1. Introduction
Micro-Opto-Electro-Mechanical Systems (MOEMS) based biosensor devices [
1] are optoelectronic detection devices that use chemical and biological species for detection and quantification of targets of interest based upon the presence, activity or the concentration of the specimen. The advent of microfluidics and its integration into MOEMS has given rise to several biosensing techniques, the prominent of them being fluorescence based detection [
2] due to various reasons such as high sensitivity, advantages with non-invasive sensing, possibility to carry out time resolved study etc. [
3].
In-situ medical diagnosis and Point-of-Care (POC) testing necessitate the development of fully integrated Lab-on-a-chip type devices for several practical and readily available biosensing applications and biodetection of protein-protein interactions, antigen-antibody non-covalent binding, DNA sequencing etc. Microfluidics is an essential component of a biosensor unit because of the inherent advantages, such as smaller reagent volume handling, transportation and the means of introducing the biological element into the biosensor system in a controlled manner. Thus, the key to the success of synthesizing a Micro-Total-Analysis System (μTAS) [
4] or a Lab-on-a-chip based biosensor lies in the optimum integration of microfluidics with other complimentary modules such as micro-electrical, micro-thermal and microphotonic elements.
One of the important factors which affect microfluidics based biosensing is the problem of immobilization of the biomolecules onto the surface of scrutiny. In general, immobilization is the technique used for the physical or chemical fixation of cells, organelles, enzymes, or other proteins (e.g. monoclonal antibodies) onto a solid support, in order to increase their stability and make possible their repeated or continued use [
5]. Several immobilization techniques have been reported in the literature for binding different biological molecules onto the surface [
6-
10]. However, selection of a suitable material and method that facilitate biomolecule immobilization with the microfluidic channel surface is essential for the success of Lab-on-a-chip.
In the present work, a microfluidic chip has been fabricated on silicon platform and hybrid integrated with a spectrometer-on-chip for fluorescence based biosensing. Silicon [
11] is one of the popular substrates used for fabrication of microfluidic channels as it is very common material used in MEMS industry due to its compatibility with Complimentary Metal Oxide Semiconductor (CMOS) technology. Many works have been carried out in fabrication of silicon based microfluidic chips [
12-
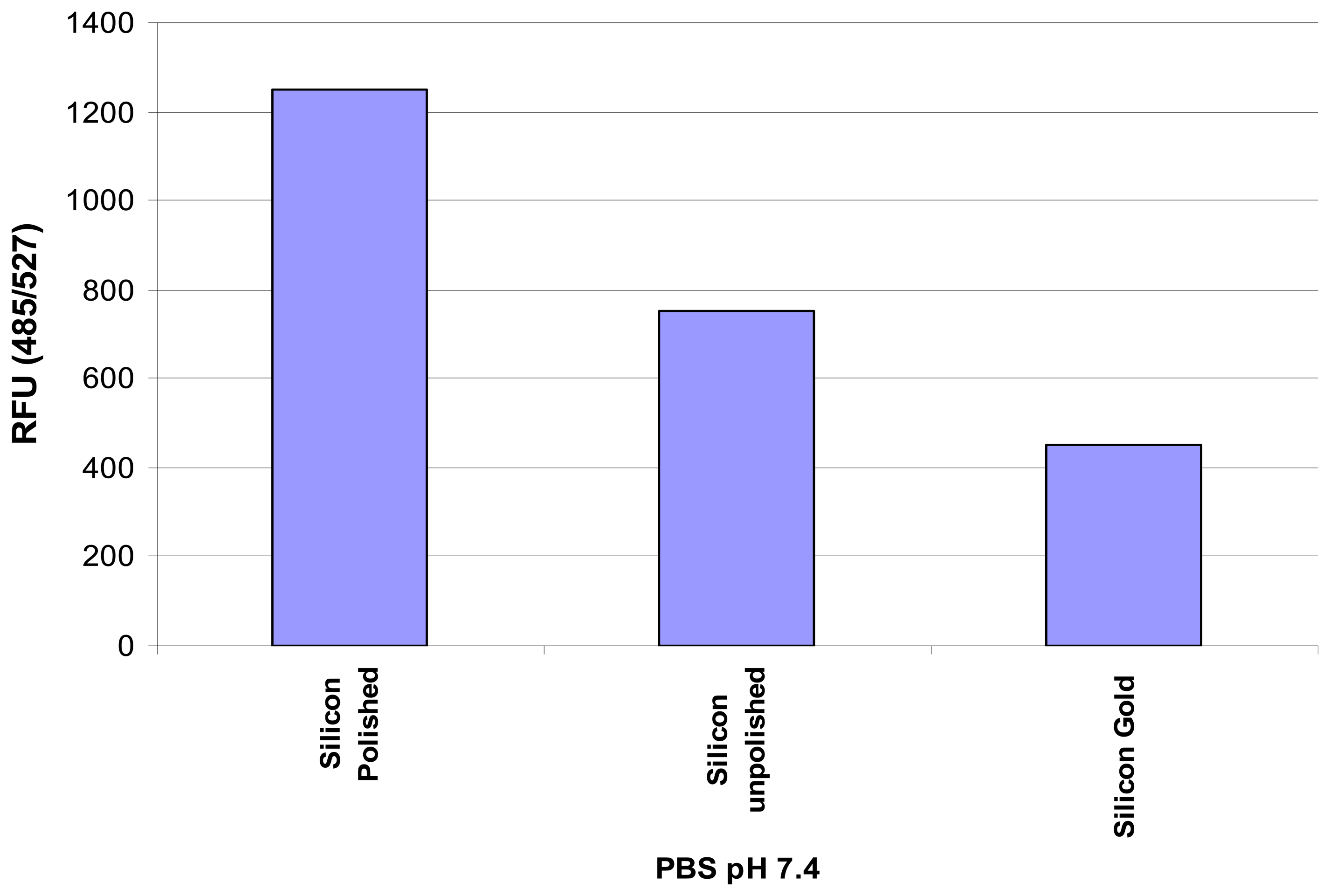
14]. But, in order to verify the compatibility of silicon for the present biological application, preliminary evaluation of the binding capacity of immune molecules to different silicon based microfluidic supports were carried out. Three different solid silicon supports, namely, unpolished, polished, and gold coated, were taken and the capacity of antibodies to bind to these surfaces was analyzed. Fluorescent (FITC labeled) antibody suspended in pH 7.4 was directly adsorbed on the different surfaces. After 30 minutes of incubation at room temperature, the supports were washed in Phosphate Buffer Solution (PBS) and the fluorescence was relatively quantified. Binding to microplates used for fluorescence assays was analyzed as a control. The Relative Fluorescence Units (RFU) measured for the three silicon based substrates are summarized in
Figure 1. RFU (485/527) indicates the excitation around 485 nm wavelength of light with peak fluorescence emission wavelength of 527 nm. Binding to the polished silicon was observed to be higher than the other substrates.
