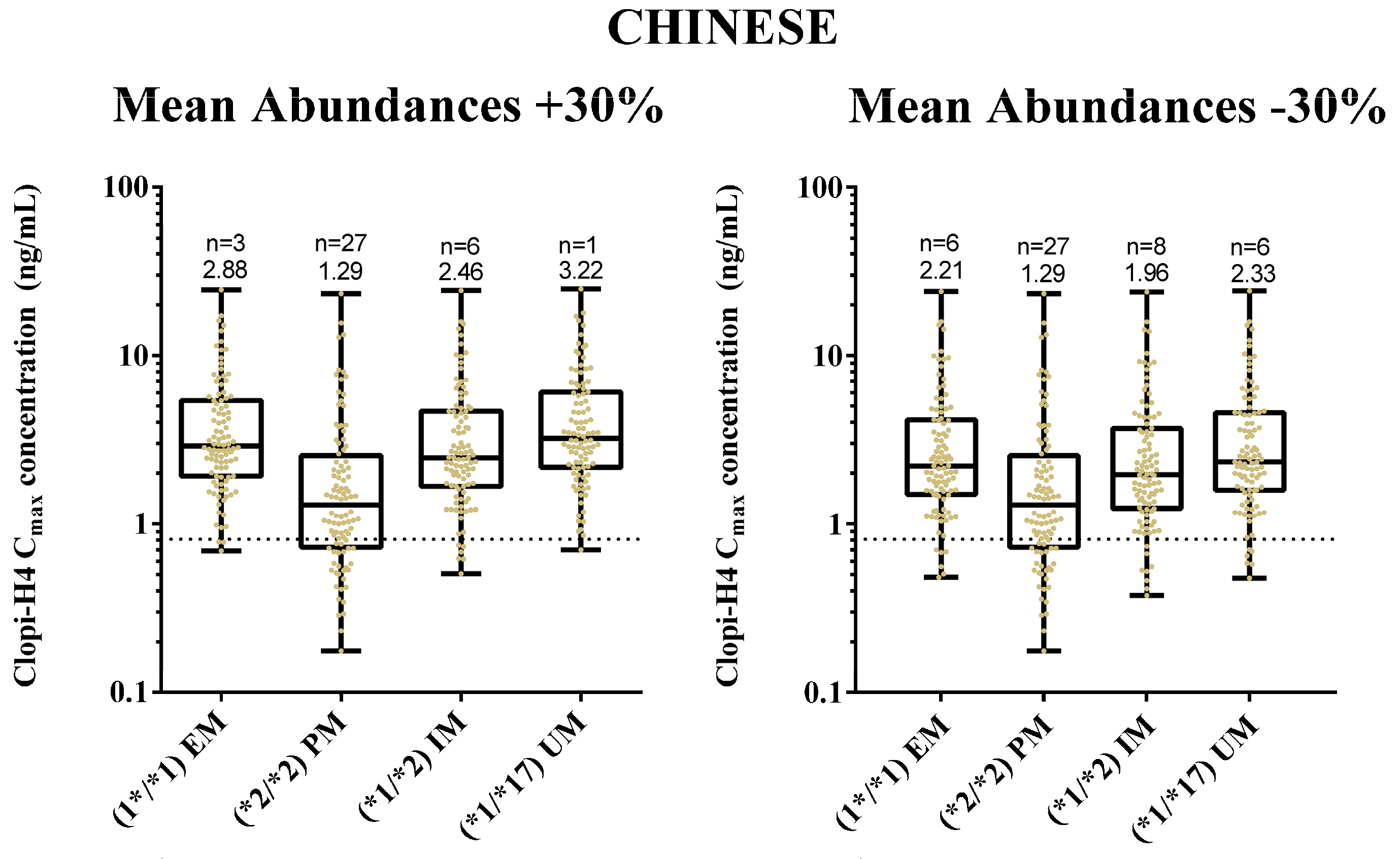
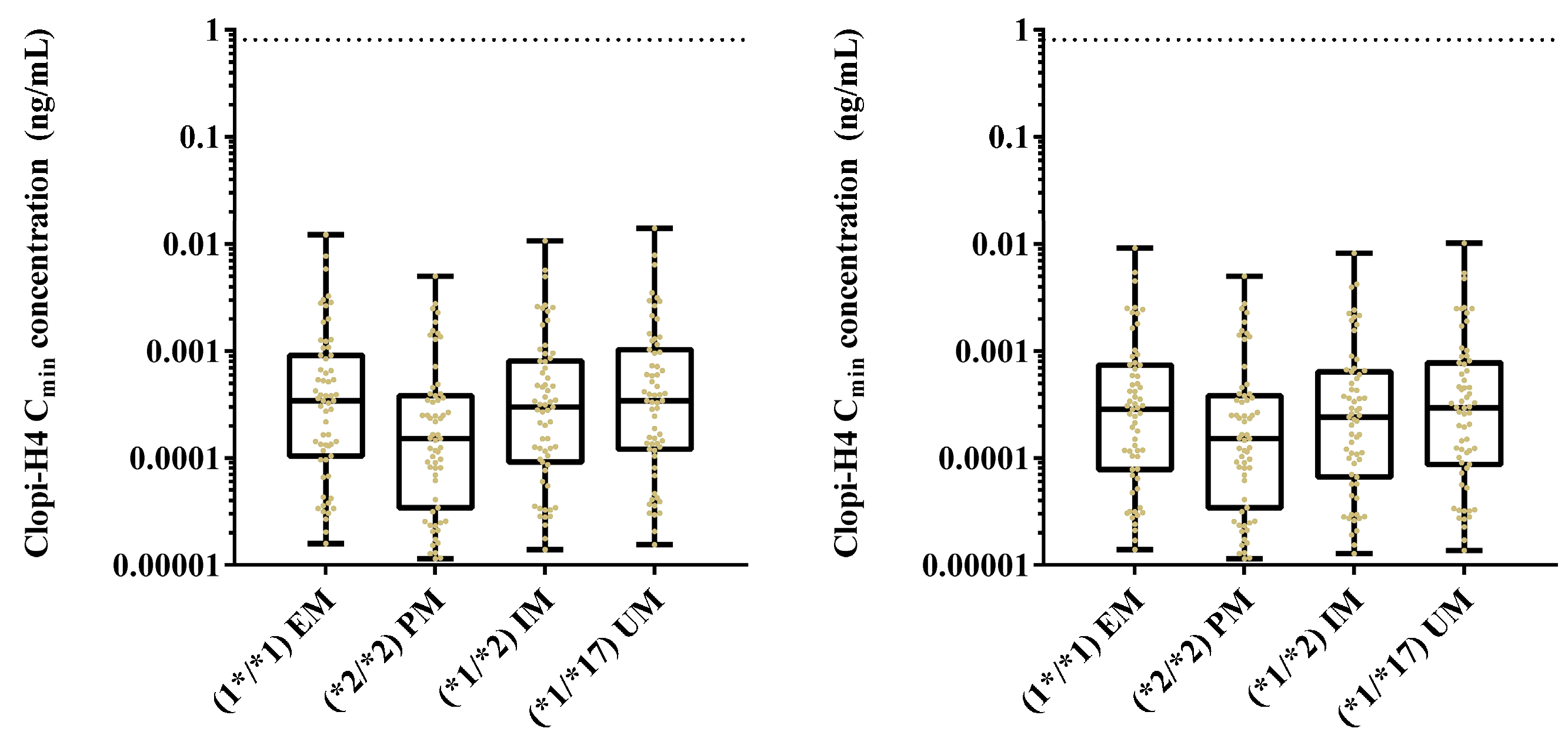
2.4. Step 4: Sensitivity Analysis for CYP2C19 Hepatic Abundances
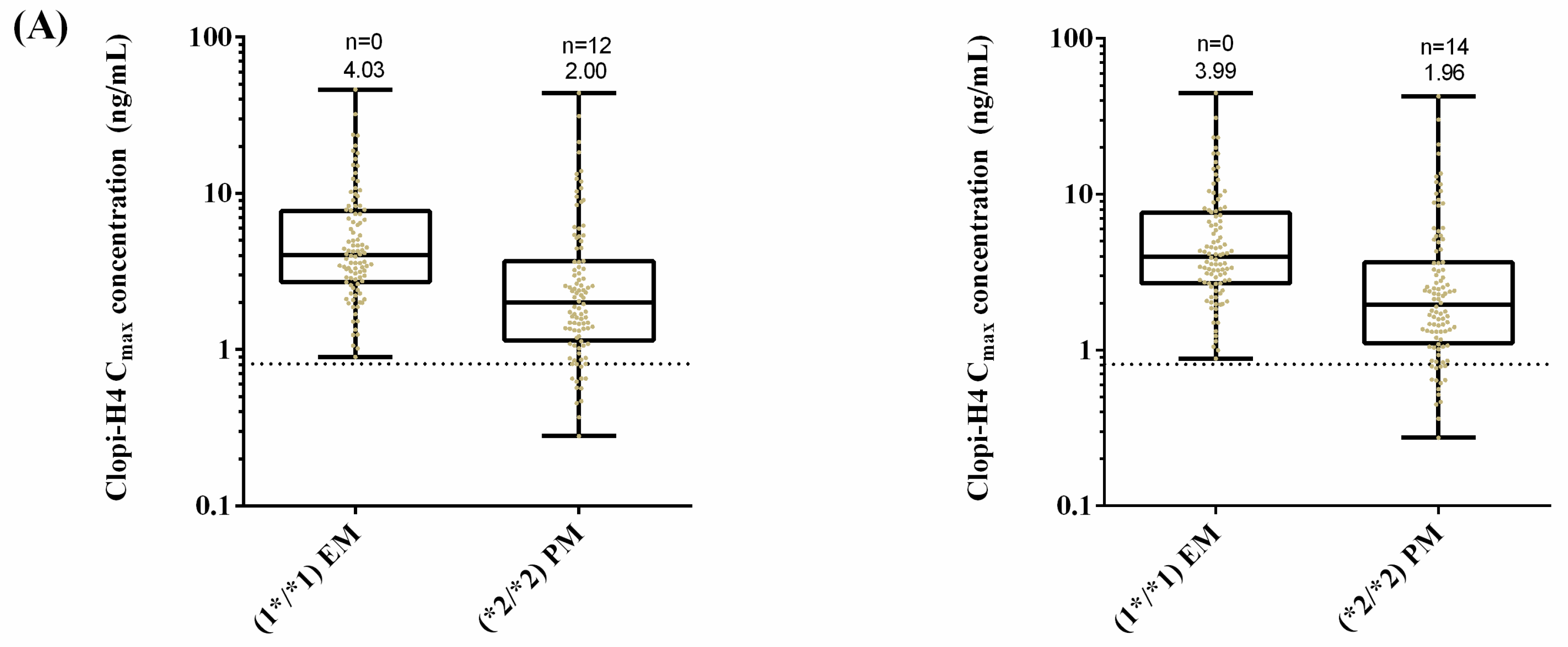
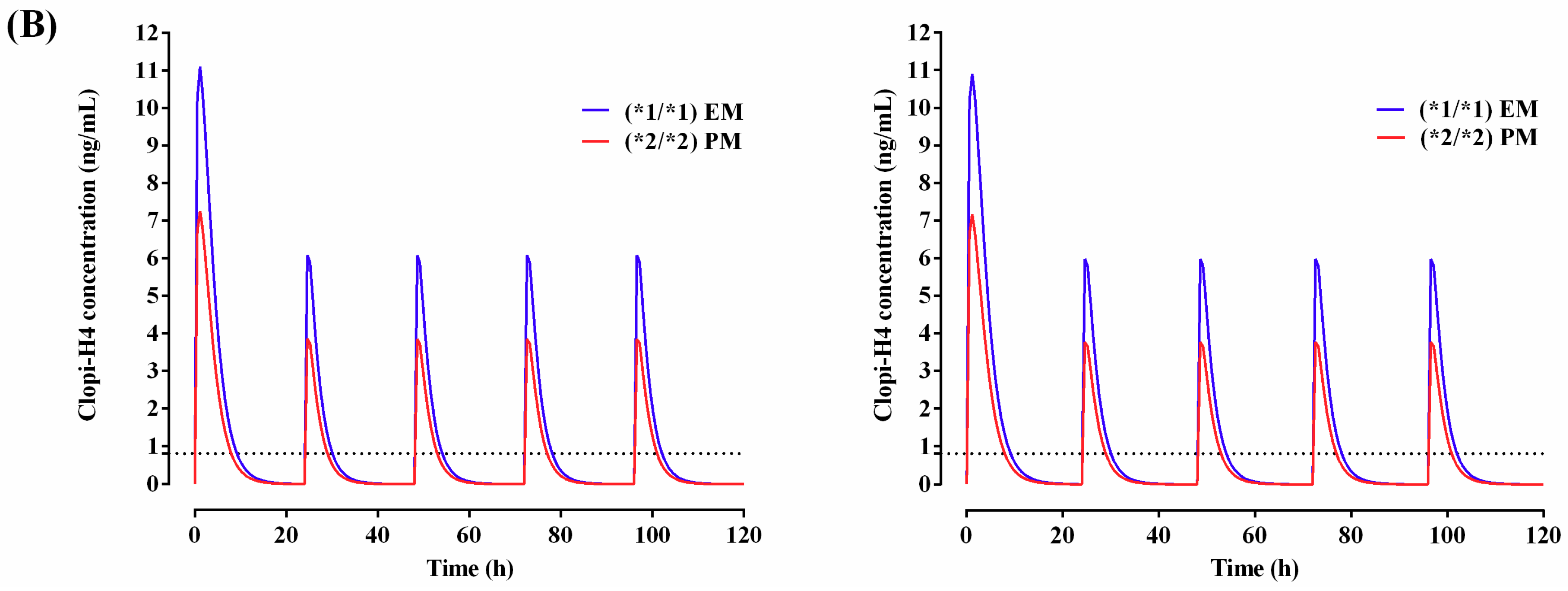
2.5. Step 5: Dose Optimization in CYP2C19 Poor Metabolisers
3. Discussion
3.1. Step 1: Malaysian Population Development
3.2. Step 2: Adult Simulations: Validation with Repaglinide, Tramadol and Rosuvastatin
3.3. Step 3: Prediction of the Impact of CYP2C19 Polymorphisms on Clopidogrel Pharmacokinetics in Malaysians
3.4. Step 4: Sensitivity Analysis for CYP2C19 Hepatic Abundances
3.5. Step 5: Dose Optimisation in CYP2C19 Poor Metabolisers
3.6. Study Limitations and Future Directions for Clopidogrel Use in Malaysia
4. Materials and Methods
4.1. Model Development
4.1.1. Step 1: Malaysian Population Development
Compound Selection and Clinical Studies
