Gastroprotective Effects of Sulphated Polysaccharides from the Alga Caulerpa mexicana Reducing Ethanol-Induced Gastric Damage
Abstract
:1. Introduction
2. Results
2.1. Effect of Cm-SP on Ethanol-Induced Gastric Damage
2.2. Histological Assessment
2.3. Effect of Cm-SP on Malondialdehyde (MDA), Glutathione (GSH), and NO2/NO3 Levels
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Marine Alga and Extraction of SP
4.3. Effect of Cm-SP on Ethanol-Induced Gastric Damage
4.4. Histological Assessment
4.5. Determination of GSH Level
4.6. Determination of MDA Level
4.7. Determination of NO2/NO3 Level
4.8. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amandeep, K.; Robin, S.; Ramica, S.; Sunil, K. Peptic ulcer: A review on etiology and pathogenesis. Int. Res. J. Pharm. 2012, 3, 34–38. [Google Scholar]
- Kwiecien, S.; Brzozowski, T.; Konturek, S.J. Effects of reactive oxygen species action on gastric mucosa in various models of mucosal injury. J. Physiol. Pharmacol. 2002, 53, 39–50. [Google Scholar] [PubMed]
- Nayeb-Hashemia, H.; Kaunitz, J.D. Gastroduodenal mucosal defense. Curr. Opin. Gastroenterol. 2009, 25, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K. Gastric cytoprotection by prostaglandin E2 and prostacyclin: Relationship to EP1 and IP receptors. J. Physiol. Pharmacol. 2014, 65, 3–14. [Google Scholar] [PubMed]
- Szabo, S. Gastric cytoprotection is still relevant. J. Gastroenterol. Hepatol. 2014, 29, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Peskar, B.M. Role of cyclooxygenase isoforms in gastric mucosal defence. J. Physiol. 2001, 95, 3–9. [Google Scholar] [CrossRef]
- Wijesinghe, W.A.J.P.; Jeon, Y.-J. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Sun, H.; Mao, W.; Fang, F.; Li, H. Polysaccharides from marine green seaweed Ulva species and their characteristics. Agro Food Ind. Hi Tech 2007, 18, 28–29. [Google Scholar]
- Carneiro, J.G.; Rodrigues, J.A.G.; Teles, F.B.; Cavalcante, A.B.D.; Benevides, N.M.B. Analysis of some chemical nutrients in four Brazilian tropical seaweeds. Acta Sci. Biol. Sci. 2014, 36, 137–145. [Google Scholar] [CrossRef]
- Wijesinghe, W.A.J.P.; Jeon, Y.-J. Biological activities and potential cosmeceutical applications of bioactive components from brown seaweeds: A review. Phytochem. Rev. 2011, 10, 431–443. [Google Scholar] [CrossRef]