The above experiment carried out with the incubation of the immune molecules onto different substrates provides the confidence in fabricating silicon based microfluidic channels onto which biomolecules can be immobilized. Polished silicon is well suited to the present application for practical and biological reasons such as ease of fabrication, cost effectiveness, ease of immobilization with biological molecules and its crystalline structure that offers compatibility with different microfabrication processes. Later on, it has also been explained as to how smooth surface on silicon can be obtained by microfabrication through anisotropic etching.
The following sections of the paper give a detailed description of the microfluidic channel design, Finite Element Analysis of the flow behavior within the channels, fabrication technique of the microfluidic channel, experiments and fluorescence detection results achieved on the hybrid integrated optical microfluidic setup. This paper also explains as to how the results of flow controlled enzyme immobilization can be used for fabricating a Lab-on-a-chip device, by the hybrid attachment of the microfluidic device with a Spectrometer-on-chip.
2. Design, fabrication and packaging of microfluidic chip
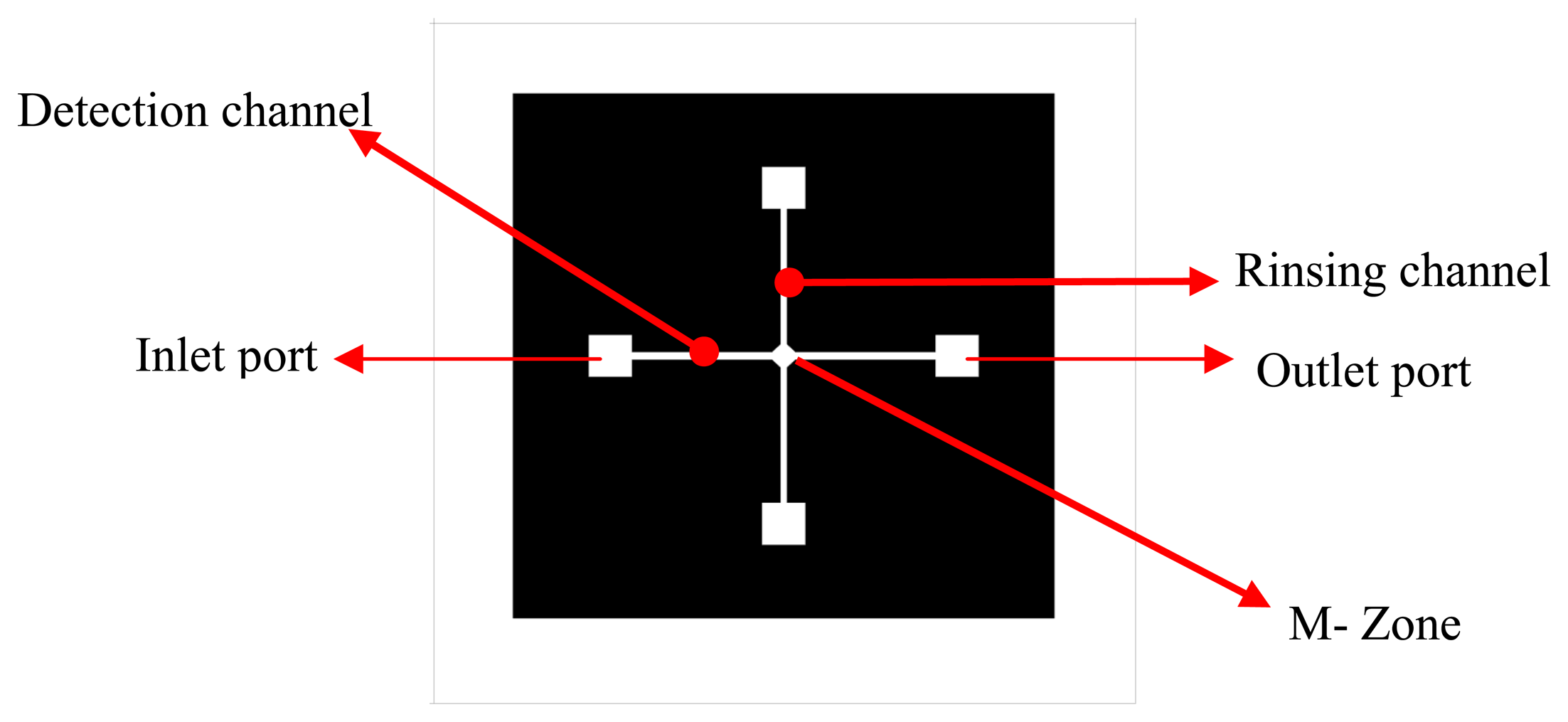
Several channel configurations were designed and Finite Element Analysis was carried out for the flow behavior. The final design of the microfluidic chip basically consisted of two channels, namely, the detection channel and the rinsing channel with a mixing zone (M-Zone), as shown in
Figure 2.
In order to study the flow behavior within the microfluidic channels, Navier-Stokes and continuity equations given in
equations (1) and
(2) were solved, assuming a 2-dimensional no-slip pressure driven flow that is invariant with respect to the height and having a constant velocity at the input, with the outlet being at ambient pressure. The incompressible Navier-Stokes and continuity equations [
15] are given as
wherein,
u⃗(
x,
y) is the velocity vector of the fluid,
p is the pressure,
ρ is the fluid density,
γ is the kinematic viscosity and
. The above equations were solved using Finite Element Modeling (FEM) with FEMLAB [
16] for several channel geometries of the channel, and the corroboration of the FEM results was carried out with commercial software, FLUENT [
17].
