A Systematic Review and Meta-Analysis of the In Vivo Haemodynamic Effects of Δ9-Tetrahydrocannabinol
Abstract
1. Introduction
2. Results
2.1. Effect of THC Treatment on Haemodynamics
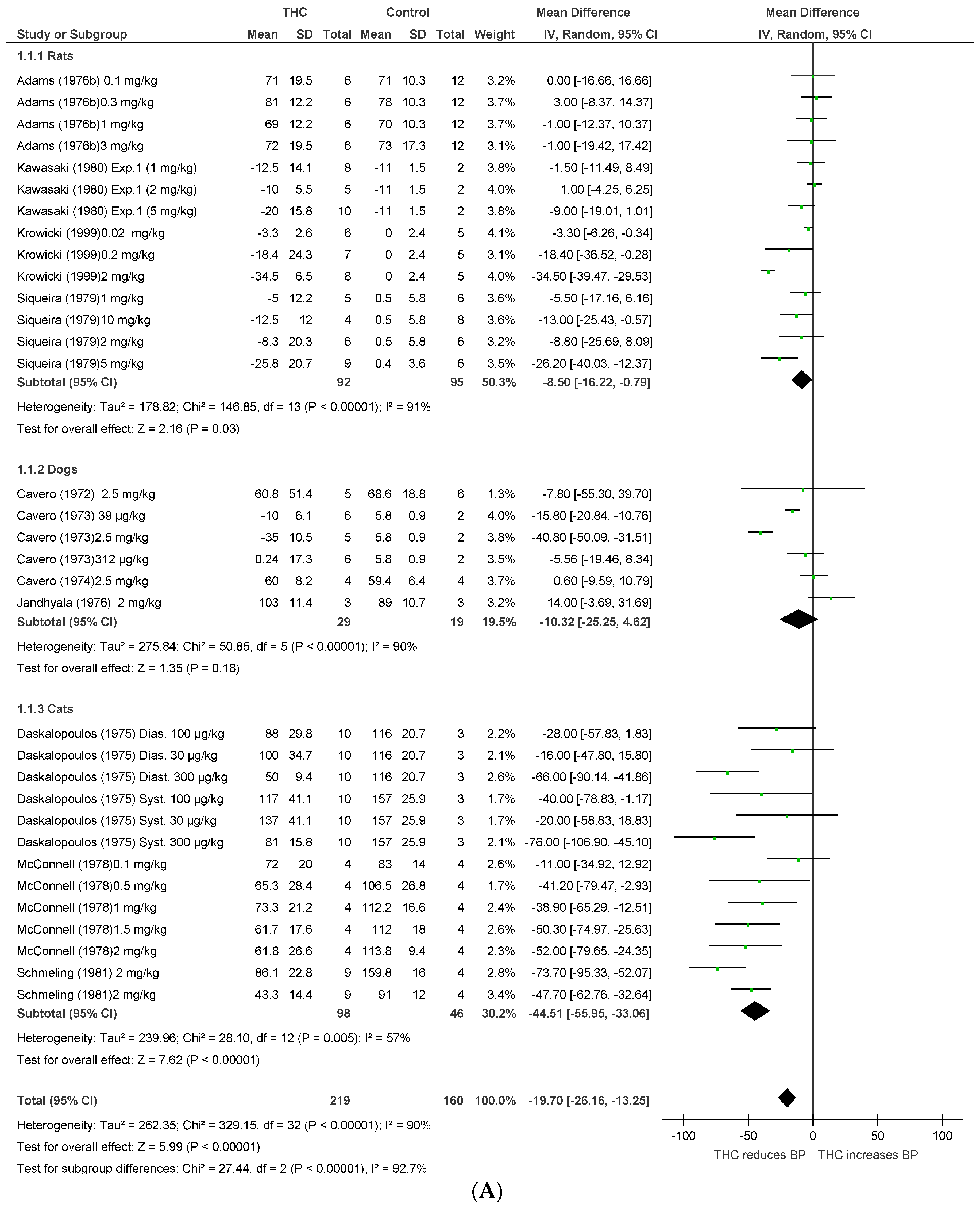
2.1.1. Anaesthetised Animals
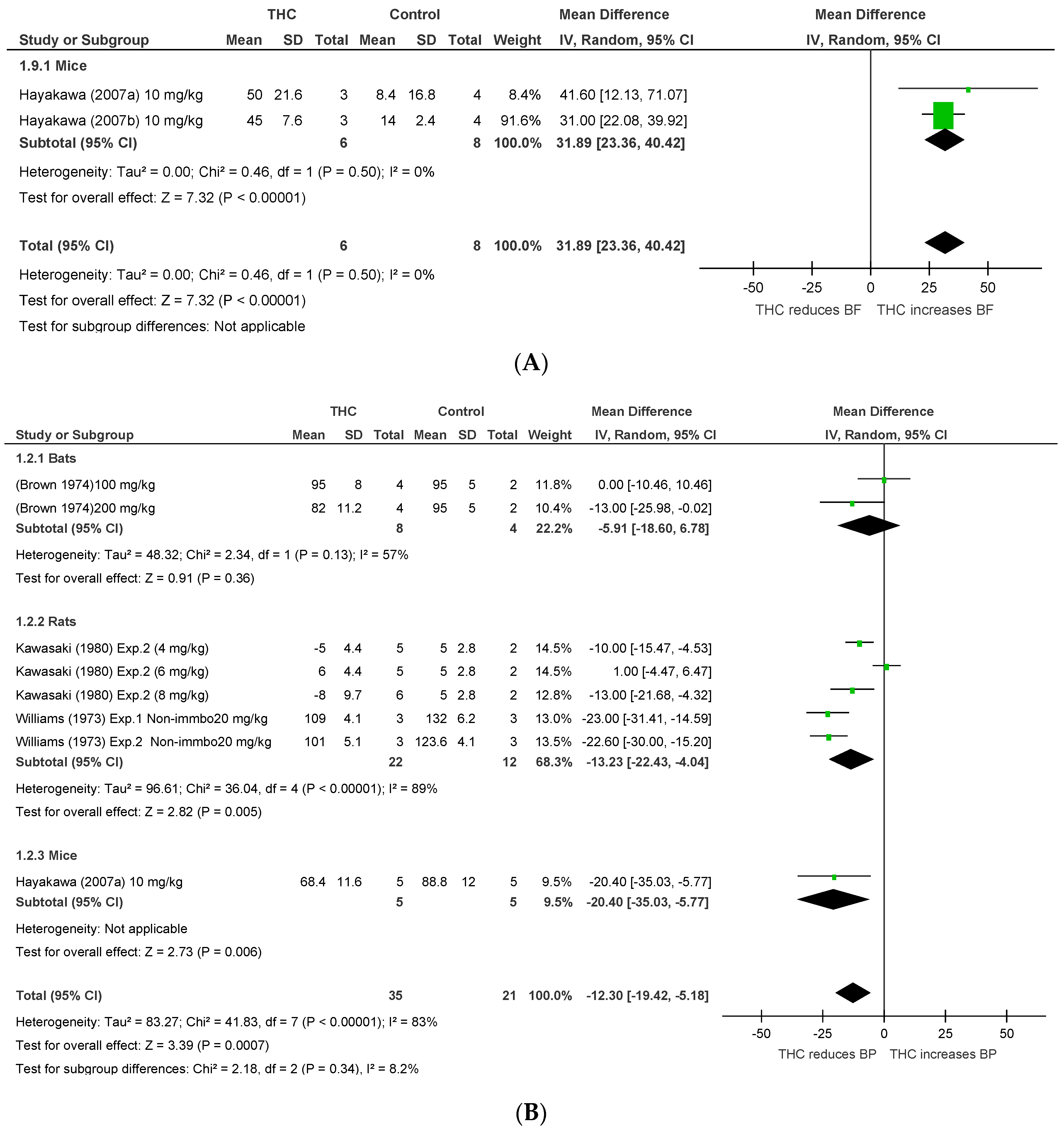
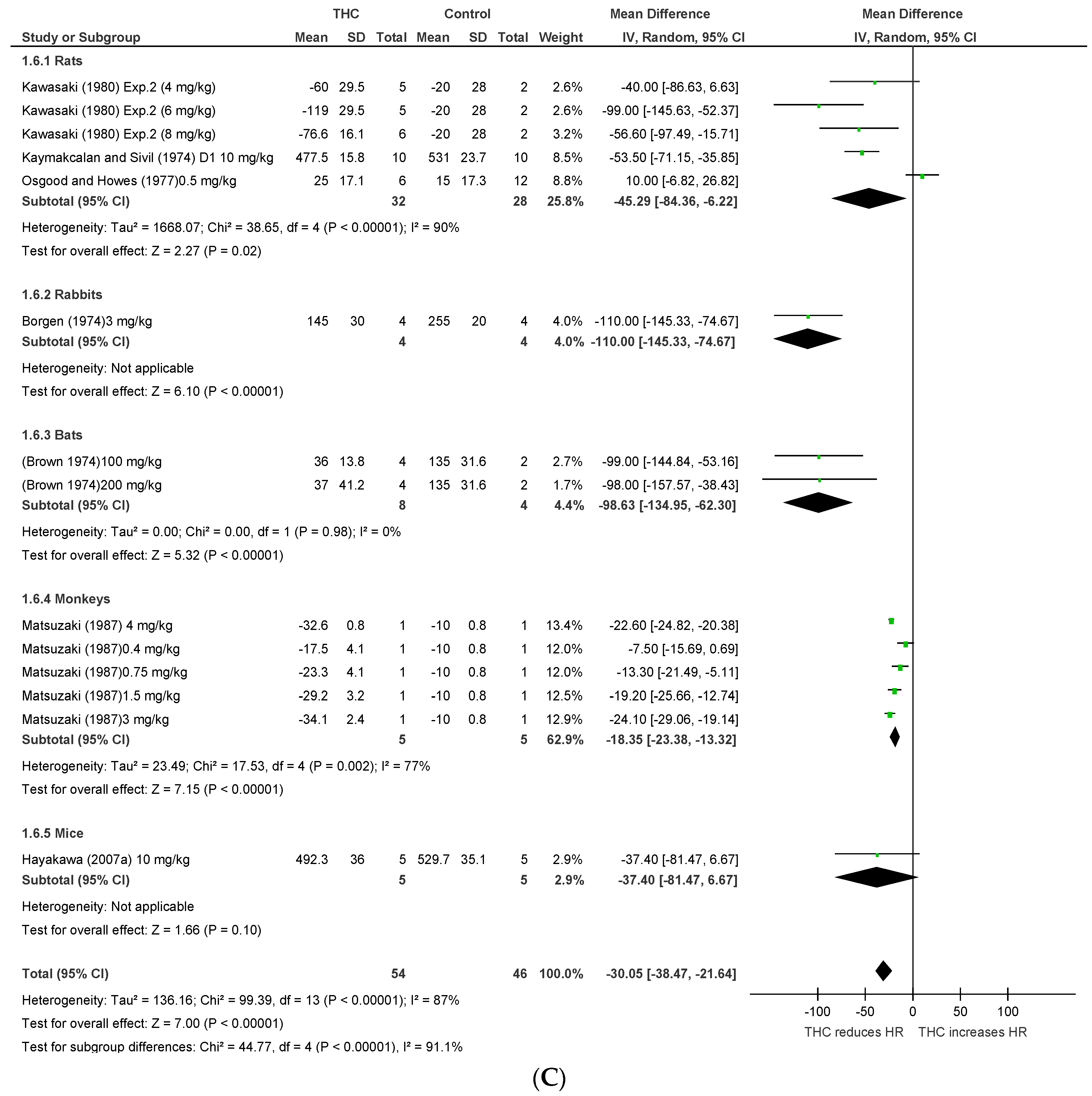
2.1.2. Conscious Animals
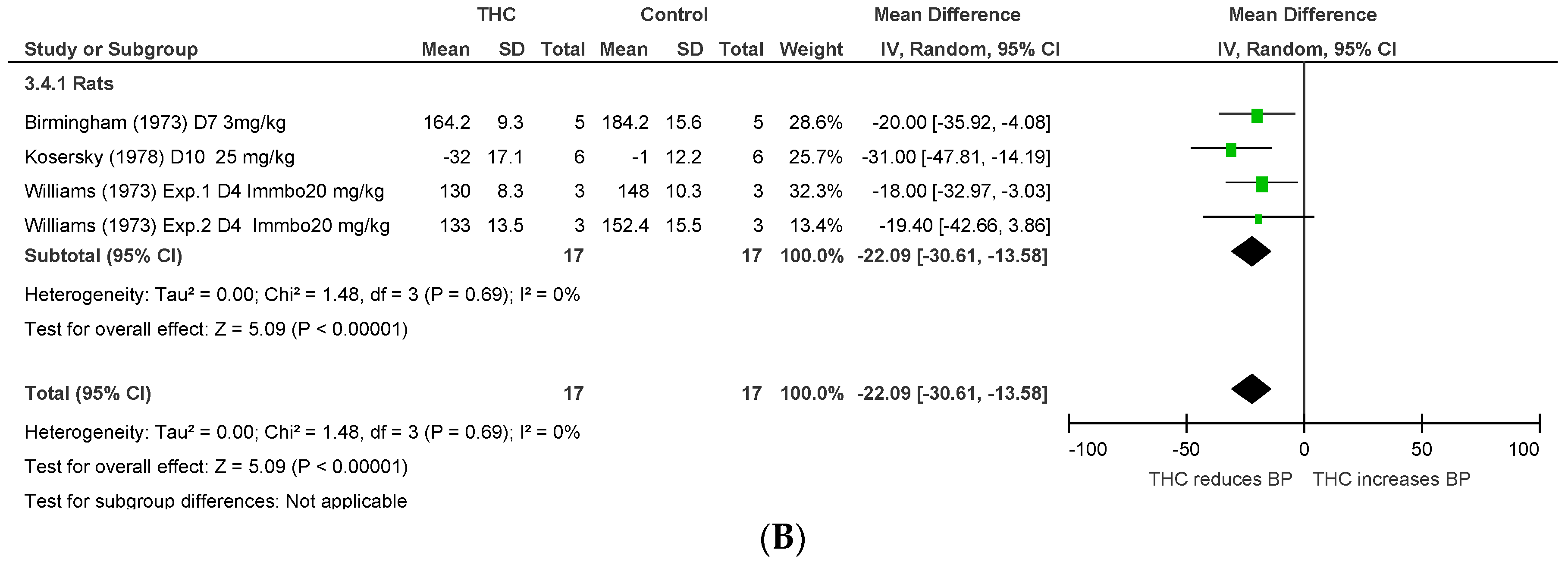
2.1.3. Conscious Animal Models of Stress or Hypertension
2.1.4. Human Studies
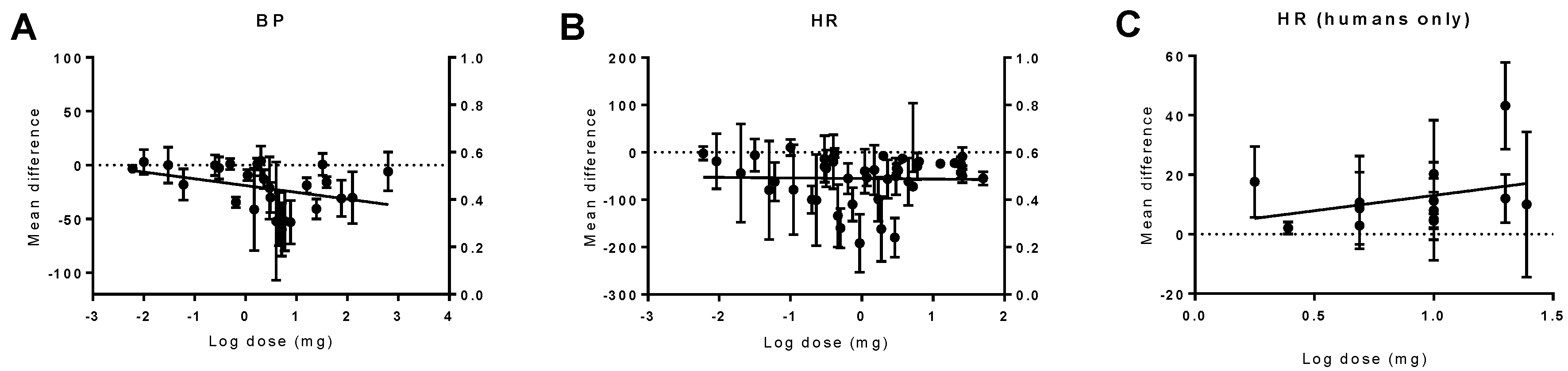
2.2. Dose–Response to THC
2.3. Quality
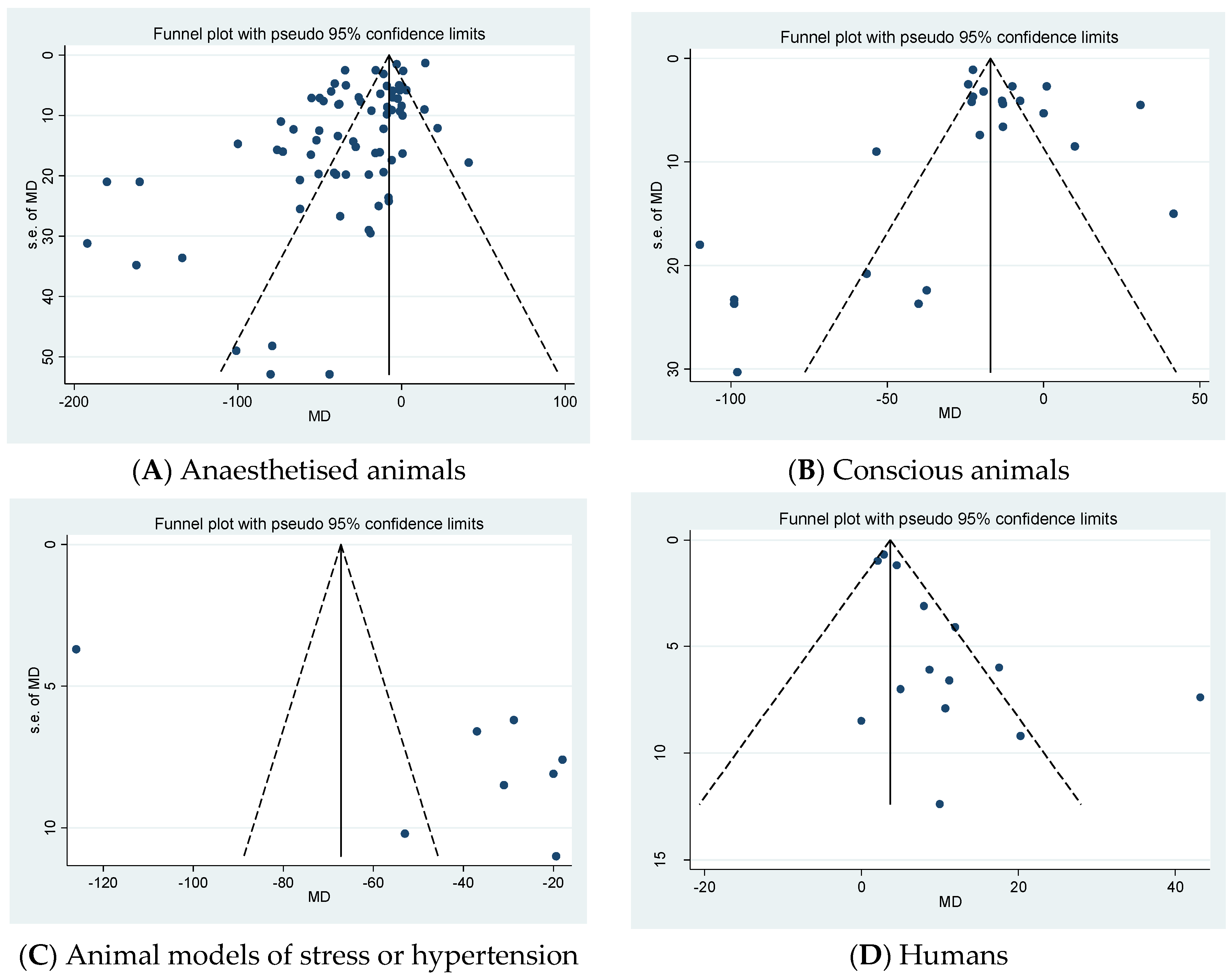
2.4. Publication Bias
3. Discussion
4. Materials and Methods
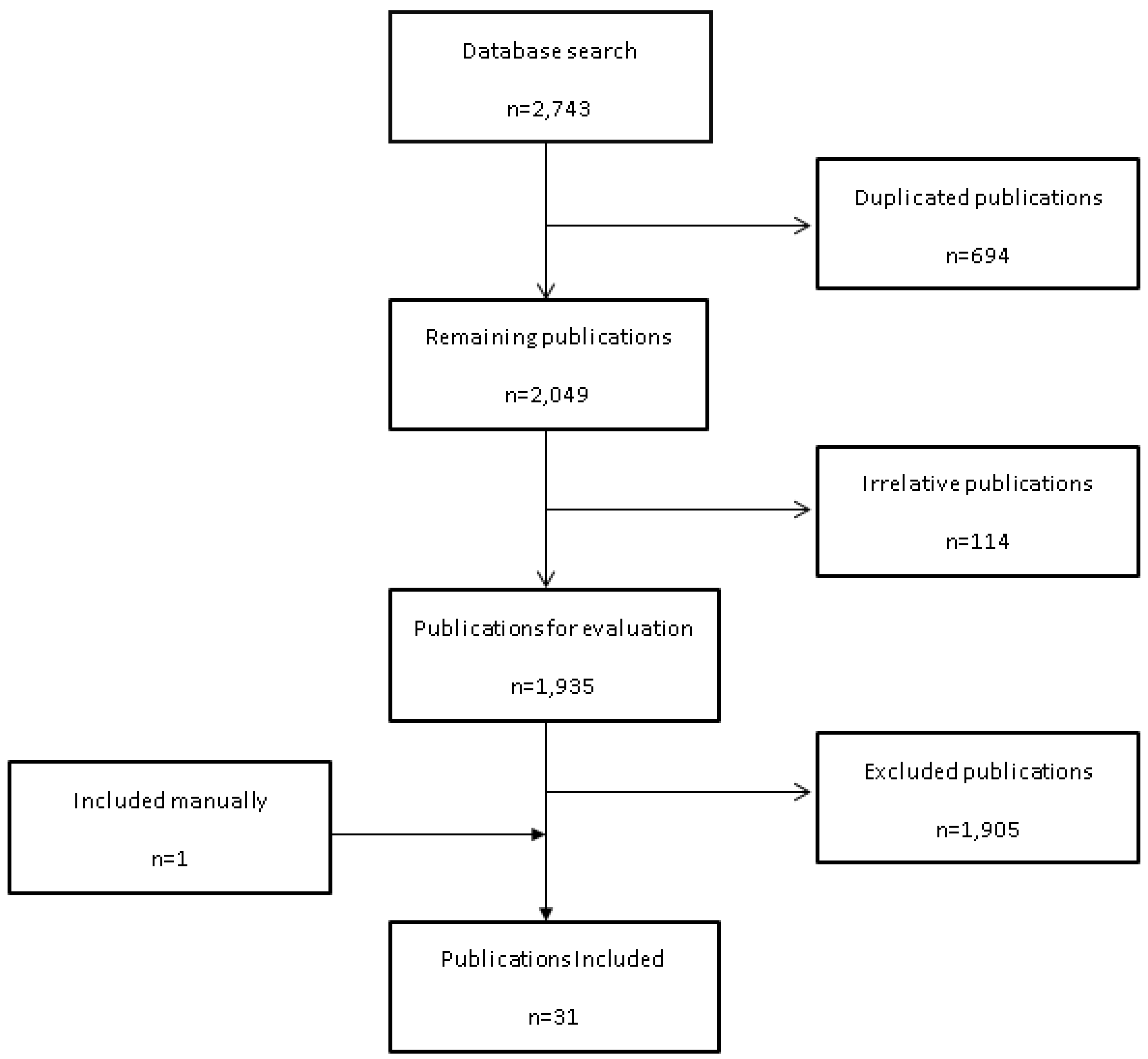
4.1. Search Strategy
4.2. Data Acquisition
4.3. Quality
4.4. Data Analysis
Author Contributions
Conflicts of Interest
Appendix A
References
- Gaoni, Y.; Mechoulam, R. Isolation, Structure, and Partial Synthesis of an Active Constituent of Hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647. [Google Scholar] [CrossRef]
- Pertwee, R.G. Pharmacological actions of cannabinoids. Handb. Exp. Pharmacol. 2005, 20, 1–51. [Google Scholar]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D.; Page, J.; Dunn, E.; Bradshaw, H.B. Delta(9) -Tetrahydrocannabinol and N-arachidonyl glycine are full agonists at GPR18 receptors and induce migration in human endometrial HEC-1B cells. Br. J. Pharmacol. 2012, 165, 2414–2424. [Google Scholar] [CrossRef] [PubMed]
- Ben Amar, M. Cannabinoids in medicine: A review of their therapeutic potential. J. Ethnopharmacol. 2006, 105, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Lastres-Becker, I.; Molina-Holgado, F.; Ramos, J.A.; Mechoulam, R.; Fernandez-Ruiz, J. Cannabinoids provide neuroprotection against 6-hydroxydopamine toxicity in vivo and in vitro: Relevance to Parkinson’s disease. Neurobiol. Dis. 2005, 19, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Buccellato, E.; Carretta, D.; Utan, A.; Cavina, C.; Speroni, E.; Grassi, G.; Candeletti, S.; Romualdi, P. Acute and chronic cannabinoid extracts administration affects motor function in a CREAE model of multiple sclerosis. J Ethnopharmacol. 2011, 133, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for Medical Use: A Systematic Review and Meta-analysis. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef] [PubMed]
- Todaro, B. Cannabinoids in the treatment of chemotherapy-induced nausea and vomiting. J. Natl. Compr. Cancer Netw. 2012, 10, 487–492. [Google Scholar] [CrossRef]
- Abrams, D.I.; Guzman, M. Cannabis in cancer care. Clin. Pharmacol. Ther. 2015, 97, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Fleming, I.; Schermer, B.; Popp, R.; Busse, R. Inhibition of the production of endothelium-derived hyperpolarizing factor by cannabinoid receptor agonists. Br. J. Pharmacol. 1999, 126, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Zygmunt, P.M.; Andersson, D.A.; Högestätt, E.D. Δ9Tetrahydrocannabinol and Cannabinol Activate Capsaicin-Sensitive Sensory Nerves via a CB1 and CB2 Cannabinoid Receptor-Independent Mechanism. J. Neurosci. 2002, 22, 4720–4727. [Google Scholar] [PubMed]
