Drug Repurposing for Schistosomiasis: Combinations of Drugs or Biomolecules
Abstract
1. Introduction
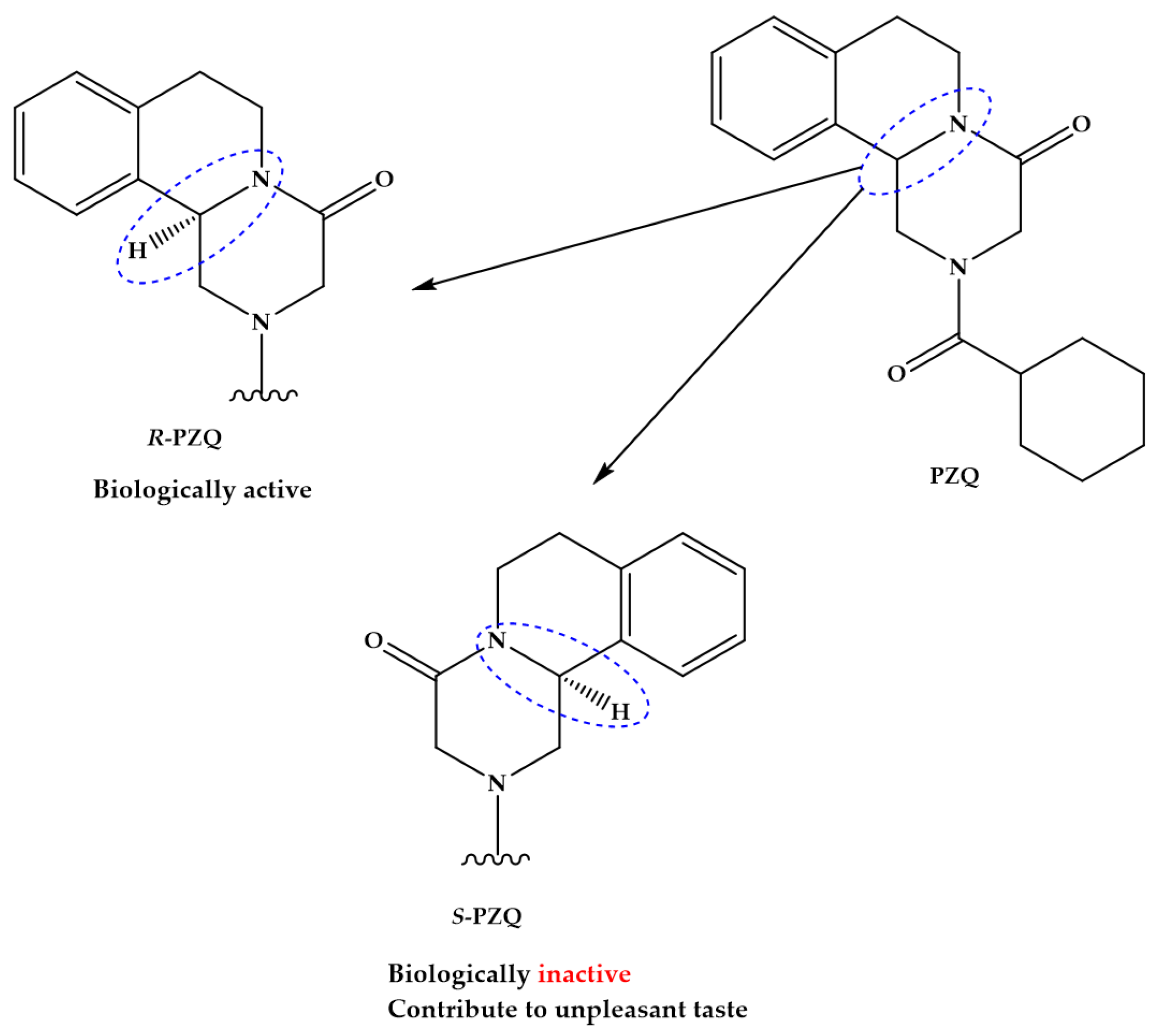
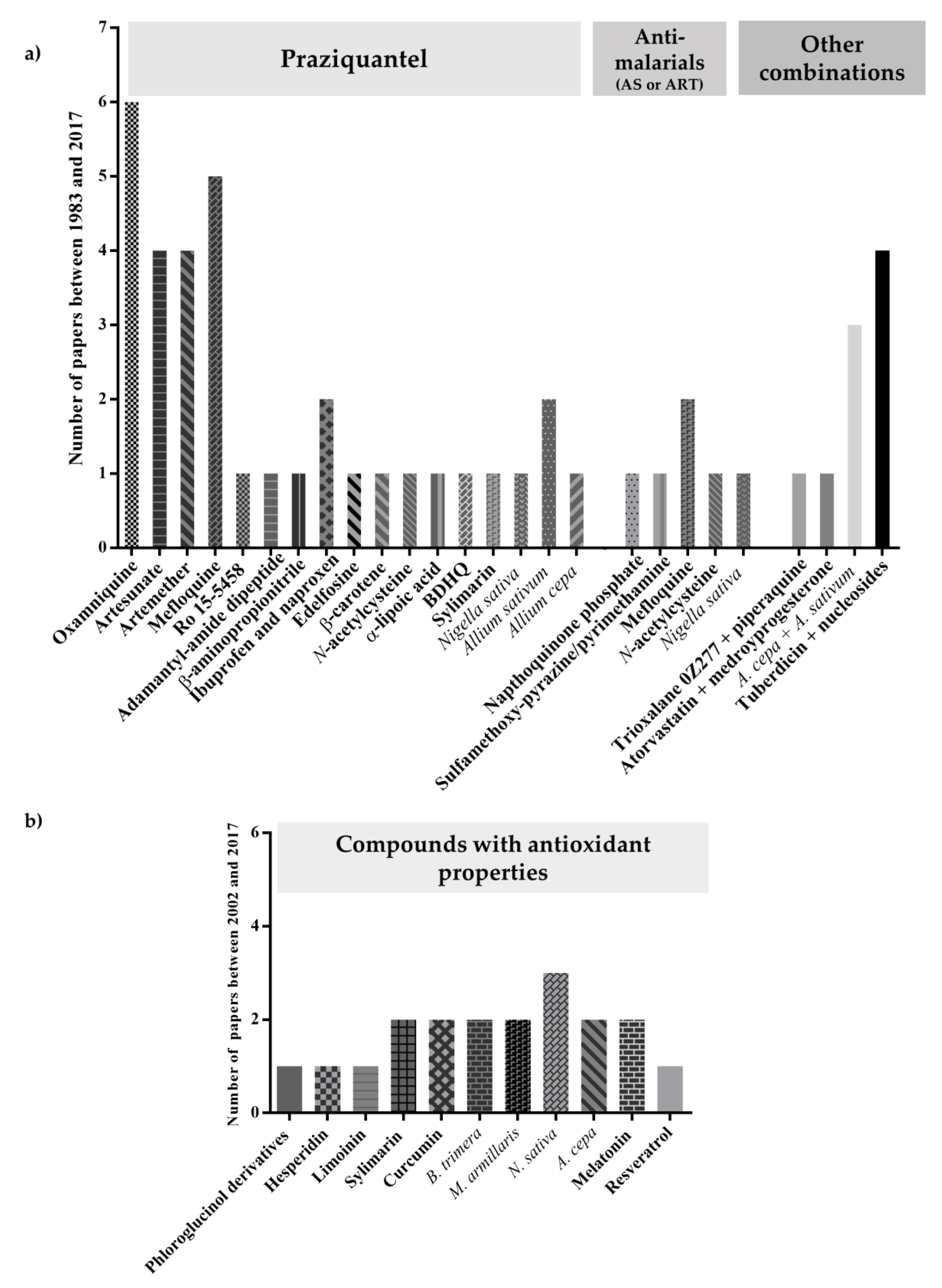
2. Praziquantel: Mainstay Chemotherapy against Schistosomiasis
3. Treatment of Schistosomiasis: Anthelminthic Drugs Alone or Combined
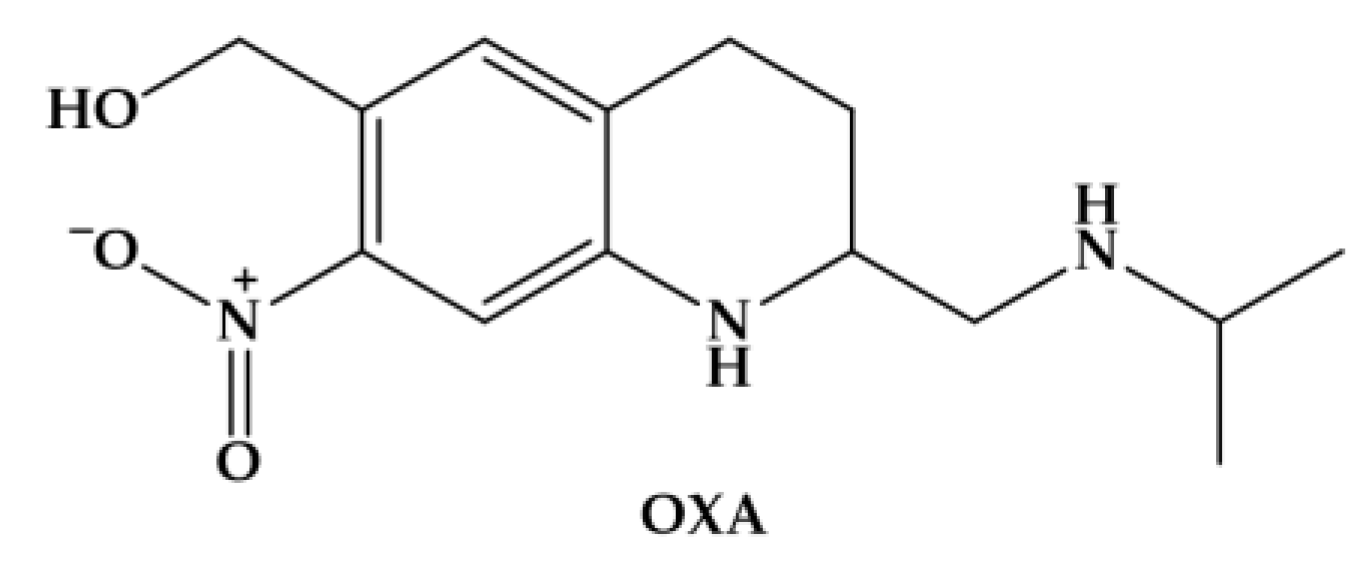
3.1. Oxamniquine
3.2. Antimalarials
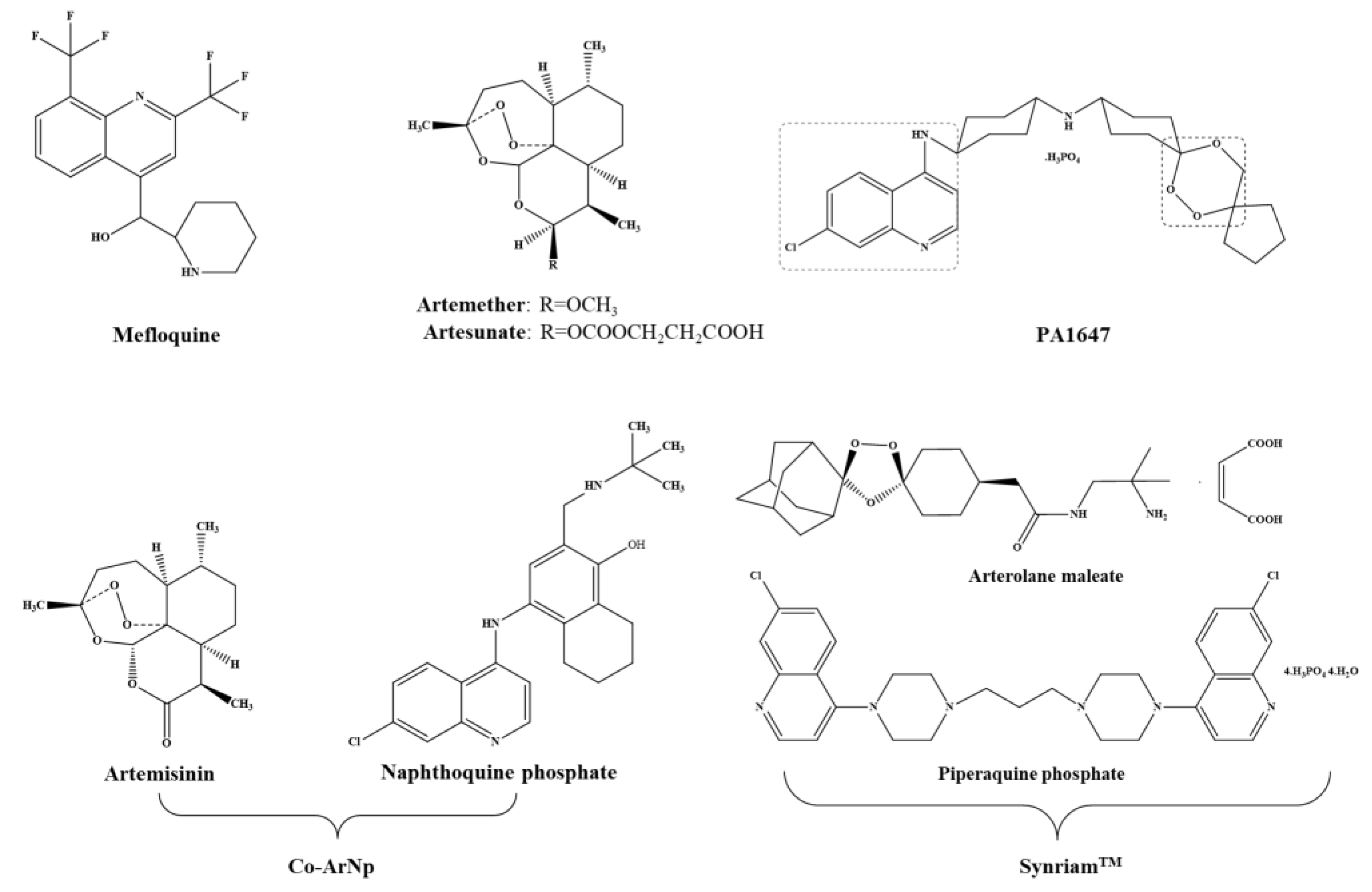
3.2.1. Artemisinins Derivatives
Arthemeter
Artesunate
Artesunate Combined with Sulfamethoxypyrazine/Pyrimethamine
Artemisinin and Naphthoquine Phosphate
3.2.2. Mefloquine
3.2.3. Trioxalanes/Trioxaquines
Synriam™
PA1647
3.3. Other Combinations
3.3.1. Nucleosides
3.3.2. Acridine Derivatives
3.3.3. Anti-Inflammatory Agents
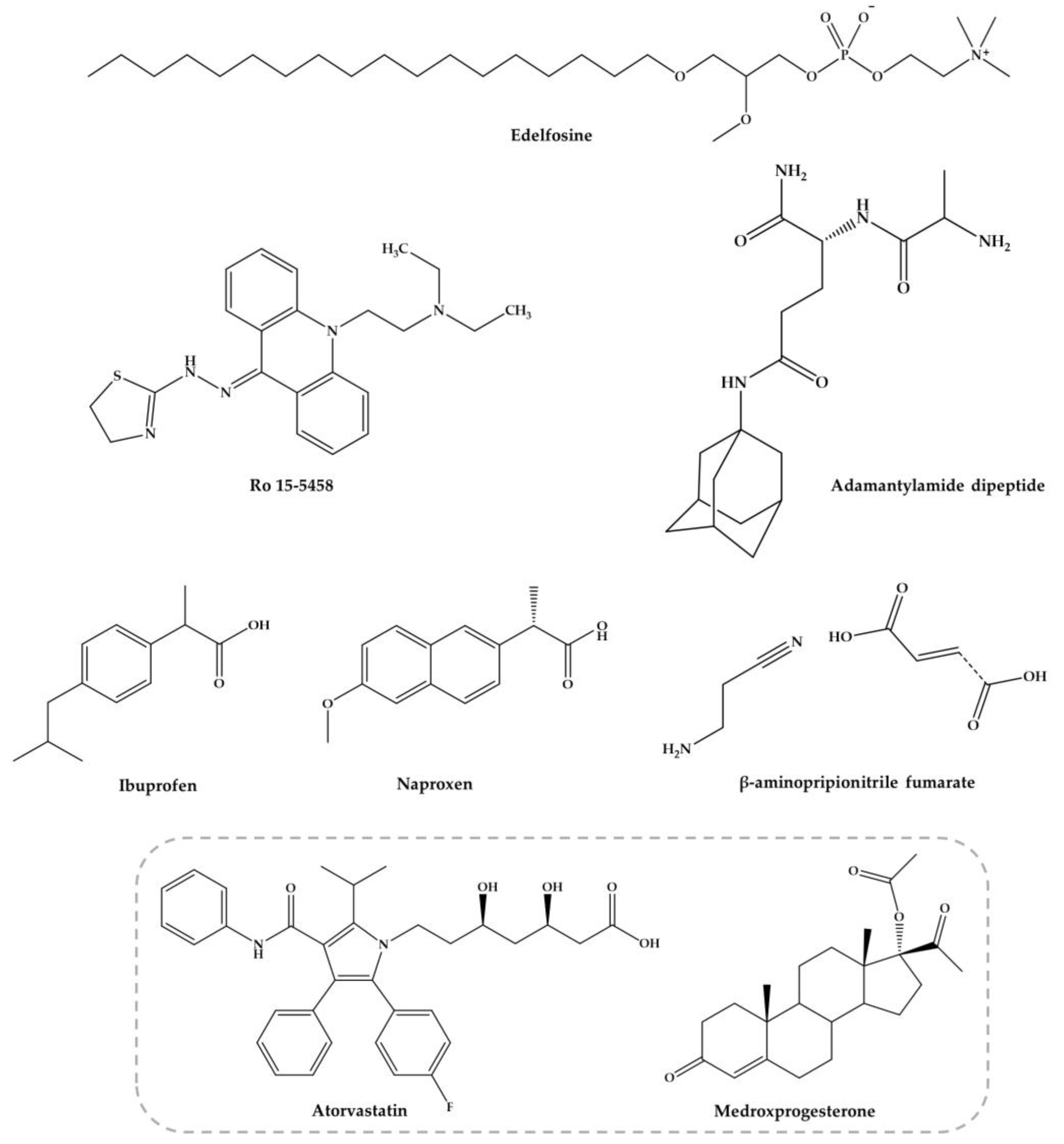
3.3.4. Edelfosine
3.3.5. Antifibrotic Agent, β-Aminopropionitrile
3.3.6. Adamatylamide Dipeptide
3.3.7. Atorvastatin and Medroxyprogesterone Acetate
4. Antioxidants: A New Chemotherapy against Schistosomiasis?
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- GDB 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef]
- Hotez, P.J.; Alvarado, M.; Basáñez, M.-G.; Bollinger, I.; Bourne, I.; Boussinesq, M.; Brooker, S.J.; Brown, A.S.; Buckle, G.; Budke, C.M.; et al. The global burden of disease study 2010: Interpretation and implications for the neglected tropical diseases. PLoS Negl. Trop. Dis. 2014, 8, e2865. [Google Scholar] [CrossRef] [PubMed]
- Steinmann, P.; Keiser, J.; Bos, R.; Tanner, M.; Utzinger, J. Schistosomiasis and water-resources development: Systematic review, meta-analysis and estimates of people at risk. Lancet Infect. Dis. 2006, 6, 411–425. [Google Scholar] [CrossRef]
- King, C.H. Parasites and poverty: The case of schistosomiasis. Acta Trop. 2010, 113, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Gray, D.J.; McManus, D.P.; Li, Y.; Williams, G.M.; Bergquist, R.; Ross, A.G. Schistosomiasis elimination: Lessons from the past guide the future. Lancet Infect. Dis. 2010, 10, 733–736. [Google Scholar] [CrossRef]
- Schistosomiasis: Number of people treated worldwide in 2013. Wkly. Epidemiol. Rec. 2015, 90, 25–32.
- Schistosomiasis: Number of people treated worldwide in 2015. Wkly. Epidemiol. Rec. 2016, 91, 585–600.
- Ross, A.G.P.; Bartley, P.B.; Sleigh, A.C.; Olds, R.; Li, Y.; Williams, G.M.; McManus, P. Schistosomiasis. N. Engl. J. Med. 2002, 346, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Brindley, P.J.; Correia da Costa, J.M.; Sripa, B. Why does helminths cause cancer? Trends Cancer 2015, 1, 174–182. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research On Cancer. A review of human carcinogens. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization/International Agency for Research on Cancer: Geneve, Switzerland, 2012; Volume 100(B), pp. 1–441. [Google Scholar]
- Holtfreter, M.C.; Moné, H.; Müller-Stöver, I.; Mouhadi, G.; Ritcher, J. Schistosoma haematobium infections acquired in Corsica, France, August 2013. Euro Surveill. 2014, 19, 20821. [Google Scholar] [CrossRef] [PubMed]
- Barsoum, R.S.; Esmat, G.; El-Baz, T. Human schistosomiasis: Clinical perspective: Review. J. Adv. Res. 2013, 4, 433–444. [Google Scholar] [CrossRef] [PubMed]
- King, C.H. Schistosomiasis: Challenge and opportunities. In Institute of Medicine (US) Forum on Microbial Threaths. The Causes and Impacts of Neglected Tropical and Zoonotic Diseases: Opportunities for Integrated Intervention Strategies; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Utzinger, J.; Keiser, J.; Shuhua, X.; Tanner, M.; Singer, B.H. Combination chemotherapy of schistosomiasis in laboratory studies and clinical trials. Antimicrob. Agent. Chemother. 2003, 47, 1487–1495. [Google Scholar] [CrossRef]
- World Health Organization. Prevention and Control of Schistosomiasis and Soil-Transmitted Helminthiasis: Report of a WHO Expert Committee; WHO Technical Report Series No. 912; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Beaumier, C.M.; Gillespie, P.M.; Hotez, P.J.; Bottazzi, M.E. New vaccines for neglected parasitic diseases and dengue. Transl. Res. 2013, 162, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Cioli, D.; Pica-Mattocia, L.; Basso, A.; Guidi, A. Schistosomiasis control: Praziquantel forever? Mol. Biochem. Parasitol. 2014, 195, 23–29. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Sustaining the drive to overcome the global impact of neglected tropical diseases. In Second WHO Report on Neglected Tropical Diseases; Crompton, D.W.T., Ed.; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- Olliaro, P.; Delgado-Romero, P.; Keiser, J. The little we know about the pharmacokinetics and pharmacodynamics of praziquantel (racemate and R-enantiomer). J. Antimicrob. Chemother. 2014, 69, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, C.R. Chemotherapy of schistosomiasis: Present and future. Curr. Opin. Chem. Biol. 2007, 11, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Nalugwa, A.; Nuwaha, F.; Tukahebwa, E.M.; Olsen, A. Single versus double dose praziquantel comparison on efficacy and Schistosoma mansoni re-infection in preschool-age children in Uganda: A randomized controlled trial. PLoS Negl. Trop. Dis. 2015, 9, e0003796. [Google Scholar] [CrossRef] [PubMed]
- Meyer, T.; Sekljic, H.; Fuchs, S.; Bothe, H.; Schollmeyer, D.; Miculka, C. Taste, a new incentive to switch to (R)-Praziquantel in schistosomiasis treatment. PLoS Negl. Trop. Dis. 2009, 3, e357. [Google Scholar] [CrossRef] [PubMed]
- Andrews, P. Praziquantel: Mechanisms of antischistosomal activity. Pharmacol. Therapeut. 1985, 29, 129–156. [Google Scholar] [CrossRef]
- Xiao, S.; Catto, B.A.; Webster, L.T. Effects of praziquantel on different developmental stages of Schistosoma mansoni. J. Infect. Dis. 1985, 151, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Valle, C.; Troiani, A.R.; Festucci, A.; Pica-Mattocia, L.; Liberti, P.; Wolstenholme, A.; Franclow, K.; Doenhoff, M.J.; Cioli, D. Sequence and level of endogenous expression of calcium channel beta subunits in Schistosoma mansoni displaying different susceptibilities to praziquantel. Mol. Biochem. Parasitol. 2003, 130, 111–115. [Google Scholar] [CrossRef]
- Angelucci, F.; Basso, A.; Belielli, A.; Brunori, M.; Pica-Mattocia, L.; Valle, C. The anti-schistosomal drug praziquantel is an adenosine antagonist. Parasitology 2007, 134, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Tallina, H.; El Ridi, R. Praziquantel binds Schistosoma mansoni adult worm actin. Int. J. Antimicrob. Agents 2007, 29, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Troiani, A.R.; Pica-Mattocia, L.; Valle, C.; Cioli, D.; Mignogna, G.; Ronketti, F.; Todd, M. Is actin a praziquantel receptor? Int. J. Antimicrob. Agents 2007, 30, 280–281. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.D.; Cupit, P.M.; Gunaratne, G.S.; McCorvy, J.D.; Yang, Y.; Stoltz, K.; Webb, T.R.; Dosa, P.I.; Roth, B.L.; Abagyan, R.; Cunningham, C.; Marchant, J.S. The anthelmintic praziquantel is a human serotoninergic G-protein-coupled receptor ligand. Nat. Commun. 2017, 8, 1910. [Google Scholar] [CrossRef] [PubMed]
- Vale, N.; Gouveia, M.J.; Rinaldi, G.; Brindley, P.J.; Gärtner, F.; Correia da Costa, J.M. Praziquantel for schistosomiasis: Single-drug metabolism revisited, mode of action, and resistance. Antimicrob. Agents Chemother. 2017, 61, e02582-16. [Google Scholar] [CrossRef] [PubMed]
- Fallon, P.G. Schistosome resistance to praziquantel. Drug Resist. Updat. 1998, 1, 236–241. [Google Scholar] [CrossRef]