4.1.2. Step 2: Adult Simulations: Validation with Repaglinide, Tramadol and Rosuvastatin
4.1.3. Step 3: Prediction of the Impact of CYP2C19 Polymorphisms on Clopidogrel Pharmacokinetics in Malaysians
4.1.4. Step 4: Sensitivity Analysis for CYP2C19 Hepatic Abundances
4.1.5. Step 5: Dose Optimisation in CYP2C19 Poor Metabolisers
4.2. Data Analysis
4.3. Predictive Performance
4.4. Visual Predictive Checks
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ho, M. Current Population Estimates, Malaysia, 2014–2016; The Office of Chief Statistician Malaysia, Department of Statistics Malaysia: Putrajaya, Malaysia, 2016. [Google Scholar]
- National Pharmaceutical Regulatory Agency, Ministry of Health Malaysia. National Centre for Adverse Drug Reaction Monitoring Annual Report; National Pharmaceutical Regulatory Agency: Putrajaya, Malaysia, 2018. [Google Scholar]
- Xie, H.G.; Kim, R.B.; Wood, A.J.; Michael Stein, C.M. Molecular basis of ethnic differences in drug disposition and response. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 815–850. [Google Scholar] [CrossRef] [PubMed]
- Yusoff, N.; Saleem, M.; Nagaya, D.; Yahaya, B.; Rasmaizatul Akma, R.; Moosa, N.; Ismail, R.; Tan, C. Cross-ethnic distribution of clinically relevant Cyp2c19 genotypes and haplotypes. J. Pharm. Pharmacoproteomics 2015, 6, 147. [Google Scholar] [CrossRef]
- Yang, Y.; Wong, L.; Lee, T.; Mustafa, A.; Mohamed, Z.; Lang, C.C. Genetic polymorphism of cytochrome P450 2C19 in healthy Malaysian subjects. Br. J. Clin. Pharmacol. 2004, 58, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Health Facts 2017, Ministry of Health Malaysia, Putrajaya, Malaysia. Available online: http://www.moh.gov.my/english.php/pages/view/56 (accessed on 9 July 2018).