- Rodrigues, J.A.G.; Vanderlei, E.D.S.O.; Quinderé, A.L.G.; Fontes, B.P.; Queiroz, I.N.L.de.; Benevides, N.M.B. Extraction and anticoagulant activity of sulfated polysaccharides from Caulerpa cupressoides var. lycopodium (Vahl) C. Agardh (Chlorophyceae). Acta Sci. BioLog. Sci. 2011, 33, 133–140. [Google Scholar]
- Quinderé, A.L.G.; Santos, G.R.C.; Oliveira, S.N.M.C.G.; Glauser, B.F.; Fontes, B.P.; Queiroz, I.N.L.; Benevides, N.M.; Pomin, V.H.; Mourão, P.A. Is the antithrombotic effect of sulfated galactans independent of serpin? J. Thromb. Haemost. 2014, 12, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.B.; Frota, A.F.; Sousa, R.S.; Cezario, N.A.; Santos, T.B.; Souza, L.M.F.; Coura, C.O.; Monteiro, V.S.; Cristino Filho, G.; Vasconcelos, S.M.; et al. Neuroprotective effects of sulphated agaran from marine alga gracilaria cornea in rat 6-hydroxydopamine parkinson’s disease model: Behavioural, neurochemical and transcriptional alterations. Basic Clin. Pharmacol. Toxicol. 2017, 120, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Coura, C.O.; de Araújo, I.W.F.; Vanderlei, E.S.O.; Rodrigues, J.A.G.; Quinderé, A.L.G.; Fontes, B.P.; de Queiroz, I.N.L.; de Menezes, D.B.; Bezerra, M.M.; e Silva, A.A.R.; et al. Antinociceptive and anti-inflammatory activities of sulphated polysaccharides from the red seaweed gracilaria cornea. Basic Clin. Pharmacol. Toxicol. 2011, 110, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.A.G.; Vanderlei, E.S.; Silva, L.M.; de Araújo, I.W.F.; de Queiroz, I.N.L.; de Paula, G.A.; Abreu, T.M.; Ribeiro, N.A.; Bezerra, M.M.; Chaves, H.V.; et al. Antinociceptive and anti-inflammatory activities of a sulfated polysaccharide isolated from the green seaweed Caulerpa cupressoides. Pharmacol. Rep. 2012, 64, 282–292. [Google Scholar] [CrossRef]
- Ribeiro, N.A.; Abreu, T.M.; Chaves, H.V.; Bezerra, M.M.; Monteiro, H.S.A.; Jorge, R.J.B.; Benevides, N.M.B. Sulfated polysaccharides isolated from the green seaweed Caulerpa racemosa plays antinociceptive and anti-inflammatory activities in a way dependent on HO-1 pathway activation. Inflamm. Res. 2014, 63, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, J.G.; Rodrigues, J.A.G.; de Sousa Oliveira Vanderlei, E.; Souza, R.B.; Quinderé, A.L.G.; Coura, C.O.; de Araújo, I.W.F.; Chaves, H.V.; Bezerra, M.M.; Benevides, N.M.B. Peripheral antinociception and anti-inflammatory effects of sulphated polysaccharides from the alga caulerpa mexicana. Basic Clin. Pharmacol. Toxicol. 