- Wang, W.; Wang, L.; Liang, Y. Susceptibility or resistance of praziquantel in human schistosome: A review. Parasitol. Res. 2012, 111, 1871–1877. [Google Scholar] [CrossRef] [PubMed]
- Cioli, D. Praziquantel: Is there any real resistance and are there alternatives? Curr. Opin. Infect. Dis. 2000, 13, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Brindley, P.J.; Hotez, P.J. Break out: Urogenital schistosomiasis and Schistosoma haematobium infection in the post-genomic era. PLoS Negl. Trop. Dis. 2013, 7, e1961. [Google Scholar] [CrossRef] [PubMed]
- Trainor-Moss, S.; Mutapi, F. Schistosomiasis therapeutics: Whats in the pipeline? Expert Rev. Clin. Pharmacol. 2016, 9, 157–160. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Merrifield, M.; Hotez, P.J.; Beaumier, C.M.; Gillspie, P.; Strych, U.; Hayward, T.; Bottazzi, M.E. Advancing a vaccine to prevent human schistosomiasis. Vaccine 2016, 34, 2988–2991. [Google Scholar] [CrossRef] [PubMed]
- Doenhoff, M.J.; Cioli, D.; Utzinger, J. Praziquantel: Mechanism of action, resistance and new derivatives for schistosomiasis. Curr. Opin. Infect. Dis. 2008, 21, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; William, S.; Herdtweck, E.; Botros, S.; Dömling, A. MCR synthesis of praziquantel derivatives. Chem. Biol. Drug Des. 2012, 79, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Kerantzas, C.A.; Jacobs, W.R., Jr. Origins of combination therapy for tuberculosis: Lessons for future antimicrobial development and application. mBio 2017, 8, e01686-16. [Google Scholar] [CrossRef] [PubMed]
- Maenza, J.; Flexner, C. Combination antiretroviral therapy for HIV infection. Am. Fam. Physician 1998, 57, 2789–2798. [Google Scholar] [PubMed]
- Wells, T.N.C.; van Huijsduijnen, R.H.; van Voortris, W.C. Malaria medicines: A glass half full? Nat. Rev. Drug Dis. 2015, 14, 424–442. [Google Scholar] [CrossRef] [PubMed]
- Richards, H.C.; Foster, R. A new series of 2-aminomethyltetrahydroquinoline derivatives displaying schistosomicidal activity in rodents and primates. Nature 1969, 222, 581–582. [Google Scholar] [CrossRef] [PubMed]
- Fewick, A.; Savioli, L.; Engles, D.; Robert Bergquist, N.; Todd, M.H. Drugs for the control of parasitic diseases: Current status and development in schistosomiasis. Trends Parasitol. 2003, 19, 509–515. [Google Scholar] [CrossRef]
- Foster, R.; Cheetam, R.L.; King, D.F. The action of UK 3883, a novel 2-aminomethyltetrahydroquinoline derivative, against mature schistosomes in rodents and primates. Ann. Trop. Med. Parasitol. 1971, 65, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Pica-Mattoccia, L.; Carlini, D.; Guidi, A.; Cimica, V.; Vigorosi, F.; Cioli, D. The schistosome enzyme that activates oxamniquine has the characteristics of a sulfotransferase. Mem. Inst. Oswaldo Cruz 2006, 101 (Suppl. 1), 307–312. [Google Scholar] [CrossRef] [PubMed]
- Valentim, C.L.L.; Cioli, D.; Chevalier, F.D.; Cao, X.; Taylor, A.B.; Holloway, S.P.; Pica-Mattocia, L.; Guidi, A.; Basso, A.; Tsai, I.J.; et al. Genetic and molecular basis of drug resistance and species-specific drug action in schistosome parasites. Science 2013, 342, 1385–1389. [Google Scholar] [CrossRef] [PubMed]
- Foster, R. A review of clinical experience with oxamniquine. Trans. R. Soc. Trop. Med. Hyg. 1987, 89, 55–59. [Google Scholar] [CrossRef]
- Abdul-Ghani, R.; Loufty, N.; el-Sahn, A.; Hassan, A. Current chemotherapy arsenal for schistosomiasis mansoni: Alternatives and challenges. Parasitol. Res. 2009, 104, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.R.; Brammer, K.W. The treatment of experimental schistosomiasis with a combination of oxamniquine and praziquantel. Trans. R. Soc. Med. Trop. Hyg. 1983, 77, 39–40. [Google Scholar] [CrossRef]
- Botros, S.; Soliman, A.; El-Gawhary, N. Selim, N.; Guirguis, N. Effect of combined low dose of praziquantel and oxamniquine on different stages of schistosome maturity. Trans. R. Soc. Trop. Med. Hyg. 1989, 83, 86–89. [Google Scholar] [CrossRef]
- Delgado, V.S.; Suárez, D.P.; Cesar, I.M.; Incani, R.N. Experimental chemotherapy of Schistosoma mansoni with praziquantel and oxamniquine: Differential effect on single of combined formulations of drugs on various strains and on both sexes of the parasite. Parasitol. Res. 1992, 78, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Pugh, R.N.; Teesdale, C.H. Synergy of concurrent low dose of oxamniquine and praziquantel in schistosomiasis. Br. Med. J. 1983, 287, 877–878. [Google Scholar] [CrossRef][Green Version]
- Creasey, A.M.; Taylor, P.; Thomas, J.E. Dosage trial of a combination of oxamniquine and praziquantel in the treatment of schistosomiasis in Zimbabwean schoolchildren. Cent. Afr. J. Med. 1986, 32, 165–167. [Google Scholar] [PubMed]
- Utzinger, J.; Xiao, S.H.; N’Goran, E.K.; Bergquist, R.; Tanner, M. The potential of artemether for the control of schistosomiasis. Int. J. Parasitol. 2001, 31, 1549–1562. [Google Scholar] [CrossRef]
- Xiao, S.H.; Hotez, P.J.; Tanner, T. Artemether, an effective new agent for chemoprophylaxis against schistosomiasis in China: Its in vivo effect on biochemical metabolism of the Asian schistosome. Southeast. Asian J. Trop. Med. Public Health 2000, 31, 724–732. [Google Scholar]
- El-Lakkany, N.M.; el-Din, S.H. Hemin enhances the in vivo efficacy of artemether against juvenile and adult S. mansoni in mice. Parasitol. Res. 2013, 112, 2005–2015. [Google Scholar] [CrossRef] [PubMed]
- Utzinger, J.; Xiao, S.H.; Tanner, M.; Keiser, J. Artemisinins for schistosomiasis and beyond. Curr. Opin. Investig. Drugs 2007, 8, 105–116. [Google Scholar] [PubMed]
- Shuhua, X.; Chollet, J.; Weiss, N.A.; Bergquist, R.N.; Tanner, M. Preventive effect of artemether in experimental animals infected with Schistosoma mansoni. Parasitol. Int. 2000, 49, 19–24. [Google Scholar] [CrossRef]
- Pérez del Villar, L.; Burguillo, F.J.; López-Abán, J.; Muro, A. Systematic review and meta-analysis of artemisinin based therapies for the treatment and prevention of schistosomiasis. PLoS ONE 2012, 7, e45867. [Google Scholar] [CrossRef] [PubMed]
- Shuhua, X.; Jiqing, Y.; Jinying, M.; Huifang, M.; Peiying, J.; Tanner, M. Effect of praziquantel together with artemether on Schistosoma japonicum parasites of different ages in rabbits. Parasitol. Int. 2000, 49, 25–30. [Google Scholar] [CrossRef]
- Utzinger, J.; Chollet, J.; You, J.; Mei, J.; Tanner, M.; Xiao, S. Effect of combined treatment with praziquantel and artemether on Schistosoma japonicum and Schistosoma mansoni in experimentally infected animals. Acta Trop. 2001, 80, 9–18. [Google Scholar] [CrossRef]
- Mamhoud, M.R.; Botros, S.S. Artemether as adjuvant therapy to praziquantel in murine Egyptian schistosomiasis mansoni. J. Parasitol. 2005, 91, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.-Y.; McManus, D.P.; Gray, D.J.; Balen, J.; Luo, X.S.; He, Y.K.; Ellis, M.; Williams, G.M.; Li, Y.S. A randomized, double-blind, placebo-controlled trial of safety and efficacy of combined praziquantel and artemether treatment for acute schistosomiasis japonica in China. Bull. World Health Org. 2008, 86, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Abdin, A.A.; Ashour, D.S.; Shoheib, Z.S. Artesunate effect on schistosome thioredoxin glutathione reductase and cytochrome c peroxidase as new molecular targets in Schistosoma mansoni-infected mice. Biomed. Environ. Sci. 2013, 26, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, A.N.; Davioud-Charvet, E.; Sayed, A.A.; Califf, L.L.; Dessolin, J.; Arnér, E.S.J.; Williams, D.L. Thioredoxin glutathione reductase from Schistosoma mansoni: An essential parasite enzyme and a key drug target. PLoS Med. 2007, 4, e264. [Google Scholar]