- Cattaneo, M. Response variability to clopidogrel: Is tailored treatment, based on laboratory testing, the right solution? J. Thromb. Haemost. 2012, 10, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Topol, E.J. Scientific and therapeutic advances in antiplatelet therapy. Nat. Rev. Drug Discov. 2003, 2, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Simon, T.; Bhatt, D.L.; Bergougnan, L.; Farenc, C.; Pearson, K.; Perrin, L.; Vicaut, E.; Lacreta, F.; Hurbin, F.; Dubar, M. Genetic polymorphisms and the impact of a higher clopidogrel dose regimen on active metabolite exposure and antiplatelet response in healthy subjects. Clin. Pharmacol. Ther. 2011, 90, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Mega, J.L.; Close, S.L.; Wiviott, S.D.; Shen, L.; Hockett, R.D.; Brandt, J.T.; Walker, J.R.; Antman, E.M.; Macias, W.; Braunwald, E. Cytochrome p-450 polymorphisms and response to clopidogrel. N. Engl. J. Med. 2009, 360, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Park, P.; Hong, S.; Park, J.Y. The effect of CYP2C19 polymorphism on the pharmacokinetics and pharmacodynamics of clopidogrel: A possible mechanism for clopidogrel resistance. Clin. Pharmacol. Ther. 2008, 84, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Holmes, D.R., Jr.; Dehmer, G.J.; Kaul, S.; Leifer, D.; O‘Gara, P.T.; Stein, C.M. ACCF/AHA clopidogrel clinical alert: Approaches to the fda “boxed warning”: A report of the american college of cardiology foundation task force on clinical expert consensus documents and the american heart association endorsed by the society for cardiovascular angiography and interventions and the society of thoracic surgeons. J. Am. Coll. Cardiol. 2010, 56, 321–341. [Google Scholar] [PubMed]
- Kazui, M.; Nishiya, Y.; Ishizuka, T.; Hagihara, K.; Farid, N.A.; Okazaki, O.; Ikeda, T.; Kurihara, A. Identification of the human cytochrome p450 enzymes involved in the two oxidative steps in the bioactivation of clopidogrel to its pharmacologically active metabolite. Drug Metab. Dispos. Biol. Fate Chem. 2010, 38, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Djebli, N.; Fabre, D.; Boulenc, X.; Fabre, G.; Sultan, E.; Hurbin, F. Physiologically-based pharmacokinetic modeling for sequential metabolism: Effect of CYP2C19 genetic polymorphism on clopidogrel and clopidogrel active metabolite pharmacokinetics. Drug Metab. Dispos. 2015, 43, 510–522. [Google Scholar] [CrossRef] [PubMed]
- Cappola, T.P.; Margulies, K.B. Functional genomics applied to cardiovascular medicine. Circulation 2011, 124, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.Q.; Wang, D.G.; Yang, H.; Cao, H. Cytochrome P450 CYP 2C19*2 associated with adverse 1-year cardiovascular events in patients with acute coronary syndrome. PLoS ONE 2015, 10, e0132561. [Google Scholar] [CrossRef] [PubMed]
- Shuldiner, A.R.; O’Connell, J.R.; Bliden, K.P.; Gandhi, A.; Ryan, K.; Horenstein, R.B.; Damcott, C.M.; Pakyz, R.; Tantry, U.S.; Gibson, Q.; et al. Association of cytochrome P450 2C19 genotype with the antiplatelet effect and clinical efficacy of clopidogrel therapy. JAMA 2009, 302, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Brandt, J.T.; Close, S.L.; Iturria, S.J.; Payne, C.D.; Farid, N.A.; Ernest, C.S., 2nd; Lachno, D.R.; Salazar, D.; Winters, K.J. Common polymorphisms of CYP2C19 and CYP2C9 affect the pharmacokinetic and pharmacodynamic response to clopidogrel but not prasugrel. J. Thromb. Haemost. 2007, 5, 2429–2436. [Google Scholar] [CrossRef] [PubMed]
- Home Page of the Pharmacogene Variation (Pharmvar) Consortium. Available online: https://www.pharmvar.org/gene/CYP2C19 (accessed on 9 July 2018).
- Yin, T.; Miyata, T. Pharmacogenomics of clopidogrel: Evidence and perspectives. Thromb. Res. 2011, 128, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.M.; Sheau Chin, L.; Azri Mohamed Noor, D.; Sk Abdul Kader, M.A.; Kah Hay, Y.; Ibrahim, B. The personalization of clopidogrel antiplatelet therapy: The role of integrative pharmacogenetics and pharmacometabolomics. Cardiol. Res. Pract. 2017, 2017, 8062796. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.A.; Sangkuhl, K.; Stein, C.M.; Hulot, J.S.; Mega, J.L.; Roden, D.M.; Klein, T.E.; Sabatine, M.S.; Johnson, J.A.; Shuldiner, A.R. Clinical pharmacogenetics implementation consortium guidelines for cyp2c19 genotype and clopidogrel therapy: 2013 update. Clin. Pharmacol. Ther. 2013, 94, 317–323. [Google Scholar] [CrossRef] [PubMed]
- De Morais, S.; Wilkinson, G.R.; Blaisdell, J.; Nakamura, K.; Meyer, U.A.; Goldstein, J.A. The major genetic defect responsible for the polymorphism of s-mephenytoin metabolism in humans. J. Biol. Chem. 1994, 269, 15419–15422. [Google Scholar] [PubMed]
- De Morais, S.; Wilkinson, G.R.; Blaisdell, J.; Meyer, U.A.; Nakamura, K.; Goldstein, J.A. Identification of a new genetic defect responsible for the polymorphism of (s)-mephenytoin metabolism in Japanese. Mol. Pharmacol. 1994, 46, 594–598. [Google Scholar] [PubMed]
- Desta, Z.; Zhao, X.; Shin, J.-G.; Flockhart, D.A. Clinical significance of the cytochrome P450 2C19 genetic polymorphism. Clin. Pharmacokinet. 2002, 41, 913–958. [Google Scholar] [CrossRef] [PubMed]
- Cacabelos, R.; Martinez-Bouza, R.; Carril, J.C.; Fernandez-Novoa, L.; Lombardi, V.; Carrera, I.; Corzo, L.; McKay, A. Genomics and pharmacogenomics of brain disorders. Curr. Pharm. Biotechnol. 2012, 13, 674–725. [Google Scholar] [CrossRef] [PubMed]