2014, 115, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Bitencourt, M.A.O.; Silva, H.M.D.; Abílio, G.M.F.; Miranda, G.E.C.; Moura, A.M.A.; de Araújo-Júnior, J.X.; Silveira, E.J.D.; Santos, B.V.O.; Souto, J.T. Anti-inflammatory effects of methanolic extract of green algae Caulerpa mexicana in a murine model of ulcerative colitis. Rev. Bras. Farm. 2015, 25, 677–682. [Google Scholar] [CrossRef]
- Gomes, A.S.; Gadelha, G.G.; Lima, S.J.; Garcia, J.A.; Medeiros, J.V.R.; Havt, A.; Lima, A.A.; Ribeiro, R.A.; Brito, G.A.C.; Cunha, F.Q.; et al. Gastroprotective effect of heme-oxygenase 1/biliverdin/CO pathway in ethanol-induced gastric damage in mice. Eur. J. Pharmacol. 2010, 642, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Barrachina, M.D.; Panés, J.; Esplugues, J.V. Role of nitric oxide in gastrointestinal inflammatory and ulcerative diseases: Perspective for drugs development. Curr. Pharm. Des. 2001, 7, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Tarnawski, A.; Ahluwalia, A.K.; Jones, M. Gastric cytoprotection beyond prostaglandins: Cellular and molecular mechanisms of gastroprotective and ulcer healing actions of antacids. Curr. Pharm. Des. 2012, 19, 126–132. [Google Scholar]
- Kwiecien, S.; Jasnos, K.; Magierowski, M.; Sliwowski, Z.; Pajdo, R.; Brzozowski, B.; Mach, T.; Wojcik, D.; Brzozowski, T. Lipid peroxidation, reactive oxygen species and antioxidative factors in the pathogenesis of gastric mucosal lesions and mechanism of protection against oxidative stress-induced gastric injury. J. Physiol. Pharmacol. 2014, 65, 613–622. [Google Scholar] [PubMed]
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A sustainable functional food for complementary and alternative therapy. Trends Food Sci. Technol. 2012, 23, 83–96. [Google Scholar] [CrossRef]
- Damasceno, S.R.B.; Rodrigues, J.C.; Silva, R.O.; Nicolau, L.A.D.; Chaves, L.S.; Freitas, A.L.P.; Souza, M.H.L.P.; Barbosa, A.L.R.; Medeiros, J.-V.R. Role of the NO/KATP pathway in the protective effect of a sulfated-polysaccharide fraction from the algae Hypnea musciformis against ethanol-induced gastric damage in mice. Rev. Bras. Farm. 2013, 23, 320–328. [Google Scholar] [CrossRef]