- De Clercq, D.; Vercruysse, J.; Verlé, P.; Kongs, A.; Diop, M. What is the effect of combining artesunate and praziquantel in the treatment of schistosomiasis mansoni infections? Trop. Med. Int. Health 2000, 5, 744–746. [Google Scholar] [CrossRef] [PubMed]
- Borrmann, S.; Szlekák, N.; Faucher, J.F.; Matsiegui, P.B.; Neubauer, R.; Binder, R.K.; Lell, B.; Kremsner, P.G. Artesunate and praziquantel for the treatment of Schistosoma haematobium infections: A double-blind, randomized, placebo-controlled study. J. Infect. Dis. 2001, 184, 1363–1366. [Google Scholar] [CrossRef] [PubMed]
- Inyang-Etoh, P.C.; Ejezie, G.C.; Useh, M.F.; Inyang-Etoh, E.C. Efficacy of a combination of praziquantel and artesunate in the treatment of urinary schistosomiasis in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Yunusa, E.U.; Awosan, K.J.; Ibrahim, M.T.O.; Ahmed, M.I. Comparative cure rates of artesunate-praziquantel combination and praziquantel monotherapy in the control of urinary schistosomiasis among “Almajiri” school children in Sokoto, Nigeria. Int. Res. J. Basic Clin. Stud. 2016, 5, 5–9. [Google Scholar] [CrossRef]
- Sissoko, M.S.; Dabo, A.; Traoré, H.; Diallo, M.; Traoré, B.; Konaté, D.; Niaré, B.; Diakité, M.; Kamaté, B.; Traoré, A.; et al. Efficacy of artesunate+sulfamethoxypyrazine/pyrimethamine versus praziquantel in the treatment of Schistosoma haematobium in children. PLoS ONE 2009, 4, e6732. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cao, W.; Shan, C.; Zhang, M.; Li, G.F.; Ding, D.B.; Shi, Y.L.; Wu, B.A. Naphtoquine phosphate and its combination with artemisinine. Acta Trop. 2004, 89, 375–381. [Google Scholar] [CrossRef] [PubMed]
- El-Beshishi, S.M.; Taman, A.; El-Malky, M.; Azab, M.S.; El-Hawary, A.K.; El-Tantawy, D.A. First insight into the effect of single oral dose therapy with artemsinin-naphthoquine phosphate combination in a mouse model of Schistosoma mansoni infection. Int. J. Parasitol. 2013, 43, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E.A.; White, N.J. Artemisinin-based combinations. Curr. Opin. Infect. Dis. 2005, 18, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Van Nassauw, L.; Toovey, S.; Van Op den Bosch, J.; Timmermans, J.P.; Vercruysse, J. Schistosomicidal activity of antimalarial drug mefloquine, in Schistosoma mansoni-infected mice. Travel Med. Infect. Dis. 2008, 6, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Chollet, J.; Xiao, S.H.; Mei, J-Y.; Jiao, P-Y.; Utzinger, J.; Tanner, M. Mefloquine-an aminoalcohol with promising antischistosomal properties in mice. PLoS Negl. Trop. Dis. 2009, 3, e350. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.H.; May, J.Y.; Jiao, P.Y. Further study on mefloquine concerning several aspects in experimental treatment of mice and hamsters infected with Schistosoma japonicum. Parasitol. Res. 2009, 106, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Mei, J.; Jiao, P. Effect of mefloquine administrated orally at single, multiple, or combined with artemether, artesunate, or praziquantel in treatment of mice infected with Schistosoma japonicum. Parasitol. Res. 2011, 108, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Manneck, T.; Vargas, M. Interactions of mefloquine with praziquantel in the Schistosoma mansoni mouse model and in vitro. J. Antimicrob. Chemother. 2011, 66, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- El-Lakkany, N.M.; Seif el-Din, H.; Sabra, A.A.; Hammam, O.A. Pharmacodynamics of mefloquine and praziquantel combination therapy in mice harboring juvenile and adult S. mansoni. Mem. Inst. Oswaldo Cruz 2011, 106, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Fattah, N.S.; Ahmed, N.S. Evaluation of mefloquine-praziquantel combination therapy in pre-patent and patent S. mansoni infection in mice. Sci. Parasitol. 2011, 12, 139–149. [Google Scholar]
- Keiser, J.; N’Guessan, N.A.; Adoubryn, K.D.; Silué, K.D.; Vounatsou, P.; Hatz, C.; Utzinger, J.; N’Goran, E.K. Efficacy and safety of mefloquine, artesunate, mefloquine artesunate and praziquantel against Schistosoma haematobium: Randomized, exploratory open-label trial. Clin. Infect. Dis. 2010, 50, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Silué, K.D.; Adiossan, L.K.; N’Guessan, N.A.; Monsan, N.; Utzinger, J.; N’Goran, E.K. Praziquantel, mefloquine-praziquantel, and mefloquine-artesunate-praziquantel against Schistosoma haematobium. A randomized, exploratory, open-label trial. PLoS Negl. Trop. Dis. 2014, 8, e2975. [Google Scholar] [CrossRef] [PubMed]
- Patil, C.Y.; Katare, S.S.; Baig, M.S.; Doifode, S. Fixed dose combination of arterolane and piperaquine: A newer prospect in antimalarial therapy. Ann. Med. Health Sci. 2014, 4, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Mossallam, S.F.; Amer, E.I.; El-Faham, M.H. Efficacy of Synriam™, a new antimalarial combination of OZ277 and piperaquine, against different developmental stages of Schistosoma mansoni. Acta Trop. 2015, 143, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Dechy-Cabaret, O.; Benoit-Vical, F.; Robert, A.; Meunier, B. Preparation and antimalarial activities of “Trioxaquines”, new modular molecules with a trioxane skeleton linked to a 4-aminoquinoline. ChemBioChem 2000, 1, 281–283. [Google Scholar] [CrossRef]
- Coslidan, F.; Fraisse, L.; Pellet, A.; Guillou, F.; Mordmüller, B.; Kremsner, P.G.; Moreno, A.; Mazier, D.; Maffrand, J.-P.; Meunier, B. Selection of a trioxaquine as an antimalarial drug candidate. Proc. Natl. Acad. Sci. USA 2008, 105, 17579–17584. [Google Scholar] [CrossRef] [PubMed]
- Portela, J.; Boissier, J.; Gourbal, B.; Pradines, V.; Collière, V.; Coslédan, F.; Meunier, B.; Robert, A. Antischistosomal activity of trioxaquines: In vivo efficacy and mechanism of action on Schistosoma mansoni. PLoS Negl. Trop. Dis. 2012, 6, e1474. [Google Scholar] [CrossRef] [PubMed]
- El-Kouni, M.H.; Diop, D.; Cha, S. Combination therapy of schistosomiasis by tubercidin and nitrobenzylthionisine 5′-monophosphate. Proc. Natl. Acad. Sci. USA 1983, 80, 6667–6680. [Google Scholar] [CrossRef] [PubMed]
- El-Kouni, M.H.; Knopf, P.M.; Cha, S. Combination therapy of Schistosoma japonicum by tubercidin and nitrobenzylthioinosine 5′-monophospahte. Biochem. Pharmacol. 1985, 34, 3921–3923. [Google Scholar] [CrossRef]
- El-Kouni, M.H. Efficacy of combination therapy with tubercidin and nitrobenzylthioinoisine 5’-monophosphate against chronic and advanced stages of schistosomiasis. Biochem. Parasitol. 1991, 41, 815–820. [Google Scholar] [CrossRef]
- El-Kouni, M.H.; Messier, N.J.; Cha, S. Treatment of schistosomiasis by purine nucleoside analogues in combination with nucleoside transport inhibitors. Biochem. Pharmacol. 1987, 36, 3815–3821. [Google Scholar] [CrossRef]
- Senft, A.W.; Crabtree, G.W. Purine metabolism in the schistosomes: Potential targets for chemotherapy. Pharmcol. Ther. 1983, 20, 341–356. [Google Scholar] [CrossRef]
- Sulaiman, S.M.; Ali, H.M.; Homeida, M.M.; Bennett, J.L. Efficacy of a new Hoffmann-La Roche compound (Ro 15-5458) against Schistosoma mansoni (Gezira strain, Sudan) in vervet monkeys (Cercopithecus aethiops). Trop. Med. Parasitol. 1989, 40, 335–336. [Google Scholar] [PubMed]