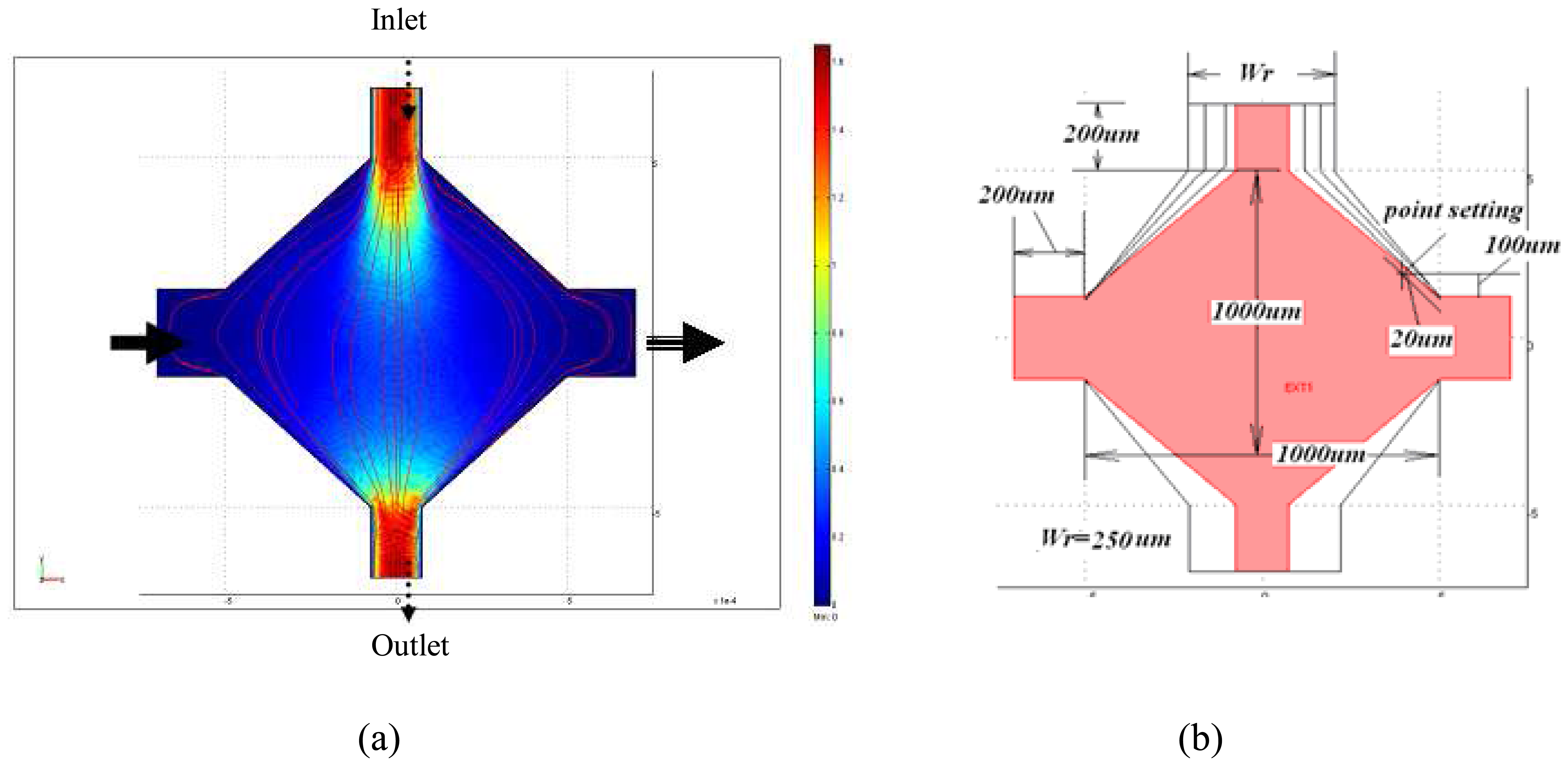
From the FEM results, the possible stagnation flow zones within the microfluidic channels were studied. The flow behavior for different channel dimensions was analyzed and suitable design geometry and parameters were applied for the microfluidic channel, as shown in
Figure 3.
The advantages of fabricating microfluidic channels with silicon have been mentioned before. In the experimental results presented in
Figure 1, it was observed that the biological specimen immobilized to the surface of polished silicon was better than other silicon surfaces. In order to obtain smooth surface [
18] for better binding of the biomolecules, in the present work, the microfluidic channels were fabricated by anisotropic etching technique using Tetra Methyl Ammonium Hydroxide (TMAH).
The microfabrication results have been presented in the form of Scanning Electron Microscope (SEM) images, as shown in
Figure 4 and
Figure 5. It can be seen that smooth anisotropic walls have been obtained on the microfluidic channel which is essential for undisrupted fluid flow and improved surface adsorption.
Packaging of the microfluidic chip consists of sealing the top with a transparent cover and introducing external microfluidic tubes thereby creating provisions for fluid inlet and outlet. In order to explore different packaging feasibilities on silicon based microfluidic channels and to study the surface affinity of biomolecules with different microfluidic channel surfaces, two types of chips were prepared for the experiments with two different top cover materials. In one of the chips, the channels were partially sputtered with gold and were packaged with a transparent polycarbonate top cover (these chips are called SGP -
Silicon-
Gold-
Plastic chips). Polycarbonate is a flexible material and it is 2mm thick, so tubes were inserted into the drilled holes and fixed with glue on SGP chip. The non gold sputtered chips were packaged with Pyrex top cover (called SiPy -
Silicon-
Pyrex chips). Steel metal connectors that fit perfectly into the holes on the Pyrex top cover were designed and fabricated by Computer Numerical Controlled (CNC) machining. These connectors were glued to the Pyrex wafer and connected to the external microfluidic circuit so as to package the SiPy chips. The SGP and SiPy are as shown in
Figure 6.
