Aerosol Jet Printed Ion-Selective Electrodes for Potassium Detection
Abstract
1. Introduction
2. Materials and Methods
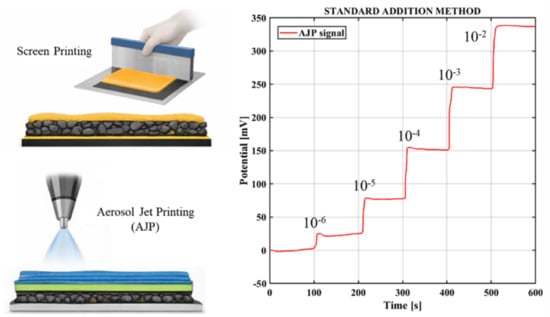
2.1. Sensor Description and Fabrication
2.2. Morphological Characterization
2.3. Measurement Setup
- -
- working electrode: printed or commercial K+ selective sensor;
- -
- reference electrode: integrated Ag/AgCl;
- -
- counter electrode: carbon trace.
2.4. Preparation of Standard Solutions for Tests
2.5. Interference Test
3. Experimental Results
3.1. Optical Characterization
3.2. Tests of Sensors in Distilled H2O
3.3. Effects of Interferents
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gao, F.; Liu, C.; Zhang, L.; Liu, T.; Wang, Z.; Song, Z.; Cai, H.; Fang, Z.; Chen, J.; Wang, J.; et al. Wearable and fl exible electrochemical sensors for sweat analysis: A review. Microsyst. Nanoeng. 2023, 9, 1. [Google Scholar] [CrossRef]
- Crespo, G.A. Electrochimica Acta Recent Advances in Ion-selective membrane electrodes for in situ environmental water analysis. Electrochim. Acta 2017, 245, 1023–1034. [Google Scholar] [CrossRef]
- Polidori, G.; Tonello, S.; Serpelloni, M. Ion-Selective All-Solid-State Printed Sensors: A Systematic Review. IEEE Sens. J. 2024, 24, 7375–7394. [Google Scholar] [CrossRef]
- Hyun, T. High-Performance Potassium-Selective Biosensor Platform Based on Resistive Coupling of a-IGZO Coplanar-Gate Thin-Film Transistor. Int. J. Mol. Sci. 2023, 24, 6164. [Google Scholar] [CrossRef]
- Yu, B.S.; Nie, L.H.; Yao, S.Z. Ion chromatographic study of sodium, potassium and ammonium in human body fluids with bulk acoustic wave detection. J. Chromatogr. B Biomed. Appl. 1997, 693, 43–49. [Google Scholar] [CrossRef]
- Korek, E.M.; Kounoupioti, E.; Brederlow, R. Manufacturing of Flexible, Impedimetric Potassium Sensors. IEEE Sens. Lett. 2023, 7, 1–4. [Google Scholar] [CrossRef]
- Sajid, M.; Justyna, P. Green analytical chemistry metrics: A review. Talanta 2022, 238, 123046. [Google Scholar] [CrossRef]
- An, Q.; Gan, S.; Xu, J.; Bao, Y.; Wu, T.; Kong, H.; Zhong, L.; Ma, Y.; Song, Z.; Niu, L. Electrochemistry Communications Full communication A multichannel electrochemical all-solid-state wearable potentiometric sensor for real-time sweat ion monitoring. Electrochem. Commun. 2019, 107, 106553. [Google Scholar] [CrossRef]
- Ping, J.; Wang, Y.; Wu, J.; Ying, Y. Development of an all-solid-state potassium ion-selective electrode using graphene as the solid-contact transducer. Electrochem. Commun. 2011, 13, 1529–1532. [Google Scholar] [CrossRef]
- Shao, Y.; Ying, Y.; Ping, J. Recent advances in solid-contact ion-selective electrodes: Functional materials, transduction mechanisms, and development trends. Chem. Soc. Rev. 2020, 49, 4405–4465. [Google Scholar] [CrossRef] [PubMed]
- Mirabootalebi, S.O.; Liu, Y. Recent advances in nanomaterial-based solid-contact ion-selective electrodes. Analyst 2024, 149, 3694–3710. [Google Scholar] [CrossRef] [PubMed]
- Macho, S.; Bobacka, J.; Rius, F.X. Transduction Mechanism of Carbon Nanotubes in Solid-Contact Ion-Selective Electrodes. Talanta 2009, 81, 676–681. [Google Scholar] [CrossRef]