- Kamel, G.; Metwally, A.; Guirguis, F.; Nessim, N.G.; Noseir, M. Effect of a combination of the new antischistosomal drug Ro 15-5458 and praziquantel on different strains of Schistosoma mansoni infected mice. Arzneim-Forsch Drug Res. 2000, 50, 391–394. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.M.; Salama, M.A.; Abou-Basha, L. Experimental murine schistomiasis: Reduced hepatic morbidity after, pre- and/or post-infection treatment with ibuprofen or diclofenac sodium. Ann. Trop. Med. Parasitol. 1995, 89, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.R.; Zoheiry, M.M.K.; Nosseir, M.M.F. Effect of combined chemotherapy and anti-inflammatory drugs on murine schistosomiasis. Arzneim-Forsch Drug Res. 2002, 52, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Yepes, E.; Varela, M.R.E.; López-Abán, J.; Rojas-Caraballo, J.; Muro, A.; Mollinedo, F. Inhibition of granulomatous inflammation and prophylactic treatment of schistosomiasis with a combination of edelfosine and praziquantel. PLoS Negl. Trop. Dis. 2015, 9, e0003893. [Google Scholar] [CrossRef] [PubMed]
- Mollinedo, F.; Gajate, C.; Morales, A.I.; Canto-Jañez, E.; Justies, N.; Collía, F.; Rivas, J.V.; Modolell, M.; Iglesias, A. Novel anti-inflammatory action of edelfosine lacking toxicity with protective effect in experimental colitis. J. Pharmacol. Exp. Ther. 2009, 329, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Abramowski, P.; Otto, B.; Martin, R. The orally available, synthetic ether lipid edelfosine inhibits T cell proliferation and induces a type I interferon response. PLoS ONE 2014, 9, e91970. [Google Scholar] [CrossRef] [PubMed]
- Caldas, I.R.; Campi-Azevedo, A.C.; Oliveira, L.F.; Silveira, A.M.; Gazzinelli, G. Human schistosomiasis mansoni: Immune responses during acute and chronic phases of the infection. Acta Trop. 2008, 108, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.J.; MacDonald, A.S. The immunobiology of schistosomiasis. Nat. Rev. Immunol. 2002, 2, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Yepes, E.; Varela-M, R.E.; López-Abán, J.; Habib Dakir, E.L.; Mollinedo, F.; Muro, A. In vitro and in vivo antischistosomal activity of the alkylphospholipid analog edelfosine. PLoS ONE 2014, 9, e109431. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.I.; Ali, I.; Nessim, N.G.; Amer, N.M.; Abd el Kader el Khafif, M.; Ashour, A.; el Mohandes, M. Treatment of acute schistosomiasis mansoni with praziquantel and an antifibrotic agent in mice. Arzneim-Forsch Drug Res. 2003, 53, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Giboda, M.; Zenka, J.; Julis, I.; Vítovec, J. Experimental schistosomiasis mansoni: Modulation of granulomas by inhibition of collagen cross-link formation. Preliminary report. Ann. Trop. Med. Parasitol. 1992, 86, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Botros, S.; Mahmoud, M.; Hamman, O.; Salah, F.; Zidek, Z.; Masek, K. Effect of adamantylamide dipeptide as adjuvant therapy to praziquantel in mice infected with different S. mansoni isolates. APMIS 2006, 114, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.F.M.; Ibrahim, M.M. Antischistosomal action of atorvastatin alone and concurrently with medroxyprogesterone acetate on Schistosoma haematobium harboured in hamster: Surface ultrastructure and parasitological study. Acta Trop. 2005, 93, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Z.; Foster, L.; Bennett, J.L. Antischistosomal action of mevinolin: Evidence that 3-hydroxy-methylglutaryl-coenzyme a reductase activity in Schistosoma mansoni is vital for parasite survival. Naunyn-Schmiedebergs Arch. Pharmacol. 1990, 342, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Rojo-Arreola, L.; Long, T.; Asarnow, D.; Suzuki, B.M.; Singh, R.; Caffrey, C.R. Chemical and genetic validation of the statin drug target to treat the helminth disease, schistosomiasis. PLoS ONE 2014, 9, e87594. [Google Scholar] [CrossRef] [PubMed]
- Rizk, M.; Ibhraim, N.; El-Rigal, N. Comparative in vivo antioxidant levels in Schistosoma mansoni mice treated with praziquantel or the essential oil Melaleuca armillaris leaves. Pak. J. Biol. Sci. 2012, 15, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Eraky, M.A.; El-Kholy, A.A.E.; Rashed, G.A.E.; Hammam, O.A.; Moharam, A.F.; Abou-Ouf, E.A.; Aly, N.S.; Kishik, S.M.; Abdallah, K.F.; Hamdan, D.I. Dose-response relationship in Schistosoma mansoni juvenile and adult stages following limonin treatment in experimentally infected mice. Parasitol. Res. 2016, 115, 4045–4054. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, L.G.; Kapadia, G.J.; Tonuci, L.R.S.; Caixeta, S.C.; Parreira, N.A.; Rodrigues, V.; Da Silva Filho, A.A. In vitro schistosomicidal effects of some phloroglucinol derivatives from Dryopteris species against Schistosoma mansoni adult worms. Parasitol. Res. 2010, 10, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Allam, G.; Abuelsaad, A.S. In vitro and in vivo effects of hesperidin treatment on adult worms of Schistosoma mansoni. J. Helminthol. 2014, 88, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hafeez, E.H.; Ahmad, A.K.; Abdulla, A.M.; Aabdel-Wahab, S.; Mosalem, F.A. Therapeutic effect of alpha lipoic acid combined with praziquantel on liver fibrosis induced by Schistosoma mansoni challenged mice. Parasitol. Res. 2012, 111, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Soliman, R.H.; Ismail, O.A.; Badr, M.S.; Nasr, S.M. Resveratrol ameliorates oxidative stress and organ dysfunction in Schistosoma mansoni infected mice. Exp. Parasitol. 2017, 174, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Rehder, V.L.G.; Oliveira, A.S.S.; Júnior, Í.M.; de Carvalho, J.E.; de Ruiz, A.L.; Jeraldo Vde, L.; Linhares, A.X.; Allegretti, S.M. Schistosoma mansoni: In vitro schistosomicidal activity of essential oil of Baccharis trímera (less) DC. Exp. Parasitol. 2012, 132, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Rehder, V.L.G.; Oliveira, A.S.; Jeraldo Vde, L.; Linhares, A.X.; Allegretti, S.M. Antihelminthic activity in vitro and in vivo of Baccharis trímera (less) DC against immature and adult worms of Schistosoma mansoni. Exp. Parasitol. 2014, 139, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.F.M.; El-Shennawy, W.S. Evaluation of the protective effect of two antioxidative agents in mice experimentally infected with Schistosoma mansoni: Haematological and histopathological aspects. Pak. J. Biol. Sci. 2003, 6, 887–897. [Google Scholar] [CrossRef]
- Soliman, M.F.M.; El Shenawy, N.S.; El Arabi, S.E. Schistosoma mansoni: Melatonin enhances efficacy of cercarial and soluble antigen in the induction of protective immunity against infection in the hamster. Exp. Parasitol. 2008, 119, 291–295. [Google Scholar] [CrossRef] [PubMed]
- El-Sokkary, G.H.; Omar, H.M.; Hassanein, A.M.M.; Cuzzocrea, S.; Reiter, R.J. Melatonin reduces oxidative damage and increases survival of mice infected with Schistosoma mansoni. Free Rad. Biol. Med. 2002, 32, 319–332. [Google Scholar] [CrossRef]
- El-Shennawy, A.M.; Mohamed, A.H.; Abass, M. Studies on parasitologic and haematologic activities of an enaminone derivative of 4-hydroxyquinolin-2(1H)-one against murine schistosomiasis mansoni. MedGenMed 2007, 9, 15. [Google Scholar] [PubMed]
- Ebeid, J.I.; Mohammed, A.R.; Hussein, N.A.; El-Shennawy, A.; Noshy, M.; Abbas, M. In vivo antioxidant and antigenotoxic evaluation of an enaminone derivative BDHQ combined with praziquantel in uninfected and Schistosoma mansoni infected mice. J. Appl. Pharm. Sci. 2014, 4, 25–33. [Google Scholar] [CrossRef]