- Cacabelos, R.; Martinez, R.; Fernandez-Novoa, L.; Carril, J.C.; Lombardi, V.; Carrera, I.; Corzo, L.; Tellado, I.; Leszek, J.; McKay, A.; et al. Genomics of dementia: Apoe- and CYP2D6-related pharmacogenetics. Int. J. Alzheimer’s Dis. 2012, 2012, 518901. [Google Scholar]
- Mega, J.L.; Simon, T.; Collet, J.P.; Anderson, J.L.; Antman, E.M.; Bliden, K.; Cannon, C.P.; Danchin, N.; Giusti, B.; Gurbel, P.; et al. Reduced-function CYP2C19 genotype and risk of adverse clinical outcomes among patients treated with clopidogrel predominantly for PCI: A meta-analysis. JAMA 2010, 304, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Mejin, M.; Tiong, W.N.; Lai, L.Y.; Tiong, L.L.; Bujang, A.M.; Hwang, S.S.; Ong, T.K.; Fong, A.Y. CYP2C19 genotypes and their impact on clopidogrel responsiveness in percutaneous coronary intervention. Int. J. Clin. Pharm. 2013, 35, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Gurbel, P.A.; Tantry, U.S. Do platelet function testing and genotyping improve outcome in patients treated with antithrombotic agents?: Platelet function testing and genotyping improve outcome in patients treated with antithrombotic agents. Circulation 2012, 125, 1276–1287. [Google Scholar] [CrossRef] [PubMed]
- Krishna, V.; Diamond, G.A.; Kaul, S. Do platelet function testing and genotyping improve outcome in patients treated with antithrombotic agents?: The role of platelet reactivity and genotype testing in the prevention of atherothrombotic cardiovascular events remains unproven. Circulation 2012, 125, 1288–1303. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.S.; Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Jameson, J.L.; Longo, D.L. Precision medicine--personalized, problematic, and promising. N. Engl. J. Med. 2015, 372, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Advancing Regulatory Science for Public Health—A Framework for FDA’S Regulatory Science Initiative; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2010.
- U.S. Food and Drug Administration. Guidance for industry clinical lactation studies—Study design, data analysis, and recommendations for labeling. Fed. Regist. 2005, 70, 6697. [Google Scholar]
- Zhang, L.; Reynolds, K.S.; Zhao, P.; Huang, S.-M. Drug interactions evaluation: An integrated part of risk assessment of therapeutics. Toxicol. Appl. Pharmacol. 2010, 243, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Lee, S.L.; Lawrence, X.Y. Mechanistic approaches to predicting oral drug absorption. AAPS J. 2009, 11, 217–224. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Drug Development and Drug Interactions; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2011.
- U.S. Food and Drug Administration. Physiologically Based Pharmacokinetic Analyses—Format and Content. Guidance for Industry; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2016. [Google Scholar]
- Olafuyi, O.; Coleman, M.; Badhan, R.K.S. The application of physiologically based pharmacokinetic modelling to assess the impact of antiretroviral-mediated drug-drug interactions on piperaquine antimalarial therapy during pregnancy. Biopharm. Drug Dispos. 2017, 38, 464–478. [Google Scholar] [CrossRef] [PubMed]
- Olafuyi, O.; Coleman, M.; Badhan, R.K.S. Development of a paediatric physiologically based pharmacokinetic model to assess the impact of drug-drug interactions in tuberculosis co-infected malaria subjects: A case study with artemether-lumefantrine and the CYP3A4-inducer rifampicin. Eur. J. Pharm. Sci. 2017, 106, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Hatorp, V. Clinical pharmacokinetics and pharmacodynamics of repaglinide. Clin. Pharmacokinet. 2002, 41, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Hatorp, V.; Huang, W.-C.; Strange, P. Repaglinide pharmacokinetics in healthy young adult and elderly subjects. Clin. Ther. 1999, 21, 702–710. [Google Scholar] [CrossRef]
- Zhai, X.-J.; Hu, K.; Chen, F.; Lu, Y.-N. Comparative bioavailability and tolerability of a single 2-mg dose of 2 repaglinide tablet formulations in fasting, healthy chinese male volunteers: An open-label, randomized-sequence, 2-period crossover study. Curr. Ther. Res. 2013, 75, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Ruzilawati, A.; Gan, S. CYP3A4 genetic polymorphism influences repaglinide’s pharmacokinetics. Pharmacology 2010, 85, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.; Ismail, R.; Adnan, W.W.; Wan, Z. Correlation of tramadol pharmacokinetics and CYP2D6* 10 genotype in malaysian subjects. J. Pharm. Biomed. Anal. 2002, 30, 189–195. [Google Scholar] [CrossRef]