- O’Sullivan, S.E.; Kendall, D.A.; Randall, M.D. The effects of Δ(9)-tetrahydrocannabinol in rat mesenteric vasculature, and its interactions with the endocannabinoid anandamide. Br. J. Pharmacol. 2005, 145, 514–526. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.E.; Kendall, D.A.; Randall, M.D. Vascular effects of delta 9-tetrahydrocannabinol (THC), anandamide and N-arachidonoyldopamine (NADA) in the rat isolated aorta. Eur. J. Pharmacol 2005, 507, 211–221. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.E.; Tarling, E.J.; Bennett, A.J.; Kendall, D.A.; Randall, M.D. Novel time-dependent vascular actions of Delta9-tetrahydrocannabinol mediated by peroxisome proliferator-activated receptor gamma. Biochem. Biophys. Res. Commun. 2005, 337, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Kaymakcalan, S.; Turker, R.K. The evidence of the release of prostaglandin-like material from rabbit kidney and guinea-pig lung by (minus)-trans-delta9-tetrahydrocannabinol. J. Pharm. Pharmacol. 1975, 27, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.; Kendall, D.A.; Ralevic, V. Characterization of cannabinoid modulation of sensory neurotransmission in the rat isolated mesenteric arterial bed. J. Pharmacol. Exp. Ther. 2004, 311, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, P.P.; Lapa, A.J.; Lima-Landman, M.T.; Valle, J.R. Vasoconstriction induced by delta 9-tetrahydrocannabinol on the perfused rabbit ear artery. Arch. Int. Pharmacodyn. Ther. 1981, 252, 253–261. [Google Scholar] [PubMed]
- Cavero, I.; Lokhandwala, M.F.; Buckley, J.P.; Jandhyala, B.S. The effect of (−)-Δ9-trans-tetrahydrocannabinol on myocardial contractility and venous return in anesthetized dogs. Eur. J. Pharmacol. 1974, 29, 74–82. [Google Scholar] [CrossRef]
- Brown, D.J.; Miller, F.N.; Longnecker, D.E.; Greenwald, E.K.; Harris, P.D.; Forney, R.B. The influence of delta 9-tetrahydrocannabinol on cardiovascular and subcutaneous microcirculatory systems in the bat. J. Pharmacol. Exp. Ther. 1974, 188, 624–629. [Google Scholar] [PubMed]
- Beaumont, H.; Jensen, J.; Carlsson, A.; Ruth, M.; Lehmann, A.; Boeckxstaens, G. Effect of delta9-tetrahydrocannabinol, a cannabinoid receptor agonist, on the triggering of transient lower oesophageal sphincter relaxations in dogs and humans. Br. J. Pharmacol. 2009, 156, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Osgood, P.F.; Howes, J.F. Δ9-tetrahydrocannabinol and dimethylheptylpyran induced tachycardia in the conscious rat. Life Sci. 1977, 21, 1329–1335. [Google Scholar] [CrossRef]
- Kawasaki, H.; Watanabe, S.; Oishi, R.; Ueki, S. Effects of delta-9-tetrahydrocannabinol on the cardiovascular system, and pressor and behavioral responses to brain stimulation in rats. Jpn. J. Pharmacol. 1980, 30, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, S.W.; Lapa, A.J.; Ribeiro do Valle, J. The triple effect induced by delta 9-tetrahydrocannabinol on the rat blood pressure. Eur. J. Pharmacol. 1979, 58, 351–357. [Google Scholar] [CrossRef]
- Cavero, I.; Ertel, R.; Buckley, J.P.; Jandhyala, B.S. Effects of (−)-Δ9-trans-tetrahydrocannabinol on regional blood flow in anesthetized dogs. Eur. J. Pharmacol. 1972, 20, 373–376. [Google Scholar] [CrossRef]
- Cavero, I.; Buckley, J.P.; Jandhyala, B.S. Hemodynamic and myocardial effects of (−)-Δ9-trans-tetrahydrocannabinol in anesthetized dogs. Eur. J. Pharmacol. 1973, 24, 243–251. [Google Scholar] [CrossRef]