- Mata-Santos, H.A.; Lino, F.G.; Rocha, C.C.; Paiva, C.N.; Castelo Branco, M.T.; Pyrrho Ados, S. Silymarin treatment reduces granuloma and hepatic fibrosis in experimental schistosomiasis. Parasitol. Res. 2010, 107, 1424–1434. [Google Scholar] [CrossRef] [PubMed]
- El-Lakkany, N.M.; Hammam, O.A.; El-Maadawy, W.H.; Badawy, A.A.; Ain-Shoka, A.A.; Ebeid, F.A. Anti-inflammatory/anti-fibrotic effects of the hepatoprotective silymarin and the schistosomicide praziquantel against Schistosoma mansoni-induced liver fibrosis. Parasites Vector 2012, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Wan, K.; Wang, P.; Zhang, L. In vivo and in vitro activity of oil extract of garlic (Allium sativum Linnaeus) against Schistosoma japonicum cercariae. Rev. Soc. Bras. Med. Trop. 2017, 50, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Kamel, R.O.A.; El-Shinnawy, N.A. Immunomodulatory effect of garlic oil extract on Schistosoma mansoni infected mice. Asian Pac. J. Trop. Med. 2015, 8, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Metawally, N.S. Potency of Allium sativum and Allium cepa oils against Schitosoma mansoni infection in mice. Egypt. J. Hosp. Med. 2006, 23, 319–322. [Google Scholar]
- Mantawy, M.M.; Ay, H.F.; Rizk, M.Z. Therapeutic effects of Allium sativum and Allium cepa in Schistosoma mansoni experimental infection. Rev. Inst. Med. Trop. 2011, 53, 155–163. [Google Scholar] [CrossRef]
- Mantawy, M.M.; Aly, H.F.; Zayed, N.; Fahmy, Z.H. Antioxidant and schistosomicidal effect of Allium sativum and Allium cepa against Schistosoma mansoni different stages. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 69–80. [Google Scholar] [PubMed]
- Mohamed, A.M.; Metwally, N.M.; Mahmoud, S.S. Sativa seeds against Schistosoma mansoni different stages. Mem. Inst. Oswaldo Cruz 2005, 100, 205–211. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mahmoud, M.R.; El-Abhar, H.S.; Saleh, S. The effect of Nigella sativa oil against the liver damage induced by S. mansoni infection in mice. J. Enthopharmacol. 2002, 79, 1–11. [Google Scholar] [CrossRef]
- Sheir, S.K.; Maghraby, A.M.; Mohamed, A.H.; Osman, G.Y.; Al-Qormuti, S.A. Immunomodulatory and ameliorative role of Nigella sativa oil on Schistosoma mansoni infected mice. Can. J. Pure Appl. Sci. 2015, 9, 3345–3355. [Google Scholar]
- El-Shennawy, N.S.; Soliman, M.F.M.; Reyad, S.I. The effect of antioxidant properties of aqueous garlic extract and Nigella sativa as anti-schistosomiasis agents in mice. Rev. Inst. Med. Trop. 2008, 50, 26–36. [Google Scholar] [CrossRef]
- El-Ansary, A.K.; Ahmed, S.A.; Aly, S.A. Antischistosomal and liver protective effects of curcuma longa extract in Schistosoma mansoni infected mice. Indian J. Exp. Biol. 2007, 45, 791–801. [Google Scholar] [PubMed]
- Allam, G. Immunomodulatory effects of curcumin treatment on murine schistosomiasis mansoni. Immunobiology 2009, 214, 712–727. [Google Scholar] [CrossRef] [PubMed]
- De Paula Aguiar, D.; Brunetto, M.M.M.; Rezende, M.E.; Graciano de Paula, R.; Ferreira, P.M.; Afonso, A.; Belo, S.; Tomie Ouchida, A.; Curti, C.; Cunha, W.R.; et al. Curcumin generates oxidative stress and induces apoptosis in adult Schistosoma mansoni worms. PLoS ONE 2016, 11, e0167135. [Google Scholar] [CrossRef] [PubMed]
- Seif el-Din, S.H.; Ebeid, F.A.; Badawy, A.A.; Ezzat, A.R. Protective effects of β-carotene; N-acetylcysteine with or without praziquantel treatment in Schistosoma mansoni-infected mice. Egypt. J. Schistosomiasis Infect. Endem. Dis. 2006, 28, 67–90. [Google Scholar]
- Aires, A.L.; Alburquerque, M.C.P.A.; Silva, R.A.; Schirato, G.V.; de Pontes Filho, N.T.; de Araújo, S.B.; Souza, V.M.; Costa, V.M.; Malagueño, E. Immunohistopathological changes in murine schistosomiasis mansoni under the influence of N-acetyl-l-cysteine. Parasitol. Res. 2012, 111, 1569–1578. [Google Scholar] [CrossRef] [PubMed]
- Seif el-Din, S.H.; Al-Hroob, A.M.; Ebeid, F.A. Schistosoma mansoni: N-acetylcysteine downregulates oxidative stress and enhances the antischistosomal activity of artemether in mice. Exp. Parasitol. 2011, 128, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Patocka, N.; Sharma, N.; Rashid, R.; Ribeiro, P. Serotonin signaling in Schistosoma mansoni: A serotonin-activated G protein-coupled receptor controls parasite movement. PLoS Pathog. 2014, 10, e1003878. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.; Patocka, N. Neurotransmitter transporters in schistosomes: Structure, function and prospects for drug discovery. Parasitol. Int. 2013, 62, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.-G.; Brindley, P.J.; Wang, S.-Y.; Chen, Z. Schistosoma genomics: New perspectives on schistosome biology and host-parasite interaction. Annu. Rev. Genom. Hum. Genet. 2009, 10, 211–240. [Google Scholar] [CrossRef]
- Erasmus, D.A. A comparative study of the reproductive system of mature, immature and “unisexual” female Schistosoma mansoni. Parasitology 1973, 67, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Gharib, B.; Abd-Allah, D.M.; Dessein, H.; De Reggi, M. Development of eosinophil peroxidase activity and concomitant alteration of the antioxidant defenses in the liver of mice infected with Schistosoma mansoni. J. Hepatol. 1999, 30, 594–602. [Google Scholar] [CrossRef]
- Caulfield, J.P.; Lenzi, H.L.; Elsas, P.; Dessein, A.J. Ultrastructure of the attack eosinophils stimulated by blood mononuclear cell products on schistosomula of Schistosoma mansoni. Am. J. Pathol. 1985, 120, 380–390. [Google Scholar] [PubMed]
- Elsammak, M.Y.; Al-Sharkaweey, R.M.; Ragab, M.S.; Amin, G.A.; Kandil, M.H. IL-4 and reactive oxygen species are elevated in Egyptian patients affected with schistosomal liver disease. Parasite Immunol. 2008, 30, 603–609. [Google Scholar] [CrossRef] [PubMed]
- McCormick, M.L.; Metwalli, A.; Railsback, M.A.; Weinstock, J.V.; Britigan, B.E. Eosinophils from schistosome-induced hepatic granulomas produce superoxide and hydroxyl radical. J. Immunol. 1996, 157, 5009–5015. [Google Scholar] [PubMed]
- Williams, D.L.; Bonilla, M.; Gladyshev, V.N.; Salinas, G. Thioredoxin glutathione reductase-dependent redox networks in Platyhelminth parasites. Antioxid. Redox Signal. 2013, 19, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Mei, H.; Lo Verde, P.T. Schistosoma mansoni: Cloning the gene encoding glutathione peroxidase. Exp. Parasitol. 1995, 80, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Olveda, D.U.; Olved, R.M.; McManus, D.P.; Cai, P.; Chau, T.N.P.; Lam, A.K.; Li, Y.; Harn, D.A.; Vinluam, M.L.; Ross, A.G.P. The chronic enteropathogenic disease schistosomiasis. Int. J. Inf. Dis. 2014, 28, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Stadecker, M.J.; Villanueva, P.O.F. Accessory cell signals regulate Th-cell responses from basic immunology to a model of helminthic disease. Immunol. Today 1994, 15, 571–573. [Google Scholar] [CrossRef]
- Liang, Y.J.; Luo, J.; Yuan, Q.; Zheng, D.; Li, Y.-P.; Shi, L.; Zhou, Y.; Cheng, A.-L.; Ren, Y.-Y.; Sun, K.-Y.; et al. New insight into the antifibrotic effects of praziquantel on mice in infection with Schistosoma japonicum. PLoS ONE 2011, 6, e20247. [Google Scholar] [CrossRef] [PubMed]
- Tawfeek, G.M.; Alafifi, A.M.; Azmy, M.F. Immunological indicators of morbidity in human schistosomiasis mansoni: Role of vascular endothelial growth and anti-soluble egg antigen IgG4 in disease progression. J. Egypt. Soc. Parasitol. 2003, 33, 597–614. [Google Scholar] [PubMed]