- T’jollyn, H.; Snoeys, J.; Vermeulen, A.; Michelet, R.; Cuyckens, F.; Mannens, G.; Van Peer, A.; Annaert, P.; Allegaert, K.; Van Bocxlaer, J. Physiologically based pharmacokinetic predictions of tramadol exposure throughout pediatric life: An analysis of the different clearance contributors with emphasis on CYP2D6 maturation. AAPS J. 2015, 17, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Ryan, S.; Birmingham, B.; Zalikowski, J.; March, R.; Ambrose, H.; Moore, R.; Lee, C.; Chen, Y.; Schneck, D. Rosuvastatin pharmacokinetics and pharmacogenetics in white and asian subjects residing in the same environment. Clin. Pharmacol. Ther. 2005, 78, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Karaźniewicz-Łada, M.; Danielak, D.; Burchardt, P.; Kruszyna, Ł.; Komosa, A.; Lesiak, M.; Główka, F. Clinical pharmacokinetics of clopidogrel and its metabolites in patients with cardiovascular diseases. Clin. Pharmacokinet. 2014, 53, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Naghavi, M.; Allen, C.; Barber, R.M.; Bhutta, Z.A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; Coates, M.M.; et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Reddy, K.S.; Yusuf, S. Emerging epidemic of cardiovascular disease in developing countries. Circulation 1998, 97, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Greving, J.P.; Buskens, E.; Koffijberg, H.; Algra, A. Cost-effectiveness of aspirin treatment in the primary prevention of cardiovascular disease events in subgroups based on age, gender, and varying cardiovascular risk. Circulation 2008, 117, 2875–2883. [Google Scholar] [CrossRef] [PubMed]
- Luk, H.H.; Pang, J.; Li, L.S.W.; Ng, M. Use of antiplatelet drugs in the stroke unit of a hong kong hospital. Pharm. World Sci. 2005, 27, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, S.; Fukuuchi, Y.; Yamaguchi, T. The safety and efficacy of clopidogrel versus ticlopidine in japanese stroke patients: Combined results of two phase iii, multicenter, randomized clinical trials. J. Neurol. 2009, 256, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.B.; Lee, K.A.; Lee, K.Y. Cytochrome P450 2C19 polymorphism is associated with reduced clopidogrel response in cerebrovascular disease. Yonsei Med. J. 2011, 52, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Man, M.; Farmen, M.; Dumaual, C.; Teng, C.H.; Moser, B.; Irie, S.; Noh, G.J.; Njau, R.; Close, S.; Wise, S. Genetic variation in metabolizing enzyme and transporter genes: Comprehensive assessment in 3 major East Asian subpopulations with comparison to Caucasians and Africans. J. Clin. Pharmacol. 2010, 50, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Gaedigk, A. Interethnic differences of drug-metabolizing enzymes. Int. J. Clin. Pharmacol. Ther. 2000, 38, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jin, J.Y.; Mukadam, S.; Malhi, V.; Kenny, J.R. Application of IVIVE and PBPK modeling in prospective prediction of clinical pharmacokinetics: Strategy and approach during the drug discovery phase with four case studies. Biopharm. Drug Dispos. 2012, 33, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Budha, N.R.; Leabman, M.; Jin, J.Y.; Wada, D.R.; Baruch, A.; Peng, K.; Tingley, W.G.; Davis, J.D. Modeling and simulation to support phase 2 dose selection for RG7652, a fully human monoclonal antibody against proprotein convertase subtilisin/kexin type 9. AAPS J. 2015, 17, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Cecchelli, R.; Berezowski, V.; Lundquist, S.; Culot, M.; Renftel, M.; Dehouck, M.-P.; Fenart, L. Modelling of the blood-brain barrier in drug discovery and development. Nat. Rev. Drug Discov. 2007, 6, 650–661. [Google Scholar] [CrossRef] [PubMed]
- Zakaria, Z.; Badhan, R.K. The impact of CYP2B6 polymorphisms on the interactions of efavirenz with lumefantrine: Implications for paediatric antimalarial therapy. Eur. J. Pharm. Sci. 2018, 119, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Bhindi, R.; Ormerod, O.; Newton, J.; Banning, A.; Testa, L. Interaction between statins and clopidogrel: Is there anything clinically relevant? QJM 2008, 101, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Rolan, P. Plasma protein binding displacement interactions—Why are they still regarded as clinically important? Br. J. Clin. Pharmacol. 1994, 37, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Hamzah, A.A.; Bakar, Z.; Sani, N.A.; Tan, J.; Damanhuri, M.A.; Aripin, K.; Rani, M.M.; Noh, N.; Razali, R.; Mohamad, M. Relationship between education and cognitive performance among healthy malay adults. Sains Malays. 2016, 45, 1371–1379. [Google Scholar]
- Khor, G.; Duraisamy, G.; Loh, S.; Green, T.; Skeaff, C. Dietary and blood folate status of malaysian women of childbearing age. Asia Pac. J. Clin. Nutr. 2006, 15, 341–349. [Google Scholar] [PubMed]
- Lim, K.G. A review of adult obesity research in malaysia. Med. J. Malays. 2016, 71, 1–19. [Google Scholar]
- Wang, X.-Q.; Shen, C.-L.; Wang, B.-N.; Huang, X.-H.; Hu, Z.-L.; Li, J. Genetic polymorphisms of CYP2C19*2 and ABCB1 C3435T affect the pharmacokinetic and pharmacodynamic responses to clopidogrel in 401 patients with acute coronary syndrome. Gene 2015, 558, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.Y.; Tan, K.; Tan, H.-C.; Huan, P.-T.; Li, B.; Phua, Q.-H.; Lee, H.-K.; Lee, C.-H.; Low, A.; Becker, R.C. CYP2C19 and PON1 polymorphisms regulating clopidogrel bioactivation in chinese, malay and indian subjects. Pharmacogenomics 2012, 13, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Rosdi, R.A.; Yusoff, N.M.; Ismail, R.; Choon, T.S.; Musa, N.; Yusoff, S. A minireview of CYP2C9 and CYP2C19 single nucleotide polymorphisms (SNPS) among malaysian populations. J. Biomed. Clin. Sci. 2017, 2, 47–52. [Google Scholar]