3. Experimental setup
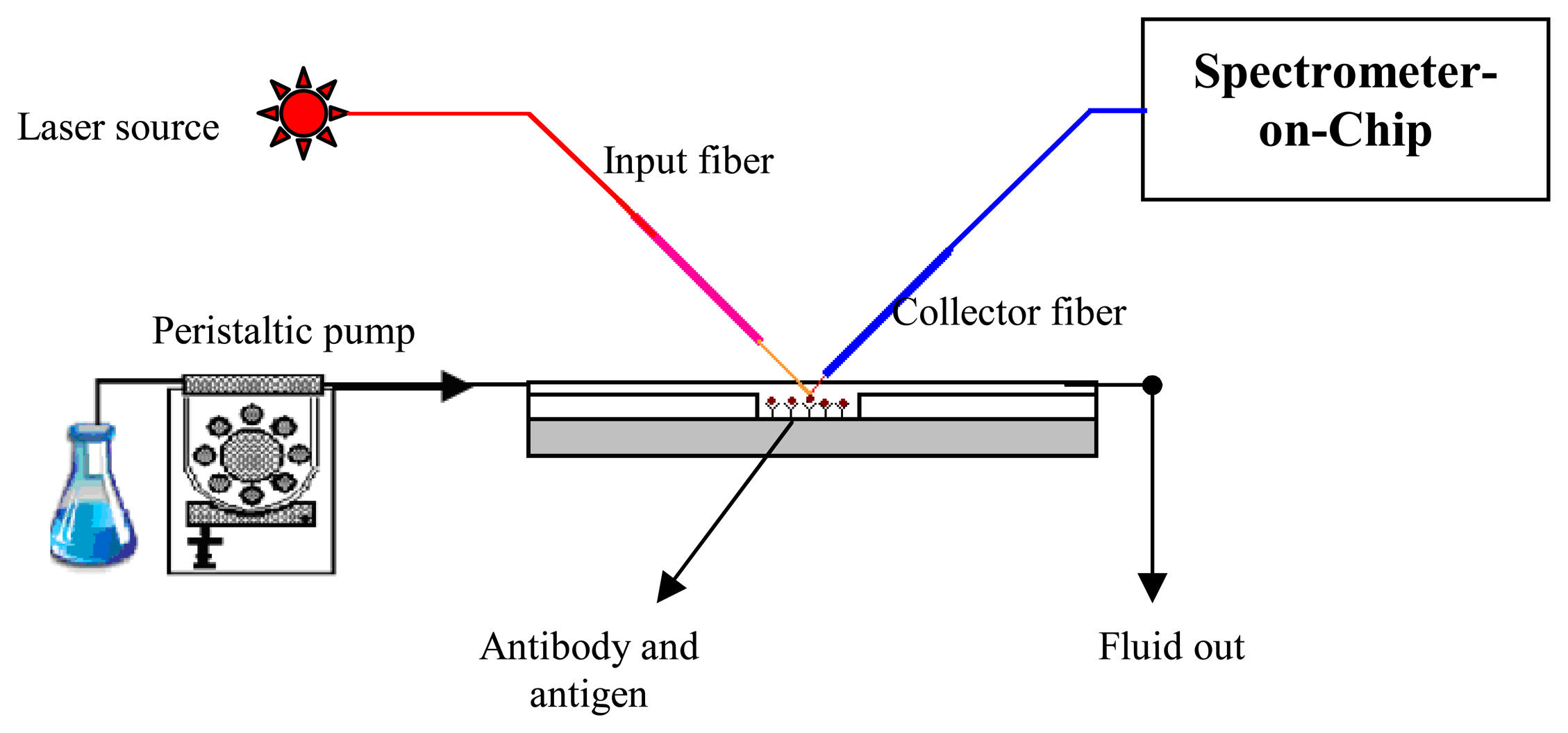
The bio-optical fluorescence testing not only involved the detection of antigen-antibody but also the characterization of different parameters such as the flow rate, surface stiction, rinsing time, concentration of the reagents to be used to optimize the fluorescent signal etc. In a hybrid integrated setup, the fluorescent signal obtained is a function of different parameters, such as, intensity of the input light, positioning of the collector fiber relative to the fluorescing region etc. Adequate fluorescent intensity is essential for the signals to be sensed by the spectrometer. Hence, it was necessary to optimize these parameters for controlled fluorescence study. The bio-optical testing scheme is shown in
Figure 7 and
Figure 8 shows the actual test setup used.
The hybrid integrated setup consisted of a standard SMF28 coupled SMA input fiber mounted on a vertical positioner. A diode laser source was used as the input light source emitting light in the range of 640 nm. The microfluidic chip was mounted on a nano-positioner so that the position of the chip with respect to the input fiber could be finitely adjusted. The fluorescent signals were collected by SMA fiber of 50 μm core diameter, mounted on a 45° platform. This fiber was coupled with a spectrometer at the other end in order to carry out further spectral analysis. Controlled fluid injection into the microfluidic channel was achieved using a peristaltic pump. Fiber position was then adjusted to obtain optimal fluorescence.
4. Experiment
10% antigen sheep IgG was prepared to 1% concentration, by diluting 25 ml of this sample with 225 ml of Phosphate Buffer Solution (PBS). Four different concentrations of the antibody (Alexafluor-647, 2 mg/ml, pH 7.5 in 0.1 M NaP and 0.1 M NaCl, with 5 mM azide) were prepared: 1%, 2%, 10%, and 20%, by diluting the antibody with PBS. 10% Bovine Serum Albumin (BSA) in Phosphate Buffer Solution (PBS) was used as the blocker buffer solution and Isopropyl Alcohol (IPA) was used to rinse the microfluidic channel.
Initially, de-ionized water was passed into the microfluidic channel and the residence time taken by the fluid to reach the mixing zone was calculated by repeating the procedure with different liquids such as IPA and BSA. The initial tests for fluorescence detection were carried out and the distance between the fluorescence collector and the microfluidic channel was optimized. Flow visualization was also carried out with Alexafluor 647 particles in order to establish the flow characteristics. The duration of flow for different fluids and the sequence in which the experiments were carried out is listed in
Table 1.