- Cavero, I.; Solomon, T.; Buckley, J.P.; Jandhyala, B.S. Studies on the bradycardia induced by (−)-delta9-trans-tetrahydrocannabinol in anesthetized dogs. Eur. J. Pharmacol. 1973, 22, 263–269. [Google Scholar] [CrossRef]
- Daskalopoulos, N.; Schmitt, H.; Laubie, M. Action of delta 9 tetrahydrocannabinol on the central cardiovascular regulation: Mechanism and localization. Lencephale 1975, 1, 121–132. [Google Scholar]
- Adams, M.D.; Earnhardt, J.T.; Dewey, W.L.; Harris, L.S. Vasoconstrictor actions of delta8- and delta9-tetrahydrocannabinol in the rat. J. Pharmacol. Exp. Ther. 1976, 196, 649–656. [Google Scholar] [PubMed]
- Jandhyala, B.S.; Malloy, K.P.; Buckley, J.P. Effects of chronic administration of delta9-tetrahydrocannabinol on the heart rate of mongrel dogs. Res. Commun. Chem. Pathol. Pharmacol. 1976, 14, 201–204. [Google Scholar] [PubMed]
- Jandhyala, B.S.; Buckley, J.P. Autonomic and cardiovascular effects of chronic delta9-tetrahydrocannabinol administration in mongrel dogs. Res. Commun. Chem. Pathol. Pharmacol. 1977, 16, 593–607. [Google Scholar] [PubMed]
- Jandhyala, B.S. Effects of prolonged administration of delta 9-tetrahydrocannabinol on the autonomic and cardiovascular function and regional hemodynamics in mongrel dogs. Res. Commun. Chem. Pathol. Pharmacol. 1978, 20, 489–508. [Google Scholar] [PubMed]
- McConnell, W.R.; Dewey, W.L.; Harris, L.S.; Borzelleca, J.F. A study of the effect of delta 9-tetrahydrocannabinol (delta 9-THC) on mammalian salivary flow. J. Pharmacol. Exp. Ther. 1978, 206, 567–573. [Google Scholar] [PubMed]
- Schmeling, W.T.; Hosko, M.J.; Hardman, H.F. Potentials evoked in the intermediolateral column by hypothalamic stimulation—Suppression by Δ9-tetrahydrocannabinol. Life Sci. 1981, 29, 673–680. [Google Scholar] [CrossRef]
- Estrada, U.; Brase, D.A.; Martin, B.R.; Dewey, W.L. Cardiovascular effects of delta 9- and delta 9(11)-tetrahydrocannabinol and their interaction with epinephrine. Life Sci. 1987, 41, 79–87. [Google Scholar] [CrossRef]
- Krowicki, Z.K.; Moerschbaecher, J.M.; Winsauer, P.J.; Digavalli, S.V.; Hornby, P.J. Delta9-tetrahydrocannabinol inhibits gastric motility in the rat through cannabinoid CB1 receptors. Eur. J. Pharmacol. 1999, 371, 187–196. [Google Scholar] [CrossRef]
- Kaymakcalan, S.; Sivil, S. Lack of tolerance to the bradycardic effect of delta 9-trans-tetrahydrocannabinol in rats. Pharmacology 1974, 12, 290–295. [Google Scholar] [PubMed]
- Borgen, L.A.; Davis, W.M. Cannabidiol interaction with delta9-tetrahydrocannabinol. Res. Commun. Chem. Pathol. Pharmacol. 1974, 7, 663–670. [Google Scholar] [PubMed]
- Matsuzaki, M.; Casella, G.A.; Ratner, M. delta 9-Tetrahydrocannabinol: EEG changes, bradycardia and hypothermia in the rhesus monkey. Brain Res. Bull. 1987, 19, 223–229. [Google Scholar] [CrossRef]