- Wang, H.-Y.; Chen, X.; Jiang, J.; Shi, J.; Hu, P. Evaluating a physiologically based pharmacokinetic model for predicting the pharmacokinetics of midazolam in chinese after oral administration. Acta Pharmacol. Sin. 2016, 37, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Cleary, Y.; Parrott, N.; Hu, P.; Weber, C.; Wang, Y.; Yin, O.Q.; Shi, J. Evaluating a physiologically based pharmacokinetic model for prediction of omeprazole clearance and assessing ethnic sensitivity in CYP2C19 metabolic pathway. Eur. J. Clin. Pharmacol. 2015, 71, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.S.; Basri, H.B.; Hin, L.P.; Stanslas, J. Genetic polymorphisms and drug interactions leading to clopidogrel resistance: Why the asian population requires special attention. Int. J. Neurosci. 2013, 123, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.D.; Wang, W.; Yang, M.; Zhang, K.; Chen, J.; Qiao, S.; Yan, H.; Wu, Y.; Huang, X.; Xu, B.; et al. Randomized comparisons of double-dose clopidogrel or adjunctive cilostazol versus standard dual antiplatelet in patients with high posttreatment platelet reactivity: Results of the creative trial. Circulation 2018, 137, 2231–2245. [Google Scholar] [CrossRef] [PubMed]
- Bossard, M.; Granger, C.B.; Tanguay, J.F.; Montalescot, G.; Faxon, D.P.; Jolly, S.S.; Widimsky, P.; Niemela, K.; Steg, P.G.; Natarajan, M.K.; et al. Double-dose versus standard-dose clopidogrel according to smoking status among patients with acute coronary syndromes undergoing percutaneous coronary intervention. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, Y.; Wang, L.; Geng, Y.; Gu, J.; Hao, Q.; Wang, H.; Qi, P. Effects of dual-dose clopidogrel, clopidogrel combined with tongxinluo capsule, and ticagrelor on patients with coronary heart disease and CYP2C19*2 gene mutation after percutaneous coronary interventions (PCI). Med. Sci. Monit. 2017, 23, 3824–3830. [Google Scholar] [CrossRef] [PubMed]
- Collet, J.-P.; Hulot, J.-S.; Anzaha, G.; Pena, A.; Chastre, T.; Caron, C.; Silvain, J.; Cayla, G.; Bellemain-Appaix, A.; Vignalou, J.-B.; et al. High doses of clopidogrel to overcome genetic resistance. JACC Cardiovasc. Interv. 2011, 4, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tang, H.L.; Hu, Y.F.; Xie, H.G. The gain-of-function variant allele CYP2C19*17: A double-edged sword between thrombosis and bleeding in clopidogrel-treated patients. J. Thromb. Haemost. 2012, 10, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Zabalza, M.; Subirana, I.; Sala, J.; Lluis-Ganella, C.; Lucas, G.; Tomas, M.; Masia, R.; Marrugat, J.; Brugada, R.; Elosua, R. Meta-analyses of the association between cytochrome CYP2C19 loss- and gain-of-function polymorphisms and cardiovascular outcomes in patients with coronary artery disease treated with clopidogrel. Heart 2012, 98, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Bauer, T.; Bouman, H.J.; van Werkum, J.W.; Ford, N.F.; ten Berg, J.M.; Taubert, D. Impact of CYP2C19 variant genotypes on clinical efficacy of antiplatelet treatment with clopidogrel: Systematic review and meta-analysis. BMJ 2011, 343, d4588. [Google Scholar] [CrossRef] [PubMed]
- Wilffert, B.; Swen, J.; Mulder, H.; Touw, D.; Maitland-Van der Zee, A.-H.; Deneer, V.; KNMP Working Group Pharmacogenetics. From evidence based medicine to mechanism based medicine. Reviewing the role of pharmacogenetics. Int. J. Clin. Pharm. 2013, 35, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Goh, L.L.; Lim, C.W.; Sim, W.C.; Toh, L.X.; Leong, K.P. Analysis of genetic variation in CYP450 genes for clinical implementation. PLoS ONE 2017, 12, e0169233. [Google Scholar] [CrossRef] [PubMed]
- Varma, M.V.S.; Lai, Y.; Kimoto, E.; Goosen, T.C.; El-Kattan, A.F.; Kumar, V. Mechanistic modeling to predict the transporter- and enzyme-mediated drug-drug interactions of repaglinide. Pharm. Res. 2013, 30, 1188–1199. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.H.; Park, W.S.; Han, S.; Park, G.J.; Lee, J.; Hong, T.; Jeon, S.; Yim, D.S. Physiologically-based pharmacokinetic predictions of intestinal BCRP-mediated drug interactions of rosuvastatin in Koreans. Korean J. Physiol. Pharmacol. 2018, 22, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Edginton, A.N.; Schmitt, W.; Willmann, S. Development and evaluation of a generic physiologically based pharmacokinetic model for children. Clin. Pharmacokinet. 2006, 45, 1013–1034. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, G.; Hattis, D.; Russ, A.; Sonawane, B. Physiologically based pharmacokinetic (PBPK) modeling of caffeine and theophylline in neonates and adults: Implications for assessing children’s risks from environmental agents. J. Toxicol. Environ. Health Part A 2004, 67, 297–329. [Google Scholar] [CrossRef] [PubMed]
- Parrott, N.; Davies, B.; Hoffmann, G.; Koerner, A.; Lave, T.; Prinssen, E.; Theogaraj, E.; Singer, T. Development of a physiologically based model for oseltamivir and simulation of pharmacokinetics in neonates and infants. Clin. Pharmacokinet. 2011, 50, 613–623. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Summary Minutes of the Advisory Committee for Pharmaceutical Science and Clinical Pharmacology. Available online: https://wayback.archive-it.org/7993/20170403224110/https://www.fda.gov/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/AdvisoryCommitteeforPharmaceuticalScienceandClinicalPharmacology/ucm286697.htm (accessed on 29 May 2018).