- Silva, R.O.; Santos, G.M.P.D.; Nicolau, L.A.D.; Lucetti, L.T.; Santana, A.P.M.; Chaves, L.D.S.; Barros, F.C.N.; Freitas, A.L.P.; Medeiros, J. V. R. Sulfated-Polysaccharide fraction from red algae gracilaria caudata protects mice gut against ethanol-induced damage. Mar Drugs. 2011, 9, 2188–2200. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.-J.; Kwon, M.-J.; Kim, I.-H.; Nam, T.-J. The effect of polysaccharide extracted from the marine alga capsosiphon fulvescens on ethanol administration. Food Chem. Toxicol. 2008, 46, 2653–2657. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.-Y.; Hwang, H.-J.; Kim, I.-H.; Nam, T.-J. Protective effects of a polysaccharide from Hizikia fusiformis against ethanol toxicity in rats. Food Chem. Toxicol. 2009, 47, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Hase, S.; Takeeda, M.; Nakashima, M.; Yokota, A. Prostaglandin EP receptor subtypes and gastric cytoprotection. InflammoPharmacology 2002, 10, 303–312. [Google Scholar] [CrossRef]
- Brzozowski, T.; Konturek, P.C.; Konturek, S.J.; Brzozowska, I.; Pawlik, T. Role of prostaglandins in gastroprotection and gastric adaptation. J. Physiol. Pharmacol. 2005, 56, 33–55. [Google Scholar] [PubMed]
- Wiley, J.W. The many faces of nitric oxide: Cytotoxic, cytoprotective or both. Neurogastroenterol. Motil. 2007, 19, 541–544. [Google Scholar] [CrossRef] [PubMed]
- Magierowska, K.; Magierowski, M.; Surmiak, M.; Adamski, J.; Mazur-Bialy, A.; Pajdo, R.; Sliwowski, Z.; Kwiecien, S.; Brzozowski, T. The protective role of carbon monoxide (CO) produced by heme oxygenases and derived from the CO-releasing molecule CORM-2 in the pathogenesis of stress-induced gastric lesions: Evidence for non-involvement of nitric oxide (NO). Int. J. Mol. Sci. 2016, 17, 442. [Google Scholar] [CrossRef] [PubMed]