Controlled volume of the antigen was first passed into the microfluidic channel with a peristaltic pump and was allowed to remain in the mixing zone for 5 minutes, so that the antigen binds itself to the surface of the channel. The antigen was then rinsed with PBS and blocker buffer was passed through. PBS was passed again followed by the antibody. The channel was rinsed with PBS and the fluorescence readings were noted. This fluorescence was due to the antibody binding on to the antigen alone and not due to the stray antibody particles. Once the fluorescence readings were recorded, the channel was flushed with IPA and the procedure was repeated with different concentrations of the antibody. The flow was maintained at a velocity of 0.0375 m/s and rinsing was done at 0.065 m/s.The experiment was then repeated for different antibody concentrations on SiPy and SGP.
5. Results
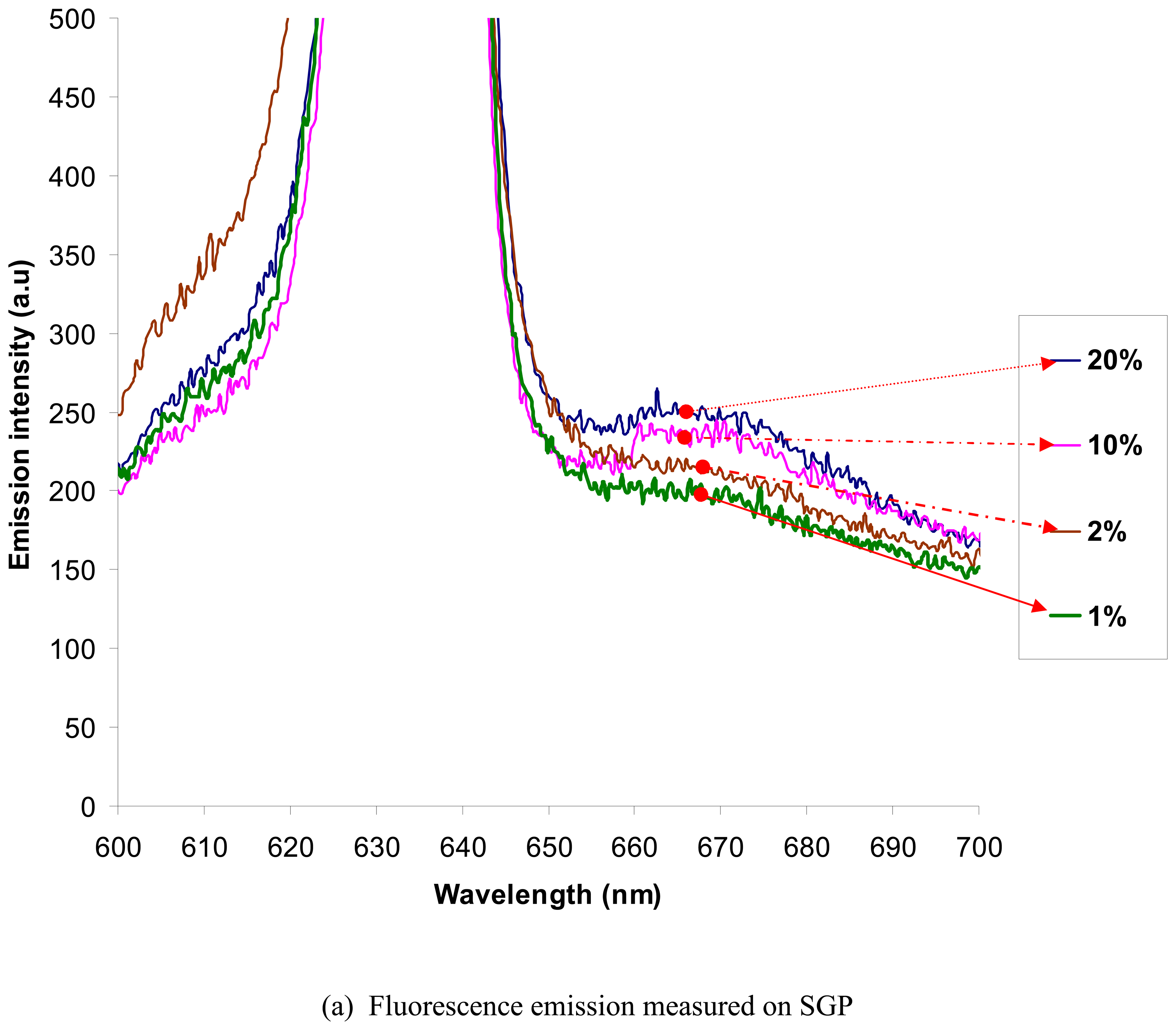
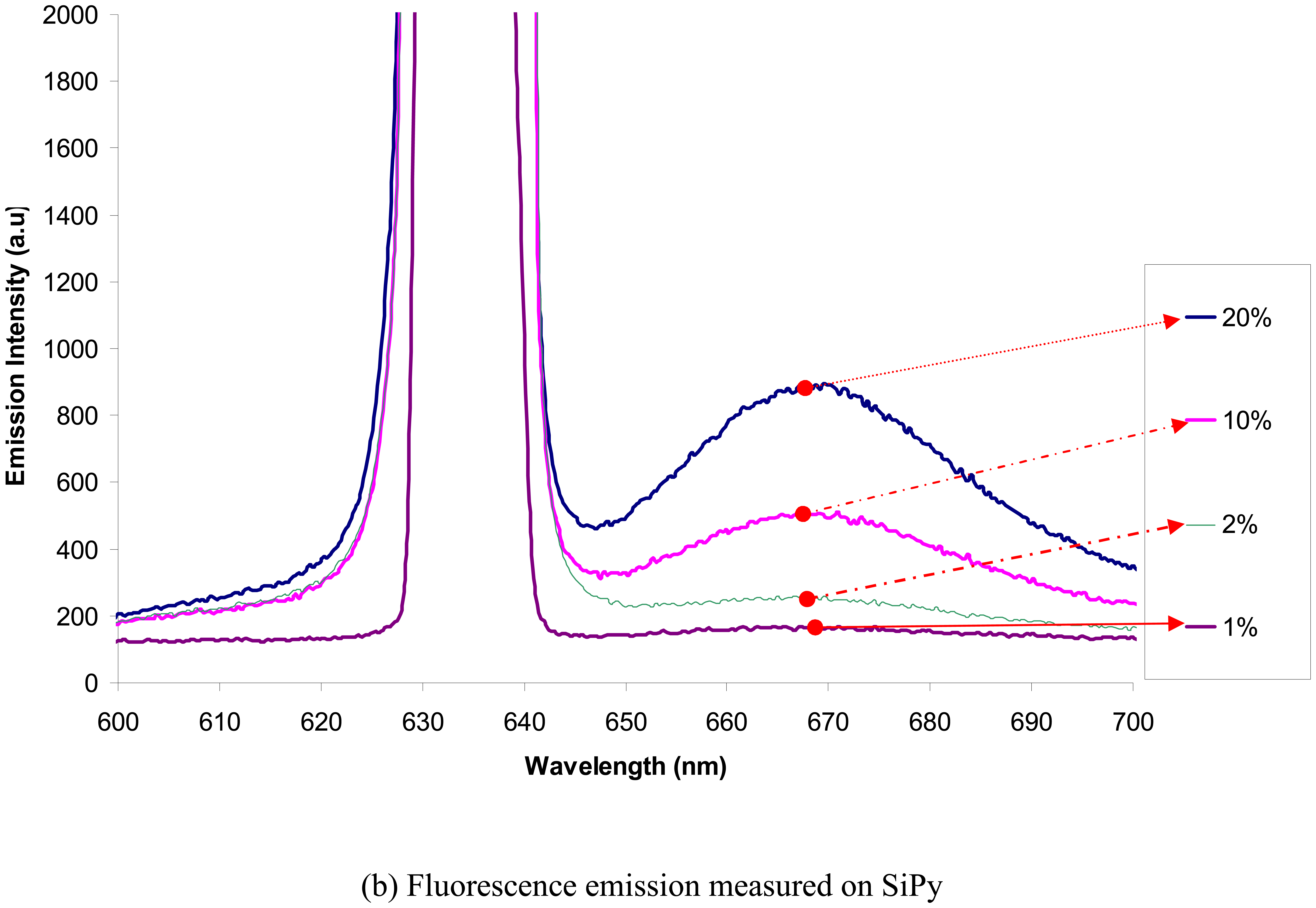
The fluorescence spectral response on SGP and SiPy are as plotted in