- Chen, K.Y.; Kachhadiya, J.; Muhtasim, S.; Cai, S.; Huang, J.; Andrews, J. Underground Ink: Printed Electronics Enabling Electrochemical Sensing in Soil. Micromachines 2024, 15, 625. [Google Scholar] [CrossRef]
- Moya, A.; Gabriel, G.; Villa, R.; del Campo, F.J. Inkjet-printed electrochemical sensors. Curr. Opin. Electrochem. 2017, 3, 29–39. [Google Scholar] [CrossRef]
- Coltro, W.K.T.; Janegitz, B.C. Screen-Printing vs. Additive Manufacturing Approaches: Recent Aspects and Trends Involving the Fabrication of Electrochemical Sensors. Anal. Chem. 2025, 97, 1482–1494. [Google Scholar] [CrossRef]
- Hayat, A.; Marty, J.L. Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring. Sensors 2014, 14, 10432–10453. [Google Scholar] [CrossRef]
- Kamyshny, A.; Magdassi, S. Conductive nanomaterials for 2D and 3D printed flexible electronics. Chem. Soc. Rev. 2019, 48, 1712–1740. [Google Scholar] [CrossRef] [PubMed]
- Rocha, D.P.; Oliveira, P.R.; Janegitz, B.C.; Bonacin, J.A.; Richter, E.M.; Munoz, R.A.A. Analytica Chimica Acta Additive-manufactured (3D-printed) electrochemical sensors: A critical review. Anal. Chim. Acta 2020, 1118, 73–91. [Google Scholar] [CrossRef]
- Song, D.; Mao, C.; Jiang, C.; Liang, R.; Lisak, G. Trends in Analytical Chemistry Printed potentiometric sensors. Trends Anal. Chem. 2025, 192, 118420. [Google Scholar] [CrossRef]
- Jeong, H.; Lee, J.H.; Kim, S.; Han, S.; Moon, H. Optimization of process parameters in micro—Scale pneumatic aerosol jet printing for high—Yield precise electrodes. Sci. Rep. 2023, 13, 21297. [Google Scholar] [CrossRef]
- Secor, E.B. Principles of aerosol jet printing. Flex. Print. Electron. 2018, 3, 35002. [Google Scholar] [CrossRef]
- Fapanni, T.; Sardini, E.; Serpelloni, M.; Tonello, S. 3D Electrochemical Sensor and Microstructuration Using Aerosol Jet Printing. Sensors 2021, 21, 7820. [Google Scholar] [CrossRef]
- Di Novo, N.G.; Cantù, E.; Tonello, S.; Sardini, E.; Serpelloni, M. Support-Material-Free Microfluidics on an Electrochemical Sensors Platform by Aerosol Jet Printing. Sensors 2019, 19, 1842. [Google Scholar] [CrossRef]
- Wilkinson, N.J.; Smith, M.A.A.; Kay, R.W.; Harris, R.A. A review of aerosol jet printing—A non-traditional hybrid process for micro-manufacturing. Int. J. Adv. Manuf. Technol. 2019, 105, 4599–4619. [Google Scholar] [CrossRef]
- Rojas, D.; Torricelli, D. 3D-Printed Transducers for Solid Contact Potentiometric Ion Sensors: Improving Reproducibility by Fabrication Automation. Anal. Chem. 2024, 96, 15572–15580. [Google Scholar] [CrossRef]
- Bao, C.; Kaur, M.; Kim, W.S. Toward a highly selective artificial saliva sensor using printed hybrid field effect transistors. Sens. Actuators B Chem. 2019, 285, 186–192. [Google Scholar] [CrossRef]
- Sophocleous, M.; Contat-Rodrigo, L.; García-Breijo, E.; Georgiou, J. Towards solid-state, thick-film K+ and Na+ ion sensors for soil quality assessment. In Proceedings of the 2020 IEEE SENSORS, Rotterdam, The Netherlands, 25–28 October 2020; pp. 31–34. [Google Scholar] [CrossRef]
- Polidori, G. Ion-Selective Sensors with Interdigitated Electrodes by Aerosol Jet Printing for Biomedical and Industrial Fields: A Preliminary Investigation. In Proceedings of the 2025 IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd4.0 & IoT), Castelldefels, Spain, 1–3 July 2025; pp. 145–150. [Google Scholar] [CrossRef]