- Hayakawa, K.; Mishima, K.; Nozako, M.; Hazekawa, M.; Irie, K.; Fujioka, M.; Orito, K.; Abe, K.; Hasebe, N.; Egashira, N.; et al. Delayed treatment with cannabidiol has a cerebroprotective action via a cannabinoid receptor-independent myeloperoxidase-inhibiting mechanism. J. Neurochem. 2007, 102, 1488–1496. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Mishima, K.; Nozako, M.; Ogata, A.; Hazekawa, M.; Liu, A.X.; Fujioka, M.; Abe, K.; Hasebe, N.; Egashira, N.; et al. Repeated treatment with cannabidiol but not Delta9-tetrahydrocannabinol has a neuroprotective effect without the development of tolerance. Neuropharmacology 2007, 52, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.B.; Ng, L.K.Y.; Lamprecht, F.; Roth, K.; Kopin, I.J. Δ9-Tetrahydrocannabinol: A hypotensive effect in rats. Psychopharmacologia 1973, 28, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Birmingham, M.K. Reduction by 9-tetrahydrocannabinol in the blood pressure of hypertensive rats bearing regenerated adrenal glands. Br. J. Pharmacol. 1973, 48, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Kosersky, D.S. Antihypertensive effects of delta9-tetrahydrocannabinol. Arch. Int. Pharmacodyn. Ther. 1978, 233, 76–81. [Google Scholar] [PubMed]
- Karniol, I.G.; Carlini, E.A. Comparative Studies in Man and in Laboratory Animals on Δ8-and Δ9-trans-Tetrahydrocannabinol. Pharmacology 1973, 9, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Karniol, I.G.; Shirakawa, I.; Takahashi, R.N.; Knobel, E.; Musty, R.E. Effects of delta9-tetrahydrocannabinol and cannabinol in man. Pharmacology 1975, 13, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, B.D.; Bickel, P.; Dittrich, A. Changes of simple somatic parameters by delta-9-trans-tetrahydrocannabinol (delta-9-THC) in a double-blind-study. Short communication. Arzneimittelforschung 1976, 26, 1614–1616. [Google Scholar] [PubMed]
- Haney, M. Opioid antagonism of cannabinoid effects: Differences between marijuana smokers and nonmarijuana smokers. Neuropsychopharmacology 2007, 32, 1391–1403. [Google Scholar] [CrossRef] [PubMed]
- Klooker, T.K.; Leliefeld, K.E.; Van Den Wijngaard, R.M.; Boeckxstaens, G.E. The cannabinoid receptor agonist delta-9-tetrahydrocannabinol does not affect visceral sensitivity to rectal distension in healthy volunteers and IBS patients. Neurogastroenterol. Motil. 2011, 23, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Lake, K.D.; Compton, D.R.; Varga, K.; Martin, B.R.; Kunos, G. Cannabinoid-induced hypotension and bradycardia in rats mediated by CB1-like cannabinoid receptors. J. Pharmacol. Exp. Ther. 1997, 281, 1030–1037. [Google Scholar] [PubMed]
- Strougo, A.; Zuurman, L.; Roy, C.; Pinquier, J.L.; van Gerven, J.M.; Cohen, A.F.; Schoemaker, R.C. Modelling of the concentration—Effect relationship of THC on central nervous system parameters and heart rate—Insight into its mechanisms of action and a tool for clinical research and development of cannabinoids. J. Psychopharmacol. 2008, 22, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Schwope, D.M.; Bosker, W.M.; Ramaekers, J.G.; Gorelick, D.A.; Huestis, M.A. Psychomotor performance, subjective and physiological effects and whole blood Delta(9)-tetrahydrocannabinol concentrations in heavy, chronic cannabis smokers following acute smoked cannabis. J. Anal. Toxicol. 2012, 36, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.J.; Wilson, W.H.; Humphreys, D.; Lowe, J.V.; Wiethe, K.E. Middle cerebral artery velocity during upright posture after marijuana smoking. Acta Psychiatr. Scand. 1992, 86, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.J.; Wilson, W.H.; Humphreys, D.F.; Lowe, J.V.; Wiethe, K.E. Changes in middle cerebral artery velocity after marijuana. Biol. Psychiatry 1992, 32, 164–169. [Google Scholar] [CrossRef]