- Magierowska, K.; Magierowski, M.; Hubalewska-Mazgaj, M.; Adamski, J.; Surmiak, M.; Sliwowski, Z.; Kwiecien, S.; Brzozowski, T. Carbon monoxide (CO) released from tricarbonyldichlororuthenium (II) dimer (CORM-2) in gastroprotection against experimental ethanol-induced gastric damage. PLoS ONE 2015, 10, e0140493. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Muñoz, R.; Montiel-Ruíz, C.; Vázquez-Martínez, O. Gastric mucosal cell proliferation in ethanol-induced chronic mucosal injury is related to oxidative stress and lipid peroxidation in rats. Lab. Investig. 2000, 80, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Kalayci, M.; Kocdor, M.A.; Kuloglu, T.; Sahin, I.; Sarac, M.; Aksoy, A.; Yardim, M.; Dalkilic, S.; Gursu, O.; Aydin, S.; et al. Comparison of the therapeutic effects of sildenafil citrate, heparin and neuropeptides in a rat model of acetic acid-induced gastric ulcer. Life Sci. 2017, 186, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Brzozowski, T.; Magierowska, K.; Magierowski, M.; Ptak-Belowska, A.; Pajdo, R.; Kwiecien, S.; Olszanecki, R.; Korbut, R. Recent advances in the gastric mucosal protection against stress-induced gastric lesions. Importance of renin-angiotensin vasoactive metabolites, gaseous mediators and appetite peptides. Curr. Pharm. Des. 2017, 23, 1. [Google Scholar]
- Farias, W.R.L.; Valente, A.-P.; Pereira, M.S.; Mourao, P.A.S. Structure and anticoagulant activity of sulfated galactans: Isolation of a unique sulfated galactan from the red algaebotryocladia occidentalis and comparison of its anticoagulant action with that of sulfated galactans from invertebrates. J. BioLog. Chem. 2000, 275, 29299–29307. [Google Scholar] [CrossRef] [PubMed]
- Laine, L.; Weinstein, W.M. Histology of alcoholic hemorrhagic gastritis: A prospective evaluation. Gastroenterology 1988, 94, 1254–1262. [Google Scholar] [CrossRef]
- Sedlak, J.; Lindsay, R.H. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid Peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar] [PubMed]
- Green, L.C.; Tannenbaum, S.R.; Goldman, P. Nitrate synthesis in the germfree and conventional rat. Science 1981, 212, 56. [Google Scholar] [CrossRef] [PubMed]
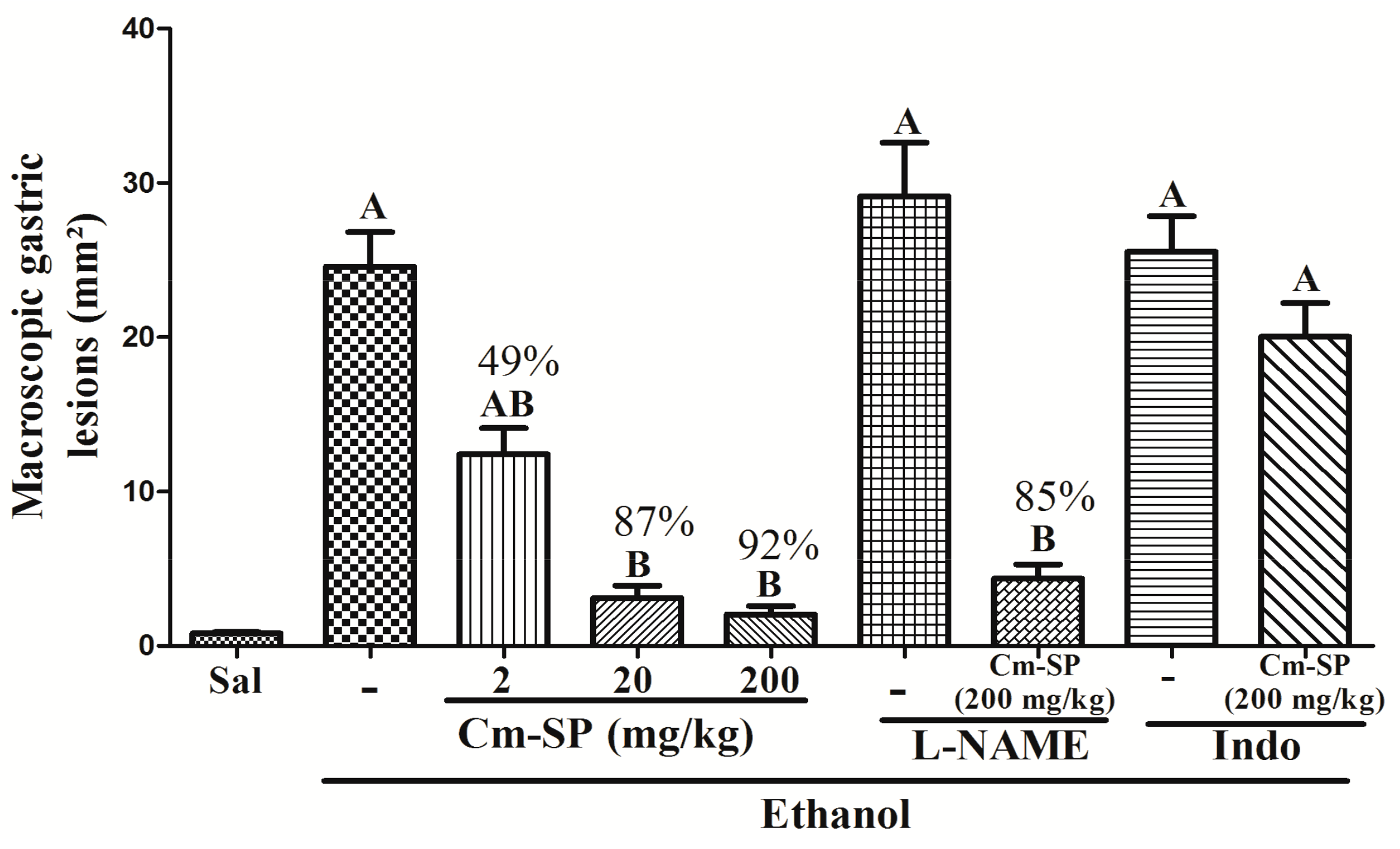
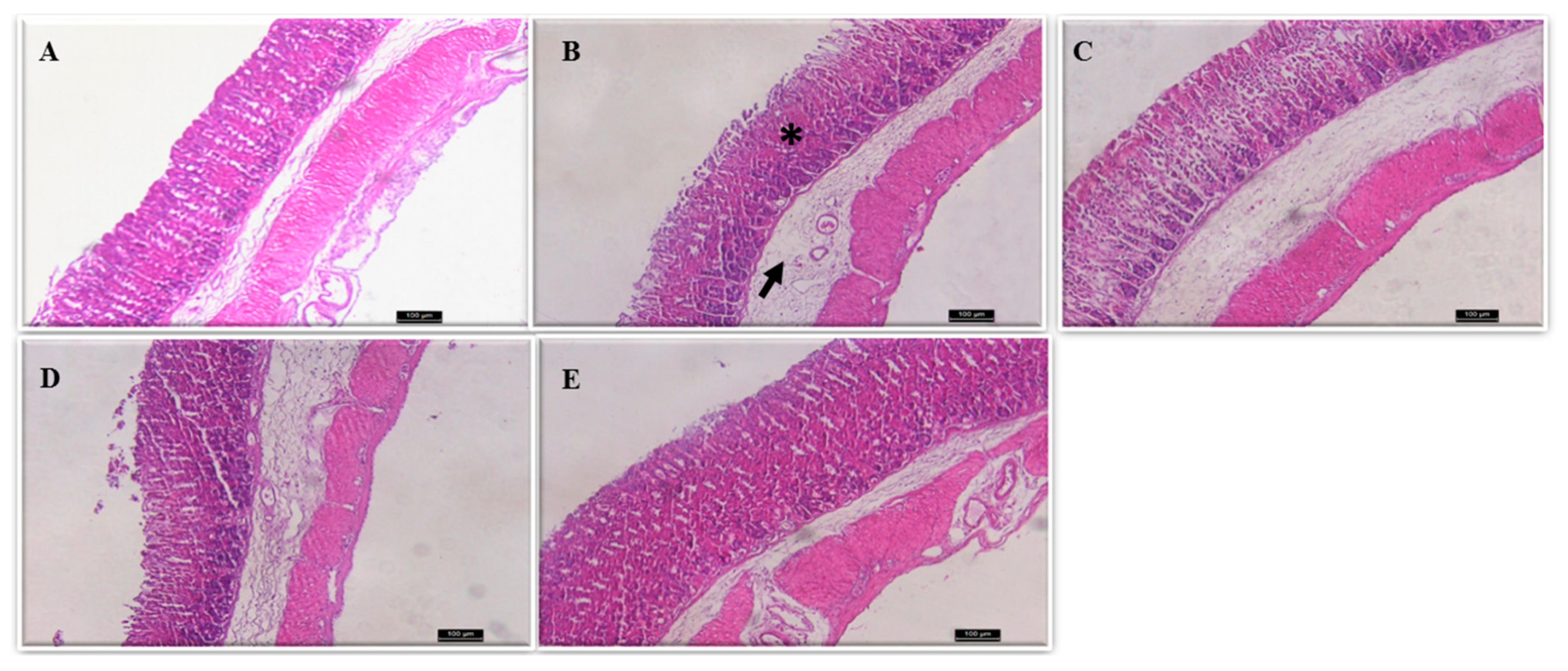
| Experimental Group (N = 6) | Hemorrhagic Lesion (Score 0–4) | Oedema (Score 0–4) | Erosion (Loss of Cell Architecture) (Score 0–3) | Cell Infiltrate (Score 0–3) | Total (Scores 14) |
|---|---|---|---|---|---|
| Saline | 0 | 0 | 0 | 0 | 0 |
| Ethanol | 4 (3–4) A | 2 (2–4) A | 2.5 (2–3) A | 2 (2–3) A | 10.5 (9–14) A |