- Bakker, E. Selectivity of Liquid Membrane Ion—Selec tive Electrodes. Electroanalysis 1996, 9, 7–12. [Google Scholar] [CrossRef]
- Polidori, G.; Tonello, S.; Serpelloni, M.; Giamberini, M.; Montane, X.; Reina, J.A. Preliminary Study on Printed Membrane by Aerosol Jet for Ion Detection in Industrial Field. In Proceedings of the 2024 IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd4.0 & IoT), Firenze, Italy, 29–31 May 2024; pp. 1–6. [Google Scholar] [CrossRef]
- Yi, H.; Liu, Y.; Cao, H.; Luo, J.; Dong, X.; An, J. Material and process integrated innovations in Aerosol Jet Printing: A review. Mater. Today 2025, 91, 431–458. [Google Scholar] [CrossRef]
- Novell, M.; Parrilla, M.; Crespo, A.; Rius, F.X.; Andrade, F.J. Paper-Based Ion-Selective Potentiometric Sensors. Anal. Chem. 2012, 84, 4695–4702. Available online: https://pubs.acs.org/doi/10.1021/ac202979j (accessed on 7 May 2026). [CrossRef]
- Lemos, S.G.; Nogueira, A.R.A.; Torre-Neto, A.; Parra, A.; Artigas, J.; Alonso, J. In-soil potassium sensor system. J. Agric. Food Chem. 2004, 52, 5810–5815. [Google Scholar] [CrossRef]
- Manjunatha, J.G. A novel voltammetric method for the enhanced detection of the food additive tartrazine using an electrochemical sensor. Heliyon 2018, 4, e00986. [Google Scholar] [CrossRef]
- Lindsay, A.E.; Hare, D.O. The development of an electrochemical sensor for the determination of cyanide in physiological solutions. Anal. Chim. Acta 2006, 558, 158–163. [Google Scholar] [CrossRef]
- Durst, R.A. Ion-Selective Electrodes—The Early Years. Electroanalysis 2012, 24, 15–22. [Google Scholar] [CrossRef]
- Bobacka, J.; Ivaska, A.; Lewenstam, A. Potentiometric Ion Sensors. Chem. Rev. 2008, 108, 329–351. [Google Scholar] [CrossRef]
- Baranwal, J.; Barse, B.; Gatto, G.; Broncova, G. Electrochemical Sensors and Their Applications: A Review. Chemosensors 2022, 10, 363. [Google Scholar] [CrossRef]
- Chen, J.; Chung, H.; Hsu, C.; Tsai, D.; Kumar, A.S.; Zen, J. A disposable single-use electrochemical sensor for the detection of uric acid in human whole blood. Sens. Actuators B Chem. 2005, 110, 364–369. [Google Scholar] [CrossRef]
- Buck, R.P.; Lindner, E. Recomendations for nomenclature of ion-selective electrodes. Pure Appl. Chem. 1994, 66, 2527–2536. [Google Scholar] [CrossRef]
- Chumbimuni-torres, K.Y.; Rubinova, N.; Radu, A.; Lauro, T.; Bakker, E. Solid Contact Potentiometric Sensors for Trace Level Measurements. Anal. Chem. 2006, 78, 1318–1322. [Google Scholar] [CrossRef]
- Bobacka, J. Conducting Polymer-Based Solid-State Ion-Selective Electrodes. Electroanalysis 2006, 18, 7–18. [Google Scholar] [CrossRef]
- Ceresa, A.; Bakker, E.; Hattendorf, B.; Gu, D. Potentiometric Polymeric Membrane Electrodes for Measurement of Environmental Samples at Trace Levels: New Requirements for Selectivities and Measuring Protocols, and Comparison with ICPMS. Anal. Chem. 2001, 73, 343–351. [Google Scholar] [CrossRef]