- Klumpers, L.E.; Roy, C.; Ferron, G.; Turpault, S.; Poitiers, F.; Pinquier, J.L.; van Hasselt, J.G.; Zuurman, L.; Erwich, F.A.; van Gerven, J.M. Surinabant, a selective cannabinoid receptor type 1 antagonist, inhibits Delta9-tetrahydrocannabinol-induced central nervous system and heart rate effects in humans. Br. J. Clin. Pharmacol. 2013, 76, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Morrison, P.D.; Fusar-Poli, P.; Martin-Santos, R.; Borgwardt, S.; Winton-Brown, T.; Nosarti, C.; CM, O.C.; Seal, M.; Allen, P.; et al. Opposite effects of delta-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacology 2010, 35, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Niesink, R.J.; van Laar, M.W. Does Cannabidiol Protect Against Adverse Psychological Effects of THC? Front. Psychiatry 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Karniol, I.G.; Shirakawa, I.; Kasinski, N.; Pfeferman, A.; Carlini, E.A. Cannabidiol interferes with the effects of delta 9-tetrahydrocannabinol in man. Eur. J. Pharmacol. 1974, 28, 172–177. [Google Scholar] [CrossRef]
- Stroke Therapy Academic Industry Roundtable. Recommendations for standards regarding preclinical neuroprotective and restorative drug development. Stroke 1999, 30, 2752–2758. [Google Scholar]
- England, T.J.; Hind, W.H.; Rasid, N.A.; O’Sullivan, S.E. Cannabinoids in experimental stroke: A systematic review and meta-analysis. J. Cereb. Blood Flow Metab. 2015, 35, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Sultan, S.R.; Millar, S.A.; England, T.J.; O’Sullivan, S.E. A Systematic Review and Meta-Analysis of the Haemodynamic Effects of Cannabidiol. Front. Pharmacol. 2017, 8, 81. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Neukirchen, M.; Kienbaum, P. Sympathetic nervous system: Evaluation and importance for clinical general anesthesia. Anesthesiology 2008, 109, 1113–1131. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]




| Author & Year | Study Description | Species, Model (Anaesthetic & Route) | Sample Size | THC Dose | THC Route | Time of THC Administration | Time of Haemodynamic Measurements | Basal Parameters * | Outcomes and Comments |
|---|---|---|---|---|---|---|---|---|---|
| Anaesthetised animals | |||||||||
| Cavero 1972 [25] | Investigate the haemodynamic effects of THC | Dogs Anaesthetised (pentobarbital, iv) | 11 | 2.5 mg/kg | i.v. | Post-anaesthesia | Continues for 30 m post-drug | - | THC altered distribution of regional BF, and reduced HR and BP. |
| Cavero 1973a [26] | Investigate the haemodynamic effects of THC | Dogs Anaesthetised (pentobarbital, iv) | 23 | 39 µg/kg–2.5 mg/kg | i.v. | Post-anaesthesia | Continues for 2 h post-drug | C: HR:169, BP:91.7; T: HR:165.7, BP:93.5 | THC caused reduction in HR and BP mediated via central nervous system. |
| Cavero 1973b [27] | Characterise the mechanism of action of THC on HR | Dogs Anaesthetised (pentobarbital, iv) | 29 | 39 µg/kg–5 mg/kg | i.v. | Post-anaesthesia | Continues for 140 m post-drug | - | THC induced reduction in HR through alteration of autonomic innervation to myocardium. |