- Akhiani, A.A.; Nilsson, L.A.; Ouchterlony, O. Intranasal administration of Schistosoma mansoni adult worm antigen in combination with cholera toxin induces a Th2 cell response. Parasite Immunol. 1997, 19, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Ashour, A.A.; Ahmed, S.A.; Maghraby, A.S.; Zahran, H.G. Immunoprophylactic effect of single and mixed schistosomal antigens of Schistosoma mansoni infected mice. Egypt. J. Hosp. Med. 2004, 14, 86–103. [Google Scholar]
- Afifi, M.A.; El-Wakil, H.S.; Abdel-Ghaffar, M.M.; Mohamed, R.T. Application of adult worm and lung-stage antigens to immunize against Schistosoma mansoni using cytokines as adjuvants. J. Egypt. Soc. Parasitol. 2006, 36, 251–262. [Google Scholar]
- Vale, N.; Gouveia, M.J.; Rinaldi, G.; Santos, J.; Santos, L.L.; Brindley, P.J.; da Costa, J.M. The role of estradiol metabolism in urogenital schistosomiasis-induced bladder cancer. Tumor Biol. 2017, 39, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, M.J.; Santos, J.; Brindley, P.J.; Rinaldi, G.; Lopes, C.; Santos, L.L.; da Costa, J.M.; Vale, N. Estrogen-like metabolites and DNA-adducts in urogenital schistosomiasis-associated bladder cancer. Cancer Lett. 2015, 359, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Correia da Costa, J.M.; Vale, N.; Gouveia, M.J.; Botelho, M.C.; Sripa, B.; Santos, L.L.; Santos, J.H.; Rinaldi, G.; Brindley, P.J. Schistosome and liver fluke derived catechol-estrogens and helminth associated cancer. Front. Genet. 2014, 5, 444. [Google Scholar] [CrossRef] [PubMed]
- Pinlaor, S.; Yongvanit, P.; Prakobwong, S.; Kaewasamut, B.; Khoontawad, J.; Pinlaor, P.; Hiraku, Y. Curcumin reduces oxidative and nitrative DNA damage through balancing of oxidant-antioxidant status in hamster infected with Opisthorchis viverrini. Mol. Nutr. Food Res. 2009, 53, 1316–1328. [Google Scholar] [CrossRef] [PubMed]
- Zahid, M.; Saeed, H.; Beseler, C.; Rogan, E.G.; Cavalieri, E.L. Resveratrol and N-acetylcysteine block the cancer initiating step in MCF-10F cells. Free Rad. Biol. Med. 2001, 50, 78–85. [Google Scholar] [CrossRef] [PubMed]

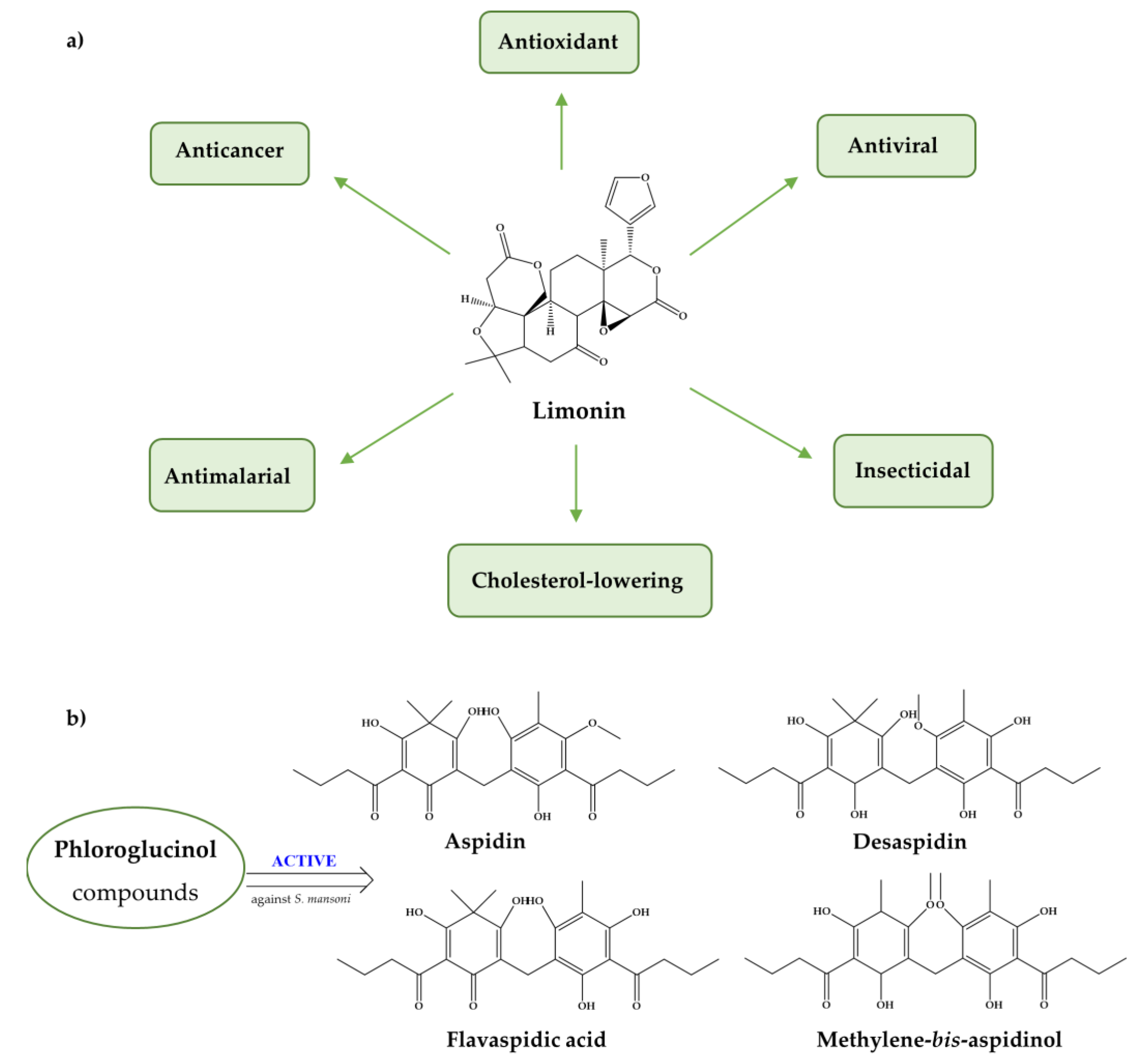
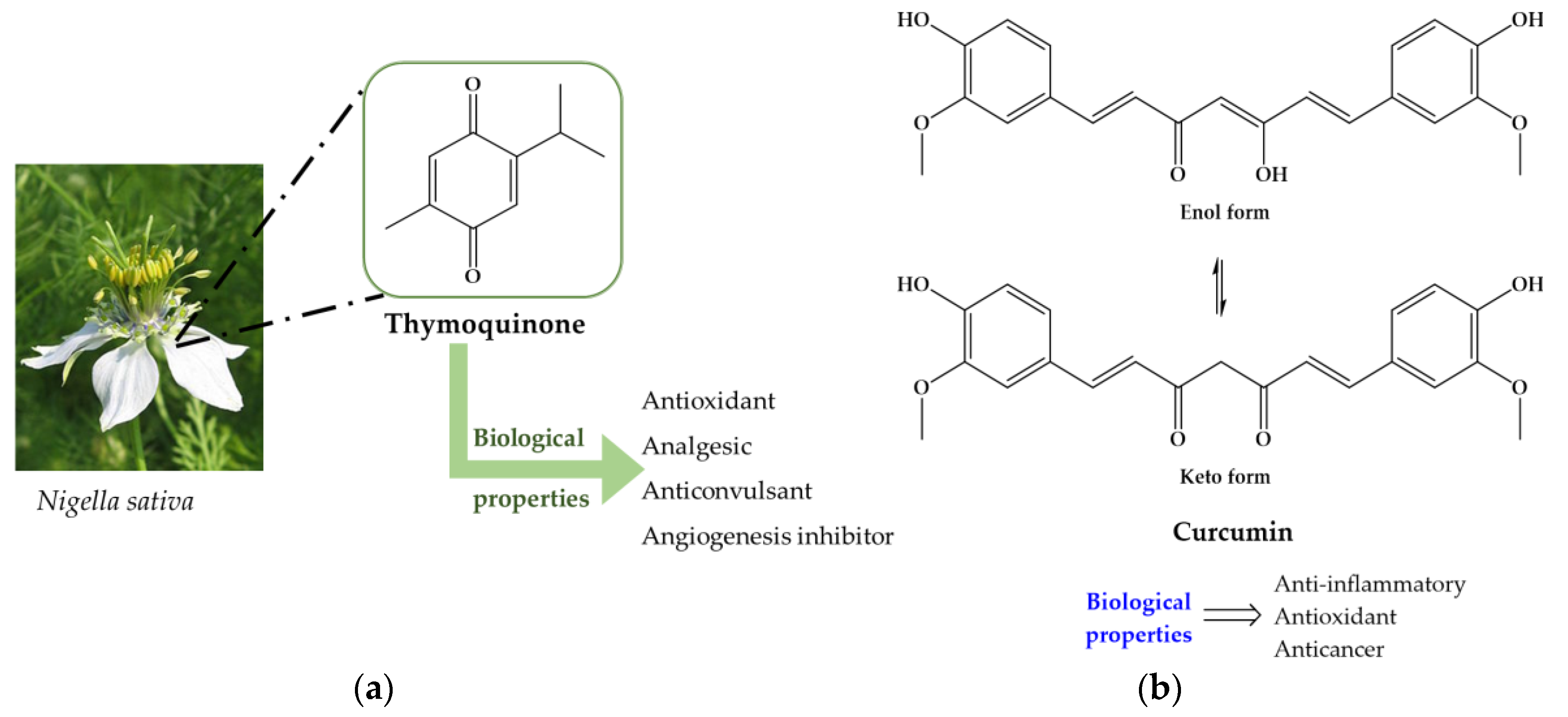
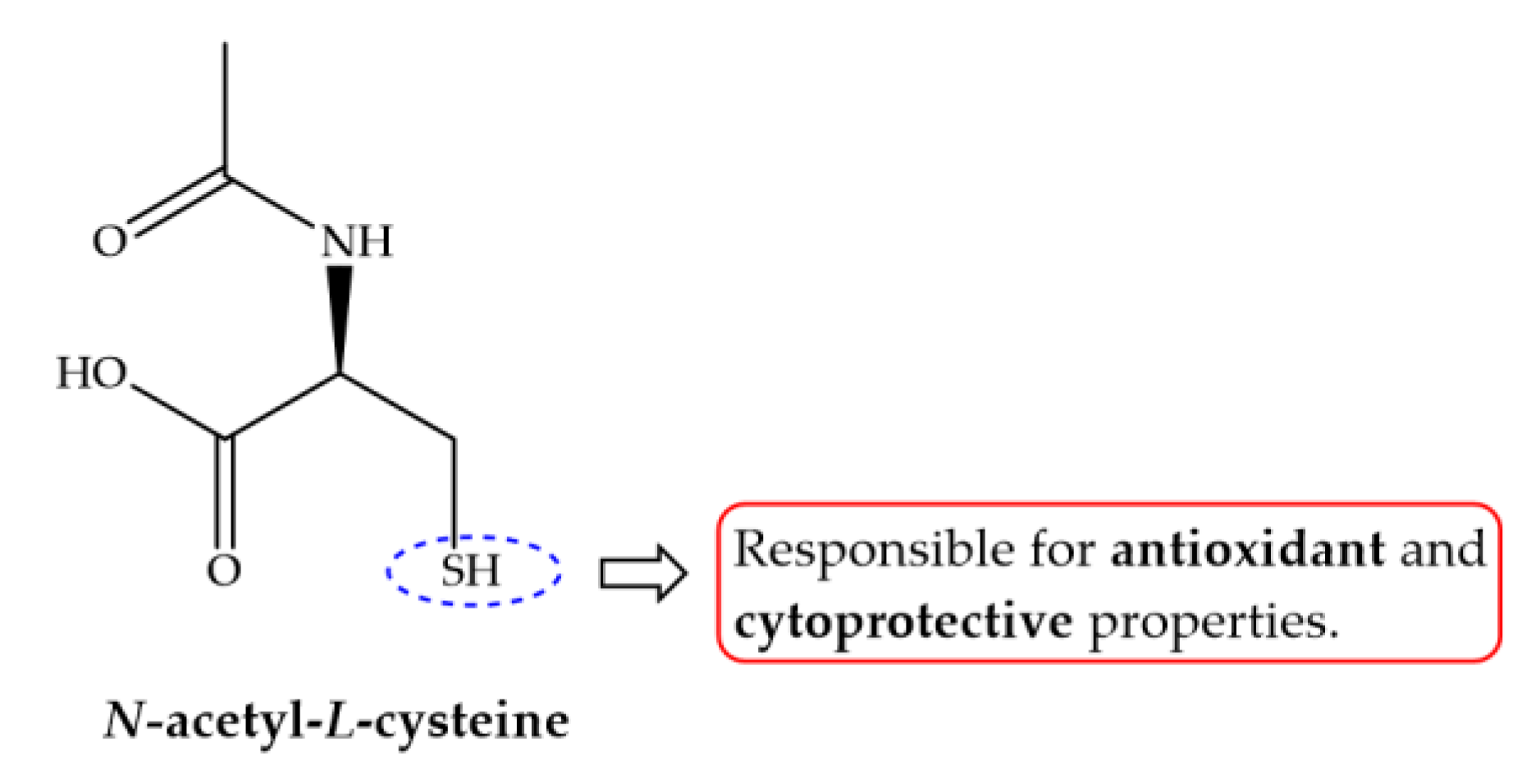
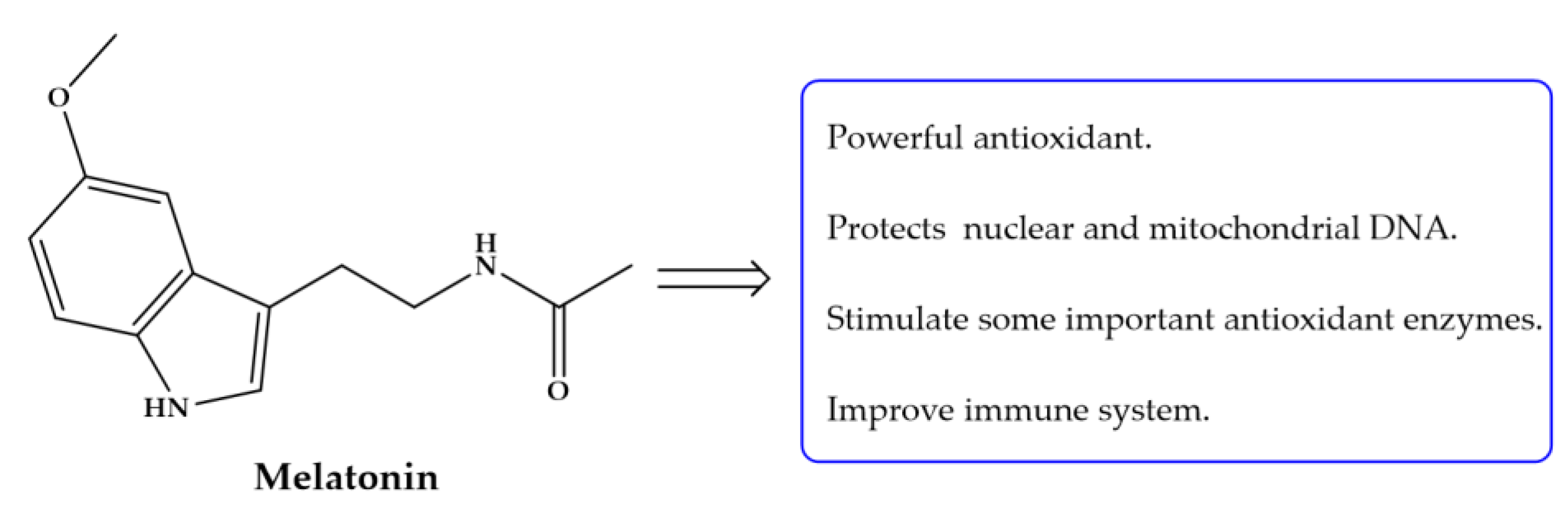
| Compound | Aim/Study | Treatment | Findings/Outcomes | Ref. |
|---|---|---|---|---|
| M. armillaris | Antischistosomal and antioxidant activity of essential oil on normal and infected S. mansoni mice in comparison to PZQ. | Oil 150 mg/kg orally from second week p.i., twice week for 6 weeks; PZQ 600 mg/kg, orally for 2 consecutive days, 8 weeks p.i. | Administration of PZQ and M. armillaris ameliorate the levels of GSH and lipid peroxides (MDA); Restored the activities of SOD and catalase; M. armillaris enhance antioxidant defense system reducing disease complications. | [111] |
| Limonin | Antischistosomal activity in vitro and in vivo harboring juvenile and adult S. mansoni worms. | Oral administration in a single dose of 50 or 100 mg/kg on day 21 p.i.; Same dose given on 56 p.i. | Limonin is more effective against immature stages rather adult worms also induce tegument alterations; Reduction of worm burden: at day 21 p.i. 70.0% and 83.3%; and day 56 p.i. 41.09 and 60.27%. Significant reduction in the hepatic (34.90% and 47.16%) and intestinal (46.67% and 56.1%) tissue egg load associated the oogram pattern with elevated dead egg levels; Also, ameliorate hepatic pathology with reduction in size and numbers of granulomas. | [112] |
| Pholoro-glucinol derivatives | Evaluation in vitro schistosomicidal activity of aspidin (AS), flavaspidic acid (FAA), methylene-bis-aspidinol (MbA) and desaspidin (DA) against S. mansoni adult worms. | AP-25 to 100 μM FAA-50 and 100 μM MbA-100 μM DA-25 to 100 μM | AP and FAA decrease motor activity with tegumental alterations while MBA and DA also decrease motor activity but without tegumental alterations. At highest concentrations viability of worms were similar to positive controls (PZQ); Egg production and the development of eggs produced were inhibited; Probably, in vitro activity is related to the inhibition of oxidative phosphorylation pathways. | [113] |
| Hesperidin | Evaluation of antischistosomal activity in vitro and in vivo and compared to PZQ. Effect on parasite antigens. Treatments were administered on 6th week p.i. | In vitro: 50, 100 and 200 μg/mL. In vivo: Hesp-600 mg/kg bw (6 injections, 2 injections per week for 3 consecutive week); PZQ (2 consecutive days with 500 mg/kg bw. | In vitro: At highest concentration, all males and females were dead while lower concentration had moderate effect. No activity on oogram pattern was seen. In vivo: Reduction of numbers of males, females and possibly worm pairs and total worm burden counts (47.5%) but was not higher than PZQ; significantly reduced tissue egg load. Augmented the mouse IgG response against soluble worm antigen protein, soluble egg antigen and cercarial preparation of S. mansoni. | [114] |
| α-Lipoic acid | Study combined effect of ALA with PZQ on liver fibrosis induced by S. mansoni infection in mice. | PZQ-500 mg/kg divided into 2 doses 9 weeks p.i.: PZQ (same described) + ALA (single dose 30 mg/kg) daily for two months. | Combine regimen results in reduction in the worm burden (ALA: 7.63 ± 1.49; PZQ: 6.13 ± 1.89; PZQ + ALA: 36.50 ± 10.80), egg count and granuloma size. Recovered the level serum of ALT, AST and GGT and increased the tissue level of GSH and decreased MDA (biomarkers of antioxidant function and stress oxidative, respectively). | [115] |
| Resveratrol | Investigate effect of Resv on oxidative stress imposed on liver, lung, kidney, brain and spleen of S. mansoni-infected mice. | 20 mg/kg once daily for 2 weeks | Improvement of lipid metabolism and antioxidant profile by Resv which were not only restricted to liver but also other vital organs. Specific biomarkers of lung and brain homeostasis also showed remarkable improvement. | [116] |
| B. trimera | Assessment of antischistosomal activity agaisnt S. mansoni adult worms in vitro. | 4 concentrations of 24, 48, 91 and 130 µg/mL. | Antischistosomal activity at highest concentrations with significant reductions in motility; Total inhibition in egg laying when parasites were exposure to sub-lethal concentrations and separations of all couples. Morphological changes on the tegument of worm’s males and females. | [117] |
| In vitro and in vivo efficacy of aqueous fraction and dichloromethane extracts against schistosomula, juvenile and adult worms of S. mansoni. | In vitro: Same as above In vivo: Single doses 40 and 200 mg/kg of B. trimera and PZQ 4 after 3 and 30 days p.i. and 60 p.i. | In vitro: Similar results described. In vivo: B. trimera exhibits major schistosomicidal effects in vivo against immature and adult worms (significantly female worm, 68–75%, reduction and number of eggs/g in faeces); Significant reduction in relation to number and size of granulomas. | [118] | |
| Melatonin | Assessment protection against oxidative stress induced by schistosomiasis mansoni. | 3.55 mg/kg daily for 30 consecutive days starting from first day p.i. | Decreased in total leukocyte count: Markedly reduced the fibrotic areas, small diameter of granuloma with few collagen fiber depositions; ameliorate liver architecture and glycogen content. | [119] |