Figure 9(a) and 9(b) respectively. The peak at 670 nm is the result of fluorescence emitted by the fluorophores due to the excitation at 640 nm. It can be clearly observed from the fluorescent spectral response that the number of antibody molecules that adhere onto the silicon surface is much higher than the gold sputtered surface.
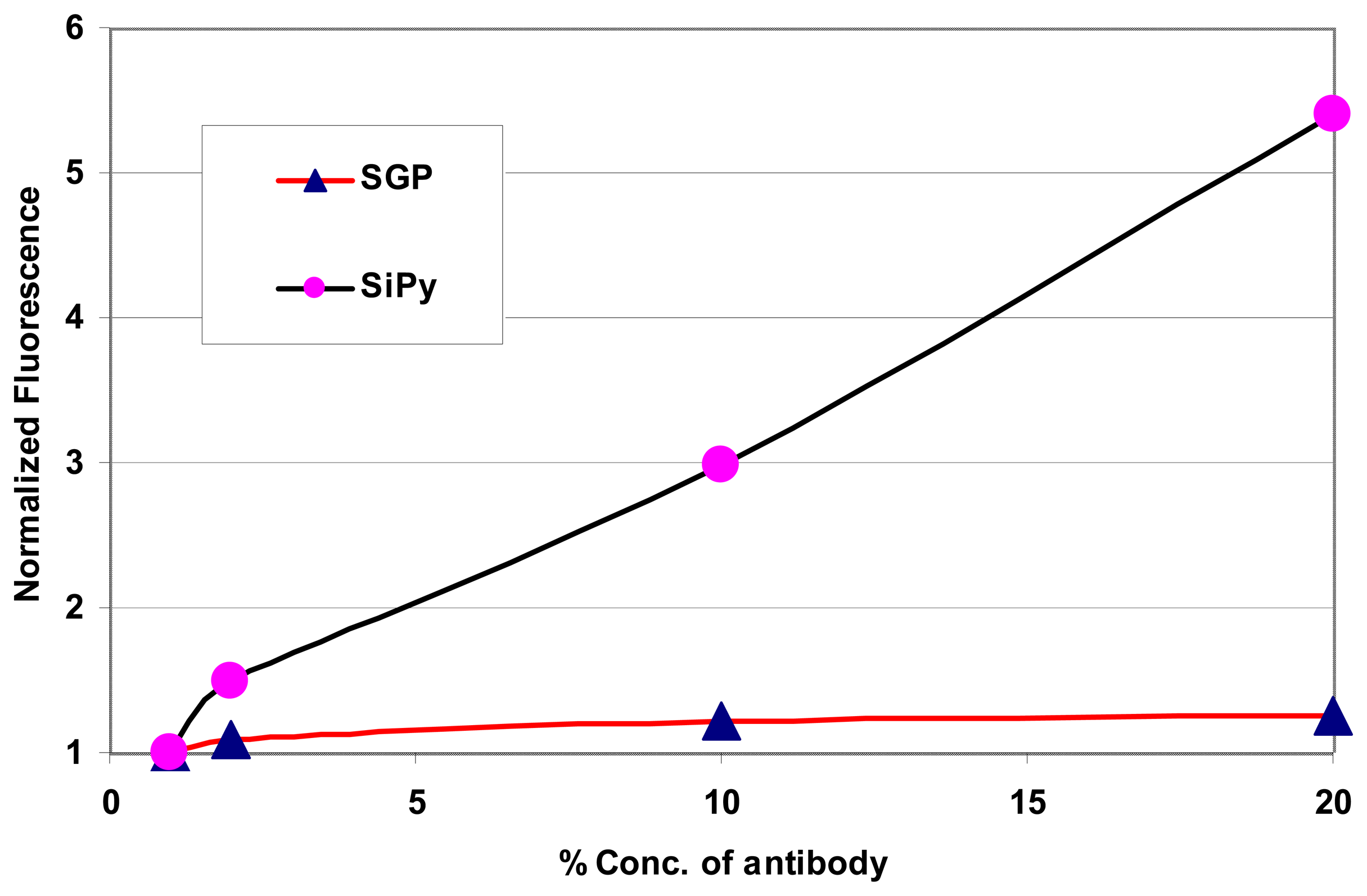
Once the fluorescence readings due to the immobilized antigen-antibody were recorded, the channels were then rinsed for 300 s and the residual fluorescence due to the remnant antigen-antibody was studied on both the platforms. A comparison of the fluorescence detected in both the SiPy and the SGP for different concentration of antibodies at different wavelengths is as shown in
Figure 10. In order to alleviate any discrepancies that may arise due to the study of absolute fluorescence values, given that fluorescence measurement is a function of several factors, the fluorescence unit is normalized with respect to the fluorescence observed with the lowest concentration of the antibody for the specific microfluidic chip, namely SGP and SiPy. It can be seen from
Figure 10 that the fluorescence emission from the antibodies immobilized onto the SiPy chips is comparatively much higher than the SGP chips.