| Cavero 1974 [19] | Investigate the effect of THC on venous return | Dogs (heart bypass) Anaesthetised (dibucaine, spinal) | 8 | 2.5 mg/kg | i.v. | Post-anaesthesia | Pre-drug and continues for 30 m post-drug | C: HR:156, BP:85.8; T: HR:147, BP:85. | THC caused reduction in HR and BP, and reduced venous return. |
| Daskalopoulos 1975 [28] | Investigate the mechanism of THC on CV system | Cats Anaesthetised (urethane, iv) | 40 | 30–300 µg/kg | i.v. | Post-anaesthesia | 20 m post-drug | - | THC reduced HR and BP mediated via central nervous system. |
| Adams 1976 [29] | Examined the CV effects of THC | Rats Anaesthetised (urethane, ip) | 72 | 0.1–3 mg/kg | i.v. | Post-anaesthesia | Continues for 30 min post-drug | C: HR:316.2, BP:76.2; T: HR:314.8, BP:73.5. | THC caused reduction in HR and biphasic BP response (↑ BP followed by ↓ BP), suggesting that THC depressed CV reflex functions. |
| Jandhyala 1976 [30] | Evaluated possible interaction with THC on HR | Dogs Anaesthetised (pentobarbital) | 12 | 1 mg/kg | s.c. | Twice/day for 7 days Pre-anaesthesia | On the 7th day post-anaesthesia | - | Chronic THC antagonised the elevation in HR induced by the anaesthetic agent via vagal stimulation. |
| Jandhyala 1977 [31] | Determined chronic administration of THC on CV function | Dogs Anaesthetised (pentobarbital) | 16 | 1 mg/kg | s.c. | Twice/day for 7 days Pre-anaesthesia | On the 7th day post-anaesthesia | - | Chronic THC had no effect on haemodynamics. |
| Jandhyala 1978 [32] | Investigated prolonged THC effects on CV system | Dogs Anaesthetised (pentobarbital) | 16 | 2 mg/kg | s.c. | Single dose per day for 35 days | On the 35th day post-anaesthesia | - | Chronic THC increased BF in femoral and mesenteric arteries with no effect on HR or BP. |
| McConnell 1978 [33] | Examined the effects of THC on salivary flow | Cats Anaesthetised (urethane & pentobarbital, ip) | 20 | 0.1–2 mg/kg | i.v. | Post-anaesthesia | Continues for 1 h post-drug | - | THC had no effect in stimulated salivary flow of cats. THC caused a reduction in HR and BP. |
| Siqueira 1979 [24] | Clarify the triple BP response post-THC | Rats Anaesthetised (urethane, ip) | 50 | 1–10 mg/kg | i.v. | Post-anaesthesia | Continues for 70 m post-drug | - | THC induced triphasic BP response (↓ BP via vagal stimulation, then ↑ BP not dependent on sympathetic activity followed by ↓ BP due to central decrease in sympathetic tone). |
| Kawasaki 1980 [23] | Investigated the effect of THC on the CV system and behavior changes | Rats Anaesthetised (urethane, ip) | 29 | 1–5 mg/kg | i.v. | Post-anaesthesia | Continues for 70 m post-drug | - | THC induced CV effects (↓ HR and ↑ BP) through vagal activity, and influence behavior changes to brain stimulation. |
| Schmeling 1981 [34] | Investigated the effect of THC on hypothalamus | Cats Anaesthetised (urethane, ip) | 12 | 2 mg/kg | i.v. | Post-anaesthesia | Continues for 30 m post-drug | - | THC produced significant reductions in HR and BP and attenuated the pressor response threshold suggesting that THC reduces sympathetic activity. |