| Establish an immunization program using S. mansoni adult worm antigen and cercarial antigen alone or concurrently with Mel in attempt to enhance efficacy against infection in mice. | 30 μg/mL CAP or SWAP on first day and 20 μg/mL on 4th day p.i.; On 7th day all hamsters were infected. Mel same regimen as above. | Mel alone did not result decrease of worm burden reductions (CAP: 538%; CAP + Mel: 67.01%; SWAP: 56.4% and SWAP + Mel: 99.3%). Highly significant reductions in egg load in liver and alteration of oogram pattern: high percentage of immature eggs and few dead eggs. Improved the oxidative status in the immunized groups. No antibody response was observed in the groups immunized with SWAP + Mel while low antibody level was observed in CAP + Mel. | [120] | |
| Investigate oxidative processes in mice infected with S. mansoni | 10 mg/kg, 2 weeks after S. mansoni infection until end of experiment; or daily for 30 days | Mel did not restore glutathione levels (although were tendencies for that); Increase SOD activity (but not statistically significant); Reduction of AST levels; Reduction of granuloma formation and highly protective against pathological changes not only in liver but kidney; Mel has multiple direct and indirect antioxidant actions and its ability to stimulate antioxidative enzymes and mitochondrial oxidative phosphorylation. | [121] | |
| 4-Hydroxy-quinolin-2(1H)-one (BDHQ) | Evaluation potential activity on murine schistosomiasis. For that mice were sacrificed on different weeks p.i.: 3 (for schistosomula) and 6 (for adult worms) | BDHQ: Lower dose—10 mg/mL for consecutive days; Higher dose on same regimen; PZQ: 2 times of 500 mg/kg 2 consecutive days on different weeks. | Antischistosomal activity against immature and mature worms; Destructive effects on the female and male genital systems; Antischistosomal activity may be due to its mixed cellular and humoral immunologic mechanisms, as demonstrated by the significant increase of serum levels of IgE and IFN-γ. | [122] |
| 4-Hydroxy-quinolin-2(1H)-one (BDHQ) | Evaluation of antioxidant and antigenotoxic effects alone or combined with PZQ. | PZQ, 0 or 500 mg/kg BDHQ, 600 mg/kg PZQ (250 mg/kg) + BDHQ (300 mg/kg) for 2 consecutive days | BDHQ alone or combined resulted in highly significant reduction in total worm burden (7 weeks p.i. PZQ: 86.37%, BDHQ: 79.22%; PZQ + BDHQ: 91.84%; 9 weeks PZQ: 94.72%, BDHQ: 92.32%; PZQ + BDHQ: 95.54%), associated with significant reduction in the hepatic tissue egg load; Drugs alone reduced the granuloma size and inflammatory cells. These parameters were improved with combine regimen; Significant decrease in MDA level accompanied with highly increase in NOx level with combine regimen, in addition to increase in the activities of both SOD and CAT; Remarkable significant decrease in % DNA fragmentation reaching a level close to control; These suggest a synergistic action attributed to different mechanism of action of both drugs that achieved the same or higher levels of efficacy using smaller doses of either agent. | [123] |
| Sylimarin | Assessment of parasitological and biochemical parameters on S. mansoni infection in mice. | 10, 20 or 25 doses of 10 mg/kg Syl suspended on carboxymethyl-cellulose at 55 days p.i. | Did not show antischistosomal activity; Reduced granulomatous and hepatic fibrosis. At acute schistosomiasis may result in a mild course of murine schistosomiasis and minimize the deleterious effects. | [124] |
| Anti-inflammatory/antifibrotic effect alone and combined with PZQ. | Syl: —4th week p.i. (3 weeks before PZQ therapy) —12th week p.i. (5 weeks after PZQ); PZQ (7th week p.i.) Syl + PZQ | Syl alone: Partial decrease of worm burden (26.55 and 39.39%) and decrease hepatic tissue egg load with an increase in percentage of dead ova; Modulation of granuloma size and conservation of hepatic GSH. PZQ: Complete eradication of worm, egg and alleviated liver inflammation and fibrosis; Combine regime: Improvement of liver function and histopathology whether acute and chronic infection may due to a combine action of anti-inflammatory, anti-fibrotic actions, in addition to the antioxidant properties of silymarin. Syl did not interfere or affect the antischistosomal activity of PZQ. Worm burden reduction 97–100%. | [125] | |
| A. sativum | Antischistosomal activity against S. japonicum cercariae in vitro and in vivo. | In vitro: 10−2 to 10−6 (v/v) concentration. In vivo: Pre-treated with garlic, then mice were infected. | Garlic oil displays marked activity agaisnts S. japonicum cercariae and may be used as agent to prevent S. japonicum (pre-exposure garlic oil at 10−4 and high showed total inhibition of infection). | [126] |
| A. sativum | Assess potency and the immunomodulatory response in enhancing the host immune system caused by S. mansoni in mice at different stages of worm. | 100 mg/kg body weight from 1 to 7 days p.i., 14 to 21 or 1 to 42 days p.i. | Morphologic alterations in the parasite tegument; significant decrease in worm burden, hepatic and inestinal ova count. Decline in granuloma number and diameter; Reduction in serum TNF-α, ICAM-1, IgG and IgM after 7 and 42 days p.i.; garlic oil enhance host immune system. | [127] |
| Ability of both oils to offser infectivity as well as metabolic disturbances induced by S. mansoni infection | 5 mL/kg body daily separately for 8 weeks on healthy control and infected groups. On infected groups oil were given 24 h p.i. | Reduced worm burden (garlic: 67.56% and onion: 75.97%) and ova count; normalized liver functions enzymes; effect may be induced by improving the immunological host immune system and their antioxidant activities. | [128] | |
| A. sativum+ A. cepa | Effect of both oils alone and mixed or concurrently with PZQ on biochemical parameters of experimentally infected S. mansoni mice. | A. sativum or A. Cepa, 2 g/100 g body weight daily for 45 consecutive days. PZQ: 500 mg/kg bw on 2 successive days 45 days p.i. | Significant reduction in worm burden (PZQ: 95.8%; onion: 66.29%; PZQ + onion: 99.1%; garlic: 73.41; garlic + PZQ: 99.3%; garlic + onion: 74.63; garlic + onion + PZQ: 99.7%); Reduction hepatic and intestinal eggs and oogram count; Suppression in granuloma tissue formation and diminutive histopathological changes; Improvement of liver architecture and attenuated the decrease of tissue antioxidant enzymes | [129] |
| Antischistosomal activity in vitro against S. mansoni miracidia, cercariae, schistosomula and adult worm. Effect in vivo on lipid peroxidase and antioxidant enzymes. | In vitro: 0.5–5 ppm In vivo: Same described above. | Lethal effect of both antioxidant against all developmental stages; Inhibition of coupling; Powerful reducing capacity demonstrated in DPHH radical scavenging and NO; Both plants enhance host antioxidant system indicated by lowering in lipid peroxide and stimulation of SOD, CAT, GR, TrxR and SDH enzyme levels which could turn render parasite vulnerable. | [130] | |
| Antischistosomal activity against miracidia, cercariae and adult worms in vitro. Effect on some antioxidants enzymes. | In vitro: Serial concentrations (0.5–5 ppm) for miracidia and cercariae. Adult worms, 10–110 ppm. | Antischistosomal activity against miracida and cercariae; Separation of coupled worms; Inhibition of egg laying by adult female worms; Significant inhibition of parasitic antioxidant enzymes (SOD, GR and GPX) and enzymes glucose metabolism (HK and G-6-PDH), higher in males than females. | [131] | |
| Nigella sativa | Effect in protection against oxidative stress in experimentally infected mice with S. mansoni. | N. sativa oil (1.14 g/kg orally) for 30 consecutive days from first day p.i. | No suppressive effect on granuloma formation in intestine; Did not improve the liver architecture; Noticeable degree of protection represented in less severe pathological changes, particularly the frequency of inflammatory reactions. | [126] |