| Ethnicity | Age (years) | Height (m) | Weight (kg) | BMI (kg/m2) | |
|---|---|---|---|---|---|
| Malay | Mean | 57.75 | 1.63 | 69.79 | 26.19 |
| Median | 57.6 | 1.63 | 69.5 | 25.78 | |
| N | 23114 | 10250 | 12193 | 10170 | |
| SD | 11.62 | 0.08 | 14.24 | 4.35 | |
| Malaysian Chinese | Mean | 62.89 | 1.63 | 67.17 | 25.11 |
| Median | 63.1 | 1.63 | 66 | 24.79 | |
| N | 9929 | 4111 | 5259 | 4086 | |
| SD | 12.04 | 0.08 | 13.18 | 3.84 | |
| Malaysian Indian | Mean | 57.7 | 1.63 | 69.61 | 25.95 |
| Median | 57.3 | 1.64 | 69 | 25.52 | |
| N | 9167 | 4257 | 4809 | 4229 | |
| SD | 11.91 | 0.09 | 13.91 | 4.25 |
| Compound | Validation | CL (L/h) | Cmax (ng/mL) | tmax (h) | AUC (ng/mL·h) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Observed | Predicted | Observed | Predicted | Observed | Predicted | Observed | Predicted | ||
| Repaglinide | Hatorp et al., (2002) (Healthy volunteers) | - | 12.17 (10.03–14.77) | 26.0 µg/L | 32.9 (16.4–47.0) µg/L | 0.83 (0.31–1.35) | 0.54 (0.18 -0.91) | 152.4 (62.8–242.0) µg/L·h | 70.89 (15.51–73.82) µg/L·h |
| Hatorp et al., (1999) (Single dose) (Healthy volunteers) | - | 13.9 (10.33–18.70) | 47.9 (15.9–79.9) | 26.2 (19.61–35.0) | 0.8 (0.2–1.4) | 0.47 (0.31–0.60) | 69.0 (61.2–76.8) | 54.87 (21.57–106.1) | |
| Hatorp et al., (1999) (Multiple doses) (Healthy volunteers) | - | 15.32 (12.24–20.57) | 58.5 (8.1–108.9) | 26.39 (11.53–46.85) | 0.6 (0.5–0.7) | 0.48 (0.32–0.61) | 98.1 (84.54–111.66) | 56.8 (22.59–109.1) | |
| Zhai et al., (2013) (Chinese) 1 | - | 56.66 (21.10–146.00) (Malaysian Chinese) | 20.0 (14.9–25.1) | 18.5 (8.79–28.0) (Malaysian Chinese) | 1.2 (0.5–1.9) | 0.93 (0.82–1.08) (Malaysian Chinese) | 46.3 (31.2–61.4) | 39.51 (16.21–74.56) (Malaysian Chinese) | |
| Ruzilawati et al., (2010) (Malay) | 11.82 (7.86–15.78) | 10.41 (5.29–15.53) | 83.56 (55.63–111.49) | 67.5 (34.4–118) | 0.62 (0.24–1.00) | 0.58 (0.20–0.96) | 340.66 (226.14–455.18) | 151 (33.61–196) | |
| Tramadol | Gan et al., (2002) 2 (Malaysian) | 19.3 (13.1–25.5) | 19.24 (13.31–33.11) (Malay) | - | - | - | - | 5078.4 (3117.3–7039.5) | 4389 (2915–6128) (Malay) |
| 18.36 (11.73–30.19) (Malaysian Chinese) | - | - | - | - | 4716 (3201–6658) (Malaysian Chinese) | ||||
| Rosuvastatin | Lee et al., (2005) (Malay, Chinese, Healthy volunteers Caucasians) | - | 21.87 (19.49–24.53) (Malay) | 50.0 (42.2–59.3) (Malay) | 49.91 (42.68–58.36) (Malay) | 3.00 (0.50–5.00) (Malay) | 3.01 (2.51–3.61) (Malay) | 413 (354–482) (Malay) | 552.84 (473.51–645.46) (Malay) |
| 21.28 (19.17–23.63) (Chinese) | 59.1 (49.8–70.1) (Chinese) | 53.83 (42.01–57.35) (Chinese) | 3.00 (0.50–5.00) (Chinese) | 2.90 (2.43–3.45) (Chinese) | 500 (428–583) (Chinese) | 553.18 (475.13–644.05) (Chinese) | |||
| 23.99 (22.40–25.70) (Healthy volunteers) | 25.0 (21.1–29.6) | 13.49 (10.55–14.28) | 5.00 (1.00–8.00) | 2.99 (2.15–3.16) | 216 (186–252) | 116.15 (98.70–136.68) |
| Biochemistry | Malay | Malaysian Chinese |
|---|---|---|
| Haematocrit (%) | M: 43 b F: 38 c | M: 45.3 F: 40.5 |
| AAG (g/L) | M: 0.65 a F: 0.64 a | M: 0.65 F: 0.64 |
| HSA (g/L) | M: 47.3 b F: 46.3 b | M: 50.34 F: 49.38 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zakaria, Z.H.; Fong, A.Y.Y.; Badhan, R.K.S. Clopidogrel Pharmacokinetics in Malaysian Population Groups: The Impact of Inter-Ethnic Variability. Pharmaceuticals 2018, 11, 74. https://doi.org/10.3390/ph11030074
Zakaria ZH, Fong AYY, Badhan RKS. Clopidogrel Pharmacokinetics in Malaysian Population Groups: The Impact of Inter-Ethnic Variability. Pharmaceuticals. 2018; 11(3):74. https://doi.org/10.3390/ph11030074
Chicago/Turabian StyleZakaria, Zaril H., Alan Y. Y. Fong, and Raj K. S. Badhan. 2018. "Clopidogrel Pharmacokinetics in Malaysian Population Groups: The Impact of Inter-Ethnic Variability" Pharmaceuticals 11, no. 3: 74. https://doi.org/10.3390/ph11030074
APA StyleZakaria, Z. H., Fong, A. Y. Y., & Badhan, R. K. S. (2018). Clopidogrel Pharmacokinetics in Malaysian Population Groups: The Impact of Inter-Ethnic Variability. Pharmaceuticals, 11(3), 74. https://doi.org/10.3390/ph11030074