It is thus very evident from the graphs in
Figure 10 that even after rinsing the channels, silicon surface retains sufficient amount of antigen molecules which proves that the immobilization of antigen with silicon is superior compared to the surface adsorption with gold coated channels, thereby confirming the results of the incubation experiment shown in
Figure 1 within a microfluidic channel environment.
In order to utilize the microfluidic chip as a Lab-on-a-chip device, a Spectrometer-On-Chip [
19,
20] was hybrid integrated with a SiPy microfluidic chip and the fluorescence measurements were carried out using the experimental setup shown in
Figure 8. The importance of using the spectrometer-on-chip (SOC) was that the size of the SOC being less than 4 cm
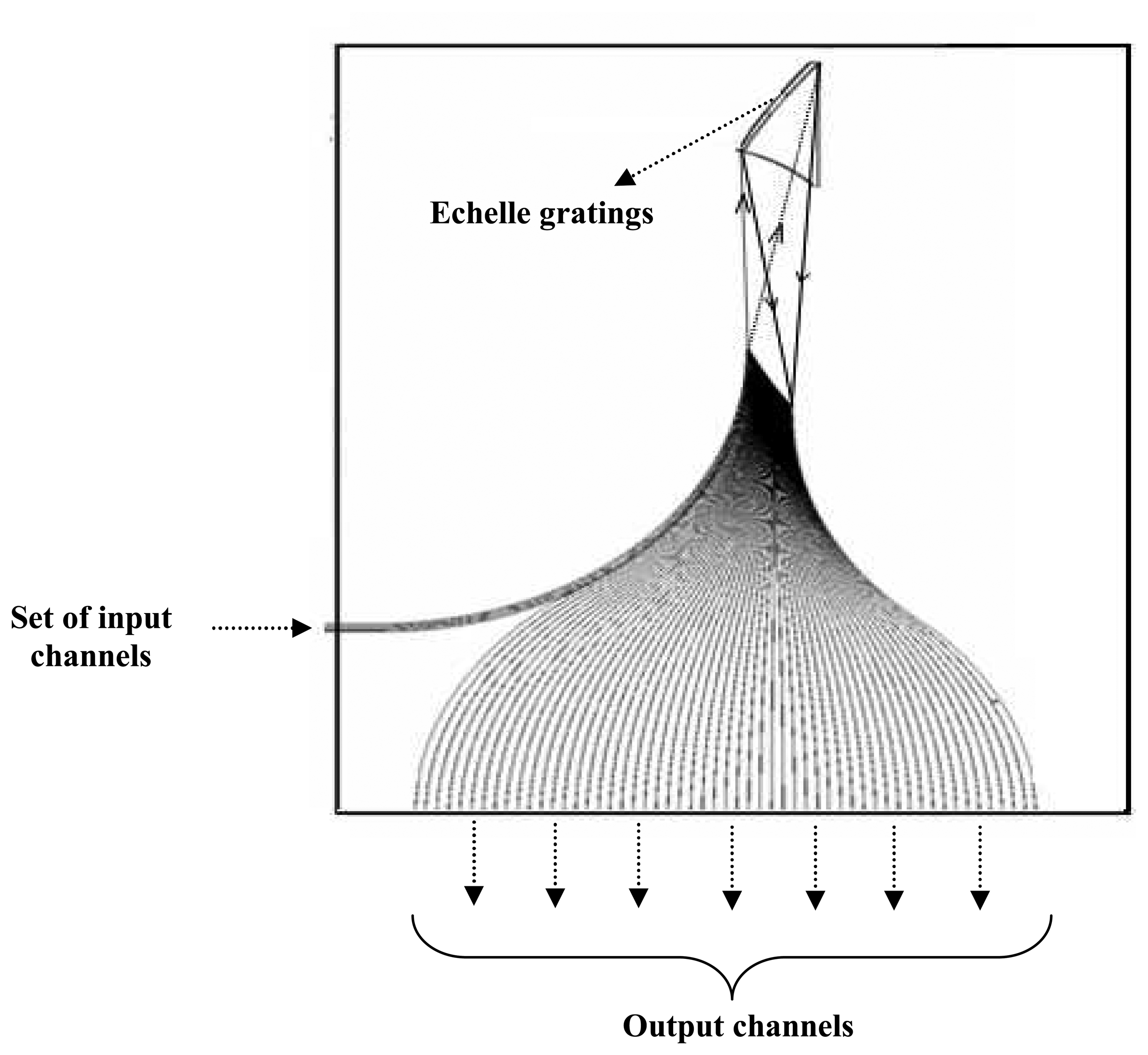
2, is very suitable for integration with the microfluidic device to form a Lab-on-a-chip device. The schematic working principle of the spectrometer-on-chip is illustrated in
Figure 11. The Spectrometer-On-Chip was designed based on Echelle grating fabricated with Silica-on-Silicon platform.