| Study effect the oil on liver functions and antioxidant ability on experimentally infected mice with S. mansoni. | 2.5 and 5 mL/kg orally either alone or in combination with PZQ (500 mg/kg for 2 consecutive days) | N. sativa alone: Reduce the number of S. mansoni worms in the liver; Total worm burden: 22% e 32%, respectively, while PZQ: 98%; Decreased total number of ova deposited; Increased the number of dead ova; Reduced the granuloma markedly; Partially correct alterations in serum levels of ALT, GGT, activity as well as the Ab content. Failed in the liver restore either LPD and GSH content or LDH (lactate dehydrogenase) and SOD activity to normal level. N. sativa + PZQ: Improved most parameters with most prominent effect was further lowering in dead ova number over that produced by PZQ. Total worm burden: 98% and 99%. | [132] | |
| Nigella sativa | Investigate immune mechanism possibly involved in the amelioration of histopathological changes in liver of S. mansoni infected mice treated alone or in combination with ART or PZQ. | N. sativa: orally with 0.2 mg/kg of body weight for 4 weeks starting from 1st day p.i. ART: intramuscularly single dose of 300 mg/kg of body weight after 49 days p.i. PZQ: 500 mg/kg for 2 consecutive days | N. sativa as well as the combination of ART or PZQ resulted in significant increase in IL-2, IL-12 and TNF-α activities in S. mansoni infected mice as well as treatment of NS in non-infected. N. sativa in combination with ART or PZQ accelerate healing pathological granulomatous lesions of liver architecture and improved host immunity by stimulating cytokines. | [133] |
| Antischistosomal activity and antioxidant effects of NS alone or combined with garlic extracts on experimentally S. mansoni infected mice. | Garlic extract 125 mg/kg p.i. and NS oil 0.2 mg/kg alone or combine for successive 28 days, starting 1st day p.i. | All treatment regimens significantly affected oogram pattern: treatment with compounds alone resulted in reduction of percentage of mature eggs while combine regimen resulted in increase of percentage of dead eggs. Administration of garlic extract prevent GSH depletion on infected mice. Combine regimen had more significant effect on serum enzymes (AST and ALP). | [134] | |
| Curcumin | Assess curative effect of oil extract in liver cells of S. mansoni infected mice in compaison to PZQ | PZQ: 500 mg/kg by 2 consecutive days Extract: 300 mg/kg bw after one month p.i., twice a week for 2 months | Curcumin normalize the concentration of protein, glucose, AMP-deaminase and adenosine deaminase which were altered by infection Lowered pyruvate kinase level while PZQ induce more elevation; More potent rather PZQ in reducing egg count but no lowering worm burden. Most likely, antifecundity effect of curcumin might be involve in impairment or adult worms. | [135] |
| Evaluation of schistosomicidal activity in vivo and immunomodulation of granulomatous inflammation and liver pathology in acute S. mansoni infection. | Total dose 400 mg/kg bw divided into 16 injections (2 injections per week for 8 consecutive weeks) starting from the first week of infection. | Effective in reducing worm (44.4%) and tissue-egg burdens; Reduction granuloma volume and liver collagen (79%); Restore hepatic enzymes activities to normal levels and enhanced catalase activity; Low serum level of both IL-12 and TNF-α; Augmented specific IgG and IgG1 responses against both SWAP and SEA.; It modulates cellular and humoral responses. | [136] | |
| Evaluation its role on induction of apoptosis and oxidative stress in couples of adult S. mansoni worms in vitro | 1.56 to 100 μM incubated for 6, 12 or 24 h. | Significantly decreases the viability of adult female and male worms; Induce separation of couples and morphological alteration on mitochondria; Induce formation of SOD and increase its activity in adult worms; Alters several oxidative stress parameters in adult worms such decrease of GST, GR and GPX culminating in the oxidation of protein: Generates oxidative stress followed by an apoptotic-like event in adult worms, which ultimate leads to their dead. | [137] | |
| β-carotene | Evaluation the protective effect on experimentally S. mansoni infected mice and on major enzymes activities involved in liver redox. | PZQ, 7 weeks p.i., 500 mg/kg (full dose) or PZQ ED50 74.64 mg/kg βC, 2.7 mg/kg, 1 week before infection. βC + PZQ ED50 as mentioned | Produced significant reduction in worm burden (total number of worms: PZQ: 11.57 ± 0.59; PZQ (full dose): 0.46 ± 0.14; βC: 17.64 ± 1.11; βC + PZQ: 8.38 ± 0.51) accompanied with increase of dead ova and decrease in percentage of mature ova; reduced liver granuloma diameter. Combined regimen improved these parameters. Combined regimen improved the effect of antioxidant enzymes (as GPX and GST) and increase serum ALT and GGT. βC has protective effects against liver fibrosis which may be due to ability to encounter or minimize the formation of schistosomal products. | [138] |
| N-Acetyl-cysteine | Study immunopathological changes in murine schistosomiasis alone or in combination with PZQ. | NAC (200 mg/kg/day on 1st day after infection for acute phase; On 45th for the intermediate; 59 and 75th for chronic phases. PZQ (100 mg/kg) from 45th to 49th day p.i. | NAC alone did not present any schistosomicidal activity; animals treated with NAC and/or PZQ showed a reduction in the size of granulomas and those treated with NAC exhibited a lower degree of fibrosis. NAC functions as a direct scavenger of NO and peroxinitrite which are related to reductions of IFN-γ levels and increasing of IL-10 synthesis; Induce an immunomodulatory effect and reduce liver damage during granulomatous inflammation. | [139] |
| Investigate ability of NAC to enhance potential of ART against adult S. mansoni worms and evaluates protective role on oxidative stress. | NAC-300 mg/kg 5 days a week for 4 weeks ART-300 mg/kg 7 weeks p.i. NAC + ART (as described) | Combine regimen approximately recovered levels of serum enzymes, content of GSH and activities. Decrease the total number of worms and hepatic ova count. ART alone produce valuable modulations in the hepatic activities; NAC may prevent experimental liver injury by modulating and enhancing GSH content and GSH-dependent antioxidant enzyme activities. Total worms: ART: 7.6 ± 1.5; NAC: 17.7 ± 1.5; NAC + ART: 3.3 ± 1.1. | [140] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouveia, M.J.; Brindley, P.J.; Gärtner, F.; Costa, J.M.C.d.; Vale, N. Drug Repurposing for Schistosomiasis: Combinations of Drugs or Biomolecules. Pharmaceuticals 2018, 11, 15. https://doi.org/10.3390/ph11010015
Gouveia MJ, Brindley PJ, Gärtner F, Costa JMCd, Vale N. Drug Repurposing for Schistosomiasis: Combinations of Drugs or Biomolecules. Pharmaceuticals. 2018; 11(1):15. https://doi.org/10.3390/ph11010015
Chicago/Turabian StyleGouveia, Maria João, Paul J. Brindley, Fátima Gärtner, José M. Correia da Costa, and Nuno Vale. 2018. "Drug Repurposing for Schistosomiasis: Combinations of Drugs or Biomolecules" Pharmaceuticals 11, no. 1: 15. https://doi.org/10.3390/ph11010015
APA StyleGouveia, M. J., Brindley, P. J., Gärtner, F., Costa, J. M. C. d., & Vale, N. (2018). Drug Repurposing for Schistosomiasis: Combinations of Drugs or Biomolecules. Pharmaceuticals, 11(1), 15. https://doi.org/10.3390/ph11010015