| Cm-SP (2 mg/kg) | 1 (0–1) B | 2 (0–3) | 1 (0–2) | 0 (0–1) B | 4 (0–7) |
| Cm-SP (20 mg/kg) | 0 (0–1) B | 0 (0–3) | 0 (0–3) | 0 (0–2) | 0 (0–9) |
| Cm-SP (200 kg/mg) | 0 (0–0) B | 0 (0–2) B | 0 (0–1) B | 0 (0–2) | 0 (0–5) B |
| Experimental Groups | MDA | GSH | NO2/NO3 |
|---|---|---|---|
| Saline | 152.30 ± 9.91 | 31.81 ± 8.48 | 16.05 ± 3.24 |
| Ethanol | 391.42 ± 23.06 A | 20.96 ± 1.87 A | 28.02 ± 2.74 A |
| Cm-SP (200 mg/kg) | 153.30 ± 8.28 B | 38.26 ± 0.57 B | 23.14 ± 3.52 |
| L-NAME | 541.60 ± 49.02 | 19.28 ± 3.74 | 7.52 ± 1.68 |
| L-NAME + Cm-SP (200 mg/kg) | 383.0 ± 23.97 C | 32.35 ± 1.56 C | 8.66 ± 1.62 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carneiro, J.G.; Holanda, T.D.B.L.; Quinderé, A.L.G.; Frota, A.F.; Soares, V.V.M.; Sousa, R.S.d.; Carneiro, M.A.; Martins, D.S.; Gomes Duarte, A.S.; Benevides, N.M.B. Gastroprotective Effects of Sulphated Polysaccharides from the Alga Caulerpa mexicana Reducing Ethanol-Induced Gastric Damage. Pharmaceuticals 2018, 11, 6. https://doi.org/10.3390/ph11010006
Carneiro JG, Holanda TDBL, Quinderé ALG, Frota AF, Soares VVM, Sousa RSd, Carneiro MA, Martins DS, Gomes Duarte AS, Benevides NMB. Gastroprotective Effects of Sulphated Polysaccharides from the Alga Caulerpa mexicana Reducing Ethanol-Induced Gastric Damage. Pharmaceuticals. 2018; 11(1):6. https://doi.org/10.3390/ph11010006
Chicago/Turabian StyleCarneiro, José Gerardo, Ticiana De Brito Lima Holanda, Ana Luíza Gomes Quinderé, Annyta Fernandes Frota, Vitória Virgínia Magalhães Soares, Rayane Siqueira de Sousa, Manuela Araújo Carneiro, Dainesy Santos Martins, Antoniella Souza Gomes Duarte, and Norma Maria Barros Benevides. 2018. "Gastroprotective Effects of Sulphated Polysaccharides from the Alga Caulerpa mexicana Reducing Ethanol-Induced Gastric Damage" Pharmaceuticals 11, no. 1: 6. https://doi.org/10.3390/ph11010006
APA StyleCarneiro, J. G., Holanda, T. D. B. L., Quinderé, A. L. G., Frota, A. F., Soares, V. V. M., Sousa, R. S. d., Carneiro, M. A., Martins, D. S., Gomes Duarte, A. S., & Benevides, N. M. B. (2018). Gastroprotective Effects of Sulphated Polysaccharides from the Alga Caulerpa mexicana Reducing Ethanol-Induced Gastric Damage. Pharmaceuticals, 11(1), 6. https://doi.org/10.3390/ph11010006