| Estrada 1987 [35] | Investigated the CV effects of THC | Rats Anaesthetised (pentobarbital, ip) | 28 | 0.078–5 mg/kg | i.v. | Post-anaesthesia | 3-12 min post-drug | - | THC produced adverse effects on the CV system (↓ HR and ↓ BP) |
| Krowicki 1999 [36] | Investigated whether CB1 activation by THC inhibits gastric motor function | Rats Anaesthetised (ketamine and xylazine) | 36 | 0.02–2 mg/kg | i.v. | Post-anaesthesia | Continues for 10 m post-drug | - | THC decreased gastric motor function, HR, and BP via autonomic effects mediated by CB1. |
| Conscious animals | |||||||||
| Kaymakcalan 1974 [37] | Investigated chronic effects of THC on HR | Rats Conscious | 20 | 10 mg/kg | s.c. | Single dose per day for 16 days | Hourly interval to 6 h on the 1st, 4th, 8th and 16th days | - | THC produced marked reduction in HR |
| Borgen 1974 [38] | Examined possible interaction of CBD on THC effects | Rabbits Conscious | 8 | 3 mg/kg | i.v. | Pre-test | Pre-drug and hourly interval to 7 h post-drug | C: HR:264; T: HR:276 | CBD reduced the hypothermic effect of THC and attenuated the depressant effects of THC on respiration, rectal temperature and HR |
| Brown 1974 [20] | Investigated CV response to THC | Bats Conscious | 12 | 100 and 200 mg/kg | i.p. | Pre-test | Pre-drug and continues for 145 m post-drug | C: HR:436, BP:101; T: HR:390, BP:114 | THC induced hypothermia and reduction in HR and BP. |
| Osgood 1977 [22] | Investigated THC effects on HR | Rats Conscious | 18 | 0.5 mg/kg | i.p. | Pre-test | Continues for 30 m post-drug | - | THC had minimal effect on BP and caused an increase in HR, which may be related to central mediation release of epinephrine from adrenal gland. |
| Kawasaki 1980 [23] | Investigated the effects of THC on the CV system and behavior changes | Rats Conscious | 21 | 4–8 mg/kg | i.p. | Pre-test | Continues for 2 h post-drug | - | THC induced CV effects (↓ HR and ↑ BP) through vagal activity, and influenced behavior changes to brain stimulation. |
| Matsuzaki 1987 [39] | Examined the effects of THC on EEG, body temperature, and HR | Monkeys Conscious | 6 | 0.4–4 mg/kg | i.p. | Pre-test | Continues for 5 h post-drug | - | THC induced reduction in HR and hypothermia and induced responses of EGG along with behavioral depression and alertness. |
| Hayakawa 2007a [40] | Investigated CBD and THC effects on ischemic brain damage | Stroke Mice Conscious | 17 | 10 mg/kg | i.p. | Pre-, 3 and 4 h post-occlusion, and 1 and 2 h post-reperfusion | BP and HR: pre-reperfusion. CBF: continued 4 h post-occlusion and 1 post-reperfusion | - | Pre and post-ischemic treatment with CBD induced neuroprotection, whereas only preischemic treatment with THC induced neuroprotection. THC increased CBF with no effects on BP or HR |
| Hayakawa 2007b [41] | Explored the development of tolerance of THC and CBD neuroprotection | Stroke Mice Conscious | 7 | 10 mg/kg | i.p. | Pre-occlusion and 3 h post-occlusion. Single dose per day for 14 days | During 4 h and on day 14 post-occlusion | - | Repeated treatment with CBD, but not THC, induced neuroprotection with development of tolerance. THC increased CBF on day 1 only with no effects on BP or HR. |
| Stress and hypertensive animal models | |||||||||