Herein, the set of input waveguides/channels of the SOC, specifically designed for a particular wavelength, terminate at the boundary of slab waveguide region where the light diverges in the waveguide plane and illuminates the grating. Light is diffracted backward from the concave grating and is focused onto the output waveguides/channels, which are arrayed along the Rowland circle with a spacing chosen to give the desired channel separation. In the present spectrometer-on-chip device, the input channel, channel 4 was used and the output for different wavelengths of light was obtained from channel 11 – 18, in which channel 11 was the output fiber for emission from Alexafluor fluorescent particles at 670 nm.
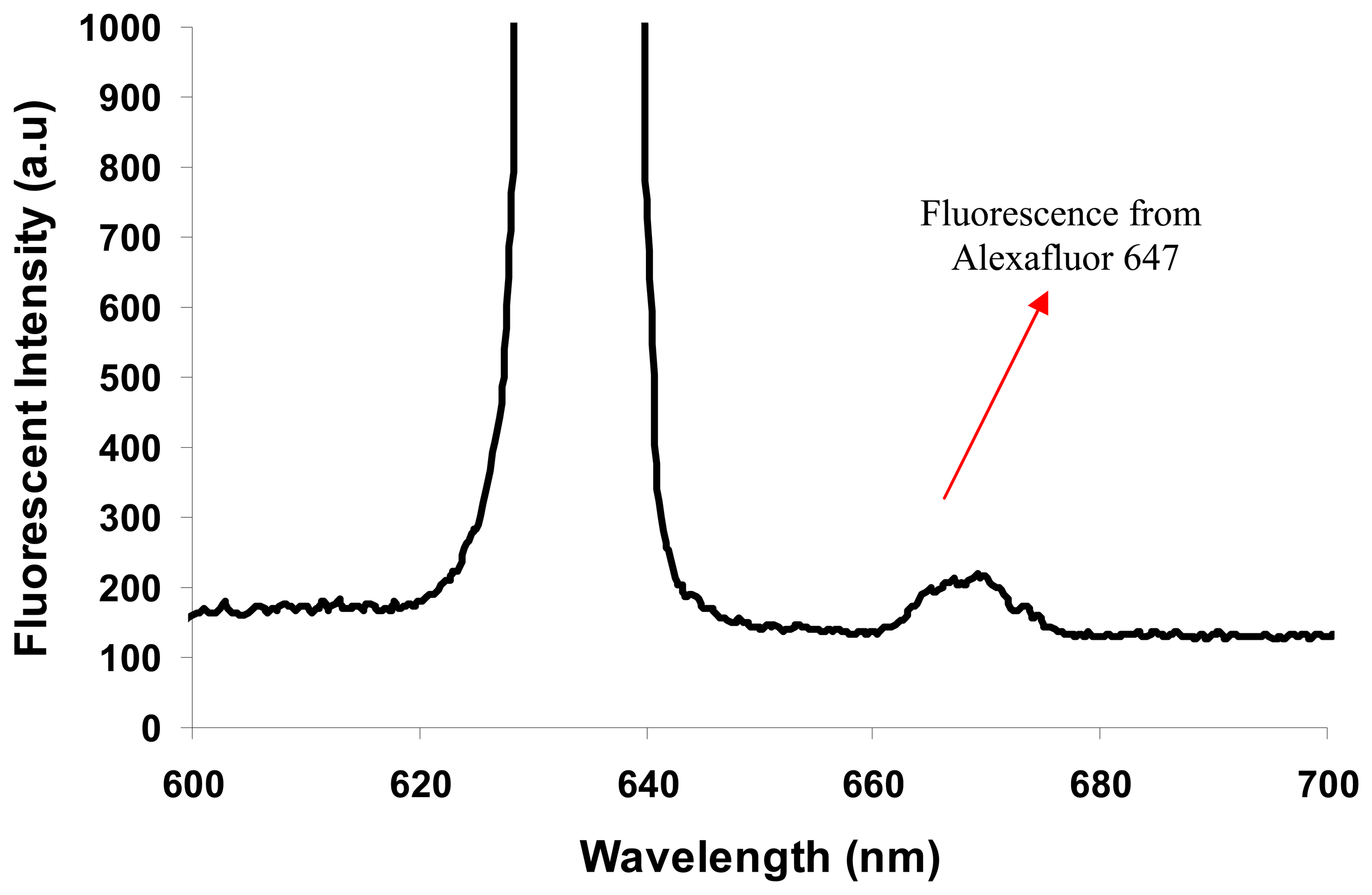
Figure 12 shows the spectral curve obtained from Chennal-11 of the SOC with the hybrid integrated setup.
The peak at 635 nm is observed due to the input light from the laser source. However, the peak at 670 nm is due to the fluorescence from the Alexafluor 647 particles. It is clear that the spectrometer-on-chip is able to detect the aimed fluorescence of Alexafluor 647 in channel 11 at a wavelength of 670 nm. One could improve the intensity of light detected by SOC by further improving the coupling with input channel-4 using a more integrated setup, and this would enable the fabrication of an integrated opto-microfluidic Lab-on-a-chip device, which can be used for real-time Point-of-Care testing applications.
6. Conclusion
Integrated Opto-microfluidics offers a lot of advantages to carry out high throughput analysis for various biomedical applications. In this work, the feasibility of using silicon based microfluidic chip for Lab-on-a-chip type applications has been demonstrated successfully through hybrid attachment of the device with spectrometer-on-chip for fluorescence based biodetection. Microfluidic channels on silicon platform can be easily bulk microfabricated in different geometries, thereby providing a highly cost-effective alternative for other materials used for such applications. Important aspects of the present work include immobilization of the antigen molecules onto the surface of the silicon microfluidic channels without the need for specialized surface treatments, simple and inexpensive microfluidic packaging techniques with pyrex and polycarbonate materials, which could now be applied in standard packaging technologies for silicon based microfluidic chips, and the hybrid integration of silicon based microfluidic chip and silica-on-silicon based spectrometer-on-chip, opening up the feasibility of fabricating a monolithically integrated Lab-on-a-chip type device that could be used for in-situ biomedical applications and Point-of-Care testing (POCT) for rapid pathogenic detections and for other chemical and biological sensing.