- Tonello, S.; Abate, G.; Borghetti, M.; Lopomo, N.F.; Serpelloni, M.; Sardini, E. How to Assess the Measurement Performance of Mobile/Wearable Point-of-Care Testing Devices? A Systematic Review Addressing Sweat Analysis. Electronics 2022, 11, 761. [Google Scholar] [CrossRef]
- Tonello, S.; Fapanni, T.; Bonaldo, S.; Giorgi, G.; Narduzzi, C.; Paccagnella, A.; Serpelloni, M.; Sardini, E.; Carrara, S. Amperometric Measurements by a Novel Aerosol Jet Printed Flexible Sensor for Wearable Applications. IEEE Trans. Instrum. Meas. 2023, 72, 1–12. [Google Scholar] [CrossRef]
- Fan, Y.; Qian, X.; Wang, X.; Funk, T.; Herman, B.; McCutcheon, J.R.; Li, B. Enhancing long-term accuracy and durability of wastewater monitoring using electrosprayed ultra-thin solid-state ion selective membrane sensors. J. Memb. Sci. 2022, 643, 119997. [Google Scholar] [CrossRef]
- Serbest, B.; Kara, S.G.; Alpay, R.; Ataşer, T.; Kınacı, B.; Sönmez, N.A.; Özçelik, S. Aerosol Jet Printing of Flexible Transparent Conductive Silver Nanowire Electrodes: Effects of Printing Cycles. J. Electron. Mater. 2025, 54, 1245–1253. [Google Scholar] [CrossRef]











| Sensor Type | Conductive Substrate | Conductive Materials | PVC Membrane | Deposition Method |
|---|---|---|---|---|
| KION | Carbon | Conductive additives (e.g., PEDOT/MWCNTs, not declared) | Provided by the manufacturer | Screen printing |
| AJP | Carbon | MWCNTs (printed) | Printed | Aerosol Jet Printing |
| Additions | Time (s) | Added Volume (mL) | Concentration of Added Solution (M) | Concentration of Final Solution (M) |
|---|---|---|---|---|
| 1 | 0 | 0.01 | 10−2 | 10−6 |
| 2 | 100 | 0.09 | 10−2 | 10−5 |
| 3 | 200 | 0.026 | 0.34 | 10−4 |
| 4 | 300 | 0.266 | 0.34 | 10−3 |
| 5 | 400 | 0.92 | 1 | 10−2 |
| Sensor | Slope (mV/dec) | RSD (%) | Nernst Deviation (%) | R2 | t90 (s) |
|---|---|---|---|---|---|
| KION | 57.28 ± 5.07 | 8.86 | 3.18 | 0.9742 | 26.44 |
| AJP | 57.91 ± 5.07 | 8.75 | 2.11 | 0.9554 | 23.53 |
| Sensor | Slope (mV/dec) | RSD (%) | ND (%) | R2 | t90 (s) |
|---|---|---|---|---|---|
| KION | 61.75 ± 11.39 | 18.45 | 4.38 | 0.9755 | 31.14 |
| AJP | 56.41 ± 16.99 | 30.12 | 4.64 | 0.9598 | 21.10 |
| Type of Sensor | Analyte | Sensitivity (mV/dec) | R2 |
|---|---|---|---|
| KION | K+ | 57.28 ± 5.07 | 0.9742 |
| NH4+ | 20.60 ± 4.86 | 0.884 | |
| Na+ | 33.95 ± 8.95 | 0.936 | |
| Urea | 5.89 ± 2.77 | 0.847 | |
| AJP | K+ | 57.91 ± 5.07 | 0.9554 |
| NH4+ | 19.64 ± 7.35 | 0.942 | |
| Na+ | 24.37 ± 1.39 | 0.936 | |
| Urea | 10.46 ± 3.71 | 0.817 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Polidori, G.; Sardini, E.; Serpelloni, M. Aerosol Jet Printed Ion-Selective Electrodes for Potassium Detection. Sensors 2026, 26, 3053. https://doi.org/10.3390/s26103053
Polidori G, Sardini E, Serpelloni M. Aerosol Jet Printed Ion-Selective Electrodes for Potassium Detection. Sensors. 2026; 26(10):3053. https://doi.org/10.3390/s26103053
Chicago/Turabian StylePolidori, Giorgia, Emilio Sardini, and Mauro Serpelloni. 2026. "Aerosol Jet Printed Ion-Selective Electrodes for Potassium Detection" Sensors 26, no. 10: 3053. https://doi.org/10.3390/s26103053
APA StylePolidori, G., Sardini, E., & Serpelloni, M. (2026). Aerosol Jet Printed Ion-Selective Electrodes for Potassium Detection. Sensors, 26(10), 3053. https://doi.org/10.3390/s26103053