| Williams 1973 [42] | Studied the effects of THC on BP | Rats Stress | 30 | 20 mg/kg | s.c. | Single dose per day for 4 days | Pre-drug, 4 h, 48 and 96 h post-drug | C: BP:128; T: BP:129 | THC reduced BP |
| Birmingham 1973 [43] | Studies the effects of THC on BP | Rats Hypertensive | 10 | 3 mg/kg | i.p. | Single dose per day for 7 days | Hourly to 5 h for 7 days | - | THC reduced BP |
| Kosersky 1978 [44] | Examined the antihypertensive effects of THC | Rats Hypertensive | 12 | 25 mg/kg | Oral | Single dose per day for 10 days | 4 h and every day for 14 days post-drug | - | THC effectively reduced BP to the same degree over the treatment period. |
| Humans | |||||||||
| Karniol 1973 [45] | Compared the effects of 8-THC and 9-THC | Human Healthy | 21 | 5–20 mg | Inhale | Pre-test | Avrg. of 20 m post-drug | C: HR:82; T: HR:85 | 9-THC was twice as active as 8-THC in increasing HR and caused more subjective symptoms. |
| Karniol 1975 [46] | Examined the interaction between THC and CBN | Human Healthy | 5 (M) | 25 mg | Oral | Pre-test | 50, 70 and 160 m post-drug | - | THC induced increase in HR and psychological effects. No change on THC effects when combined with CBN |
| Zimmer 1976 [47] | Examined changes of somatic parameters post-THC | Human Healthy | 36 | 250 µg/kg | Oral | Pre-test | Pre-drug and 4 h post-drug | C: HR:87.9, BP:127.5; T: HR:89, BP:123 | THC raised HR with no changes on other parameters including BP |
| Haney 2007 [48] | Determined the effects of naltrexone in combination with THC | Human Healthy | 21 (11 M & 10 F) | 2.5–10 mg | Oral | Pre-test | Continues for 6 h post-drug | - | Naltrexone enhanced intoxication effects of THC; THC increased HR |
| Beaumont 2009 [21] | Evaluated whether THC has inhibitory effect on transient esophageal sphincter | Human Healthy | 18 (M) | 10 and 20 mg | Oral | Pre-test | Continues for 4 h post-drug | C: HR:59; T: HR:59 | THC inhibited the increased induced meal transient esophageal sphincter relaxation. THC increased HR and decreased BP |
| Klooker 2011 [49] | Assessed the effect of THC on rectal sensation | Human Healthy and IBD | 10 and 12 | 5 and 10 mg | Oral | Pre-test | Continues for 105 m post-drug | - | THC had no effect on rectal perception to distension. THC increased HR with no effect on BP |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sultan, S.R.; Millar, S.A.; O’Sullivan, S.E.; England, T.J. A Systematic Review and Meta-Analysis of the In Vivo Haemodynamic Effects of Δ9-Tetrahydrocannabinol. Pharmaceuticals 2018, 11, 13. https://doi.org/10.3390/ph11010013
Sultan SR, Millar SA, O’Sullivan SE, England TJ. A Systematic Review and Meta-Analysis of the In Vivo Haemodynamic Effects of Δ9-Tetrahydrocannabinol. Pharmaceuticals. 2018; 11(1):13. https://doi.org/10.3390/ph11010013
Chicago/Turabian StyleSultan, Salahaden R., Sophie A. Millar, Saoirse E. O’Sullivan, and Timothy J. England. 2018. "A Systematic Review and Meta-Analysis of the In Vivo Haemodynamic Effects of Δ9-Tetrahydrocannabinol" Pharmaceuticals 11, no. 1: 13. https://doi.org/10.3390/ph11010013
APA StyleSultan, S. R., Millar, S. A., O’Sullivan, S. E., & England, T. J. (2018). A Systematic Review and Meta-Analysis of the In Vivo Haemodynamic Effects of Δ9-Tetrahydrocannabinol. Pharmaceuticals, 11(1), 13. https://doi.org/10.3390/ph11010013





